Abstract
The recent success of pancreatic islet transplantation has generated considerable enthusiasm. To better understand the quality and characteristics of human islets used for transplantation, we performed detailed analysis of islet architecture and composition using confocal laser scanning microscopy. Human islets from six separate isolations provided by three different islet isolation centers were compared with isolated mouse and non-human primate islets. As expected from histological sections of murine pancreas, in isolated murine islets α and δ cells resided at the periphery of the β-cell core. However, human islets were markedly different in that α, β, and δ cells were dispersed throughout the islet. This pattern of cell distribution was present in all human islet preparations and islets of various sizes and was also seen in histological sections of human pancreas. The architecture of isolated non-human primate islets was very similar to that of human islets. Using an image analysis program, we calculated the volume of α, β, and δ cells. In contrast to murine islets, we found that populations of islet cell types varied considerably in human islets. The results indicate that human islets not only are quite heterogeneous in terms of cell composition but also have a substantially different architecture from widely studied murine islets.
P
Materials and Methods
Animals
Adult C57BL/6 mice were obtained from the Jackson Laboratory (Bar Harbor, ME), adult B6D2 F1 mice were purchased from Harlan (Indianapolis, IN), and adult wild-type mice on mixed background of 129/Sv and Black Swiss (Offield et al. 1996) were used.
Mouse Islet Isolation
Islets from mice of three different strains were isolated by dissection of the splenic portion of pancreas followed by digestion with collagenase P (Boehringer Mannheim; Indianapolis, IN) (Brissova et al. 2002). Groups of two pancreata were digested in 2 mg collagenase/pancreas in Hanks’ balanced salt solution (0.6 mg collagenase/ml) for 6–9 min at 37C using a wrist-action shaker. Mouse islets were subjected to immunohistochemical analysis immediately after isolation.
Human and Non-human Primate Islets
Human islets were prepared by the Cell Processing Unit, Department of Transfusion Medicine, National Institutes of Health (Hirshberg et al. 2003a) or obtained through the Juvenile Diabetes Research Foundation Human Islet Distribution Program [islets studied were provided by the Diabetes Institute for Immunology and Transplantation at the University of Minnesota (Bernhard J. Hering, Jeffrey Ansite, and Hui-Jian Zhang) and The Diabetes Research Institute Islet Cell Resource at the University of Miami (Camilo Ricordi)]. Non-human primate islets from rhesus macaques were prepared by the Islet and Autoimmunity Branch of the NIH (Hirshberg et al. 2002a,b). After isolation, human and non-human primate islets were shipped in CMRL media by overnight courier to Vanderbilt University and cultured for an additional 24 hr in CMRL media, 95% CO2/5% O2 at 37C. Human and non-human primate islets were subjected to immunohistochemical analysis ≃48 hr after isolation (approximate time required for islet shipping). At the same time, the health of human islet preparations was examined in the cell perifusion system (Wang et al. 1997) and perifusate fractions were assayed for insulin by radioimmunoassay (Brissova et al. 2002). Four hundred-islet equivalents were perifused at 1 ml/min flow rate and 3-min fractions were collected after a 30-min equilibration period in 2.8 mM glucose. In addition to 16.8 mM glucose, two islet preparations were also tested for their responsiveness to a combination of 16.8 mM glucose + 250 μM IBMX and 2.8 mM glucose + 300 μM tolbutamide.
Antibodies
Guinea pig anti-human insulin IgG (1:1000) and rabbit antiglucagon IgG (1:5000) were from Linco Research, Inc. (St. Charles, MO), sheep anti-somatostatin IgG (1:1000) was from American Research Products, Inc. (Belmont, MA). The antigens were visualized using appropriate secondary antibodies conjugated with Cy2, Cy3, and Cy5 fluorophores (1:1000) from Jackson ImmunoResearch Laboratories, Inc. (West Grove, PA).
Immunocytochemistry Performed on Histological Sections
Dissected adult mouse pancreata were rinsed in ice-cold 10 mM PBS and fixed in freshly prepared 4% paraformaldehyde (Electron Microscopy Sciences; Washington, PA)/100 mM PBS for 1.5 hr on ice. Following fixation, the tissues were washed four to six times with 100 mM PBS over a period of 2 hr and then equilibrated in 30% sucrose/10 mM PBS overnight at 4C. The tissues were cryopreserved in optimum cutting temperature compound (VWR Scientific Products; Willard, OH) at -80C, and 8-μm sections were mounted on charged slides. Human pancreatic tissue obtained from the NIH (Hirshberg et al. 2003a) was processed the same way as the mouse pancreas. Cryosections were permeabilized in 0.2% Triton X-100 for 10 min at room temperature, blocked with 5% normal donkey serum (Jackson ImmunoResearch Laboratories, Inc.) for 1.5 hr, and then incubated with primary antibodies overnight at 4C. Secondary antibodies were applied to the tissue sections for 1 hr at room temperature. Both primary and secondary antibodies were diluted in 10 mM PBS containing 1% BSA and 0.1% Triton X-100. Digital images of the 8-μm cryosections mounted with AquaPoly/Mount (Polysciences; Warrington, PA) were acquired with a MagnaFire digital camera (Optronics; Goleta, CA) connected to an Olympus BX-41 fluorescence microscope (Olympus; Tokyo, Japan).
Immunocytochemistry Performed on Wholemount Islets
Freshly isolated islets were attached to MatTek dishes (Cat. #P35G-0–14-C; MatTek Corporation, Ashland, MA) precoated with CELL-TAK adhesive (Becton Dickinson Lab-ware; Bedford, MA) and fixed in 4% paraformaldehyde/10 mM PBS for 25 min at room temperature. To ensure that the entire islet was optically sectioned, we chose islets ranging from 60 μm to 100 μm in (z) dimension for study. Because islets are known for not having a perfectly spherical shape, their size in (x) and (y) dimensions varied as much as from 60 μm to 200 μm. The fixation was followed by three 30-min washes in 10 mM PBS and 3-hr permeabilization with 0.3% Triton-X 100/10 mM PBS. The islets were blocked with 5% normal donkey serum/0.15% Triton-X 100/10 mM PBS overnight at 4C and then equilibrated in antibody dilution buffer twice for 20 min at room temperature. The primary and secondary antibodies were diluted in 1% BSA/ 0.2% Triton X-100/10 mM PBS and the incubations were carried out for 24 hr at 4C. The islets were mounted with AquaPoly/Mount (Polysciences). Samples were subjected to optical sectioning at 1-μm increments in axial (z) dimension using a Zeiss LSM410 confocal laser scanning microscope (Carl Zeiss; Jena, Germany).
Quantification of Endocrine Cell Types in the Isolated Islets
Optical sections of isolated islets were analyzed and three-dimensionally (3-D) reconstructed using MetaMorph v6.1 software (Universal Imaging Corporation; Downington, PA). Calibrated RGB overlays of the z-stacks for each individual islet were color thresholded, and then the contribution of islet cells labeled in red (VR, α cells), green (VG, β cells), and blue (VB, δ cells) to the islet volume (V = VR + VG + VB; μm3) was measured. The population of each islet cell type was expressed as a fraction of the total islet volume: VR/V; VG/V; VB/V (%).
Statistical Analysis
Unpaired t-test was used to compare populations of β, α, and δ cells in mouse and human islets.
Results
Confocal Microscopy of Mouse, Human, and Non-human Primate Islets
Laser scanning confocal microscopy, unlike conventional fluorescence microscopy, collects light from a single focal plane. It scans the specimen point-by-point, line-by-line and assembles the pixel information into a single image. By moving the focal plane in axial (z) dimension, single images (or optical slices) can be put together and built up a three-dimensional stack (see schematic in Figure 1). In our case, the optical sections through islets in axial (z) dimension were spaced by 1 μm. In this way, we systematically examined (a) six separate isolations of human islets (n=32 islets) provided by three different human islet isolation centers, (b) two separate isolations of non-human primate islets (n=9), and (c) three separate isolations of mouse islets (n=28 islets).
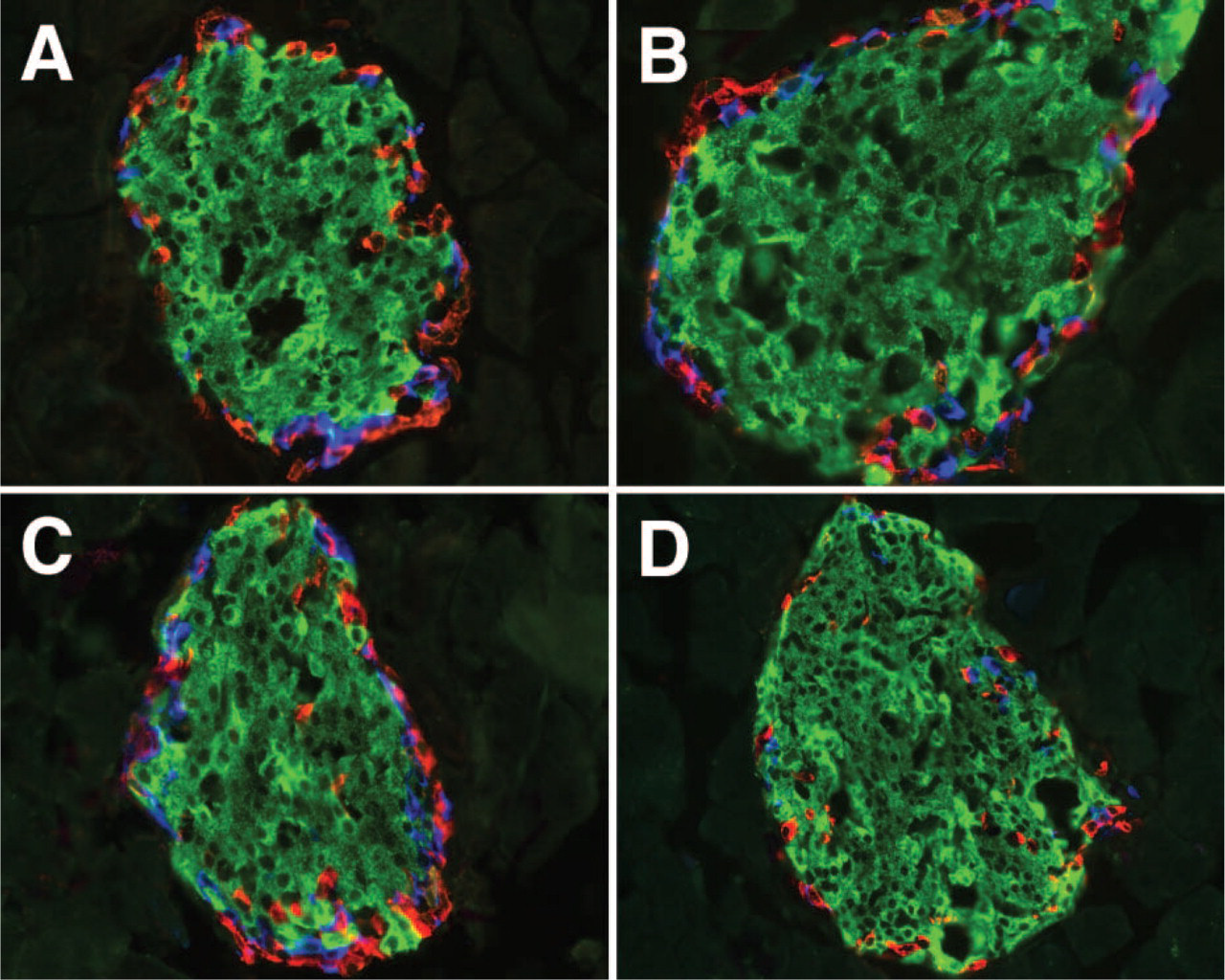
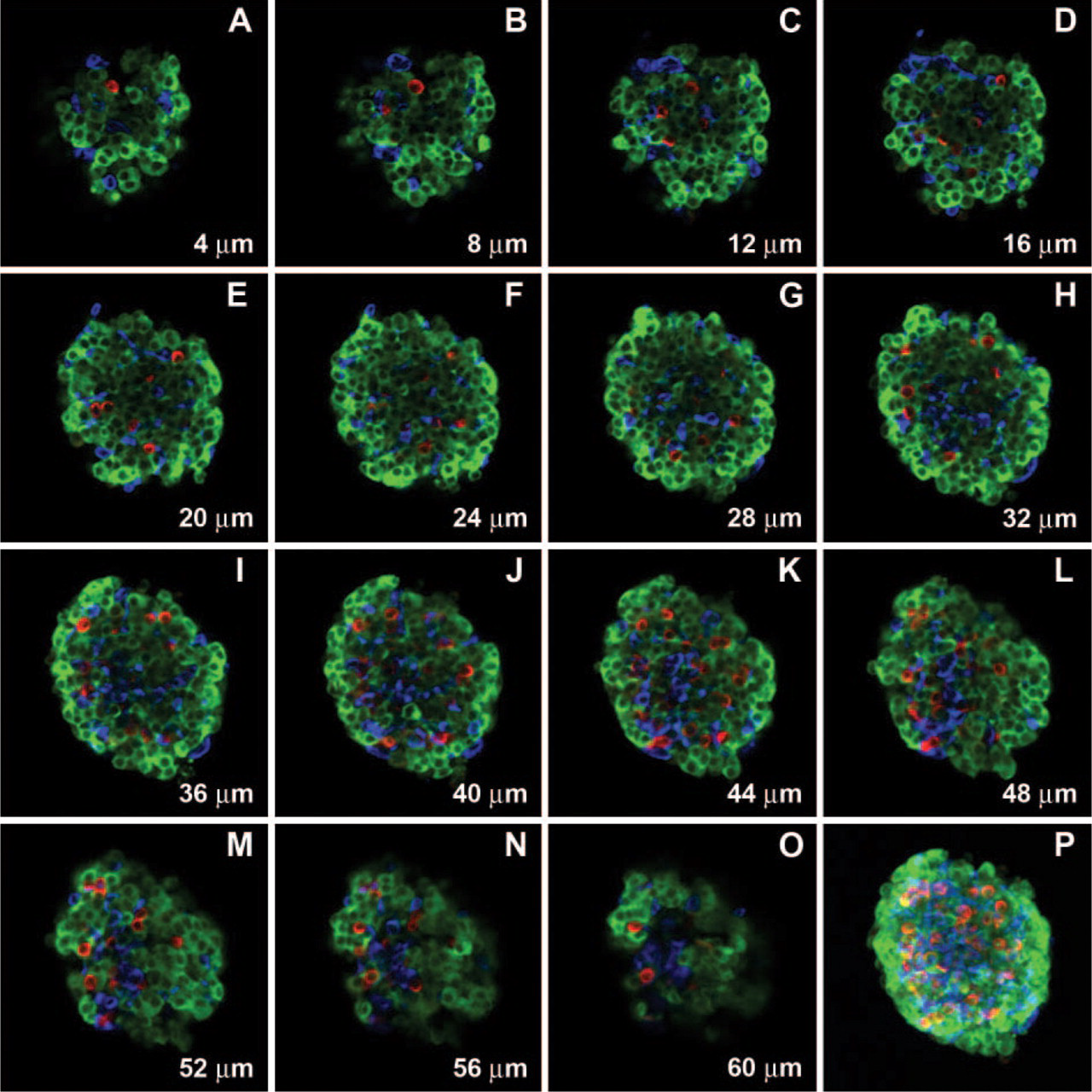
A representative series of optical sections through an isolated mouse islet is shown in Figure 2. Figures 2A-2O represent every fifth optical section of the stack, whereas Figure 2P shows 0° 3-D projection with respect to the y-axis of the whole stack. As demonstrated by individual optical sections in Figures 2A-2O, the core of the mouse islets was almost exclusively composed of β cells, whereas α and δ cells were localized at the islet periphery. A very similar islet cell distribution pattern was seen in the histological sections of the mouse pancreas (Figure 3) with β cells in the islet core and non-β cells forming the mantel. Figures 4A-4O show a representative series of optical sections through an isolated human islet. Unlike in murine islets, α and δ cells were dispersed throughout the islet rather than being confined to the islet periphery (Figures 4A-4O). It is noteworthy that in many human islets the outermost cells were β cells, whereas α and δ cells were localized throughout the islet interior. The architecture of isolated non-human primate islets was very similar to that of human islets (Figure 5). The dispersed distribution of α and δ cells demonstrated by optical sections of isolated human islets was present in all islet preparations and in islets of various sizes. Non-β cells were also seen dispersed throughout the islets in histological sections of human pancreas (Figure 6) where acinar cells still surrounded islets, thus ruling out the possibility that the dispersed cell distribution in isolated human islets resulted from pancreas digestion process. In addition, it appeared from both optical and histological sections that the number of α cells might be greater in human islets in comparison to mouse islets.
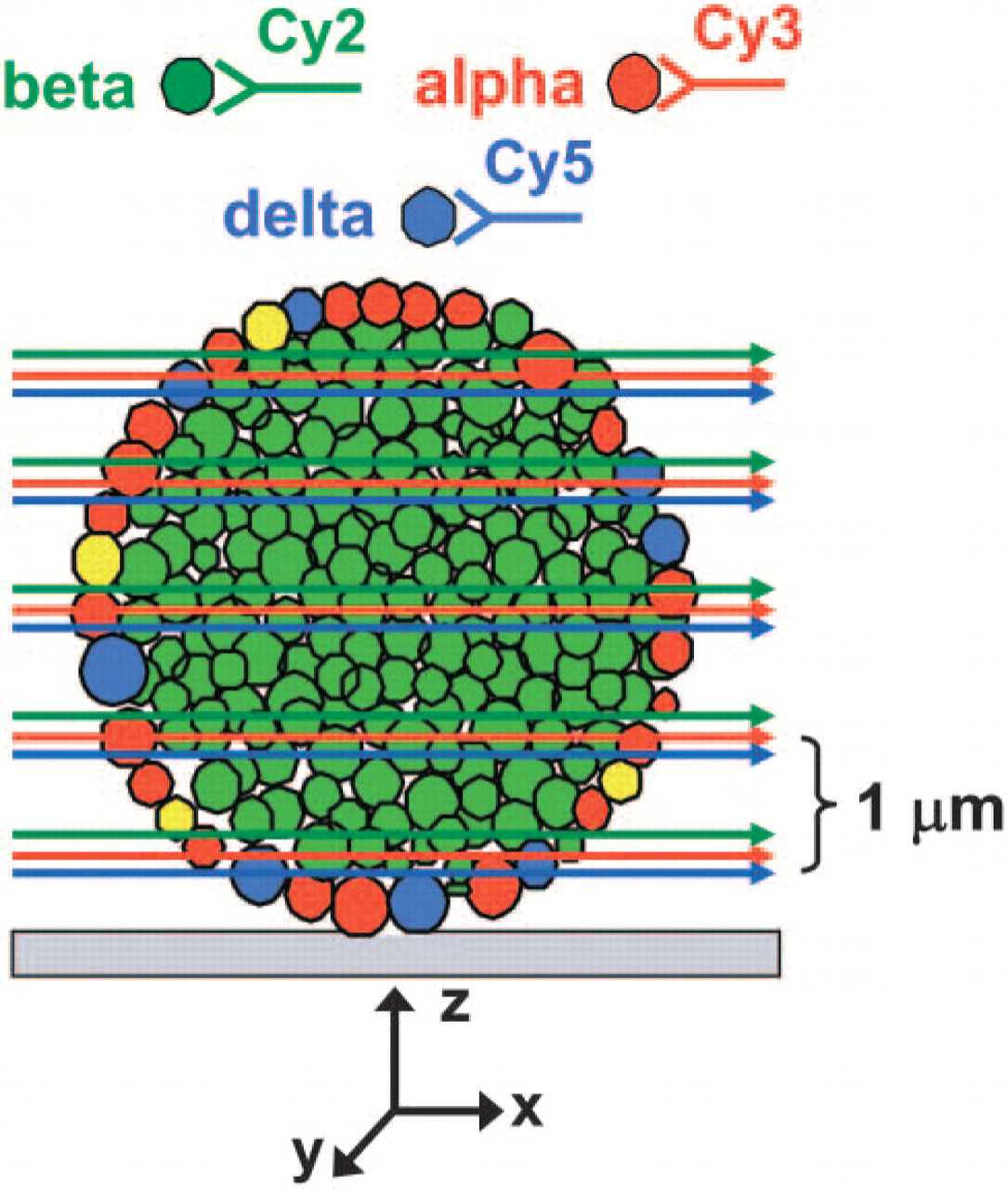
Schematic representation of optical sectioning of isolated islets by confocal laser scanning microscopy. Islet cell types are illustrated in four different colors: β cells, green; α cells, red; δ cells, blue; PP cells, yellow. Antibodies applied to islet hormones for islet cell labeling are shown schematically at the top. Red, green, and blue arrows represent image overlay of α, β, and δ cells in a single focal plane (optical slice). x,y,z refer to axis. Optical slices through islet were acquired by moving focal plane (x,y) along z-axis from the bottom to the top of the islet at 1-μm increments. Using image analysis software, individual optical sections were assembled into a three-dimensional (3-D) stack and projected as a 0° view with respect to the y-axis (head-on projection).
Endocrine Cell Populations in Isolated Mouse and Human Islets
To address the question whether the cell composition of isolated human islets differed from that of murine islets, we subjected their optical sections to image analysis by MetaMorph v6.1 software (Universal Imaging Corporation). In sharp contrast to murine islets, the β-cell population varied considerably in human islets (ranging from 28% to 75%), as did the population of glucagon- (ranging from 10% to 65%) and somatostatin-producing cells (ranging from 1.2% to 22%) (Figure 7). An example of the differences in cell composition is shown in Figures 7C and 7D. The isolated human islets contained 53.9 ± 2.5% β cells, 34.4 ± 2.5% α cells, and 10.4 ± 0.9% δ cells, which was statistically different from the composition of mouse islets where β cells represented 75.4 ± 1.2%, α cells were 18.7 ± 0.9%, and δ cells were 5.9 ± 0.5% (Figure 7). The results with murine islets are quite similar to the morphometric analysis on histological sections of mouse islets in pancreatic sections, in which we reported that mouse islets were composed of 80% β cells, 14% α cells, and 6% δ cells (Brissova et al. 2002). Butler and colleagues (2003) applied a similar approach to the analysis of histological sections from 17 normal human subjects. These investigators found that the relative β-cell volume/islet in human islets was 52.0 ± 4.1%. The data suggest that the composition of isolated islets determined from 3-D reconstructed optical sections on the basis of islet cell volume correlates well with that derived from morphometric analysis of histological sections and confirms the utility of this approach.
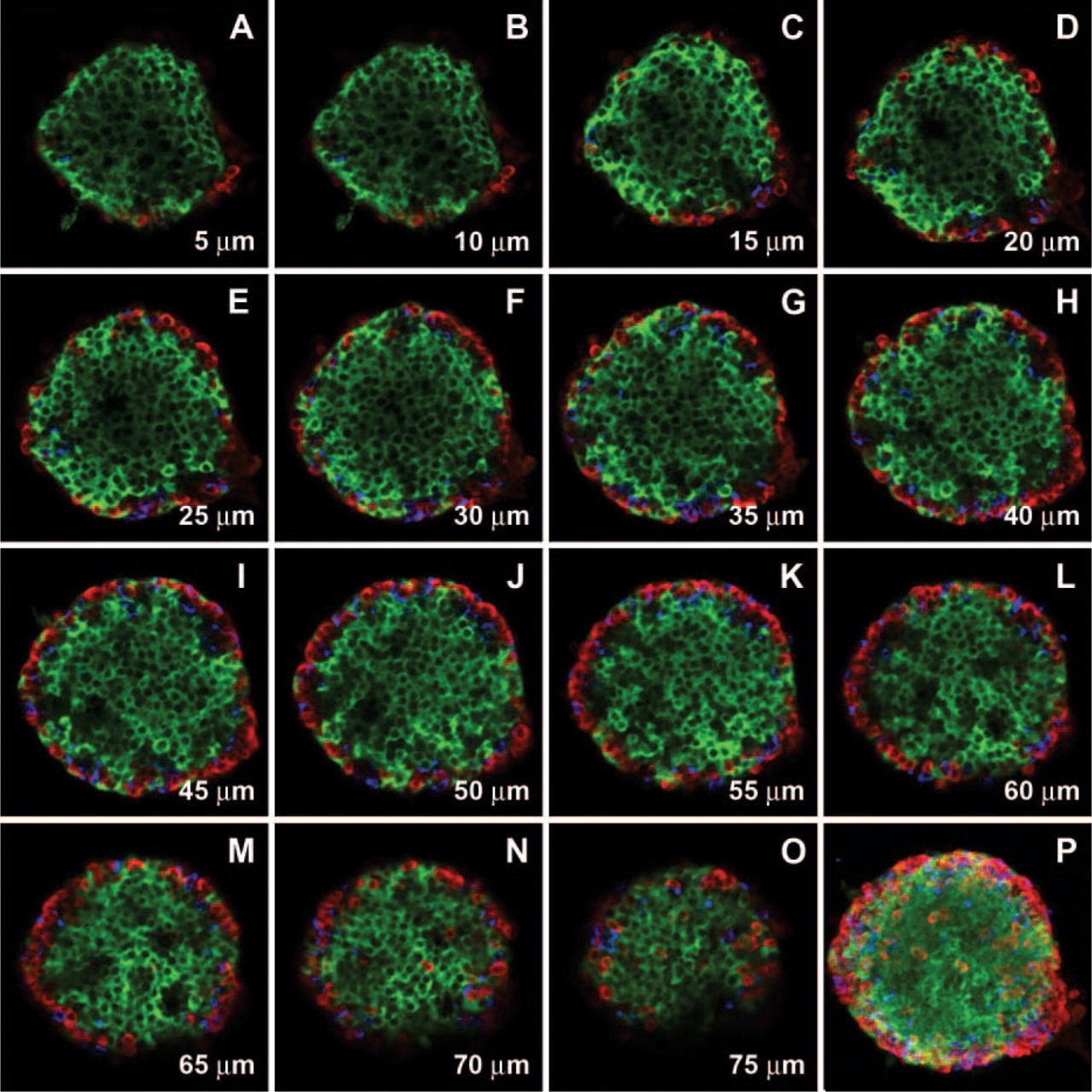
Mouse islet architecture. Series of optical sections through the entire isolated mouse islet was acquired at 1-μm intervals in the axial (z) dimension. (
Histology of mouse pancreas. Histological sections (
Viability and Function of Human Islet Preparations
Each human islet preparation was subjected to quality assessment in a cell perifusion system. In all six preparations, insulin secretion in response to 16.8 mM glucose rose from 1 ng/ml of insulin (baseline) to 3–10 ng/ml. The representative insulin secretory profile of one human islet preparation is shown in Figure 8. These data indicate that isolated human islets used in our studies were viable and functional.
Discussion
Using laser scanning confocal microscopy, this study found that human islets and mouse islets differ considerably in architecture and composition. Histological sections from mouse and rat islets demonstrate a characteristic architecture where the core of the islets consists of insulin-producing β cells, whereas the other three islet cell types, α, δ, and PP cells, are localized around the islet perimeter (Beatens et al. 1979; Cirulli et al. 1994; Bennett et al. 1996; Esni et al. 1999; Kulkarni et al. 1999; Xu et al. 1999; Gannon et al. 2000; Brissova et al. 2002). In several murine studies, deviation from this stereotypical architecture is associated with defects in islet morphogenesis. For example, overexpression of HNF-6 in islets causes a phenotype where non-β cells no longer reside at the islet periphery but become dispersed throughout the islet core (Gannon et al. 2000). There is some evidence that regulated expression of cell adhesion molecules, such as CAM and cadherin families, is implicated in budding of endocrine cells from the pancreatic ducts and correct sorting of endocrine cell types within the islets during the cluster formation (Rouiller et al. 1990; Cirulli et al. 1994; Dahl et al. 1996; Esni et al. 1999). Rouiller and colleagues (1990) showed that N-CAM levels are ≃2-fold higher in murine non-β cells than β cells, and heterozygote and null mutant mice for NCAM have α cells infiltrating into the islet core (Esni et al. 1999). By serial optical sectioning through the entire mouse islets, we were able to conclusively demonstrate that normal adult murine islets have a β-cell core and non-β-cell mantle; this architecture confirms observations with discrete histological sections of the mouse pancreata.
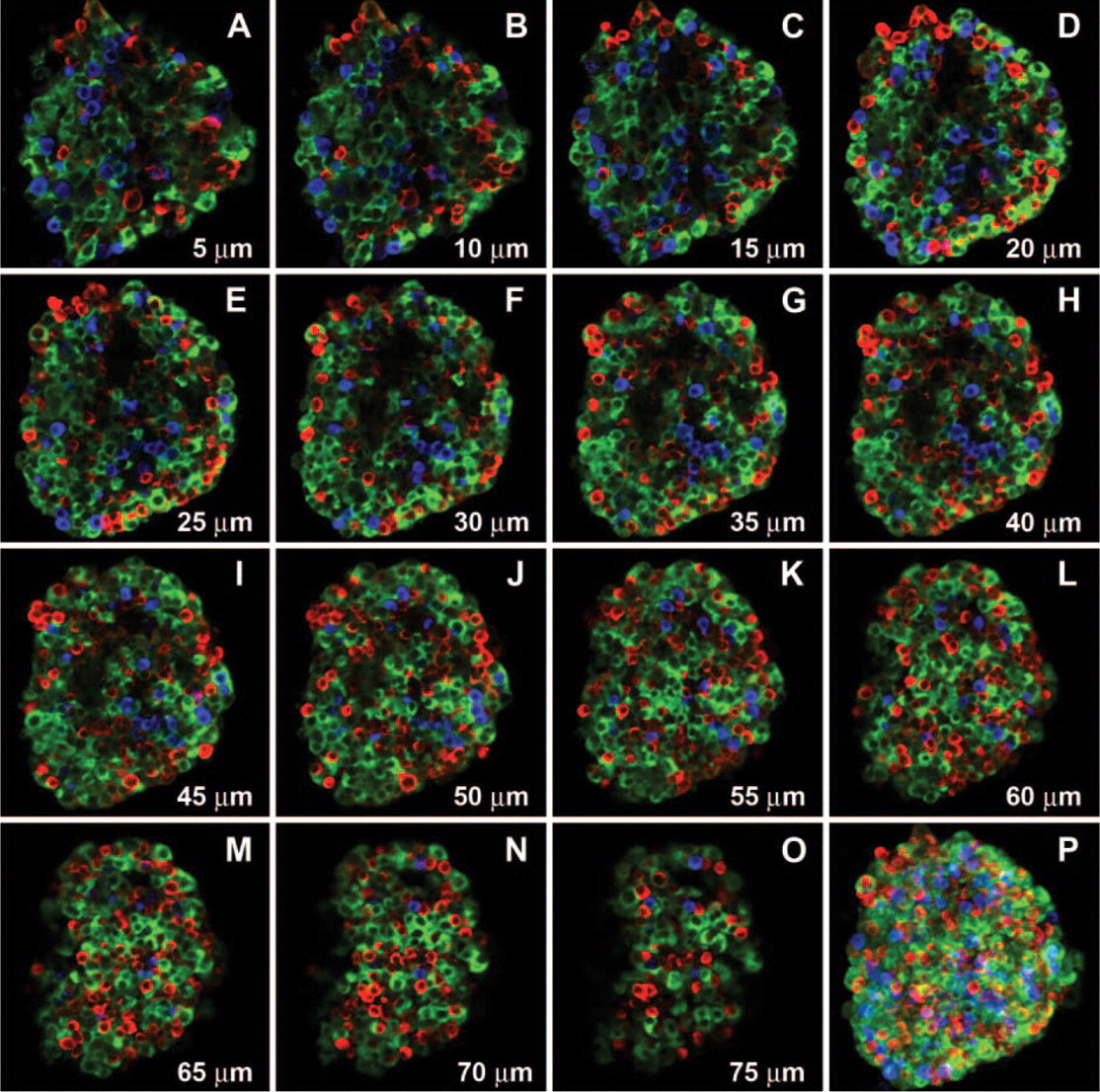
Human islet architecture. Series of optical sections through the entire isolated human islet was acquired at 1-μm intervals in the axial (z) dimension.
Histological sections of the human pancreas, however, have a different appearance than those of the mouse or rat. Micrographs of human islets published by Orci and colleagues (1976), who at that time studied changes in islet morphology associated with type 1 diabetes, suggest that α and δ cells in the adult human islets are dispersed throughout the islet rather than being confined to its periphery. More recent data from the study by Dubois and colleagues (2000), who were interested in PPARγ expression in normal human pancreatic islets, also suggest that α and δ cells in adult human islets are arranged in a less stereotypical fashion than in mouse islets. Although histological sections of the human pancreas suggest a distinct architecture for human islets compared with rodent islets, without a serial sectioning through an entire islet, one cannot be certain whether the sections were collected from the mantle (more intermingling of β and non-β cells) or a deeper portion of the islet comprising the core. Using confocal laser scanning microscopy, Brelje and colleagues (1989) previously described the presence of α, δ, and PP cells deeper within the isolated human islets. However, their study was limited by the depth to which they were able to section into the specimens. By acquisition of serial optical sections through the entire isolated human islets, we were able to demonstrate that human islets lack the typical core–mantle architecture of mouse/rat islets and that β cells are rather intermingled with α and δ cells. Furthermore, β cells are commonly on the surface of isolated human islets. This observation was consistent across six different human islet preparations (from three different human islet isolation centers) and islets of various sizes. Interestingly, non-human primate islets displayed architecture similar to human islets.
Non-human primate islet architecture. Series of optical sections through entire isolated non-human primate islet was acquired at 1-μm intervals in the axial (z) dimension.
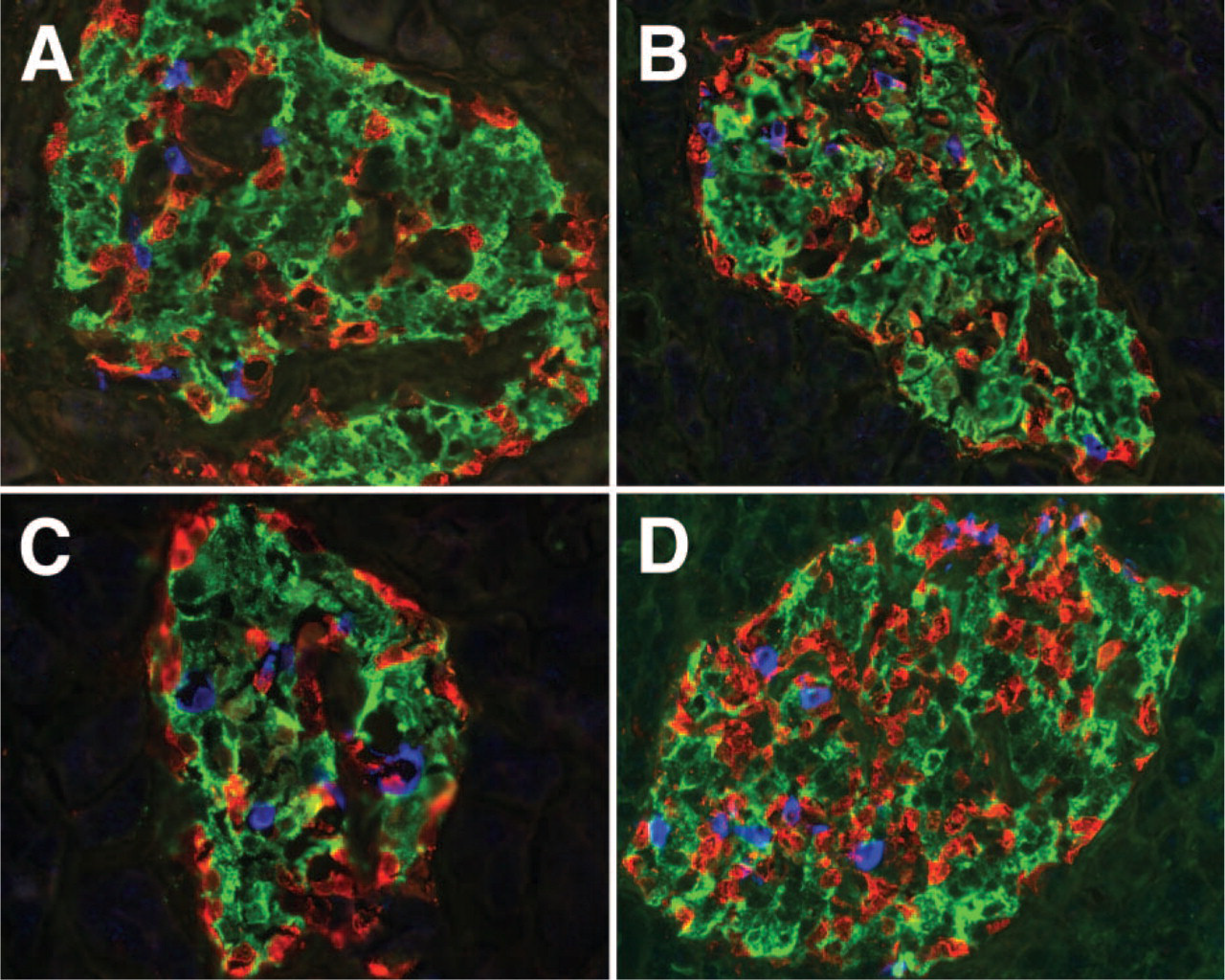
Histology of human pancreas. Histological sections (8 μm) of human pancreas were stained for islet hormones. Micrographs of four different human islets show the intermingling of β and non-β cells.
Our data indicate that human islets not only have an architecture distinct from that of rodent islets, but that endocrine cell populations are also quite different in human islets. In the human pancreas, endocrine cell populations were previously examined with respect to their location in the gland (Orci et al. 1976; Gersell et al. 1979; Malaisse-Lagae et al. 1979), but it is not known what populations of endocrine cell types are present in individual islets. For example, Malaisse-Lagae and colleagues (1979), who identified a PP-rich lobe in the adult human pancreas, noticed a rather significant variation in β- and α-cell populations between PP-rich and PP-poor pancreatic regions. An independent study by Gersell and colleagues (1979) confirmed partition of the human pancreas into PP-rich and PP-poor pancreatic regions by radioimmunoassay and in addition found that a similar phenomenon exists in the canine pancreas. In our studies we selected islets isolated from the total pancreas; therefore, we cannot ascertain the original islet location. However, because we examined a number of islets randomly selected from different islet preparations isolated at three different isolation centers, it seems unlikely that our islets reflect an ascertainment bias. It is highly unlikely that out of an estimated 6,000,000 human islets we would have randomly picked 32 islets that were significantly architecturally and compositionally different from 28 randomly picked mouse islets (three different isolations, 200 islets/isolation). Furthermore, the architecture of intact islet correlated with histological sections of human pancreas. Thus, we feel that our results are representative of islets isolated for human islet transplantation; this concept is supported by the observation that similar islet cell populations were seen in dispersed islets used for human islet transplantation (Shapiro et al. 2000). In addition, from work by Orci and colleagues, it appears that endocrine cell populations in the human pancreas may change throughout life (Malaisse-Lagae et al. 1979; Orci et al. 1979). In infant pancreas, they reported a higher abundance of δ cells, which happens at the expense of PP cells in the PP-rich region and β cells in the PP-poor region. This has not been examined in detail in the pancreas of other species.
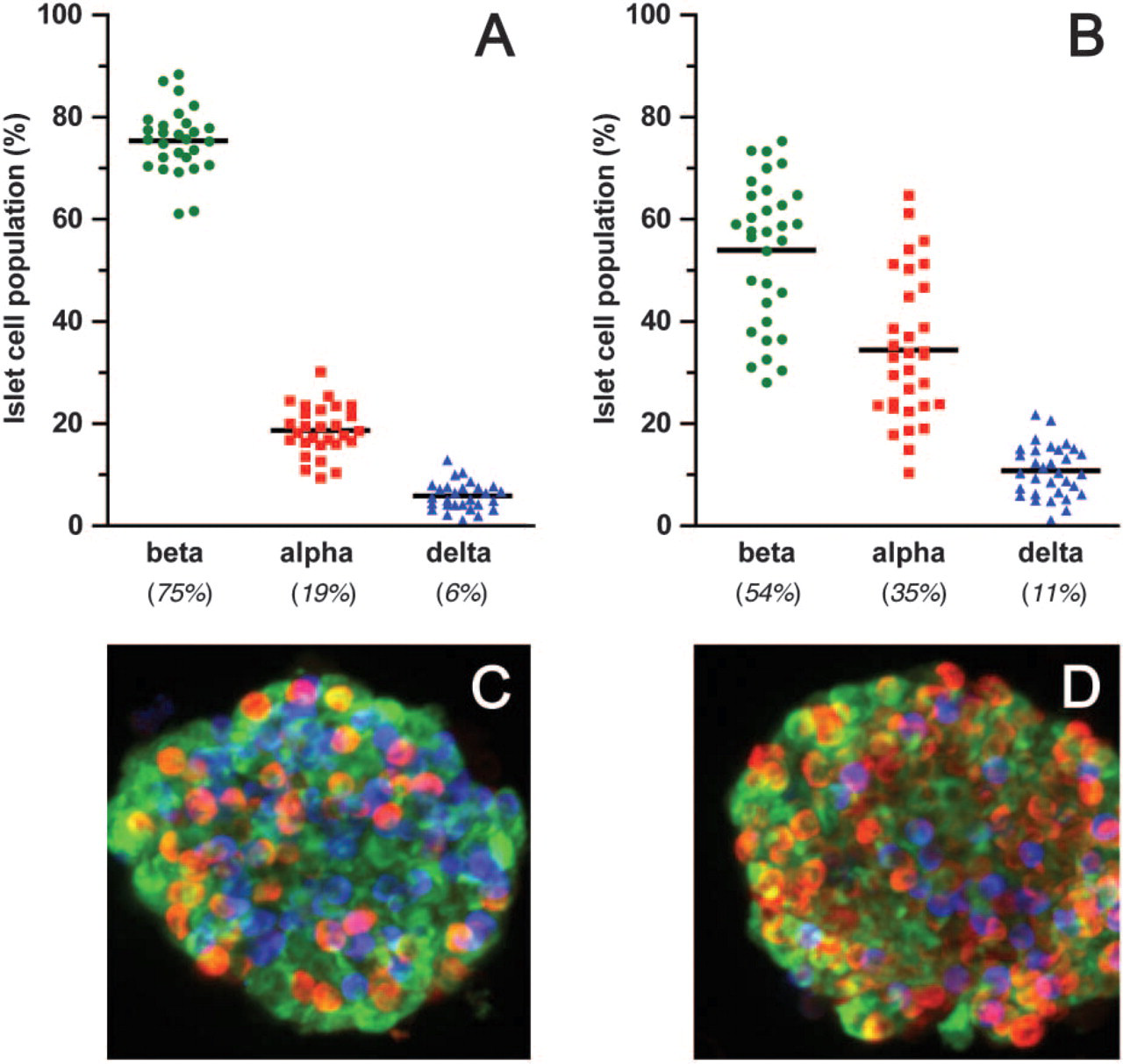
Endocrine cell populations in mouse and human islets. Optical sections of entire mouse (n=28) and human (n=32) islets were 3-D reconstructed and analyzed for the cellular composition using MetaMorph v6.1 software (Universal Imaging Corporation).
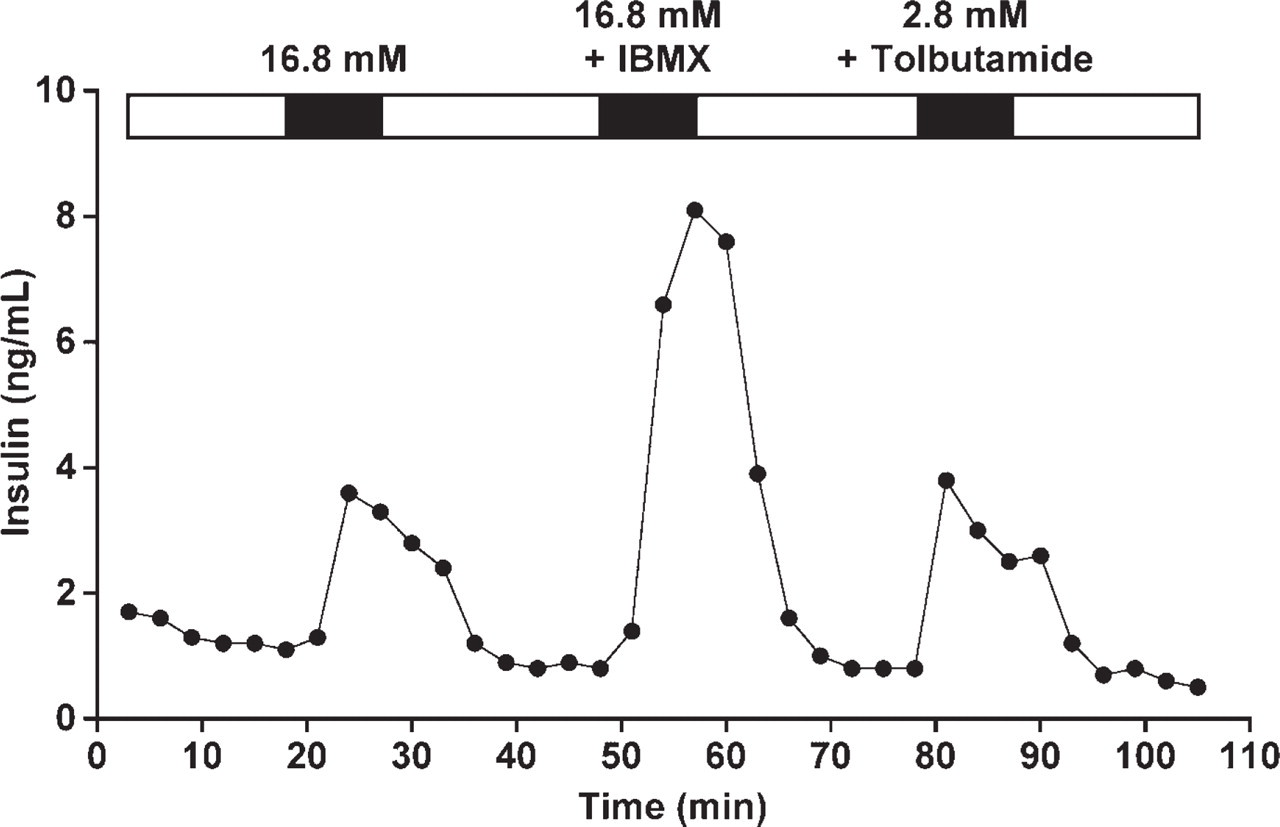
Insulin secretion by isolated human islets. Insulin secretion by human islets was measured in a cell perifusion system in response to the secretagogues shown at the top of the graph (indicated by black bars). White bars indicate basal glucose periods (2.8 mM) between the stimuli.
Similar heterogeneity of endocrine cell types was observed when Redecker and colleagues (1992) studied the microanatomy of canine islets and used semithin sections (up to 1000 sections/islet) to reconstruct the islets. A large number of serial histological sections (up to 77 sections/islet) were also used for the reconstruction of individual rat islets and the examination of their endocrine cell populations (Beatens et al. 1979). These investigators found that PP-rich islets were located in the ventral pancreas, whereas islets in the dorsal pancreas had a lower abundance of PP cells. However, the population of β cells in the PP-rich and PP-poor rat islets was remarkably similar (82.5 vs 82.0%), and only the α-cell population varied. The homogeneous β-cell distribution in the rat islets contrasts with the fluctuation of β and α cells that we found within individual human islets and that was previously described in PP-rich and PP-poor regions of the human pancreas. The murine islets examined in our study were very similar to the observations with rat islets (Beatens et al. 1979), not only by their architecture but also in terms of uniformity in the cellular composition. Thus, these results suggest that islet composition in higher mammals (human, non-human primate, and canine) is more diverse than in rodent islets.
Our findings have several theoretical and practical implications: (a) factors that control human islet development and structure may differ from those in the mouse and thus we encourage caution in extrapolating findings in rodent models directly to higher species including man; (b) beta cells in isolated human islets are often on the islet surface and thus may be more accessible than rodent beta cells (for example, for gene transfer approaches); and (c) human islet heterogeneity may impact assessments of islet quality for transplantation and determinations of insulin secretory capacity.
Footnotes
Acknowledgements
This study was supported by a Merit Review Award from the VA Research Service, research grants from the National Institutes of Health (DK-55233, DK-63439, and DK-62641) and the Juvenile Diabetes Research Foundation International, and the Vanderbilt Diabetes Research and Training Center (NIH DK20593). The Human Islet Distribution Program of the Juvenile Diabetes Research Foundation was crucial for obtaining some of the human islets used in these studies.
We thank Drs. Camillo Ricordi [The Diabetes Research Institute Islet Cell Resource (ICR) at the University of Miami] and Bernhard Hering (Diabetes Institute for Immunology and Transplantation at the University of Minnesota) for assistance in providing human islets for study.
