Abstract
Extensive remodeling of the extracellular matrix (ECM) occurs in inflammatory tissues. The celiac lesion in the small intestine is characterized by inflammation accompanied by profound morphological alterations. We used immunohistochemistry to determine the distribution of laminin, fibronectin, and tenascin isoforms in small intestinal biopsies of untreated patients with celiac disease. In normal mucosa, the distribution of laminin isoforms defines three epithelial basement membrane (BM) zones. We found that the organization of these zones was maintained in the celiac mucosa. Thus, components of laminin-5 (α3 and β3) were found in the surface epithelial BM, laminin α2 chain was found selectively at crypt bottoms, and laminin α5 chain was the sole α-type chain in middle crypt BMs. Likewise, the distribution of fibronectin and tenascin resembled that of the normal gut. The organization of pericryptal fibroblasts and lamina propria smooth muscle strands, as defined by immunostaining for α-smooth muscle actin, also remained unchanged in the celiac mucosa. Unexpectedly, major ECM changes were not detected in the celiac lesion.
C
Profound alterations in extracellular matrix (ECM) molecules and their cell surface receptors have been found in a number of inflammatory processes. The characterization of such alterations is important not only because they reflect the disease processes but because cell–ECM interactions are regulators of these pathological sequences (Smith et al. 1997).
Accompanied by extensive immunopathology, the celiac mucosa displays marked morphological alterations (Marsh and Crowe 1995). We hypothesized that these processes would be reflected in the distribution of ECM molecules. To investigate this, we compared the distribution of laminin (Ln), fibronectin (Fn), and tenascin (Tn) polypeptides in active celiac intestinal mucosa. Lns are important in this respect because they influence enterocyte differentiation (Beaulieu 1997). In the small intestinal epithelial basement membrane (BM), different Ln chains show differential distribution along the crypt–villous axis (Beaulieu and Vachon 1994; Simon–Assmann et al. 1994; Perreault et al. 1995; Leivo et al. 1996). Fns and Tns were studied because their distribution is modulated in many inflammatory diseases. Furthermore, the regulation of Fn and Tn expression may be important because in the normal intestinal mucosa they are found in opposing gradients along the crypt–villous axis (Quaroni et al. 1978; Thor et al. 1987). In fact, Tn has been proposed to be important for cell shedding at the villous tip (Probstmeier et al. 1990).
In this study we compared the distribution of Ln, Fn, and Tn isoforms and organization of mucosal smooth muscle components in celiac disease and in normal human child and adult small intestinal mucosa by immunohistochemistry.
Materials and Methods
Patients and Controls
Small intestinal biopsy specimens from 13 patients with untreated celiac disease were available for the study. Nine were pediatric patients aged 0.9–10.3 years. Indications for biopsy were gastrointestinal complaints and/or growth failure, and increased levels of endomysium or reticulin antibodies. Four specimens were from adult healthy hospital personnel aged 39.5–48.9 years, who were found to have increased levels of endomysium antibodies in a screening study (Kolho et al. unpublished data). All had had minor gastrointestinal complaints and had an increased level of IgA and/or IgG anti-gliadin antibodies. Biopsy specimens from pediatric patients were taken with a Crosby–Kugler biopsy device at the level of ligamentum Treitz or distal to it. Adult patients were biopsied with a gastroscope from the distal duodenum. Eleven of the biopsies showed total villous atrophy and two had a villous length of <180 μm. All had an elevated density of intraepithelial lymphocytes and γ/δ+ cells. All 13 patients had a favorable clinical response to a gluten-free diet.
Four controls were specimens from pediatric patients aged 3.2–13.8 years. They were taken from children with minor gastrointestinal complaints. An additional indication for biopsy was an increased level of IgG anti-gliadin antibodies in three patients. IgA–endomysium and IgA–gliadin antibodies, mucosal morphology, and density of intraepithelial lymphocytes and γ/δ+ cells were all normal. Four samples of normal adult small intestine were obtained from surgical specimens of the Jorvi Hospital (Espoo, Finland). Both biopsy and surgical samples were immediately frozen in liquid nitrogen and stored at −80C until use.
The patient samples used in this study represented active celiac lesions with typical mucosal morphology and marked infiltration of surface epithelium with both α/β and γ/δ TCR+ lymphocytes (Savilahti et al. 1997). The immunohistochemical results were consistent throughout the samples, except for the distribution of Ln-5 components (see below). Both pediatric and adult celiac and control patients were included in the study. The results were consistent in children and in adults.
Mucosal Histology and Density of Intraepithelial Lymphocytes
MAbs TCR 1 (T Cell Diagnostics; Woburn, MA) and F1 (T Cell Diagnostics) were used at a dilution of 1:100, and anti-Leu4 (anti-CD3; Beckton–Dickinson, Mountain View, CA) at a dilution of 1:400. Eight-μm cryostat sections were incubated with MAbs and then treated with 0.5% hydrogen peroxide for 20 min to block endogenous peroxidase activity.
The bound MAbs were detected with a Vectastain Elite ABC kit (PK-6102; Vector Laboratories, Burlingame, CA). Mucosal histology and the density of intraepithelial lymphocytes were evaluated independently from the other results of the study. The number of stained cells was counted as cells/mm of epithelium with a light microscope through a calibrated graticule (0.053 mm) at ×100 magnification (Savilahti et al. 1997).
Ethical Considerations
Biopsy specimens from pediatric patients were taken because of clinical indications. From the adults, informed consent was obtained for the screening study of endomysium antibodies in hospital personnel and subsequently for gastroduodenoscopy. The screening study has been approved by the Ethical committee of the Medical Department of Helsinki University Central Hospital.
Antibodies
The following MAbs were used to localize ECM proteins: Ln α2 chain (clone 5H2) (Leivo and Engvall 1988), Ln α3 (BM-2) (Rousselle et al. 1991), Ln α5 (4C7) (Engvall et al. 1990), Ln β1 (4E10 and LnB1) (Engvall et al. 1990; Virtanen et al. 1997), Ln β2 (C4) (Hunter et al. 1989), Ln β3 (6F12) (Marinkovich et al. 1992), Ln γ1 (2E8) (Engvall et al. 1990), extracellular domain A-containing fibronectin (EDA-Fn; 52DH1) (Vartio et al. 1987), extracellular domain B-containing fibronectin (EDB-Fn; BC-1) (Carnemolla et al. 1989), oncofetal Fn (FDC-6) (Matsuura and Hakomori 1985), Tn (100EB2, reacting with the fourth and fifth Fn-like repeats and thus detecting all Tns) (Howeedy et al. 1990), and Tn-A1A4 (BC-2 reacting with Tn isoforms containing repeats A1 and A4) (Balza et al. 1993). Other MAbs were used to detect α-smooth muscle actin (1A4; Sigma Chemical, St Louis, MO) (Skalli et al. 1986) and proliferating cells (Ki-67; Serotec, Bichester, UK) (Brown and Gatter 1990). Polyclonal antisera against Ln-α1 chain (Tiger et al. 1997), Ln-1 (1141) (Liesi et al. 1983), Ln-5 (4101) (Rousselle et al. 1991), and desmin (Virtanen et al. 1986) were used. Mucosal capillaries were identified using Ulex europaeus lectin-I (UEA-I; Vector Laboratories, San Mateo, CA) (Holthöfer et al. 1982).
Indirect Immunofluorescence Microscopy
Frozen sections were cut at 5 μm and fixed in acetone at −20C for 10 min. The sections were then exposed to the primary and subsequently to the secondary antibodies at room temperature for 30 min. The secondary antibodies were fluorescein isothiocyanate-coupled goat anti-mouse IgG, fluorescein isothiocyanate-coupled goat anti-rat IgG, and tetramethylrhodamine-coupled goat anti-rabbit IgG sera (all from Jackson Immunoresearch; West Grove, CA). Controls were carried out omitting the primary antibodies. The sections were embedded in sodium-veronal/glycerol buffer (1:1; pH 8.4), or Mowiol in the case of lectins, and viewed with a Leitz Aristoplan microscope using appropriate filters. Intensity of the immunoreaction was evaluated on a 4-point scale: –, negative; +, weak; + +, moderate;
Summary of the distribution of Ln chains in normal and celiac intestinal mucosaa
a–, negative; +, weak; ++, moderate; + + +, strong.
Results
The results are summarized in Tables 1 and 2 and in Figure 5.
Laminins in Epithelial BMs in Celiac Mucosa
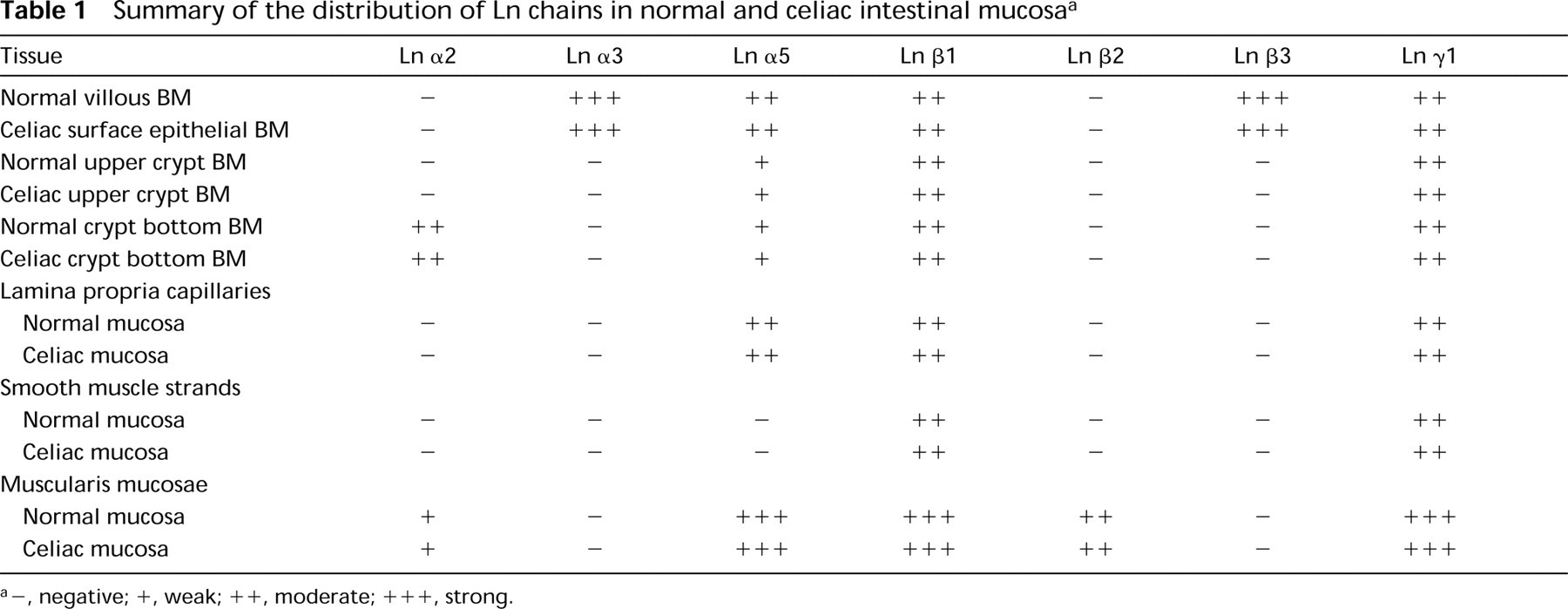
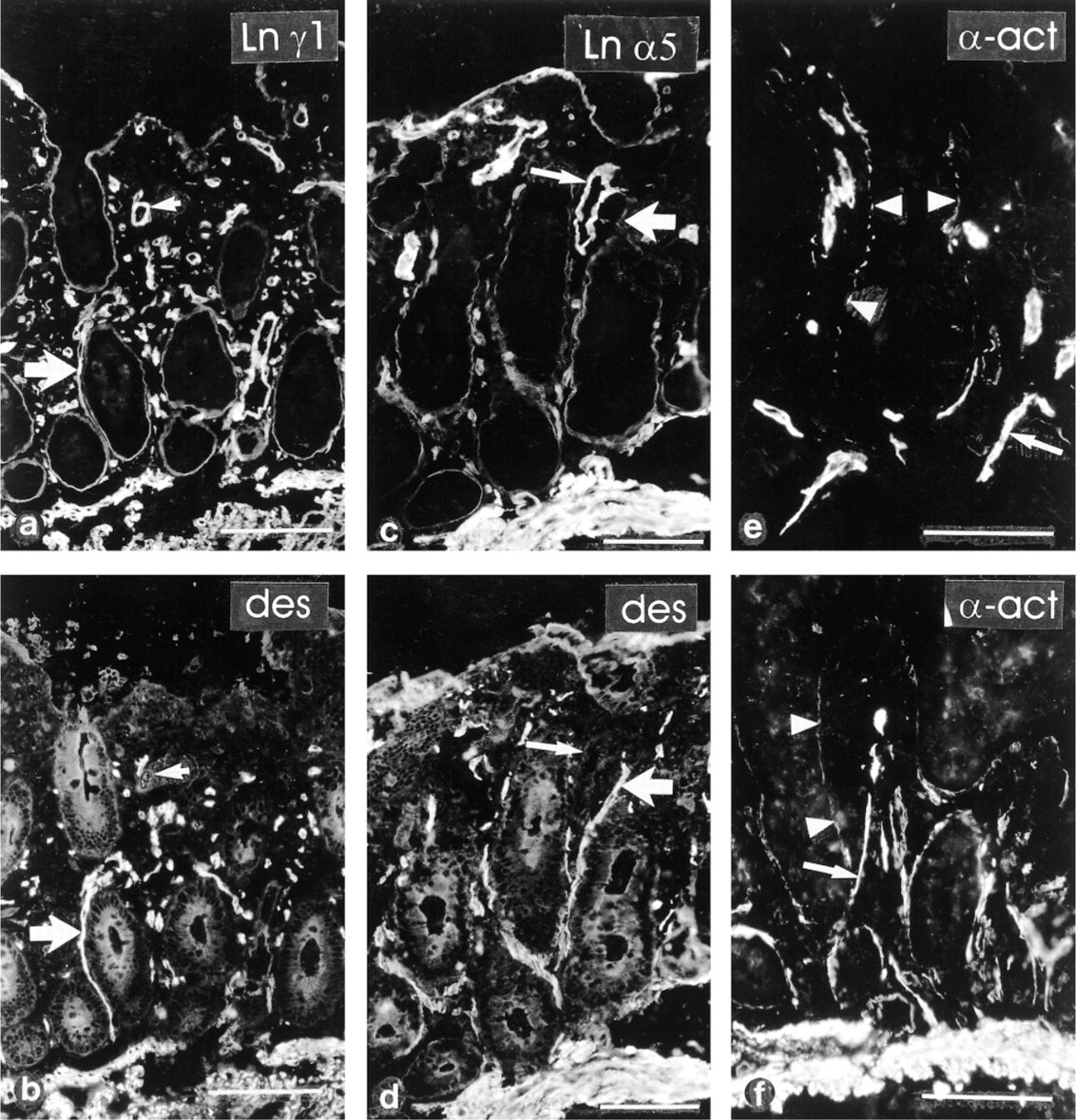
Immunoreactivity for Ln α5 (Figure 1a), β1 (Figure 1b), and γ1 (Figure 4a) chains, components of Ln-10, was detected in a linear formation along BMs of surface epithelium and the full length of the crypts. Immunoreactivity for Ln α5 chain was weaker along the crypt than the surface epithelial BMs. MAb to Ln α2 (Figure 1c) reacted selectively with BM surrounding crypt bottoms, excluding middle and upper crypt and surface epithelial BMs. Immunoreactivity for Ln α1 (Figure 1d) or β2 chains (not shown) was not detected in the epithelial BM or lamina propria. Occasional subepithelial myofibroblasts, also immunoreactive for α-actinin and desmin, reacted with Ln α3, α5, β1 (Figure 1b), and γ1 MAbs.
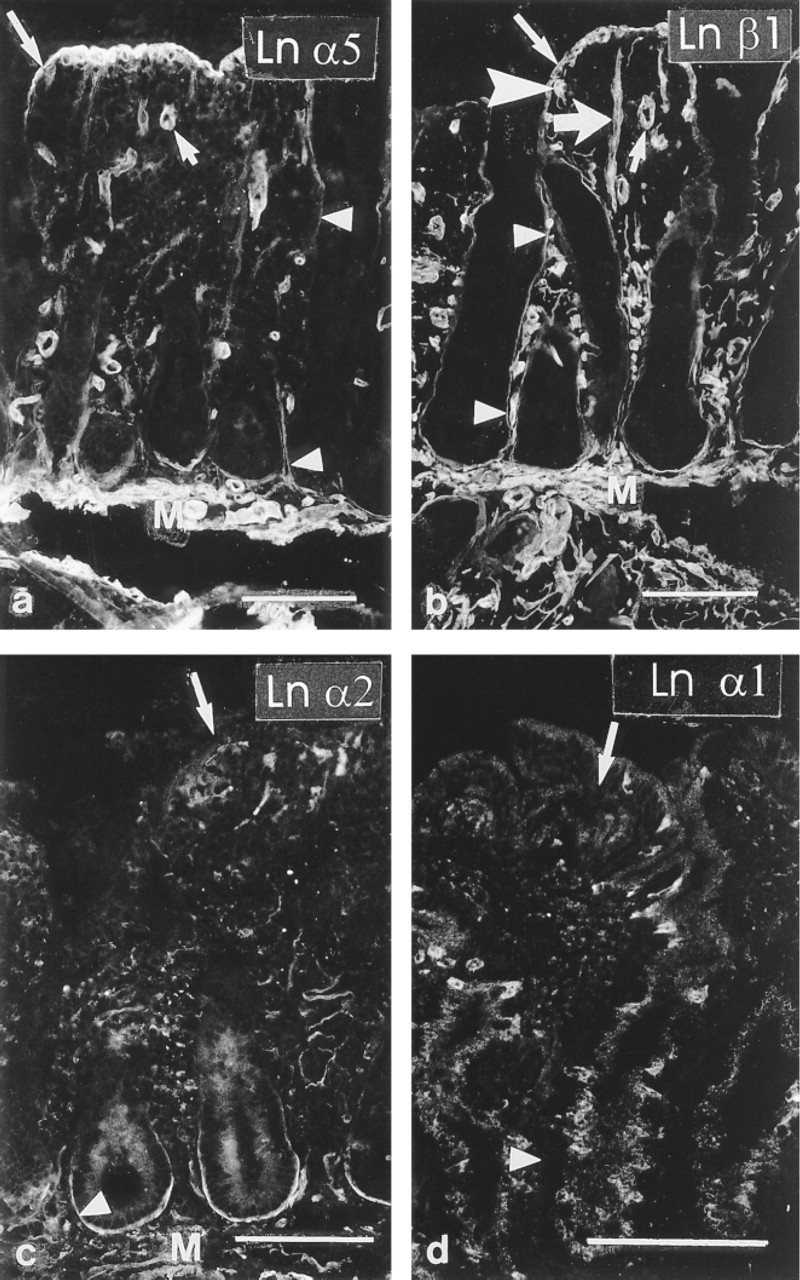
MAbs detecting Ln β3 and α3 chains (Figure 2a and 2b) and the antiserum against Ln-5 (Ln α3β3γ2; 2c and 2d) reacted distinctly with the BM of surface epithelium, with weak immunoreactivity sometimes extending to upper crypt BMs. In tangential sections, the immunoreactivity was often spotty (Figure 2b). In double immunostaining experiments, there was no overlap of the proliferative cell compartment, as defined by immunoreaction for the MAb Ki-67, and the distribution of Ln-5 (Figure 2c and 2d). In two of the 13 samples, weak immunoreactivity using Ln-5 antiserum and Ln α3 and β3 chain MAbs was detected in crypt BMs, including crypt bottoms, in addition to the surface epithelial BM.
Fibronectins and Tenascins in Celiac Mucosa
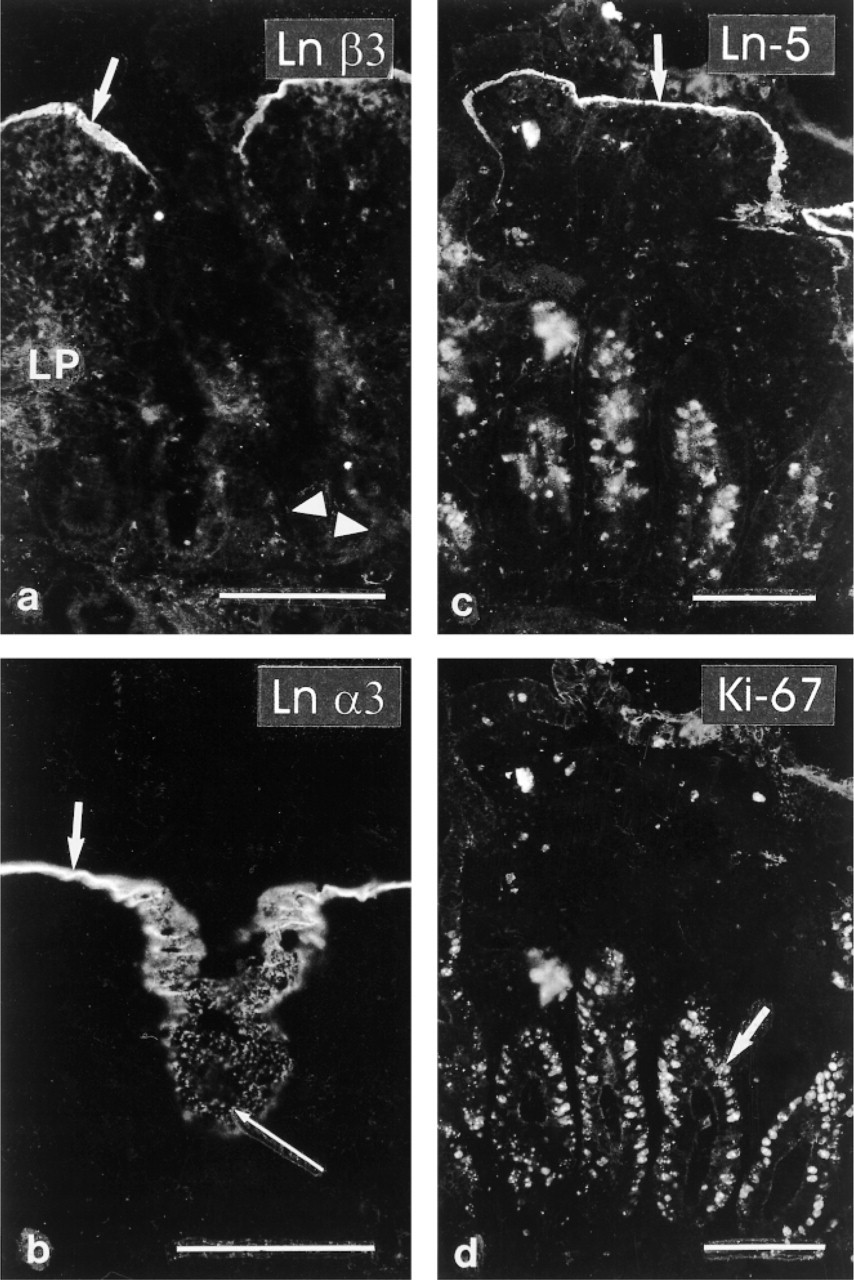
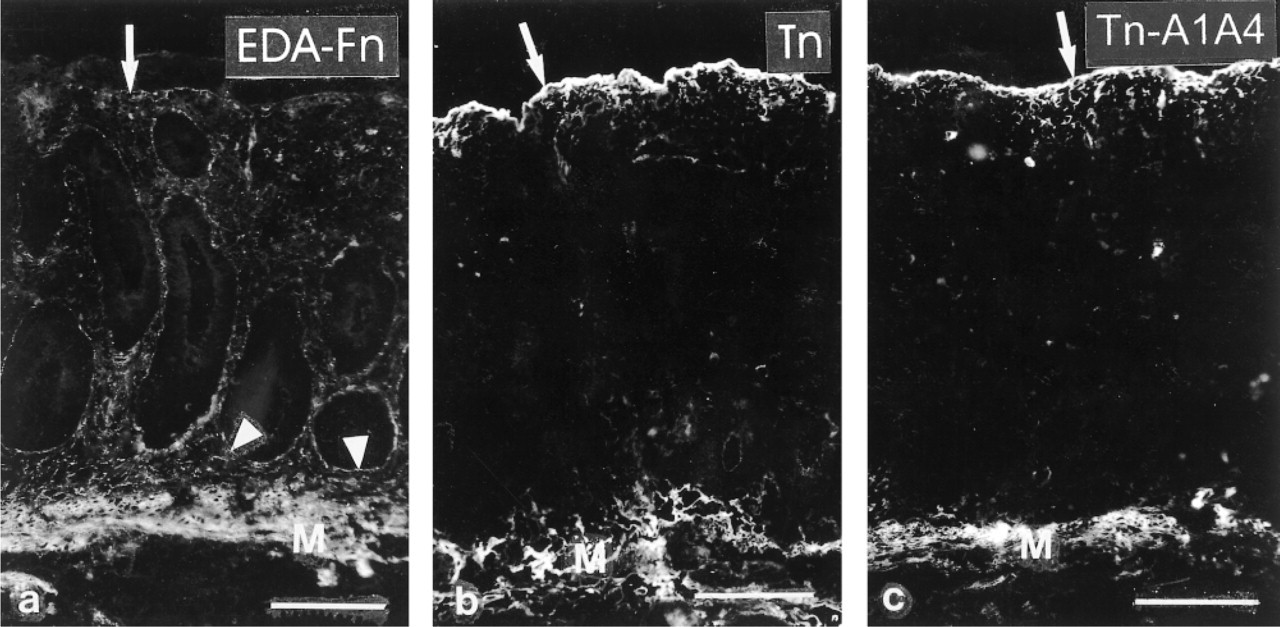
An MAb recognizing EDA-Fn (Figure 3a) reacted with crypt BMs but inconsistently with upper crypt and surface epithelial BM regions. In addition, a weak fibrillar meshwork was seen throughout the lamina propria mesenchyme. The EDB-Fn and Onc-Fn MAbs (not shown) detected sparse weakly immunoreactive cells in the lamina propria but otherwise failed to react with epithelial BMs or the lamina propria stroma.
Both anti-Tn MAbs (Figure 3b and 3c) reacted distinctly with the BM zone of surface epithelium as well as upper crypts, but failed to reveal immunoreactivity in the stroma of lamina propria. No distinct differences in the distribution of the different Tn splicing isoforms were noted.
Distribution of Fn and Tn isoforms in normal and celiac small intestinal mucosaa

a–, negative; +, weak; + +, moderate; + + +, strong.
Frozen sections of celiac intestinal mucosa. (
Mucosal Smooth Muscle, Pericryptal Fibroblasts, and Capillaries
Ln β1 (Figure 1b) and γ1 (Figure 4a and 4b) chain MAbs reacted with strand-like formations within the lamina propria mesenchyme in the celiac patient samples. These strands also reacted with desmin antiserum (Figures 4a-4d) and with an MAb to α-smooth muscle actin (Figure 4e), but not with UEA-I lectin (not shown) that detects endothelial cells, indicating that the strands contain smooth muscle and are distinct from capillaries. In normal small intestine, smooth muscle strands extending from the muscularis mucosae into the villous mesenchyme reacted similarly (Figure 4e and 4f), suggesting that the strands were corresponding structures. Immunoreactivity for Ln α1, α2, α3, α5 (Figure 4c and 4d), β2, or β3 chains was not found in the strands. Slight immunoreactivity for α-smooth muscle actin located at the epithelial–mesenchymal interface extended all the way from crypt bottoms to the surface epithelium, identifying pericryptal fibroblasts (Figure 4e and 4f). Muscularis mucosae displayed distinct immunoreactivity for Ln α5 (Figure 4c and 4d), β1 (Figure 1b), β2 (not shown), and γ1 (Figure 4a and 4b) chains as well for EDA-Fn (Figure 3a) and both Tn MAbs (Figure 3b and 3c). However, antibodies against Ln α1, α3 and β3 chains and against EDB- and Onc-Fn did not react, and Ln α2 (Figure 1c) chain MAb revealed only scattered immunoreactivity in muscularis mucosae.
Celiac mucosa. (
Capillaries of lamina propria, identified by double labeling with UEA-I, displayed immunoreactivity for components of Ln-10: Ln α5 (Figure 1a and 4c), β1 (Figure 1b), and γ1 (Figure 4a), but not for α1, α2, α3, β2, or β3 chains.
Discussion
Extensive remodeling of the ECM, detectable by immunohistochemistry, is often found in inflammatory lesions, e.g., in inflammatory bowel disease (MacDonald et al. 1990; Riedl et al. 1992), gastritis associated with Helicobacter pylori infection (Tiitta et al. 1994), periodontal disease (Haapasalmi et al. 1995), glomerulonephritis (Border and Noble 1994), wound healing (Raghow 1994), asthma (Laitinen et al. 1997), and gastrointestinal neoplastic infiltration (Riedl et al. 1992; Hauptmann et al. 1995).
Celiac mucosa. (
The morphology of the celiac lesion is characterized by crypt hyperplasia and villous atrophy, coupled with an increase in the volume of lamina propria (Mäki and Collin 1997). Extensive activation of immunologically active cells, the cytokine network, and other mediators, although not yet fully elucidated, occurs in celiac disease (reviewed in Marsh and Crowe 1995; Przemioslo and Ciclitira 1996). With respect to the ECM, increased deposition of collagen and thickening of the epithelial BM (Araya and Walker–Smith 1975), as well as tissue edema (Marsh and Hinde 1986), have been detected in mucosa of celiac disease patients. Furthermore, increased mRNA for matrix metalloproteinases was found in intestinal biopsy samples from patients with celiac disease (Daum et al. 1999).
This study addresses the question of whether ECM alterations can be detected in the celiac lesion by immunohistochemistry. One pertinent limitation of this kind of study is that it gives us only a snapshot of a dynamic process of ECM turnover. However, immunohistochemical changes can be detected in the other above-mentioned inflammatory lesions, and we expected to find likewise in the celiac mucosa. Given the multitude of known ECM molecules, another obvious limitation is that they cannot all be covered in a single study. Here we chose to investigate the distribution of Lns, Fn, and Tn because they are all differentially expressed along the crypt–villous axis in the normal gut, and have been suggested to influence enterocyte differentiation (see above). Another family of ECM molecules that requires further study in this context is that of the collagen IV isoforms. Their expression has been shown to be regulated independently of laminins in both normal and neoplastic gut (Perreault et al. 1998; Hewitt et al. 1997).
The main finding of this study is that untreated celiac intestinal mucosa displays a surprisingly normal distribution of ECM molecules.
It should be noted that MAb 4C7, formerly believed to recognize the Ln α1 chain, was recently shown to recognize the human Ln α5 polypeptide (Tiger et al. 1997; Church and Aplin 1998). Ln isoforms containing this α-type chain are therefore found ubiquitously throughout normal and celiac intestinal epithelial BM. In accordance with earlier studies using in situ hybridization suggesting sparse distribution of the Ln α1 mRNA in many tissues (Ekblom 1996), we found no Ln α 1 protein in normal or celiac small intestinal mucosa.
The Epithelial Basement Membrane
In the normal small intestinal epithelium, the exclusive localization of specific Ln isoforms defines three zones within the epithelial BM of the crypt–villous axis (Beaulieu and Vachon 1994; Leivo et al. 1996). (I) The villous zone is characterized by expression of Ln α3 and β3 chains, components of Ln-5. (II) The middle and upper crypt BMs contain α5 as their sole α-type chain, lacking both α2 and α3. (III) The α2 chain is found exclusively at the crypt bottoms. These three zones were also identified in the celiac epithelium (Table 1; Figure 5). The BM at the bottom of the crypts was characterized by immunoreactivity for Ln α2, implying the presence of Ln-2 (α2β1γ1), and BM of surface epithelium was defined as homologous to that of normal villous epithelium by its immunoreactivity for α3 and β3, components of Ln-5. The expansion of the crypt compartment that occurs in the celiac lesion (Mäki and Collin 1997) appears to correspond to elongation of Zone II-type BM, and presumably also the corresponding epithelium.
(
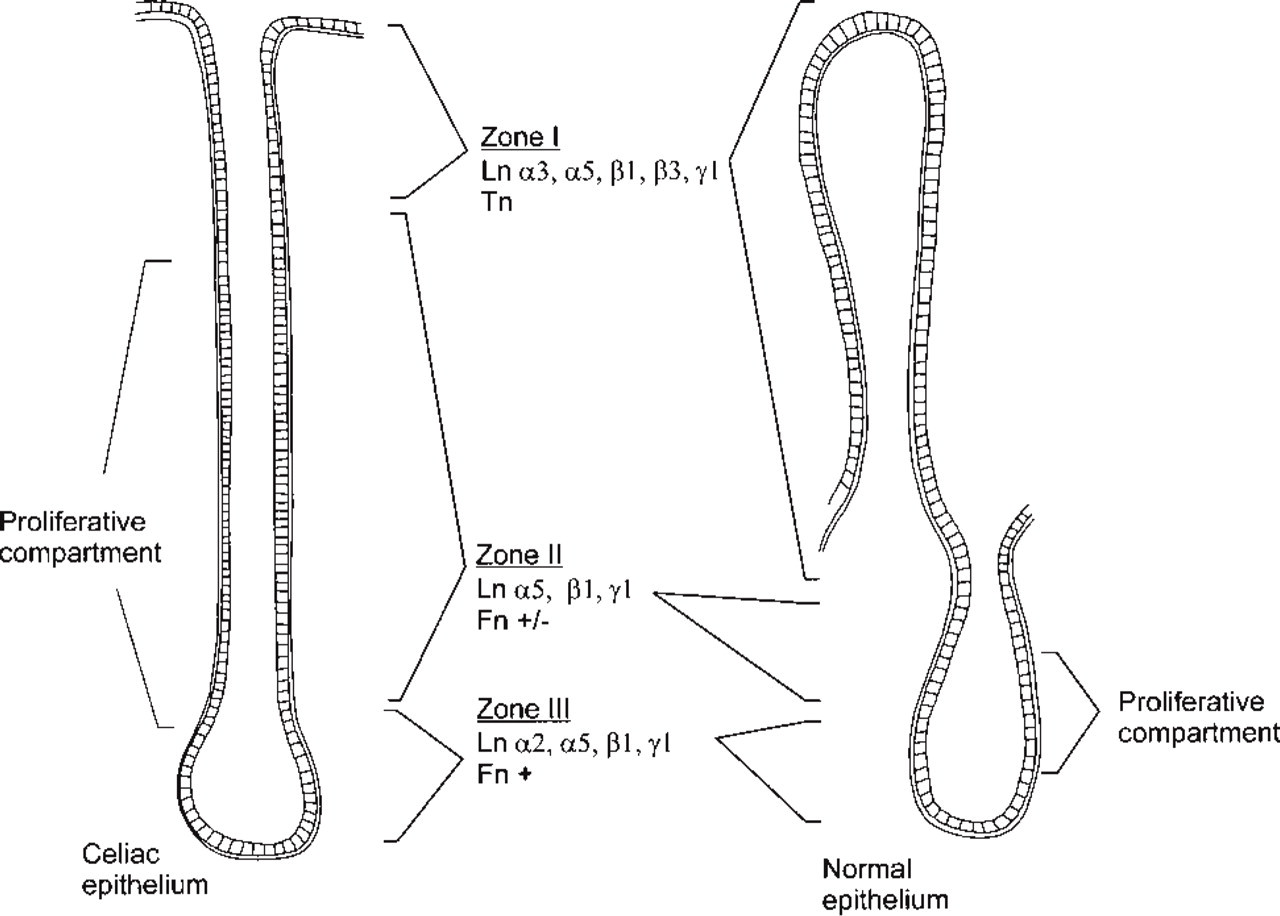
Diagram summarizing the distribution of ECM proteins in celiac (left) and normal (right) intestinal mucosa. Only the epithelial BMs are depicted, and the distribution of ECM proteins in lamina propria is omitted for clarity.
The celiac mucosa is characterized by hypertrophy of crypts and expansion of the proliferative cell compartment. However, by double immunostaining using MAb Ki-67 we showed that the proliferative cell compartment did not extend to the surface epithelial BM zone that contains Ln-5. In addition, the organization of the celiac epithelium resembles that of the normal gut (Leivo et al. 1996). Samples from two patients, however, deviated from the pattern: weak immunoreaction for Ln α3 and β3 chains and Ln-5 was detected also in crypt BMs.
Normal gut mucosa displays a striking gradient of increasing Tn expression from crypt bottom to villous tip, apparently involved in cell shedding at the villous tip (Thor et al. 1987; Probstmeier et al. 1990), contrasting with the opposite gradient of Fn (Quaroni et al. 1978). Even this organization is maintained in the celiac mucosa (Table 2). Both Tn MAbs reacted only at the BM zone of the surface epithelium, whereas crypt BM and lamina propria were negative. Also parallelling the normal gut, immunoreactivity for EDA-Fn was detected in crypt BM but less was found in the surface epithelial BM, which corresponds to the villous compartment of normal mucosa. The orderly Fn and Tn expression is striking, given that increased synthesis of these molecules in particular accompanies a wide variety of inflammatory and reactive tissue processes (Koukoulis et al. 1991; Border and Noble 1994; Hauptmann et al. 1995; Laitinen et al. 1997).
The specialization of the intestinal BM along the crypt–villous axis is believed to reflect and modulate enterocyte differentiation (Beaulieu 1997). Our results imply that despite the extensive morphological alterations of the mucosa in celiac disease, at least these differentiative processes remain intact.
Lamina Propria
Two other aspects of lamina propria tissue architecture merit mention: the smooth muscle scaffolding of small intestinal villi and the pericryptal fibroblastic sheath. In celiac mucosa, we detected abundant strands of desmin and α-smooth muscle actin-positive cells extending from the muscularis mucosae towards the mucosal surface, similar to the smooth muscle bundles of normal villi (Joyce et al. 1987). These structures displayed distinct immunoreactivity for the Ln β1 and γ1 chains, but the α-type chain could not be identified using α1, α2, α3, or α5 antibodies. The Ln α4 chain, which has been found in murine intestinal mesenchyme and specifically in mucosal smooth muscle (Lefebvre et al. 1999), may be the unidentified α-type chain also found in human intestine.
In the normal gut, a pericryptal fibroblastic sheath envelops crypts, extending to the villous tips at the epithelial–mesenchymal interface. It has been suggested that these cells control epithelial cell proliferation and immunological functions (Göke et al. 1997), and may therefore be important with respect to celiac disease. Our results imply that the morphological organization of lamina propria smooth muscle strands and of the pericryptal fibroblastic sheath, as detected by indirect immunofluorescence microscopy for α-smooth muscle actin, remains intact in the celiac mucosa.
In summary, we found that the spatially restricted distribution of Lns, Fn, and Tn along the crypt-villous axis and the organization of the lamina propria smooth muscle scaffolding and of the pericryptal fibroblastic sheath resemble those seen in normal intestinal mucosa. Surprisingly, our results suggest that despite the profound changes in morphology coupled with the active immunopathological processes, major ECM changes do not occur in celiac disease.
Footnotes
Acknowledgments
We are indebted to Eva Engvall, Sen-itiroh Hakomori, Joshua Sanes, and Luciano Zardi for kind gifts of antibodies. The skillful technical assistance of Hannu Kamppinen, Reijo Karppinen, Marja-Leena Piironen, Sirkku Kristianssen, and Outi Rauanheimo is acknowledged.
