Abstract
L1 cell adhesion molecule (L1CAM) is a transmembrane molecule belonging to the L1 protein family. It has shown to be a key player in axonal guidance in the course of neuronal development. Furthermore, L1CAM is also crucial for the establishment of the enteric and urogenital organs and is aberrantly expressed in cancer originating in these organs. Carcinogenesis and embryogenesis follow a lot of similar molecular pathways, but unfortunately, comprehensive data on L1CAM expression and localization in human developing organs are lacking so far. In the present study we, therefore, examined the spatiotemporal distribution of L1CAM in the early human fetal period (weeks 8–12 of gestation) by means of immunohistochemistry and in situ hybridization (ISH). In the epithelia of the gastrointestinal organs, L1CAM localization cannot be observed in the examined stages most likely due to their advanced polarization and differentiation. Despite these results, our ISH data indicate weak L1CAM expression, but only in few epithelial cells. The genital tracts, however, are distinctly L1CAM positive throughout the entire fetal period. We, therefore, conclude that in embryogenesis L1CAM is crucial for further differentiation of epithelia.
Introduction
In 1984, Rathjen and Schachner identified a new cell adhesion molecule (L1 cell adhesion molecule [L1CAM]) in neurons that turned out to be a key player in the neuronal development of mice where it governs neuronal outgrowth, migration, and axonal guidance. 1 L1CAM is a transmembrane protein belonging to the immunoglobulin superfamily, interacting with several ligands such as integrins, neuropilins, contactin 1 and 2, CD24, as well as with its shed form. 2
Apart from its established function, studies in rodents and cell lines showed that L1CAM has an essential role in the formation and maintenance of epithelia, which is achieved via two contrary modes of action: On the one hand, L1CAM is important for both the establishment of cell-cell complexes and the polarization of epithelial cells. On the other hand, it is crucial for cell migration during development. 3 Thus, L1CAM is an important issue in the formation of the gastrointestinal and urogenital organs, and its critical role in this process has been emphasized in studies of congenital defects. It has been shown that inherent functional defects of L1CAM are associated with failure of the proper development of the renal system and the autonomous enteric nervous system. 3 This is corroborated by further studies revealing an association between aganglionic diseases of the enteric system and L1CAM dysfunction.4,5
Beside its developmental effects, the significance of L1CAM in malignant diseases has considerably increased over the years.6,7 Overexpression of L1CAM in cancerous tissue, for instance, is associated with a poor histological differentiation of the tissues, accelerated cell cycle and metastasis, all together responsible for a poor clinical outcome.3,6-9 Furthermore, studies on various types of cancer of the gastrointestinal and urogenital systems have emphasized the connection between an aberrant upregulation of L1CAM protein, enhanced epithelial-mesenchymal transition (EMT), and consequently the metastatic capacity of these malignancies. 10
It is well known that embryogenesis and carcinogenesis share numerous similar molecular pathways, and cancer development is frequently correlated with a disrupted embryonic program.11-13Cancerous tissues often reexpress molecules which have significant functions in embryonic and fetal morphogenesis and are consecutively silenced in healthy adult tissue. Such molecules to which L1CAM belongs gain importance in diagnosis and prognosis as well as antibody-based therapy of carcinomas. Nonetheless, further comprehensive studies are needed to gain an insight into the role of L1CAM during development as well as during carcinogenesis before L1CAM can be used in daily clinical routine.
Several studies14-17 focused on L1CAM localization in organ systems but were exclusively conducted in rodents, avian, and cell lines. In the present study we, therefore, aimed at the spatiotemporal localization and expression of L1CAM in humans. We focused on the enteric and urogenital system and investigated samples from gestational weeks 8 to 12. Furthermore, we examined adults to assess the spatiotemporal alterations during the aging process of tissues.
Materials and Methods
Embryonic, Fetal, and Adult Human Specimens
Human embryos and fetuses (n = 24) from week (w) 8 to the end of w12 after gestation (4/w8; 4/w9; 4/w10; 6/w11; 6/w12), correlating to the anatomical age were examined. They were obtained as parts of legal abortions according to Austrian law (§ 97 StGB) and with informed written parental consent and were approved by the ethical committee of Innsbruck Medical University, Austria, according to the Declaration of Helsinki. The specimens showed no macroscopic abnormalities, and their embryological age was defined by measurement of the crown–rump length (CRL), external and internal morphology, and the estimated gynecological age. Adult human specimens (n = 4) were obtained from fresh cadavers (post-mortem time was less than 8 hr) with no known dysfunction regarding the examined tissue. Tissue from a newborn specimen was obtained from the archival collection of the Division of Clinical and Functional Anatomy Innsbruck. These bodies were donated to the Division of Clinical and Functional Anatomy of the Medical University of Innsbruck by people who had given their informed consent for their use for scientific and educational purposes prior to death.18,19 Due to immediate anonymization of the cadavers, no certificate of non-objection was needed.
Tissue Preparation for Histology, Immunohistochemistry, and In Situ Hybridization on Paraffin Sections
Embryos/fetuses immediately after abortion and tissue samples (enteric and urogenital system) of adult human specimens were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS, 0.1 M), pH 7.4, overnight at 4C, followed by rinsing in PBS. Samples were then dehydrated and paraffin embedded using a routine histological infiltration processor (Shandon Excelsior ES Tissue Processor; Thermo Fisher Scientific, Waltham, MA). Serial sections (4 µm) were made on a HM 355S microtome (Microm, Walldorf, Germany) and mounted on SuperFrost Plus slides (Menzel, Braunschweig, Germany). Sections were dried overnight at room temperature. Subsequently, the slides were incubated at 60 C for 2 hr to adhere the sections firmly to the glass slides. For histological analysis, every 10th section of a complete series from each anatomical age was stained with hematoxylin and eosin (H&E) (Shandon Varistain 24-4, Histocom Vienna, Austria).
Antibody
The L1CAM-specific MAb L1-14.10 (10µg/ml) was provided by Peter Altevogt, Skin Cancer Unit 300G, German Cancer Research Centre, Heidelberg, Germany. The monoclonal antibody L1-14.10 was obtained after immunization of mice with human L1-Fc protein comprising the ectodomain of L1. 20
Immunohistochemistry
IHC was performed on paraffin sections in a Ventana Roche Discovery XT Immunostainer (Mannheim, Germany) according to the DAB-Map discovery research standard procedure. Antigen retrieval was initiated by heat-induced epitope unmasking, while sections were immersed in ethylenediaminetetraacetic acid buffer (Cell Conditioning Solution CC1, Ventana 950-124). A standardized staining protocol for L1CAM on formalin-fixed, paraffin-embedded tissues was used. 21 In brief, sections were incubated with the primary antibody (1:5000) at 37C for 1 hr. After that, a biotinylated cocktail of goat anti-mouse IgG, goat anti-mouse IgM, and goat anti-rabbit IgG as well as a protein block (Discovery Universal Secondary Antibody, Ventana 760-4250) was applied at room temperature for 32 min. Detection of the antigen–antibody complex was achieved using the DAB-Map Detection Kit (Ventana 760-124) with the standard streptavidin–biotin–peroxidase complex system and diaminobenzidine (DAB) as a substrate with copper enhancement. Sections were subsequently counterstaining with hematoxylin (Ventana 760-2021), dehydrated in graded ethanol series, cleared in xylene, and mounted permanently with Entellan (Merck, Darmstadt, Germany).
Brain samples served as positive controls and were included in each experiment. In addition, peripheral neuronal structures in the urogenital and enteric system revealed L1CAM localization and thus served as an internal positive control. Negative controls were obtained by substituting the primary antibody with a reaction buffer or an isotype matching control IgGs. These controls yielded no staining.
In Situ Hybridization
In situ hybridization (ISH) was performed on paraffin sections in a Ventana Roche Discovery Immunostainer according to the “research ISH BlueMap XT” standard procedure. For pretreatment, ISH Proteinase 3 (Ventana 780-4149) and RiboCC—a citrate-based buffer (pH 6.0)—(Ventana 760-107) were used. Sections were incubated with a L1CAM-specific antisense oligonucleotide which reads as follows: TGG ACA ACG TAG ATG TAG GCA. Oligonucleotides were tailed with DIG-11-dUTP and recombinant terminal transferase according to the manufacture’s protocol (DIG Oligonucleotide Tailing Kit, 2nd generation, Roche 03353583910). Hybridization conditions were adjusted using Ventana guidance for standard protocols with regular RiboMap kit (Ventana 760-102) conditions. In brief, hybridization was performed at 52C for 6 hr, followed by 2 washes with 2.0× saline sodium citrate (SSC; Ribowash, Ventana 760-105) at 52C, each for 8 min, followed by 1 wash with 0.1× SSC at 52C for 8 min. Sections were subsequently incubated with a mouse monoclonal anti-DIG/biotonylated antibody. The detection was achieved using the BlueMap Detection Kit (Ventana 760-120), and sections were counterstained with nuclear fast red (Red Counterstain II, Ventana 780-2218) for 4 min. Sections were dried, cleared in xylene, and cover-slipped.
Hybridization probes were designed by use of Ensemble Genome Browser (http://www.ensembl.org/index.html) and Exiqon’s “Custom LNA™ mRNA Detection Probe design tool” (http://www.exiqon.com/ls). Consequently, the attained nucleotide sequence was compared with sequence databases, and the statistical significance of these matches was calculated (http://blast.ncbi.nlm.nih.gov/Blast.cgi?CMD=Web&PAGE_TYPE=BlastHome). This query revealed 100% for Homo sapiens L1CAM messenger RNA (mRNA) transcript variants. The sense oligonucleotide sequence served as negative control. The controls yielded no labeling.
Results
In the present investigation, we basically focused on the L1CAM expression and localization in epithelia of the digestive and reproductive organs. In several images, L1CAM positive nerve fibers were also shown; they were not specifically mentioned in the result section, but rather served as internal positive control.
ISH was only performed on older developmental stages (w11 and 12, respectively) due to the fact that only few samples/sections from younger stages were available (see Material and Methods) which we used for IHC and H&E staining.
IHC in Digestive Organs in the Period From Week (W) 8–12
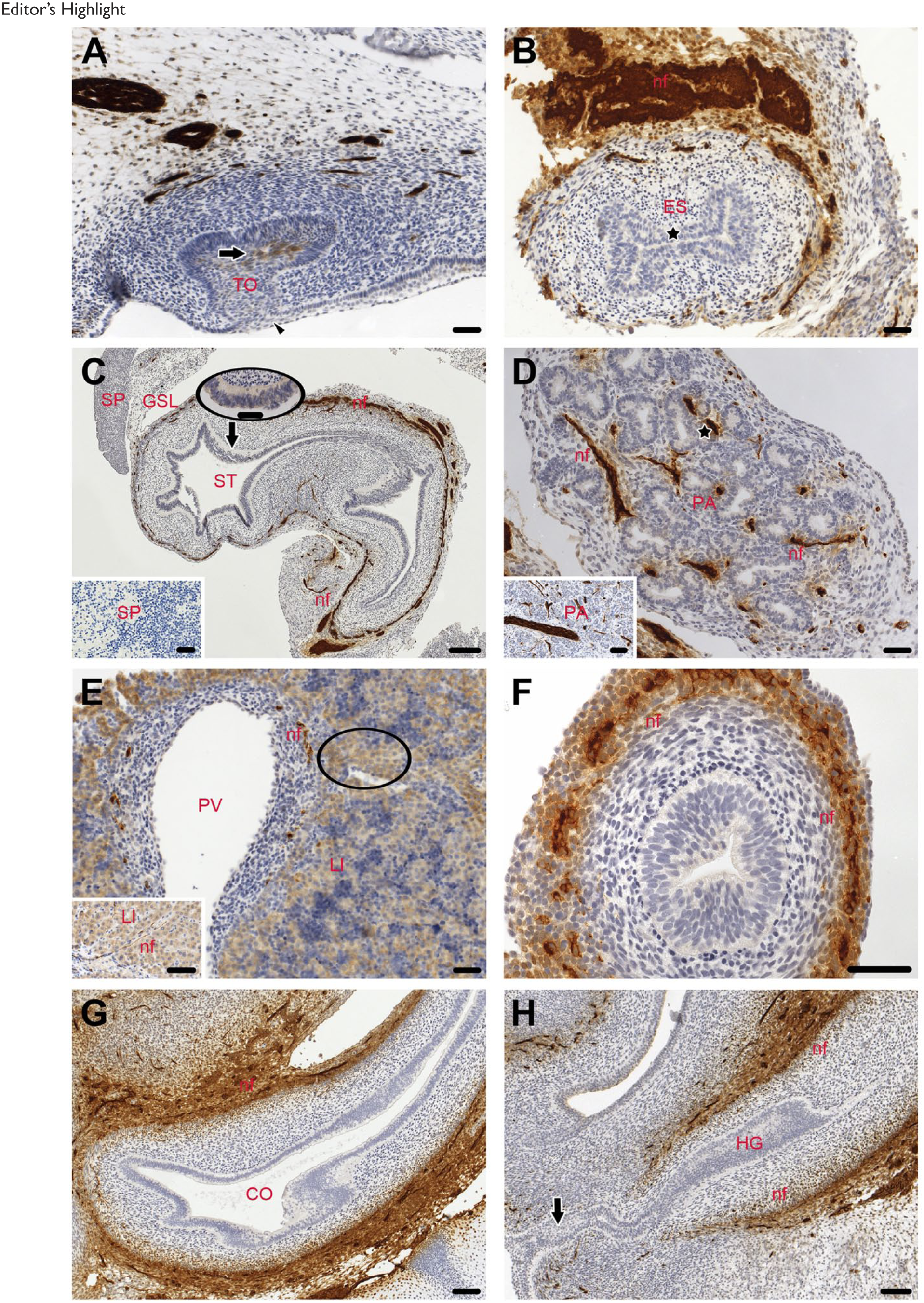
In the oral cavity, the deciduous teeth were not yet visible throughout the examined gestational period. They were in the cap stadium, defined by a mesenchymal densification surrounded by a columnar epithelium which was connected to the squamous epithelium of the lips (Fig. 1A arrowhead). L1CAM staining was only visible in the mesenchyme of the dental papilla anlage (Fig. 1A arrow).
Editor’s Highlight
At w10, the esophagus was the first time canalized and showed a slit-like lumen, lined by a squamous epithelium (Fig. 1B asterisk) that was L1CAM negative up to w12.
The endodermal-derived stomach was composed of a columnar epithelium and revealed no staining for L1CAM from w8 until w12 (Fig. 1C). Next to the stomach the spleen, a mesodermal derivate, was present and they were connected via the gastrosplenic ligament. Both the spleen and the ligament were L1CAM negative. Similarly, in adults neither the spleen (Fig. 1C inset) nor the gastric epithelium (data not shown) showed L1CAM staining.
Next we investigated the glands (pancreas and liver). In the pancreas at w8, the excretory ducts and the adjacent tubules (asterisk), which originate from the pancreatic bud epithelium, were already present and displayed no staining for L1CAM. Furthermore, the mesenchyme was L1CAM negative but nerve fibers showed a strong staining for L1CAM (Fig. 1D). This was also seen in the adult pancreas (Fig. 1D inset). Throughout the examined period, the hepatocytes displayed a weak L1CAM staining in their membranes. The unspecific faint cytoplasmic staining in numerous hepatocytes was presumable due to high concentrations of endogenous biotin. A similar staining pattern was observed in the adults.
Subsequently, we examined the intestine. In the entire intestine (small and large intestine), L1CAM was not localized in the epithelia from w8 to 12 (Fig. 1F and G). In addition, no labeling was detectable in the hindgut, the lumen of which was closed by an epithelium plug 22 (Fig. 1H arrow).
Taken together, the epithelia of the digestive organs clearly displayed no staining for L1CAM. One explanation could be that the protein concentration was below the detection range of the antibody, and we therefore performed ISH in order to show whether or not L1CAM mRNA was expressed.
ISH in the Digestive Organs in w11 and 12
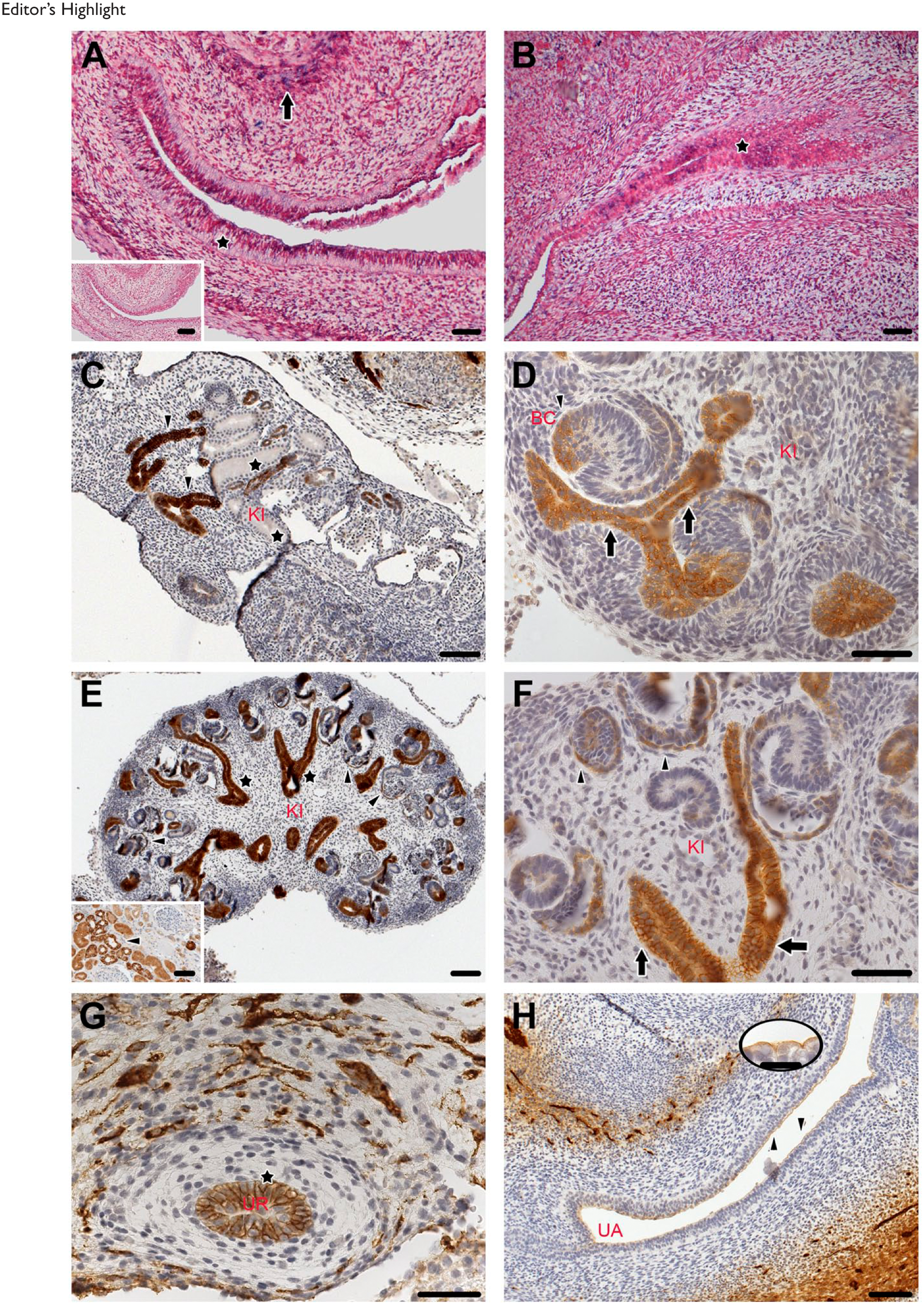
In accordance to our IHC data (Fig. 1C), L1CAM expression was visible in the myenteric plexus of the stomach (Fig. 2A arrow). Furthermore, a weak L1CAM expression was present in few epithelial cells of the stomach (Fig. 2A asterisk), and the hindgut (Fig. 2B asterisk). However, we want to emphasize that the majority of the cells were not stained. Control sense oligonucleotides revealed no reactivity under the same hybridization conditions (Fig. 2A inset).
Editor’s Highlight
IHC in the Urinary System in the Period From w8–12
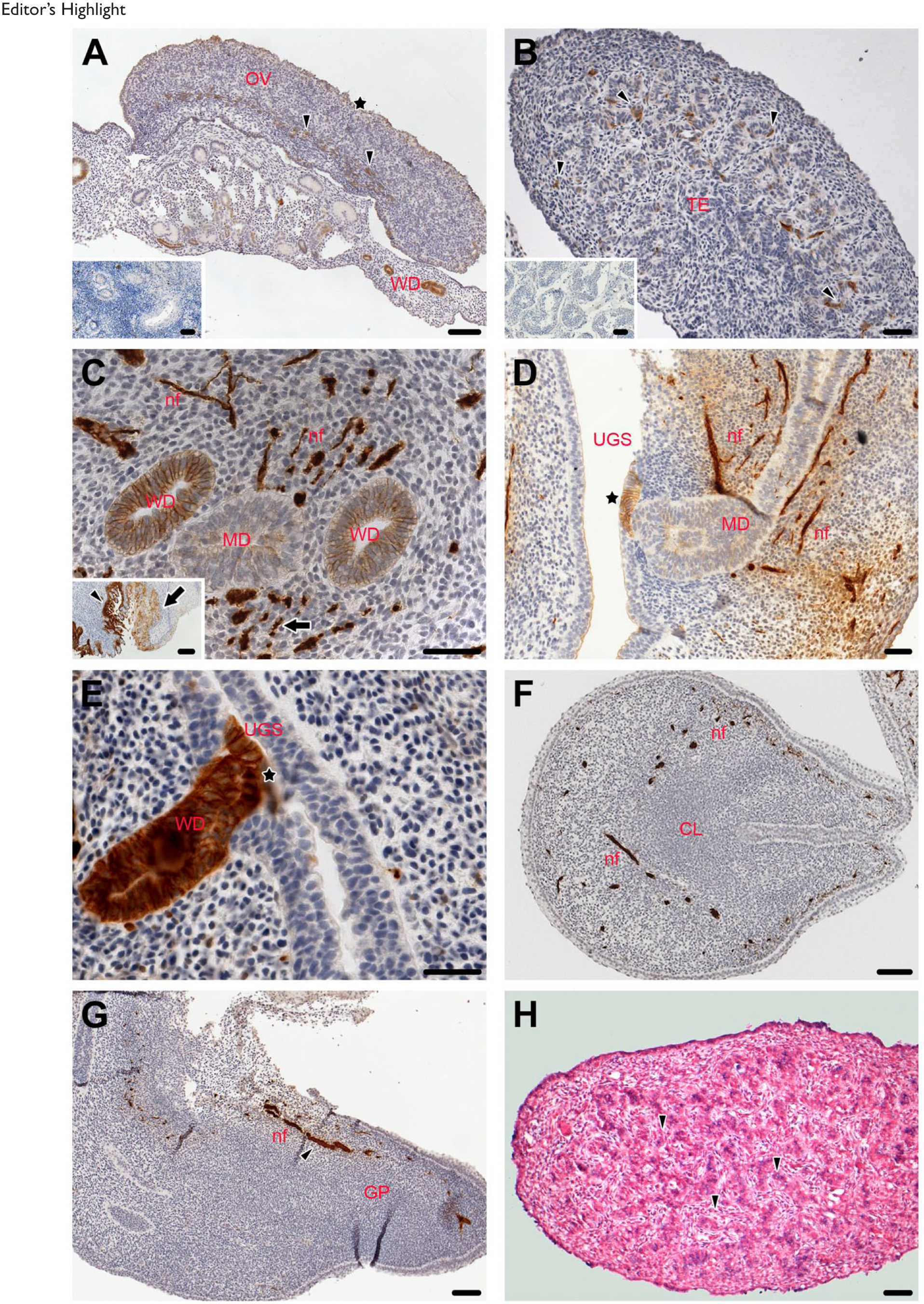
From w8 to 12, the kidney developed from meso- to metanephros which was accompanied by maturation of the nephrons. At w8, the regressive excretory canaliculi and glomeruli were visible and showed no immunoreactivity for L1CAM (asterisks). Simultaneously to the regression, the first tubules of the metanephros were observed, revealing a strong L1CAM staining (Fig. 2C arrowheads). At w9, the Bowman capsule (arrowhead) and the adjacent tubules (arrows), which derive from the metanephric blastema, exhibited intensive L1CAM labeling (Fig. 2D). At w10, the metanephros had a calix-like structure and the persistent nephrogenic tubules were now present. The collecting ducts, which are derivatives of the Wolffian ducts (WDs), displayed a strong immunoreaction for L1CAM (asterisks). The sheet of the Bowman capsule also exhibited L1CAM localization (Fig. 2E and F arrowheads). Similarly in adults, the collecting ducts revealed distinct L1CAM staining (Fig. 2E inset and arrowheads). The transitional epithelium of the ureter displayed strong L1CAM staining (Fig. 2G asterisk). Furthermore, the superficial epithelial cells of the developing urethra, which arises from the urogenital sinus, showed a distinct luminal staining for L1CAM (Fig. 3D arrowheads and oval inset).
Editor’s Highlight
IHC in Reproductive Organs in the Period From w8–12
From w8 to 12, the WD showed evidence for L1CAM immunostaining. In addition, the serosa surrounding the ovary (asterisk) as well as the medulla and, respectively, the ovary adjacent to the mesonephros were L1CAM positive (Fig. 3A arrowheads). In the male gonads, clusters of L1CAM positive cells (presumable Leydig cells) were visible (Fig. 3B arrowheads). In contrast, in adult no L1CAM stained cells were noted (Fig. 3B inset). In both sexes, the WD revealed strong immunoreaction for L1CAM, whereas the Muellerian duct (MD) displayed only a faint L1CAM staining (Fig. 3C). In the female newborn specimen, distinct L1CAM labeling was noted in the entire columnar epithelium of the endocervix (Fig. 3C inset, arrowhead), whereas in the squamous epithelium of the ectocervix only a moderate localization of the protein was visible (Fig. 3C inset, arrow). In the female fetuses, at the contact site between the urogenital sinus (UGS) and the MD, an accumulation of L1CAM positive cells occurred (Fig. 3D asterisk). In the male fetuses, at the juncture of the WD and the UGS, the epithelium of both revealed intense L1CAM staining (Fig. 3F asterisk). In the fetal clitoris, the epithelium revealed no staining for L1CAM (Fig. 3F). The dorsal penile nerve (arrowhead) displayed intense immunoreactivity in its course to the glans penis (Fig. 3G).
ISH in Reproductive Organs in the Period From w8–12
In the fetal testis, in concert with our IHC data (see above) clusters of cells (presumable Leydig cells) expressed L1CAM (Fig. 3H arrowheads).
Discussion
Apart from other factors, L1CAM seems to play a key role during embryonic development where it controls growth of cells and participates in the morphogenesis and integrity of the tissues.23,24 In the early 2000 years, upregulation of L1CAM was also found in human diseases such as solid epithelial tumors. 25 Based on these intriguing findings, many extensive studies followed, revealing that this molecule is involved in tumor formation and metastasis particularly in gastrointestinal and urogenital carcinomas of adults26-31 (Table 1). Apart from the fact that L1CAM is a significant prognostic marker for tumors, it has been considered a possible target for an antibody-based therapy,32-34 and current studies aim at the implementation of L1CAM in daily clinical use. Before that, however, can be achieved, the basic mechanism of L1CAM has to be thoroughly elucidated, including the exact role of L1CAM in the human organogenesis.
L1CAM Distribution in Embryonic/Foetal Tissue and Malignant tumors.

Abbreviations: IHC, immunohistochemistry; ISH, in situ hybridization; PDAC, pancreatic ductal adenocarcinoma; RCC, renal clear cell carcinoma.
Accordingly, in our present study we focused on the early, human fetal development (w8–12). Our data provide evidence that L1CAM is present in the epithelia of urogenital system (uriniferous structures = tubules and collecting ducts of the kidney, the renal pelvis, the ureter, and the urethra), but is absent or below the detection range in the gastrointestinal system. In adults, without any diseases, L1CAM was exclusively observed in the epithelia of the kidney. For IHC, we used the monoclonal antibody L1-14.10 that works perfectly on formalin-fixed human tissue and has improved immunohistochemical results. 20 It is well established that during embryonic development, mesenchymal cells undergo transition from motile, spindle-shaped cells to rows of polarized, stationary cells that are in close contact with each other, and that way form an epithelium.48,49 For integrity of the cell–cell contacts, L1CAM, among others, is required. 50 This biological process is termed mesenchymal–epithelial transition (MET), and it is reversible in cancers where stationary cells become motile. Nevertheless, MET also plays a part in metastasis, where it is responsible for colonization and establishment of metastases that regain epithelial functions to integrate the cancerous tissue into the invaded organ. 51
In our study, the organs of the gastrointestinal and uriniferous system are already formed, but clearly further differentiation and maturation occur in the course of development. In the gastrointestinal organs, the epithelia were L1CAM negative at w8–12, and we therefore conclude that they are already well developed and cell adhesion as well as apicobasal polarity is already established. Interestingly, L1CAM mRNA was found in the digestive organs, but this was only seen in few cells, displaying a weak staining. Consequently, we suggest that the protein synthesis had ceased in early fetal development.
Contrary to the gastrointestinal organs, L1CAM could be observed in the uriniferous structures. In these structures, the development progresses until the late fetal period (last third of pregnancy) and therefore we believe that L1CAM is crucial for these maturation processes. This is strengthened by Debiec et al., 52 who showed that in Lagomorpha L1CAM is involved in the continuous morphogenesis of the branching tubules of the kidney and the establishment of their specialized epithelium.
In the fetal urethra, the epithelium showed distinct immunoreactivity for L1CAM at its luminal side. This area (apical side of the superficial cells) is characterized by rigid membranous plaques which are essential to maintain impermeability and integrity of the urothelium 53 ; L1CAM might contribute to the maintenance of these plaques; however, this hypothesis is poorly supported by our data.
Apart from the MET pathway, L1CAM has shown to govern an epithelial-to-mesenchymal transition (EMT) during both embryogenesis and carcinogenesis. 10 In embryogenesis, EMT facilitates differentiation and establishment of organs. 49 A prerequisite for this process (EMT) is delamination of cells from their epithelium and breach of the lamina basalis, resulting in loss of the epithelial characteristics. Consequently, cells become motile and migrate over long distances to form tissues and organs on predetermined sites. 54 EMT, for instance, has been described in the kidney, liver, and pancreas of mammals during early embryonic development.55-58 Similar pathways are present in carcinomas of various organs where L1CAM converts epithelial cells into a migrating phenotype via the stimulation of the beta-catenin pathway and inhibition of adherens junction formation. 59 Moreover, abolition of the cell–cell contacts is triggered by a cleavage of the L1CAM ectodomain via the metalloproteinases ADAM 10 and 17.60,61 Taken together, EMT enables metastasis for cancer progression. Both processes, EMT and MET, are reflected in the mesonephric duct and its derivates, which all show strong epithelial L1CAM staining. The mesonephric duct is the scaffold for morphologically and functionally different cell types and is initially formed via EMT followed by MET. 62 Kujat et al. (1995) 63 demonstrated that in the fully differentiated urogenital tract of male mice and bulls, L1CAM is present and obviously enhances resistance and impermeability of the epithelium. Similarly, we could show that the epithelia of both the ureter and the urethra were L1CAM positive.
In conclusion, the present study indicates that in advanced differentiated epithelia such as those of the gastrointestinal system, L1CAM localization vanishes. In epithelia, however, which undergo further development such as those of the urogenital system, L1CAM is further needed for their fully establishment. Besides its important effects, L1CAM obviously plays a role in disintegration of epithelia and thus initiates tumor formation.
Footnotes
Acknowledgements
We thank A. Knab from the Division of Clinical and Functional Anatomy, Medical University Innsbruck, Austria, for technical assistance and C. Siemon for careful reading and correction of the manuscript. We acknowledged the excellent support of Dr. H. Wolf in the conservation of the human embryological and fetal samples. We also thank individuals who donated their bodies and tissues for the advancement of education and research. We dedicate this work to our friend and co-worker Mario Bitsche who passed away during the course of this work.
Author Contributions
EJP developed the concept, collected the data, analyzed the data, and wrote the manuscript. NC developed the concept, participated in data interpretation, and revising the manuscript. MJB analyzed the data and revised the manuscript. MB developed the concept, collected the data, and analyzed the data. MZ collected the data, analyzed the data, and revised the manuscript. JD participated in establishment of the methods and participated in data interpretation. KK participated in data interpretation and revised the manuscript. PA developed concept, participated in data interpretation, and revised the manuscript. A-GZ developed the concept, participated in data interpretation, and revised the manuscript. HF developed the concept, participated in data interpretation, and revised the manuscript. All authors read and approved the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Department of Anatomy, Histology and Embryology, Division of Clinical and Functional Anatomy, Medical University Innsbruck. The primary antibody used in this study was a donation of Peter Altevogt, Skin Cancer Unit 300G, German Cancer Research Centre, Heidelberg, Germany & Department of Dermatology, Venereology and Allergology University Medical Center Mannheim, Ruprecht-Karl University of Heidelberg, Germany.
