Abstract
Protein tyrosine phosphatase-interacting protein 51 (PTPIP51) expression was analyzed in proliferating and differentiating human myogenic cells cultured in vitro. Satellite cell cultures derived from four different individuals were used in this study. To analyze the expression of PTPIP51, myoblasts were cultured under conditions promoting either proliferation or differentiation. In addition, further differentiation of already-differentiated myobtubes was inhibited by resubmitting the cells to conditions promoting proliferation. PTPIP51 protein and mRNA were investigated in samples taken at defined time intervals by immunostaining, immunoblotting, in situ hybridization, and PCR. Image analyses of fluorescence immuno-stainings were used to quantify PTPIP51 in cultured myoblasts and myotubes. Myoblasts grown in the presence of epidermal and fibroblast growth factors (EGF and FGF), both promoting proliferation, expressed PTPIP51 on a basic level. Differentiation to multinuclear myotubes displayed a linear increase in PTPIP51 expression. The rise in PTPIP51 protein was paralleled by an augmented expression of muscle-specific proteins, namely, sarcoplasmic reticulum Ca2+ ATPase and myosin heavy-chain protein, both linked to a progressive state of myotubal differentiation. This differentiation-induced increase in PTPIP51 was partly reversible by resubmission of differentiated myotubes to conditions boosting proliferation. The results clearly point toward a strong association between PTPIP51 expression and differentiation in human muscle cells.
Immunohistochemical screening of mammalian tissues revealed the expression of PTPIP51 antigen in various tissues and organs (Stenzinger et al. 2005). Maerker et al. (2008) found PTPIP51 protein to be involved in mammalian development. PTPIP51 mRNA and protein could also be traced in various carcinomas (Lv et al. 2006; Koch et al. 2008). Conspicuously, PTPIP51 was observed in cells undergoing differentiation, such as gametes and terminally differentiating keratinocytes (Stenzinger et al. 2006).
Preliminary studies investigating PTPIP51 in primary rat satellite cell cultures isolated from rat tibialis anterior muscles revealed an equally low PTPIP51 antigen concentration in all cells. These results were in contrast to earlier in situ observations made in the rat tibialis anterior muscle. Sections of the tibialis anterior muscle stained for PTPIP51 antigen displayed a certain proportion of muscle fibers showing strong PTPIP51 immunoreactivity, whereas other fibers displayed low immunostaining. The expression of high amounts of PTPIP51 could be linked to the fast-contracting fiber type (Stenzinger et al. 2005). Because all cultured myoblasts from rat tibialis anterior muscle express PTPIP51 at an equally low level, there has to be a selective up-regulation of PTPIP51 in the course of differentiation of those fibers expressing high levels of PTPIP51 in the adult skeletal muscle. To learn more about the regulation of PTPIP51 expression in the developing muscle, we investigated PTPIP51 expression during the course of myoblast-to-myofiber differentiation. We used human myoblast cultures, which were cultivated either under the influence of epidermal and fibroblast growth factors (EGF and FGF) or in the absence of either of these factors, thus initiating differentiation. Additionally, differentiated myotubes were resubmitted to the influence of EGF and FGF, thus promoting proliferation. Time-dependent changes in the expression of PTPIP51 were monitored by analyzing samples from various time points. Therefore, we were able to provide evidence for a differentiation-dependent expression of PTPIP51 in human skeletal muscle cells.
Materials and Methods
Cell Culture
Four human myoblast cultures (rectus femoris and tibialis anterior muscles) were obtained from the muscle tissue culture collection at the Friedrich-Bauer Institute (Department of Neurology, Ludwig-Maximilians University, Munich, Germany). The muscle tissue culture collection is part of the German network on muscular dystrophies (MD-NET, service structure S1, 01GM0601) funded by the German Ministry of Education and Research (BMBF; Bonn, Germany).
Cultivation
All cells were kept at 37C in a humidified atmosphere of 5% CO2. For proliferation, cells were cultivated on dishes or on culture slides in skeletal growth muscle medium (PromoCell; Heidelberg, Germany) supplemented with 10 μg hEGF, 1 μg hbFGF, 50 mg fetuin, 10 mg insulin, 400 μg dexamethason, 50 mg gentamycin, 50 μg amphotericin, 100,000 U penicillin, 100 mg streptomycin, and 20% fetal calf serum (FCS) (HyClone; Bonn, Germany) per liter.
When cells reached subconfluency, differentiation was initiated by switching the medium to DMEM high-glucose medium (PAA; Marburg, Germany) including 4.5 g glucose, 100,000 U penicillin, 100 mg streptomycin, and 3% FCS per liter.
Samples for immunostaining were taken at different time intervals before and after onset of differentiation, fixed in methanol (-20C) for 10 min, and subsequently stored at −20C until use.
For reproliferation experiments, each culture was divided into two samples. One sample continued to grow in differentiation medium and was used as a control, whereas further differentiation of the second sample was inhibited by a switch to proliferation medium. At defined time intervals, coverslips of each group were fixed with methanol and stored until use.
PTPIP51 Antibody Production
For details of antibody production see Stenzinger et al. (2005).
Immunohistochemistry
Immunohistochemical stainings were performed according to a standard protocol. Cells were fixed at −20C with methanol for 10 min. The primary polyclonal rabbit antibody against the PTPIP51 antigen was used in a dilution of 1:250 for immunocytochemical staining and visualized with Alexa Fluor 555 (Molecular Probes; Leiden, The Netherlands) as secondary antibody. Primary monoclonal antibodies (mouse) used for double staining experiments were as follows: MIB-1/Ki-67 (DakoCytomation; Glostrup, Denmark), SERCA1, SERCA2, My32 (Sigma; St. Louis, MO), and PTP1B (Calbiochem; Darmstadt, Germany). Mouse monoclonal antibodies were visualized with Alexa Fluor 488 (Molecular Probes). Nuclei were stained with 4′-6-diamidino-2-phenylindole (DAPI).
Double immunostainings were performed as follows: The rabbit polyclonal antibody against PTPIP51 was used in combination with one of the mouse monoclonal antibodies in the incubation medium. Simultaneous primary antibody incubation was done for 24 hr at room temperature. After several washing steps, the cells were incubated with both secondary antibodies (Alexa Fluor 555, goat anti-rabbit IgG and Alexa Fluor 488, goat anti-mouse IgG) simultaneously for 1 hr at room temperature. After further washing steps, the coverslips were mounted onto glass slides. Omission of the first antibody was used as an internal negative control.
The specificity of the PTPIP51 antibody was established by preabsorbing the antibody for 24 hr at 4C with its specific antigen prior to immunostaining.
Pictures were taken with an Axioplan 2 fluorescence microscope equipped with Plan-Apochromat objectives (Carl Zeiss; Jena, Germany) for photo documentation. For visualization of the secondary antibody, Alexa Fluor 555, an excitation filter with a spectrum of 530–560 nm, and an emission filter with a spectrum 572.5–647 nm were used. Alexa Fluor 488 was visualized by an excitation filter with a range of 460–500 nm and an emission filter with a range of 512–542 nm.
Microscope settings were adjusted by using the negative control as baseline value.
Semiquantification of Immunofluorescence Intensities
For semiquantifying immunfluorescence intensities, immunostaining intensities of mononuclear myoblasts and multinuclear myotubes (>2 nuclei) were registered for encircled single cells with the Leica histology program. Intensities of 150–200 cells/group were measured. The averaged values registered for undifferentiated mononucleated cells were set as 100%. The averaged values registered for multinucleated myotubes were set in relation to the value registered for mononucleated cells. The resulting differences in PTPIP51 immunofluorescence were plotted in a diagram.

Preabsorption of the protein tyrosine phosphatase-interacting protein 51 (PTPIP51) antibody with samples of cultured human muscle cells differentiating for 6 days. (
Immunoblotting
Cells were lysed in Laemmli buffer (Laemmli 1970) and subsequently separated on a 10% SDS-PAGE gel. Transfer on an Immobilon P membrane (Millipore; Billerica, MA) was performed according to Towbin et al. (1979). The membrane was blocked with 10% fat-free milk powder in PBS and subsequently incubated for 1 hr with antibodies diluted in 0.5% fat-free milk powder in PBS. The incubation with polyclonal anti-PTPIP51 was done overnight at 4C. Alkaline phosphatase—conjugated anti-rabbit immunoglobulins were applied for 1 hr at room temperature, and the reaction was visualized with the SigmaFast BCIP/NBT substrate. A prestained molecular mass marker (Fermentas; Glen Burnie, MD) was used for calibration.
PCR
Quantitative RT-PCR (qRT-PCR) was performed on an iCycler using SYBR Green Supermix (BioRad; Munich, Germany) to visualize the respective amplicons. Per sample, 2 μl cDNA was used for amplification of PTPIP51. Cycling conditions were 95C for 3 min, followed by 40 cycles of 95C for 30 sec, 55C for 30 sec, and 72C for 30 sec. The following primers were employed (MWG; Ebersberg, Germany): 5′gcaggtggtgctatcaggtc3′ as forward primer and 5′agctccagggccaacttcatc3′ as reverse primer, resulting in a 232-bp amplification product. PCR products were visualized by agarose gel electrophoresis. Amplification of a 90-bp β-actin product served as positive control, and negative controls included samples lacking reverse transcriptase.
ISH
ISH was performed as described previously (Steger et al. 1998, 2000) using a stringency to achieve hybridization only with a homology greater than 80%. Samples of muscle cells were partially digested with proteinase K, postfixed in 4% paraformaldehyde, and exposed to 20% acetic acid. After prehybridization in 20% glycerol, sections were covered with the digoxigenin (DIG)-labeled sense or antisense cRNA probes. Production of DIG-labeled cRNA probes was performed as described previously (Steger et al. 1998). PCR conditions were 94C for 3 min, 66C for 30 sec, 72C for 60 sec, and 72C for 8 min using 5′gtctctcacctgagctggctagctaga3′ as forward primer and 5′ggatagccaaatcctctcctccttcgtga3′ as reverse primer. The 247-nucleotide PCR product (nt position 1126–1372, corresponding to aa 376–457) of the human PTPIP51 gene, was subcloned in pGEM-T (Promega; Heidelberg, Germany). Plasmids were transformed in the XL1-Blue Escherichia coli strain (Stratagene; Heidelberg, Germany) and extracted by column purification (Qiagen; Hilden, Germany). In vitro transcription of DIG-labeled PTPIP51-cRNA was performed using the RNA-DIG labeling mix (Boehringer Mannheim; Mannheim, Germany) and RNA polymerases T3 and SP6. Vectors containing the PTPIP51 inserts were digested with NcoI and NotI (New England Biolabs; Frankfurt, Germany) for the production of sense cRNA and antisense cRNA, respectively. Both cRNAs were used at a dilution of 1:100 (the optimal concentration was determined by a dot blot test) in hybridization buffer containing 50% deionized formamide, 10% dextran sulfate, saline sodium citrate (SSC), Denhardt's solution, 10 mg/ml salmon sperm DNA, and 10 mg/ml yeast tRNA. Hybridization was performed overnight at 37C in a humidified chamber containing 50% formamide in SSC. After blocking with 3% BSA, sections were incubated with the anti-DIG Fab antibody conjugated to alkaline phosphatase (Boehringer Mannheim) overnight at 4C. Staining was visualized by developing sections with nitro blue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate in a humidified chamber protected from light. Alternatively, the sections were incubated with anti-DIG-fluorescein Fab fragments (Roche; Mannheim, Germany). For each test, negative controls were performed using DIG-labeled cRNA sense probes.
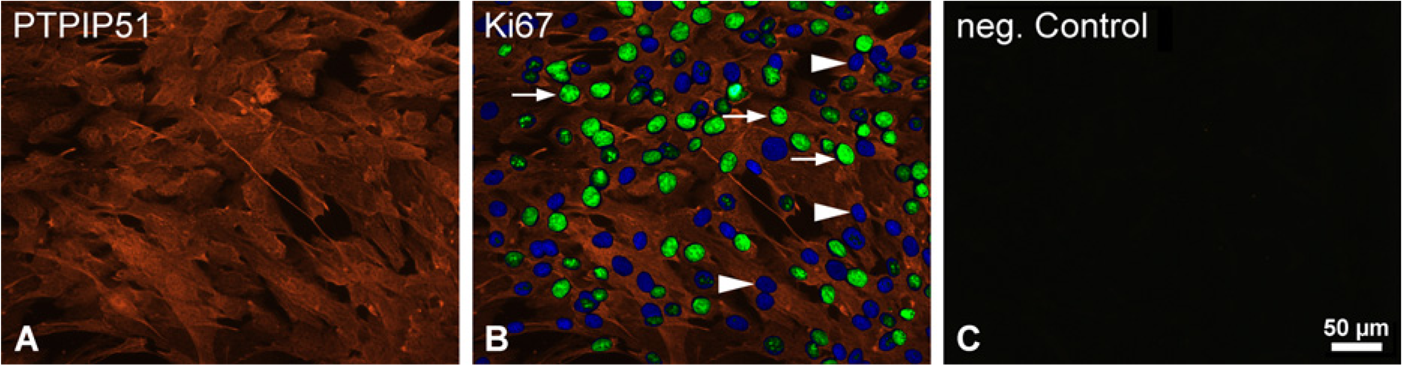
Immunohistochemical double staining of PTPIP51 and Ki67 of human muscle cells proliferating for 4 days. (
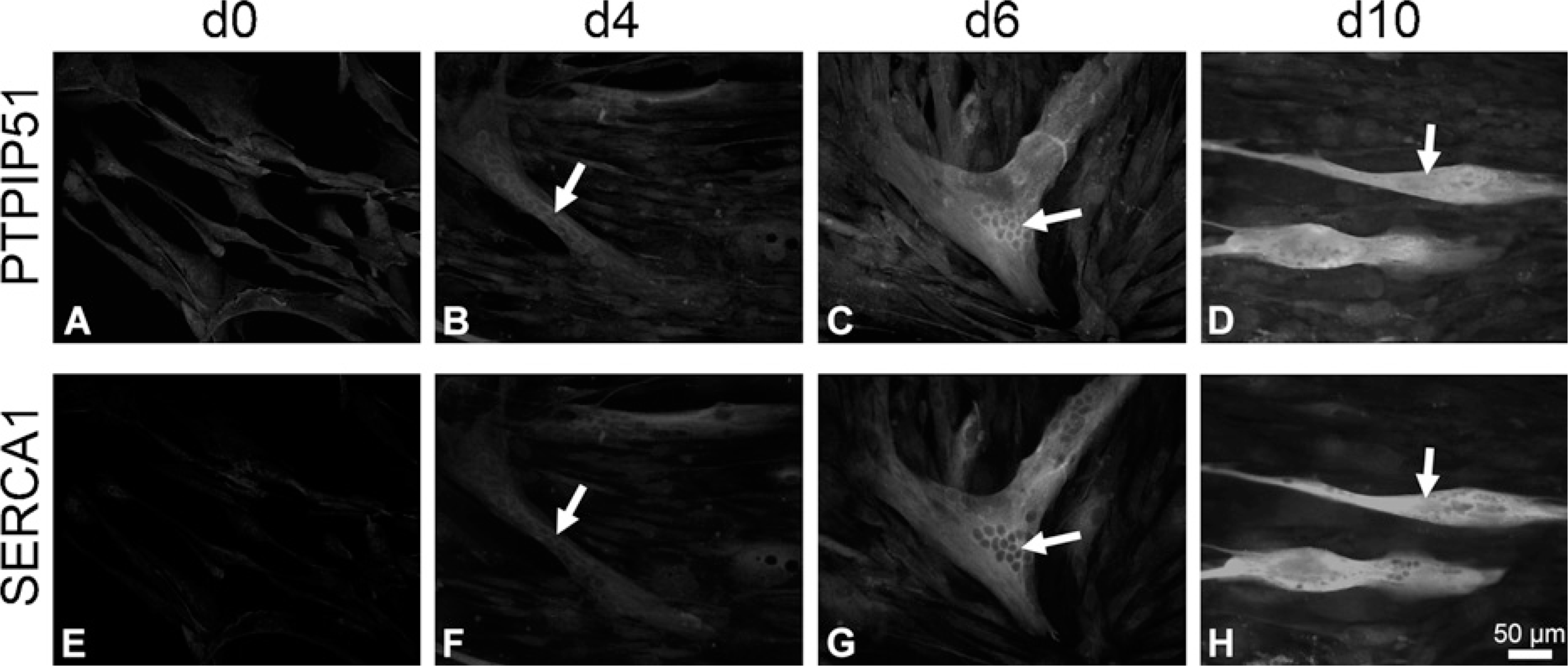
Immunohistochemical double staining of PTPIP51 and SERCA1 in proliferating (d0) and 4, 6, and 10 days differentiating (d4, d6, d10) human muscle cells. (
Results
PTPIP51 expression was analyzed in myotubes and myoblasts of four different muscle cell cultures derived from human rectus femoris and tibialis anterior muscles. In a first step, the specifity of the PTPIP51 immunoreaction was controlled through preabsorption experiments. As shown in Figure 1B, preabsorbing the antibody with its antigen resulted in the complete loss of positive PTPIP51 labeling in myotubes and myoblasts. Controls stained with non-pretreated antibody displayed an unchanged immunoreaction (Figure 1A).
Cultured myoblasts stained for PTPIP51 antigen displayed only a low immunostaining (Figure 2). Using an unsynchronized culture, the myoblasts were undergoing different phases of the cell cycle. Double immunostainings using the antibody against PTPIP51 in combination with an antibody against the mitotic core protein Ki67 revealed the same level of PTPIP51 in myoblasts in the mitotic phases G1, S, G2, and M (Figure 2). Cells in G0 phase, marked by nuclear DAPI staining, but not displaying Ki67 staining (Figure 2B), did not differ in their PTPIP51 staining from cells undergoing mitosis.
Myogenic cells cultured for various time intervals under the influence of the proliferation factors EGF and FGF displayed low immunostaining of the PTPIP51 antigen.
The effect of differentiation on PTPIP51 expression in myogenic cells was studied by comparing cells grown under conditions of proliferation with cells grown under conditions of differentiation.
Proliferating myoblast cultures, grown to near confluency, were analyzed for PTPIP51 at various time points (Figure 3A; day 0 of differentiation). PTPIP51 levels remained unchanged, unless submitting the myoblasts to the differentiation medium. The changes observed in PTPIP51 were monitored on days 4, 6, and 10 of differentiation (Figure 3B—D). In parallel, differentiation status was controlled by double immunostainings using antibodies against PTPIP51 antigen in combination with antibodies against either sarcoplasmic reticulum Ca2+ ATPase (SERCA1 and 2) or muscle fiber—specific myosin heavy-chain protein (MHC/My32). SERCA and MHC proteins are not expressed unless fusion of myoblasts to myotubes takes place and differentiation starts. Corresponding data for days 4, 6, and 10 are presented for SERCA1 in Figure 3E, F, and H and SERCA2 in Figure 4E, F, and H.
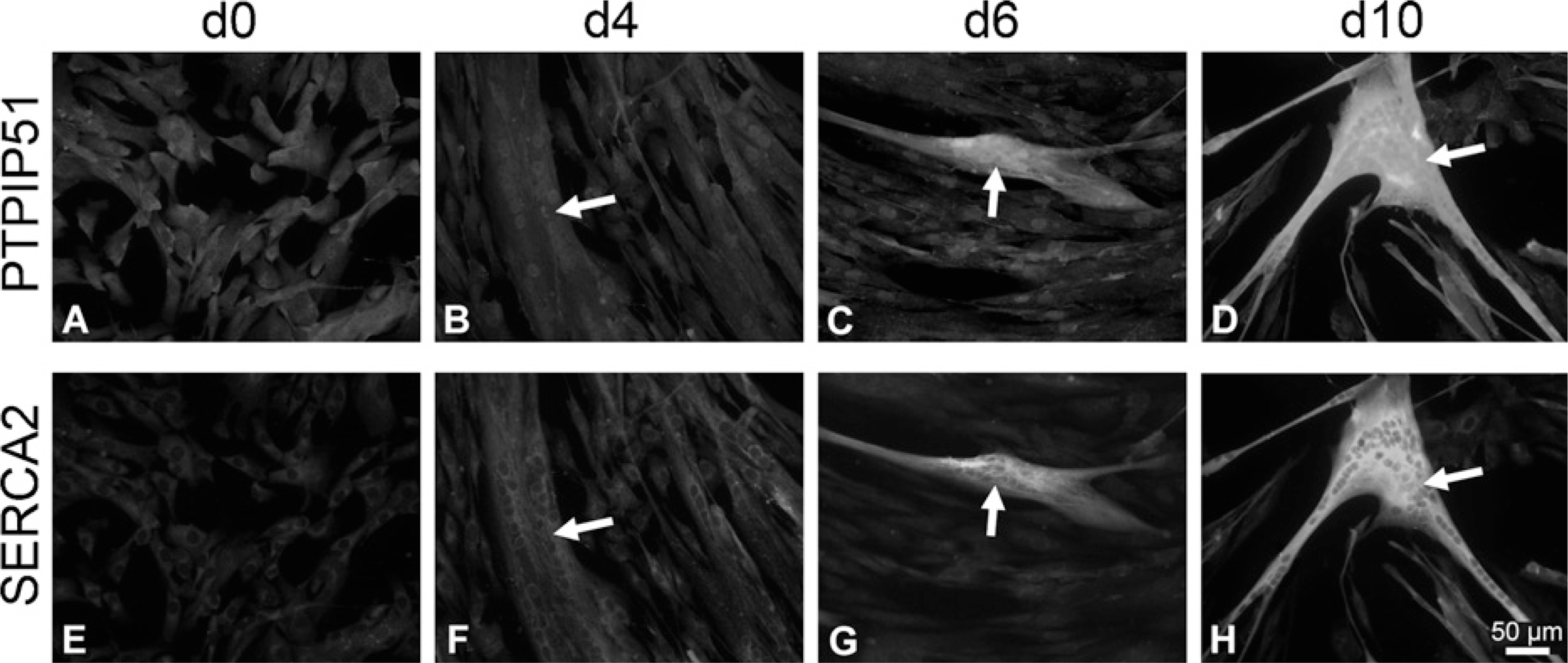
Immunohistochemical double staining of PTPIP51 and SERCA2 in proliferating (d0) and 4, 6, and 10 days differentiating (d4, d6, d10) human muscle cells. (
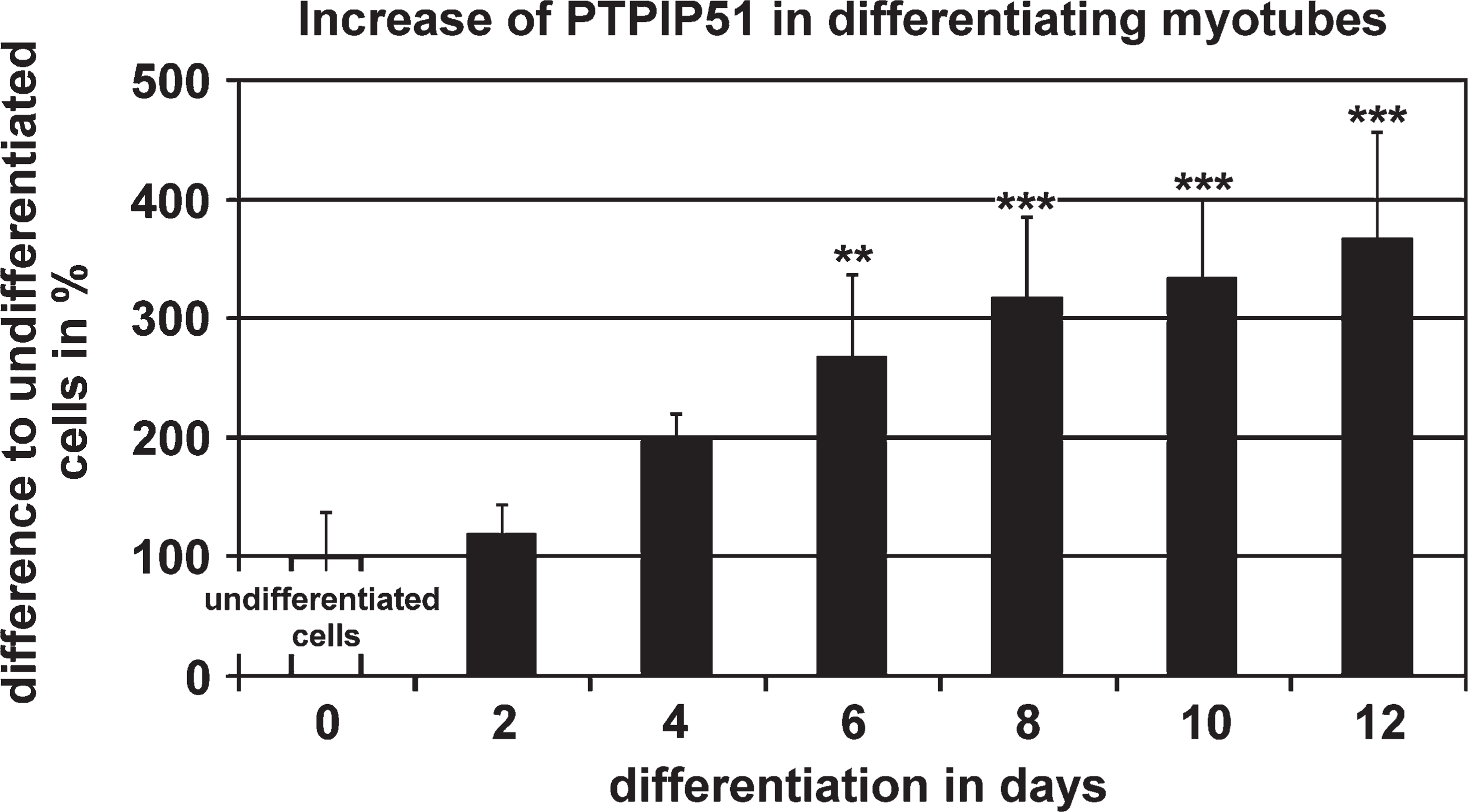
Semiquantitative analysis of PTPIP51 immune fluorescence intensities measured in myoblasts (d0) compared with myotubes grown under conditions of differentiation for 2, 4, 6, 8, 10, and 12 days (d2–d12). Data are presented as means ± standard deviation. ∗∗ p<0.01 and ∗∗∗ p<0.001 vs undifferentiated myoblasts.
Because My32-MHC and SERCA1 are specific for fibers of the fast type and SERCA2 is restricted to slow fibers, we were able to investigate the evolving fiber type of the emerging myotubes. PTPIP51 is associated with myotubes staining for both the fast and slow types (Figures 3 and 4). Because myotubes displayed both types of proteins specific for both the slow and fast types, they were identified as fibers of hybrid type.
Semiquantification of fluorescence staining intensities displayed a continuous linear increase in PTPIP51 in multinucleated myotubes undergoing differentiation compared with the values of proliferating mononucleated myoblasts (Figure 5).
Within 12 days after the onset of fusion, PTPIP51 fluorescence values registered for myotubes were 3-fold higher compared with values of non-differentiated, mononuclear myoblasts (Figure 5).
In situ hybridization experiments corroborated these results on the mRNA level in myoblasts and myotubes grown for 10 days under conditions of differentiation. PTPIP51 mRNA in mononucleated myoblasts displayed only a low level of mRNA, whereas differentiated multi-nucleated myotubes showed a strong mRNA reaction (Figure 6).
Submitting myotubes grown for 14 days in differentiation medium to proliferation medium for an additional time of 10 days resulted in a markedly reduced PTPIP51 staining pattern (Figure 7C). Cells from the same batch grown permanently in differentiation medium and harvested at identical time intervals served as controls (Figure 7B).
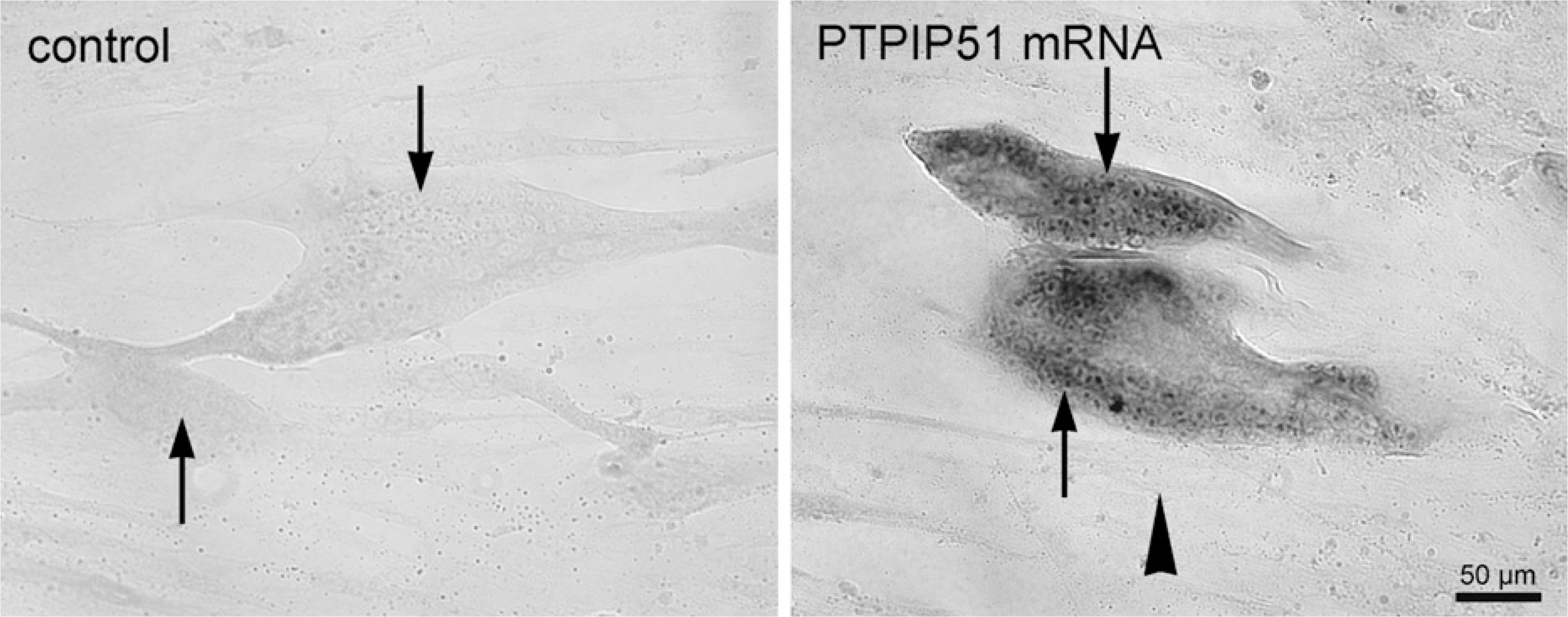
In situ hybridization of PTPIP51 mRNA (black) in differentiated (arrow) and undifferentiated (arrowhead) human muscle cells. (Left) Sense. (Right) Antisense.
Immunostaining of PTPIP51 in muscle cells. (
Semiquantification of PTPIP51 staining intensities in myotubes regrown under the influence of EGF and FGF, thus promoting proliferation, revealed a loss of more than 50% in PTPIP51 staining compared with values registered for control cells grown in parallel in differentiation medium (Figure 8). Data were standardized to PTPIP51 reaction intensities of the mononucleated myoblasts.
When samples of proliferating myoblasts were probed by RT-PCR, differentiated and differentiation-inhibited myotubes confirmed the expression of PTPIP51 mRNA during proliferation and differentiation (Figure 9). Additionally, qRT-PCR revealed a significant increase in PTPIP51 mRNA in differentiated myotubes compared with proliferating myoblasts. Inhibition of differentiation significantly reduced PTPIP51 mRNA expression compared with the mRNA level of differentiating myo-tubes (Figure 10).
Immunoblotting of extracts from cultured myoblasts grown under proliferating conditions revealed bands with a molecular mass of ∼52 kDa and 65 kDa (Figure 11, D0). Differentiation for 13 days led to an additional band with an approximate molecular mass of 85 kDa (Figure 11, d13). When differentiation of myotubes was inhibited by switching them to proliferation medium, this 85-kDa PTPIP51 form disappeared (Figure 11, d13+10).
Possible interactions with PTP1B were investigated by double immunostaining techniques in myoblasts and myotubes differentiated for 8 days. All myoblasts and myotubes expressed PTPIP51 and PTP1B, but only partial colocalization could be detected (Figure 12).
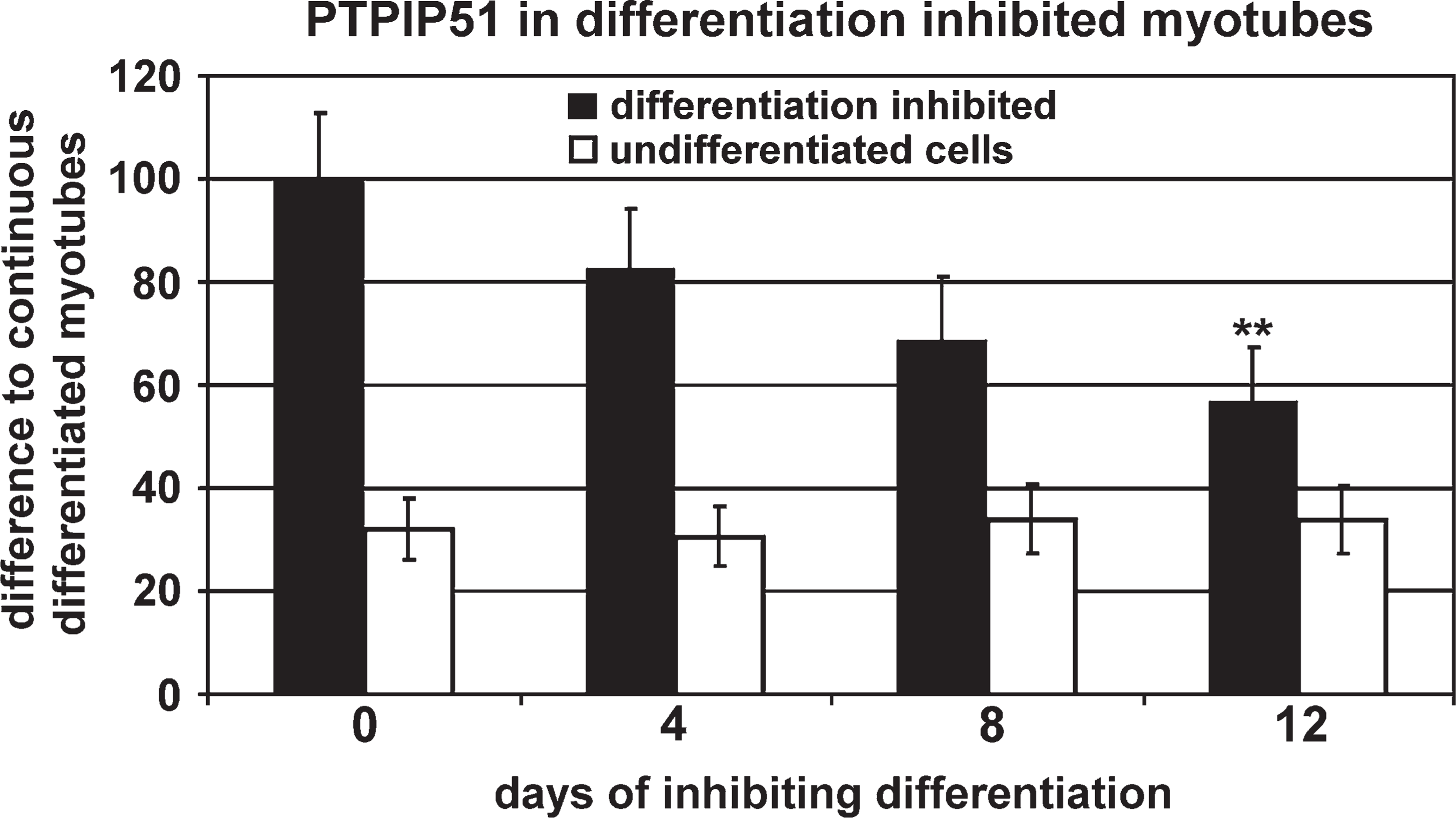
Semiquantitative analysis of PTPIP51 immunofluorescence intensities measured in myotubes cultivated for 14 days in proliferation medium and then cultivated for another 4, 8, and 12 days in proliferation medium (differentiation-inhibited, black bars) related to PTPIP51 immunofluorescence intensities measured in myotubes permanently cultivated in differentiation medium (corresponding to 100%). Basic PTPIP51 immunoreaction in undifferentiated cells permanently grown in differentiation medium relative to the immunoreaction of myotubes continuously cultivated in differentiation medium (white bars). Data are presented as means ± standard deviation. ∗∗ p<0.01 vs continuous differentiated myotubes.
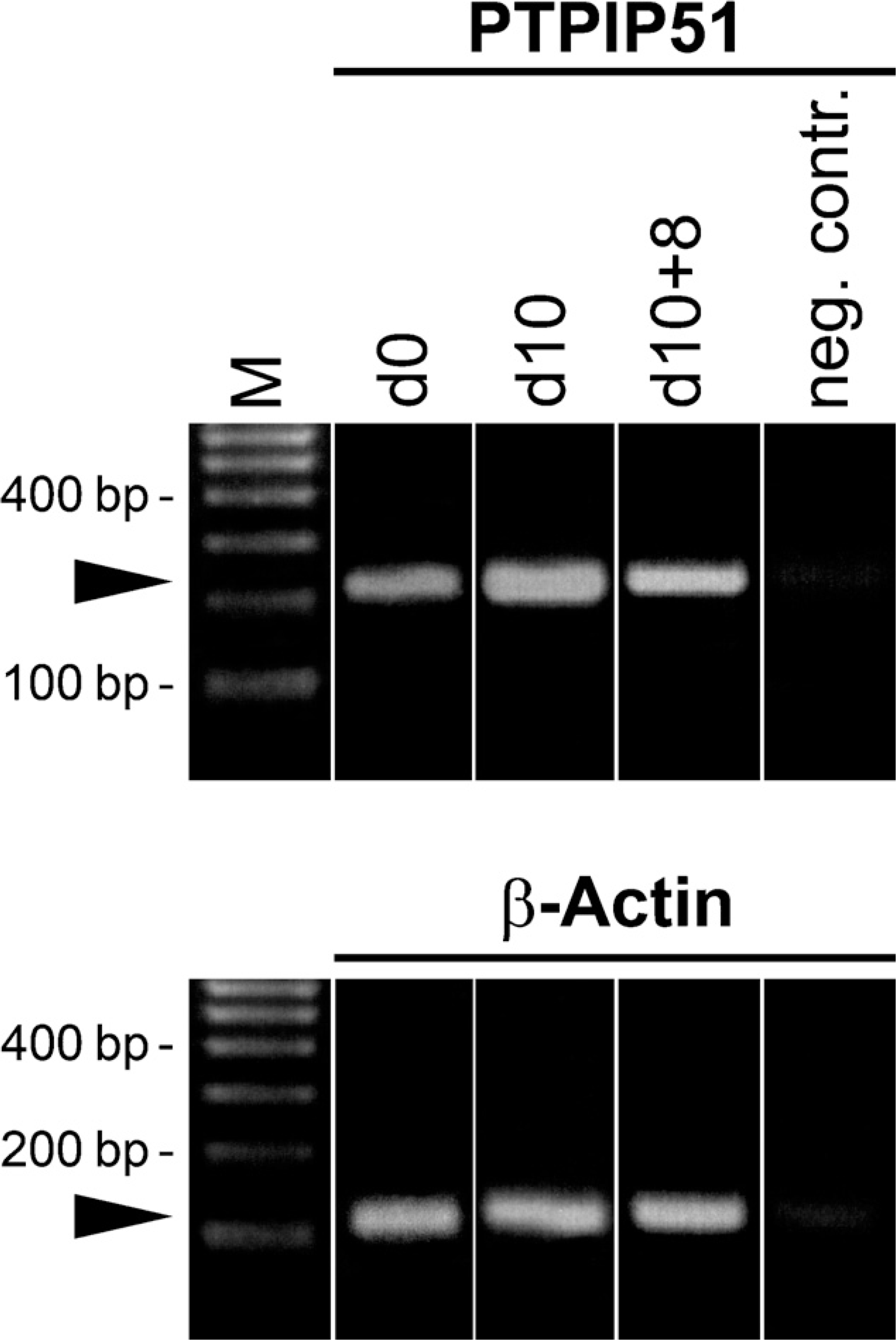
RT-PCR of PTPIP51. Effects of proliferation on PTPIP51 mRNA expression. Cells were grown for 8 days in proliferation medium (d0), differentiation [10 days in differentiation medium (d10)], and proliferation inhibition [10 days differentiation and subsequent growth for 8 days in proliferation medium (d10+8)]. β-Actin was amplified as an internal positive control, and probes lacking reverse transcriptase served as negative controls.
In human muscle cells, PTPIP51 could not be associated to specific subcellular compartments, neither of mitochondria nor of the endoplasmic reticulum, as analyzed by colocalizing antibodies against Complex 5 and protein disulphide isomerase with the PTPIP51 antigen (data not shown).
Discussion
The expression of PTPIP51 was shown in cultured human muscle cells. Proliferating human myoblasts exhibited a low concentration of PTPIP51. The expression of PTPIP51 was not linked to any mitotic processes, inasmuch as no association to mitotic core protein Ki67 or to mitotic figures was detected. Terminally differentiated myotubes, marked by their expression of muscle-specific proteins, such as SR Ca2+ ATPase and myofibrillar myosin heavy-chain (Strohman et al. 1974; Stromer et al. 1974; Puente et al. 2004), showed high levels of PTPIP51 and mRNA. Sarcoplasmic reticulum proteins (Harrer et al. 1995; Stenoien et al. 2007) and MHC proteins (Muller et al. 1996; Furmanczyk and Quinn 2003; Takata et al. 2007) are sequentially expressed in the course of terminal differentiation. The SR Ca2+ ATPase isoform is dependent on the fiber type. Using antibodies against both isoforms, all cultured human myotubes were identified as hybrid fiber types, which share characteristics of both slow and fast types.
Mouly et al. (1993) reported that fused myotubes of human satellite cell cultures are of the hybrid type. They express embryonic and fetal MHC isoforms in addition to adult fast and slow MHC isoforms (Edom et al. 1994; LaFramboise et al. 2003). PTPIP51 is expressed in all in vitro—differentiated myotubes of mixed MHC type; thus, its expression cannot be linked to a specific MHC isoform.
In cultures of rat as well as mouse myogenic cells, MHC protein and mRNA expression were not evident prior to the onset of differentiation (Shani et al. 1981; Pin and Merrifield 1993) followed by a continuous linear increase (Caravatti et al. 1982). This also holds true for the expression of PTPIP51 in human satellite cell cultures.
Stenoien et al. (2007) observed a linear increase in both SERCA proteins as cells were differentiating into myotubes. In our study, the expression of SERCA1 and −2 isoforms paralleled PTPIP51 expression during terminal differentiation.
Terminal muscle differentiation depends on numerous phosphorylation processes (Puente et al. 2004, 2006), including MHC proteins, which during differentiation are tyrosine phosphorylated by c-Src kinase (Goel and Dey 2002; Harney et al. 2005). C-Src kinase is also known to interact with PTPIP51 (Schreiner et al., unpublished data).
C-Src is not only important for mitogen signal transduction, but also is known to be essential for muscle fiber differentiation (Lu et al. 2002). Dephosphorylation of c-Src kinase leads to an accelerated differentiation seemingly by an activation of p38 mitogen-activated protein kinase (MAPK), which enhances the transcriptional activities of MyoD and myocyte enhancer binding factor2 (Lu et al. 2002; Lim et al. 2007).
Proliferation of myoblasts was promoted by the growth factors EGF and bFGF, both known to promote the proliferation of myoblasts by stimulating the c-Src—dependent Ras/Raf/MEK/ERK pathway. The inactivation of the Raf/ERK pathway, however, does not promote myogenic differentiation (Jones et al. 2001). The Raf/MEK/ERK pathway is stage-specifically inhibited during muscle differentiation by Akt (Rommel et al. 1999). PTPIP51 is known to regulate ERK activity at the level of Raf. Yu et al. (2008) suggested that PTPIP51 could be a regulator of the Raf/MEK/ERK cascade through a modulation of Raf-1 by 14-3-3. It is tempting to assume that PTPIP51 is involved in muscle differentiation via the regulation of Raf, inasmuch as low levels of Raf signaling lead to accelerated differentiation (DeChant et al. 2002).
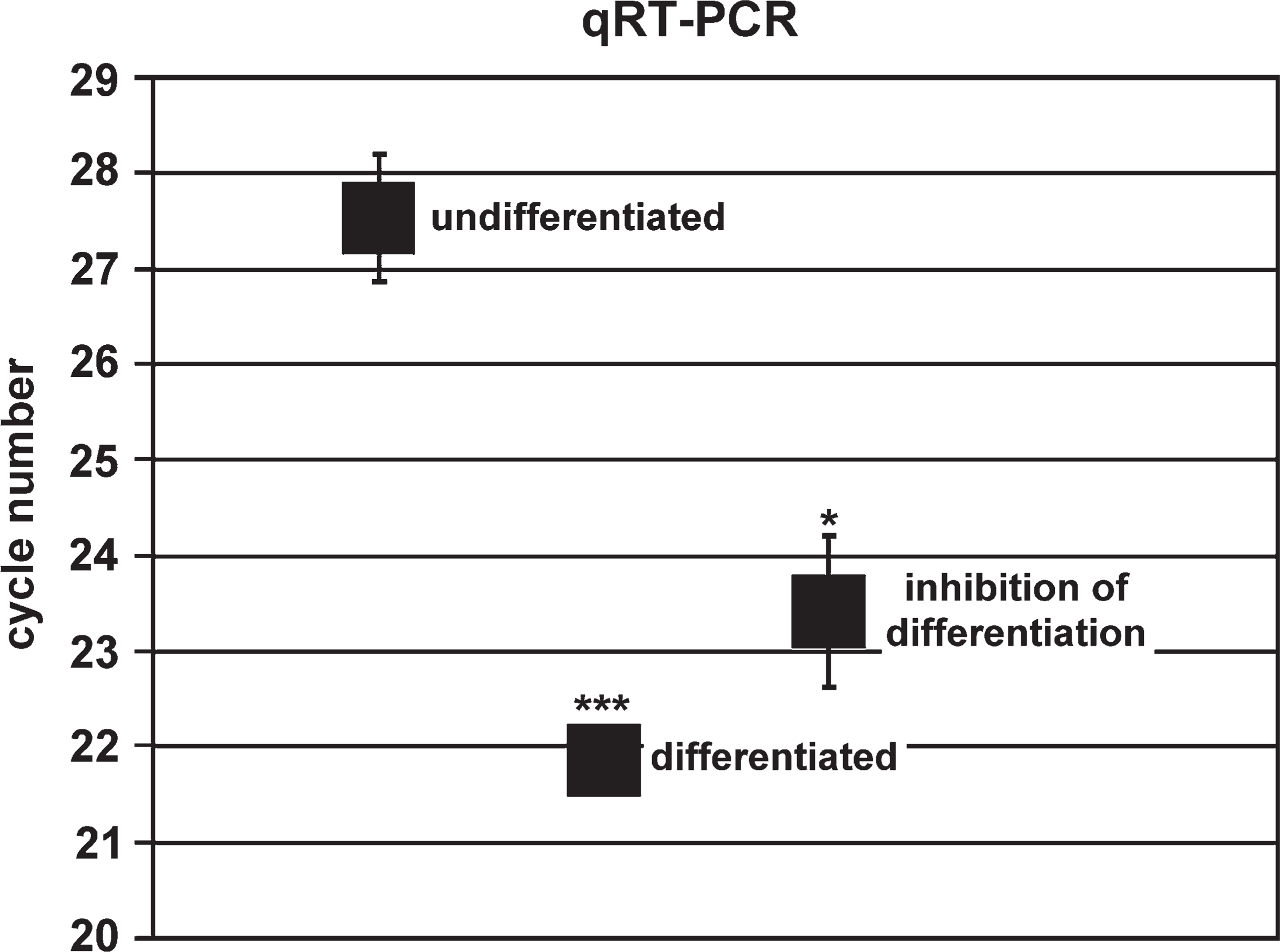
Quantitative RT-PCR (qRT-PCR). The quantification was performed by a light cycler. The graph shows the cycle number, which was needed to achieve significant PTPIP51 mRNA values (crossing point with the baseline). Mean values are given for proliferating cells, cells differentiated for 8 days, and cells differentiated for 8 days with subsequent cultivation for 4 days in proliferation medium. Data are presented as means ± standard deviation. ∗ p<0.05 vs continuous differentiated myotubes and ∗∗∗ p<0.001 vs undifferentiated myoblasts.
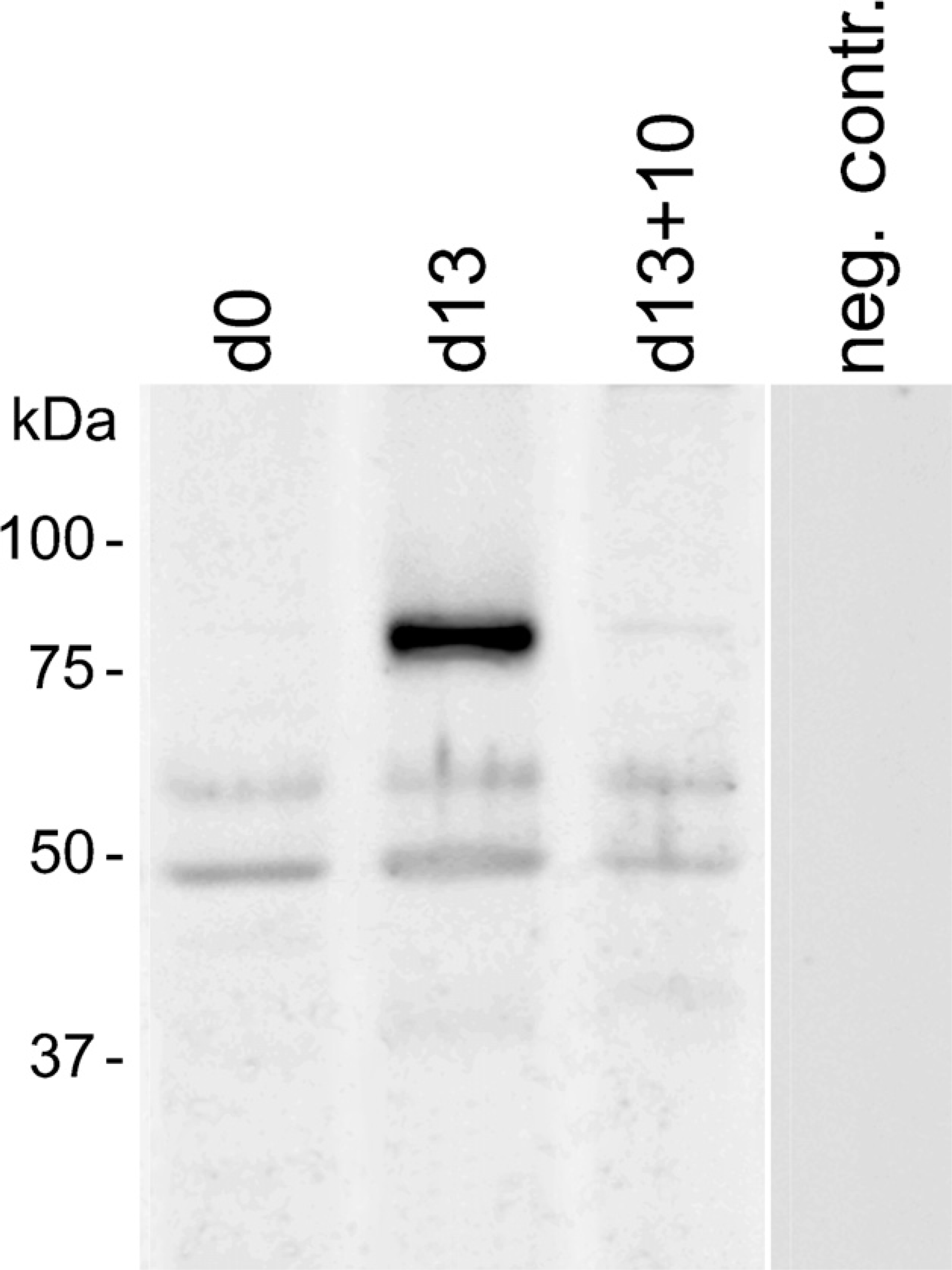
Immunoblotting of PTPIP51 in proliferating (d0), differentiating (d13) and differentiation-inhibited for 10 days (d13+10) cultured human muscle cells.
The fact that Raf/MEK/MAPK negatively affects skeletal myogenesis and muscle-specific transcription and differentiation in multinucleated myocytes (Wang et al. 2004) could explain the effect exerted on multi-nucleated myotubes by resubmission to EGF and FGF, which results in a reduced PTPIP51 expression. Investigating a probable interaction of PTPIP51 and Raf in skeletal muscle will be a subject for further studies.
bFGF acts as a potent myoblast mitogen by suppressing the expression of muscle-specific proteins. Mediated by MAPkinase, FGF inhibits differentiation, repressing the expression of the muscle-specific transcription factors MyoD and myogenin (Tortorella et al. 2001). EGF, like FGF, is known to induce proliferation in muscle cells (Doumit et al. 1993; Yablonka-Reuveni 1995) by activating c-Src kinase, MAPK, and non-receptor protein tyrosine kinase, all tyrosine-phosphorylating enzymes (Belsches et al. 1997; Biscardi et al. 2000; Fuller et al. 2008).
Reapplication of EGF and FGF prevented further differentiation, an effect that has been described before (Lassar et al. 1994; Yablonka-Reuveni 1995; Vachon et al. 1996), resulting in the activation of the STAT3/RAS/MAPK pathway (Megeney et al. 1996; Bottcher and Niehrs 2005). Phosphorylation of STAT3 by protein kinase C (PKC) activates proliferation and inhibits differentiation (Kataoka et al. 2003; Jo et al. 2005).
Stenzinger et al. (2006) reported a downregulation of PTPIP51 expression in keratinocytes that had been exposed to physiological concentrations of EGF. Our present study corroborates this observation by showing the downregulation of formerly high PTPIP51 expression in myotubes when these were resubmitted to EGF and bFGF.
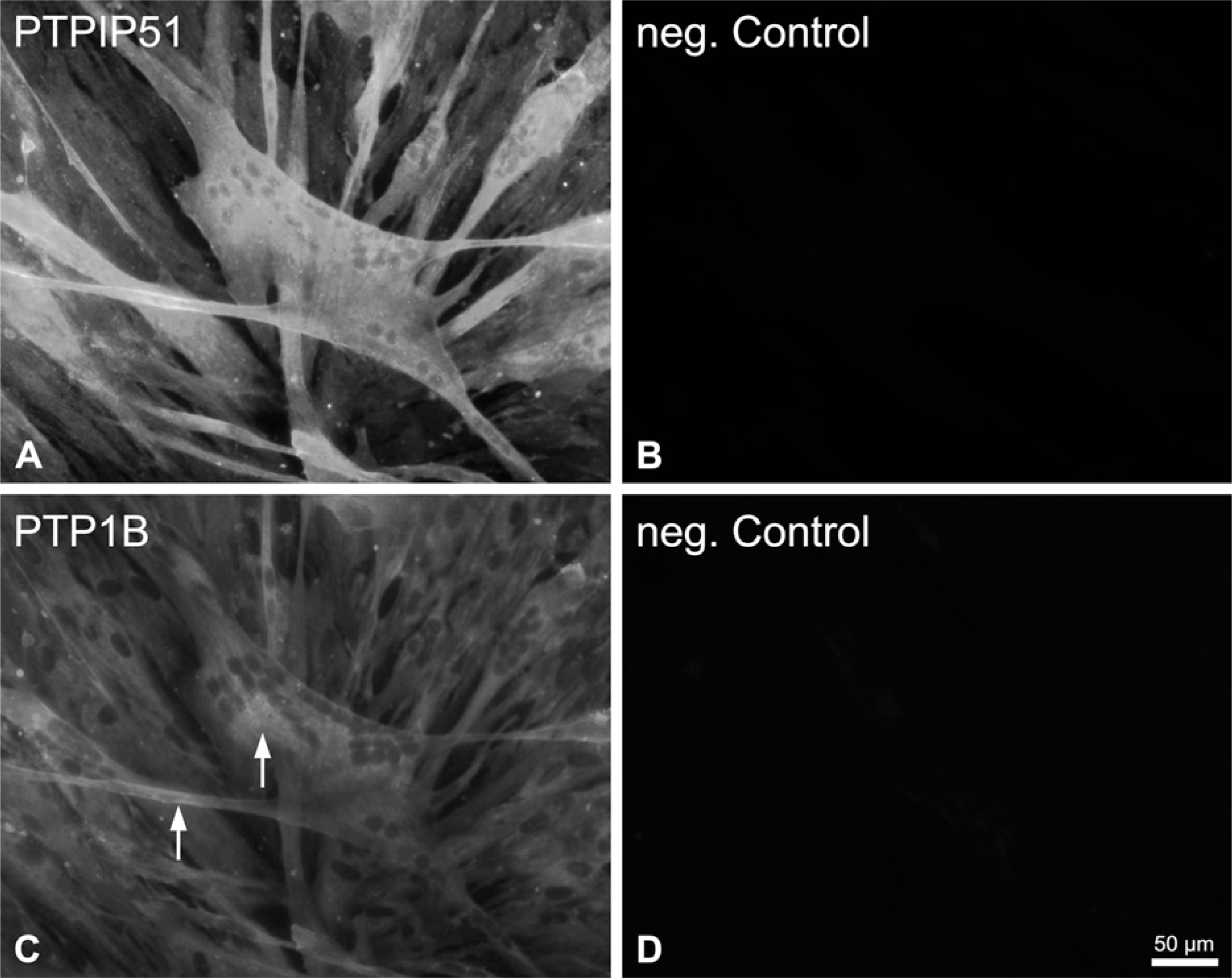
Immunohistochemicalstaining of PTPIP51 and protein tyrosine phosphatase 1B (PTP1B) in differentiated and undifferentiated mononuclear human muscle cells. (
PTPIP51 revealed a cytoplasmic localization in proliferating myoblasts. The pattern of PTPIP51 expression in differentiating myotubes matched that of SERCA protein, thus indicating a possible interaction with structures of the sarcoplasmic reticulum (SR). Considering the membrane target sequence of the full-length protein, a possible integration of this PTPIP51 isoform into SR membranes is likely. Because PKC, a theoretical interaction partner of PTPIP51 (PROSCAN in Expasy.ch), is involved in the regulation of SR receptor activity (Berchtold et al. 2000), a potential implication of PTPIP51 in the regulation of SR proteins is possible.
Proliferating cells revealed PTPIP51 forms with approximate molecular mass of 52 kDa and 65 kDa. Differentiating myotubes had an additional form with an apparent molecular mass of 85 kDa. This high-molecular-mass form was downregulated in multinucleated myotubes by suppressing differentiation and inducing proliferation by reapplication of EGF and FGF. The 52-kDa protein corresponds to the full-length form of PTPIP51 with 470 amino acids. Higher molecular mass, like 65 kDa and 85 kDa, can result from posttranscriptional modifications. This is in accordance with earlier observations of Mostafa (unpublished data), who reported an 85-kDa PTPIP51 form that was modified by glycosylation. Posttranslational modifications are common mechanisms to increase protein efficiency and stability (Umar et al. 1996; Meek and Knippschild 2003; Roberts et al. 2008). Thus, the 85-kDa protein might be a stabilized form of PTPIP51 accumulating in differentiating myotubes.
PTP1B, the in vitro interaction partner of PTPIP51, is also expressed in muscle cells (Delibegovic et al. 2007). PTP1B is involved in FGF action and negatively regulates EGF-induced signaling in various cell types by dephosphorylating the EGF receptor (Chang et al. 2006; Mertins et al. 2008). Thus, in cultured smooth-muscle cells, PTP1B suppresses cell proliferation and motility (Shimizu et al. 2001). PTP1B protein was also expressed in low concentrations in cultured human myoblasts as well as in myotubes, displaying partial colocalization with PTPIP51. Therefore, a possible interaction of PTPIP51 with PTP1B in cultured myocytes seems unlikely.
In summary, PTPIP51 expression in myogenic cells is downregulated in the presence of EFG and FGF, thus confirming previous results (Stenzinger et al. 2006). Omission of EGF and FGF does not necessarily result in an upregulation of PTPIP51 protein, inasmuch as mono-nucleated cells grown in the absence of these factors still express PTPIP51, only on a base level. Upregulation of the PTPIP51 protein is linked to processes proceeding after fusion of myoblasts in the course of terminal differentiation. This is probably due to factors or signaling pathways that also regulate MHC and SERCA protein expression. An involvement of PTPIP51 in signal transduction during differentiation could be mediated through its interaction with either PKC, c-Src kinase, or Raf-ERK. Further experiments are needed to identify the factors regulating PTPIP51 expression as well as its interaction partners during muscle cell differentiation.
Footnotes
Acknowledgements
The authors thank Dr. P. Grant for linguistic revision.
