Abstract
We have previously shown that postischemic injury to renal allografts results in profound impairment of p-aminohippuric acid (PAH) extraction. To elucidate the cellular integrity of the human organic anion transporter 1 (hOAT1) in postischemic acute renal failure (ARF), immunohistochemical analysis of hOAT1 was performed in cadaveric renal allografts using confocal microscopy for three-dimensional reconstruction of serial optical images. Biopsy samples were obtained from 10 cadaveric renal allografts 1 hr after reperfusion during transplant operation. Control tissues were obtained from four living donors of healthy kidneys immediately before an arterial clamp was applied to the renal artery. Control tissues demonstrated hOAT1 distributed to basolateral membrane of proximal tubule cells. In contrast, maldistribution of hOAT1 to cytoplasm and/or diminution of the protein was noted in cadaveric allografts. Characteristics of maldistribution were variable: disappearance of lateral distribution, diffuse cytoplasmic aggregates, apical cytoplasmic aggregates, and disappearance of the staining. In addition, iothalamate and PAH clearances were performed on posttransplant days 3–7 in 18 recipients of a cadaveric renal allograft. PAH clearance was depressed <250 ml/min in all but three subjects. We conclude that reperfused, transplanted kidneys exhibit maldistribution of hOAT1 in proximal tubule cells, resulting in impairment of PAH clearance. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
Accumulation of uremic toxins in ARF is attributable to two phenomena. The first is a profound reduction in the glomerular filtration rate (GFR). Alejandro et al. (1995) have shown this phenomenon in the freshly implanted renal allograft to be predominantly caused by marked depression of the transglomerular hydraulic pressure gradient, the outward driving force for filtration. In addition, backleak of filtrate through damaged tubule walls further lowers the “effective” GFR (Kwon et al. 1998). A second mechanism of diminished renal solute clearance in postischemic ARF is impairment of tubule secretory function. The role of tubular secretion in total renal solute clearance has often been disregarded, even though tubular secretion plays a major role in the clearance of many solutes; for example, 80–90% of renal PAH (para-aminohippurate; a prototype of organic anions) excretion occurs via tubular secretion. Because of its abundant extraction by the kidney, PAH is widely used to measure renal plasma flow. We have shown that PAH extraction is profoundly depressed in sustained ARF, despite no difference in renal plasma flow in recovering and sustained ARF subjects and cannot be used for this purpose (Corrigan et al. 1999). However, the underlying mechanism explaining the decreased PAH extraction has not been clarified. We studied renal allografts of cadaveric origin 1 hr after reperfusion of the allograft. At this time, a marked injury to proximal tubule cells is manifest by altered ultrastructure and a loss of normal cell polarity (Alejandro et al. 1995; Kwon et al. 1998, 1999).
We accordingly selected this model of ischemia-reperfusion injury in humans to study the fate of human organic anion transporter 1 (hOAT1), which has the highest affinity for PAH among human renal OAT isoforms and is located on the basolateral membrane of proximal tubule cells (Hosoyamada et al. 1999; Lu et al. 1999; Cha et al. 2001; Sun et al. 2001; Russel et al. 2002). We used confocal microscopy to perform a three-dimensional (3-D) reconstruction of serial optical images for detailed characterization of hOAT1. Kidney tissue was obtained by biopsy in the operating room 60 min after reperfusion of the allograft. Our findings provide the basis for this first report of hOAT1 in human kidneys after ischemic injury.
Materials and Methods
Subjects
The study group was comprised of 24 consecutive consenting recipients of a cadaveric renal allograft. None of these recipients had acute rejection or other medical or surgical complications during the first two posttransplant weeks, allowing ischemia-reperfusion injury to the allograft to be evaluated in isolation. Informed consent was obtained as approved previously by Indiana University-Purdue University in Indianapolis and Clarian Institutional Review Boards (#0005–09) and the Committee for Research in Human Subjects at Stanford University. Patients' ages ranged from 27 to 70 years. The corresponding age range of the donors varied between 16 and 59 years. Characteristics of subjects are shown in Table 1.
Protocol
A biopsy of the allograft was performed approximately 1 hr after completion of the vascular anastomosis and reperfusion of the transplanted kidney in 10 of the renal allograft recipients.
Patients' characteristics

Mean ± SEM.
Graft function was monitored daily by collecting 24-hr urine samples following surgery for creatinine clearance (Clcr), a surrogate for effective GFR. Physiological evaluation using iothalamate and PAH was performed in 18 subjects on days 3–7 after transplant. On the day of the physiology study, each patient was on prednisone, mycophenolate mofetil, tacrolimus, or cyclosporine, clotrimazole troche, sulfamethoxazole-trimethoprim, valganciclovir, famotidine, and antihypertensive medication such as metoprolol.
Control tissues were obtained from four living donor kidneys at the time of transplantation, immediately before an arterial clamp was applied to the kidney, and from a patient with IgA nephropathy with normal renal function.
Immunohistochemistry
Antibodies. Anti-hOAT1 antibody was prepared as follows: rabbits were immunized with a purified keyhole limpet hemocyanin-conjugated synthesized peptide (CKYMVPLQA-SAQEKNGL) corresponding to the C terminus of hOAT1. Polyclonal antibodies against this peptide were affinity purified (Covance Research Products; Denver, PA) from the serum of immunized rabbits for use at a dilution of 1:1000. Antibody specificity was confirmed by preabsorption of the antibody to hOAT1 peptide before immunohistochemical studies.
To identify different segments of the nephron by characteristic cellular distribution of filamentous actin (F-actin), tissue sections were dual stained with Texas red-conjugated phalloidin (Molecular Probes; Eugene, OR). In some cases, an antibody against Tamm-Horsfall protein (Accurate Chemical and Scientific Corp.; Westbury, NY) was also used at a dilution of 1:200 to identify outer medullary portion of the thick ascending limb of Henle, where S3 portion of proximal tubules may coexist.
Tissue Preparation for Immunofluorescence. Tissue fraction mainly from the cortex of renal allografts was immediately dropped into 10 ml of 2% paraformaldehyde-0.075 M lysine-0.01 M periodate fixative on ice for 30 min. After fixation, the tissue was washed three times with ice-cold PBS consisting of (in mM) 2.7 KCl, 1.5 KH2PO4, 137 NaCl, and 8 NaH2PO4. Each wash was carried out for 10 min on ice. After this step, the tissue was cryoprotected by transferring to a 50-ml conical tube containing 40 ml of 2.5 M sucrose in PBS. The tissue was allowed to remain in this solution for at least 48 hr at 4C, until the tissue was removed from the 2.5-M sucrose solution, immersed in OCT cryoembedding compound (Miles; Naperville, IL), frozeninliquid N2, and stored at −80C.
Immunofluorescence Staining. The frozen tissue block was mounted onto chucks and sectioned using a cryotome (CM 3050; Leica, Nussloch, Germany). Six-μm-thick sections were transferred onto “ProbeOn Plus” glass slides from Fisher Scientific. Frozen sections were extracted for 10 min at room temperature with cytoskeleton buffer consisting of 50 mM NaCl, 300 mM sucrose, 10 mM piperazine-N, N'-bis (2-ethane-sulfonic acid) (pH 6.8), 3 mM MgCl, 0.5% Triton X-100, and 1 mM phenylmethylsulfonyl fluoride (PMSF). Slides were then washed twice with PBS at room temperature. Each wash was carried out for 10 min. After this step, slides were incubated in blocking solution for 2 hr at room temperature in a humidified chamber. The blocking solution consisted of PBS containing 20% normal goat serum, 0.2% BSA, 50 mM NH4Cl, 25 mM glycine, and 25 mM lysine. After the slides were blocked, they were washed twice with PBS containing 0.2% BSA. Washes were carried out for 10 min at room temperature. Slides were then incubated with the affinity-purified anti-hOAT1 antibody solution at a dilution of 1:1000 overnight at 4C in a humidified chamber. The primary antibody was diluted in PBS containing 20% normal goat serum and 0.2% BSA. The following day, slides were again washed twice with PBS containing 0.2% BSA. Both washes were conducted for 10 min at room temperature. Slides were then incubated with fluorescein-conjugated anti-rabbit antibody solution and Texas red-conjugated phalloidin for 2 hr at room temperature in a humidified chamber for double-labeling experiments. Texas red-conjugated phalloidin was used to identify proximal tubules. Secondary antibodies were diluted 1:200 in PBS containing 20% normal goat serum and 0.2% BSA. After the secondary antibody incubation, slides were washed twice in PBS containing 0.2% BSA as above and then mounted with glass coverslips in PBS containing 16.7% Mowiol (Calbiochem), 33% glycerol, and 0.1% paraphenylene diamine. Slides were viewed using the MRC-1024 confocal microscope (BioRad; Hercules, CA) with a ×60 water objective lens.
Image Analysis. To evaluate hOAT1, serial images were taken at 0.2-μm intervals throughout the 6-μm depth of tissue sections using confocal microscopy. 3-D reconstructions of these images were generated with Metamorph software (Universal Imaging; West Chester, PA). Each 3-D rotating video image was evaluated to characterize the distribution of hOAT1.
Clearance Study. Clearance study using PAH and iothala-mate was performed on average on postoperative day 4 ± 1 (mean ± SD). A priming dose of PAH (6 mg/kg) and iothalalmate (900 mg) was followed by a continuous infusion calculated tomaintain plasma PAH and iothalamate concentrations constant. After a 60-min equilibration period, four timed-urine collections were made. Blood samples were drawn to bracket each urine collection. Effective PAH and iothalamate clearances (ClPAH and Clioth) were calculated as the average of the four individual clearances. Assuming that PAH is filtered at the glomerular level and leaked back across the damaged tubule walls in the same fashion as iothalamate, net tubular PAH secretion (TPAH) was derived by subtracting Clioth from ClPAH.
A high-pressure liquid chromatography (HPLC) system with an ultraviolet light detector was used to assay iothalamate and PAH at 236 nm (#LC-6A; Shimadzu Instruments, Columbia, MD). Ultrafiltrates of serum and diluted urine were injected onto a C18 reverse-phase column (Ultrasphere 5 μ; Beckman Instruments, San Ramon, CA). The mobile phase was 3.5% acetonitrile in 10 mM triethylamine, pH 3.0. Flow rate was 1.0 ml/min. Iothalamate and PAH concentrations were determined from the peak areas of each solute, corresponding to column retention times of 14 and 10 min, respectively (Lemley et al. 2000).
Statistical Analysis. To examine the extent of correlation between Clioth and TPAH, correlation coefficient (r2) was assessed. All results are expressed as mean ± 1 SE.
Results
Clinical Features
Clinical characteristics of the allografts and patient population are summarized in Table 1. Total and warm ischemic times were 1573.8 ± 72.2 and 37.4 ± 1.9 min, respectively.
Distribution of hOAT1 Protein
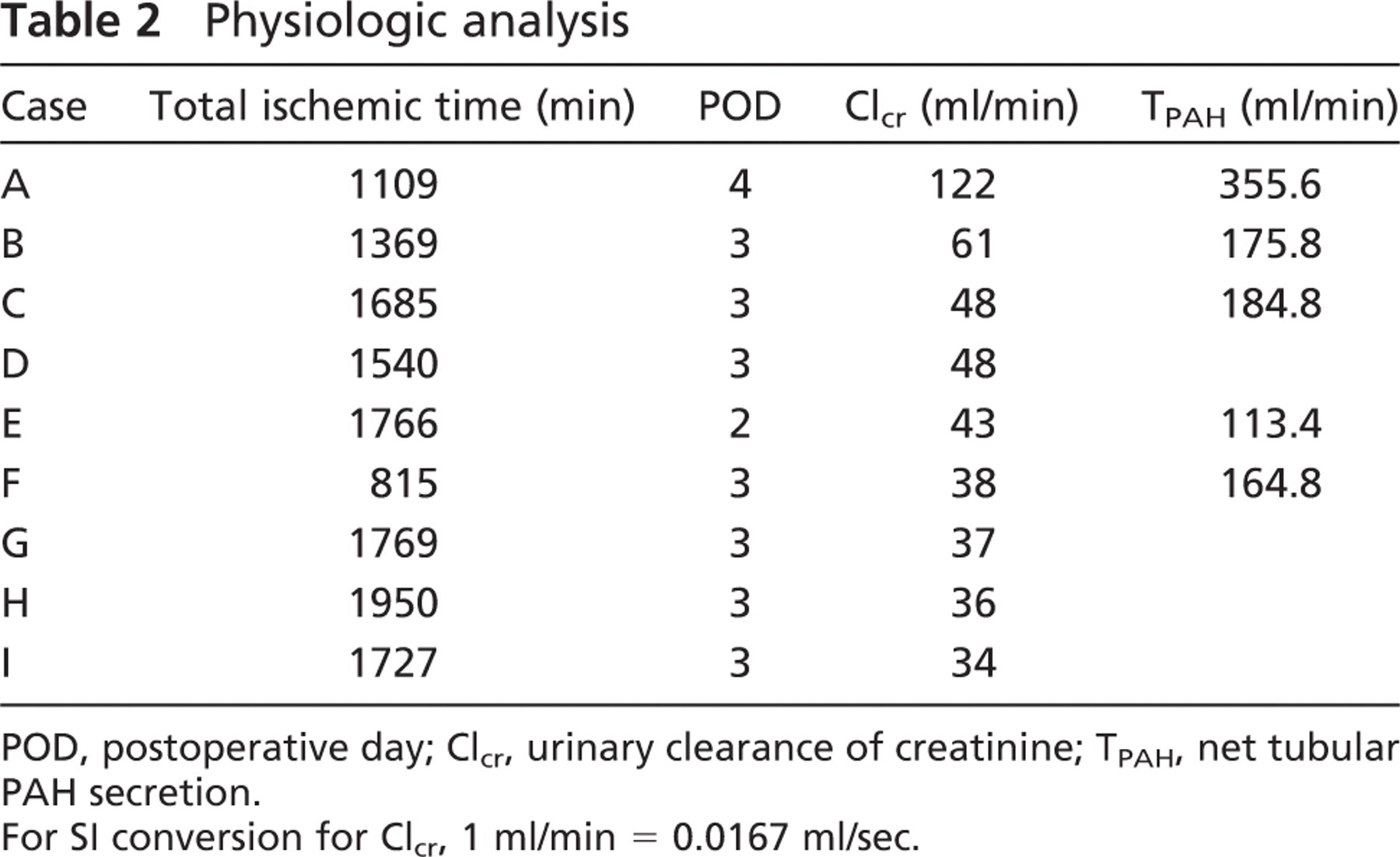
Staining for hOAT1 is localized to the basolateral membrane of proximal tubule cells in control tissues (Figure 1 and Supplemental Movie 1). Staining is present all throughout the proximal tubule segments. Proximal tubule segments are identified by the presence of brush border shown in F-actin staining (shown in red, Figure 1, right panel). hOAT1 staining in proximal tubule cells frequently appears as a stringy pattern that corresponds to a tangential view of lateral membrane interdigitation (arrowhead, Figure 1, left panel). Tissues from cadaveric renal allografts demonstrate maldistribution of hOAT1 at the subcellular level. The maldistribution pattern varies among individuals and among cells. Heterogeneity is more pronounced among individuals. Excluding one case containing only medullary portion of the kidney, the findings of hOAT1 distribution in nine cadaveric renal allografts are described as follows. Each individual case is described following the order of magnitude of postischemic renal injury, as judged by Clcr on posttransplant days 2–4, from the highest to the lowest. Representative images are illustrated in Figure 2-Figure 4 following a sequence of the changes in hOAT1 location that we hypothesize to reflect its redistribution after ischemia and reperfusion. One alphabetical letter indicates one subject. Physiological characteristics of individual cases are shown in Table 2.
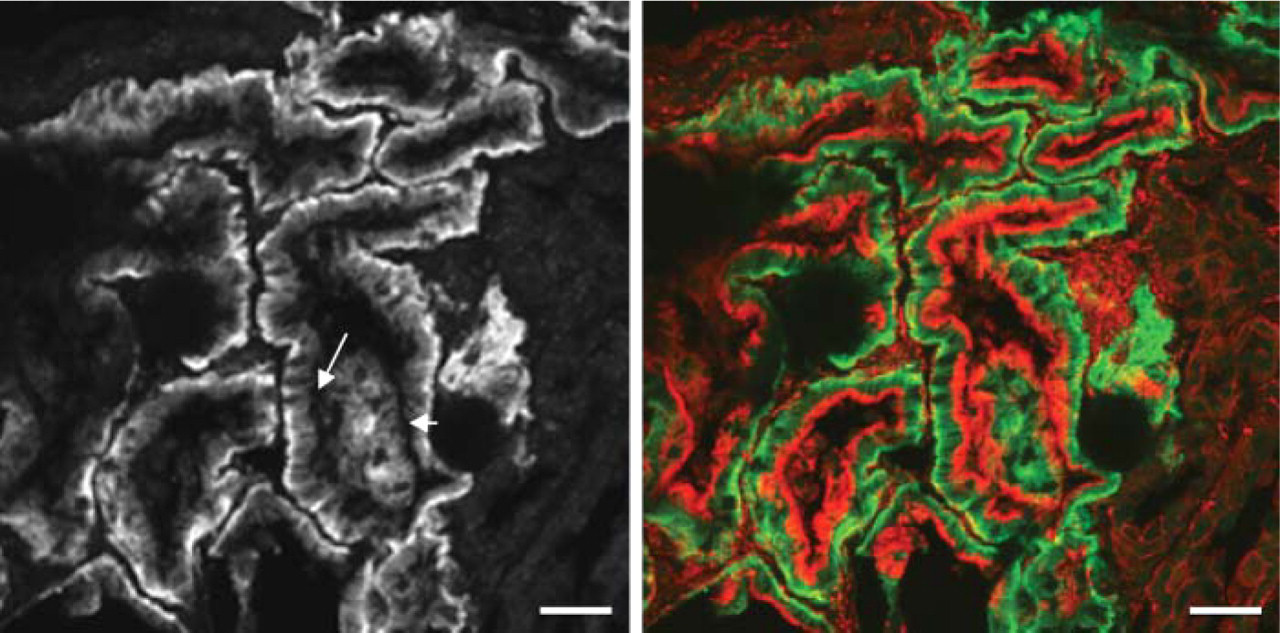
Fluorescence microscopy of human organic anion transporter 1 (hOAT1) in control tissue showing basolateral distribution in proximal tubule cells (left panel and green in right panel). Kidney tissue was dual stained with Texas red-conjugated phalloidin for F-actin (right panel). Proximal tubule cells are identified by the presence of brush border. hOAT1 is present in all proximal tubule cells (right panel). Arrows in the left panel indicate basolateral membrane (arrow) and a tangential view of basolateral interdigitation (arrowhead) stained for hOAT1. Bar = 20 μm.
Case A. Staining for hOAT1 is distributed to the basolateral membrane domain of proximal tubule cells with some fine cytoplasmic dots and occasional intraluminal aggregates (Figure 2A). In some proximal tubule cells, lateral and basal staining is markedly diminished with more fine cytoplasmic dots. Some areas of the tissue section show only patchy basal and faint lateral staining for hOAT1 with no cytoplasmic dots of the staining.
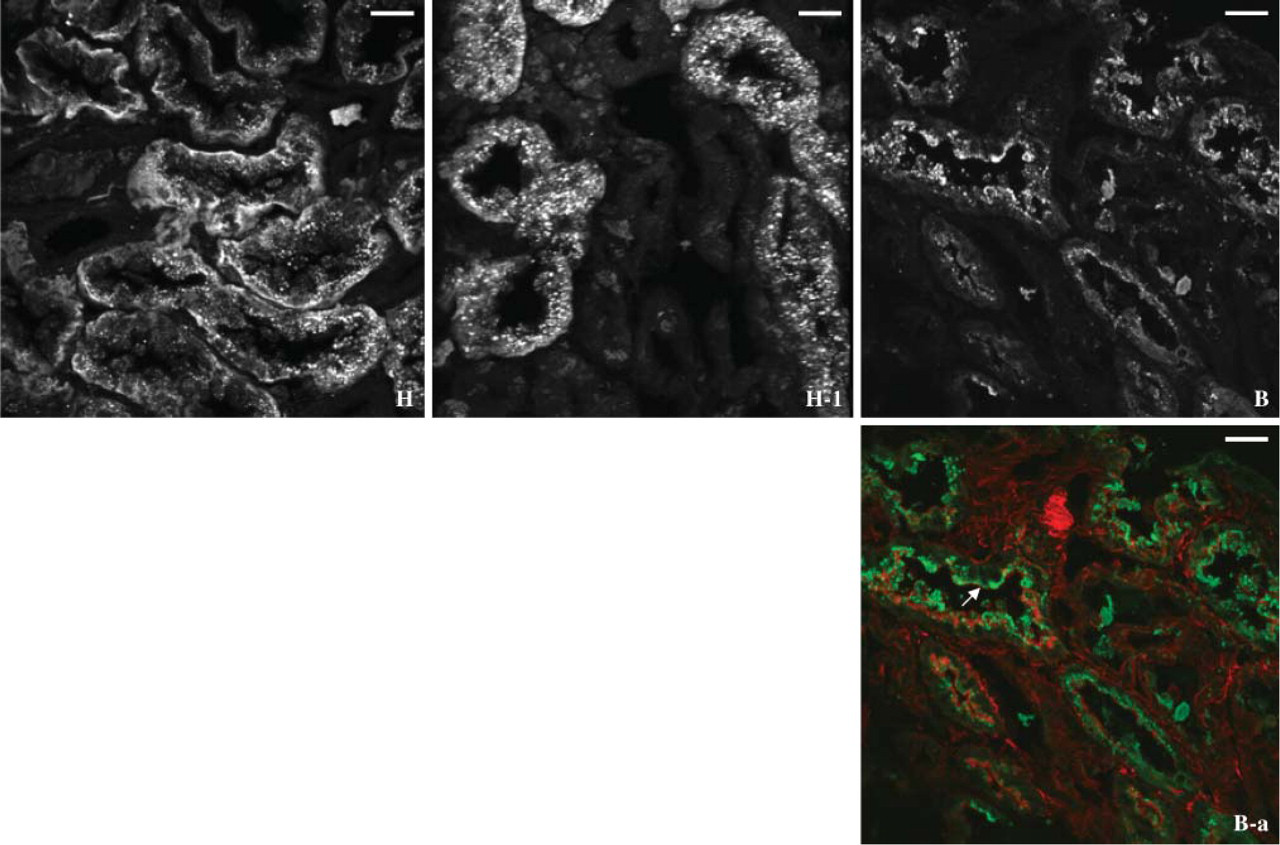
Case B. Most of the staining for hOAT1 is present in the form of large coarse aggregates (Figure 3B). The aggregates are mainly redistributed to the apical cytoplasm, judged by dual staining for F-actin (Figure 3B-a, shown in red). An orange color would be seen if hOAT1 was colocalized with F-actin in the brush border.
Case C. hOAT1 staining is distributed to the baso-lateral membrane domain of proximal tubule cells. Intensity of basal staining is heterogeneous and the lateral membrane staining is discerned only in a few areas. Rarely, fine dots of the cytoplasmic staining are also observed (Figure 2C).
Case D. Strong basal staining for hOAT1 is observed with faint lateral membrane staining. Fine dots of the staining for hOAT1 are scattered in the cytoplasm of many proximal tubule cells (Figure 2D).
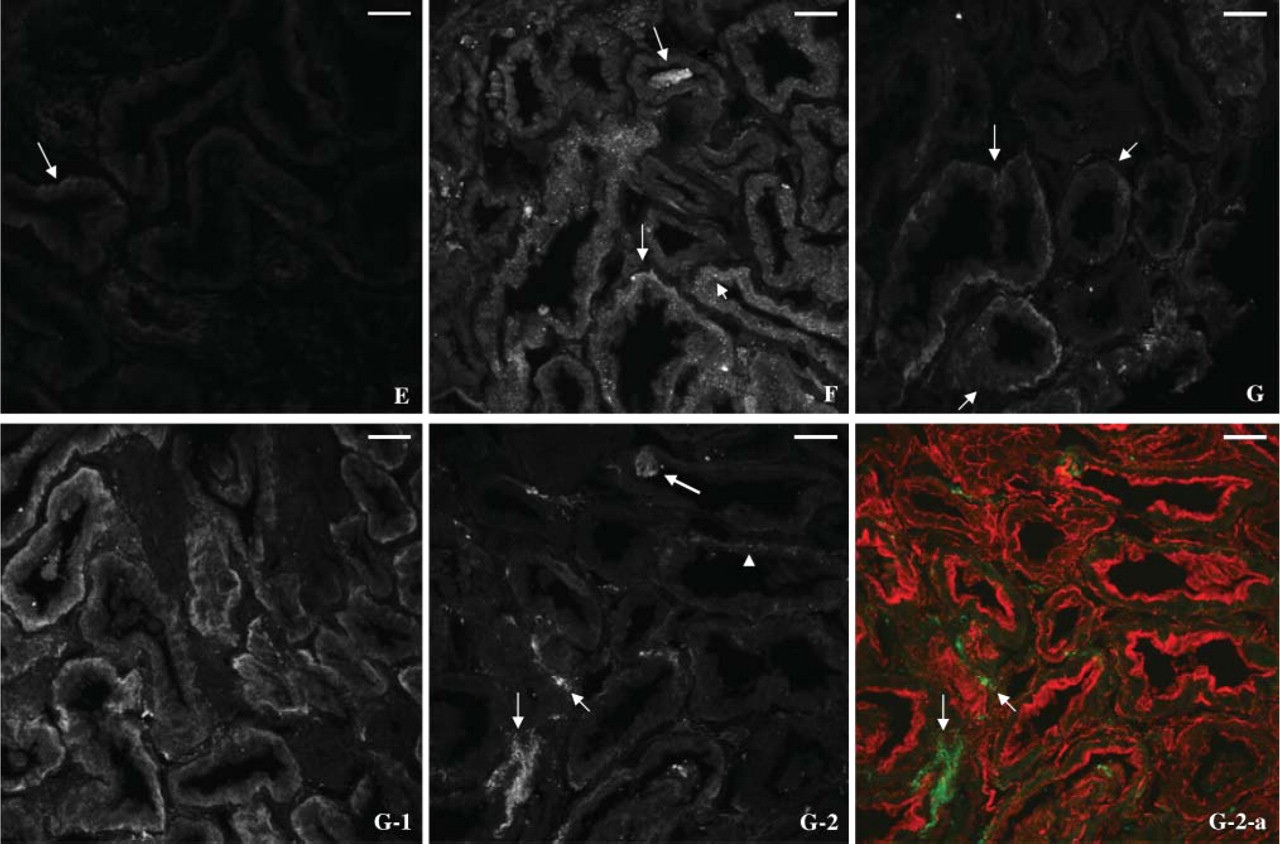
Case E. Staining for hOAT1 is almost completely absent in the tissue section except that very faint patchy basal staining is rarely observed (Figure 4E).
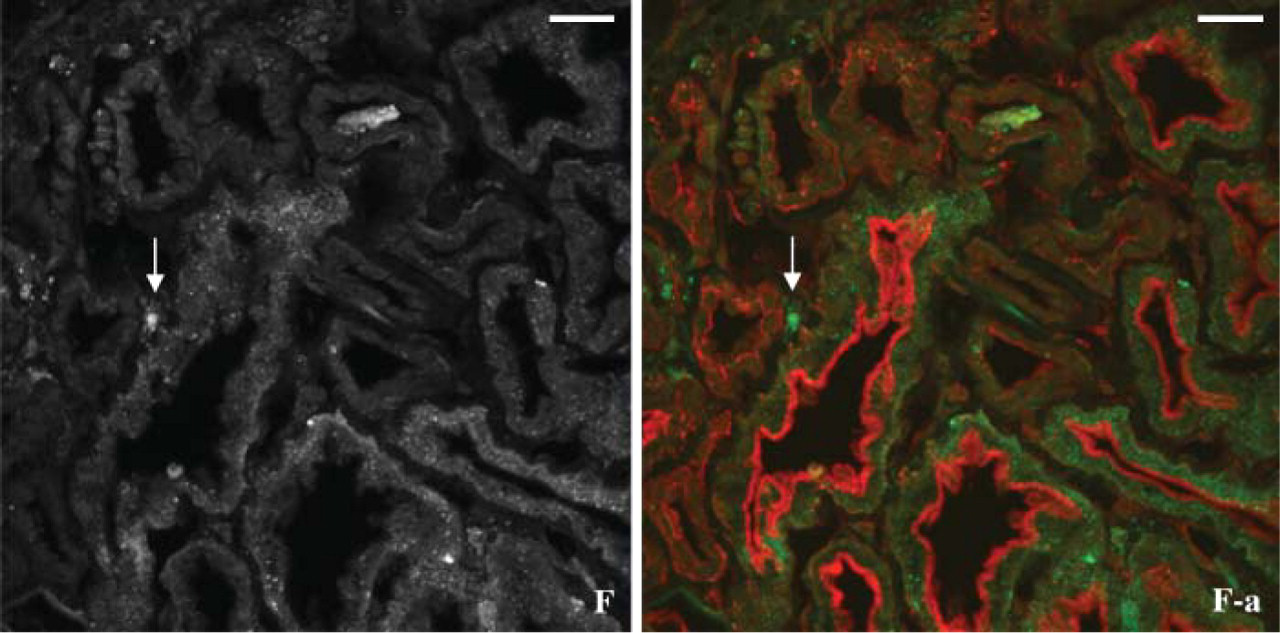
Case F. Most of the staining for hOAT1 is observed as fine diffuse cytoplasmic aggregates (Figure 4F and Supplemental Movie 2). Also, diffuse cytoplasmic staining is above the level of background. Occasional patchy basal staining for hOAT1 is also noted as well as intra-luminal aggregates. Interestingly, occasional patchy staining is also detected in peritubular interstitial area (Figure 4F, Figure 5F, and Figure 5F-a, shown in green; Supplemental Movie 2).
Case G. Staining characteristics are heterogeneous with diminished overall intensity of the staining (Figures 4G, G-1, and G-2). Some proximal tubules show patchy basal and almost no lateral staining (Figure 4G). Some proximal tubule cells show heterogeneous basal distribution and faint lateral staining for hOAT1 (Figure 4G-1). Strikingly, no staining for hOAT1 is observed in some proximal tubule cells except for a few fine cytoplasmic dots and intraluminal aggregates, but with occasional positive staining in the peritubular area (Figure 4G-2 and 4G-2-a, shown in green).
Case H. Staining for hOAT1 is remarkable for diffuse large coarse cytoplasmic aggregates with almost no basolateral membrane staining (Figure 3H-1). In some proximal tubule segments, patchy basal staining is also observed (Figure 3H and Supplemental Movie 3). Cytoplasmic aggregates are variable in size.
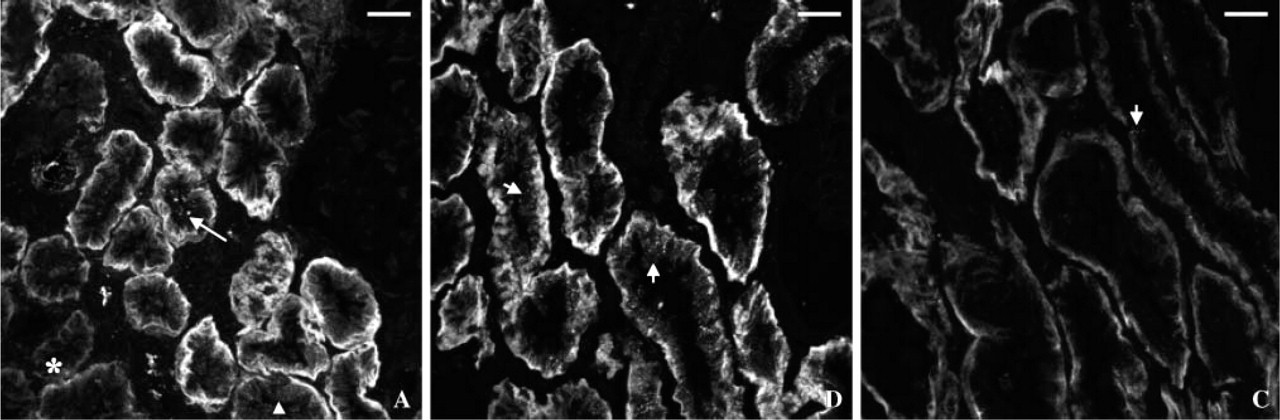
Fluorescence microscopy of hOAT1 in human cadaveric renal allografts. Case
Fluorescence microscopy of hOAT1 in human cadaveric renal allografts; cases
Case I. Staining for hOAT1 is distributed to the basal membrane and is also present in cytoplasmic aggregates (not shown).
Renal Function
Five subjects (A, B, C, E, and F) had an intraoperative graft biopsy and also had undergone postoperative physiological assessment for TPAH. Their TPAH was 355, 175, 184, 113, and 164 ml/min in A, B, C, E, and F, respectively (Table 2). Case A, who demonstrated the best-preserved basal and lateral hOAT1 distribution among subjects, manifested the highest TPAH, 355 ml/min. Case E, who revealed almost no staining for hOAT1, had the lowest TPAH, 113 ml/min. Cases B, C, and F showed TPAH of 175, 184, and 164 ml/min with variable hOAT1 distribution: apical cytoplasmic aggregates in variable sizes, diminished basolateral distribution with rare cytoplasmic aggregates, and diffuse fine cytoplasmic aggregates with faint basal staining, respectively.
In 18 subjects who underwent the physiology study using iothalamate and PAH, the degree of correlation between Clioth and TPAH was examined. As shown in Figure 6, the values of TPAH were quite variable, even at the same level of Clioth, with r 2 = 0.471.
Discussion
In the present study we have shown that, in the freshly transplanted human renal allograft, hOAT1 is maldistributed. An ensuing impairment of organic anion transport results in reduction in renal tubular PAH secretion.
Glomerular filtration has become the dominant measure of renal function in clinical medicine. But the focus on filtration, indeed its virtual identification with kidney function in practice, may have distracted physiologists from the study of tubular organic anion transport and tubular secretion. Many substances are cleared more rapidly by tubular secretion than by glomerular filtration. Some such substances are probably toxins whose levels are maintained low by normal kidney function (Hostetter and Meyer 2004). Reduction in solute clearance through glomerular filtration in ARF has also been the major focus of most investigators. In acute renal failure, tubular secretion of organic solutes may be markedly depressed and contribute to the uremic state.
Fluorescence microscopy of hOAT1 in human cadaveric renal allografts. Case
The kidney plays an essential role in the elimination of numerous organic anions, which are a group of chemically heterogeneous substances with carbon backbones and negative charges at physiological pH. Organic anions include numerous exogenous and endogenous compounds and their metabolites. Examples are antibiotics (penicillin, cephalosporin, sulfa drugs, acyclovir), various chemotherapeutic agents used to treat malignancy, diuretics, non-steroidal anti-inflammatory drugs, angiotensin-converting enzyme inhibitors, radiocontrast agents, drug metabolites, toxicants and their metabolites, prostaglandins, bile acids, and creatinine (Moller and Sheikh 1983; Russel et al. 2002; Burckhardt and Burckhardt 2003). Some of these organic anions, for example, indoxyl sulfate, 3-carboxy-4-methyl-5-propyl-2-furanpropionate, indoleacetate, hippurate, and p-cresol have been proposed to contribute to the uremic syndrome (Gulyassy et al. 1987; Kawashima et al. 1987; Spustova et al. 1989; Vanholder et al. 1995; Niwa 1996; Ringoir 1997; Vanholder and De Smet 1999; Dzurik et al. 2001; Vanholder et al. 2001; Motojima et al. 2002; Ohtsuki et al. 2002; Tsutsumi et al. 2002; Satoh et al. 2003; Vanholder et al. 2003; Dou et al. 2004). Recently, transport of these organic anions by rOAT1/hOAT1 has been demonstrated in kidney epithelial cell lines (Deguchi et al. 2004).
Physiologic analysis
POD, postoperative day; Clcr, urinary clearance of creatinine; TPAH, net tubular PAH secretion.
For SI conversion for Clcr, 1 ml/min = 0.0167 ml/sec.
Fluorescence microscopy of hOAT1 in a human cadaveric renal allograft, case
In the kidney, proximal tubule cells actively secrete organic anions into the urine via organic anion transporters, which possess broad substrate specificity (Moller and Sheikh 1983; Russel et al. 2002). Recently, a family of organic anion transporters (OAT1-6) has been cloned. Our understanding of their molecular and functional characteristics has increased (Lopez-Nieto et al. 1997; Sekine et al. 1997, 1998, 2006; Hosoyamada et al.1999; Kusuhara et al. 1999; Cha et al. 2000; You 2002; Monte et al. 2004; Youngblood and Sweet 2004; Anzai et al. 2005, 2006; Schnabolk et al. 2006). hOAT1 is a prototypical organic anion transporter with the highest affinity for PAH among the OAT isoforms; transport affinity (Km) of hOAT1 for PAH is 5–9.3 μM, whereas Km of hOAT3 for PAH is 87 μM and OAT2 is present predominantly in the liver. It has been reported that human kidneys contain approximately 2-fold more hOAT3 than hOAT1 in mRNA expression and protein amount (Motohashi et al 2002). However, hOAT1 has been recognized as the major OAT in human kidneys in view of its high affinity for PAH. It is a classical PAH/dicarboxylate exchanger mediating high-affinity uptake of PAH by an active sodium-dependent tertiary process in the basolateral membrane of proximal tubule cells (Hosoyamada et al. 1999; Lu et al 1999; Cha et al. 2001; Sun et al. 2001; Sweet et al. 2001; Russel et al. 2002). It has been reported that four OAT1 isoforms (hOAT1-1, 2, 3, and 4) are constitutively expressed in human kidneys, and hOAT1-2 is the main transcript in kidney cortex and contributes to the excretion of PAH (Bahn et al. 2004). Twenty single nucleotide polymorphisms in hOAT1 were identified in genomic DNA from individuals of African, Asian, and Caucasian origin, but kinetic analysis indicated that the Km for PAH was unchanged in the variants (Bleasby et al. 2005). Previous reports suggested that OAT1 and OAT3 were predominantly expressed in males compared with females in experimental animals (Cerrutti et al. 2002; Buist and Klaassen 2004; Ljubojevic et al. 2004). Gender difference has not yet been known in humans. The current study included all female and male subjects to illustrate all the dynamic alterations of hOAT1 after ischemia in human kidneys. Relatively little is known about the apical transport of PAH. Smeets et al. (2004) reported that multidrug-resistance proteins (MRP) 2 and 4, predominantly MRP4, were localized to the apical membrane and mediated ATP-dependent PAH transport. It has also been reported that PAH uptake decreases in rat renal cortical slices associated with loss of basolateral OAT1 (Crljen et al. 2005). In addition, downregulation of renal sodium-dependent phosphate cotransporter NaPi2 (Npt2) has been reported in experimental rats with acute renal failure due to ischemia and reperfusion (Rubinger et al. 2005). Post-ischemic injury to human renal allografts resulted in profound impairment of PAH extraction, even with the comparable renal blood flow. This finding strongly suggested that organic anion transporters were dysfunctional after an ischemic insult (Corrigan et al. 1999). However, the pathobiology of organic anion transporters in postischemic ARF has not been clarified in humans.
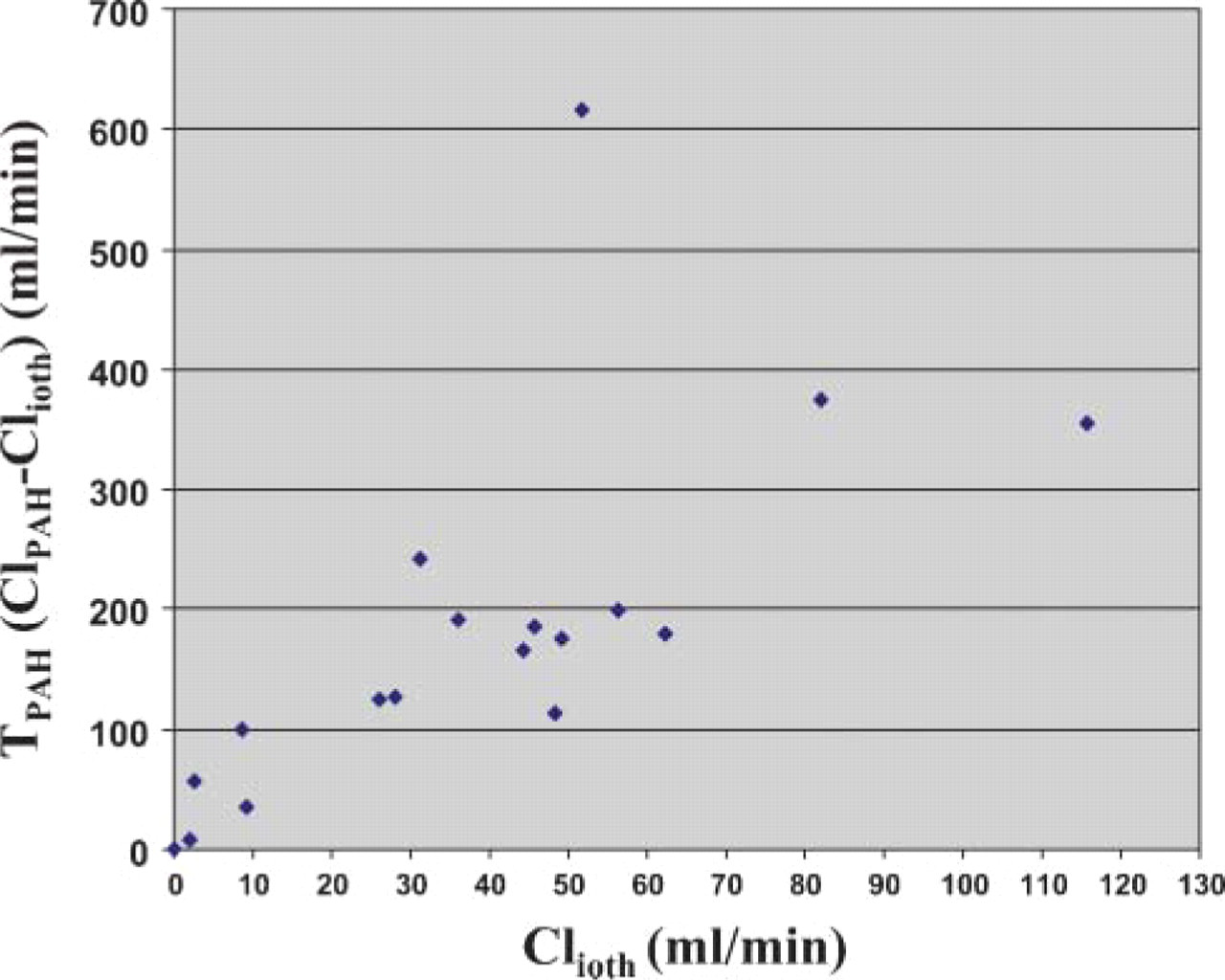
Physiological analysis of cadaveric renal allografts during the first week of transplantation showing a poor correlation between net tubular PAH secretion (TPAH) and iothalamate clearance (Clioth). r 2 = 0.471.
We hypothesized that an alteration in proximal tubule cell OAT1 after ischemia could disrupt tubular organic anion secretion, resulting in the reduction in total renal solute clearance. Freshly transplanted cadaveric renal allografts were analyzed because they have been shown to invariably exhibit an ischemia-reperfusion injury, which results in a form of postischemic ARF known as “delayed graft function” in humans (Alejandro et al. 1995; Kwon et al. 1998, 1999; Corrigan et al. 1999; Hladunewich et al. 2003). During the transplant procedure, a previously healthy kidney sustains a period of cold and warm ischemia before implantation and reperfusion. In contrast to native kidney ARF in humans, there are typically no confounding factors such as sepsis, multiorgan failure, or exposure to nephro-toxins after the initial measurable period of ischemia. As many as 50% of all grafts fail to function promptly following the operation (Halloran and Hunsicker 2001). Each recipient of a renal allograft in our study was hemodynamically stable without other active medical or surgical conditions that could cause independent and confounding damage to the kidney within the first two posttransplant weeks. Using biopsy tissues obtained from the cadaveric renal allografts following an average of 26 hr of ischemia and ~60 min of reperfusion, we performed immunohistochemical analysis of hOAT1. Physiological analysis of the allografts was also performed during the first posttransplant week to estimate for effective GFR and TPAH. On the day of physiology study, subjects were on medications such as sulfameth-oxazole and valganciclovir, which were transported by OAT1, possibly competing with PAH secretion. Nonetheless, it is reasonable to compare TPAH among the subjects because all subjects were on the same medical regimen at the time of the study.
It has been shown that OAT1 is exclusively expressed in the basolateral membrane of S2 and S3 segments of proximal tubules in rats (Tojo et al. 1999; Kojima et al 2002). A previous report demonstrated that hOAT1 was localized to the basolateral membrane of proximal tubules in human renal cortex (Motohashi et al. 2002). The current study also reveals that, in human kidneys, hOAT1 is distributed to the basolateral membrane of proximal tubule cells throughout all three of the proximal tubule segments. Cadaveric renal allografts that had sustained ischemic damage showed variable dynamic alterations in hOAT1 distribution (Figure 2-Figure 5). Polarized renal tubular epithelial cells display distinct patterns of protein distribution in their apical and basolateral membrane domain, which is essential to vectorial transport of solutes. hOAT1 examined in the current study is normally distributed to the basolateral membrane domain (Figure 1). Following our hypothetical sequence, hOAT1 loses its normal basolateral membrane localization after ischemia and reperfusion. The dislodged hOAT1 is present in forms of cytoplasmic aggregates, appearing first as fine granules and later as larger coarse irregular clumps. Cytoplasmic aggregates are localized to the apical cytoplasm in an advanced stage. Thereafter, hOAT1 can almost completely disappear. Its reappearance takes forms of fine cytoplasmic granules. Then, hOAT1 begins to be redistributed in the basal membrane domain and later in the lateral membrane. At this stage, the pattern of hOAT1 distribution becomes very heterogeneous. Interestingly, positive staining for hOAT1 is also detected in a peritubular location. Peritubular cells stained for hOAT1 may be peritubular capillary cells, although that remains to be confirmed. Based on our findings, we propose that each subject may represent a different stage of dynamic alteration of hOAT1 after ischemia and reperfusion. Many of the findings resemble a demonstration of similar maldistribution of Na+/K+-ATPase from the normal basolateral membrane to the cytoplasm after ischemia (Kwon et al. 1999). hOAT1 staining in the present study differs from that of Na+/K+-ATPase mainly in that cytoplasmic aggregates are more prominent from ischemia-reperfusion injury. Whether protein-sorting pathways are cell-type specific or protein specific during recovery is not entirely clear. Nonetheless, three sites of protein sorting were identified: the trans-Golgi network, endosomes, and the plasma membrane (Nelson and Rodriguez-Boulan 2004). Our hypothetical sequence of sorting of the newly formed hOAT1 follows the general concept.
It has been reported that protein kinase C activation downregulates hOAT1-mediated transport through carrier internalization in oocytes and HEK293 cells (Wolff et al. 2003). Whether or not protein kinase C plays a role in the maldistribution of hOAT1 we observed in human kidneys after ischemia-reperfusion remains to be clarified.
Five organic anion transporters, hOAT1-5 have been isolated from human kidney and liver. hOAT1, 2, and 3 are expressed in the kidney and hOAT2, 4, 5 are expressed in the liver. OAT2 is predominantly expressed in the liver (Sun et al. 2001). hOAT1 is known to have 10–20 times higher affinity for PAH than that of OAT3 (Hosoyamada et al. 1999; Lu et al. 1999; Cha et al. 2001). Recently, a remarkable reduction of PAH secretion has been reported in OAT1 knockout mice (Eraly et al. 2006). The profound reduction of TPAH in postischemic ARF may be due to the maldistribution/dysfunction of hOAT1 as observed in our study. Interestingly, our subjects with maldistributed or absent hOAT1 (subjects B, C, E, and F) still showed some TPAH (Table 2). We can only speculate that there may be enough functional hOAT1 left in the basolateral membrane to secrete such an amount of PAH and/or other isoform, most likely, OAT3 may have contributed to the PAH secretion.
Of note, TPAH was quite variable even at the same level of Clioth as shown in Figure 6. This finding suggests that tubular damage reflected by impaired TPAH and dissipation of glomerular filtration may not necessarily correlate with each other after ischemia-reperfusion.
To summarize, we have shown that human kidneys exhibit abnormal expression and cellular localization of hOAT1 after an ischemic insult. We propose that an ensuing decrease in tubular organic anion secretion may contribute to the uremic syndrome that accompanies postischemic ARF.
Footnotes
Acknowledgements
This study was supported by Department of Medicine, Indiana University School of Medicine, Indianapolis, Indiana.
The authors gratefully acknowledge the help of Scott Herring and Sharon Henson with recruiting subjects at Indiana University. The authors thank Drs. Mark Pescovitz, Martin L. Milgrom, Ronald S. Filo, and Jonathan Fridell of Indiana University for allowing us to obtain tissue samples intraoperatively. We also thank Dr. Bryan D. Myers at Stanford University for helpful suggestions.
