Abstract
Introduction
Renal interstitial fibrosis (RIF) is a common pathological change in the progression of chronic kidney disease (CKD) to end-stage kidney disease. During RIF, myofibroblasts (MFb) are activated and can produce a large amount of abnormal extracellular matrix (ECM) under the action of fibroblast-promoting factors.1–4 Abnormal content of ECM can impair the normal structure of kidney tissue, promote the formation of fiber scar, and then lead to irreversible damage to the kidney structure and function.5–7 The epithelial-mesenchymal transition (EMT) of renal tubular epithelial cells (RTECs) is an important source of myofibroblasts during RIF and a major pathological mechanism leading to the massive accumulation of ECM.8,9 However, the mechanism of EMT in RTECs has not been fully elucidated.
Interleukin-33 (IL-33) is a member of the interleukin-1 cytokine superfamily and is involved in the fibrosis of the skin, liver, kidney, lung, and other tissues through its membrane receptor suppression of tumorigenicity 2 (ST2). 10 IL-33 can promote the occurrence of skin fibrosis by increasing the expression of ECM-related proteins. 11 One clinical study has shown that the IL-33 and ST2 levels in patients with idiopathic pulmonary fibrosis were significantly increased and were directly proportional to disease severity. 12 The IL-33/ST2 axis can induce the proliferation and activation of alveolar epithelial cells, resulting in excessive deposition of ECM and promoting the occurrence of pulmonary fibrosis (PF).13,14 The IL-33/ST2 axis is also reported to promote the occurrence of hepatic fibrosis via the nuclear factor kappa-B (NF-κB) signalling pathway. 15 The expressions of IL-33 and ST2 are significantly increased in the kidney tissues of RIF induced by unilateral ureteral obstruction (UUO), which could be alleviated by IL-33 knockout. 16 However, the underlying mechanism remains unknown, which is worthy of further investigation.
The transcription factor Twist1 belongs to the basic helix-loop-helix superfamily, which can bind to the E-box region of various gene promoters and is involved in various biological processes such as embryonic development, EMT, and fibrosis. 17 Twist1 expression can be transcriptionally upregulated by NF-κB p65 overexpression in mammary epithelial cells. 18 In contrast, the inhibition of Twist1 expression can eliminate NF-κB p65-induced cell migration, invasion, and EMT characteristics. 19 Moreover, NF-κB p65 nuclear translocation increases Twist1 expression, while the knockdown or inhibition of NF-κB p65 decreases Twist1 expression. 20 These findings support that Twist1 is a downstream target gene of NF-κB, mediating NF-κB activation-induced cellular EMT. Our previous study also found that Twist1 was highly expressed in bleomycin-mediated PF, and transforming growth factor-beta 1 (TGF-β1) mediated EMT in type II alveolar epithelial cells was related to the activation of the Notch-1/Twist1 signaling pathway. 21 A recent study has found that targeted suppression of NF-κB-mediated Twist1 expression can inhibit the proliferation and EMT of kidney fibroblasts, thus alleviating RIF. 22 Therefore, in this study, we investigated whether IL-33 mediates the EMT of RTECs by activating the NF-κB/Twist1 signalling pathway. An in vivo EMT model induced by UUO and an in vitro EMT model induced by IL-33 were established. Our findings may provide new evidence for the pathogenesis of RIF.
Materials and methods
Animal experiments
Male C57BL/6 mice (8 weeks, 22 ± 2 g) were obtained from the SiPeiFu (SPF) Biotechnology Co., Ltd (certificate No: SCXK (jing) 2019-0010; Beijing, China). The animal experiments were divided into four groups: Sham-operated group (Sham), UUO group (UUO); UUO + IL-33 group (mice treated with recombinant IL-33(0.3 μg/10 g; #3626-ML-010, R&D Systems, Emeryville, CA, USA), 23 UUO + IL-33 Ab group (mice treated with anti-IL-33 antibody (1 μg/10 g; #AF3626, R&D Systems), 24 n = 12 in each group (Four mice are used for pathological testing, and the remaining eight are used for biochemical or molecular biological indicators testing). UUO-induced RIF model was prepared according to the reference method. 7 Recombinant IL-33 or anti-IL-33 antibody was intraperitoneally injected every other day from day 1 to day 21 after UUO. As Sham or UUO group, the same amount of IgG of rabbit (#AB-105-C, R&D Systems) was given intraperitoneally every other day.
Renal function analysis and measurement of serum IL-33 concentration
On the 22nd day, the eyeballs of mice were removed for blood collection and centrifugation for serum collection after anesthesia with 10% chloral hydrate (i.p., 300 mg/kg; 23100, Sigma, St. Louis, MO, USA). The concentration of blood urea nitrogen (BUN) (#BU6121) and serum creatinine (Scr) (#CR6113) and IL-33 (#M3300) were determined according to the manufacturer’s instructions. The BUN and Scr assay kits were purchased from Beijing Leadman Biochemical Co., Ltd (Beijing, China) and the IL-33 ELISA kit was purchased from R&D Systems (Emeryville, CA, USA).
Histological and immunohistochemical analyses
According to previous research methods, 19 the renal tissue was fixed with 4% paraformaldehyde and prepared into 4 μm paraffin sections. Hematoxylin-eosin (HE) staining and Masson’s trichromatic staining (KeyGEN Biotech, Nanjing, Jiangsu, China) were performed to observe the kidney histopathological changes and collagen deposition under light microscope. Protein expressions of ST2 (1:500, ab25877, Abcam, Hong Kong, China) in renal tissue were detected by streptavidin biotin complex-alkaline phosphatase (SABC-AP) immunohistochemistry, the positive expression of ST2 were yellow to brownish yellow particles. Under a digital microscope (DS-Ri1, Nikon, Shanghai, China) (magnification, ×400), 10 pictures were randomly taken with non-medullary field of view and no overlap, and then analyzed the positive expression of ST2 with Image-Pro plus 6.0 (Media Cybernetics, Rockville, Maryland, USA).
Cell culture and treatment
The human RTECs (HK-2) were obtained from BOSTER (#CX0044, Wuhan, Hubei, China). To verify the effect of NF-κB inhibitor pyrrolidine dithiocarbamate (PDTC) (P8765, Sigma) on IL-33 induced EMT in RTECs. The cells were divided into control (Con), IL-33 (100 ng/mL), dimethyl sulfoxide (DMSO) and PDTC (100 μM) group. The cells were treated with DMSO or PDTC for 2 h and then IL-33 for 48 h. To research the effect of ST2 siRNA on IL-33-induced EMT in HK-2 cells. The cells were divided into Con, Illinois-33, siRNA negative control (NC) and siRNA ST2 (si ST2) group. The cells were treated with si NC or si ST2 for 2 h and then IL-33 for 48 h. Each group has three compound holes, and each independent experiment is repeated three times.
small interference RNA (siRNA) Transfection
The ST2 siRNA and negative control siRNA were designed according to the mouse ST2 cDNA sequence by GenePharma (Suzhou, Jiangsu, China). The si ST2 sequences: sense, 5′- CCGUGAAAGUUGCUAUUGATT-3′ and antisense, 5′- UCAAUAGCAACUUUCACGGTT -3′. The cell transfection was performed according to siRNA and Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) instructions.
Immunofluorescence staining
After cells were treated according to the above cell experiment protocol and fixed in 4% paraformaldehyde. Immunostaining solution (Triton X-100) was incubated for 10 min. QuickBlock™ blocking buffer (P0260, Beyotime, Shanghai, China) was added and closed for 10 min. The blocking buffer was discarded, and anti-E-Cadherin monoclonal antibody (#14472, CST, Boston, MA, USA) and anti-α-smooth muscle actin (α-SMA) antibody (#19245, CST) were added and incubated at 4°C for 24 h. Fluorescently labeled secondary antibodies (A0460, A0423, Beyotime) were added and incubated for 2 h away from light. The anti-fluorescence quenched sealing solution (including DAPI) was added and placed for 5 min, then Laser scanning confocal microscope system (Leica, Heidelberg, Baden-Württemberg, Germany) was performed and photographs were taken.
RT-qPCR
Total RNA was extracted from mouse kidneys and RTECs in enzyme-free environment using the TRI Reagent® (T9424, Sigma). The PCR reaction conditions refer to our previous research results. 20 The following sense and antisense primers were used: collagen I (mouse), Forward: 5′- TGAACGTGACCAAAAACCAA-3′ and Reverse: 5′-GCAGAAAAGGCAGCATTAGG-3′; collagen I (human), Forward: 5′-CCAAATCTGTCTCCCCAGAA-3′ and Reverse: 5′- TCAAAAACGAAGGGGGAGATG-3′;; collagen III (mouse), forward 5′-GCCCACAGCCTTCTACACCT-3′, reverse 5′-GCCAGGGTC ACCATTTCTC-3′; collagen III (human), forward 5′-GGGAACAACTTGATGGTGCT-3′, reverse 5′-CCTCCTTCAACAGCTTCCTG-3′; E-cadherin (mouse), Forward: 5′-CAAGGACAGCCTTCTTTTCG-3′ and Reverse: 5′-TGGACTTCAGCGTCACTTT-3′; E-cadherin (human), Forward: 5′- TGCCCAGAAAATGAAAAAGG-3′ and Reverse: 5′- GTGTATGTGGCAATGCGTTC-3′; α-SMA (mouse), Forward: 5′- ACTGGGACGACATGGAAAAG-3′ and Reverse: 5′- TACATGGCTGGGACATTGAA-3′; α-SMA (human), Forward: 5′- ACTGGGACGACATGGAAAAG-3′ and Reverse: 5′- TACATGGCTGGGACATTGAA-3′; Vimentin (mouse), Forward: 5′-ATGCTTCTCTGGCACGTCTT-3′ and Reverse: 5′-AGCCACGCTTTCATACTGCT-3′; Vimentin (human), Forward: 5′- GAGAACTTTGCCGTTGAAGC-3′ and Reverse: 5′- TCCAGCAGCTTCCTGTAGGT-3′; GAPDH (mouse), Forward:5′-ACCCAGAAGACTGTGGATGG-3′ and Reverse: 5′-CACATTGGGGGTAGGAACAC-3; GAPDH (human), Forward:5′- CGACCACTTTGTCAAGCTCA-3′ and Reverse: 5′- AGGGTCTACATGGCAACTG-3.
Western blot analysis
Total proteins were extracted from Renal tissues or RTECs with Cell lysis buffer for Western (P0013, Beyotime) and Nuclear Protein Extraction Kit (R0050, Solarbio, Beijing, China) at low temperature. Western Blot was performed according to previous research methods. 25 The antibody information is as follows: collagen I (ab6308), collagen III (ab7778) and Histone H3 (ab1791) were purchased from Abcam (Hong Kong, China); Twist1 (#69366) and Vimentin (#5741) were obtained from CST (MA, USA); I-kappa-B-alpha (IκB-α) (AI096), phospho-IKB-α (p-IκB-α) (AF1870), NF-κB p65 (AF0246), phospho-NF-κB p65 (p-NF-κB p65) (AF5875), glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (AF2819) and secondary antibodies (A0208, A0216) were obtained Beyotime Biotech. Inc. (Shanghai, China). The gray values of each protein strip were measured by Western blot analysis function of Image Studio Lite software (LI-COR Biotechnology, Nebraska, USA).
Statistical analysis
The results were presented as means ± SEM. One-Way ANOVA analysis of variance was used to compare the means between multiple groups, and the LSD method was used to compare the means between two groups with SPSS 19.0 software. p < 0.05 was considered statistically significant.
Results
IL-33 exacerbates renal dysfunction and renal interstitial fibrosis caused by UUO
After UUO for 21 days, the renal function of mice was significantly decreased (the levels of Scr and BUN were significantly increased), and IL-33 significantly aggravated the renal function damage induced by UUO, but anti-IL-33 antibody could significantly alleviate the damage of renal function (Figure 1(a)). Pathological examination showed that the renal interstitial structure was disordered and the area was significantly widened, the renal tubules were atrophic and disappeared in large sheets, a lot of inflammatory cells were infiltrated and blue-stained collagen fibers were deposited after UUO. The levels of type I and III of collagen in the ECM of UUO mice was significantly increased. Exogenous IL-33 further aggravated the above pathological damage, further increased the deposition of collagen and the levels of type I and III of collagen. However, the anti-IL-33 antibody effect in the opposite way to IL-33 (Figure 1(b)–(e)). Effect of Interleukin-33 (IL-33) or IL-33 antibody (Ab) on unilateral ureteral obstruction (UUO)-induced renal interstitial fibrosis in mice. (a) The levels of serum creatinine (Scr) and blood urea nitrogen (BUN) were measured by sarcosine oxidase method and urease glutamate dehydrogenase method. (b) Hematoxylin-eosin on paraffin section of kidney tissue in mice. (c) Masson staining on paraffin section of kidney tissue in mice. (d) and (e) The expression of collagen I and collagen III mRNA and protein were determined by qPCR and Western blot. Data are means ± SEM n = 8. **p < 0.01 vs. Sham; #p < 0.05, ##p < 0.01 vs. UUO.
IL-33 aggravates UUO-induced EMT in mice
In accordance with previous studies, the levels of IL-33 and ST2 were significantly increased in serum and (or) kidney tissue of UUO mice. However, exogenous IL-33 significantly up-regulated ST2 expression, while anti-IL-33 antibody significantly down-regulated ST2 expression after UUO (Figure 2). In the process of RIFs after UUO, some RTECs gradually lose the characteristics of epithelial cells and acquire mesenchymal cell phenotype, that is EMT.
26
Our results also showed that the level of E-cadherin was down-regulated and the levels of α-SMA and Vimentin were up-regulated in kidney tissues of UUO mice. But more importantly, we found that exogenous IL-33 aggravated UUO-induced EMT, while anti-IL-33 antibody attenuated UUO-induced EMT (Figure 3). The protein expression levels of Interleukin-33 (IL-33) and suppression of tumorigenicity 2 (ST2) in mice. (a) and (b) The expression of IL-33 and ST2 protein levels were determined by Western blot. (c) Serum IL-33 level was measured by enzyme linked immunosorbent assay. (d) Immunohistochemical staining of ST2. (e) Quantification of ST2 positive cells. Data are means ± SEM n = 8. **p < 0.01 vs. Sham; #p < 0.05, ##p < 0.01 vs. UUO. Effect of Interleukin-33 (IL-33) or IL-33 antibody (Ab) on unilateral ureteral obstruction (UUO)-induced epithelial-mesenchymal transition in mice. (a) The expression of E-cadherin, Vimentin and α-smooth muscle actin (α-SMA) mRNA were determined by qPCR. (b) The expression of E-cadherin, Vimentin and α-SMA protein were determined by Western blot. Data are means ± SEM n = 8. **p < 0.01 vs. Sham; #p < 0.05, ##p < 0.01 vs. UUO.

IL-33 increases UUO-induced NF-κB activation and Twist1 expression
Previous study has demonstrated that IL-33 protein levels and NF-κB p65 activity obviously high in renal tissues of diabetic in rats.
27
The present results showed that the p-IκBα and p-NF-κB p65 were significantly high, and NF-κB p65 nuclear translocation and Twist1 nuclear expression were obviously increased induced by UUO. Treatment with IL-33 further significantly up-regulated IκBα and NF-κB p65 phosphorylated level, NF-κB p65 nuclear translocation and Twist1 expression after UUO. But the IκBα and NF-κB p65 phosphorylated level, NF-κB p65 nuclear translocation and Twist1 nuclear expression were attenuated by anti-IL-33 antibody in UUO mice (Figure 4). Effect of Interleukin-33 (IL-33) or IL-33 antibody (Ab) on unilateral ureteral obstruction (UUO)-induced NF-κB pathway activation and Twist1 protein expression. (a) and (b) p-IκBα and p-NF-κB p65 levels in whole tissue lysates were determined by Western blot, GAPDH was the endogenous control for whole tissue lysates. (c) and (d) The expression of NF-κB p65 and Twist1 in nuclear fractions were determined by Western blot, Histone H3 was the endogenous control for nuclear fractions. Data are means ± SEM n = 8. **p < 0.01 vs. Sham; #p < 0.05, ##p < 0.01 vs. UUO.
Inhibition of NF-κB activation reverses IL-33 induced Twist1 expression and EMT in RTECs
Studies have reported that the activation of NF-κB signaling is closely related to EMT of RTECs.
28
The western blot and Immunofluorescence staining results showed that, when NF-κB inhibitor PDTC was used to reduce IκBα and NF-κB p65 phosphorylated level and NF-κB nuclear transfer, it could significantly inhibit the nuclear expression of Twist1 induced by IL-33, but there was no effect on the ST2 expression (Figure 5). In addition, we also found that the content of E-cadherin obviously decreased while the content of α-SMA and Vimentin significantly increased, which together with the immunofluorescence results indicated that IL-33 could induce EMT in RTECs. We further found that the PDTC significantly reversed IL-33-induced EMT in RTECs compared with the IL-33group (Figure 6). Pyrrolidine dithiocarbamate (PDTC) decreased Interleukin-33 (IL-33) induced NF-κB pathway activation and Twist1 expression in cultured renal tubular epithelial cells. (a), (b) and (f) p-IκBα and p-NF-κB p65 levels and ST2 expression in whole tissue lysates were determined by Western blot. (c) and (e) The expression of NF-κB p65 and Twist1 in nuclear fractions were determined by Western blot. (d) Immunofluorescence analysis of p-NF-κB p65 (red) expression following PDTC and IL-33 treatment and staining with DAPI (blue) for DNA-blue. The values are means ± SEM from three independent experiments in vitro. **p < 0.01 vs. Con; #p < 0.05, ##p < 0.01 vs. IL-33. Pyrrolidine dithiocarbamate (PDTC) inhibited Interleukin-33 (IL-33) induced epithelial-mesenchymal transition in cultured renal tubular epithelial cells. (a) E-cadherin and α-SMA immunofluorescence staining of renal tubular epithelial cells was performed, with imaging for E-cadherin (red) and α-smooth muscle actin (α-SMA) (green) and staining with DAPI (blue) for DNA-blue. (b) The expression of E-cadherin, Vimentin and α-SMA mRNA were determined by qPCR. (c) The expression of E-cadherin, Vimentin and α-SMA protein were determined by Western blot. The values are means ± SEM from three independent experiments in vitro. **p < 0.01 vs. Con; #p < 0.05, ##p < 0.01 vs. IL-33.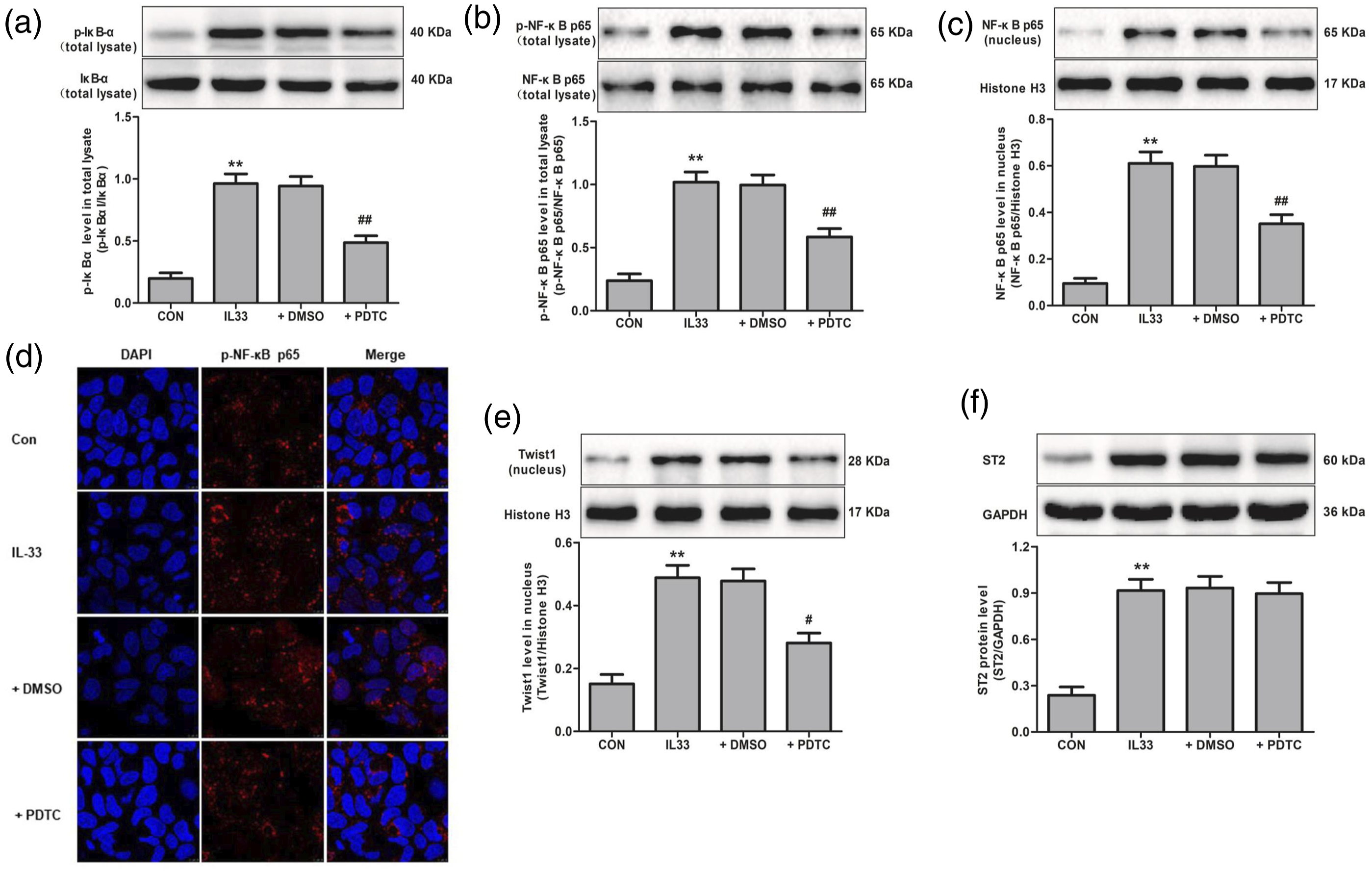

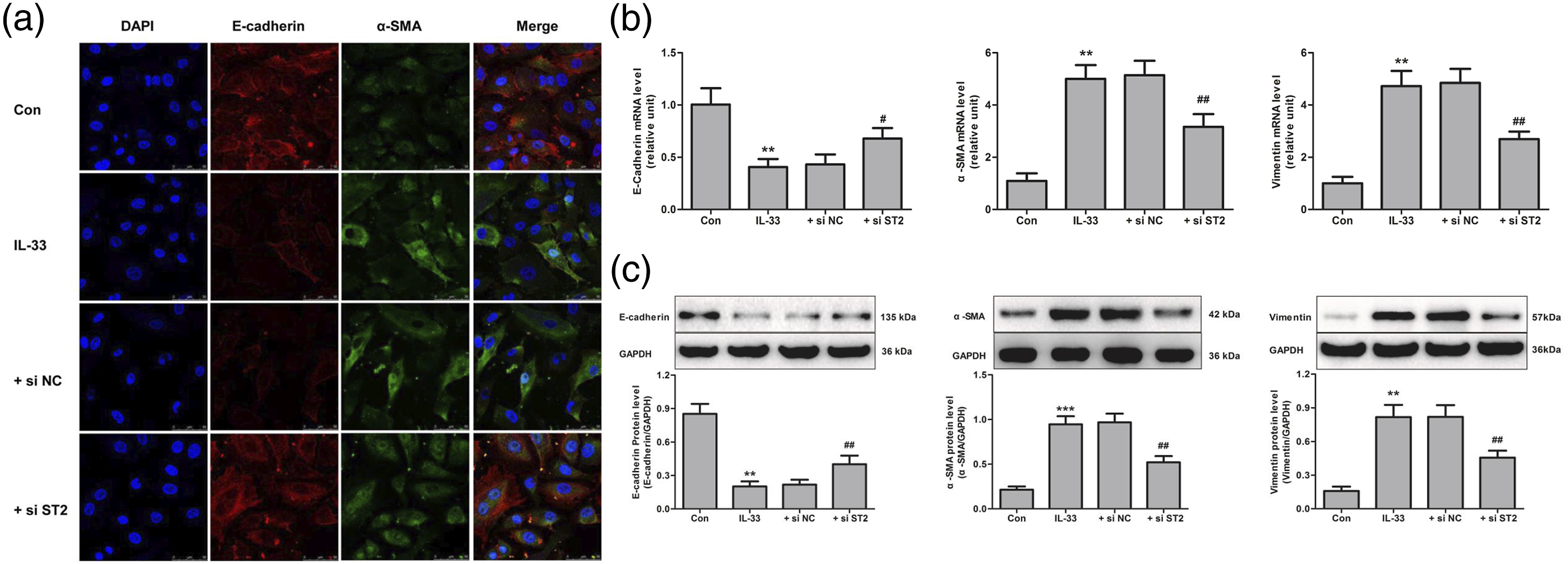
ST2 mediates NF-κB activation, Twist1 expression and EMT in RTECs induced by IL-33
Studies have found that after binding to its ST2 receptor, IL-33 activates the NF-κB signaling pathway causing immune responses.
29
As shown in Figure 7(a)–(e), ST2 siRNA inhibited IL-33 induced increased IκBα and NF-κB p65 phosphorylated level and NF-κB p65 nuclear translocation. Importantly, we also found that ST2 siRNA reversed IL-33 mediated increased of Twist1 nuclear expression (Figure 7(f)). Accordingly, ST2 siRNA also reversed IL-33 induced EMT in RTECs (Figure 8). These results indicate that ST2/IL-33 signaling facilitates NF-κB pathway activation, Twist1 expression and then may promote EMT of RTECs. Suppression of tumorigenicity 2 (ST2) knockdown reversed Interleukin-33 (IL-33) induced NF-κB pathway activation and Twist1 expression in cultured renal tubular epithelial cells. (a), (b) and (c) ST2 expression and p-IκBα, p-NF-κB p65 levels in whole tissue lysates were determined by Western blot. (d) and (f) The expression of NF-κB p65 and Twist1 in nuclear fractions were determined by Western blot. (e) Immunofluorescence analysis of p-NF-κB p65 (red) expression following PDTC and IL-33 treatment and staining with DAPI (blue) for DNA-blue. The values are means ± SEM from three independent experiments in vitro. **p < 0.01 vs. Con; #p < 0.05, ##p < 0.01 vs. IL-33. si, small interference; NC, negative control. Suppression of tumorigenicity 2 (ST2) knockdown reversed Interleukin-33 (IL-33) induced epithelial-mesenchymal transition in cultured renal tubular epithelial cells. (a) E-cadherin and α-smooth muscle actin (α-SMA) immunofluorescence staining of renal tubular epithelial cells was performed, with imaging for E-cadherin (red) and α-SMA (green) and staining with DAPI (blue) for DNA-blue. (b) The expression of E-cadherin, Vimentin and α-SMA mRNA were determined by qPCR. (c) The expression of E-cadherin, Vimentin and α-SMA protein were determined by Western blot. The values are means ± SEM from three independent experiments in vitro. **p < 0.01 vs. Con; #p < 0.05, ##p < 0.01 vs. IL-33.
Inhibition NF-κB activation or knockdown of ST2 suppresses IL-33 induced type I and III of collagen expression in RTECs
Previous study found that IL-33 can promote collagen expression in cultured A549 cells.
30
We found that IL-33 can significantly increase type I and III of collagen expression in cultured RTECs compared with control group for the first time. But when pre-treatment of NF-κB inhibitor PDTC or ST2 siRNA both reversed the effect of IL-33 on type I and III of collagen expression o (Figure 9). Pyrrolidine dithiocarbamate (PDTC) or suppression of tumorigenicity 2 (ST2) knockdown inhibited Interleukin-33 (IL-33) induced collagen I and collagen III expression in cultured renal tubular epithelial cells. (a) The expression of collagen I and collagen III mRNA were determined by qPCR. (b) The expression of collagen I and collagen III protein were determined by Western blot. The values are means ± SEM from three independent experiments in vitro. **p < 0.01 vs. Con; #p < 0.05, ##p < 0.01 vs. IL-33.
Discussion
The RIF is characterized by inflammatory cell infiltration, inflammatory response, the activation of myofibroblasts, and the accumulation and degradation of ECM, etc.6 Therefore, exploring the pathogenesis of RIF, developing effective methods for the treatment of RIF, and delaying the occurrence of end-stage kidney failure are the most concerned issues in the treatment of kidney diseases at present. The UUO model is established by ligating the unilateral ureter, which leads to urine retention and kidney volume increase, followed by the dilatation/atrophy of kidney tubules, inflammatory cell infiltration, fibroblast proliferation and activation, and collagen secretion, which eventually results in structural damages in kidney tissues. 31 We also found that kidney function was significantly impaired in UUO mice, andpathological examination showed atrophic kidney tubules, disordered kidney interstitial structure, and widened kidney interstitial area. The inflammatory cell infiltration, collagen fiber deposition, and expressions of type I and III collagens were significantly increased after UUO. It has been reported that the levels of IL-33 and ST2 were significantly increased in mice with UUO-induced RIF, and knockout of IL-33 significantly alleviated RIF induced by UUO. 16 Consistently, we found that the expressions of IL-33 and ST2 were significantly increased in the serum and (or) kidney tissues of UUO mice. In addition, we also found that exogenous IL-33 significantly aggravated UUO-induced RIF, and further increased the expressions of type I and III collagens. However, exogenous anti-IL-33 antibodies significantly reduced UUO-induced RIF and ECM deposition. In vitro, IL-33 significantly induced collagen expression in RTEC, while the knockdown of ST2 significantly inhibited IL-33-induced collagen expression. These results further suggest that the IL-33/ST2 axis may contribute to RIF in obstructive kidney disease. However, the mechanism of the IL-33/ST2 signalling pathway in promoting RIF has not been fully elucidated.
The pathogenesis of RIF is unclear. Studies have suggested that the EMT of RTECs plays an important role in the RIF of chronic kidney disease. 32 Studies also have found that kidney injury caused by toxins, drugs, ischemia, and other factors leads to the EMT of RTECs, in which the RTECs lose the epithelial phenotype and acquire the mesenchymal phenotype. This can further promote the secretion of a large number of inflammatory factors and pro-fibrotic factors, induce myofibroblasts to synthesize a large amount of ECM, and promote the development of RIF.33,34 It has been reported that RIF induced by UUO could promote EMT. 35 This is consistent with our results that E-cadherin was significantly decreased whereas α-SMA and Vimentin were significantly increased in the kidney tissues of UUO mice, indicating that there is EMT in RTECs after UUO. It was found that IL-33 expression was significantly increased in the kidney interstitial cells with positive expressions of α-SMA and Vimentin, suggesting that IL-33 may be involved in the EMT of RTECs. 36 In our study, we found that the levels of IL-33 and ST2 were significantly increased in the kidney tissues of UUO mice. More importantly, we further found that exogenous IL-33 could aggravate the UUO-induced EMT, while anti-IL-33 antibodies could significantly attenuate the UUO-induced EMT. In vitro, the knockdown of ST2 significantly inhibited IL-33-mediated EMT in RTECs. These results further confirm that the IL-33/ST2 axis is involved in the EMT process in RTECs.
NF-κB activity can promote the activation of fibroblasts and the occurrence of RIF, and inhibition of the NF-κB signalling pathway can effectively improve RIF.37,38 The basic helix-loop-helix transcription factor Twist1 regulates mouseembryonic development and participates in the EMT of fibrotic diease.17,18 It has been found that the Twist1 expression was significantly increased in patients with idiopathic pulmonary fibrosis. 39 Other studies have shown that activated NF-κB is involved in the EMT of RTECs by promoting Twist1 expression and the inhibition of Twist1 expression can inhibit the proliferation and EMT of kidney fibroblasts to alleviate RIF.19,20 The binding of IL-33 with ST2 can activate the downstream NF-κB, which then participates in the fibrosis of the heart, liver, kidney, lung, and other tissues.40,41 In this study, we found that the p-IκBα, p-NF-κB p65, nuclear translocation of NF-κB p65, and nuclear expression of Twist1 were significantly up-regulated in the kidney tissues of UUO mice. These changes were further enhanced by treatment with exogenous IL-33. However, treatment with anti-IL-33 antibodies significantly reduced the phosphorylation levels of IκBα and NF-κB p65, nuclear translocation of NF-κB p65, and nuclear expression of Twist1 in UUO mice. In vitro, IL-33 significantly increased the phosphorylation levels of IκBα and NF-κB p65 in RTECs and obviously increased the nuclear translocation of NF-κB p65 and the nuclear expression of Twist1. However, these effects of IL-33 were abolished by ST2 knockdown. In addition, the inhibition of p-IκBα, p-NF-κB p65, and NF-κB p65 nuclear translocation by PDTC significantly reduced the nuclear expression of Twist1, but there was no significant effect on the ST2 expression. These findings suggest that the IL-33/ST2 axis may promote RIF by activating the NF-κB signalling pathway and inducing Twist1 expression, leading to the EMT of RTECs.
However, in our study there are some limitations. Firstly, our animal study was poorly designed, missing the IL-33 alone group. Second, In vitro, we did not overexpress ST2 or NF-κB p65 to further validate the role of NF-κB/Twist1 signalling pathway in the EMT of RTECs. Third, how the NF-κB/Twist1 signalling pathway upregulates the expression of Twist1 deserves further exploration. Finally, this study needs to be future confirmed by clinical studies.
Conclusions
Taken together, IL-33 can bind to ST2 to promote the expression of Twist1 by activating the NF-κB signalling pathway, thereby inducing the EMT transformation of RTECs and leading to RIF. Therefore, the intervention of the IL-33/ST2 axis and its downstream signaling pathway may provide novel strategies for the treatment of RIF.
Footnotes
Acknowledgements
The authors would like to thank Anhui Provincial Department of Education, Anhui Provincial Health Commission and the National Natural Science Foundation of China for their support.
Author contributions
Ziyu Zhang and Wei Xu performed the experiments. Ziyu Zhang, Wengqiang Gu and Zepeng Li analyzed the data. Xianwei Li and Ziyu Zhang designed the study, and wrote and revised the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 81900531) and Key Projects of Anhui University Natural Science Foundation (No. KJ2021ZD0106) and Key Projects of Anhui Provincial Health Commission (No. 2021zy033).
Ethical approval
The study was obtained permission from the Animal Welfare and Ethics Committee of Wannan Medical College (NO. LLSC-2022-249).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
