Abstract
Epithelial-mesenchymal transition (EMT) is a critical step in renal fibrosis. It has been recently reported that a transcription factor, Twist, plays a pivotal role in metastasis of breast tumors by inducing EMT. In this study, we examined whether Twist relates to renal fibrogenesis including EMT of tubular epithelia, evaluating Twist expression level in the unilateral ureteral obstruction (UUO) model. Kidneys of mice subjected to UUO were harvested 1, 3, 7, and 10 days after obstruction. Compared with control kidneys, Twist mRNA-level significantly increased 3 days after UUO (UUO day 3 kidney) and further augmented until 10 days after UUO. Twist expression increased in tubular epithelia of the dilated tubules and the expanded interstitial areas of UUO kidneys, where cell-proliferating appearances were frequently found in a time-dependent manner. Although a part of tubular cells in whole nephron segment were immunopositive for Twist in UUO day 7 kidneys, tubular epithelia downstream of nephron more frequently expressed Twist than upstream of nephron. In UUO day 7 kidneys, some tubular epithelia were confirmed to coexpress Twist and fibroblast-specific protein-1, a marker for EMT, indicating that Twist is involved in tubular EMT under pathological state. Twist was expressed also in a number of α-smooth muscle actin-positive myofibroblasts located in the expanded interstitial area of UUO kidneys. From these findings, the present investigation suggests that Twist is associated with tubular EMT, proliferation of myofibroblasts, and subsequent renal fibrosis in obstructed kidneys.
Keywords
T
Whereas the role of myofibroblasts in renal fibrosis is widely accepted, their origins and activation process in the fibrous kidney remain largely obscure and controversial. Because myofibroblasts are localized in the interstitial area in vivo, they often seem to result from the activation of resident fibroblasts in renal interstitial area. However, the pioneer study by Strutz et al. (1995) showed that renal tubular epithelial cells could express fibroblast-specific protein-1 (FSP1), one of the fibroblast-associated markers, under pathological conditions, indicating the possibility that tubular epithelial cells undergo epithelial-mesenchymal transition (EMT). FSP1 is a cytoskeleton-associated, calcium-binding protein normally expressed in fibroblasts but not in tubular epithelia (Strutz et al. 1995). Epithelial cells sit on and adhere to basement membrane that provides architectural stability for cell-cell contact emblematic of this phenotype. When the disruptionofbasement membrane occurs under pathological conditions, epithelia begin to express various cytokines such as TGF-β, EGF, and FGF-2 that initiate EMT (Zeisberg et al. 2001; Iwano and Neilson 2004). Tubular EMT, by definition, is a series of processes in which renal tubular cells lose their epithelial phenotype and acquire new characteristic features of mesenchymal cells. This phenotypical conversion not only illustrates the plasticity of mature, differentiated kidney epithelial cells but is also basically included in the pathogenesis of various types of chronic renal disease (Iwano et al. 2002; Yang et al. 2002; Zeisberg et al. 2003). Tubular epithelial EMT is one of the most plausible explanations for the replenishment of fibroblasts required for renal fibrogenesis. Iwano et al. (2002) provided direct evidence that a large fraction of interstitial fibroblasts derive from tubular epithelial cells under the pathological stress of renal fibrosis.
In carcinomas, the metastatic process is thought to consist of many distinct steps. The first step, invasion, requires neoplastic epithelial cells to lose cell-cell adhesion and to gain motility, which enables them to invade adjacent tissue. In the second step, tumor cells penetrate through the endothelium of blood or lymphatic vessels to enter the systemic circulation. EMT is recognized as an essential process for tumor metastasis (Thiery 2002). Several transcription factors including Snail, Slug, and SIP1 have been reported to be important for regulation of EMT in metastatic processes (Kang and Massague 2004). Recently, the transcription factor Twist, a master regulator of embryonic morphogenesis, was newly identified as an important factor of promoting EMT in breast cancers (Yang et al. 2004). Yang et al. (2004) demonstrated that ectopic expression of Twist potently induces EMT in Madin-Darby canine kidney epithelial cells (MDCK) and immortalized human mammary epithelial cells under culture.
In the present study we examine whether the expression level of Twist is increased in fibrous kidneys at mRNA level and clarify the localization of Twist in fibrous kidneys using immunohistochemistry (IHC). Furthermore, we discuss whether a transcription factor, Twist, is associated with tubular EMT and renal fibro-genesis in obstructed kidney.
Materials and Methods
Animal Model
Male C57BL/6 mice weighing 20–25 g at 8 weeks of age received water and standard mouse chow. Animal handling conformed to the guidelines for care and use of experimental animals established by the Ethical Committee of Animal Experiments of Tsurumi University School of Dental Medicine. Unilateral ureteral obstruction (UUO) was performed using an established procedure (Chevalier et al. 1999). Briefly, under ether narcosis, complete ureteral obstruction was performed by double ligating the left ureter using 3-0 silk after a left lateral abdominal incision. Mice were sacrificed 1, 3, 7, and 10 days (n = 3 mice per group) after obstruction. The left kidneys harvested 1, 3, 7, and 10 days after ureteral ligation were defined as UUO day 1, UUO day 3, UUO day 7, and UUO day 10 kidney, respectively. Similarly, we defined the contralateral right kidneys harvested 1, 3, 7, and 10 days after UUO asCont day 1, Cont day 3, Cont day 7, and Cont day 10 kidney, respectively. Cont day 1 kidneys were used for control.
Real-time PCR
Total RNAs were extracted from both right (Cont kidney) and left kidney (UUO kidney) at different time points after UUO using TRIzol Reagent (Invitrogen; Carlsbad, CA). cDNA was synthesized from 1 μg of total RNA using a Super Script III first-strand synthesis system with Oligo dT (Invitrogen) according to the manufacturer's instruction. Synthesized cDNAs were used as templates for real-time PCR using ABI PRISM 7900HT Fast Real-Time PCR System and SYBR Green PCR Master Mix (Applied Biosystems; Foster City, CA). PCR primer, annealing temperature, and cycles were as follows: for Twist, 5′-GAA-AAT-GGA-CAG-TCT-AGA-GAC-TCT-G, 5′-GTG-GCT-GAT-TGG-CAA-GAC-CTC-TTG, 55C, 40 cycles; for Wnt-2, 5′-CAT-GGT-GGT-ACA-TGA-GAG-CTA, 5′-GGC-AAA-TAC-AAC-GCC-AGC-TGA, 55C, 40 cycles; for glyceraldehyde-3-phosphate dehydrogenase (GAPDH), 5′-GGC-AAA-TTC-AAC-GGC-ACA-GTC, 5′-GCT-GAC-AAT-CTT-GAG-TGA-GTT, 55C, 40 cycles. Primers were designed using different exons for each gene. The ratios of Twist and Wnt-2 to GAPDH mRNAs were calculated in each sample.
IHC for Twist
Mice were transcardially perfused with 0.1 M PBS followed by the fixative solution containing 4% paraformaldehyde and 15% saturated picric acid for IHC. Both Cont and UUO kidneys were harvested, cut, and immersed in the same fixative solution for 4–6 hr at 4C. For cryostat sections, a part of the kidney was immersed with 15% sucrose in 0.1 M phosphate buffer (PB) (pH 7.4) for 6 hr, 30% sucrose-PB for 6 hr, continuously after fixation. Finally, specimens were frozen in optimal cutting temperature compound (OCT; Sakura Finetechnical, Tokyo, Japan). Five-μm-thick serial cryostat sections were cut and maintained on slides at −20C. Immunolabeling was performed with a mouse monoclonal antibody against Twist (Gitelman 1997) and visualized by the streptavidin-biotin-peroxidase complex method as follows. Sections were treated with 0.3% hydrogen peroxide in 100% methanol for 20 min to block endogenous peroxidase activity and incubated with 10% BSA (Sigma; St Louis, MO) in PBS with 0.03% Triton X-100 (Wako; Tokyo, Japan) for 30 min. A primary antibody was applied overnight at appropriate dilutions (1:50) in PBS with 1% BSA (Sigma) and 0.03% Triton X-100. After that, sections were incubated with biotinylated rat anti-mouse immunoglobulins (Dako; Copenhagen, Denmark) diluted 1:600 in PBS with 1% BSA (Sigma) and Triton X-100 for 30 min and, finally, incubated with horseradish peroxidase-conjugated streptavidin (Dako) diluted 1:300 in PBS with 1% BSA (Sigma) and Triton X-100 for 30 min. Immunoreaction was visualized using 0.025% DAB (Sigma) and 0.065% sodium azide (Wako) in 0.05 M Tris-HCl buffer (pH 7.4) containing 0.01% hydrogen peroxidase for 10 min. All steps were performed at room temperature. Sections were counterstained with hematoxylin, dehydrated, and then mounted in Permount. As the negative controls, we performed the same steps of IHC for UUO kidneys without primary antibody to rule out the possibility of binding of the biotinylated secondary antibody to endogenous mouse immunoglobulin because it would be difficult to flush all the plasma out of UUO kidneys during perfusion fixation due to compression of renal parenchyma by urine accumulation.
Assessment of the Number of Tubular Epithelial Cells Expressing Twist
To evaluate the grade of Twist expression in tubular epithelia, we counted the number of tubular epithelial cells expressing Twist (Twist+ tubular cell) per 100 tubular cells of both Cont and UUO kidneys. We randomly selected 1200 tubular epithelial cells of the cortex in each Cont or UUO kidney group (n = 3 mice per group). The cortex was defined as the region extending from the capsule to the deepest glomerulus.
Assessment of the Relation Between Tubular Expression of Twist and Luminal Area of Tubules
We defined that Twist+ tubules were the tubules containing more than one Twist+ tubular cell and Twist− tubules were defined as the tubules without Twist+ tubular cells. Forty Twist+ or Twist− tubules in the cortex of UUO day 7 kidneys (n = 3 mice) were randomly selected, and the luminal area of such tubules was measured with an Image Processor and Analyser (TRI/2 D-MES; RATOC, Tokyo, Japan). The measured luminal areas of Twist+ tubules were presented as the relative value to those of Twist− tubules.
Double-immunofluorescence Staining for Twist and Other Markers
Frozen sections of both Cont and UUO day 7 kidneys prepared in the manner described above were used for double-immunofluorescence staining. Sections were preincubated with 10% BSA (Sigma) in PBS with Triton X-100 for 30 min. Primary antibodies were as follows: (1) anti-aquaporin-1 (AQP1) antibody (Chemicon; Temecula, CA), which is a specific marker for the proximal tubules and the thin descending limb of Henle's loop (Nielsen et al. 2002); (2)anti-Na/K/2Cl cotransporter (NKCC2) antibody (Alpha Diagnostic International; San Antonio, TX), which is a specific marker for the thick ascending limb of Henle's loop (Delpire and Mount 2002); (3) anti-Na/Cl cotransporter (NCC) antibody (Chemicon), which is a specific marker for the distal tubules (Delpire and Mount 2002); (4) anti-AQP2 antibody (Santa Cruz Biotechnology; Santa Cruz, CA), which is a specific marker for both the connecting tubules and the collecting ducts (Nielsen et al. 2002); (5) anti-FSP1 (FSP1 is a mouse homolog of human S100A4) antibody (Lab Vision; Fremont, CA), which is a specific EMT marker in case of epithelial expression (Okada et al. 1997); and (6) anti-αSMA antibody (Abcam; Cambridge, UK), which is a specific mesenchymal marker. All primary antibodies were generated in rabbits except anti-AQP2 antibody, which was a goat polyclonal antibody. All primary anti-bodies were diluted 1:100 in PBS with 1% BSA (Sigma) and Triton X-100. Anti-Twist antibody was generated from mice as mentioned above. Sections were incubated with the primary antibodies for 2 hr at 37C, washed in PBS with Triton X-100, and then incubated with the secondary antibodies for 1 hr at room temperature. As the secondary antibodies, we used Alexa Fluor-labeled antibodies (Molecular Probes; Eugene, OR) or FITC-conjugated anti-mouse IgG (Dako) for each primary antibody. Stained sections were mounted with Entellan (Merck KgaA; Darmstadt, Germany) and visualized by confocal laser-scanning microscope (Fluoview 500; Olympus, Tokyo, Japan). We performed immunofluorescence staining for UUO kidneys without the primary antibodies as the negative controls.
Analysis of the Degree of Tubular Twist Expression in Three Nephron Segments
In UUO day 7 kidney, we analyzed the difference in the degree of tubular Twist expression at three nephron segments as follows: (1) proximal tubule, (2) distal tubule, and (3) connecting tubule/collecting duct. We counted the number of Twist+ tubular cells per 100 tubular cells with AQP1 expression (AQP1+ tubular cells) in the cortex of UUO day 7 kidney after double-immunofluorescence staining for Twist and AQP1. Eight hundred AQP1+ tubular cells in the cortex (proximal tubular epithelia) were randomly selected and analyzed (n = 3 mice). For NCC+ or AQP2+ tubular cells in the cortex of UUO day 7 kidney, we performed the same analysis and examined the extent of tubular Twist expression in three nephron segments (n = 3 mice per segment). In the cortex, NCC+ and AQP2+ tubular cells constitute the distal tubule and connecting tubule/collecting duct, respectively.
IHC for Proliferating Cell Nuclear Antigen (PCNA)
After perfusion by the fixative solution, a part of the kidney was dehydrated and embedded in paraffin. Five-μm-thick serial paraffin sections were cut and maintained at room temperature on gelatin-coated slides until submitting to IHC. Immuno-labeling was performed with a mouse monoclonal antibody against PCNA (Dako) and visualized by the streptavidin-biotin-peroxidase complex method described above. Primary antibody was diluted 1:200 in PBS with 1% BSA (Sigma) and Triton X-100. We also performed the same steps of IHC for UUO kidneys without the primary antibody as the negative controls.
Proliferating cells were identified with nuclear expression of PCNA. We defined that PCNA+ tubules were the tubules containing at least one tubular epithelial cell expressing PCNA (PCNA+ tubular cell). To evaluate the relationship between tubular dilatation and tubular epithelial proliferation, we randomly selected 100 PCNA+ tubules in each Cont or UUO kidney group and measured the luminal areas of such tubules (n = 3 mice per group) with an Image Processor and Analyser (TRI/2 D-MES; RATOC). In control kidneys, we measured only 30 tubular luminal areas of PCNA+ tubules because we rarely found PCNA+ tubules in those kidneys (n = 3 mice). Moreover, to examine the relationship between cellular proliferation and tubular dilatation, 100 PCNA+ tubules were analyzed (tubular luminal area vs the number of PCNA+ tubular cells per one tubule) in UUO day 7 kidney (n = 3 mice). In cortical interstitial areas of both Cont and UUO kidneys, we counted the number of cells expressing PCNA (PCNA+ interstitial cells) per one high-power field (HPF) under high magnification (3400). We randomly selected 10 HPFs in the cortex of each Cont and UUO kidneys (n = 3 mice per group).
Statistical Analysis
Statistical analyses were performed with a commercially available statistical package (Dr. SPSS II). Dunnett's t-test was mainly used to determine whether there were statistically significant differences among groups. In addition, we performed the statistical analysis using linear regression for correlation (the tubular luminal area vs the number of PCNA+ tubular cells/one tubule); p<0.05 was considered statistically significant.
Results
Gene Expression for Twist and Wnt-2
UUO model has been used as one of the experimental models developing renal tubulointerstitial fibrosis (Klahr 1998). Gene expressions for Twist and Wnt-2 were examined in both Cont and UUO kidneys. Real-time PCR revealed that Twist mRNA expression significantly increased from 3 days after UUO (Figure 1A). Expression levels of Twist mRNA were 11.4 times as high in UUO day 7 kidneys and 21.5 times as high in UUO day 10 kidneys as those in control kidneys (Cont day 1 kidneys), respectively (Figure 1A). It has been reported that Wnt-2 contributed to the regulation of endogenous Twist expression in the mouse mammary gland (Howe et al. 2003). Thus, we examined mRNA levels of Wnt-2 in both Cont and UUO kidneys. Wnt-2 mRNA expression significantly increased 3 days after ureteral ligation compared with control and continued to augment until 10 days after UUO as well as Twist mRNA (Figure 1B). Expression levels of Wnt-2 mRNA in UUO day 7 and UUO day 10 kidneys were 16.5 times and 32.6 times as high as those in control kidneys, respectively (Figure 1B).
IHC for Twist
In control kidneys, Twist-labeled cells were sparsely distributed in the vascular wall and interstitial space (Figure 2A). Twist-immunoreactive products were also detected on the capsular epithelia of Bowman's capsule (Figure 2A, double arrows), whereas Twist expression was not clearly identified in the glomerulus (Figure 2A). After ureteral ligation, expression level of Twist was remarkably increased in the intertubular areas and the Bowman's capsule (Figures 2B-2D, double arrows) in a time-dependent manner. In these areas, spindle-shaped fibroblast-like cells were intensely labeled with anti-Twist antibody in UUO day 3 and UUO day 7 kidneys (Figures 2C and 2D, double arrowheads). Although we did not determine which kind of glomerular cells expressed Twist, many cells in glomerulus expressed Twist in UUO day 3 and UUO day 7 kidneys (Figures 2C and 2D, single arrowheads). We detected immuno-labeling of Twist not only in the nuclei of some cells (Figure 2A, double arrows; Figure 2D, double arrowhead) but also in the cytoplasm. Time-dependent changes of Twist expression were consistent with those of Twist mRNA obtained from real-time PCR analysis (Figure 1A). Moreover, in UUO day 3 and UUO day 7 kidneys, some tubular epithelial cells slightly expressed Twist (Figures 2C and 2D, single arrows). No specific staining was detected without anti-Twist antibody in the section of UUO day 7 kidney (Figure 2E). In the medulla, intense immunolabeling of Twist was extensively observed in the expanded interstitial space of UUO day 7 kidneys (Figure 3B, arrows), whereas Twist was sparsely expressed in intertubular space of control kidneys (Figure 3A, arrowheads).
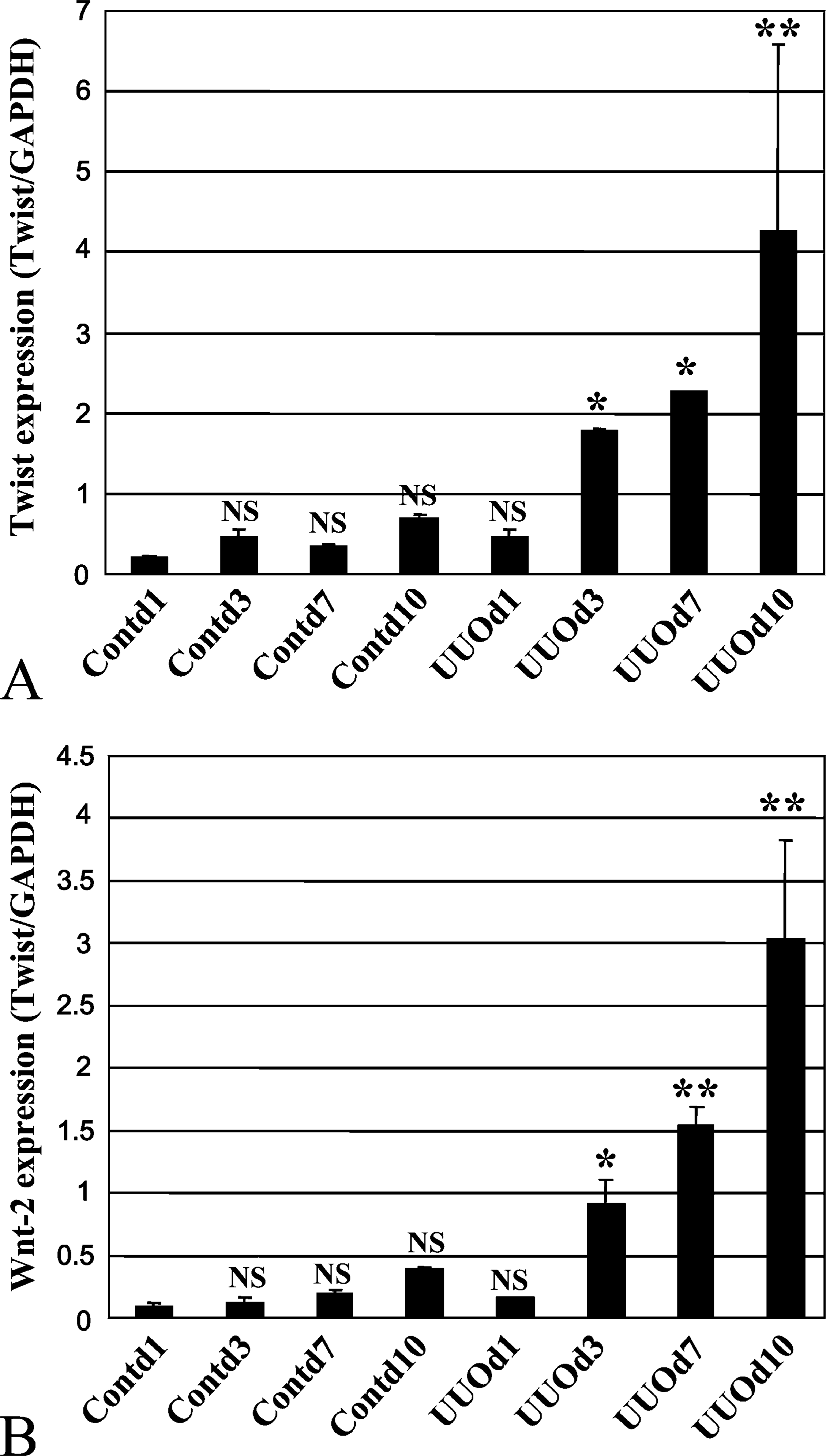
Time-dependent increases in mRNA levels of Twist and Wnt-2 in unilateral ureteral obstruction (UUO) kidneys. Real-time PCR was performed using primer sets for Twist, Wnt-2, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Transcription levels of Twist (
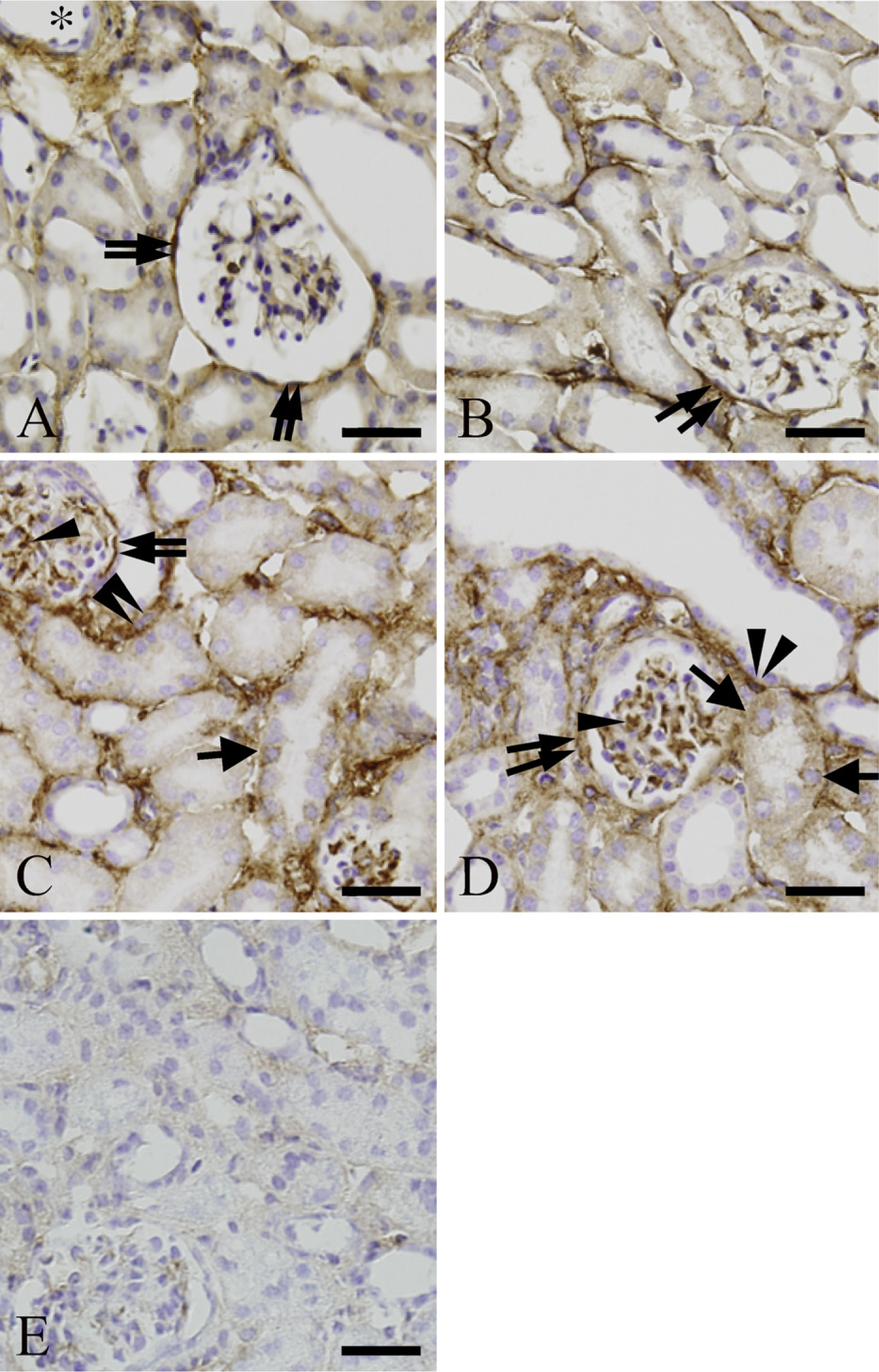
Immunolabeling of Twist in cortex of control and UUO kidneys. (
Quantification of Twist Expression
We examined the number of Twist+ tubular cells per 100 tubular cells in each Cont and UUO kidney. As shown in Figure 4A, the number of Twist+ tubular cells in UUO kidneys continued to increase until 7 days after UUO and was significantly increased in UUO day 3 and UUO day 7 kidneys compared with that in control kidneys (p<0.01).
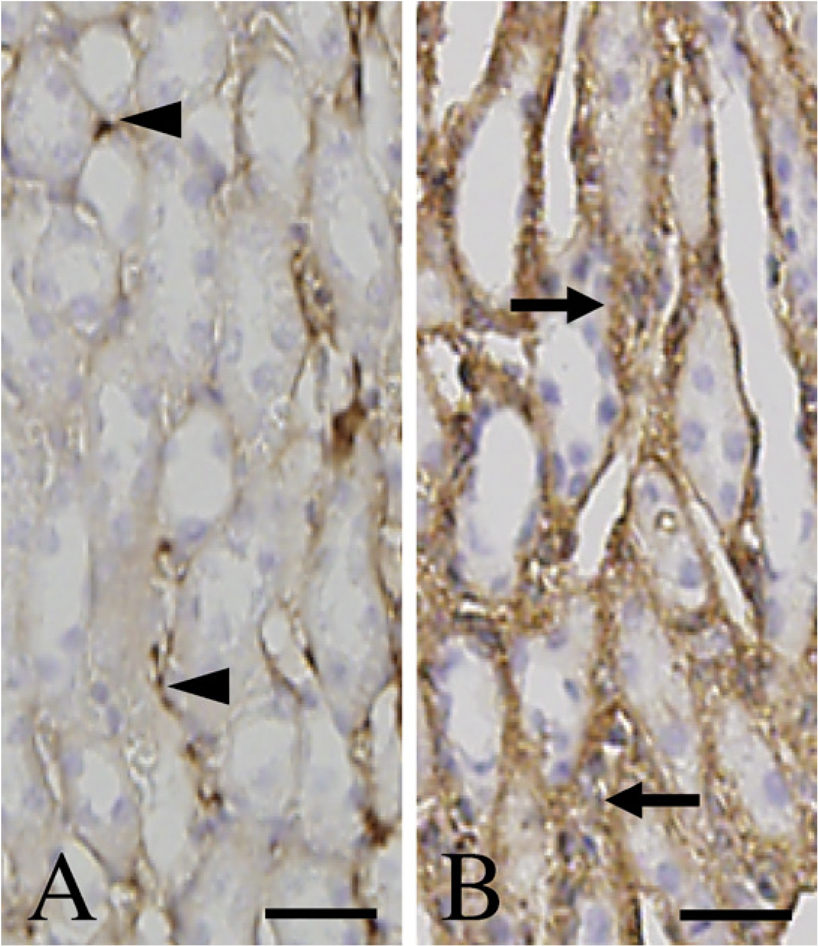
Immunolabeling of Twist in outer medulla of control and UUO day 7 kidneys. (
Next, we compared luminal areas of Twist+ tubules with those of Twist− tubules in the cortex of UUO day 7 kidneys. The luminal area of Twist+ tubules was 6.2 times as large as that of Twist− tubules (p<0.01) (Figure 4B), indicating that the luminal area of Twist+ tubules significantly enlarged compared with that of Twist− tubules.
Double-immunofluorescence Staining for Twist and Tubular Segment-specific Markers
Next we examined Twist expression at each tubular segment in UUO day 7 kidneys using double-immuno-fluorescence staining for Twist and tubular segment-specific markers because we demonstrated in Figure 4A that Twist immunoreaction on tubular epithelial cells significantly increased in both UUO day 3 and UUO day 7 kidneys. In UUO day 7 kidney, a part of AQP1+ tubular cells (Figures 5B and 5E, arrows) were simultaneously immunoreactive for Twist (Figures 5A and 5D, arrows). These results indicate that some tubular epithelia constituting the proximal tubules and the descending limb of Henle's loop expressed Twist 7 days after ureteral ligation (Figures 5C and 5F, arrows). Similarly, a part of NCC+ and NKCC2+ tubular cells (Figure 5H and Figure 6B, arrows) were also immunopositive for Twist (Figure 5G and Figure 6A, arrows) in UUO day 7 kidney, showing Twist expression in some tubular epithelia of the distal tubules and the ascending limb of Henle's loop (Figure 5I and Figure 6C, arrows). Furthermore, some AQP2+ tubular epithelial cells (Figures 6E and 6H, arrows) expressed Twist in UUO day 7 kidney (Figures 6D and 6G, arrows), indicating that Twist expression also occurred in the connecting tubules and the collecting ducts (Figures 6F and 6I, arrows). Thus, a part of tubular cells in whole nephron segment expressed Twist in UUO day 7 kidneys, whereas Twist expression was seldom detected in tubular epithelia of control kidneys shown in Figure 4A. Specific staining was absent without anti-Twist anti-body in the section of UUO day 7 kidneys (Figure 5J and Figure 6J).
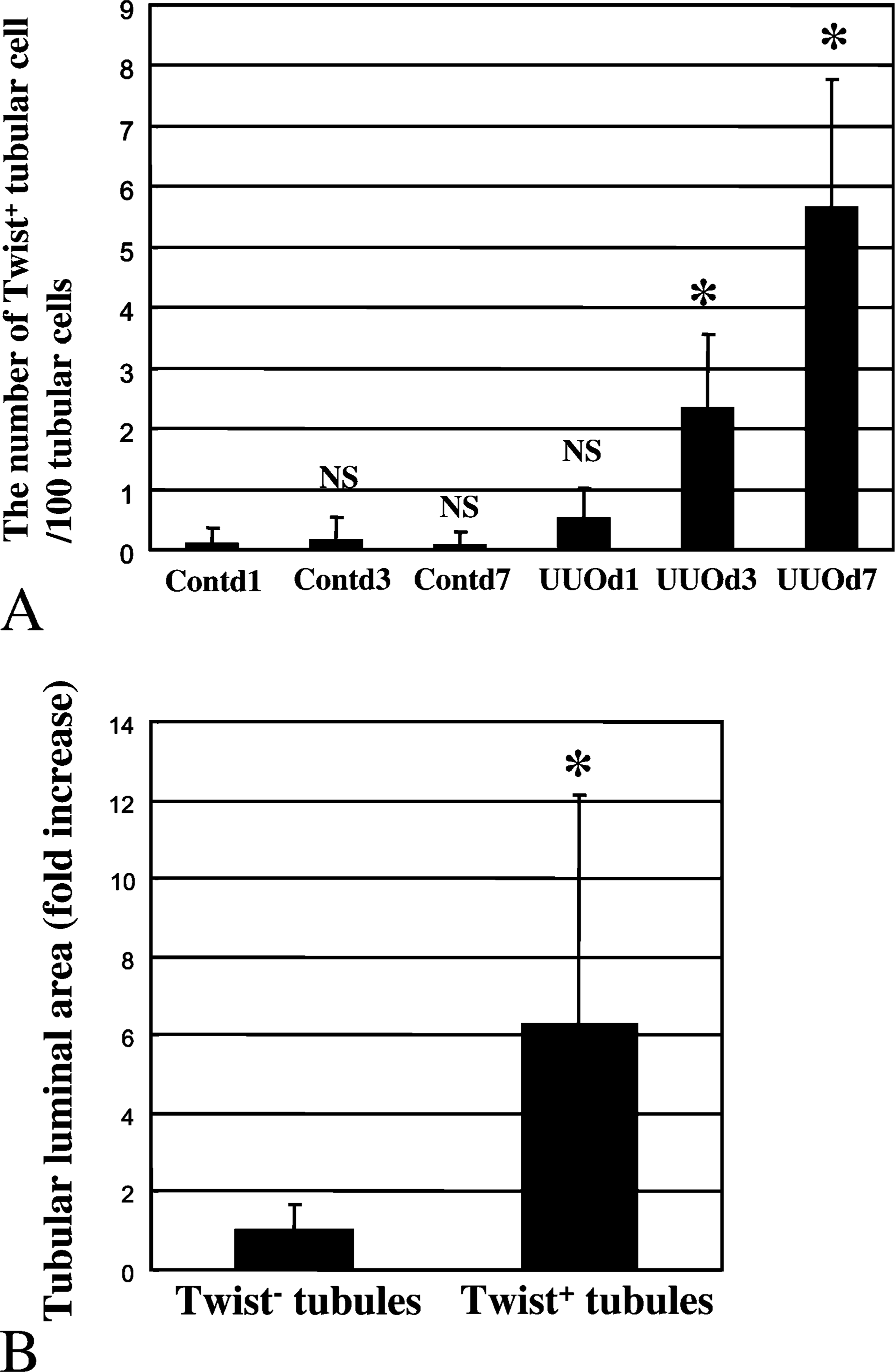
The number of Twist+ tubular cells in the cortex of both Cont and UUO kidneys (A) and the luminal area of both Twist+ tubules and Twist− tubular cells in the cortex of UUO day 7 kidney (
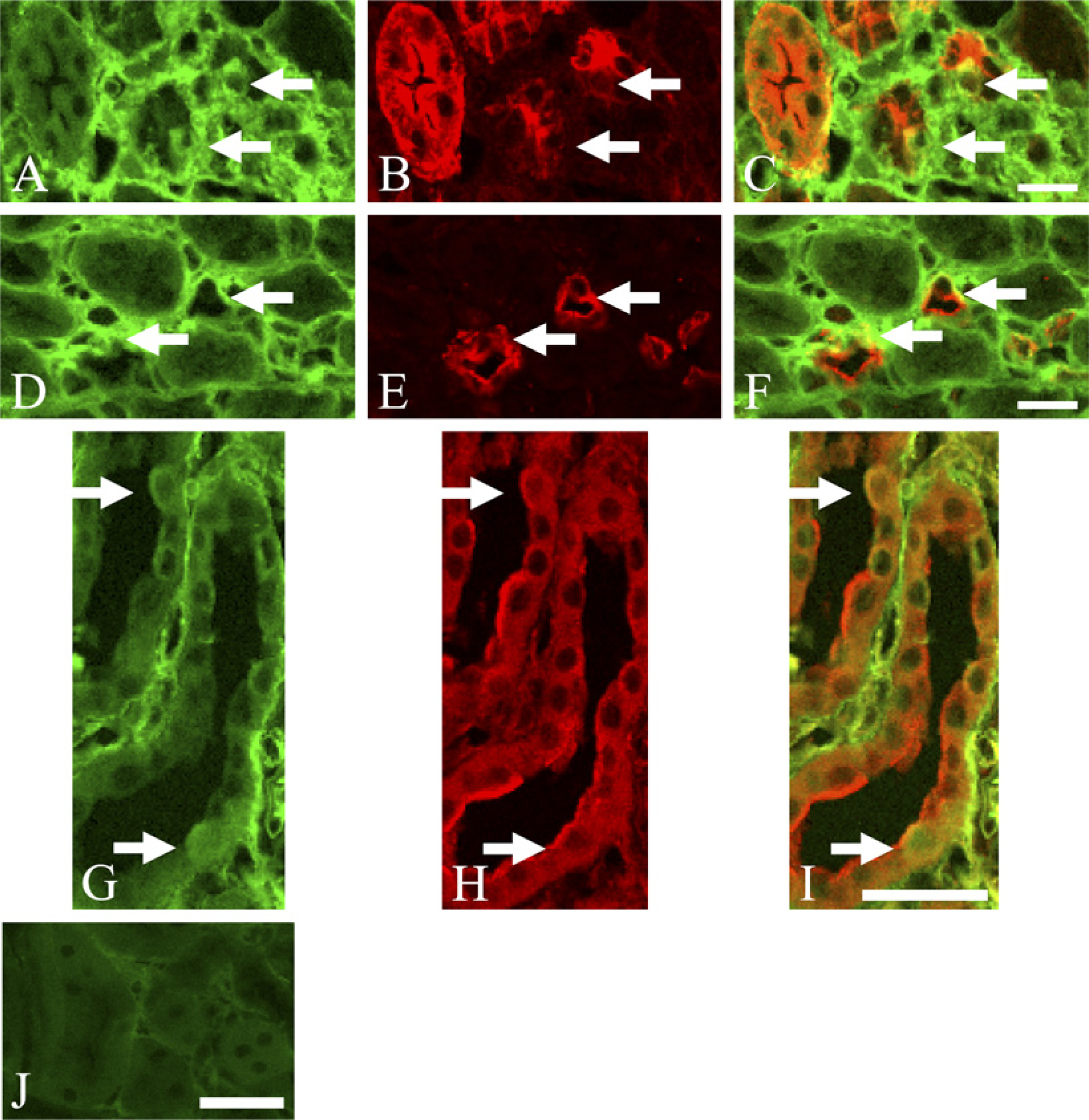
Double-immunofluorescence staining for Twist and anti-aquaporin-1 (
Analysis of the Degree of Tubular Twist Expression in Three Nephron Segments
In the cortex of UUO day 7 kidney, we analyzed the difference in the degree of tubular epithelial Twist expression at three nephron segments including the proximal tubule (AQP1+ tubule), distal tubule (NCC+ tubule), and connecting tubule/collecting duct (AQP2+ tubule). We counted the number of Twist+ tubular cells per 100 tubular cells in each nephron segment. The number of Twist+ tubular cells was 4.8 times higher in NCC+ tubular cells and 19.2 times higher in AQP2+ tubular cells than in AQP1+ tubular cells (p<0.01) (Figure 7) indicating that, in obstructed kidney, tubular epithelia in downstream segment of nephron more frequently expressed Twist than in upstream segment of nephron.
EMT of the Renal Tubular Epithelia Evaluated by Twist
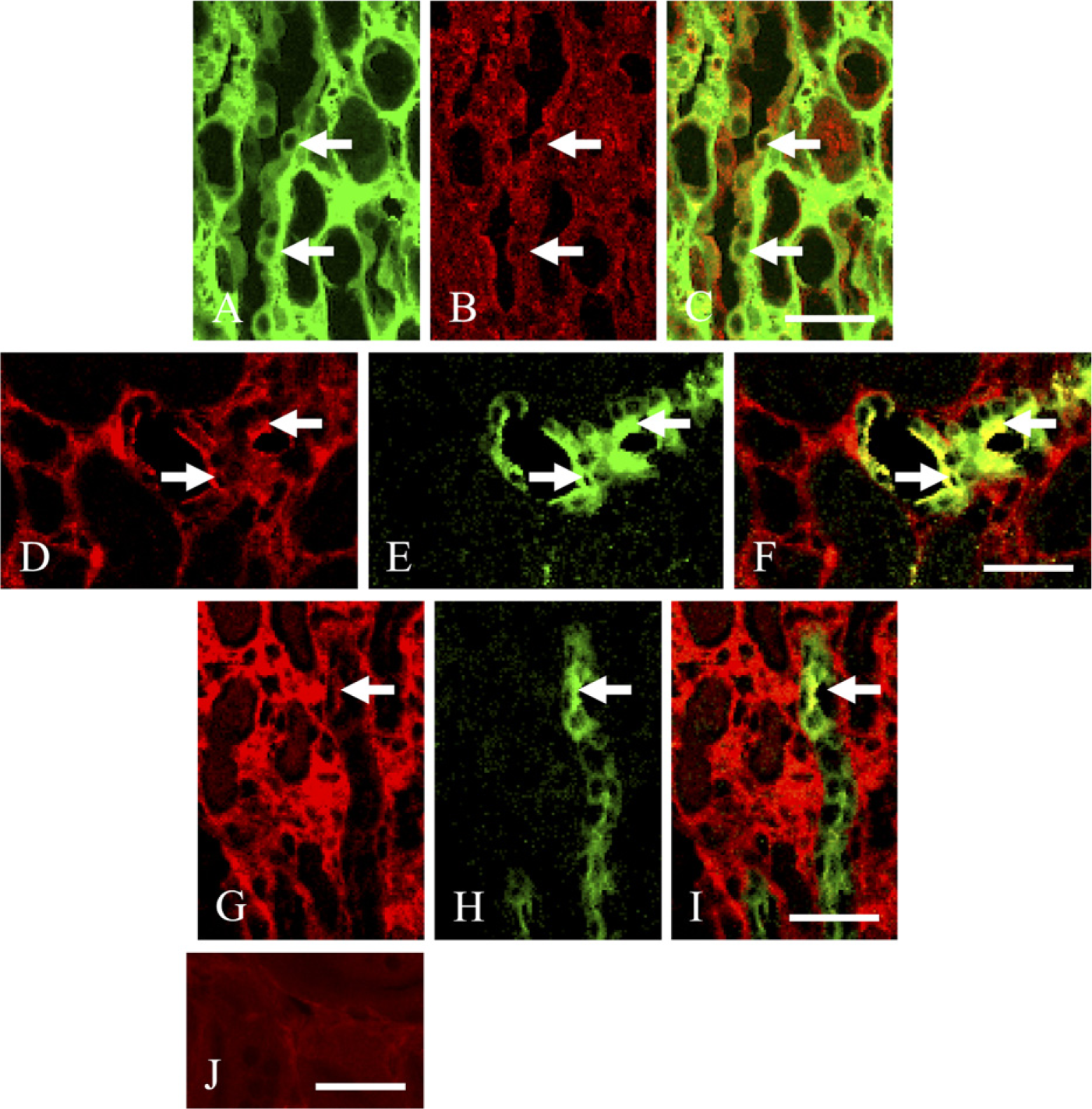
We analyzed the relationship between Twist expression and incidence of EMT in tubular epithelial cells. Expression of FSP1 is reported to identify tubular epithelial cells acquiring fibroblast phenotype in vitro (Strutz et al. 1995; Stahl and Felsen 2001). Zeisberg et al. (2003) reported that colocalization of FSP1 and the epithelial specific marker in renal tubular epithelia indicates EMT in mice with chronic renal injury. Thus, we examined whether Twist and FSP1 were coexpressed in tubular epithelial cells of UUO kidneys. We could not find any expression of Twist or FSP1 in tubular epithelia of control kidneys (Figures 8A-8C). Twist or FSP1 was only sparsely expressed in the inter-tubular area of control kidneys. Twist and FSP1 were coexpressed in some tubular epithelial cells in UUO day 7 kidneys (Figures 8D-8F, arrows). These results indicate that such epithelial cells would undergo EMT at 7 days after the ureteral ligation.
Expression of Twist in the Interstitial Space of UUO Kidneys
As demonstrated in Figures 2C and 2D, we observed a large amount of Twist expression in interstitial spaces of UUO day 3 and UUO day 7 kidneys. It is, in general, thought that αSMA+ myofibroblasts mainly synthesize interstitial extracellular matrix and induce interstitial fibrosis (Alpers et al. 1994). Therefore, we performed double-immunofluorescence staining for Twist and αSMA in UUO day 7 kidneys to inspect whether αSMA+ myofibroblasts express Twist. In control kidneys, αSMA immunoreaction was detected exclusively in the wall of vessels (data not shown), whereas we observed sparse coexpression of αSMA and Twist in intertubular areas of control kidneys (Figures 9A-9C, arrows). In contrast, we observed massive coexpression of αSMA and Twist in interstitial spaces of UUO day 7 kidneys (Figures 9D-9F, arrows), suggesting that Twist is mainly expressed in αSMA+ myofibroblasts located in interstitial spaces. We seldom detected αSMA expression in tubular epithelia of UUO day 7 kidneys (Figure 9E). Some cells in the glomerulus expressed both αSMA and Twist in UUO day 7 kidneys (Figures 9D-9F).
Double-immunofluorescence staining for Twist and NKCC2 or AQP2 in UUO day 7 kidney. (
Analysis of Cell Proliferation in Cont and UUO Kidneys
In UUO day 7 kidneys, Twist expression was frequently detected not only in tubular epithelia of dilated tubules in downstream of nephron but also in αSMA+ myofibroblasts of interstitial areas shown in Figure 4B, Figure 7, and Figure 9. Therefore, we examined PCNA expression in Cont and UUO kidneys to analyze the association between Twist expression and cellular proliferation. The luminal area of PCNA+ tubules significantly enlarged in UUO day 1 kidney compared with control (p<0.05) (Figures 10B and 10E). In UUO day 7 kidney, the luminal area of PCNA+ tubules enlarged further. The luminal area of PCNA+ tubules in UUO day 7 kidney was 15.3 times as large as that in control (p<0.01) (Figures 10D and 10E). Seven days after ureteral obstruction, there was a strong correlation between tubular dilation and tubular epithelial proliferation (r = 0.665, p<0.001) (Figure 10F). The number of PCNA+ interstitial cells significantly increased in both UUO day 3 and UUO day 7 kidneys, compared with control kidneys (Figure 10G). Thus, Twist expression might provide cell proliferation in the dilated tubules and the expanded interstitial spaces of UUO day 7 kidneys.
Discussion
Twist encodes a basic helix-loop-helix transcription factor initially found in Drosophila as one of the zygotic genes required for dorsoventral patterning and mesoderm differentiation during embryogenesis (Thisse et al. 1987; Leptin et al. 1992). The homozygous mutant Twist-null embryos die at 11.5 days with an obvious failure of neural tube closure (Chen and Behringer 1995). Twist has been well known as an essential factor in embryonic development. Actually, Twist gene has been described as being responsible for Saethre-Chotzen syndrome, an autosomal dominant craniosynostosis associated with cleft palate as well as other disturbances of the facial skeleton (El Ghouzzi et al. 1997; Howard et al. 1997). On the other hand, it has been reported that Twist plays an important role for EMT during palatogenesis (Bloch-Zupan et al. 2001). Yang et al. (2004) reported that expression of Twist in MDCK cells led to loss of cell-cell contacts and cell scattering. They observed complete loss of E-cadherin (typical epithelial marker) and appearance of mesenchymal markers including fibronectin, vimentin, and αSMA. Hence, both morphological and molecular changes in the Twist-expressing MDCK cells demonstrated that these cells had undergone an EMT. Thus, in vitro, it was confirmed that Twist contributed to the promotion of EMT in kidney epithelial cells. However, in vivo, it is unclear whether Twist relates to the occurrence of EMT in renal tubular epithelia in fibrous kidneys. Twist has never been analyzed as an EMT regulator in fibrosis of any organs. Therefore, we examined the relationship between Twist expression and renal fibrosis in UUO kidneys of adult mice.
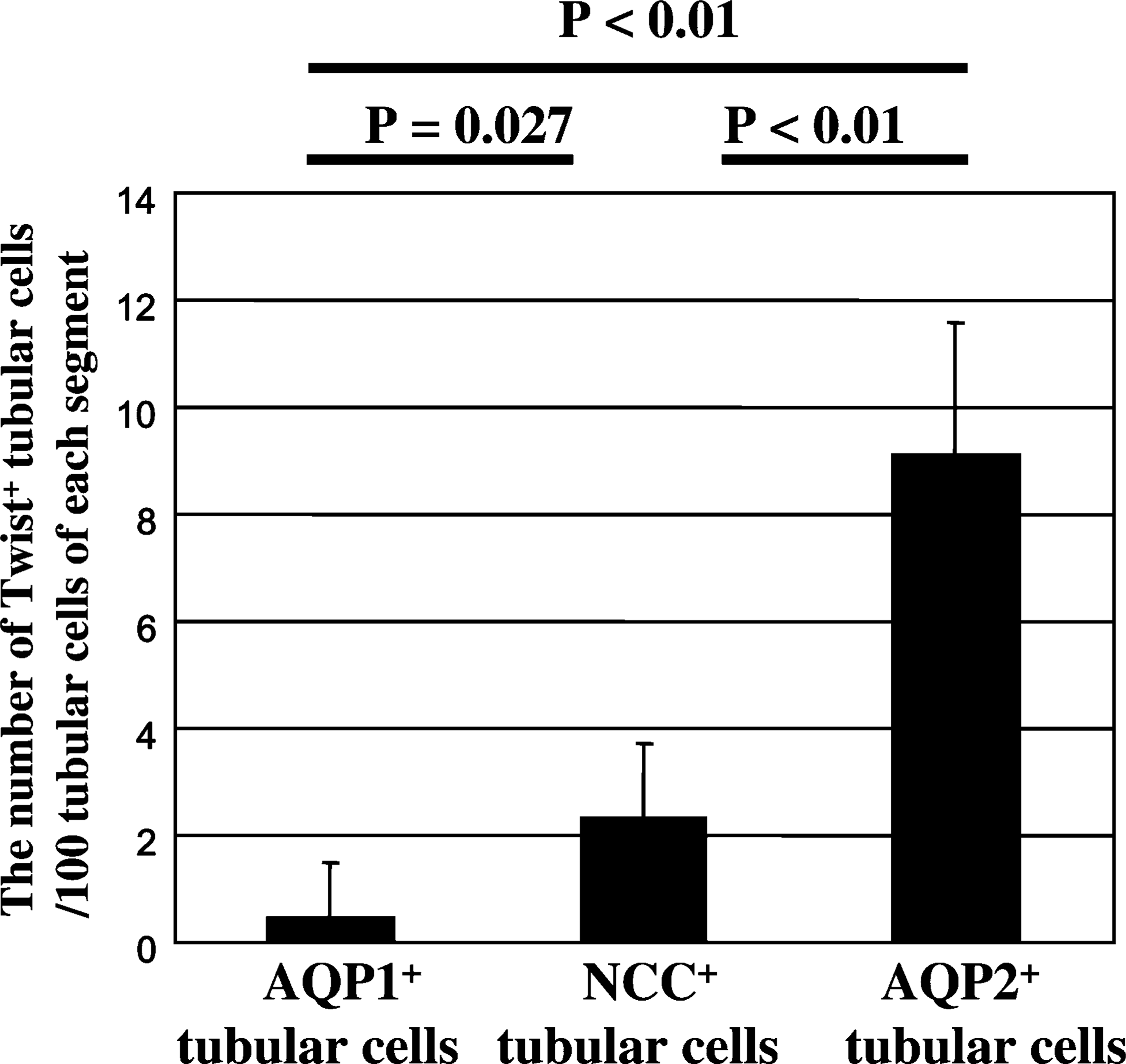
The number of tubular cells expressing Twist in three nephron segments in the cortex of UUO day 7 kidney. The number of Twist+ tubular cells per 100 tubular epithelia of each segment is counted. The results are represented as the mean ± SD (n = 3 mice). Compared with AQP1+ tubular cells, NCC+ or AQP2+ tubular cells significant frequently express Twist (AQP1+ tubular cells vs NCC+ tubular cells, p50.027, AQP1+ tubular cells vs AQP2+ tubular cells, p<0.01).
In the present study we revealed several important results about Twist expression in UUO kidneys as follows: (1) Twist transcription levels significantly increased 3 days after ureteral ligation as compared with control kidneys, (2) Twist expression increased in the tubular epithelia and in expanded interstitial spaces of UUO kidneys in a time-dependent manner, (3) tubular epithelia in downstream segment of nephron more frequently expressed Twist than in upstream segment of nephron in UUO kidneys, (4) some tubular cells coexpressed Twist and FSP1 in UUO day 7 kidney, that indicated Twist is involved in tubular EMT of obstructed kidneys, and (5) massive coexpression of Twist and αSMA was observed in interstitial spaces of UUO kidneys.
In our present study, the luminal areas of Twist+ tubules significantly enlarged, compared with those of Twist− tubules. In addition, tubular epithelia in downstream segment of nephron more frequently expressed Twist than in upstream of nephron of UUO day 7 kidneys. Ohashi et al. (2002) demonstrated that vascular endothelial growth factor (VEGF) was intensely expressed in dilated tubules 7 days after ureteral ligation. Intense VEGF expression in tubules of UUO kidneys would be a protective reaction to ischemic state of tubules due to decrease of microcirculating blood flow (Burt et al. 2007). Various kinds of stress such as hypoxic state might induce Twist expression in dilated tubules. In the present study it was shown that tubular epithelia tended to proliferate in dilated tubules of UUO kidneys. On the other hand, we previously demonstrated that tubular dilation of distal segment (from the ascending limb of Henle's loop to the collecting duct) of nephron develops tubular epithelial apoptosis caused by accumulated urine (Kida and Sato 2007). Twist expression in tubular epithelia might promote both tubular epithelial apoptosis and proliferation in dilated tubules of obstructed kidneys.
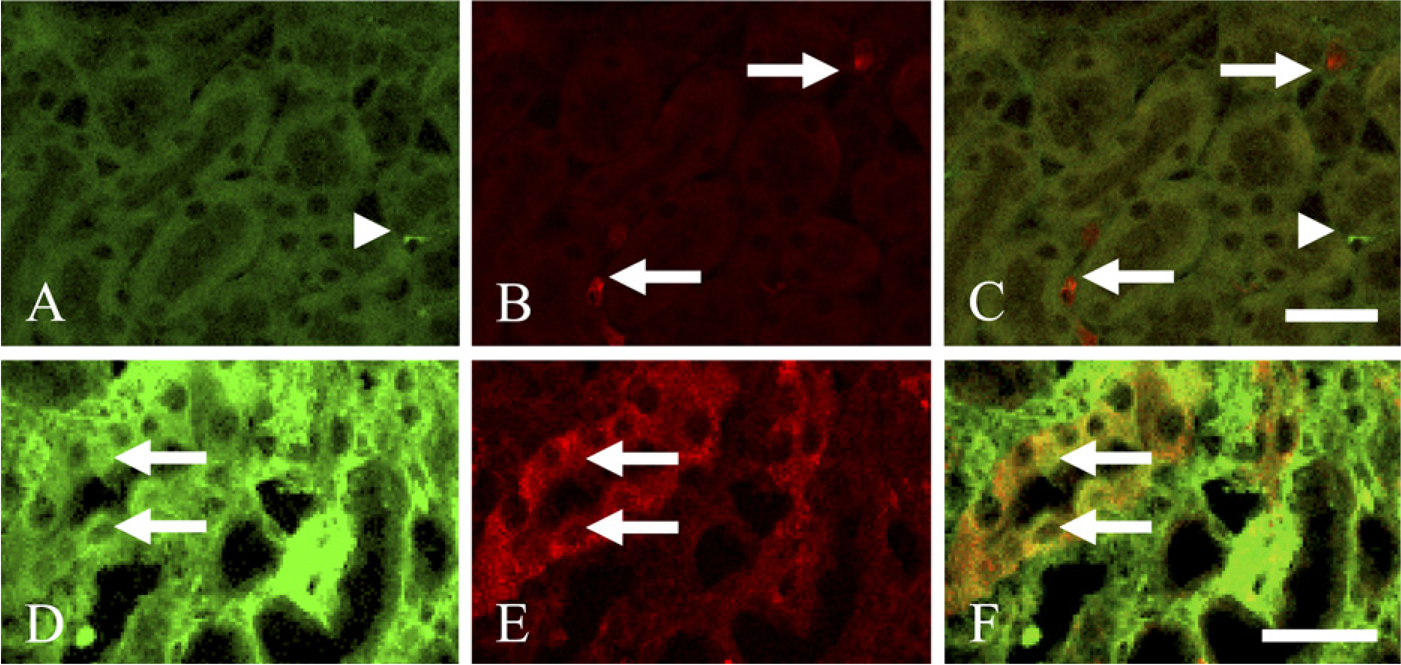
Double-immunofluorescence staining for Twist and fibroblast-specific protein 1 (FSP1) in control and UUO day 7 kidneys. (
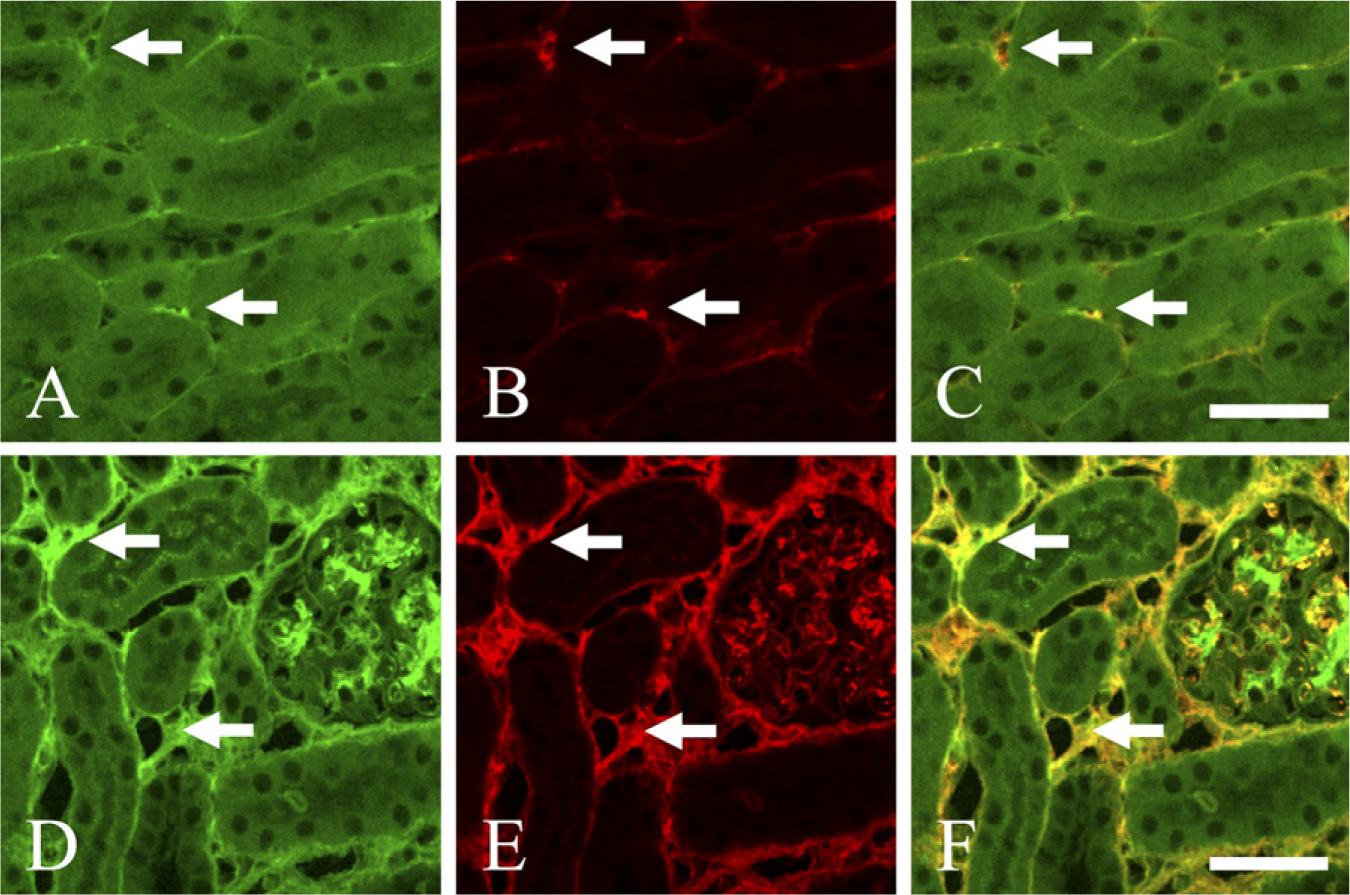
Double-immunofluorescence staining for Twist and α-smooth muscle actin (αSMA) in control and UUO day 7 kidneys. (
We found Twist+FSP1+ tubular epithelial cells in UUO day 7 kidneys, indicating that such epithelia were undergoing EMT. To our knowledge, this is the first in vivo report connecting Twist expression with renal tubular EMT in fibrous kidneys. Many investigators have understood tubular EMT as the coexpression of both αSMA and tubular marker because such tubular epithelia are regarded as a transitional stage between epithelia and mesenchyme (Yang and Liu 2001). However, according to Nishitani et al. (2005), double staining in kidney sections of patients with IgA nephropathy revealed that some tubular epithelia expressed FSP1, although no tubular epithelia were positive for αSMA, suggesting that FSP1 would be a more sensitive marker for detecting EMT of tubular epithelia than αSMA. Thus, in the present study we evaluated the occurrence of EMT in tubular cells of UUO kidneys using FSP1 instead of αSMA. In UUO day 7 kidneys we detected expression of FSP1 in some tubular epithelial cells, whereas αSMA immunoproducts were rarely observed in tubular epithelia. In UUO day 7 kidneys, αSMA expression pattern in tubular epithelia in our study was different from that reported in previous studies. The difference in mice strain might explain such disagreement of αSMA expression pattern in murine-obstructed kidneys.
Our experimental results unexpectedly demonstrated that Twist was massively expressed in many αSMA+ myofibroblasts located in the interstitial area of UUO day 7 kidneys. We showed that proliferation of interstitial cells in UUO day 7 kidneys significantly increased as compared with control kidneys. There is the possibility that Twist expression in αSMA+ myofibroblasts induces proliferation of myofibroblasts. In primary cultures of cells obtained from UUO day 25 rat kidneys, proliferation of fibroblasts significantly occurred regardless of the extent of fibrosis (Sommer et al. 1999), suggesting that fibroblasts obtained from UUO kidneys might acquire the new phenotype related to promote interstitial fibrosis. Moreover, exogenous Twist expression furthered colony formation of mouse embryonic fibroblasts (MEF) in soft agar, whereas untreated MEF were not able to form any colony, indicating that Twist-expressing MEF gained the ability to promote anchorage-independent growth (Maestro et al. 1999). It is likely that Twist might stimulate fibroblasts to change into αSMA+-activated myofibroblasts, which strongly produce extracellular matrix.
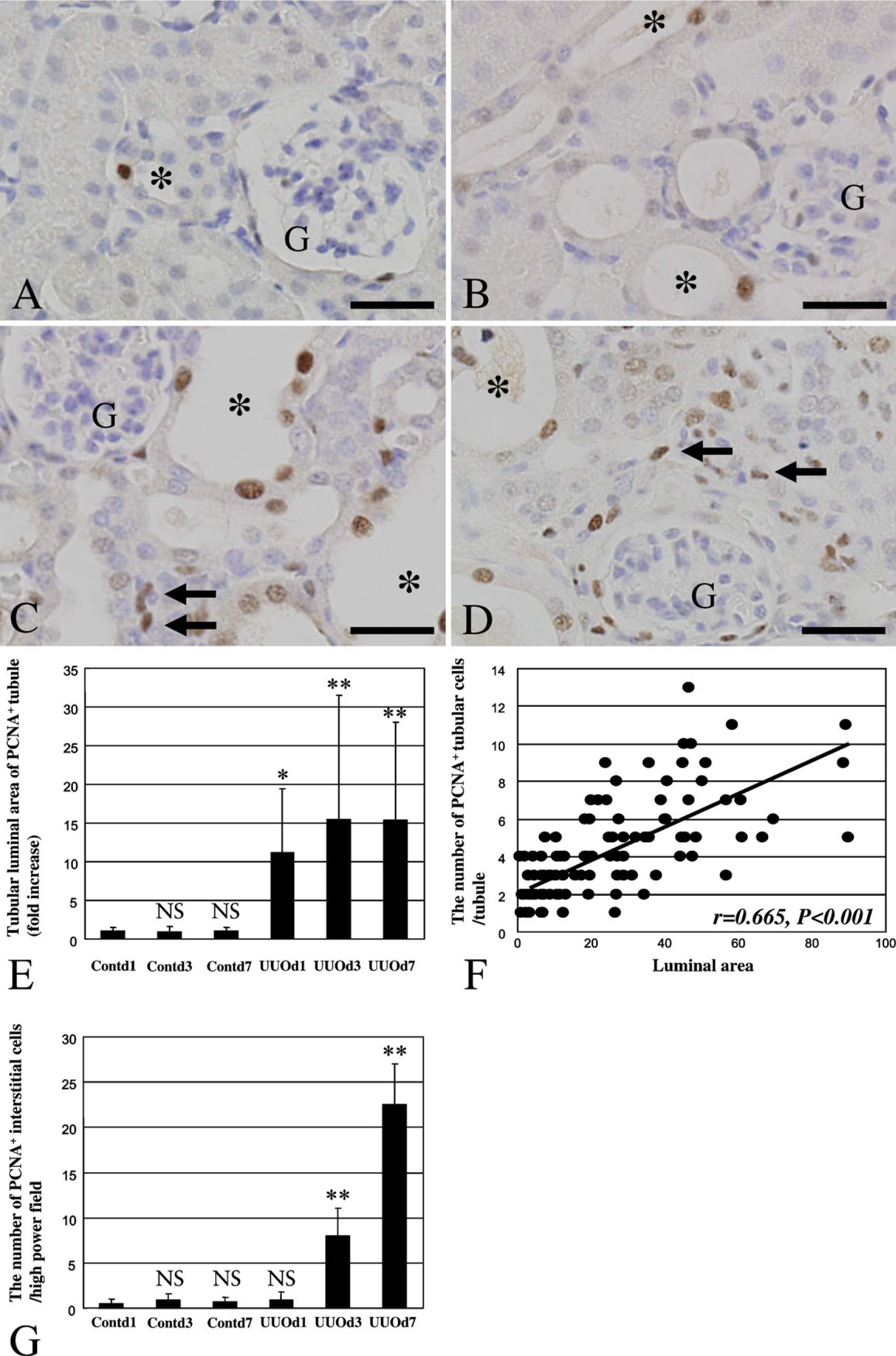
PCNA expression in Cont and UUO kidneys. (
In the present study we showed that the Wnt-2 mRNA level was increased 3 days after UUO. Thus, our present study is the first report examining the level of Wnt-2 mRNA in fibrous kidneys, although Wnt-4 was already reported to be upregulated in murine UUO kidneys (Surendran et al. 2004). Wnt-2 contributes to the regulation of endogenous Twist expression in the normal mammary gland (Howe et al. 2003). Using RT-PCR analysis, Watanabe et al. (2004) reported that both Wnt-2 and Twist mRNAs were upregulated in many breast cancers, whereas expression levels of both mRNAs were very low in most normal mammary glands. Our results were consistent with previous reports that Wnt-2 mRNA levels correlated with Twist mRNA levels. Twist directly or indirectly causes tran-scriptional repression of E-cadherin through the E-box elements on the E-cadherin promoter (Yang et al. 2004). Recently, it has been reported that CBF-A/KAP-1/FTS-1 complexes directly increase Twist expression and induce EMT in the proximal tubular epithelial cell line (Venkov et al. 2007). However, it largely remains unclear about upstream regulators or downstream signaling pathways of Twist.
Maestro et al. (1999) observed nuclear accumulation of Twist protein using a Twist-specific antibody. We found Twist-immunoreactive products not only in the nuclei but also in the cytoplasm of renal tubular epithelial cells and interstitial cells of diseased kidneys. It has already been revealed that the Twist antibody used in our present study has the precise ability of detecting mouse Twist antigen (Gitelman 1997). It may be possible that although Twist is active only in the nuclei, it also distributes in the cytoplasm. Nuclear factor-κB (NF-κB), for example, is located in the cytoplasm in an inactive form, bound to its inhibitor molecule, IκBα, in quiescent cells (Ross et al. 2004). Stimulation of cells through a variety of mechanisms triggers a cascade of signaling events that ultimately result in degradation of IκBα by the proteasome. This degradation releases active NF-κB, which then translocates into the nucleus where it binds to specific DNA sequences on its target genes (Ross et al. 2004). Although tropomodulin, another example, is highly expressed in the cytoplasm of striated muscles, Kong and Kedes (2004) found that it transfers into the nucleus and hampers the normal myogenic processes. Thus, Twist might shuttle between the cytoplasm and the nucleus. In addition, we showed that mRNA level of Twist in UUO kidneys was dramatically increased by use of real-time PCR, which supports the increase of Twist expression in UUO kidneys revealed by our IHC analysis with Twist antibody.
In summary, the present study revealed the localization of Twist and the association between Twist expression and tubular EMT in obstructed kidneys using real-time PCR and IHC. Twist is expressed both in tubular epithelia of dilated tubules and expanded interstitial spaces of obstructed kidneys. Tubular epithelia expressing Twist undergo EMT. The intense immuno-reaction of Twist was mainly recognized on αSMA+ myofibroblasts in the interstitial areas of fibrous kidneys in a time-dependent manner after ureteral ligation. Our findings suggest that Twist could be one therapeutic target for preventing progression of fibrosis occurring in diseased kidneys.
Footnotes
Acknowledgements
This work was supported in part by “Academic Frontier” Project for Private Universities: matching fund subsidy from the Ministry of Education, Cultures, Sports, Science, and Technology (2003–2007), a Grant-in-Aid for Scientific Research (No. 16659504, 17.05730) from the Ministry of Education, Cultures, Sports, Science and Technology, and a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science.
The authors are very grateful to Professor Kenjiro Wake (Liver Research Unit; Minophagen Pharmaceutical Co., Ltd., Tokyo, Japan) for his precise advice.
