Abstract
Hemicentins are recently described extracellular matrix (ECM) proteins with a single ortholog in C. elegans that assembles into discrete tracks constricting broad regions of epithelial cell contact into adhesive and flexible line-shaped junctions. There are two highly conserved hemicentin genes in most vertebrate species; however, nothing is known about the function or distribution of vertebrate hemicentins. To determine the distribution of vertebrate hemicentins, we used a polyclonal antibody to stain mouse tissue and showed that hemicentins are found in the pericellular ECM of epithelial cells in a number of tissues including embryonic trophectoderm and adult skin and tongue, in addition to the ECM of some, but not all, blood vessels. Hemicentins also assemble on multiple epithelia in the eye, including cornea, lens, and retina. The pericellular localization of vertebrate hemicentins on epithelia and other cell surfaces suggests that vertebrate hemicentins, like their nematode counterpart, are secreted ECM proteins likely to have a role in the architecture of adhesive and flexible cell junctions, particularly in tissues subject to significant amounts of mechanical stress.
E
Although a recent study has implicated human hemicentin−1 in age-related macular degenerative disease, there is nothing else known about the function or distribution of the two vertebrate hemicentins (Schultz et al. 2003). Here we present the first evidence that vertebrate hemicentins are found in the pericellular ECM of a number of epithelial cells including embryonic trophectoderm and the epidermis of skin and tongue, in addition to the ECM of some blood vessels. Hemicentin is also associated with multiple epithelia in the eye, including cornea, lens, and retina. The data suggest that vertebrate hemicentins, like their nematode counterpart, may have a role in the architecture of adhesive and flexible epithelial cell attachments, particularly in tissues subject to significant amounts of mechanical stress.
Materials and Methods
RT-PCR Detection of Hemicentin−1 and −2
RNA was extracted from adult tissue using Trizol reagent (Invitrogen; Carlsbad, CA), and cDNA was prepared from 3 μg of total RNA using SuperScript First-strand Synthesis System (Invitrogen) with random hexamer primers. For PCR, the primers used were hemicentin−1: 5′-ATGATTGCCCAGGAAGTG and 5′-CTAGACATGGGTGGGGAA; hemicentin−2: 5′-ATGACGCCTGGGGCGCAG and 5′-ATGAACTTTGGAGGCCTG. PCR products were sequenced to avoid misinterpretation due to spurious PCR products and quantitated using Image J software (NIH).
Expression and Detection of Mouse Hemicentin−1 and −2 Fusion Proteins
Fragments encoding the first 113 amino acids of hemicentin−1 and the first 230 amino acids of hemicentin−2 were amplified by RT-PCR using RNA prepared from adult mouse skin, cloned into bacterial expression vector pFLAG-MAC (Sigma; St Louis, MO), and transformed to E. coli NovaBlue competent cells (Novagen; San Diego, CA). The primers used were 5′-AGCTTATGATTGCCCAGGAAGTG, 5′-GATCTCTAGACATGGGTGGGGAA for hemicentin 1 and 5′-AGCTTATGACGCCTGGGGCGCAG, 5′-GATCCATGAACTTTGGAGGCCTG for hemicentin 2, respectively. The constructs were verified by DNA sequencing. Flag-tagged proteins were expressed by induction with 1 mM IPTG (isopropyl-β-
E. coli cell pellets were boiled in SDS-PAGE sample buffer, and extracted proteins were separated on a 16% Tris-glycine gel (Invitrogen) and transferred to nitrocellulose membrane. After blocking with 5% non-fat milk in TBST, fusion proteins were detected with rabbit anti-hemicentin antibody followed by goat anti-rabbit IgG conjugated with horseradish peroxidase (HRP) and developed using Supersignal West Pico Chemiluminescent Substrate (Pierce Biotechnology; Rockford, IL). After the membrane was stripped with 200 mM glycine-HCl (pH 2.0), the membrane was re-probed with anti-Flag-HRP antibody (Sigma).
Animal and Tissue Preparation
C57BL/6J mice (The Jackson Laboratory; Bar Harbor, ME) were bred under standard conditions and sacrificed by cervical dislocation. All tissues were harvested immediately and snap frozen with liquid nitrogen upon dissection. Appearance of the vaginal plug was designated embryonic day (E) 0.5, and embryos were either collected from E9.517.5, or pregnancies were allowed to reach term. Newborn mice were collected at postnatal (P) day 2 or 14. All animal procedures were conducted under guidelines approved by the University of Maryland Biotechnology Institute Animal Care and Use Committee.
Histological Analysis and Immunofluorescent Staining
After sample collection, tissues or embryos were frozen in optimal cutting temperature, sectioned (5–10 μm) by cryostat, and air dried for 40 min. The sections were fixed in 4% paraformaldehyde followed by acetone.
Endostatin/collagen XVIII monoclonal antibodies (kind gift from Drs. A. Marneros and B. Olsen) (Marneros et al. 2004), desmocollin−3 antibodies (kind gift of Dr. P. Koch) (Den et al. 2006), affinity-purified hemicentin antibodies, and monoclonal anti-cytokeratin 8.13 (Sigma) were used as primary antibodies to stain mouse tissue. Secondary antibodies were rhodamine-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch Laboratory; West Grove, PA) and FITC-conjugated goat anti-mouse IgG (Jackson ImmunoResearch Laboratory). Microscopic observation of immunofluorescence was performed with a confocal microscope (LSM 410; Carl Zeiss, Oberkochen, Germany).
Results
Characterization of Anti-hemicentin Antibody
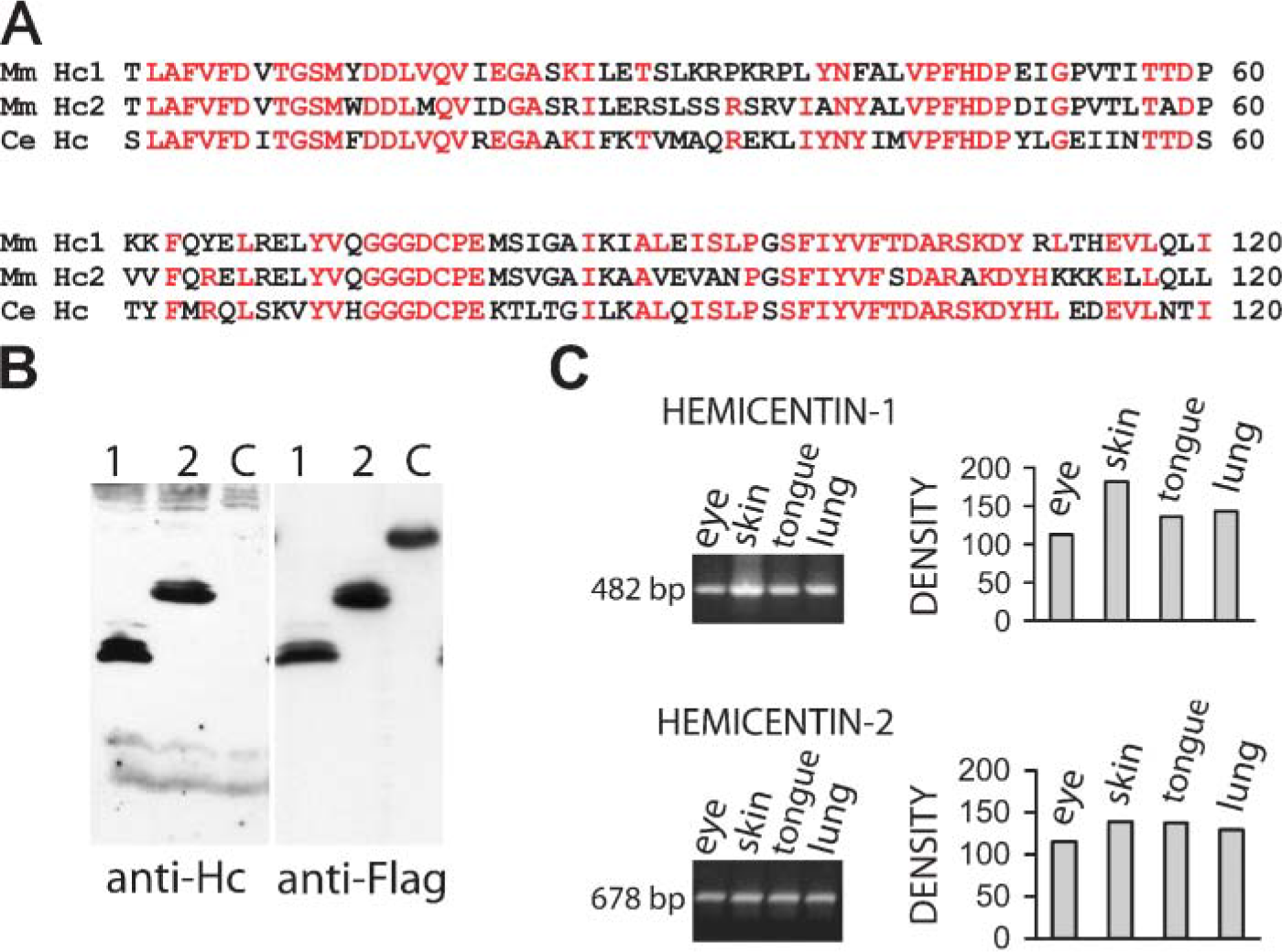
The structural organization of C. elegans hemicentin from the N-terminal is a signal sequence followed by a VWA domain, 48-tandem immunoglobulin, three EGFs, and a fibulin-like C-terminal module. The VWA domain of C. elegans hemicentin is highly conserved in mouse hemicentins−1 and −2 with 95 and 87 amino acid identities with C. elegans hemicentin out of 158, respectively (Vogel and Hedgecock 2001; Whittaker and Hynes 2002) (Figure 1A). A nematode hemicentin VWA domain: GST fusion protein was expressed in bacteria and used to immunize rabbits. Affinity-purified antibodies were characterized by Western blot and immunofluoresence on wild-type and hemicentin mutant animals (Dong et al. 2006). In Western blots, the affinity-purified antibody recognized a band of ∼600 kDa in wild-type, but not in hemicentin mutant animals (Dong et al. 2006). In immunofluoresence with C. elegans tissues, hemicentin was detected on mechanosensory neurons, tracks that connect the basement membrane of the pharynx to the basement membrane of bodywall muscle cells, and on germ cell plasma membranes in wild-type animals (not shown). These results are in complete agreement with the distribution of a functional GFP::hemicentin translational fusion that assembles on each of these tissues but not on neighboring muscle or epithelia (Vogel and Hedgecock 2001) and suggests that these antibodies can recognize native hemicentin protein found in these tissues with little background staining. The fact that this antibody recognizes a region of C. elegans hemicentin that is highly conserved in mouse hemicentin−1 and −2 (Figure 1A) suggests that this antibody might recognize these hemicentins in mouse tissues. To test this possibility, hemicentin−1 and −2 fusion proteins containing parts of the conserved VWA domain were expressed as Flag-tagged fusion proteins in bacteria. The antibody raised to the nematode hemicentin VWA domain recognized mouse hemicentin−1 and −2 fusion proteins but not an unrelated control fusion protein in Western blots of bacterial cell extracts (Figure 1B). In detergent and urea extracts of mouse skin, lung, tongue, eye, heart, and brain we were only able to detect low molecular mass (100−200 kDa) bands, even though these tissues have abundant hemicentin mRNA and antibody staining (see below). This suggests that the full-length proteins may be proteolyzed or modified in some other way that makes them difficult to detect in Western blots.
To determine whether hemicentin epitopes are detectable during mouse development, the antibody generated to the C. elegans hemicentin VWA domain was used to stain mouse embryos. At approximately 2 days postcoitus (E2) we could detect weak staining that appeared to be primarily cytoplasmic. By E2.5, an epitope that appeared to be at cell junctions in the morula was detected. In E3.5 blastocysts, intense staining was observed in a pericellular pattern around the epithelial cells found in the trophectoderm that appeared to colocalize with the distribution of the cadherin desmocollin−3 (Figure 2) (Den et al. 2006).
Hemicentin Distribution in Skin and Tongue Epithelium
To determine whether any adult tissues might contain hemicentin epitopes, we performed RT-PCR using RNA from a variety of tissues and found robust signals for hemicentins−1 and −2 in RNA obtained from skin, tongue, eye, and lung (Figure 1). When mouse skin was stained with the anti-hemicentin antibody, prominent staining was detected in the epidermis with affinity-purified immune serum but not with preimmune serum or with affinity-purified immune serum preincubated with 25 μg/ml of the fusion protein used in immunization (Figure 3). In addition, double staining with cytokeratin antibodies (Figure 3) demonstrated that the hemicentin signal appears to be extracellular. In stages of mouse development ranging from 12.5 days postcoitus (E12.5) through adulthood, staining can be observed in a pericellular pattern around individual epithelial cell surfaces [cf. immunofluorescence (Figure 3A) with hematoxylin/eosin (Figure 3B) staining].
By E17.5, embryonic skin has differentiated into distinct epidermal and dermal layers, and hair follicles have partially formed. Anti-hemicentin antibodies stain the periphery of basal and suprabasal keratinocytes, but there is little or no staining in the stratum corneum, the keratinized surface layer of epidermis (Figures 3C and 3D). At this stage, strong fluorescence is also observed in developing hair follicles. By postembryonic day 14 (P14), the skin of the newborn mouse is fully differentiated and specialized dermal structures including hair follicles, sebaceous glands, and sweat glands have become functional. Hemicentin continues to be detectable at the periphery of basal and suprabasal keratinocytes and is also detected in dermal specializations (Figures 3E and 3F), but there appears to be little staining associated with dermal fibroblasts or in the surrounding connective tissue. In hair follicles, hemicentin staining is typically located peripheral to follicular epithelial cells (Figures 3E, 3F, and 3N). Adjacent to hair follicles, fluorescence can also be detected on sebaceous glands (Figures 3E, 3F, and 3O). In this holocrine gland, hemicentin is detected only around the periphery of the flattened basal epithelial cells (Figure 3O), but there is no hemicentin staining associated with secretory cells.
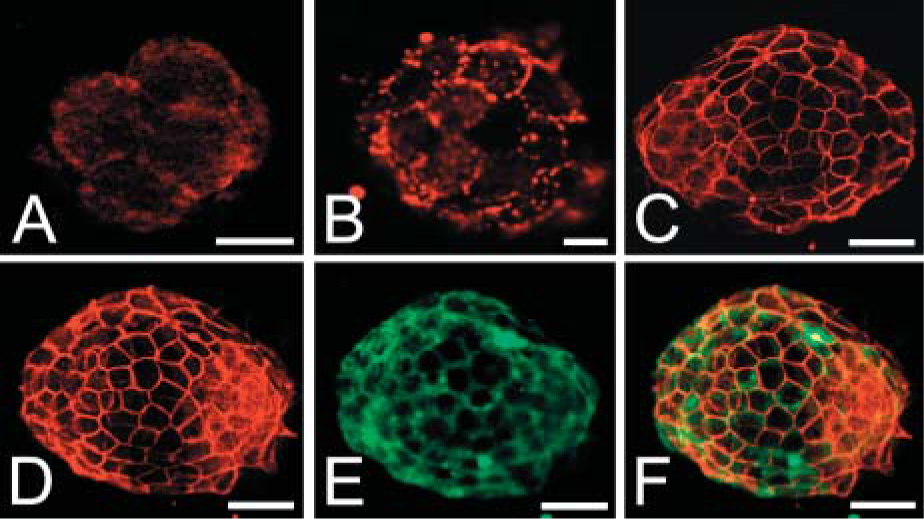
Anti-hemicentin antibody staining of mouse four- to six-cell embryo
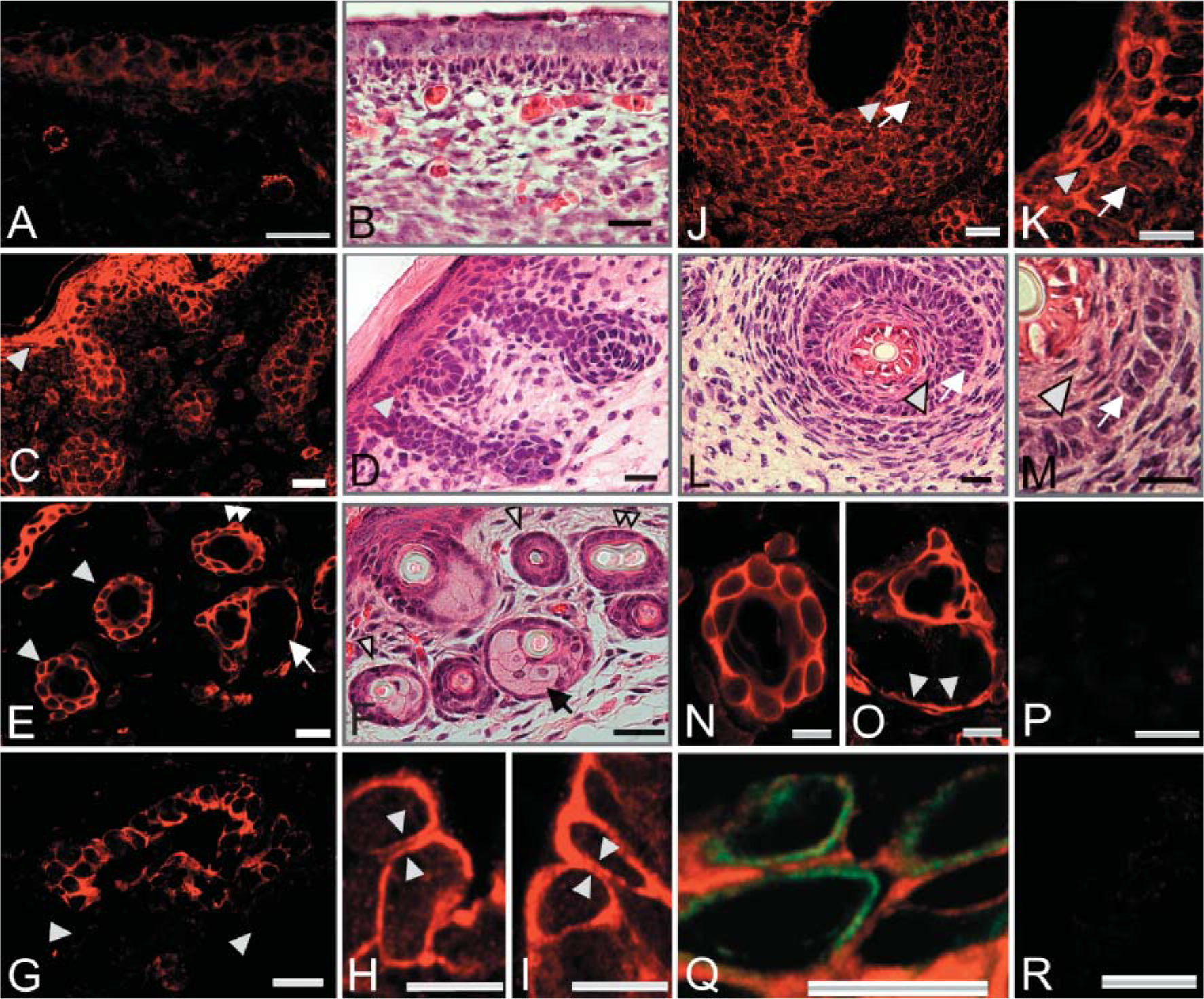
Anti-hemicentin
In sweat glands, hemicentin is detected at the periphery of epithelial cells in the secretory portion of the sweat gland tubule, but no fluorescence is associated with surrounding myoepithelial cells (arrowheads in Figures 3G-3I). Higher magnification views of secretory epithelial cells show pericellular hemicentin staining with spaces between hemicentin on neighboring cells (arrowhead pairs in Figures 3H and 3I).
In addition to its distribution in the hair follicle in mouse skin, hemicentin staining is also detected in specialized whisker hair follicles (Figures 3J-3M). On day P2, fluorescence can be detected in both inner root sheath and outer root sheath. Higher magnification views showed that the signal is mainly in pericellular ECM around squamous inner sheath epithelial cells and cuboidalouter sheath epithelial cells (Figures 3K and 3M). There is no detectable fluorescence in the center of the hair shaft.
The tongue consists of striated muscle fibers arranged in bundles that run in three dimensions wrapped in a partially keratinized epithelium. On tongue upper and lower surfaces, staining is detected around the periphery of non-keratinized epithelial cells (Figures 4A and 4B). Epithelial cells located closer to the tongue outer surfaces become keratinized, and hemicentin on epithelial cell membranes goes from being discrete lines around the periphery of cells to a series of flattened layers (Figure 4D). Hemicentin also becomes a series of flattened layers as taste bud epithelial cells keratinize (Figure 4C). Weak hemicentin staining is also detected on the surface of sensory cells in the taste bud (Figure 4C). No detectable signals are observed in taste bud neurons (Figure 4C) or in lamina propria or striated transverse and longitudinal muscle fibers (Figures 4A, 4B, and 4E).
Higher magnification views of tongue surface epithelial cells indicate that hemicentin is detected in a punctate pattern around the periphery of epithelial cells, adjacent to cell surfaces, with spaces between epithelial cells and puncta (Figure 4F).
Hemicentin Distribution in the Eye
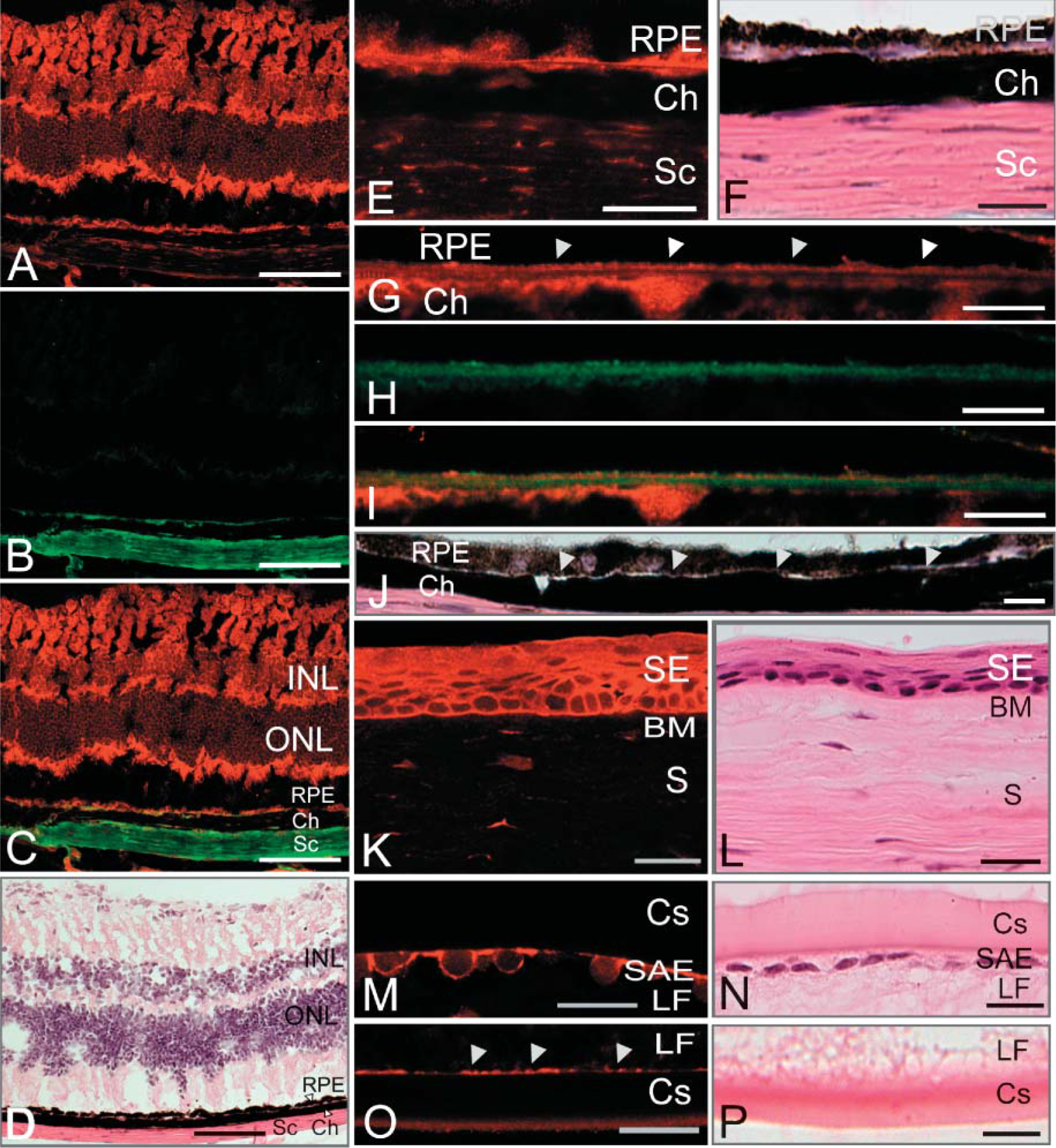
We were particularly interested in the distribution of hemicentins in the eye because human hemicentin-1 was recently implicated in age-related macular degenerative disease (Schultz et al. 2003). We compared hemicentin distribution in the eye to that of collagen XVIII, which is predominantly found in Bruch's membrane between choroid and RPE cells (Marneros et al. 2004). Hemicentin staining was detectable in the sclera and associated with the neuronal layers of the retina and Bruch's membrane (Figures 5A-5F). In Bruch's membrane, collagen XVIII is found in the central elastic layer (Marneros et al. 2004) (Figures 5H and 5I), whereas hemicentin forms a continuous, unbroken boundary distributed in the RPE and choroid basement membranes that flank the central elastic core of Bruch's membrane (Figures 5G, 5I, and 5J). In the sclera, both collagen XVIII and hemicentin are distributed in strips that run parallel to the surface of the sclera.
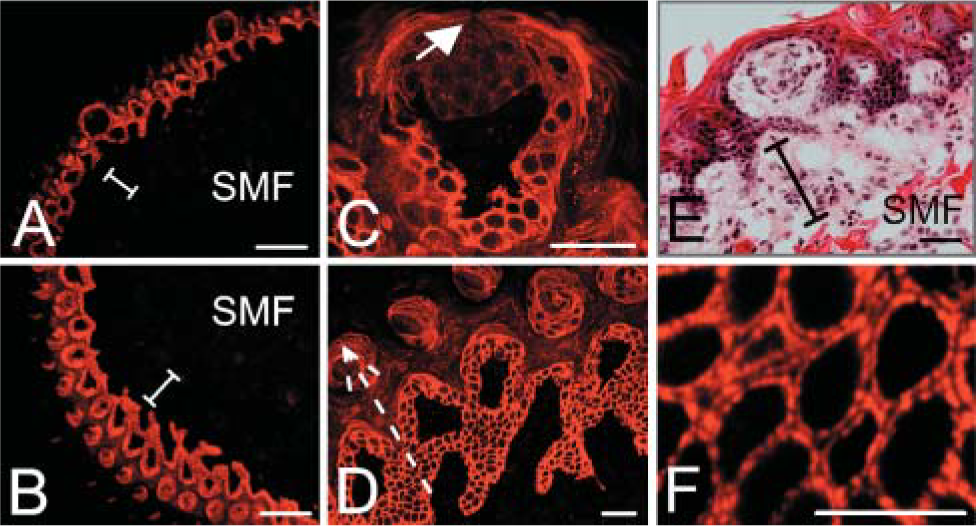
Hemicentin localization in mouse tongue epithelium. Hemicentin is detected around the periphery of non-keratinized epithelial cells on both the upper surface
The cornea consists of a non-keratinized stratified epithelium and substantia propria. The thick basement membrane between them is Bowman's membrane. In the cornea epithelium, hemicentin appears to form a lattice on the periphery of cuboidal epithelial cells closest to Bowman's membrane but gradually becomes a diffuse sheet on the squamous epithelial cells at the cornea outer surface (Figures 5K and 5L). In the substantia propria, which appears to be a histological extension of the sclera, thin wisps of hemicentin were detected.
In the lens, hemicentin staining was detected in the subcapular anterior epithelium around the periphery of lens epithelial cells but not observed in the lens capsule, a specific basement membrane enveloping the lens cell layer (Figures 5M and 5N). Lens fibers are elongated cells that extend across the entire lens from anterior epithelium, forming a major part of the lens body. At the posterior of the lens, hemicentin staining is detected along the length of the junction between lens fibers and the lens capsule basement membrane (Figures 5O and 5P).
Hemicentin Distribution in Blood Vessels
In a number of tissues, prominent hemicentin staining was observed in some, but not all, blood vessels. In the choroid of the adult eye, hemicentin was associated with the subendothelial ECM of arteriole vessels near the optic disk in addition to a number of smaller unidentified vessels (Figures 6A and 6B).
In the lungs of E17.5 mice, hemicentin is found in the pericellular ECM surrounding capillary endothelial cells and in larger blood vessels that infiltrate the walls between neighboring alveoli (Figures 6C-6F).
Approximately 9 days postcoitus, hemicentin was detected in blood vessels in the perimetrium, the outer serosal layer of the pregnant mouse uterus. Hemicentin staining was most prominent in the subendothelial ECM and the pericellular ECM of endothelial and smooth muscle cells, including what appears to be the surface of the vessel lumen (Figures 6G and 6H).
Discussion
Hemicentins are a family of secreted ECM proteins first identified in C. elegans with two orthologs in most vertebrate genomes including human and mouse (Vogel and Hedgecock 2001). In C. elegans, hemicentin is secreted by bodywall muscle and gonadal leader cells and assembles at multiple distant locations, where it forms an oriented track-like geometry whose function appears to be to constrict broad regions of cell contact into discrete line-shaped junctions. Hemicentin functions include organizing hemidesmosome-mediated attachments of mechanosensory neuron and uterine cells to the epidermis and stabilizing the plasma membranes of syncytial germ cells (Vogel and Hedgecock 2001).
Immunofluorescence
Although nothing is known about the function or distribution of the two vertebrate hemicentins, a recent study has implicated human hemicentin-1 in age-related macular degenerative disease (Schultz et al. 2003). Using an antibody designed to recognize both nematode and vertebrate hemicentins, we present evidence that hemicentins are distributed in the pericellular ECM of mouse epithelial cells in the skin, tongue, and eye and also the subendothelial ECM and pericellular ECM in some, but not all, blood vessels.
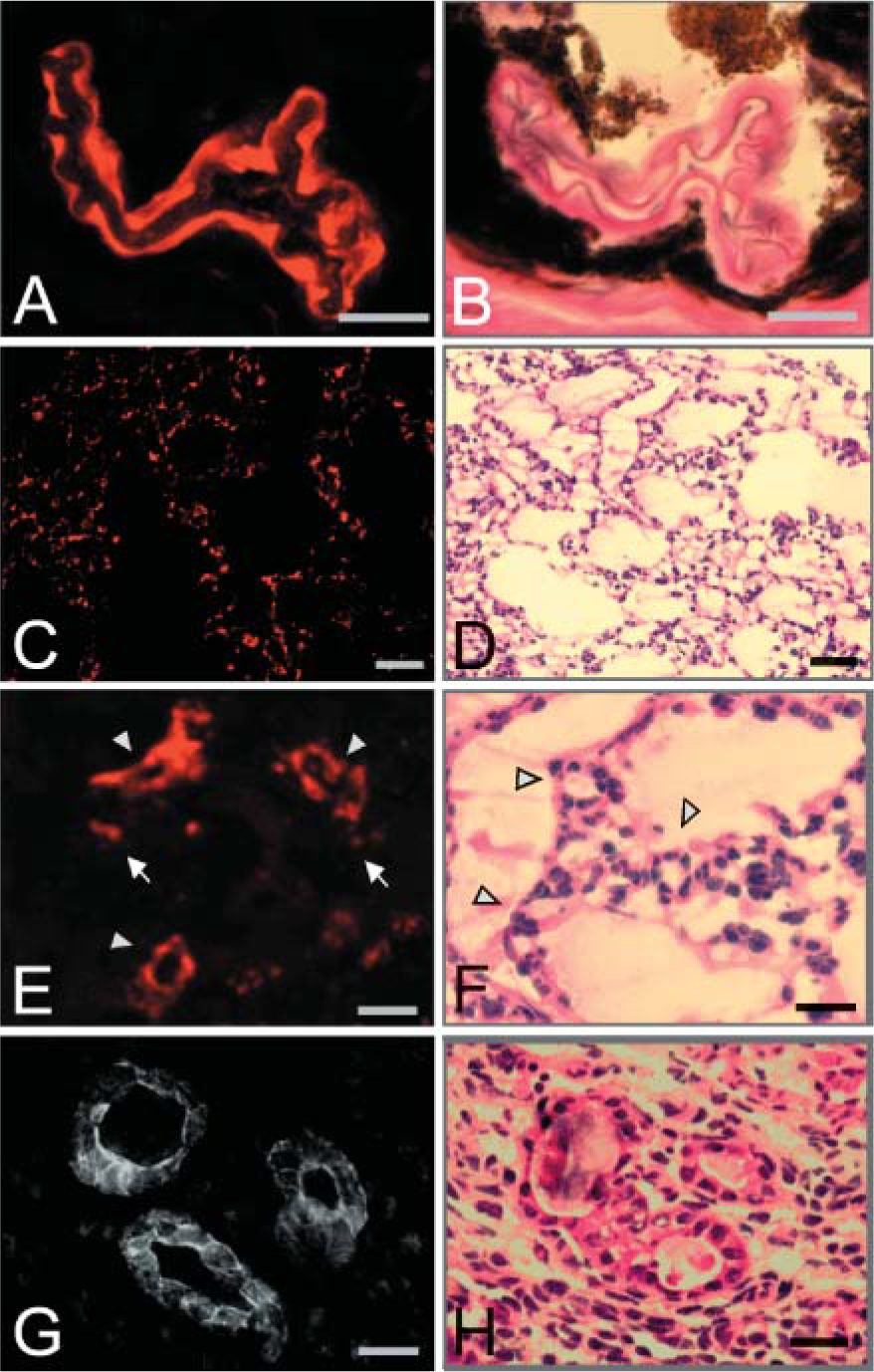
Hemicentin distribution in blood vessels.
In C. elegans, hemicentin is found in multiple regions where there is a requirement for adhesion and flexibility. Hemicentin distribution around the periphery of epithelial cells in skin and tongue suggests that hemicentins may have similar functions in vertebrates. Although epithelial cells in the eye are not subject to the same mechanical forces as those found in skin and tongue, retina, lens, and cornea are flexible tissues in the sense that they are able to accommodate modest shape changes without sustaining structural damage.
Prominent hemicentin staining is first detected in the trophectoderm, the first epithelium of developing vertebrates. Although hemicentin is located around the periphery of epithelial cells, it does not necessarily fill the ECM between cells. This is particularly noticeable as a distinct space between tracks of hemicentin staining on neighboring cells in multiple tissues, including tongue and sweat glands. Hemicentin is also absent from a number of ECMs including the ECM of Bowman's membrane, lens capsule, and dermis. The presence of hemicentin in the pericellular ECM of epithelial cells, but not in most ECMs, is consistent with hemicentin distribution in C. elegans where hemicentin is found in distinct locations at epithelial and other cellular junctions but is not found in most ECMs. This suggests that the function of hemicentins in the organization of specific epithelial cell junctions may be conserved in different organisms.
Fibulins are a family of five extracellular glycoproteins associated with basement membranes and elastic fibers in vertebrates (Argraves et al. 2003; Timpl et al. 2003). Defects in fibulin family members result in connective tissue disorders that include macular degenerative diseases and cutis laxa (Stone et al. 1999,2004; reviewed in Chu and Tsuda 2004). Fibulin-1 is a prominent component of skin, lung, and cardiovascular tissue and is essential for the morphology of endothelial cells lining capillary walls and the integrity of small blood vessels (Kostka et al. 2001). The recent demonstration that assembly of fibulin and hemicentin orthologs are codependent in C. elegans (Muriel et al. 2005) suggests that it may be interesting to determine whether hemicentin may coassemble with fibulin-1 and possibly other fibulin family members in vertebrate tissues.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health Grant GM-65184.
We thank Drs. Alexander Marneros and Bjorn Olsen for antibodies to Col XVIII and Dr. P. Koch for desmocollin-3 antibodies. We also thank Drs. Shengyun Fang, Gloria Hoffman, and Mon-Li Chu for helpful discussions.
