Abstract
L-2 cells are an immortalized cell line derived from yolk sac parietal endoderm cells, which are responsible for the production of Reichert's membrane, a thick basement membrane produced during rat gestation. Although the L-2 cells secrete all the major components of the basal lamina, they do not assemble a robust matrix in cell culture. We hypothesized that the reason L-2 cells fail to assemble a matrix in cell culture is because the concentrations of matrix components necessary for this matrix assembly do not reach a critical association concentration (CAC) under standard cell culture conditions. To limit the diffusion of secreted molecules while maintaining a nutrient-rich environment for the cells to thrive, we developed a technique that uses a dialysis membrane to limit protein diffusion in a 2-well plate format. This technique permits L-2 cells to assemble a robust matrix in as little as 24 hr that continues to be formed for at least 72 hr. This technique may address some of the physical limitations imposed by cell culture and could be readily applied to other cell types and medium conditions.
Keywords
B
L-2 cells are a tumor cell line derived from parietal endoderm cells which produce Reichert's membrane, a basement membrane transiently present during rodent development (Kulay and de Moraes 1965). Although these cells have been shown to secrete all the major components of BL, they fail to assemble a significant matrix in culture under routine conditions (Delvoye et al. 1988). However, in vivo the native parietal endoderm cells assemble a thick BL over a period of several days. One explanation for the absence of BL assembly by L-2 cells in vitro is that the volume of medium used in routine cell culture is several orders of magnitude greater than that of the immediate cellular microenvironment in vivo. Therefore, under routine cell culture conditions the secreted components are rapidly diluted into the surrounding medium. This concept led to us to consider the hypothesis that the conditions necessary for L-2 cell BL assembly require a critical assembly concentration (CAC) in the immediate vicinity of the L-2 cell surface.
To test our hypothesis, we limited the diffusion of secreted products to the immediate pericellular area while permitting the cells access to nutrients necessary for survival. Our technique uses a semipermeable membrane to limit diffusion of secreted products in a 24-well plate format. Our observations indicate that L-2 cells, which do not assemble a significant matrix under routine cell culture conditions, can be induced to assemble a robust BM matrix in 24 hr when the diffusion of secreted products is limited by a semipermeable membrane.
Materials and Methods
Reagents and Supplies
The L-2 cells were a kind gift from Ulla Wewer (Copenhagen, Denmark). The heparan sulfate proteoglycan (perlecan) antibody C17 and the s-laminin (β2 chain) antibody C4 were originally developed by Dr. Joshua Sanes (Sanes and Chiu 1983; Eldridge et al. 1986). The type IV collagen antibody was developed by Foellmer et al. (1983). These antibodies were obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICD and maintained by the University of Iowa (Department of Biological Sciences, Iowa City, IA). A polyclonal antiserum directed against laminin-1 was developed in our laboratory and recognizes all three laminin chains present in laminin-1 (Routh et al. 2002). The monoclonal antibody (MAb) 2D6 with immunoreactivity against a basement membrane proteoglycan has been previously described (McCarthy et al. 1989). Secondary antisera (Cy2-conjugated goat anti-mouse IgG, Cy3-conjugated donkey anti-rabbit IgG) were obtained from Jackson ImmunoResearch (West Grove, PA). Dialysis membrane with a MWCO of 12–14,000 kD was purchased from Spectrum Medical Industries (Houston, TX). Silicon rubber O-rings were purchased from Small Parts (Miami Lakes, FL). The 24-well plates were purchased from Becton Dickinson (Franklin Lakes, NJ).
Cell Culture Conditions
L-2 cells were maintained as previously described (Wassenhove-McCarthy and McCarthy 1999) in Dulbeco's modified eagle's medium (DMEM) containing
Immunohistochemistry and Digital Microscopy
After fixation the cells were washed three times for 10 min with PBS. The cells were blocked for 20 min with 1% BSA in PBS, followed by another PBS wash three times for 10 min. For immunostaining, the anti-laminin polyclonal antiserum was used at a 1:100 dilution in PBS. MAb supernatants were applied directly to the cells without dilution for 45 min, followed by a PBS wash three times for 10 min. The labeled secondary antiserum was diluted at 1:100 in PBS and applied to the cells for 45 min. Then the cells were washed three times for 10 min with PBS and then directly imaged within the wells to maintain the 3D dimensional organization of the assembled ECMs. Digital images of the specimens were acquired using an Olympus IX-70 microscope equipped with epifluorescent illumination optics and appropriate filters. The microscope was interfaced to a SenSys camera (Roper Industries; Tucson, AZ) whose signal was ported to a PowerMacintosh 9600 hosting the imaging acquistion/analysis software IPLab spectrum (Scanalytics; Fairfax, VA).
Results
When the diffusion of secreted products from L-2 cells was limited by the application of a dialysis membrane (MWCO 12–14,000), a robust matrix containing laminin was assembled within 24 hr (Figure 1B) and continued to be formed for 48 (Figure 1D) and 72 hr (data not shown). L-2 cells grown for 24 or 48 hr without a dialysis membrane secreted laminin but did not develop an organized laminin matrix (Figures 1A and 1C). In those wells the laminin staining had a punctate pattern. Similar patterns of matrix deposition were seen in wells immunostained for type IV collagen that contained L-2 cells grown with (Figure 1F) or without (Figure 1E) a diffusion-limiting membrane. The antibodies directed against perlecan (Figure 1G) or BM-CSPG (Figure 1H) also showed the development of pericellular matrices in cultures using the diffusion-limiting membrane. Thus, the L-2 cells assemble a BL matrix that contains at least one isoform of several common BL-associated molecules. Interestingly, the pericellular matrices immunostained with the MAb directed against the β2 laminin chain were negative for L-2 cell cultures under both growth conditions (data not shown).
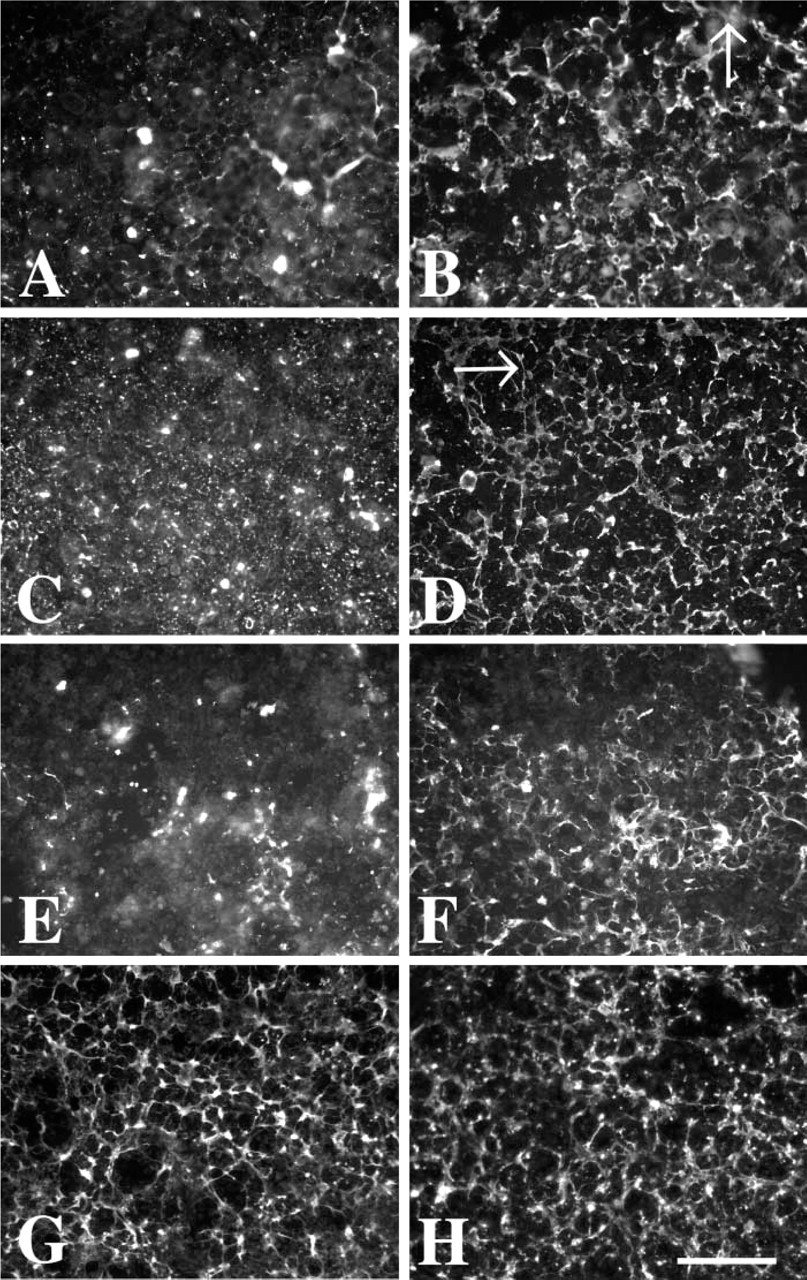
L-2 cells are induced to assemble a robust matrix by use of a dialysis membrane to limit the diffusion of secreted products. Under routine cell culture conditions, L-2 cells do not assemble a matrix, as seen by immunohistochemical labeling with laminin (
Discussion
In this report we have described a technique using a semipermeable membrane to induce the assembly of a basal lamina in a cell culture system within a 24 hr period.
We use the term BL to describe the pericellular matrices assembled by L-2 cells. In the literature, the term BM is often used by many in an interchangable manner with the term basal lamina. However, in the strictest sense, the term “basement membrane” refers to the complex ECM that underlies, for example, epithelia. Its constituent parts include the BL and the reticular lamina (Hay 1991). Because L-2 cells primarily secrete components of the BL, the pericellular matrix assembled by these cells should be considered a BL.
Although BLs have been extensively studied in animal model systems, in vitro modeling of these structures in some cell types is often difficult. Some cells, e.g., epithelial cells, readily produce a BM in culture. In light of the CAC hypothesis, this may be due to the fact that polarized epithelia will directionally/vectorially secrete BL components to their basal surface, where diffusion of these components into the surrounding medium is directly limited by the intact cellular monolayer. With other cell types, assembly occurs only after prolonged culture, or may require co-culture with a nurse cell, which can complicate in vitro modeling (Delvoye et al. 1988; Tammi et al. 2000). In addition to relying on the production of a native BL by cells, cell–matrix interactions can be studied using a commercially available “generic” pericellular matrix, such as Matrigel (Becton Dickinson; Bedford, MA). Although the Matrigel matrix contains BL proteins that facilitate cell growth, it is now accepted that many BLs are tissue-specific in their complement of matrix molecule isoforms. In many circumstances, the basement membrane proteins/proteoglycans secreted by one cell population differ in isoform composition from those present in Matrigel.
We believe that the using the CAC method has several advantages. (a) It allows cells to rapidly develop a true native BL matrix, especially those cells such as L-2 cells that appear to secrete ECM proteins from their entire cell surfaces in a non-vectorial fashion. (b) Once cells have developed their own native matrix, the culture is readily accessible for further in vitro studies with or without the diffusion-limiting membrane. (c) The CAC method allows the study of the development of a matrix by a cell line that may not readily assemble a matrix in vitro. (d) The CAC method could be applied to any situation in which limiting the diffusion of secreted products to mimic in vivo concentrations would be useful, such as studying the effect of natively secreted growth factors or chemokines.
This report further demonstrates the importance of limiting secreted protein diffusion to enhance local pericellular protein concentrations to promote matrix assembly in a cell culture system. In earlier reports Yurchenko and co-workers demonstrated that BL assembly can be recapitulated in vitro using purified basement membrane components (Yurchenco et al. 1985, 1992). In an earlier study using the L-2 cell culture system, Delvoye et al. (1988) suggested that such a mechanism might be applicable but dismissed the concept as a result of the outcome of their studies. In their report they indicated that L-2 cells could be induced to assemble a BM only when grown in the presence of fibroblasts. Parallel experiments in our laboratory also confirm the observations (data not shown) that in a co-culture system under non-diffusion limiting conditions this does occur, Although they acknowledged the possibility for the need of a precise concentration of BL components diffusing from L-2 cells to assemble a matrix, they concluded that a molecule or molecules synthesized by fibroblasts and associated with the pericellular matrix was probably responsible for the assembly of the L-2 secretred BL matrix in this model system. The deduction was based on the fact that the BL matrix secreted by L-2 cells was assembled on the fibroblast monolayer at some distance from the L-2 cells, indicating that a secretion product or products covering the clusters of fibroblasts is involved in the assembly of the BM. Our report shows that when the secreted products of L-2 cells are limited to the immediate pericellular area of the L-2 cell monolayer, a robust matrix can readily be assembled/deposited without the presence of fibroblasts. Our data therefore indicate that conditions necessary for the assembly of the matrix by L-2 cells are not limited to the fibroblast co-culturing system but rather that L-2 cells are capable of rapidly assembling their own pericellular matrix under conditions in which the diffusion of secreted macromolecules is limited to the immediate vicinity of the cells.
Although the evidence of a critical concentration of components necessary for the assembly of a matrix has been portrayed in this report, there are other avenues that will guide future work. If the concept of CAC is induced by this apparatus, one would predict that increasing the MWCO of the dialysis membrane would result in an inverse deposition of matrix. Similarly, by varying the amount of medium beneath the membrane directly in contact with the cells, the CAC hypothesis would predict that increasing the volume of medium bathing the cells would result in a decrease in the extent of assembly of the pericellular matrix.
In summary, we have developed a technique that uses a dialysis membrane to limit the diffusion of proteins secreted from L-2 cells to induce the assembly of a robust matrix within 24 hr. We propose that the membrane has caused a critical association complex (CAC) to form, concentrating components necessary for matrix assembly.
Footnotes
Acknowledgements
Supported by a research grant 1-RO1-DK48055 (KJM).
This work was submitted in partial fulfillment of the PhD degree (MEL) for the Louisiana State University Health Sciences Center, Shreveport, LA.
