Abstract
Introduction
Breast cancer, a type of solid tumour often associated with poor prognoses due to metastasis, frequently occurs in females worldwide. Metastasis is a complex multistep process that involves epithelial-mesenchymal transition (EMT), leading to the invasion of breast cancer cells to other organs. The invasion of epithelial cancer cells frequently accompanies the loss of epithelial features and the gain of mesenchymal characteristics. However, the molecular mechanisms underlying the pathogenesis of EMT remain unclear. A well-accepted and strong connection exists between malignant cancers and chronic inflammation in the tumour microenvironment (TME). Furthermore, previous reports have shown that the invasive phenotype of human carcinoma cells could be repressed by non-steroidal anti-inflammatory drugs (NSAIDs), 1 which indicates that these and other pro-resolving molecules are potential candidates for improving breast cancer therapy outcomes.
Lipoxin (LX), an important endogenous lipid mediator, is catalysed by transcellular pathways. The function of LX in inflammation and cancer has been widely investigated for several years. As an analogue of LXA4 with C-7-truncation and a shared receptor (formyl peptide receptor 2, FPR2/ALX), BML-111 is more stable and suitable for in vivo experiments. Previous research from our group revealed that BML-111 exerts inhibitory effects in hepatocarcinoma cells.2-4 The details of the mechanism of action for BML-111-mediated anti-tumour effects require further investigation.
5-lipoxygenase (5-LOX), a vital enzyme primarily expressed in leukocytes, can translocate from soluble cellular compartments to the nuclear membrane upon activation, and then catalyze the biosynthesis of leukotrienes (LTs) and specialized pro-resolving lipid mediators (SPM). 5 LTs can mediate inflammation and allergic response.6-8 Previous studies confirmed that the 5-LOX pathway is involved in the innate immune system and contributes to host defensive reactions.9-11 Furthermore, it has been reported that 5-LOX is overexpressed in malignant gastrointestinal tract cells, including the rectum, colon, pancreas, and esophagus.12-15
Here, we aimed to explore the mechanism of BML-111 on EMT and the migration of CoCl2-stimulated MCF-7 cells and BALB/c nude mice inoculated with MCF-7 in vivo. This research demonstrates that BML-111 inhibits the 5-LOX pathway to repress the EMT and migration of breast cancer cells.
Methods
Cell lines, animals, and reagents
The MCF-7 cell line was obtained from the American Type Culture Collection (ATCC, USA). Female BALB/c nude mice (18–22 g, 5–6 weeks old) were obtained from the Experimental Animal Center of Nanchang University. BML-111 (Cayman Chemical, USA) was stored at −80°C before it was diluted in a culture medium without serum. Anti-human/mouse antibodies (Santa Cruz Biotechnology, USA), including 5-LOX, 12-LOX, 15-LOX, vimentin, E-cadherin, MMP-2, MMP-9, and β-Actin, were used in this study. CoCl2 and butoxycarbonyl-Phe-Leu-Phe-Leu-Phe (Boc-2) were purchased from Sigma–Aldrich (USA) and GenScript Corporation (USA). A bicinchoninic acid protein (BCA) kit was used to determine the protein concentration of cell lysates (Pierce, USA). Cell culture medium and fetal bovine serum (FBS) were purchased from Gibco (Australia) and Gibco BRL (Switzerland), respectively. Hoechst 33258 stain was purchased from Novus (Burlington, MA, USA).
HE and immunohistochemistry of animal specimen
For the histological assay, animal specimens were embedded in paraffin after fixing in 10% buffered formalin, followed by cutting into 4 μm paraffin sections. After staining with haematoxylin and eosin (HE), a light microscope (Nikon Eclipse 90i, Tokyo, Japan) was used to evaluate conventional morphological phenotypes. For immunohistological analysis, the paraffin sections were treated with primary antibodies, including anti-5-LOX (1:100), anti-12-LOX (1:100), anti-15-LOX (1:100), anti-E-cadherin (1:100), anti-vimentin (1:100), anti-MMP-2 (1:100), and anti-MMP-9 (1:100), followed by staining with horseradish peroxidase-(HRP) conjugated secondary antibodies.
Cell culture and cell morphology
MCF-7 cells were cultured in RPMI 1640 medium with an addition of 10% heat-inactivated FBS, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES, 25 mmol/L, Sigma, USA), penicillin G (100 U/mL), and streptomycin (100 U/mL), and maintained in 5% CO2 + 95% air at 37°C, re-feeding the culture medium every 2–3 days. Cultured cells were then treated with the indicated conditions for 24 h, washed the coverslips with phosphate-buffered saline (PBS), and observed cell morphology using an inverted microscope.
Cell proliferation assay
MCF-7 cells (1 × 104 cells/well) were seeded into 96-well plates and cultured. After overnight incubation, the cells were starved in a culture medium deprived of FBS for 24 h, followed by culturing under the indicated conditions for 24 h. After treatment, 10 μL of 0.5 mg/mL MTT was added to each well of the plates and incubated for 4 h, followed by removing the supernatant and incubating the plate with 75 μL dimethyl sulfoxide (DMSO) for 15 min at 37°C. The absorbance was then measured using a wavelength of 492 nm with a micro-ELISA reader (Amersham Biosciences, USA).
Wound healing assay
MCF-7 cells were cultured to form a confluent monolayer on the surface of each well in a 6-well plate, followed by scraping a straight wound in each well with a sterile pipette tip. After treating the cells under the indicated conditions for 0 and 24 h, photographs were captured using a microscope.
Cell migration assay
Cell migration was assessed using transwells (Costar Corp., Cambridge, MA, USA) containing an extracellular matrix (50 mg filter, Sigma Chemical) and dried overnight. Meanwhile, the cells were treated under the indicated conditions and cultured for 24 h. After rehydrating the transwells for 1 h at room temperature, the treated cells were trypsinised, counted, and seeded into the upper chamber with 2 × 105 cells resuspended in RPMI 1640 containing 0.1% bovine serum albumin (BSA). The lower chamber was filled with 800 μL of RPMI 1640 medium containing 10% FBS and incubated at 37°C for 24 h. The underside of the filter was stained with HE after gently removing it, and cells on the other side of the filter were removed with a cotton bud. The cells were photographed and counted under an Olympus multifunctional microscope.
Western blotting analysis
MCF-7 cells were cultured for 24 h in 6-well plates, followed by overnight serum deprivation before treatment under the indicated conditions. The plates were then placed on ice while treated and untreated cells were harvested. Cell harvesting was performed by covering the plate with lysis buffer for 30 min, collecting the lysis buffer containing lysed cells, and centrifuging (12,000 g) cell lysates for 10 min at 4°C. After determining protein concentrations using the BCA protein assay, 10% SDS-PAGE electrophoresis was performed on 40 μg of protein extracts. After transferring the proteins onto a nitrocellulose membrane (Amersham Biosciences Inc., Piscataway, NJ), the membrane was blocked with skim milk containing Tween 20 for 1 h, then probed with antibodies, including human anti-5-LOX (1:500), anti-12-LOX (1:500), anti-15-LOX (1:500), and anti-β-Actin (1:500), at 4°C overnight. The probed membrane was then washed with TBS-Tween 20 three times, incubated with goat anti-rabbit IgG, conjugated with HRP for 1 h, and detected with enhanced chemiluminescence on an X-ray film. LabImage version 2.7.1 (Kapelan GmbH, Halle, Germany) was used to analyze the results. β-Actin was used as an internal/loading control. p-values were calculated to determine the significance of the band intensity between the control and treated groups.
Immunofluorescence
MCF-7 cells were seeded and cultured in 24-well plates to approximately 80% confluence before treatment under the indicated conditions for 24 h. This was followed by fixing the cells in cold methanol for 15 min and rinsing them with PBS three times. The fixed cells were then incubated with 0.5% Triton X-100 for 10 min to render the cells permeable. Subsequently, the cells were incubated with BSA at room temperature for 40 min before incubation with 5-LOX antibody (1:50 dilution) or PBS (negative control) at 4°C overnight. After rinsing with PBS three times, the cells were incubated with FITC or rhodamine-conjugated goat anti-rabbit or anti-mouse secondary antibody (1:100 dilution) for 1 h and washed with PBS three times. Finally, the cells were stained with Hoechst for 15 min, and fluorescent images were captured with a fluorescence microscope.
Reverse transcription polymerase chain reaction
Total RNA was extracted using TRIzol reagent (Invitrogen, Carlsbad, CA, USA), and the concentration was measured using an ultraviolet (UV) spectrophotometre (UV-1201; Shimadzu Corporation, Kyoto, Japan). For the reverse transcription reaction, 4 μg of total RNA was used to amplify the first-strand cDNA with a Thermal Cycler (Bio-Rad Laboratories, Richmond, CA, USA). Detailed descriptions of the primer sequences and reaction conditions are provided in Supplementary Table 1.
In vivo experiment
BML-111 evaluated its anti-tumour effects in vivo. All experiments involving BALB/c nude mice were approved by the Animal Care and Use Committee of Yangpu Hospital, School of Medicine, Tongji University. Based on previously published methods, 4 and considering experimental costs and statistical analysis, 20 nude mice were inoculated with MCF-7 cells into the bilateral oxters and randomly divided into two groups. After inoculation, mice were injected with PBS (MCF-7-bearing + PBS group) or BML-111 (0.1 mL, final dosage of 1 mg/kg, equals to 1/10 LD50) (MCF-7-bearing + BML-111 treated group). On day 21, after inoculation, mice were sacrificed and autopsied, with the tumours dissected. For morphological evaluation, tumour tissue slides were stained with H&E. For the immunohistochemical assays, protein expression in tumour tissues was detected by probing with anti-5-LOX, anti-12-LOX, anti-15-LOX, anti-E-cadherin, anti-vimentin, anti-MMP-2, and anti-MMP-9.
Statistical analysis
SPSS 18.0 software was used for calculating statistical significance. The data was represented as the mean ± SD of three independent experiments after one-way ANOVA analysis. A studentt test was used to compare the two groups. p < .05 was considered statistically significant.
Results
The effect of BML-111 on the morphology and viability of MCF-7 cell lines
To determine whether BML-111 inhibits the proliferation of breast cancer, MCF-7 cells were treated with 100–800 μg/L of BML-111. While BML-111 did suppress the proliferation of MCF-7 cells, these effects were not in a dose-dependent manner. We investigated the viability of MCF-7 cells following treatment with BML-111 for 6–36 h and observed that a few cells underwent necrosis after 12 h, 24 h, and 36 h; however, these effects were not time-dependent (Figure 1). Thus, we chose 400 μg/L and 24 h for the following experiments. BML-111 affected CoCl2-stimulated MCF-7 cell viability. MCF-7 cells were treated with BML-111 with different concentration and timepoint, and then incubated with MTT for 4 h and DMSO for 15 min, the absorbency was measured with amicro-ELISA reader. Shown are representative graphs of three independent experiments.
The effect of BML-111 on the EMT of MCF-7 cell lines
To explore the effect of BML-111 on EMT in MCF-7 cells, we first observed any changes in cell morphology after BML-111 treatment. As shown in Figure 2(a), when CoCl2 was administered, the epithelial morphology of the cells was lost, with a characteristic of dissociation from the epithelial clusters, and a mesenchymal phenotype was acquired, indicating that the EMT process occurred; BML-111 could block this process. However, when Boc-2 was combined with BML-111, the EMT-indicating morphology of MCF-7 cells was not inhibited by BML-111. BML-111 inhibited the epithelial-mesenchymal Transition (EMT) of CoCl2-stimulated MCF-7 cells. (a) MCF-7 cells were treated with indicated conditions. After 24 h, cells were observed by inverted microscope, Bar = 20 μm. (b1), (b2), (b3) MCF-7 cells were treated with indicated conditions. After 24 h, the expressions of E-cadherin and vimentin were detected by western blotting. *p < .05 versus Control group, #p < .05 versus CoCl2 group, and **p < .05 versus CoCl2 + BML-111 group. Shown are representative graphs of three independent experiments.
Next, we investigated epithelial and mesenchymal marker expression through western blotting. As shown in Figure 2(b), the protein level of E-cadherin was downregulated by CoCl2, while vimentin was dramatically upregulated; however, the protein level of E-cadherin was upregulated by BML-111, while vimentin was dramatically downregulated. Boc-2 co-treatment blocked the effects of BML-111.
The effect of BML-111 on the 5-LOX, 12-LOX, and 15-LOX expressions
To explore the inhibitory mechanism of BML-111 against EMT in MCF-7 cells, three isoforms of lipoxygenases (LOXs), 5-LOX, 12-LOX, and 15-LOX, were measured in response to treatment. The results showed that CoCl2 dramatically facilitated the expression of 5-LOX, which was decreased by BML-111 treatment. Moreover, the effect of BML-111 was blocked by a combined Boc-2 treatment. However, the expression of 12-LOX and 15-LOX did not change among the four treatment groups, indicating that only 5-LOX is involved in BML-111-mediated EMT in MCF-7 cells (Figure 3). BML-111 inhibited three kinds of lipoxygenases of CoCl2-stimulated MCF-7 cells (n = 3). (a) MCF-7 cells were treated with indicated conditions. After 24 h, the expressions of 5-LOX were detected by immunofluorescence, Bar = 20 μm. (b1), (b2), (b3), (b4) MCF-7 cells were treated with indicated conditions. After 24 h, the expressions of 5-LOX, 12-LOX, and 15-LOX were detected by western blotting. *p < .05 versus Control group, #p < .05 versus CoCl2 group, and **p < .05 versus CoCl2 + BML-111 group. Shown are representative graphs of three independent experiments.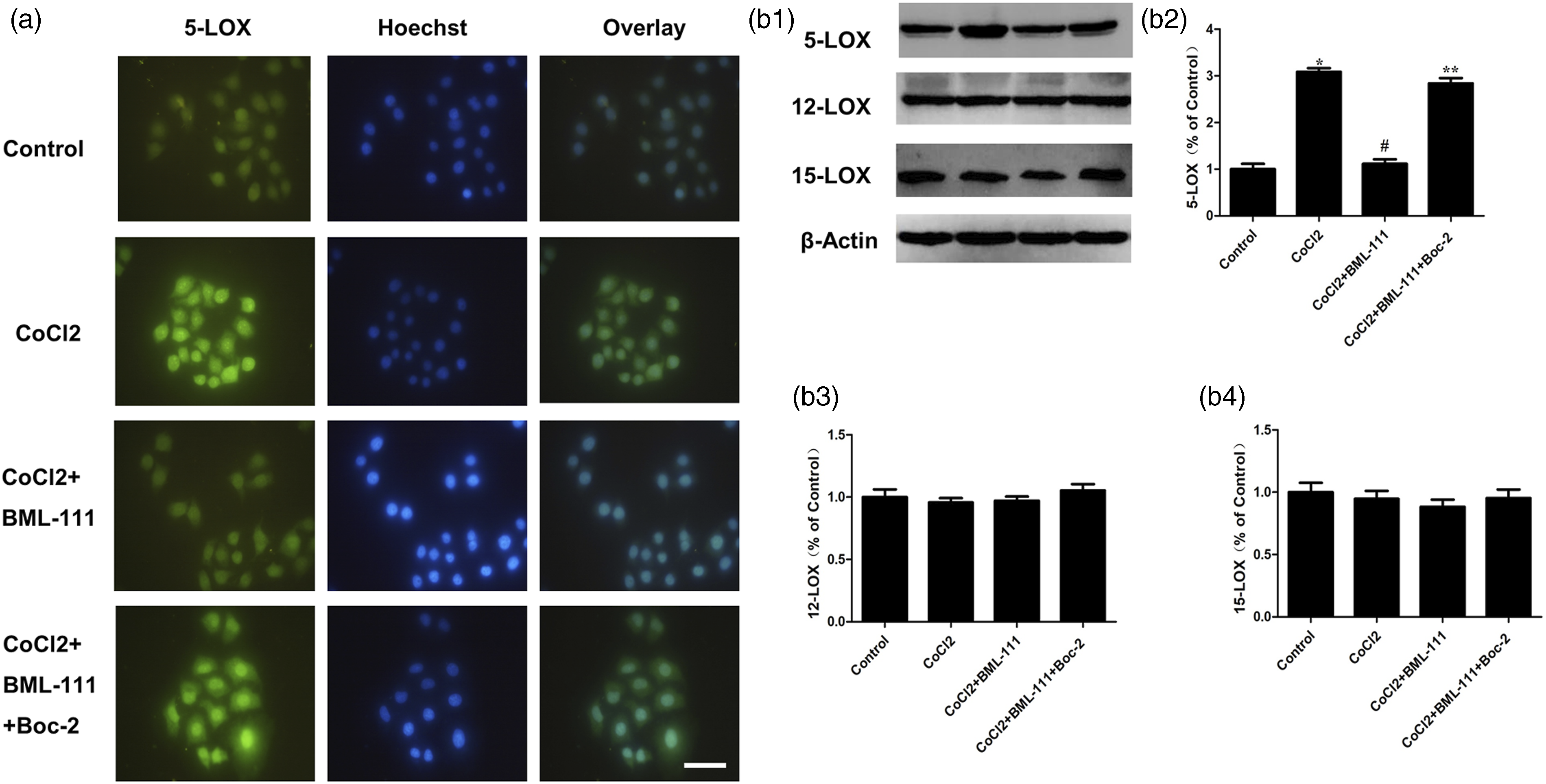
BML-111 inhibited the migration of MCF-7 cells induced by CoCl2
Wound healing and Transwell assays were conducted to investigate the migration of MCF-7 cells. The wound healing assay showed that the migration of MCF-7 cells was effectively induced by CoCl2, which BML-111 repressed; however, the effect of BML-111 was blocked by Boc-2 treatment (Figure 4(a)). BML-111 inhibited the migration of MCF-7 cells induced by CoCl2. (a1), (a2), BML-111 inhibited the migration of CoCl2-stimulated MCF-7 cells by wound healing assay. The scratch area at 0 h was arbitrarily assigned as 1.0. *p < .05 versus Control group, #p < .05 versus CoCl2 group, and **p < .05 versus CoCl2 + BML-111 group at 24 h point. (b1), (b2) BML-111 inhibited the migration of CoCl2-stimulated MCF-7 cells by transwell assay. Shown are representative images of three independent experiments. *p < .05 versus Control group, #p < .05 versus CoCl2 group, and **p < .05 versus CoCl2 + BML-111 group at 24 h point. (c1), (c2), (c3) BML-111 inhibited MMP-2 and MMP-9 expressions in CoCl2-stimulated MCF-7 cells by RT-PCR. Shown were representative graphs of three independent experiments. (d1, d2, d3) BML-111 inhibited MMP-2 and MMP-9 expressions in CoCl2-stimulated MCF-7 cells by western blot. *p < .05 versus Control group, #p < .05 versus CoCl2 group, and **p < .05 versus CoCl2 + BML-111 group. Shown are representative graphs of three independent experiments.
In the Transwell assay, the ability of MCF-7 cells to migrate through the membrane was remarkably strengthened by CoCl2 (p < .05), which was reversed by BML-111 (p < .05 vs CoCl2 group cells). Moreover, the effects of BML-111 were blocked by Boc-2 treatment (Figure 4(b)).
Next, we examined the mRNA expression of the migration and metastasis indicators, MMP-2 and MMP-9, using RT-PCR, which was dramatically enhanced by CoCl2 (p < .05 vs. control cells) (Figure 4(c1)–(c3)). However, these effects were markedly decreased by BML-111 (p < .05 vs. CoCl2 group cells).
As shown in Figure 4(d1)–(d3), we examined MMP-2 and MMP-9 protein levels through western blotting following the RT-PCR experiments. Protein expression of MMP-2 and MMP-9 was significantly increased by CoCl2 (p < .05 vs. control cells); however, these effects were decreased by BML-111 (p < .05 vs. CoCl2 group cells).
BML-111 inhibited LOXs and the expression of downstream molecules in vivo
To assess the in vivo response mediated by BML-111, tumour sizes and weights were first measured. As shown in Figure 5(a), (b1), and (b2), the tumour sizes and weights of the PBS group were much larger and heavier than those of the BML-111 group. This implies that BML-111 could repress tumour growth in BALB/c nude mice inoculated with MCF-7 cells. Effect of BML-111 on tumour morphology, 5-LOX and downstream molecule expressions in the primary tumour tissue of MCF-7 bearing BALB/C nude mice. (a) Representative images of the MCF-7 bearing nude mice before removing tumours. (b1), (b2) Representative images of the tumor tissue dissected from MCF-7 bearing nude mice. *p < .05 versus PBS group (c) The morphological feature, HE, 5-LOX, E-cadherin, vimentin, MMP-2, and MMP-9 staining were conducted, respectively. Bar = 20 μm. Shown are representative graphs of three independent experiments.
We examined the effect of BML-111 on three types of lipoxygenases in MCF-bearing BALB/c nude mice. Higher expression levels of 5-LOX and 12-LOX were observed in the tumour tissue of PBS group BALB/c nude mice (Figure 5(c)); however, BML-111 dramatically downregulated 5-LOX expression in inoculated BALB/c nude mice; however, it did not alter 12-LOX expression. Meanwhile, 15-LOX was negatively expressed in the PBS group of BALB/c nude mice, whereas BML-111 did not upregulate its expression (Figure 5(c)). Furthermore, we measured the expression levels of EMT indicators and MMPs, where BML-111 repressed the expression of vimentin, MMP-2, and MMP-9 while inducing the expression of E-cadherin.
Discussion
Chronic inflammation is involved in the pathogenesis of various cancers, including breast cancer, and is a common condition worldwide. We chose a human breast cancer cell line, MCF-7, as a model for our experiments. Anti-inflammatory drugs may exert anti-cancer effects on several cancers. 16 BML-111, an analogue of LXA4, exerts crucial functions in balancing pro-inflammatory cytokines, which may lead to a new approach for malignant tumours. As endogenous mediators of BML-111, lipoxins exert anti-inflammatory effects during tumour development.3,4,17,18 The peroxidation of arachidonic acid (AA) or linoleic acid (LA) can be catalysed by the dioxygenases, LOXs, which are involved in the inflammation of the tumour microenvironment, leading to the progression of cancers. 19 The COX and LOX metabolic pathways of AA have been implicated in carcinogenesis.20,21 Furthermore, certain human cancer cells and tissues have higher levels of 5-LOX and its metabolites. 22 COX- and LOX-generated eicosanoids could drive the proliferation of some cells, potentially driving the progression of carcinogenesis.23-25 Moreover, it might lead to possible therapeutic targets if further understanding of the molecular and cellular perturbations correlated with proliferation, transformation, and malignancy is acquired. 26
Therefore, we first explored the effects of BML-111 on MCF-7 cell proliferation. We observed that BML-111, in different concentrations, could inhibit proliferation and promote cell necrosis. This effect was observed at different time points. Moreover, EMT plays an important role in tumour pathogenesis, which can be facilitated by factors such as CoCl2, an inducer of hypoxia, leading to the induction of malignant tumour cell progression. Thus, repression of EMT-related pathways may be an effective approach for cancer treatment. Moreover, EMT can induce proteases such as MMPs to remodel the extracellular matrix (ECM) organization of the ECM. 27 Based on these results, the following in vitro and in vivo experiments were conducted.
To explore the molecular mechanisms behind BML-111 on the progression of breast cancer cells, 100 μM CoCl2 was applied, which promoted EMT in MCF-7 cells as shown by expression level changes of E-cadherin and vimentin. However, this effect was reversed by the BML-111 treatment. Furthermore, we demonstrated that BML-111 downregulated 5-LOX in vitro; however, it did not affect 12-LOX and 15-LOX. These results demonstrate that BML-111 could inhibit CoCl2-stimulated EMT in MCF-7 cells via the 5-LOX pathway in vitro.
As a vital feature of malignant tumours, neoplastic cell migration occurs via the degradation of extracellular matrix components, resulting in the migration of cells from the primary TME to a distant region. These results suggested that MCF-7 cell migration, which is enhanced by CoCl2 treatment, could be inhibited by BML-111. MMP-2 and MMP-9, two members of the endopeptidase family, are regulated by 5-LOX and play important roles in the TME, resulting in enhanced tumour migration and invasion. 28 Our results show that BML-111 downregulates the mRNA and protein levels of MMP-2 and MMP-9 in CoCl2-stimulated MCF-7 cells. Furthermore, we observed that the 5-LOX pathway was indispensable for migrating CoCl2-stimulated MCF-7 cells after treatment with BML-111 in vitro. Furthermore, the effect of BML-111 was blocked by Boc-2, an effective antagonist of the specific G-protein-coupled LXA4 receptor (FPR2/ALXR).
BML-111 and LXA4 share the same receptor (FPR2/ALXR), and the results of our previous studies demonstrated that BML-111 exerts similar functions to LXA4 in cancer cells.3,4,18 The results of in vivo experiments with MCF-7-bearing nude mice showed that the tumour size in the BML-111 group was 40% smaller than that in the control group, suggesting that BML-111 could repress tumour growth. We further identified the most important index of EMT: E-cadherin was dramatically induced, whereas 5-LOX, MMP-2, and MMP-9 were dramatically repressed in the BML-111 group. We observed that the two groups did not differ between the 12-LOX and 15-LOX expression levels. We confirmed that 5-LOX downregulated the expression of E-cadherin, and BML-111 could inhibit the EMT of breast cancer cells via the 5-LOX pathway in vivo.
Our study had several limitations. First, we selected only one cell line for the in vitro experiments, and the effect of BML-111 on breast cancer cells should be verified in other cell lines. Second, we only evaluated the effect of BML-111 on wild-type cell lines; RNA knockdown experiments should be conducted to silence 5-LOX. In-depth research needs to be carried out.
Conclusion
In summary, our data demonstrated that BML-111 effectively suppressed MCF-7 proliferation, EMT, and migration in vitro and in vivo. These results pave the way for tumour therapy, in which BML-111 exerts anti-tumour effects on breast cancer cells, and blocking 5-LOX could be a potential approach for breast cancer therapy.
Supplemental Material
Supplemental Material - BML-111, the agonist of lipoxin A4, suppresses epithelial-mesenchymal transition and migration of MCF-7 cells via regulating the lipoxygenase pathway
Supplemental Material for BML-111, the agonist of lipoxin A4, suppresses epithelial-mesenchymal transition and migration of MCF-7 cells via regulating the lipoxygenase pathway Fen Xu, Xiaoyan Zhou, Lan Lin, Jing Xu, Yu Feng, Yuanqiao He and Hua Hao in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the project of Shanghai Municipal Health Commission (No.202140232), Climb Plan of Tongji University Affiliated Yangpu Hospital (No.Ye2202105), Project of College-level Key Discipline of Tongji University Affilliated Yangpu Hospital (No.2023YJXK01) and a “Bringing in talent scientific starting project” grant of Tongji University Affiliated Yangpu Hospital.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
