Abstract
Samples of different tissues were preserved in seven fixatives for periods of time extending from 1 to 336 days, to determine which fixatives reduce the time-dependent degradation of DNA and preserve the histological structure. To achieve these results, three PCR systems were used: FGA and TC11 (both for nuclear DNA) and HV1 for mitochondrial DNA (mt-DNA). For long-term storage in combination with amplification of nuclear and mt-DNA, consistent results were obtained in Carnoy's solution and glutaraldehyde. Variable results were observed for buffered formalin; an mt-DNA product could be detected even after 3 months of fixation. In regard to comparison of the different tissues, the quantities recovered from skeletal muscles and kidneys were higher than from other tissues.
I
We chose these fixatives to investigate the influence of routinely used fixatives, fixatives no longer used, and some new fixatives that have not yet been investigated. Duration of fixation was as follows: 1, 3, 5, 7, 14, 28, 56, 84, 168, and 336 days. After this time the blocks of tissue were removed from the fixative and washed for 3 to 4 hr in isotonic saline solution. Subsequently, samples were stored at −25C until use.

Fixative: glutaraldehyde. (
DNA was extracted using the QIAamp DNA Mini Kit (Qiagen; Hilden, Germany). To test for complete, non-degraded DNA in the samples, the samples were amplified by PCR for the FGA system (∼800-bp fragments, primer FGA F88/R82). Primers were designed using the Primer3 software (http://www-genome.wi.mit.edu/cgi-bin/primer/primer3_www.cgi). The cycle conditions in a PTC-200 cycler (MJ Research; Watertown, MA) were as follows: 94C for 3 min soak, 94C for 30 sec, 60C for 30 sec, 72C for 1 min, 30 cycles, and final extension at 72C for 30 min. The forward primer used was 5′-ATG GAC AGC GAG TCT AGG GA-3’ and the reverse primer was 5′-GGG ACC ACA GCC ACA TAC TT-3′.
PCR products were made visible using electrophoresis in an agarose gel (PAGE) and silver staining. If a negative result was obtained, PCR was conducted again in which the TC11 system with a lower number of base pairs (∼150- to 170-bp fragments, primer TC11) was amplified as described by Edwards et al. (1992). Amplified samples were detected by means of laser fluorescence detection. After that, detection of mt-DNA was conducted with all the samples, including those that had produced negative TC11 results. This was done by means of PCR amplification of the HV1 region of the mt-DNA (∼220-bp fragments, primer HV1 L16190/H16410) as described by Anderson et al. (1981) and through subsequent PAGE and silver staining. For all PCR samples, 25 μg of DNA were used. As a control sample, general DNA material from the Institute of Legal Medicine was used. To investigate the influence of the fixative on the histological structures, each sample was stained using hematoxylin eosin (HE) and examined under a microscope. The most suitable substances for long-term fixation with possible subsequent DNA analysis were Carnoy's solution and glutaraldehyde. With these two solutions, it was possible to amplify and detect both nuclear DNA (with up to 800-bp fragments) and mt-DNA in each of the tissues investigated even after fixation for a period of 336 days (Figure 1).
In the phosphate-buffered formalin-fixed tissues, nuclear DNA could be detected in all tissues up to 28 days, but only up to a length of 150- to 170-bp fragments (TC11). From the 56th day of fixation, amplification of nuclear DNA was no longer possible in certain tissues (liver and brain), and on the 84th day no nuclear DNA could be detected in any of the five investigated tissues. Thus, similar results were found in regard to the duration of fixation and the detection of DNA as reported, for example, by Greer et al. (1991) and Wiegand et al. (1996). However, mt-DNA could be detected even here after a period of up to 336 days (Figure 2). The least suitable substances were found to be unbuffered formalin and potassium dichromate acetic acid. In both samples some loss was detected even after only 3 days. From the 14th day, nuclear DNA could no longer be detected. In the case of buffered formalin, mt-DNA could still be detected after 336 days. Test results for each DNA analysis are given in Table 1. When the various tissue samples were compared, skeletal muscle and kidney were particularly suitable, similar to Wiegand et al. (1996). However, these two tissues present some problems with regard to processing and cell lysis with proteinase K, because complete lysis could not be achieved. Brain tissue showed strong fluctuations in the test results, which can be attributed to uneven penetration by the respective fixatives and to differences in the number of cells. In investigating the influence of the fixatives on the histological structures, both Carnoy's solution and glutaraldehyde cause major damage to the individual tissue structures. Use of these fixatives with regard to possible forensic histological evaluation is therefore unlikely to be feasible. Potassium dichromate acetic acid was also impractical because its own color is too intense, and histological staining (HE) was no longer possible. Of all the fixatives investigated, phosphate-buffered formalin had the least influence on the histological structures of the tissues (Table 1).
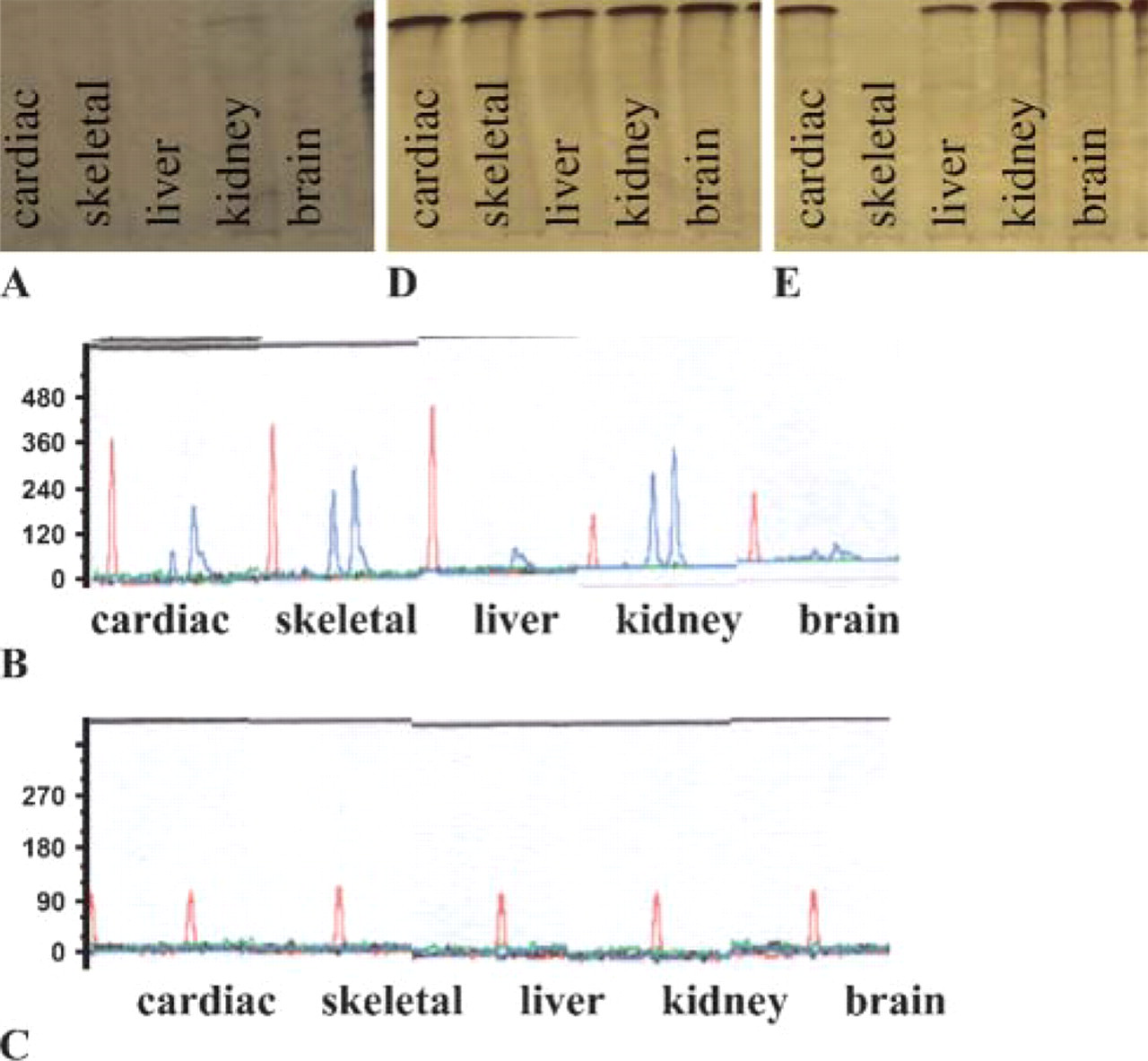
Fixative: 4% buffered formalin. Detection of DNA [PCR with FGA (
To summarize, Carnoy's solution and glutaraldehyde have clear advantages for DNA degradation. One disadvantage, however, is the very complicated process of producing these two solutions, which makes it difficult to use them in practice. A further drawback is undoubtedly the poor result obtained in histological analysis. In this regard, much better results were obtained with 4% phosphate-buffered formalin, which is already frequently used in practice. With this solution, results of DNA analysis are also satisfactory; in particular, mt-DNA could still be detected after 336 days as in the case of the aforementioned solutions. In regard to establishing identity because mt-DNA could still be detected after 1-year fixation with this fixative, it would appear that identification would be possible if comparative material was available. A similar result was obtained by Sano et al. (2000) in an investigation of fixed tissue using STR and mt-DNA primers. The remaining fixatives must be regarded as unsuitable for the purpose in question, because some revealed almost complete DNA degradation even after a few days and because histological analysis results were unsatisfactory (Greer et al. 1991; Gino et al. 2004).
Detection of nuclear DNA in the DNA systems (FGA and TC11) and of mt-DNA using the various fixatives and tissue samples
