Abstract
SWAP-70 has been demonstrated as a multiple functional signaling protein involved in formation of membrane ruffling induced by signal cascade of tyrosine kinase growth factor receptors. In the present study, the spatial and temporal expression pattern of SWAP-70 on human fetomaternal interface was investigated using specimens collected from tubal and normal pregnancies by in situ hybridization, immunohistochemistry, and Western blotting. Data showed an intense expression of SWAP-70 in trophoblasts at weeks 3–6 of fallopian implantation and at weeks 6–7 of normal pregnancy. The most intense expression was exhibited by those highly motile and invasive extravillous trophoblasts. From gestational week 8 on, the level of SWAP-70 in trophoblasts decreased significantly, and the signal was restricted in villous cytotrophoblast cells. In the in vitro cultured human trophoblast cell line, B6Tert-1, colocalization of SWAP-70 with F-actin was verified. Data in human placenta were similar to what we recently reported on rhesus monkey fetomaternal interface. Our results suggest that SWAP-70 may be involved in regulating migration and invasion of trophoblast cells during the processes of embryonic implantation and placentation in primates.
SWAP-70 is a 70-kDa protein originally isolated from activated B lymphocytes (Borggrefe et al. 1998). Its high expression was observed in activated B cells and immature mast cells, which indicated its involvement in activation, migration, and adhesion of these cells (Masat et al. 2000; Gross et al. 2002; Sivalenka and Jessberger 2004). SWAP-70 was also widely expressed in various cell types and tissues (Borggrefe et al. 1998; Hilpelä et al. 2003). Structurally, SWAP-70 contains three nuclear localization signal domains as well as a Dbl homology (DH) and a pleckstrin homology (PH) domain. DH and PH domains are often found in signaling molecules and are important for membrane targeting (Masat et al. 2000; Shinohara et al. 2002). Therefore, it was strongly proposed that SWAP-70 could act as a signaling molecule involved in both nuclear events and membrane and/or cytoplasm signalings. Recently, SWAP-70 has been identified as a new member of Rac-guanine nucleotide exchange factors (GEFs), which catalyzes phosphatidylinositol (3,4,5)-trisphosphate [PtdIns(3,4,5)P3]-dependent switching of inactive GDP-Rac to active GTP-Rac, triggering the signal cascade of tyrosine kinase receptor. SWAP-70 was further identified to be required for the formation of membrane ruffling. It could translocate from cytoplasm to cell membrane upon phosphoinositide 3-kinase (PI3K) activation by growth factor stimulation, cooperating with activated Rac and binding to filamentous actin (F-actin) directly to regulate actin organization. The process seems to commonly exist in lamellipodia protrusion and membrane ruffles during cell migration (Shinohara et al. 2002; Hilpelä et al. 2003; Ihara et al. 2006; Wakamatsu et al. 2006).
There is scant information regarding the function of SWAP-70 in reproductive processes. In our previous work, SWAP-70 gene was preferentially expressed in rhesus monkey implantation site at the very beginning of embryonic implantation (Sun et al. 2004; Li et al. 2005). We recently demonstrated that SWAP-70 was intensely expressed and colocalized with F-actin in highly motile extravillous trophoblasts on rhesus monkey fetomaternal interface during the early stage of pregnancy (Liu et al. 2006). The data seem to suggest the possible involvement of SWAP-70 in primate embryonic implantation and placentation.
Over the past decades, various aspects and molecules associated with embryonic implantation events have been explored in humans. However, owing to the restricted availability of human specimens with intact fetomaternal interface, much remains unclear concerning the expression patterns of the relevant molecules involved in the interaction between trophoblasts and the maternal endometrium, especially at the very early stage of embryonic implantation. Tubal pregnancy is a type of abnormal pregnancy in which the embryo implants in the fallopian tube rather than in the uterus. Our data and other data show that tubal pregnancy triggers normal immunoreactivity and hormonal activation of the maternal body (Earl et al. 1986; Randall et al. 1987; Marx et al. 1999), as well as giving rise to immunologically normal and hormonally active trophoblast cells (Vassiliadou and Bulmer 1998; Li et al. 2003; Qin et al. 2003; Bai et al. 2005). Furthermore, the fetomaternal compartment removed from patients with tubal pregnancy often possesses integrated fetomaternal interface, as shown in our previous studies (Li et al. 2003; Qin et al. 2003; Bai et al. 2005). Therefore, tubal pregnancy may provide a unique model to study the mechanism of human embryonic implantation, particularly from the aspect of trophoblast cells during the very early stages.
In the present study, the expression pattern and localization of SWAP-70 on the fetomaternal interface has been comparatively investigated between normal and tubal pregnancies at different gestational stages by immunohistochemistry (IHC), in situ hybridization (ISH), and Western blotting.
Materials and Methods
Human Normal Placental Tissue Preparation
Tissue collection and treatment were performed as previously described (Qiu et al. 2005). Briefly, tissues of human chorionic villi or placenta were obtained from the Beijing Haidian Hospital (Beijing, China) from patients who underwent therapeutic termination of pregnancy at weeks 6–9 and 26 or normal term delivery. Informed consent was obtained from patients, and permission was granted by the Local Ethical Committee. No special medical treatment was offered to patients before termination of pregnancy. Gestational weeks of the specimens were defined according to the morphological observation of villi as well as pathological examination, with the record of the menstrual cycle as reference. Tissue was fixed immediately in 4% paraformaldehyde (PFA) at 4C for 10 hr, gradually dehydrated in ethanol, and embedded in paraffin. Six-μm-thick sections were collected on SuperFrost Plus glass slides (Menzel-Gläser; Braunschweig, Germany). At least three cases at each gestational stage were collected.
Human Tubal Pregnant Tissue Preparation
Tissue collection and treatment were performed as previously described (Qin et al. 2003). In brief, 23 tissue specimens (gestational weeks 3–9 and week 23) were obtained during operative treatment for tubal pregnancy in Oulu University Hospital (Oulu, Finland). The project was approved by the Local Ethics Committee. Diagnosis of tubal pregnancy was based on ultrasonic and pathological examinations. All samples had histologically normal placental tissues and feto-maternal interfaces. At least three cases at each gestational stage were collected. Specimens were fixed, embedded in paraffin, and sectioned as above. IHC staining of cytokeratin was performed for the identification of trophoblast cells.
Cell Culture
Immortalized human cytotrophoblast cells, B6Tert-1, were cultured as reported (Wang et al. 2006). In brief, the cells were cultured in collagen I (Cellmatrix Type I-A; Institute of Biochemistry, Osaka, Japan)-coated flasks (Corning Inc.; Corning, NY) with FD (F12:DMEM; 1:1) medium supplemented with 10 ng/ml epidermal growth factor (EGF; Collaborative Research, Lexington, MA) and 10 μg/ml insulin (Sigma-Aldrich; St Louis, MO). Cells were incubated at 37C in an atmosphere of 5% CO2-95% air.
Preparation of Digoxigenin (DIG)-labeled Probes
DIG-labeled cRNA probes were used for ISH. Total RNA was isolated from human placenta tissues with TRIzol reagent (Gibco/BRL Life Technologies; Grand Island, NY). The cDNA fragment of human SWAP-70 was obtained by RT-PCR using the specific primers 5′-GAAGACATGTACCTAAAGCTGC-3′ (sense) and 5′-AGGACACGCTGATTCTCC-3′ (antisense). After sequence verification, the PCR fragment was constructed into pGEM-T easy vector (Promega; Madison, WI). The constructs were linearized, and DIG-labeled cRNA probes, both sense and antisense, were obtained by in vitro transcription according to the instructions of the DIG RNA Labeling System (Roche Diagnostics GmbH; Mannheim, Germany). Probes were kept at −80C at a concentration of 0.1 μg/μl.
ISH
ISH was performed as previously described (Qin et al. 2003). In brief, paraffin sections were routinely deparaffinized and rehydrated. After treatment with 0.2 N HCl, slides were denatured at 70C in 2X SSC (0.3 M sodium chloride, 0.03 M sodium citrate, pH 7.0) and digested with 4 μg/ml protein-ase K (Gibco/BRL Life Technologies). Postfixation was performed in 4% PFA followed by acetylation in triethanolamine buffer containing 0.5% acetic anhydride. Slides were pre-hybridized for 4 hr at 58C in prehybridization buffer (50% formamide, 20 mM Tris-HCl, 50 mM EDTA, tRNA Coli 0.5 mg/ml, DTT 100 mM) and further hybridized for 18 hr at 58C in fresh hybridization buffer containing 1 ng/μl antisense probe. After consecutive washes in 2X SSC and 0.1X SSC at 63C, slides were blocked with 0.5% blocking reagent (Boehringer Mannheim; Mannheim, Germany) and incubated with alkaline phosphatase-coupled anti-DIG antibodies (dilution 1:500). Color development was performed in buffer II (100 mM Tris-HCl, 100 mM NaCl, 50 mM MgCl2, pH 9.5) containing 5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium (BCIP NBT; Boehringer Mannheim). Nonspecific staining was removed by rinsing the slides with 95% EtOH. After dehydration with EtOH and xylene, slides were mounted in resin. Negative control experiments were performed by replacing the antisense probe with the sense probe in the hybridization buffer. Results were assessed based upon the evaluations of three independent observers.
IHC
IHC assay was performed as previously described (Qin et al. 2003). Briefly, paraffin sections were deparaffinized, rehydrated, and retrieved in 10 mM citrate buffer (pH 6.0). After immersion in 1% hydrogen peroxide, the sections were incubated with rabbit polyclonal antibody against SWAP-70 (5 μg/ml; kind gift of Dr. T. Borggrefe) at 4C overnight. Negative controls were performed by replacing the primary antibody with normal rabbit IgG at the same concentration. The sections were further incubated with goat anti-rabbit IgG (1:300; Vector Laboratories, Burlingame, CA) for 30 min and with horseradish peroxidase streptavidin (1:500; Vector Laboratories) for 15 min at 37C. Final visualization was performed by incubating the sections with DAB chromogen (DakoCytomation; Carpenteria, CA) solution with 0.03% H2O2 for 15 min. Counterstaining with hematoxylin was performed prior to slide mounting. Results were assessed based upon the evaluations of three independent observers.
Double-immunofluorescence Assay
Double-immunofluorescence assay was performed as previously described (Liu et al. 2006). Briefly, B6Tert-1 cells were fixed with a 1:1 mixture of ethanol-acetone. After incubation with 1% BSA (Sigma), cells were incubated with primary antibody against SWAP-70 (5 μg/ml) and the tetramethylrhodamine isothiocyanate-conjugated phalloidin (TRITC; Sigma-Aldrich), which binds specifically to F-actin. Cells were further incubated with FITC-conjugated goat anti-rabbit IgG (Santa Cruz Biotechnology; Santa Cruz, CA). After mounting, the cells were observed with a Leica TCS NT confocal system (Leica; Wetzlar, Germany). Negative control was performed by incubating the cells with normal rabbit IgG and staining cell nuclei with 0.1 mg/ml propidium iodide (Sigma-Aldrich).
Western Blotting Analysis
Soluble cytoplasmic proteins were extracted by incubating the homogenized tissues with lysis buffer (20 mM Tris with Mg2+ and K+, 1 mM DTT, 0.2% NP-40, 100 mM PMSF, 5 μg/ml aprotinin, chymostatin, leupeptin, pristine, and trypsin inhibitor) (all from Sigma-Aldrich), and the supernatants were harvested after centrifuging. Protein concentration was measured according to the method of Bradford (1976). Twenty-μg protein extract was subjected to 10% SDS-PAGE and subsequent electrotransferring to a nitrocellulose membrane (Amersham Pharmacia Biotech; Buckinghamshire, UK). The membranes were blocked with 5% non-fat milk dissolved in PBS (pH 7.4) with 0.5% Tween-20 and incubated with specific antibodies diluted in 0.1% BSA. Final visualization was achieved with HRP-linked goat anti-rabbit IgG (1:2500; W4011, Promega) and the subsequent enhanced chemiluminescence reagent (ECL; Pierce Biotechnology, Rockford, IL) according to the manufacturer's instructions.
Statistical Analysis
All experiments were repeated three times, each with at least three independent specimens per developmental stage. Western blot data were standardized with the value of actin, and the relative density was presented as mean ± SD according to three independent experiments. Statistical difference was evaluated by one-way ANOVA; p<0.05 was accepted as significant.
Results
Expression Pattern of SWAP-70 in Human Normal Placenta at Different Stages of Pregnancy
With IHC and ISH, the spatial and temporal expression of SWAP-70 in human normal placenta was demonstrated. Spatially, both mRNA and protein of SWAP-70 were observed in villous cytotrophoblast cells (VCT), syncytiotrophoblast cells (ST), and column cytotrophoblasts (CCT) (Figures 1A-1L). Temporally, the signal intensity of SWAP-70 in trophoblasts was strongest at weeks 6–7 (Figures 1A and 1B and 1G and 1H) and decreased from weeks 8 on. At week 26 and at full term, the positive signal could only be observed in VCT cells at a low level (Figures 1C-1F and 1I-1L).
The overall variation of SWAP-70 protein in human placenta tissues was further identified by Western blotting, and the result paralleled that of IHC. The relative density of SWAP-70 was relatively high at weeks 6–7 and decreased from weeks 8 on, reaching its nadir at full term of gestation. In the term placenta, the level of SWAP-70 was ~20% of that observed at weeks 6 (Figure 2).
Expression Pattern of SWAP-70 on the Fetomaternal Interface of Tubal Pregnancy During Gestational Weeks 3–9 and 23
IHC assay and ISH were performed to show the distribution pattern of SWAP-70 on the fetomaternal interface of tubal pregnancy.
Spatial Distribution Pattern of SWAP-70 on the Fetomaternal Interface of Tubal Pregnancy at Weeks 3–6. ISH and IHC revealed that both mRNA and immunoreactivity of SWAP-70 were widely expressed in various types of trophoblasts including VCT, CCT, extravillous cytotrophoblast (EVCT) invading maternal stroma (Figures 3A-3D and 3J-3M), and perivascular trophoblasts (PEVT) penetrating maternal vessels (Figures 3H and 3Q). The signal intensity increased slightly from distal CCT to EVCT and was much stronger in EVCT than in VCT (Figures 3A-3D and 3J-3M). Weak staining could be observed in some ST. Remarkably, immunostaining for SWAP-70 in trophoblast cells was observed both in cytoplasm and on the cell membrane (Figure 3Q).
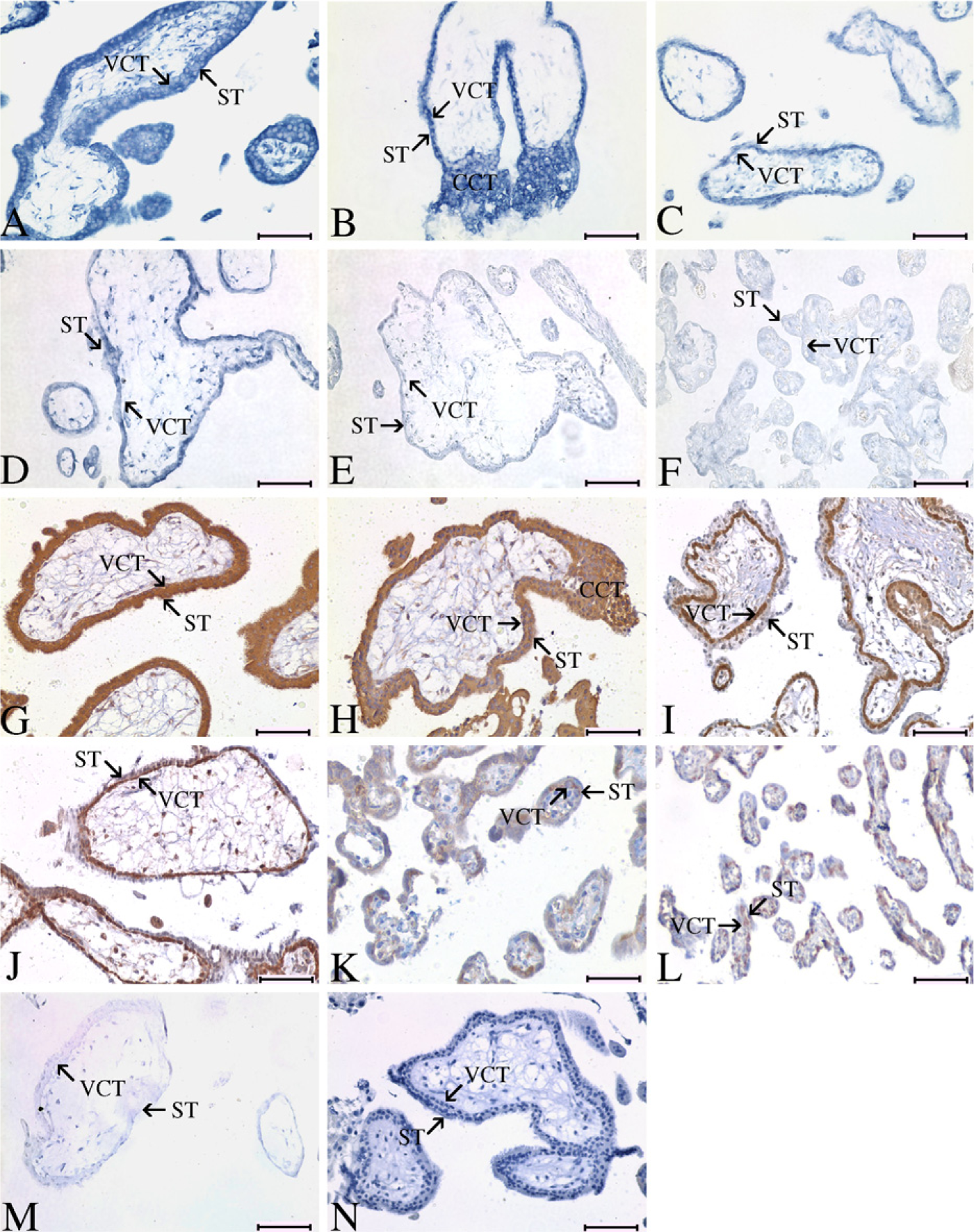
Expression pattern of SWAP-70 in human normal placenta at different stages of pregnancy. (
Temporal Alteration of SWAP-70 on the Fetomaternal Interface of Tubal Pregnancy at Different Stages. Expression of SWAP-70 in trophoblast cells showed a similar temporal change between tubal and normal intrauterine pregnancies. With ISH and IHC, it was shown that intense expression of SWAP-70 in trophoblasts was found as early as weeks 3–4 of gestation and maintained high until weeks 5–6 (Figures 3A-3D and 3J-3M). From week 8 on, the staining strength declined. At week 23 of gestation, positive signal could only be observed in VCT at a low level (Figures 3E-3G and 3N-3P).
Co-localization of SWAP-70 with F-actin in Human Trophoblasts
B6Tert-1 is an immortalized human normal cytotrophoblast cell line established in this laboratory (Wang et al. 2006). In B6Tert-1 cells, double-immunofluorescent assay revealed that SWAP-70 was exactly colocalized with F-actin in cytoplasm and membrane (Figure 4).
Discussion
The first step in human implantation involves the attachment of trophoblast to the endometrium at approximately 7 days after fertilization. After this initial interaction, trophoblast cells proliferate, invade the underlying stroma, and differentiate to form chorionic villi composed of an inner-cell layer of cytotrophoblasts and an outer layer of ST. During the third week of gestation, proliferating cytotrophoblasts at the tips of the anchoring villi breach the syncytium, forming large cellular columnsthatmigrate into the maternal deciduas. A subpopulation of these trophoblasts subsequently dissociates from the column and invades deeply into the underlying maternal tissues in which they penetrate the basal lamina of the uterine arterioles, thereby ensuring a continuous blood supply to the placenta and developing fetus (Loke and King 1996). These migrations and invasions occur in the early stage of pregnancy up to ~14 weeks of gestation, which is the most important period for the establishment of human placental circulation (Aplin 1991).
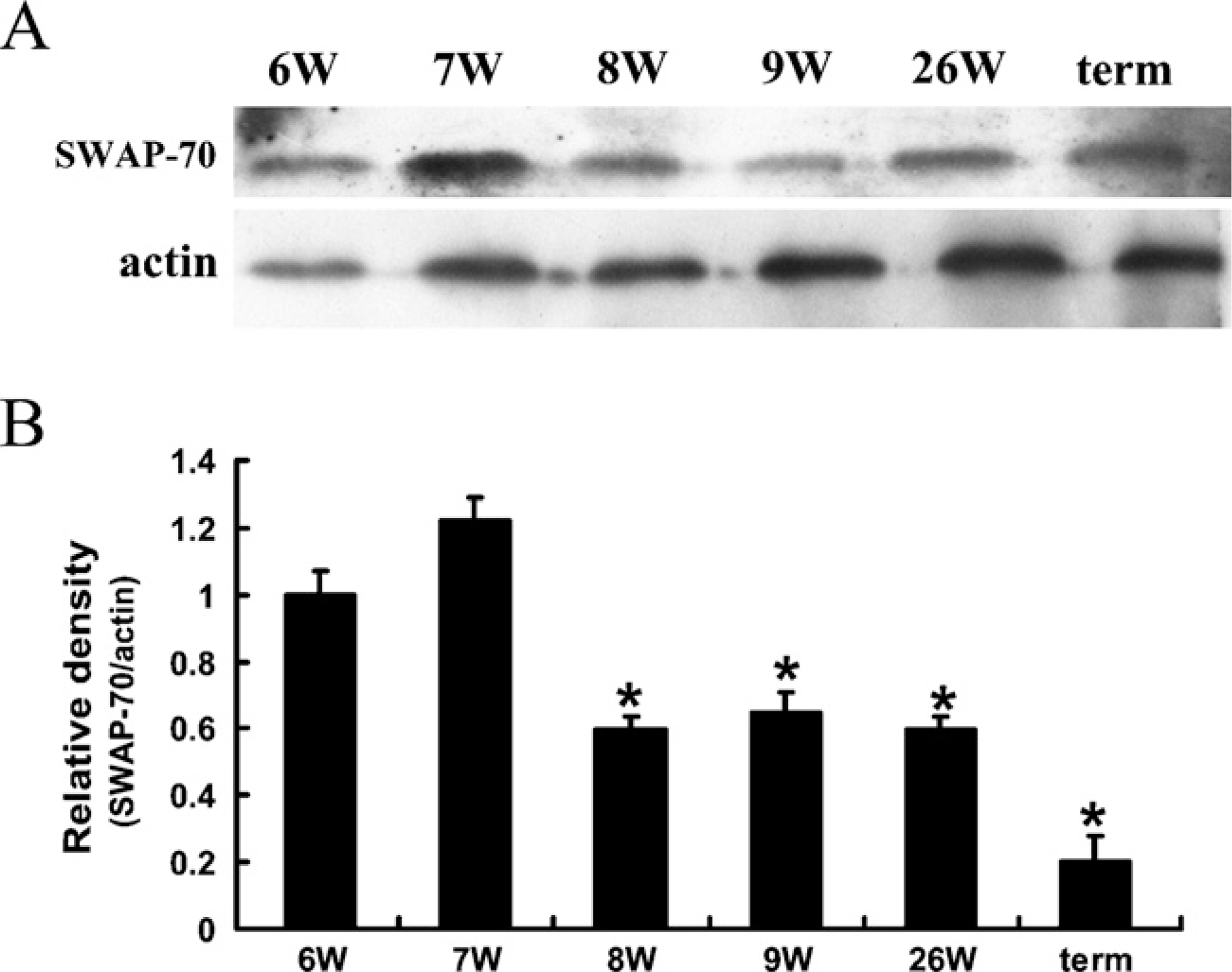
Western blotting analysis to show SWAP-70 expression in human normal placenta at different stages of gestation. (
Our present study demonstrated an intense expression of SWAP-70 in various subpopulations of trophoblasts including VCT, CCT, and EVCT at weeks 3–6 of fallopian implantation and weeks 6–7 of normal pregnancy. In addition, the notably stronger signal of SWAP-70 was mainly found in proliferative CCT and highly invasive and migratory EVCT. At the end of the first trimester, the uteroplacental circulation has been well established, and the invasiveness of trophoblast cells declines. From then on, a much lower level of SWAP-70 is restricted in VCT. The spatial and temporal change of SWAP-70 expression in human placenta suggested that SWAP-70 might be involved in some trophoblastic events during human pregnancy.
Recently, colocalization and direct binding of SWAP-70 with F-actin have been observed, and SWAP-70 was proposed to cooperate with activated Rac to induce membrane ruffling (Shinohara et al. 2002; Hilpelä et al. 2003; Ihara et al. 2006). Membrane ruffles are rapidly moving membrane protrusions usually considered as a sign of increased lamella dynamics and elevated migration levels (Ridley 1994; Small et al. 2002). Formation of membrane ruffles and lamellipodia in trophoblasts is frequently observed during embryonic implantation and placentation (Shukla et al. 1991; Fuller et al. 1994; Suenaga et al. 1996; Enders and Lopata 1999). One of our recent studies demonstrated the colocalization of SWAP-70 with F-actin on rhesus monkey fetomaternal interface at early pregnancy, especially in those highly invasive EVCT (Liu et al. 2006). It is likely that SWAP-70 may be involved in the migration of primate trophoblasts by regulating formation of membrane ruffles. In this study, intense expression of SWAP-70 in invasive EVCT cells was observed during the early stage of human pregnancy when these cells are highly motile. Colocalization of SWAP-70 with F-actin was verified in human cytotrophoblast cell line, B6Tert-1. Collectively, these observations strengthen our hypothesis that SWAP-70 may participate in the arrangement of actin filament to regulate migration and invasion of trophoblasts during embryonic implantation in the primates.
As mentioned, SWAP-70 has been demonstrated to activate Rac as a GEF and is involved in signaling downstream of PI3K-linked tyrosine kinase receptors (Shinohara et al. 2002). Among the signaling molecules activated by growth factors, PI3K and Rac have been considered to play crucial roles in regulating membrane ruffling (Ridley et al. 1992; Kotani et al. 1994; Wennstrom et al. 1994). Generally, upon stimulation of tyrosine kinase receptor by growth factor, PtdIns(3,4,5)P3 is produced under the catalysis of active PI3K. It was suggested that SWAP-70 acted as an effector or adaptor in response to PI3K activity in various cells and was involved in regulating actin cytoskeleton and cell motility by participating PtdIns(3,4,5)P3-dependent activation of Rac (Shinohara et al. 2002; Welch et al. 2003; Hilpelä et al. 2003; Ihara et al. 2006; Wakamatsu et al. 2006). It remains unclear which growth factors are the upstream stimulating molecules of SWAP-70-involved signaling pathway in human trophoblasts during implantation. Some growth factors expressed in the endometrium and trophoblast, such as epidermal growth factor (EGF) and insulin-like growth factor type I and type II (IGF-I and IGF-II), play important roles in regulation of trophoblast invasion and migration during embryonic implantation (Bass et al. 1994; McKinnon et al. 2001; Lacey et al. 2002), and these factors have been proven to affect the organization of actin cytoskeleton and induce membrane ruffling through their respective tyrosine kinase receptors (Kadowaki et al. 1986; Kotani et al. 1994; Kurokawa et al. 2004). Furthermore, there is evidence to demonstrate the involvement of EGF and IGF-I in regulation and association of some GEFs like VaV and LARG (leukemia-associated rho guanine nu-cleotide exchange factor) (Uddin et al. 1996; Becknell et al. 2003; Tamas et al. 2003). Thus, EGF, IGF-I, and/or IGF-II might be the candidates that regulate trophoblast migration through SWAP-70-activated Rac GTPase.
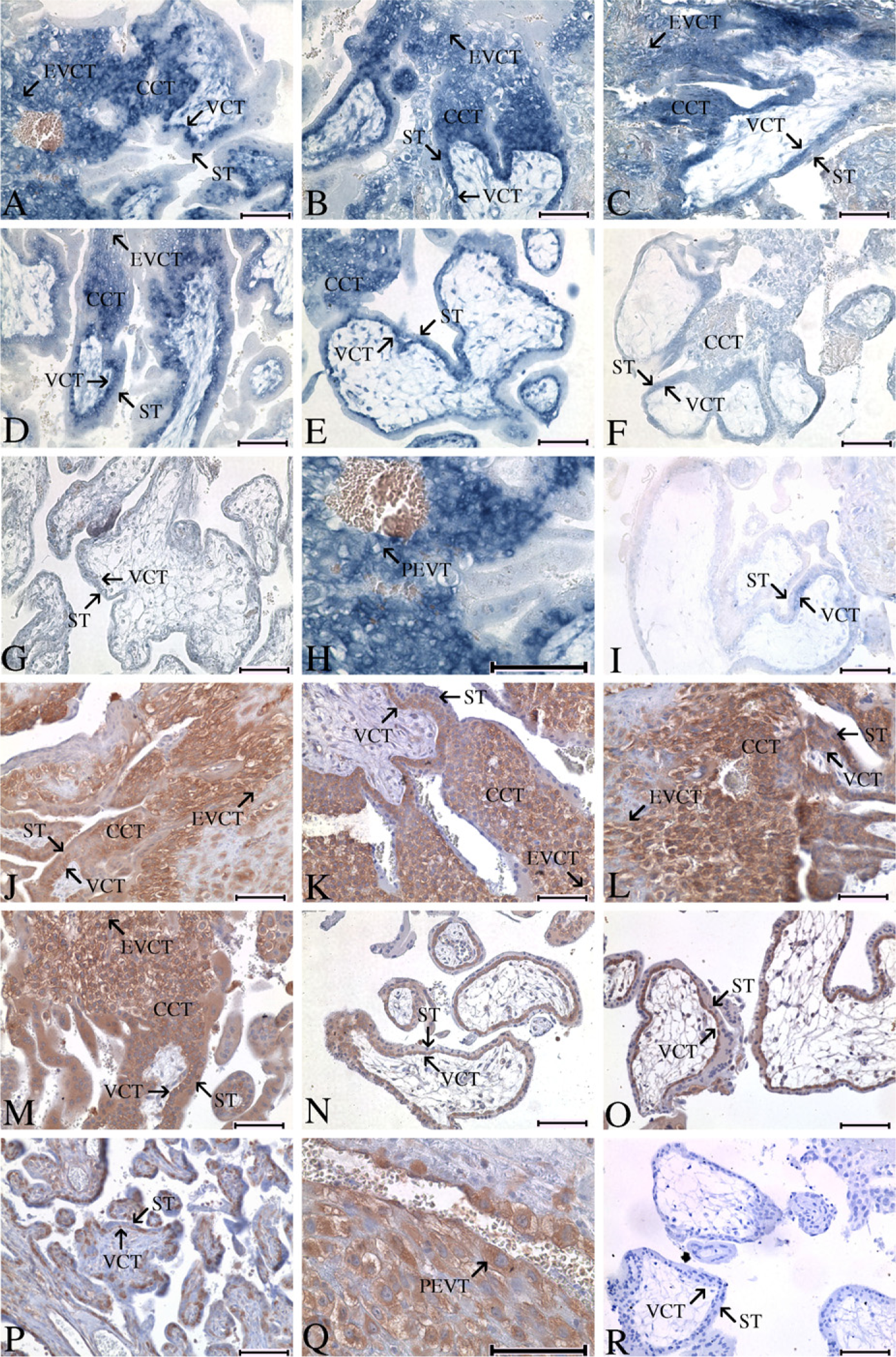
Expression pattern of SWAP-70 on the fetomaternal interface of tubal pregnancy during gestational weeks 3–9 and 23. (
In summary, this study is the first to clarify the temporal and spatial expression pattern of SWAP-70 on human fetomaternal interface and placental trophoblasts, especially during the very early stages of pregnancy, by using tubal pregnancy as a unique model. Our findings suggest that SWAP-70 may be involved in regulating motility of trophoblast cells during embryonic implantation and placentation.
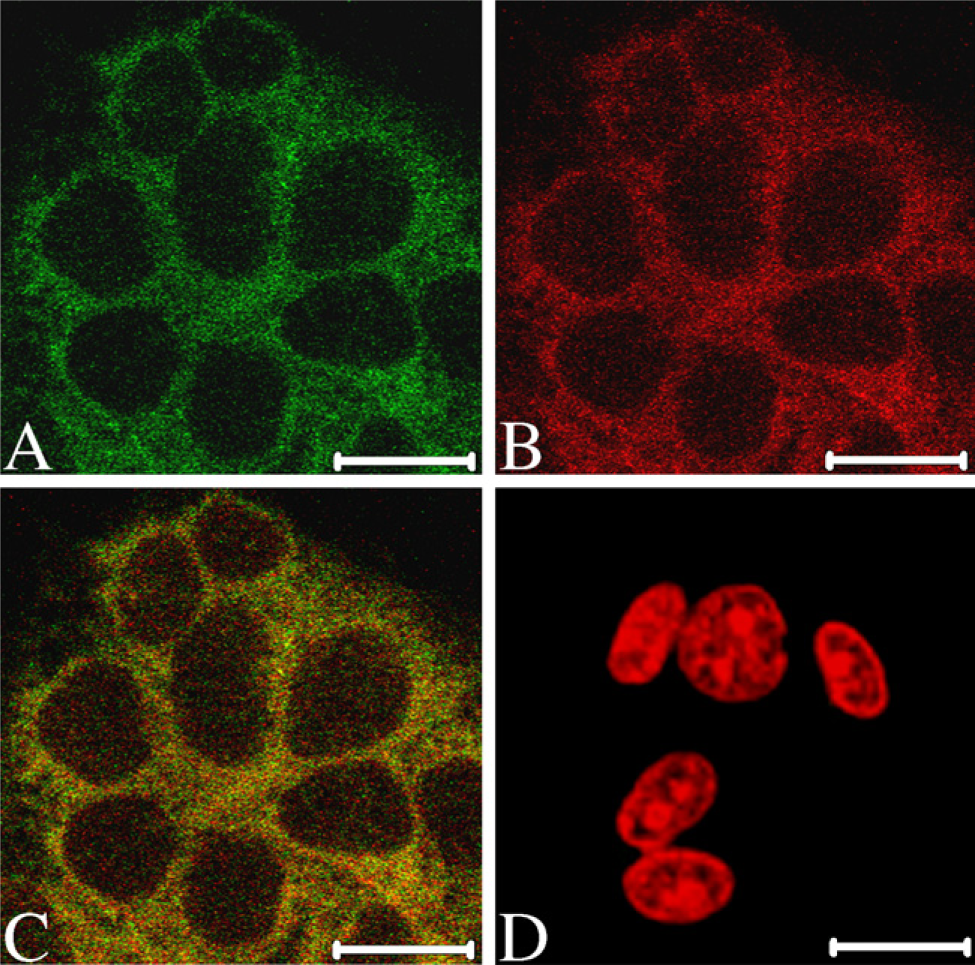
Colocalization of SWAP-70 with F-actin in human B6Tert-1 cell. (
Footnotes
Acknowledgements
The work was supported by the National Nature Science Foundation of China Project (Grant No. 30530760) and the Chinese Academy of Sciences Knowledge Innovation Program (Grant No. KSCX2-YW-R-53).
The authors thank Dr. Rolf Jessberger at Mount Sinai School of Medicine, New York, NY, for kindly providing the antibody against SWAP-70.
