Abstract
We studied the localization of laminin α1, α2, α3, α5, β1, β2, and γ1 chains and extradomain A- (EDA), EDB-, and oncofetal fibronectin by immunohistochemistry in human placental villi during placental development. The laminin α2, α5, β1, β2, and γ1 chains were detected in the trophoblastic basement membrane (BM) at all stages of gestation, suggesting the presence of laminin-2, −4, −10, and −11 trimers. The laminin α1 chain was selectively found at sites where the villous BM was in contact with proliferating cells in trophoblastic islands or columns. EDA-Fn, but not other Fn isoforms, was found in the trophoblastic BM during the first trimester. The laminin α2, β1, β2, and γ1 chains were detected in the villous stroma and capillaries throughout placental development, while the laminin α5 chain emerged distinctly during development. Extensive EDA-Fn immunoreactivity was found in first-trimester villous stroma, but distinctly fewer Fn isoforms were seen in the villous stroma during the later stages of gestation. Our results also suggest that, during the formation of new villi, laminins are not found in trophoblastic sprouts before the ingrowth of the villous mesenchyme. Rather, laminins may be deposited at the villous epithelial–mesenchymal interface. Furthermore, the results show that distinct changes occur in the localization of various laminin and Fn isoforms during the maturation of villous trophoblastic and capillary BMs.
Keywords
B
Fibronectin is a ubiquitous component of the extracellular matrix (ECM). Fibronectin is found as an intrinsic component in some BMs during development (Laitinen et al. 1991), but it is lacking in most developing and mature BMs. Isoforms containing the extradomain A (EDA) and EDB are produced by alternative splicing of mRNA (Ffrench–Constant 1995). Furthermore, alternative glycosylation within the CSIII domain creates an immunologically defined epitope, oncofetal fibronectin, which is often found in developing and malignant tissues (Matsuura and Hakomori 1985).
Earlier, we have studied the distribution of integrins and ECM molecules in the developing and term human placenta (Virtanen et al. 1988; Korhonen et al. 1991; Korhonen and Virtanen 1997). In this work we have extended these studies to the distribution of the various laminin and fibronectin isoforms in human placental villi.
Materials and Methods
Tissues
Sixteen human placentas (seven 8–10 weeks, five 12–20 weeks, and four 38–40 weeks of gestation) were aquired from spontaneous abortions due to rupture of fetal membranes, from legal abortions performed for psychosocial indications, or from normal deliveries at the Jorvi Hospital, Espoo, Finland. The tissues were frozen in liquid nitrogen and stored at −70C until use.
Antibodies
The following MAbs and antisera to laminin and fibronectin isoforms were used in the study: α1 (clone EB7, Virtanen et al. 2000), α2 (clone 5H2, Leivo and Engvall 1988), α3 (clone BM-2, Rousselle et al. 1991), α5 (clone 4C7, Engvall et al. 1986; Tiger et al. 1997) β1 (clone 4E10, Wewer et al. 1983; Engvall et al. 1990; LnB1, Virtanen et al. 1997), β2 (clone C4, Hunter et al. 1989), β3 (clone 6F12, Marinkovich et al. 1992), γ1 (clone 2E8, Engvall et al. 1986, 1990), 1141 (a rabbit antiserum to EHS laminin; Liesi et al. 1983), fibronectin (clone 52BF12 recognizing all fibronectins; Vartio et al. 1987); EDA-fibronectin (EDA-Fn, clone 52DH1; Vartio et al. 1987); EDB-fibronectin (EDB-Fn, clone BC-1 recognizing a neoepitope found in fibronectin that contains the EDB; Carnemolla et al. 1992), and Onc-fibronectin (Onc-Fn, clone FDC-6; Matsuura and Hakomori 1985). Anti-cytokeratin 19 serum (Leoncini et al. 1990) was used to identify trophoblastic cells in double immunostainings and serial sections. Polyclonal antisera to desmin (Virtanen et al. 1986) and vimentin (Virtanen et al. 1981) were used to identify the villous stroma, and capillaries were localized using Ulex europaeus agglutinin (UEA-I; Vector Laboratories, San Mateo, CA; Holthöfer et al. 1982). The optimal dilutions of the antibodies and lectins were determined by using a panel of human normal tissues.
Immunohistochemistry and Microscopy
Five-μm frozen sections were fixed in acetone at −20C for 10 min. For indirect immunofluorescence microscopy, the sections were incubated with the primary antibody and subsequently with the secondary antibodies (fluorescein isothiocyanate–coupled goat anti-mouse IgG and tetra-methylrhodamine-coupled goat anti-rabbit IgG sera; Jackson Immunoresearch, West Grove, CA) at room temperature for 30 min. Negative controls, in which sections were exposed only to the secondary antisera, did not display unspecific immunoreactivity. The specimens were embedded in sodium-veronal/glycerol buffer (1:1; pH 8.4), or Mowiol in the case of lectins, and were examined with a Leitz Aristoplan microscope equipped with appropriate filters.
The distribution of the various laminin and fibronectin chains was evaluated by indirect immunofluorescence from sets of serial sections of the placental samples. Histology of the immunofluorescence sections was evaluated by light microscopy of hematoxylin–eosin-stained adjacent sections. The nomenclature for placental structures has been adapted from Benirschke and Kaufmann (1990) and Castellucci et al. (1990). It has been shown by three-dimensional reconstructions from serial sections that “syncytial knots,” “sprouts,” and “bridges” are often mere tangential sections of tortuous villous surfaces (Cantle et al. 1987). Our observations in this study support this interpretation. To ensure that the trophoblastic sprouts were indeed genuine sprouts, the histology of individual sprouts was followed from at least six serial 5-μm sections covering a depth of 30 μm.
Results
The results are summarized in Table 1. For each aspect mentioned in the table, sections from at least three placental tissue blocks were studied. The immunofluorescence results on different tissue samples were consistent, with no major variations in fluorescence intensity or distribution.
The Villous Trophoblastic BM
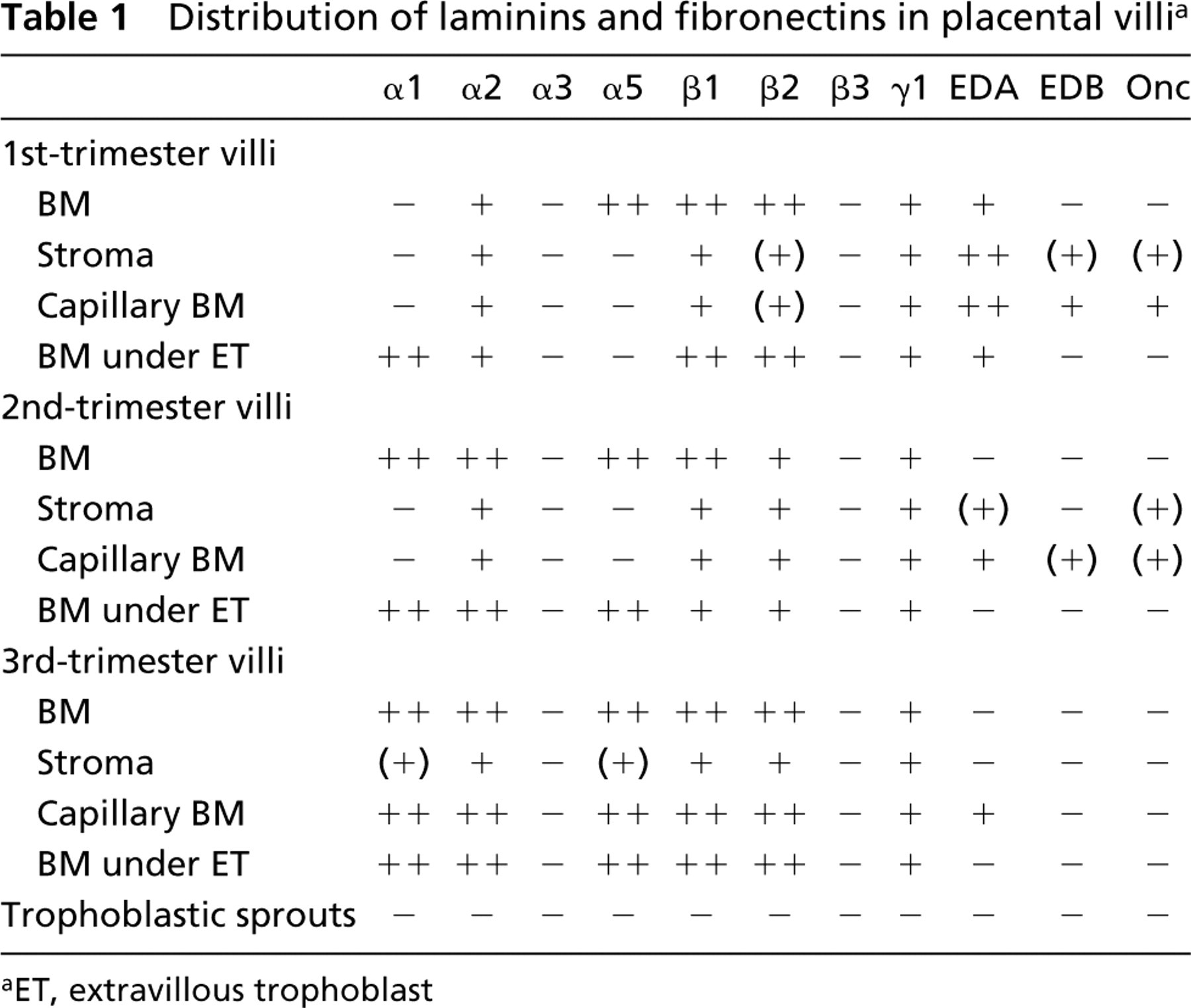
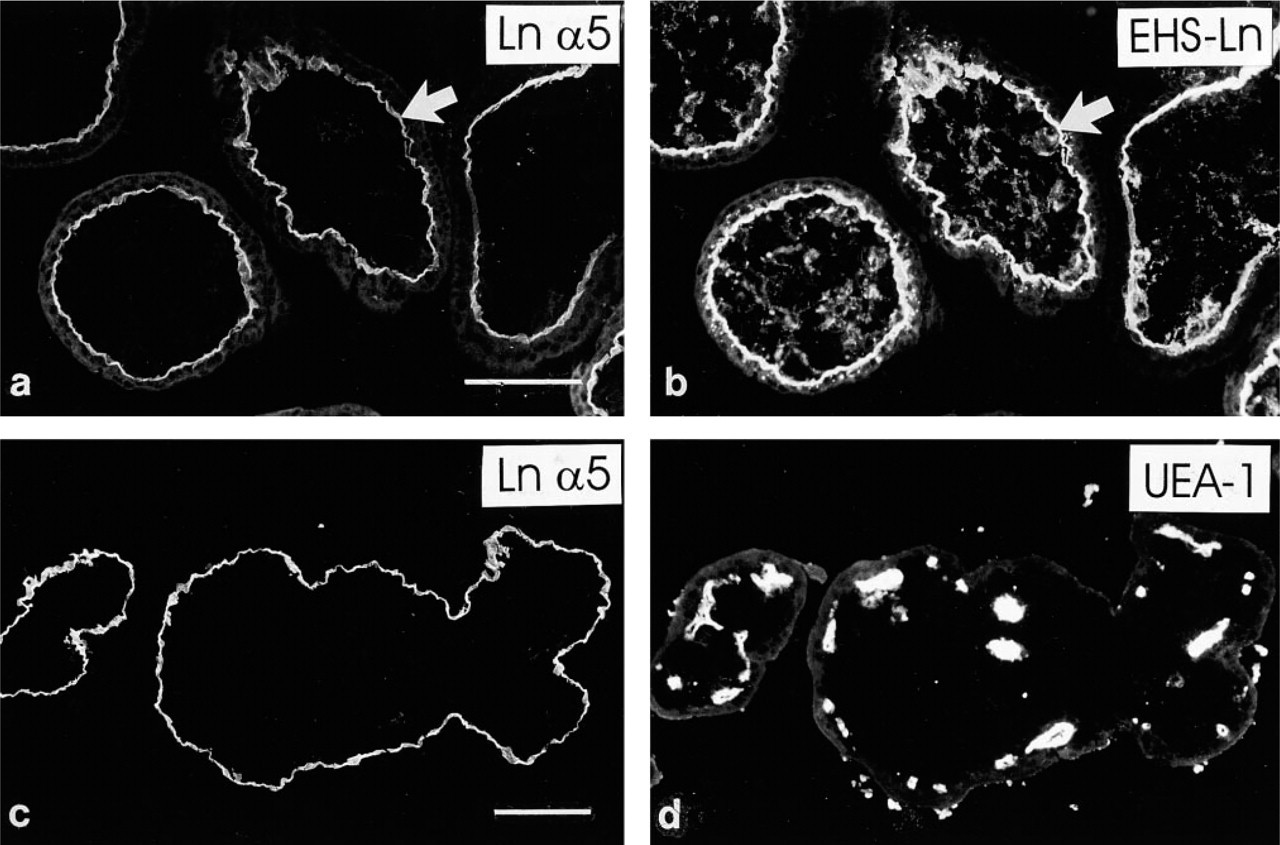
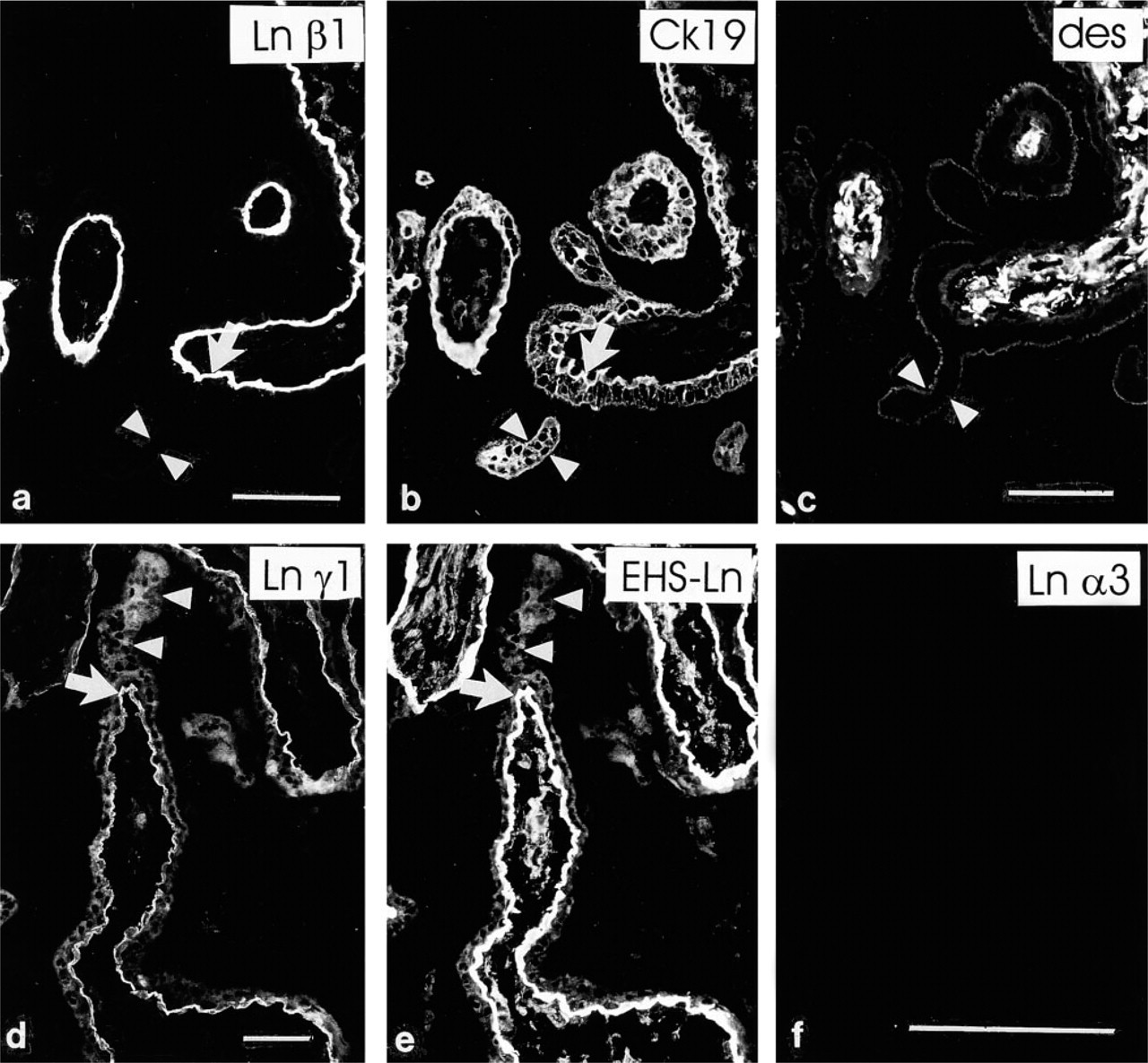
Antibodies detecting laminin β1 (Figures 1a and 1b), α5 (Figure 1c), and α2 (Figure 1d) chains reacted avidly in a linear pattern at the periphery of placental villi of all three trimesters. Double immunostainings using the anti-cytokeratin serum (Figures 1a and 1b), identifying the trophoblast, indicated that these laminin chains were found at the basal aspect of the villous trophoblast, apparently in trophoblastic BMs. Furthermore, a weaker but convincing BM-like immunoreactivity for β2 (Figure 1e) and γ1 (Figure 1f) chains was detected basally to the villous trophoblast. No distinct differences were noted in the distribution of laminin chain immunoreactivities in trophoblastic BMs from placentas at various stages of gestation.
Interestingly, distinct immunoreactivity for the laminin α1 chain was localized selectively in trophoblastic BMs at sites of formation of extravillous trophoblast.
Distribution of laminins and fibronectins in placental villi a
aET, extravillous trophoblast
Laminin chains in placental villi. (
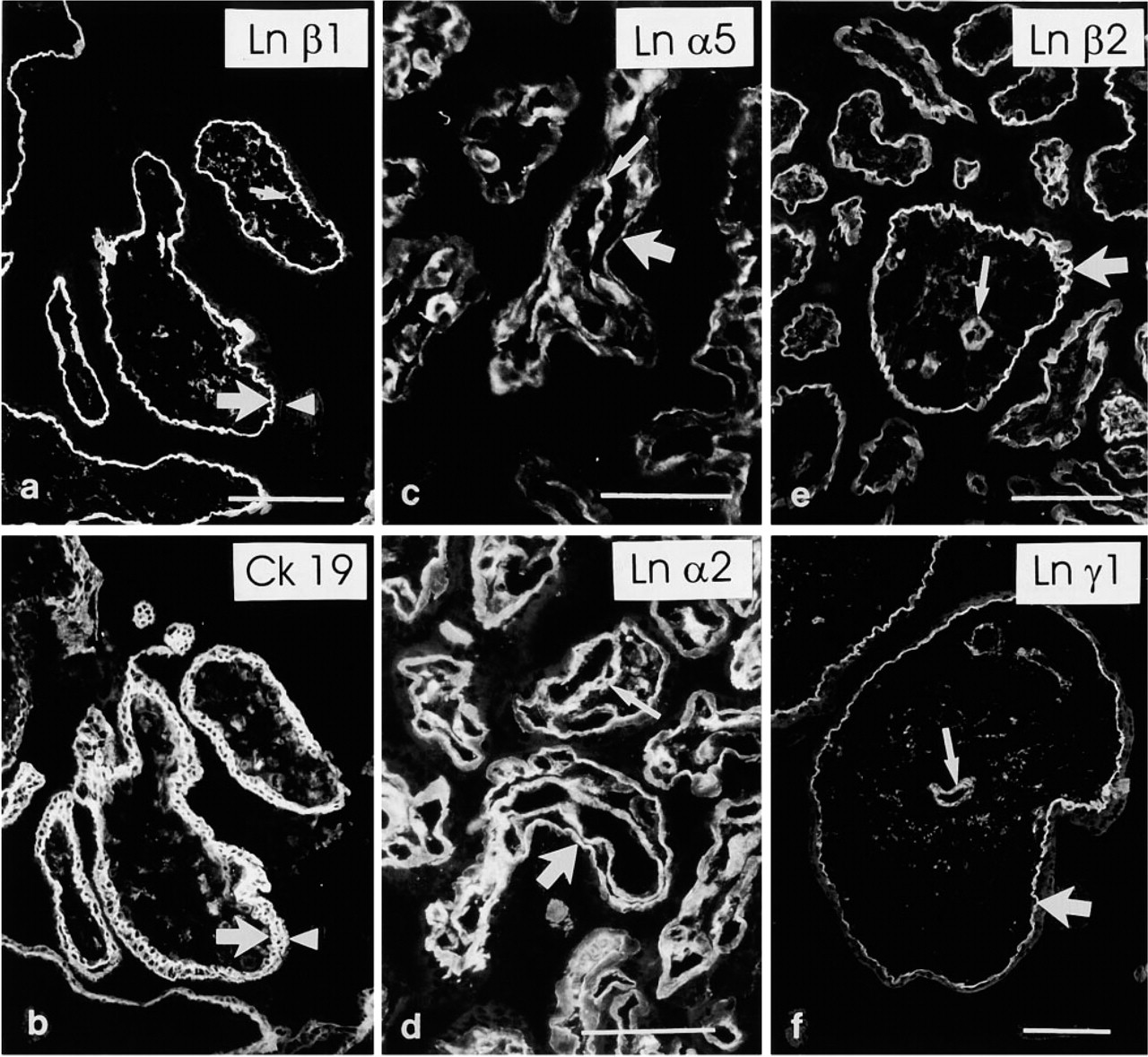
In first-trimester placentas, when extravillous trophoblast is typically found in trophoblastic cell islands, immunoreactivity for the laminin α1 chain was detected in the trophoblastic BM that is in direct contact with extravillous trophoblastic cells (Figures 2a and 2b). The rest of the villous trophoblastic BM was negative. In second-trimester placentas, extravillous trophoblast often forms anchoring trophoblastic cell columns. Analogously to first, trimester placentas, the laminin α1 chain MAb reacted avidly with the trophoblastic BM at these sites (Figures 2c–2f), whereas reactivity was not detected in BMs in other parts of villi. No immunoreactivity for the laminin α1 chain was found among the extravillous trophoblastic cells themselves within cell islands or columns. In third-trimester placentas, additional weak and irregular laminin α1 immunoreactivity was detected in the trophoblastic BM of large stem villi (not shown).
Laminin α1 chain in placenta. (
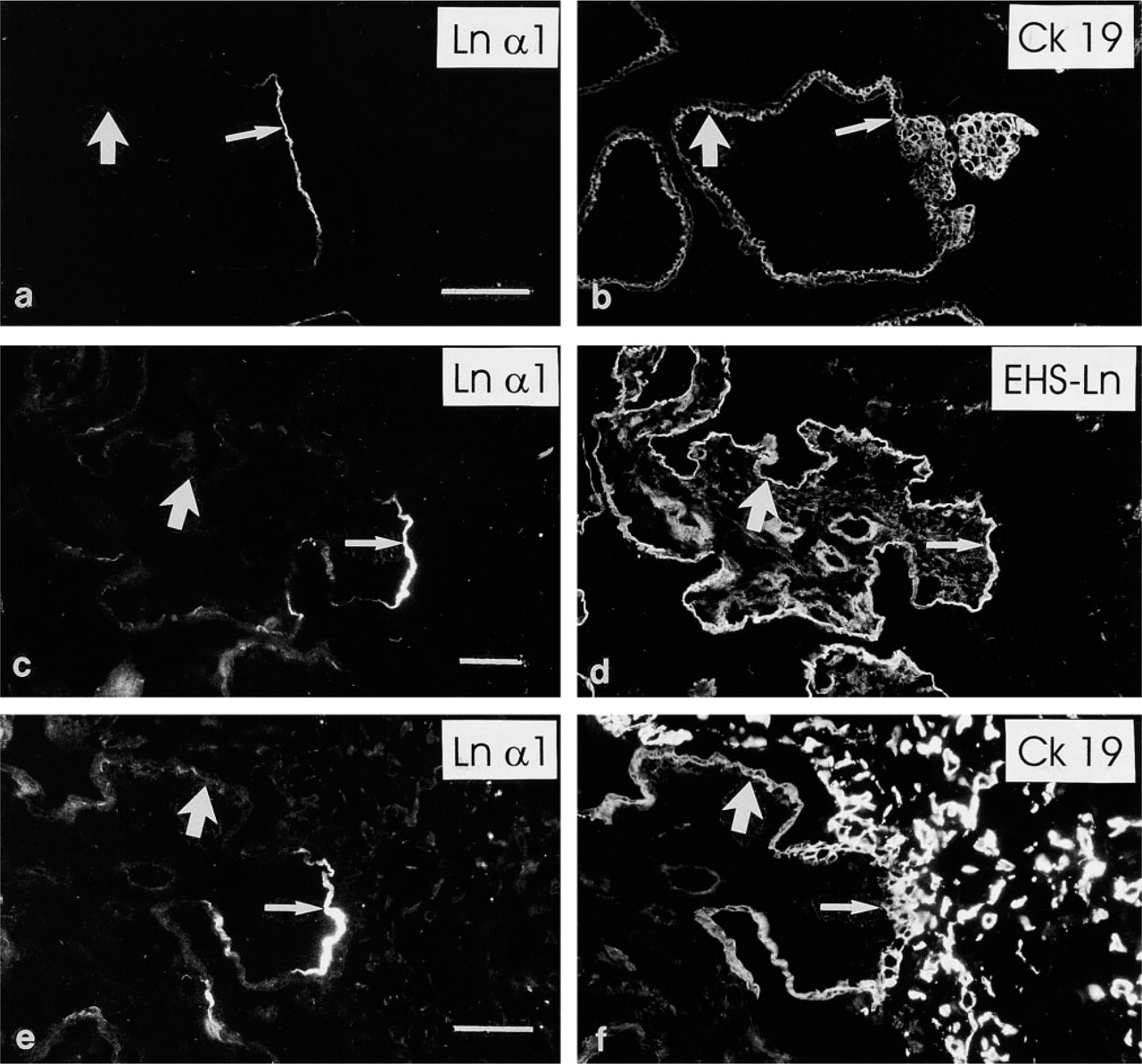
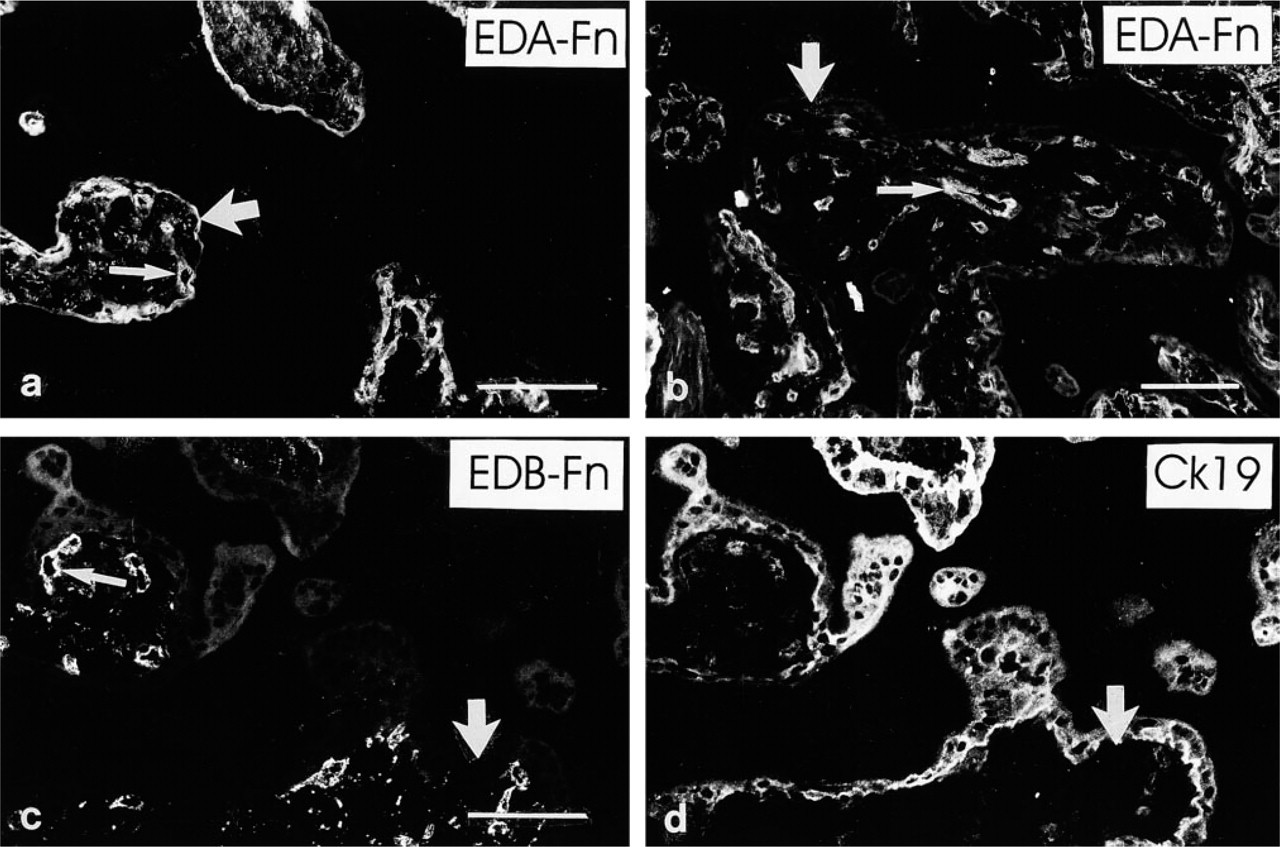
The fibronectin and EDA–fibronectin (Figure 3a) MAbs revealed linear immunoreactivity in the trophoblastic BMs of first-trimester placentas. However, these antigens were not predominantly detected in second- (Figure 3b) and third-trimester villous trophoblastic BMs. The EDB–fibronectin (Figures 3c and 3d) and oncofetal fibronectin (not shown) MAbs did not react with villous trophoblastic BMs.
Villous Stroma and Capillaries
In addition to immunoreactivity in trophoblastic BMs, distinct immunoreactivity for laminin α2 (Figure 1d) and β1 (Figure 1a) chains was found in villous stroma and capillaries. The laminin β2 (Figure 1e) and γ1 (Figure 1f) chain MAbs also reacted weakly with these structures. In contrast to the α2-, α1-, β2-, and γ1 chain MAbs, laminin α5 chain antibodies failed to react with the villous stroma and capillaries (Figures 4a–4d) of first-trimester placentas. However, intense immunoreactivity for the laminin α5 chain was detected in BMs of villous capillaries in third-trimester placentas (Figure 1c).
Fibronectins in placenta. (
The MAbs recognizing fibronectin and EDA-fibronectin revealed distinct, spotty immunoreactivity in first-trimester villous stromal tissue and reacted with villous capillaries (Figure 3a). The EDB–fibronectin (Figures 3c and 3d) and oncofetal fibronectin MAbs reacted similarly but more weakly. In second- and third-trimester placentas, the anti-EDA–fibronectin MAb (Figure 3b) reacted weakly with the villous stroma and distinctly with capillaries, while weak immunoreactivity for EDB– and oncofetal fibronectin was seen in villous capillaries (not shown).
Trophoblastic Sprouts
Trophoblastic sprouts are outgrowths from the surfaces of existing villi, believed to represent sites of formation of new ones. The presence of laminin isoforms in the sprouts was determined from serial sections of first-trimester placentas. The trophoblastic sprouts were devoid of laminin and fibronectin chain immunoreactivity, as assayed by the MAbs (Figures 5a–5d) as well as the EHS–laminin antiserum (Figure 5e). However, when a mesenchymal core, identified by double immunostaining with desmin (Figure 5c) or vimentin (not shown), was present in the sprouts defining them as villous sprouts, distinct immunoreactivity for the laminin α5, α2, β1 (Figure 5a), and weaker for β2 and γ1 (Figure 5d) chains was detected in the sprouts at the trophoblast–mesenchyme interphase and in the mesenchymal core.
We also asked whether the BM at the tip of the mesenchymal villous core ingrowth into the trophoblastic villus, which may represent a site of formation of nascent BM, differs in laminin composition from the rest of the trophoblastic BM. The distributions of the immunoreactions of each laminin chain at these locations were found to be identical with that of the polyclonal EHS–laminin antiserum (Figures 5d and 5e). This indicates that all laminin chains are found at the tip of the mesenchymal villous core ingrowth into the trophoblastic villus.
Laminin α5 chain in first-trimester placental villi. (
No immunoreactivity for the the α3 (Figure 5f) and β3 laminin chains was detected in placental villous BM or stroma at any gestational age.
Discussion
The placenta and cells derived therefrom have been a major source of human laminins for biochemical studies. Earlier studies have shown that laminin is found in BMs of placental villi as well as decidual cells (Charpin et al. 1985; Kurosawa et al. 1985; Yamada et al. 1987; Earl et al. 1990; Graf et al. 1997). Leivo et al. (1989) reported that the α2 chain is found in trophoblastic BMs. Likewise, it has been reported that fibronectin (Kurosawa et al. 1985; Yamaguchi et al. 1985; Yamada et al. 1987) and EDA–fibronectin (Virtanen et al. 1988) are found in the trophoblastic BM and villous mesenchyme and capillaries during the first trimester, but are confined to villous capillaries later during placental development. However, these studies were carried out using polyclonal antibodies or a single monoclonal antibody that detects one laminin or fibronectin chain. Therefore, it is of interest to develop a comprehensive view of the localization of laminin and fibronectin isoforms within the developing and mature human placenta.
Antibodies
The specificity of the antibodies used in our study requires comment. The MAb C4 (anti-laminin β2) has been reported to crossreact weakly with the laminin β1 chain and with an unidentified high molecular weight band in rat tissue extracts (Hunter et al. 1989; Sanes et al. 1990). We have shown, however, that the MAb C4 is specific for the laminin β2 chain in human placental tissue (Korhonen and Virtanen 1997).
Laminin chains in sprouting villi. (
The MAb 4C7, formerly believed to recognize the laminin α1 chain, has been shown to recognize laminin α5 (Tiger et al. 1997; Church and Aplin 1998). The terminology of earlier reports must be re-interpreted in this respect.
The Trophoblastic BM
In this work we demonstrate that the α2, α5, β1, β2, and γ1 laminin chains are found in the villous trophoblastic BM as well as in the stroma. In agreement with these results, laminin-2, −4, −10, and −11 (Engvall et al. 1990; Brown et al. 1994) have been isolated from human placental tissue. Earlier reports have suggested that laminin α2 and α5 chains, as well as the β1 and β2 chains, have mutually exclusive distributions in BMs of various locations (Engvall et al. 1990; Sanes et al. 1990). In placental villi, this is clearly not the case. Immunoreactivity for all these four chains was found in trophoblastic and villous capillary BMs.
Interestingly, laminin α1 chain was found selectively in trophoblastic BMs adjacent to trophoblastic cell islands and columns. A similar restricted distribution has been described for tenascin (Castellucci et al. 1991). These structures are sites of active cell division (Korhonen and Virtanen 1997), and the ECM is an important regulator of cell proliferation and differentiation (Ekblom et al. 1998). However, the significance of the selective localization of laminin α1 chain at these sites is unknown at present. These results support the hypothesis that cell islands and columns are homologous structures (Benirschke and Kaufmann 1990). The data complement our earlier results (Korhonen and Virtanen 1997) on the distribution of ECM molecules among extravillous trophoblastic cells. It appears that among the various laminin chains, only the laminin α5, α1, and γ1 chains are found both in the trophoblastic BM and within the matrix deposition zone of anchoring cell columns, whereas the α1, α2 and β2 chains are confined to the trophoblastic BM. Durbeej et al. (1996) suggested that α1 and β2 chains would have mutually exclusive localizations. At these sites, however, α1 and β2 are co-expressed, possibly allowing the presence of laminin-3. Indeed, we have recently purified laminin-3 from human placental tissue (Champliaud et al. 2000).
Furthermore, the study shows that laminin α3 and β3 chains are not found in placental tissue, suggesting that laminins-5, −6, and −7 are not present in placenta.
Earlier studies have shown that fibronectin is found in trophoblastic BMs during the first trimester but disappears later in development (Kurosawa et al. 1985; Yamaguchi et al. 1985; Yamada et al. 1987; Virtanen et al. 1988). Here we show that EDA–fibronectin is, and EDB– and oncofetal fibronectin are not, present in developing trophoblastic BMs.
Trophoblastic Sprouts
Autio–Harmainen et al. (1991) reported rich expression of laminin β1 and collagen Type IV mRNA and proteins in “cytotrophoblastic columns,” and hypothesized that the presence of these proteins is required for the ingrowth of the villous stroma during development of new villi. These results are somewhat contradictory to those presented in this study. In part, this is due to the general confusion of terminology in the field of placental histology. Villous trophoblastic sprouts are believed to represent sites of production of new villi from existing villi (Benirschke and Kaufmann 1990; Castellucci et al. 1990). By definition, they lack a mesenchymal core, and consist only of syncytio- or cytotrophoblasts. During villous development, these structures are invaded by mesenchyme and are then called villous sprouts. Our results demonstrate that laminin and fibronectin proteins are not present in pure trophoblastic sprouts but that, when mesenchymal elements invade their core, laminins and EDA–fibronectin are detected at the trophoblast–mesenchyme interface and within the mesenchymal core itself. These results imply that the prior presence of these ECM proteins may not be required for the ingrowth of the mesenchyme. Rather, it is likely that epithelial–mesen-chymal interaction is a prerequisite for the deposition of these BM components.
We also asked whether there would be differences in the distribution of the individual laminin chains at the tip of the mesenchymal ingrowth into the developing villus. We hypothesized that a differential distribution would indicate that particular laminin trimers would be deposited in the maturing trophoblastic BM before other isoforms. However, all the α2, α5, β1, β2, and γ1 laminin chains were distributed co-extensively at the tip of the mesenchymal core.
In contrast to trophoblastic sprouts, abundant ECM proteins are found among extravillous trophoblastic cells in trophoblastic columns and islands (Korhonen and Virtanen 1997). However, these are not sites of formation of new villi. We suggest that the structures alluded to by Autio–Harmainen et al. (1991) are formations of extravillous trophoblast.
Villous Stroma and Capillaries
Distinct changes in the distribution of laminin chains during the development of the villous capillaries were noted. Whereas laminin α2, β1, β2, and γ1 chains, possibly as laminin-2 and laminin-4, were found in the capillary BMs throughout placental development, α5 was seen only in the BMs of second- and third-trimester villous capillaries. Both laminin α2 and α5 chains are also found in brain capillary BMs (Jucker et al. 1996). However, capillaries of the kidney, skeletal muscle, and heart are not immunoreactive for the laminin α2 chain (Leivo and Engvall 1988; Engvall et al. 1990; Sanes et al. 1990; Virtanen et al. 1995). It is clear that capillary BMs are heterogenous with respect to their laminin composition. Our results are in contradiction to those of Leivo and Engvall (1988) as well as these of Leivo et al. (1989), who reported that laminin α2 chain is not found in BMs of villous capillaries. In the latter study, the anti-α2 chain MAb was the same as used in this study. Pepsin-digested, paraffin-embedded tissues were studied, whereas we used acetone-fixed frozen sections; this difference may account for the discrepancy.
EDB– and oncofetal fibronectin were found in capillaries in first- but not third-trimester placental villi, possibly reflecting their role in angiogenesis. Interestingly, EDB–fibronectin is specifically found in tumors and other pathological processes at sites of neovascularization (Tarli et al. 1999).
The results of this study show that several laminin and fibronectin isoforms are found in the villous trophoblastic and capillary BMs and in the villous stroma, and that the distribution of some of these ECM molecules is regulated during trophoblastic and capillary BM maturation. During outgrowth of new villous structures, laminins are not found in the trophoblastic sprouts, but they are deposited at the trophoblast–mesenchyme interface as the sprouts are invaded by mesenchyme. This suggests that epithelial–mesenchymal interaction is a prerequisite for the deposition of the villous BM components.
Footnotes
Acknowledgements
Supported by a project grant from the Medical Faculty, University of Helsinki.
We are indebted to Drs R.E. Burgeson, E. Engvall, S.-I. Hakomori, P. Liesi, V. Pallini, J. Sanes, and L. Zardi for kind gifts of antibodies. The skillful technical assistance of Mr Hannu Kamppinen, Mr Reijo Karppinen, and Ms Marja-Leena Piironen is acknowledged.
