Abstract
During early postnatal development, the myosin heavy chain (MyHC) expression pattern in equine gluteus medius muscle shows adaptation to movement and load, resulting in a decrease in the number of fast MyHC fibers and an increase in the number of slow MyHC fibers. In the present study we correlated the expression of MyHC isoforms to the expression of sarcoplasmic(endo)reticulum Ca2+-ATPase 1 and 2a (SERCA), phospholamban (PLB), calcineurin A (CnA), and calcineurin B (CnB). Gluteus medius muscle biopsies were taken at 0, 2, 4, and 48 weeks and analyzed using immunofluorescence. Both SERCA isoforms and PLB were expressed in almost all fiber types at birth. From 4 weeks of age onward, SERCA1 was exclusively expressed in fast MyHC fibers and SERCA2a and PLB in slow MyHC fibers. At all time points, CnA and CnB proteins were expressed at a basal level in all fibers, but with a higher expression level in MyHC type 1 fibers. From 4 weeks onward, expression of only CnA was also higher in MyHC type 2a and 2ad fibers. We propose a double function of calcineurin in calcium homeostasis and maintenance of slow MyHC fiber type identity. Although equine muscle is already functional at birth, expression patterns of the monitored proteins still show adaptation, depending on the MyHC fiber type.
M
In previous reports we chose the gluteus medius muscle of the horse because of its important role in locomotion to investigate postnatal changes in MyHC protein expression (Linnane et al. 1999). Major changes in fiber type composition were seen in the first 5 months after birth: a decrease in the amount of fast fibers in parallel with an increase in the amount of slow fibers (Dingboom et al. 1999,2002).
The major Ca2+-sequestering protein important for relaxation, SERCA, exists in adult skeletal muscle as two isoforms, the fast SERCA1 and the slow SERCA2a isoform (Hamalainen and Pette 1997). In adult skeletal muscles of species examined, SERCA1 is expressed in fast MyHC type 2 fibers, and SERCA2a is expressed in slow MyHC type 1 fibers. Phospholamban (PLB), the regulator of SERCA2a activity, is only expressed in adult MyHC type 1 fibers (Hamalainen and Pette 1997; Quiroz-Rothe and Rivero 2001,2004). In small mammals, SERCA2a is the predominant isoform expressed in late fetal and early neonatal skeletal muscle, but this isoform is gradually replaced by the fast-twitch isoform, SERCA1, and is lost in fast muscles as the skeletal muscle differentiates into a fast-twitch state (Brandl et al. 1987; Arai et al. 1992). In larger mammals, early postnatal spatial expression of SERCA isoforms and PLB has not been studied.
Phenotypic differentiation leading to the mature MyHC fiber identity is assumed to be partly influenced by innervation (Gunning and Hardeman 1991). In the first months after birth, mammalian muscle fibers undergo changes in innervation and MyHC expression, while adapting to the functional demands of movement and load (Essen et al. 1980; Lu et al. 1999; Allen and Leinwand 2001; Dingboom et al. 2002; Yamano et al. 2005).
The calcium-activated protein phosphatase calcineurin (Cn) has been proposed as an important regulator of muscle fiber type to translate activity at the neuromuscular junction into a specific transcriptional response driving fiber type remodeling (Oh et al. 2005). Cn activity has been shown to selectively upregulate slow fiber-specific gene promoters, and inhibition of Cn leads to a slow-to-fast fiber transformation (Berchtold et al. 2000). Most studies so far examined the amount of Cn proteins in muscle homogenates. Intriguingly, the amount of total Cn was higher in muscles containing more fast MyHC than slow MyHC (Mitchell et al. 2002). On the other hand, several studies showed that Cn activity is higher in slower muscles (Mitchell et al. 2002; Parsons et al. 2003). Cn is a heterodimeric protein phosphatase composed of a catalytic subunit A (CnA) and a regulatory subunit B (CnB). Mitchell et al. (2002) examined the amount of each subunit and showed a higher amount of CnA protein in fast muscles; in contrast, the amount of CnB protein was lower in fast muscles. Because the activity of Cn was higher in slow muscles, a complex mechanism involving regulation of expression of Cn subunits with a leading role of the amount of CnB was suggested (Mitchell et al. 2002).
Monitoring the expression levels of Cn in different MyHC muscle fiber types using serial sections provides clues about the fiber type-specific expression of these proteins (Torgan and Daniels 2006). Expression patterns of the Cn subunits per MyHC fiber type, or during adaptation as takes place in early postnatal development, are not known.
In this study we monitored expression patterns of MyHC isoforms together with expression of SERCA1/2a, PLB, CnA, and CnB on a fiber-to-fiber basis during early postnatal growth. Because the muscle is already functional at birth, our hypothesis is that the expression of SERCA isoforms and PLB would at all ages be identical to the adult expression pattern. Given the role in regulating slow fiber-type expression, we speculate that at birth the amount of Cn subunits will already be higher in slow MyHC type 1 fibers as compared with fast MyHC type 2 fibers. We also expect higher expression levels of Cn subunits in fibers changing from MyHC type 2a to type 1.
Materials and Methods
All chemicals were obtained from Merck (Amsterdam, NL) unless otherwise indicated.
Foals
The two Dutch Warmblood foals analyzed in this study remained with the mares in a paddock for 1 week after birth, after which they were kept at pasture with the mare 24 hr/day. At 22 weeks of age they were kept in a loose house with access to a small paddock. Foal #27 was a female; foal #33 was a male (weight at birth: #27, 72 kg; #33, 55 kg; at 22 weeks: #27, 288 kg; #33, 241 kg; at 48 weeks: #27, 401 kg; #33, 360 kg). Both were part of a larger project (Exercise Osteochondrosis) (van Weeren and Barneveld 1999). The study was approved by the Committee on Animal Welfare of the Faculty of Veterinary Medicine, Utrecht University, The Netherlands.
Muscle Biopsies
From each foal (#27 and #33), percutaneous muscle biopsies were taken by the same person, at 4 days (designated 27-0 and 33-0), 2 weeks (27-2, 33-2), and 4 weeks (27-4, 33-4 was lost) after birth and at the age of 11 months (27-48, 33-48) (48 weeks) (Dingboom et al. 1999). Biopsies were taken from the gluteus medius muscle on an imaginary line drawn from the coxal tuber to the sacral tuber, at one third distance from the sacral tuber, perpendicular to the skin. They were taken as deep as possible (until resistance from the iliac wing) and were taken from the same location in the muscle to ensure comparable fiber type composition (Serrano et al. 1996; Linnane et al. 1999). Samples were rolled in talcum powder, mounted on cork blocks with the use of optimal cutting temperature embedding medium, and oriented in such a way that the fibers could be sectioned transversely. All samples were stored at −80C.
Immunofluorescence Staining
Monoclonal MyHC antibodies (MAbs) used were previously shown to cross-react with horse myosin (Dingboom et al. 2002; Eizema et al. 2003). The following primary antibodies and dilutions were used: MAb slow and fast (dilution 1:2000; Sigma, St Louis, MO) recognizes the slow (type 1) and fast (type 2) isoforms, respectively. MAb A4.74 (dilution 1:50; Alexis, Kordia, Leiden, NL) recognizes MyHC type IIa (Rivero et al. 1996). MAb 412 (dilution 1:25; kind gift from Prof. A.F.M. Moorman, Amsterdam University, Amsterdam, The Netherlands) reacts with MyHC type 1 and 2d (Dingboom et al. 1999). The polyclonal antibody PP2b-a (H209, dilution 1:100; Santa Cruz Biotechnology, Santa Cruz, CA) was used to detect CnA (Bigard et al. 2000), the polyclonal PA3-025 (10Ps, dilution 1:100; Affinity BioReagents, Breda, NL) was used to detect CnB (Mitchell et al. 2002). SERCA1, SERCA2, and PLB were detected with Ab2818, Ab2817 (dilution 1:500; Abcam, Cambridge, UK), and PLB #05-205 (dilution 1:100; Upstate, Lake Placid, NY) (Quiroz-Rothe and Rivero 2001). SERCA1 antibody recognizes both the adult 1a and the neonatal 1b isoform. SERCA2 antibody recognizes both the muscle-specific 2a as well as the generally expressed 2b isoform.
Ten-μm transverse serial sections were obtained as described previously (Eizema et al. 2003). Double-labeling experiments were performed using the slow and CnA-detecting antibodies or the fast and CnB-detecting antibodies. The other antibodies were incubated on consecutive sections. The protocol was followed as described previously (Dingboom et al. 2002) with modifications enabling fluorescent detection. Slides were rinsed in PBS, pH 7.4, blocked in Teng-T (10 mM Tris, 5 mM EDTA, 0.15 M NaCl, 0.25% gelatin, and 0.05% Tween 20, pH 8.0) for 15 min, followed by rinsing in PBS and incubation overnight at room temperature (412, A4.74, SERCA2, PLB, SERCA1) or 37C (slow in combination with CnA and fast in combination with CnB). Sections were rinsed in PBS and incubated with secondary antibody goat anti-mouse, Alexa Fluor 568 (dilution 1:200; Molecular Probes, Invitrogen, Breda, NL) for 45 min in the dark. For detection of the Cn subunit expression, incubation was performed with secondary antibody goat anti-rabbit, Alexa Fluor 488 (Molecular Probes, Invitrogen), incubated together with the secondary antibody for the detection of the MyHC. Finally, sections were rinsed in large volumes of PBS, mounted in Fluorsave Reagent (Calbiochem, EMD Biosciences; Darmstadt, Germany), and left to dry at 37C (dark). Staining of the fiber perimeter was performed with a wheat glutamin antibody directly coupled to Alexa Fluor 350 (WGA350, dilution 1:500; Molecular Probes, Invitrogen). Incubation was performed together with the secondary antibody. No cross-reactivity was observed between any of the antibodies used. Omitting the primary or secondary antibodies produced no background staining (results not shown).
Analyses
Analyses were performed with a Leica DMRE microscope and Leica image software (Leica; Wetzlar, Germany). Negative controls were set on the background settings of the positive controls taken on the microscope. Between 74 and 243 fibers per biopsy were used for fiber typing and calculation of fiber type composition (mean value: 184 fibers). Fibers were classified into type 1, type 2a, type 2ad, and type 2d on the basis of their reactions with the MyHC MAbs. Expression levels were visually judged as high or low as compared with the negative controls. Two researchers have independently judged the expression levels.
Results
We analyzed expression of the Ca2+ handling proteins SERCA1, SERCA2a, and the inhibitor of SERCA2a, PLB, in biopsies from the gluteus medius muscle of young foals. Expression patterns of the Cn subunits CnA and CnB were also analyzed.
Results obtained by immunofluorescence are shown in Figure 1. Figure 1A shows the results obtained in a biopsy taken at 0 weeks of age. Figure 1B shows the results at 48 weeks of age from the same foal (#27). Some fibers are labeled according to the MyHC protein(s) they express. Table 1 presents all the data obtained at the different ages: 0, 2, 4, and 48 weeks. Expression patterns are related to the delineated MyHC fiber type; the amount (%) of each fiber type is also indicated. As previously demonstrated, the gluteus muscle of foals consists of a high percentage of fast 2ad and 2d MyHC fibers at birth, but during the first year of life the muscle becomes slower, indicated by an increase in type 1 and 2a MyHC fiber types (Table 1) (Dingboom et al. 2002). Expression of the Cn proteins was seen at a low level in all fibers; however, as can be seen in Figure 1 some fibers express the Cn proteins at a higher level. The percentage depicted in Table 1 for the Cn proteins indicates only the fibers with a higher level of Cn expression.
Expression Patterns of SERCA1, SERCA2a, and PLB
At birth, SERCA isoforms and PLB expression were not restricted to either MyHC fast type 2 or slow type 1 MyHC fibers (Figure 1; Table 1) as in adult muscle. At birth and at 2 weeks, a low number of slow fibers express SERCA1, whereas a substantial amount of fast fibers express SERCA2a and PLB especially at birth, albeit at a lower level. From 4 weeks of age onward, expression of SERCA1 was restricted to fast MyHC fibers and SERCA2a and PLB to slow fibers (Table 1). Expression patterns of SERCA2a and PLB were almost identical at all time points.
Expression Patterns of CnA and CnB
At all time points, both CnA and CnB proteins show basal expression in all fibers, with higher expression in MyHC type 1 (Figure 1, best visible in the CnB pattern). From 4 weeks onward, expression of CnA was also higher in all MyHC type 2a and 2ad fibers. CnB remained high in only type 1 fibers (Figure 1; Table 1). These expression patterns were maintained until 48 weeks of age (Table 1). Expression of CnB was uniform over the fiber area, and expression of CnA appeared at age 4 weeks and onward, more localized in dots and toward the fiber perimeter (Figure 1).
Discussion
In this paper we compared the expression pattern of the MyHC proteins with the expression pattern of SR Ca2+ handling proteins and the two subunits of Cn during the first year of life in a large mammal. The major findings are that, in contrast to our expectations, at birth until 4 weeks of age the SERCA2a and 1 isoforms are not yet fully coordinated with the expression of slow type 1 and fast type 2 MyHC, respectively. Expression of PLB is as expected almost identical to the expression of SERCA2a.
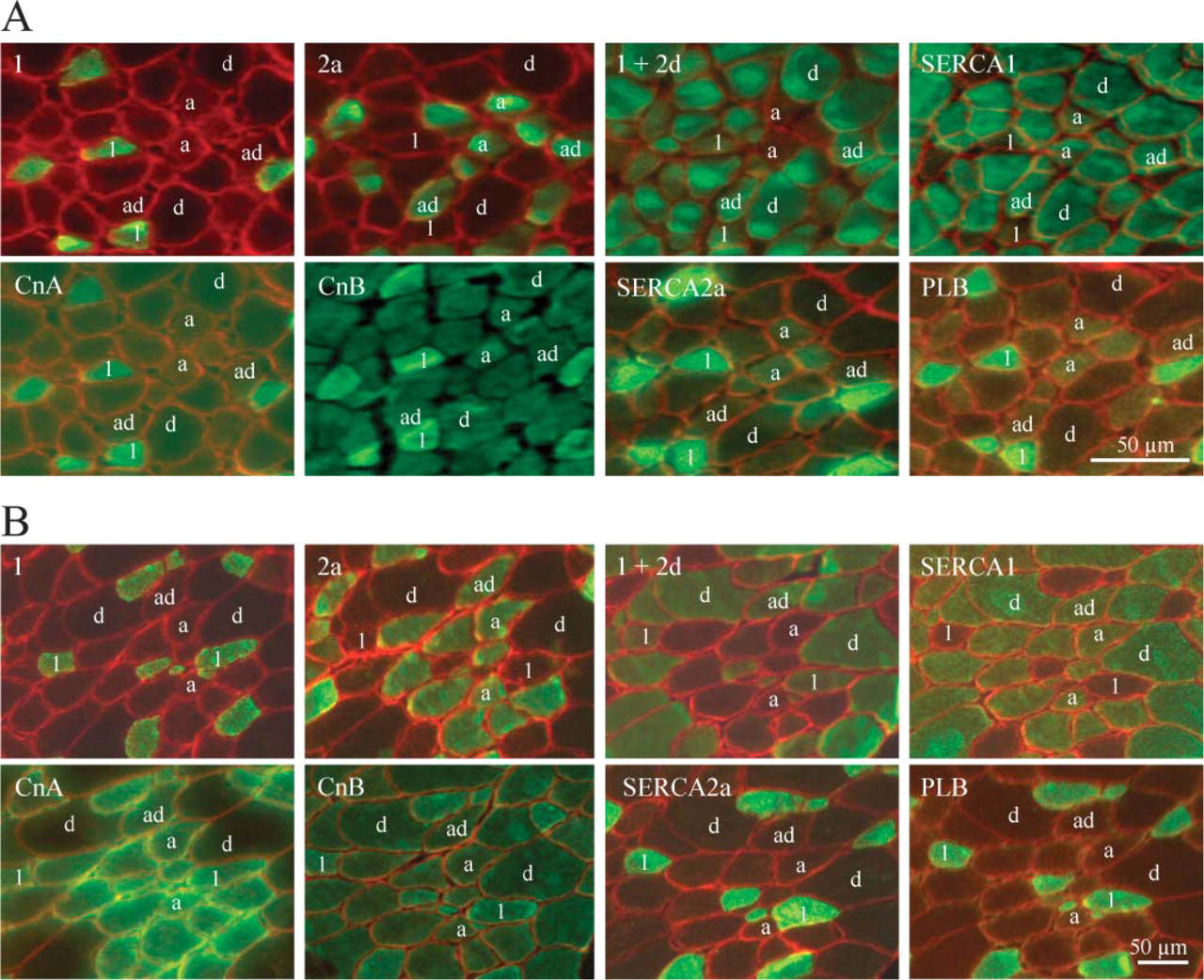
Expression of MyHC isoforms, SERCA1/2a, PLB, CnA, and CnB by immunofluorescence in serial sections of the m. gluteus medius from foal #27 at birth (27-0) (
We hypothesized that the amount of Cn subunits would be higher in slow MyHC type 1 fibers. We show here that the expression level of CnA and CnB is complex and changes within the fiber types during the first 4 weeks of life. From then on, CnA has a higher expression level in MyHC type 1, 2a, and 2ad fibers; in contrast CnB has a higher expression level only in type 1 fibers.
Figure 2 displays a schematic representation of the expression patterns seen at birth (left row) and from 4 weeks onward (adult, right row). Each circle represents a MyHC fiber type; expression level of the Ca2+ handling proteins and of the Cn subunits is indicated. Bold indicates a high expression level; normal indicates a low expression level. As demonstrated in the present study and from our previous studies, we found an increase in the amount of slow type 1 MyHC fibers together with a decrease in the fast type 2 MyHC fibers in these foals during the first 5 months after birth (Dingboom et al. 2002). Consequently, the most likely direction of change for a fiber during this time period is to express a slower type of MyHC (e.g., from 2d toward 2ad, from 2ad to 2a, etc.) according to the ‘nearest-neighbor’ rule for MyHC fiber type transitions (Pette and Staron 2001). Based on these observations and based on the SERCA expression pattern (see below), arrows in Figure 2 indicate the most likely developmental change from birth until 4 weeks of age.
Expression of CnA, CnB, SERCA2, PLB, and SERCA1 in immunohistochemically defined MyHC fibers
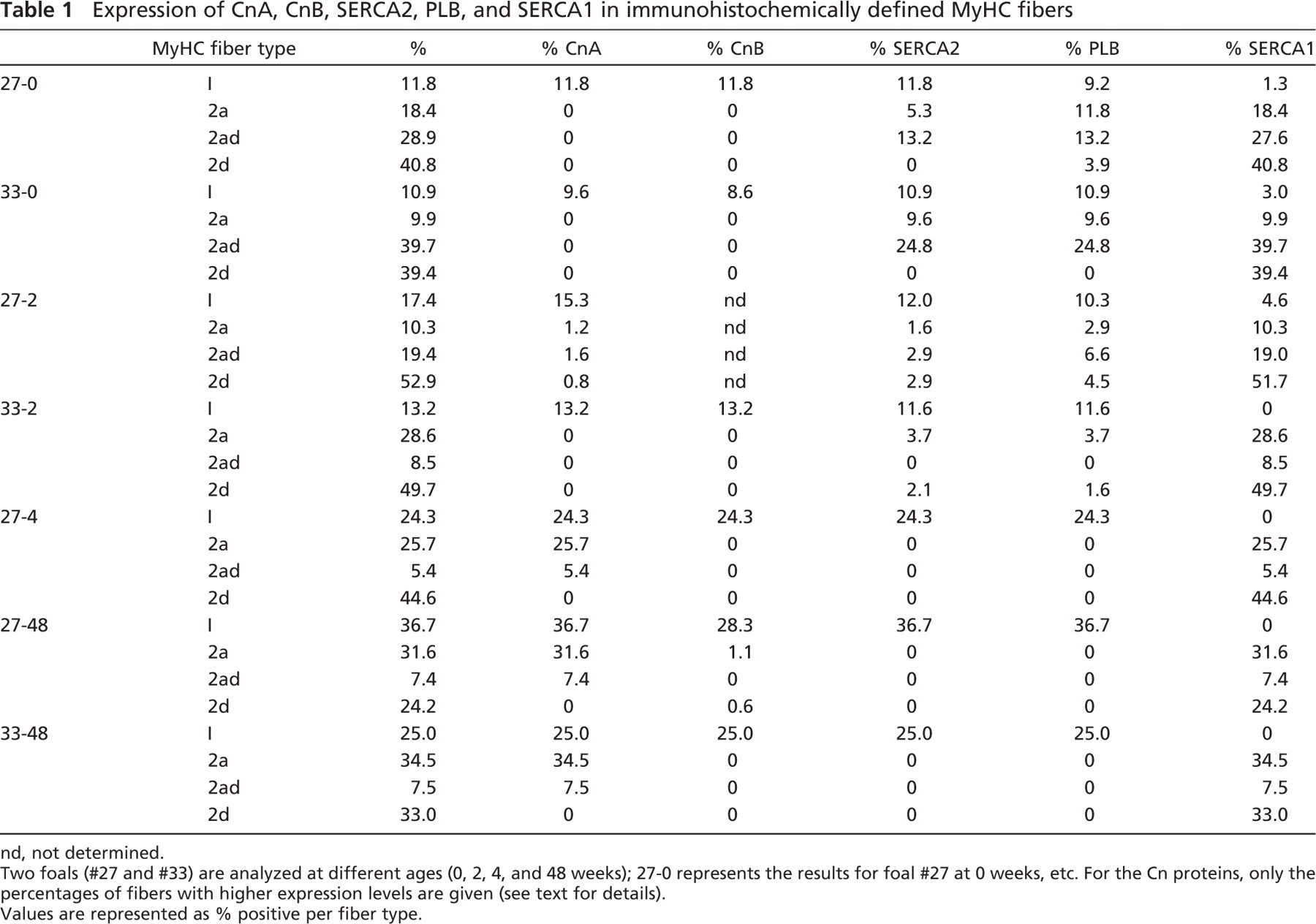
nd, not determined.
Two foals (#27 and #33) are analyzed at different ages (0, 2, 4, and 48 weeks); 27-0 represents the results for foal #27 at 0 weeks, etc. For the Cn proteins, only the percentages of fibers with higher expression levels are given (see text for details).
Values are represented as % positive per fiber type.
Expression Patterns of SERCA1, SERCA2a, and PLB
This study presents data on SERCA isoforms and PLB expression during early postnatal life in a large mammal. At the earliest ages, small differences between the expression of SERCA2a and PLB were seen. In fetal mice and pig, an uncoordinated expression of these two proteins was also observed (Arai et al. 1992; de Jonge et al. 2006), although the mechanism is as yet unknown. In small mammals, a neonatal isoform of the SERCA1 gene, the SERCA1b, is present, disappearing quickly after birth (Brandl et al. 1987; Arai et al. 1992). The antibody we used to detect SERCA1 expression binds to both the adult SERCA1a (referred to here as SERCA1) and the neonatal SERCA1b isoform; therefore, we cannot distinguish between the expressions of these isoforms.
The general process of myogenesis is similar in different species. However, the kinetics of muscle fiber development with respect to MyHC isoform expression appears to be different among species, depending on their maturity at birth (Picard et al. 2002; de Jonge et al. 2006). For small mammals, it has been described that skeletal muscles are highly undifferentiated at birth and then undergo a marked process of differentiation in attaining the adult MyHC phenotype. SERCA2a is the predominant isoform expressed in late fetal and early neonatal skeletal muscle, but this isoform is gradually replaced by the fast-twitch isoform, SERCA1, and is lost in fast muscles as the skeletal muscle differentiates into a fast-twitch state (Brandl et al. 1987; Arai et al. 1992). Fiber type differentiation in large animal species has its onset before birth, resulting in a muscle with all the different fast MyHC present at birth (Picard et al. 2002). We show that SERCA2a has, also in these differentiated fibers, a broader expression pattern in early postnatal stages. Apparently, expression of MyHC and SERCA is not completely coregulated at birth and independent of the maturity of the muscle.
Expression of MyHC and SERCA is normally highly correlated, even in experimental models in which muscle undergoes phenotypic transitions such as chronic low-frequency stimulation (Hamalainen and Pette 1997). In other models the coordination is less clear, such as in spinal cord injury (Talmadge et al. 2002), overload (Awede et al. 1999), regeneration (Zador and Wuytack 2003; Zador et al. 2005), in the early stage of hyperthyroidism (Yamada et al. 2004), or after denervation of muscles of rabbits where a transient dissociation of coordination was seen (Hamalainen and Pette 2001). From these models it is tempting to speculate that absence or diminished innervation can result in an uncoordinated expression of MyHC and SERCA. In young animals the innervation is possibly not completely matured, resulting in a dissociation of expression.
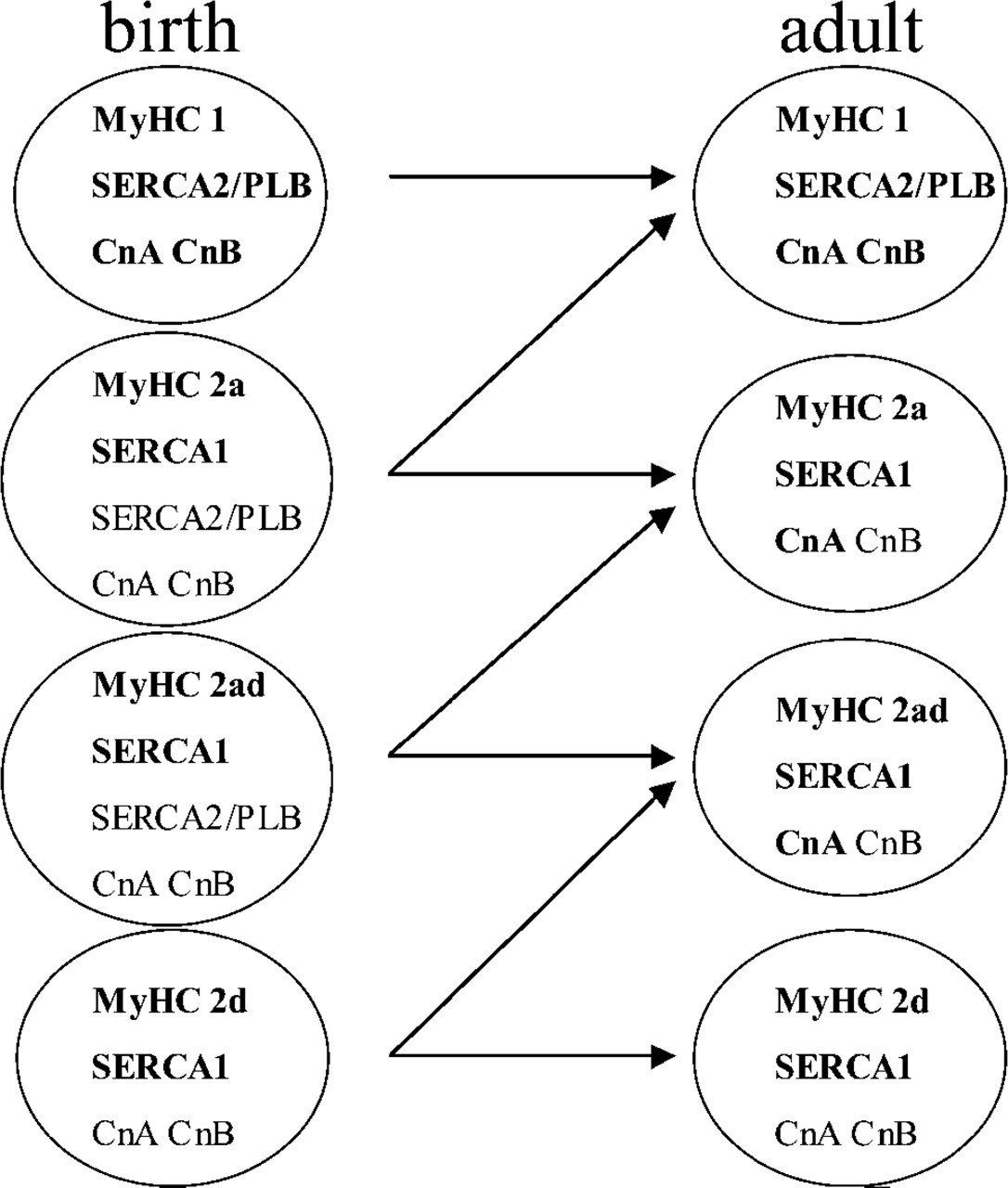
Schematic representation of the expression patterns seen at birth (left row) and from 4 weeks onward (adult, right row). Each circle represents a MyHC fiber type; indicated is the expression level of the Ca2+ handling proteins and of the Cn protein subunits. Bold indicates a high expression level; normal indicates a low expression level. Arrows indicate the most likely developmental change from birth until 4 weeks of age, according to the ‘nearest-neighbor’ rule for MyHC fiber type transitions. See text for further details.
This was also suggested by the observation that the slower muscular development of dystrophic muscle of mdx mice resulted in an increase of the discordance between the MyHC and SERCA expression early in the postnatal period (Divet et al. 2005).
Previously it was shown that the number of type 1 MyHC fibers increases during the first year of life of a foal at the expense of the fast isoforms of MyHC (Dingboom et al. 1999,2002). A change toward a different fiber type is first seen by changes in expression of cytoplasmic, mitochondrial, and sarcoplasmic proteins, e.g., seen by the expression of SERCA2a and PLB in fibers still expressing fast MyHC isoforms (Figure 2). Changes in large contractile proteins present in high amounts and with a long turnover time, such as MyHC isoforms, take more time (Everts et al. 1987; Pette and Staron 2001; Talmadge et al. 2004a). However, as stated above, regulation between the MyHC and SERCA proteins diminishes only in cases of reduced neuromuscular activity, otherwise the expression changes are coordinated.
Expression Patterns of CnA and CnB
In this paper we show for the first time the differential expression of both the CnA and CnB subunits within a muscle. The level of expression is highly fiber type specific. There is some controversy in the literature on Cn function. So far, studies have been performed in vivo and in vitro among others by using cyclosporin A, an inhibitor of Cn, or by studying expression or activity levels of CnA, CnB, or Cn. Different muscles have also been used, as well as different species. Talmadge et al. (2004b) found different roles for Cn in different muscles of the mouse. A comparison of the role of Cn in the same muscles of different species would facilitate the interpretation of the various studies. In this paper we studied Cn expression levels, not activity. The activity might also be highly fiber type specific but is not measurable within a single fiber.
Our findings corroborate with those of Delling et al. (2000), showing colocalization of activated Cn in slow fibers using a different assay. Our results are also in line with Bigard et al. (2000) showing higher expression of CnA in soleus muscle (almost exclusively composed of type 1 MyHC) as compared with the plantaris muscle (a fast muscle) of adult rats. The localization pattern obtained by Bigard et al. (2000) is, however, different from our pattern. These authors showed expression in the periphery of muscle fibers, whereas we show expression across the total fiber area. This discrepancy might be due to different staining protocols of immunohistochemistry vs immunofluorescence or to the different muscles and other species as compared with our study. In mouse muscles it is shown that Cn has specific roles depending on the muscle (Talmadge et al. 2004b). These roles might also depend on the species studied. To gain better insight into differences in Cn function, analyzing expression levels using immunofluorescence in different muscles of different species will be valuable.
Using the myogenic C2C12 cell line, it was demonstrated that Cn influences in vitro the promoter activity of MyHC 1 and 2a genes, the same fiber types in which we observed higher expression levels of CnA (Chin et al. 1998; Swoap et al. 2000; Allen et al. 2001; Allen and Leinwand 2002). Inhibitors of Cn have a reducing effect on the amount of only type 1 MyHC fibers (Berchtold et al. 2000; Serrano et al. 2001; Spangenburg and Booth 2003), in contrast to the proposed role in activating the promoter of MyHC 1 and 2a. We show that CnB is expressed higher only in type 1 MyHC fibers, explaining possibly that the amount of CnB is determining the activity of the Cn complex as suggested previously (Mitchell et al. 2002).
Additionally, we also observed basal expression of CnA and CnB in the other fiber types. This could point to a general role for Cn in maintaining calcium homeostasis in muscle fibers. Whether this role is important in all fibers or only in type 2 MyHC cannot be concluded from our data. A role in the maintenance of fiber gene programs was also suggested from studies with regeneration and transgenics (Serrano et al. 2001; Mitchell et al. 2002; Fenyvesi et al. 2004; Oh et al. 2005; Launay et al. 2006). In denervated regenerating mouse soleus, MyHC1 is reexpressed in spite of denervation, whereas Cn remains low (Launay et al. 2006). Using an elegant transgenic approach, Oh et al. (2005) showed that, in skeletal muscle of young adult mice, the contractile components of MyHC type 1 slow fibers are under the control of Cn. The conclusion of their work was that Cn is not necessary for the development of slow fiber but is necessary for maintenance of slow fibers. The prenatal role for Cn might be maintenance of muscle calcium homeostasis independent of fiber type (Mitchell et al. 2002; de Jonge et al. 2006). It would be very interesting to look at the expression pattern of foals prenatally and to compare these data with perinatal expression in mice or rats, to analyze the role of innervation and functional demand.
We propose that low expression seen in muscle fibers is necessary for maintenance of calcium homeostasis (Figure 2); high expression is necessary for maintenance of fiber type-specific gene programs (Figure 2, bold). As also seen for the SERCA expression, the expression patterns of the Cn subunits changed until the foal was 4 weeks old, again pointing toward a not yet fully matured innervation status of the muscle at birth.
Cn is a key component of the transduction pathway relaying nerve electrical activity and transcriptional regulation of slow muscle fiber-type specificity (Serrano et al. 2001), and its role in activation and maintenance of slow fibers is well established. However, the exact nature of the signaling pathway is not yet clear. Our data show that, within a muscle, substantial differences in expression exist between fiber types, underscoring the importance of spatial specificity (Torgan and Daniels 2006). From our data we propose a double function of Cn, depending on the amount of protein present, with a regulatory role for the amount of CnB.
Interestingly, it was shown in rats that using cain, a Cn inhibitor, or partial tenotomy, expression of MyHC 1 was prevented, but SERCA2a expression was unaffected in fibers of regenerating soleus muscles (Zador et al. 2005). This points to distinct regulation of expression of slow-type contractile proteins and slow type of relaxation proteins. Further studies need to focus on understanding how Cn signals integrate with other signaling pathways to coordinate muscle plasticity in response to physical activity and innervation.
We conclude that although equine muscle is already functional at birth, expression patterns of the monitored proteins still show adaptation, depending on the MyHC fiber type.
Footnotes
Acknowledgements
We thank Dr. A.F.M. Moorman and Dr. J.A.M. Korfage, University of Amsterdam, Amsterdam, The Netherlands for the generous gift of monoclonal antibodies. We are grateful for the skillful technical assistance of C.W. van der Wiel. We thanking. Anko de Graaff and Dr. Richard Wubbolts of the Center for Cell Imaging, Faculty of Veterinary Medicine, Utrecht University, Utrecht, The Netherlands for technical advice.
