Abstract
We previously identified a novel gene designated retinoid-inducible serine carboxypeptidase (RISC or Scpep1). Here we characterize a polyclonal antibody raised to Scpep1 and assess its localization in mouse cells and tissues. Western blot analysis revealed an immunospecific ∼35-kDa protein corresponding to endogenous Scpep1. This protein is smaller than the predicted ∼51-kDa, suggesting that Scpep1 is proteolytically cleaved to a mature enzyme. Immunohistochemical studies demonstrate Scpep1 expression in embryonic heart and vasculature as well as in adult aortic smooth muscle cells and endothelial cells. Scpep1 displays a broad expression pattern in adult tissues with detectable levels in epithelia of digestive tract and urinary bladder, islet of Langerhans, type II alveolar cells and macrophages of lung, macrophage-like cells of lymph nodes and spleen, Leydig cells of testis, and nerve fibers in brain and ganglia. Consistent with previous mRNA studies in kidney, Scpep1 protein is restricted to proximal convoluted tubular epithelium (PCT). Immunoelectron microscopy shows enriched Scpep1 within lysosomes of the PCT, and immunofluorescence microscopy colocalizes Scpep1 with lysosomal-associated membrane protein-2. These results suggest that Scpep1 is a widely distributed lysosomal protease requiring proteolytic cleavage for activity. The highly specific Scpep1 antibody characterized herein provides a necessary reagent for elucidating Scpep1 function.
Keywords
S
To date, SCs have been found across kingdoms, including plants, fungi, and animals. The best-studied SCs are yeast serine carboxypeptidase Y (CPY) and KEX1-encoded protein. The biosynthetic pathway of CPY from endoplasmic reticulum to vacuole has been elucidated (Jung et al. 1999), and KEX1 has been implicated in the processing of precursor proteins (Cooper and Bussey 1989). The majority of SCs are reported in the plant kingdom where they function in growth, apoptosis, brassinosteroid signaling, and seed development (Dominguez and Cejudo 1999; Li et al. 2001; Cercos et al. 2003; Schaller 2004). A battery of SCs and serine carboxypeptidase-like proteins (SCLPs) has been found to accumulate in plants such as rice where 71 SCs and SCLPs exist (Feng and Xue 2006). SCs and SCLPs in rice have a high degree of sequence similarity (92% identity in amino acid sequence level), indicating a potential case of functional redundancy (Feng and Xue 2006).
In contrast to the abundance of SC in plants, mammals possess a limited complement of SC genes with only three SCs identified to date. Partial protective/cathepsin A (PPCA, official gene symbol: Ppgb) is one of the most extensively studied mammalian SCs. Ppgb is synthesized as a 54-kDa precursor protein and then sorted into the endosomal/lysosomal compartment by mannose-6-phosphate receptor (MPR) (Morreau et al. 1992). In the acidic lysosomal environment, Ppgb monomer is composed of 32- and 20-kDa subunits linked together by disulfide bonds (Bonten et al. 1995). Ppgb forms a multienzyme complex with β-galactosidase and neuraminidase to prevent these two glycosidase proteases from degradation in the lysosome (Galjart et al. 1988). Therefore, Ppgb has two functions: catalytic activity and the protection of a lysosomal enzymatic complex, both of which comprise distinct domains in the Ppgb protein (Galjart et al. 1991). A second SC, prolylcarboxypeptidase (Prcp, angiotensinase C), has similarities to both SC and prolylendopeptidase families, is expressed in lysosomes, and cleaves substrates only after proline residues (Tan et al. 1993). Recent studies indicate Prcp, an angiotesin II-inactivating enzyme, reacts as an endothelial cell prekallikrein activator (Moreira et al. 2002; Shariat-Madar et al. 2002) A third SC, serine carboxypeptidase vitellogenic-like (Cpvl), distributes mainly in differentiated macrophages, although its exact biological role is presently unknown (Mahoney et al. 2001).
We previously identified a novel SC in a suppression subtractive hybridization screen for genes induced in SMC with retinoic acid (Chen et al. 2001). This SC, initially named retinoid-inducible SC (RISC), has been given the official gene symbol Scpep1 (for SC1). Consistent with other SCs, Scpep1 contains a putative N-terminal signal peptide followed by a putative SBD and three conserved domains comprising the catalytic triad (aa Ser-167, Asp-371, His-431) (Chen et al. 2001). Western blotting and immunocytochemistry analyses show exogenous Scpep1 accumulates around the nuclear periphery and is secreted in culture medium (Chen et al. 2001). Northern blotting and in situ hybridization studies demonstrate robust expression of Scpep1 mRNA in rat aorta, bladder, and kidney (Chen et al. 2001), although a full analysis of the tissue distribution of Scpep1 mRNA was not performed. Moreover, the properties and cell/tissue distribution of endogenous Scpep1 protein have yet to be defined. In this report, we have characterized a novel antibody raised against Scpep1 and demonstrate the localization of Scpep1 in embryonic and adult mouse tissues. Further analysis with immunoelectron and immunofluorescent microscopy provides firm evidence for Scpep1 localization to lysosomes. Results are discussed in context of other mammalian SC.
Materials and Methods
Scpep1 Antibody Generation
An affinity-purified polyclonal rabbit antibody (αScpep1) was generated from a bacterial-grown Scpep1 His-fusion protein corresponding to the C-terminal amino acids 173-452 of mouse Scpep1 (Genbank accession number NP_083299; Proteintech Group Inc., Chicago, IL). Antibodies to β-actin, α-tubulin, and FLAG were obtained from Sigma (St Louis, MO), and those to lysosome-associated membrane protein (LAMP2) were purchased from the Developmental Studies Hybridoma Bank at the University of Iowa (ABL-93; Iowa City, IA). To test the specificity of αScpep1 in Western blotting and immunohistochemistry assays, the antibody was preabsorbed to 1 mg/ml full-length Scpep1 peptide overnight at 4C prior to application.
Cell Culture
PAC1 smooth muscle cells (SMCs) (Rothman et al. 1992; Firulli et al. 1998), mouse C2C12 myoblasts (American Type Culture Collection; Rockville, MD), human coronary artery smooth muscle cells (HCASMC; Cascade Biologics Inc., Portland, OR), human HeLa cells (American Type Culture Collection), chicken SL-29 (American Type Culture Collection), Balb/c-3T3 cells (3T3) (American Type Culture Collection), and zebrafish ZF4 embryonic fibroblasts (American Type Culture Collection) were grown in Dulbecco's modified Eagle's medium (DMEM) containing high glucose and supplemented with 10% FBS. Mouse BC3H1 cells (American Type Culture Collection) were grown in DMEM containing 20% FBS. Antibiotics and antimycotics were not included in cell culture medium. Cells were grown to subconfluency prior to each experiment.
Western Blotting
Cells from 10-cm dishes and tissues from at least three male C57BL/6J mice (Jackson Laboratory; Bar Harbor, ME) were homogenized with lysis buffer (50 mM Tris-HCl, pH, 7.5, 150 mM NaCl, 1% NP-40, and 5 mM EDTA) and protease inhibitor cocktail (P8340, Sigma). Cell and tissue lysates were cleared by centrifugation at 12,000 rpm for 30 min at 4C, electrophoresed on 10% SDS-PAGE gels under reducing conditions, and then transferred to nitrocellulose membranes. Membranes were probed with antiserum to Scpep1 (1:1000), β-actin (1:3000), or α-tubulin (1:3500) overnight at 4C, followed by horseradish peroxidase-conjugated secondary antibodies (1:4000; Amersham Bioscience, Buckinghamshire, UK) for 1 hr at room temperature. Signal was detected by enhanced chemiluminescence (Pierce Biotechnology Inc.; Rockford, IL). All animal expression studies were approved by the University of Rochester School of Medicine Institutional Animal Care and Use Committee (protocol #2003-331).
Immunohistochemistry and Immunofluorescence Microscopy
Expression of Scpep1 on embryonic and adult tissues was examined by immunohistochemistry. Tissues were fixed with 4% paraformaldehyde in 0.1 M PBS (pH 7.4) overnight and then embedded in paraffin and sectioned to 6 μm. Antigen retrieval was achieved by incubating deparaffinized sections in 0.05% citraconic anhydride in a steamer for 15 min (Namimatsu et al. 2005). Sections were quenched with 3% H2O2 and blocked with 5% normal goat serum in 0.5% nonfat dry milk/Tris-HCl (pH 7.6). Sections were incubated with rabbit antiserum to Scpep1 (1:300) overnight at 4C. After rinsing in Tris-HCl (pH 7.6), sections were incubated with biotinylated goat anti-rabbit IgG (1:100) for 1 hr and with the avidin-biotin complex (Vector Laboratories; Burlingame, CA) for an additional 1 hr. The reaction product was demonstrated by 3,3V-diaminobenzidine (DAB kit; DakoCytomation, Carpinteria, CA), lightly counterstained with hematoxylin, and then observed under an Olympus BX-41 light microscope (Olympus; Melville, NY). To test the specificity of the Scpep1 antiserum, primary antibody was replaced with rabbit IgG (DakoCytomation) or preabsorbed to Scpep1 protein as above.
To validate the subcellular localization of Scpep1 in cells, HeLa cells were transduced with 300 infectious units (ifu) C-terminal FLAG-tagged Scpep1 adenovirus (Scpep1-FLAG) for 48 hr. Adenoviral Scpep1 was generated with a commercial kit (Invitrogen; Carlsbad, CA) and a large-scale preparation was done through the Vector Core Facility of the University of Pittsburgh. Double-labeling immunofluorescence for Scpep1 and a lysosomal-specific marker was performed either in normal HeLa cells or the same cells transduced with 300 ifu Scpep1-FLAG for 48 hr. Cells were then washed with cold 0.1 M PBS and fixed with 4% paraformaldehyde for 10 min at room temperature, followed by the incubation of 0.1% saponin in PBS for 15 min and then with antiserum to FLAG (1:200) and LAMP2 supernatant for 2 hr at room temperature (Chen et al. 1985). Secondary antibodies conjugated to FITC or rhodamine (1:100) were incubated for 1 hr at room temperature. Normal 3T3 cells were also used by the same staining procedure described above with αScpep1 (1:300) and LAMP2 supernatant only. Alexa Fluor 680 (Molecular Probes; Eugene, OR) and FITC were used as secondary antibodies for the detection of LAMP2 and Scpep1, respectively. Cells were examined with an Olympus BX-51 fluorescence microscope equipped with a SPOT RT-KE 2MP Color Digital Camera (Diagnostic Instruments; Sterling Heights, MI). Confocal microscopy was done with an Olympus IX70 microscope (Olympus; Tokyo, Japan). Final images were processed in Adobe Photoshop (Adobe; San Jose, CA).
Immunoelectron Microscopy
Normal male C57BL/6J mice underwent intracardiac perfusion with 4% paraformaldehyde in 0.1 M phosphate buffer (PB), pH 7.4. Kidneys were immersion fixed for an additional 2 hr, thoroughly rinsed in PBS, and sectioned at 100 μm on a vibratome. Subcellular localization of Scpep1 was performed with αScpep1 or normal IgG and immunoperoxidase techniques using avidin-biotin-peroxidase complex as described above. Stained sections were then fixed in 5% glutaraldehyde in 0.1 M PBS overnight. Tissue was postfixed in 2% osmium tetroxide for 2 hr at room temperature, dehydrated with a graded series of alcohol, and embedded in Epon 812 resin (Polyscience; Philadelphia, PA). The cortical region of the kidney was trimmed from the embedding section and cut on an ultramicrotome (Ultracut; Leica, Heidelberg, Germany). Ultrathin sections (∼90 nm) were collected on copper grids and observed under Hitachi 7100 transmission electron microscope (Hitachi; Tokyo, Japan).
Results
Immunospecificity of a Polyclonal Antibody to Scpep1 by Immunoblot and Immunoabsorption Assays
Overall amino acid homology of mouse Scpep1 is 23% (with Ppgb) and 25% (with Cpvl); there is no homology between Scpep1 and Prcp (data not shown). Much of the homology occurs in the SBD and catalytic triad (Chen et al. 2001). We therefore generated a rabbit polyclonal antibody against the C terminus (aa 173-452) of mouse Scpep1 (αScpep1) where only the last two regions of the catalytic triad reside (Figure 1A). To characterize our newly developed rabbit αScpep1, its immunospecificity for endogenous Scpep1 in cell lines was analyzed by Western blotting. Figure 1B shows that αScpep1 polyclonal antibody detects a band of ∼35 kDa in rat PAC1 SMC, mouse BC3H1, and mouse C2C12 cells, but not in HCASMC, HeLa, chicken SL-29 fibroblasts, or zebrafish ZF4 fibroblasts. The amino acid fragment (aa 173-452) of mouse Scpep1 used to generate our antibody is represented in rat (90% identity), human (82% identity), chicken (64% identity), and zebrafish (63% identity) orthologs (unpublished data). The fact that these homologies are much greater than those between Scpep1 and other SC (Ppgb and Cpvl) strongly suggests that the protein detected is Scpep1 and not another SC.
The predicted molecular weight of Scpep1 (452 aa) is ∼52 kDa (Chen et al. 2001). The presence of an ∼35-kDa protein suggests that Scpep1 may be proteolytically cleaved. This immunoreactive product is larger than that of the cleaved Ppgb proteins (Galjart et al. 1988) and smaller than the apparently uncleaved Cpvl (Mahoney et al. 2001). To confirm the immunospecificity of our new antibody, αScpep1 was preabsorbed to full-length Scpep1 protein and then applied to Western blots. Under these conditions, the ∼35-kDa band is completely abrogated in all cell lines (middle panel, Figure 1B). Together, under the conditions ofour Western blotting assay and in the cells surveyed, αScpep1 detects a specific band of 35 kDa in rat and mouse cells corresponding to endogenous Scpep1.
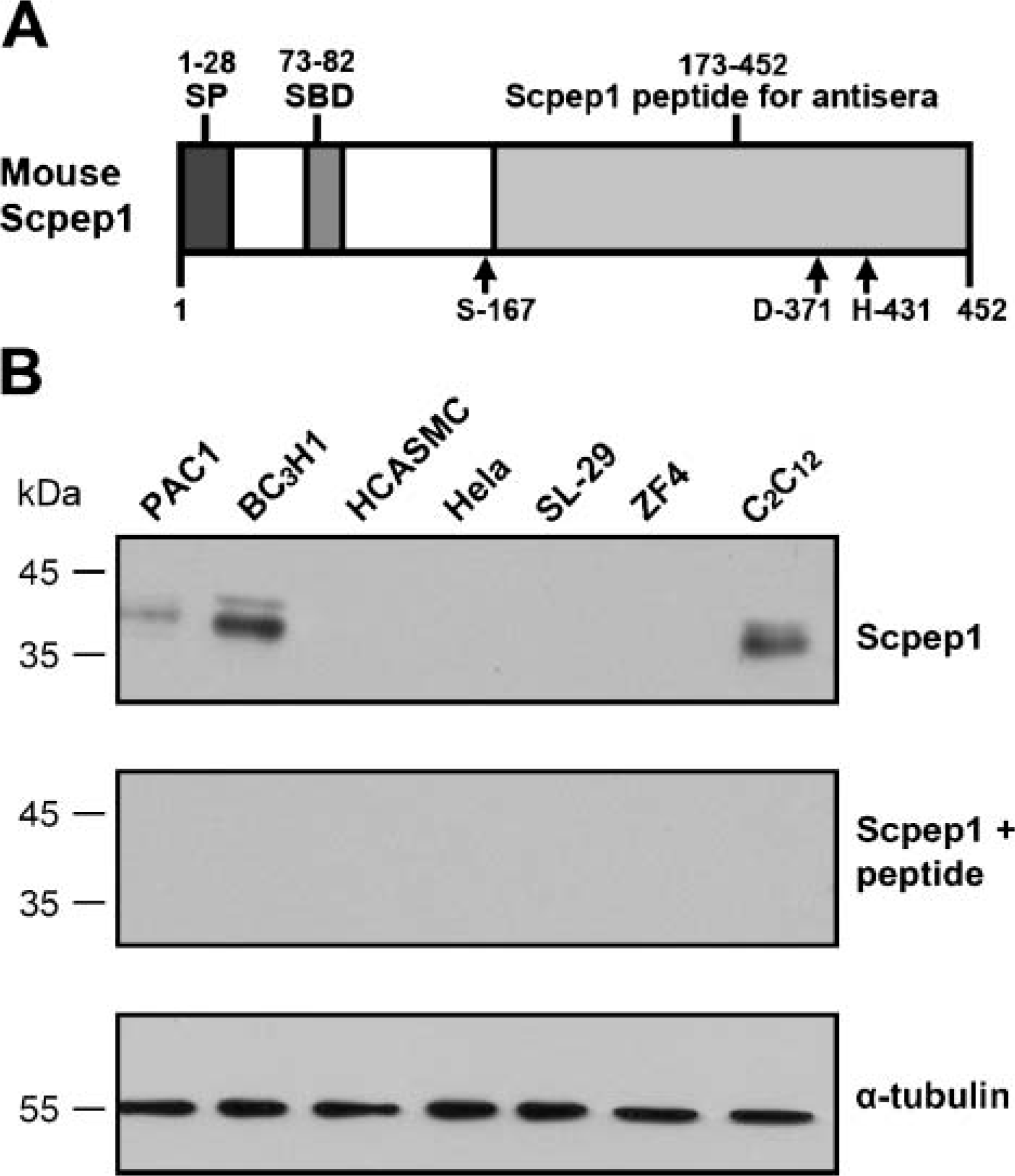
In vitro expression of retinoid-inducible serine carboxypeptidase (Scpep1). (
Expression of Scpep1 in Embryonic Mouse Tissues
To investigate the expression of Scpep1 during mouse embryonic development, staged mouse embryos (embryonic day 9.5: e9.5, e12.5, e15.5) were sagittally sectioned and immunohistochemistry performed. In the head region, Scpep1 is observed in the developing brain, choroid plexus (Figures 2A, 2D, and 2G) and ganglia (trigeminal ganglion, Figure 2G). In the thoracic region, Scpep1 is persistently observed in developing heart and aorta (Figures 2B, 2E, and 2H). Immunoreactive Scpep1 is present as early as e9.5 in trabeculae of the developing ventricular myocardium but progressively decreases in this tissue by e15.5 (Figure 2B vs. 2H). Scpep1 is also detected in the embryonic liver, esophagus, intestine, stomach, pancreas, and kidney (Figures 2C, 2F, and 2I). Together these findings indicate a wide tissue distribution of Scpep1 during mouse embryogenesis.
Expression of Scpep1 in Adult Mouse Tissues
Consistent with in vitro cell line data (Figure 1B), Western blotting with αScpep1 reveals an ∼35-kDa band in various adult tissues (Figure 3, upper panel). Interestingly, lung exhibits a slightly smaller immunoreactive band to Scpep1 (Figure 3). Expression levels of Scpep1 are high in most tissues such as bladder, esophagus, intestine, kidney, liver, pancreas, spleen, and stomach. In muscle tissues such as heart and in skeletal muscle of gastrocnemius, Scpep1 is comparatively less (Figure 3, upper panel). αScpep1 also reveals bands of larger molecular weight in brain and lung. Preabsorption of αScpep1 with full-length Scpep1 protein competes away binding to the ∼35-kDa protein in all tissues (Figure 3, middle panel). However, such preabsorption fails to compete away binding of larger molecular weight bands, indicating these are likely not to be higher molecular weight forms of Scpep1 (Figure 3, middle panel).
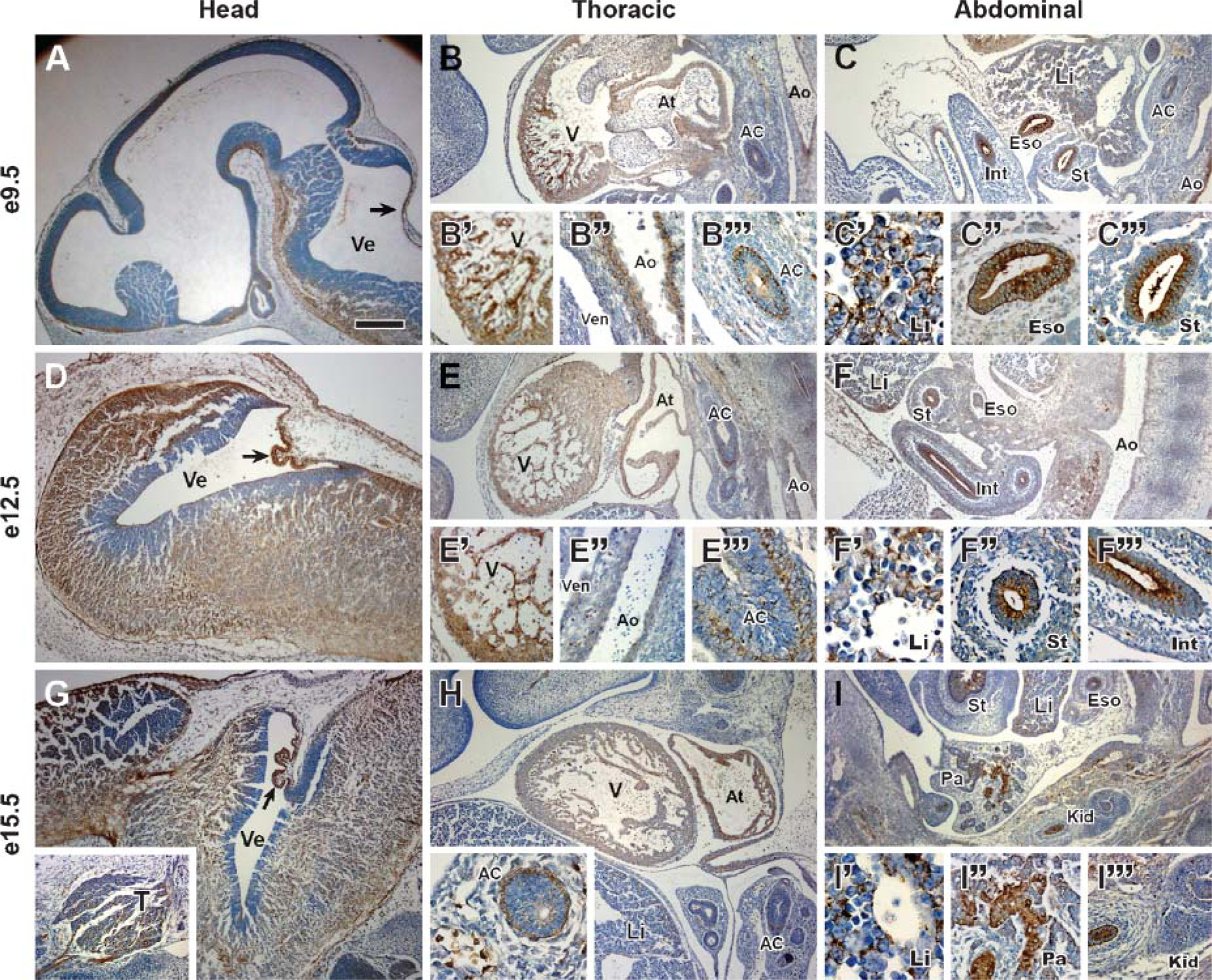
Immunohistochemistry of Scpep1 in mouse embryos. Sagittal sections at embryonic day (e9.5) (
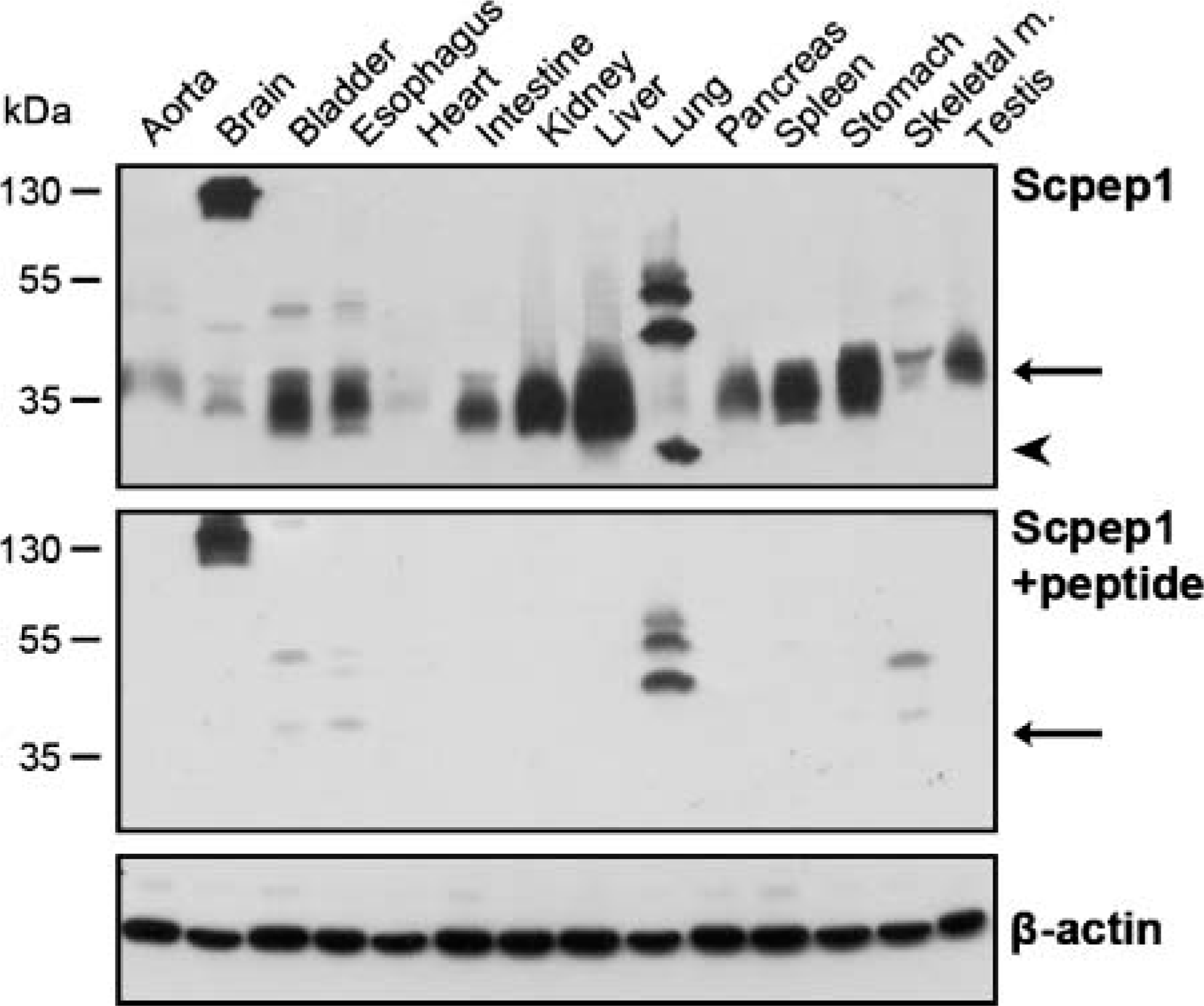
Western blot analysis of Scpep1 protein in adult mouse tissues. Upper panel, αScpep1 recognizes an ∼35-kDa band in various tissues (arrow). Arrowhead denotes smaller immunoreactive band for Scpep1 in lung. Middle panel, immunoreactivity of the ∼35-kDa band (arrow) is abolished with preabsorbed αScpep1 to full-length Scpep1 peptide. Bottom panel shows equal protein loading with β-actin control antibody.
Immunolocalization of Scpep1 in Adult Mouse Tissues
Before examining the cellular localization of Scpep1 in various adult mouse tissues, its immunospecificity was tested by immunoabsorption using the full-length Scpep1 peptide. Scpep1 is observed in SMCs of the adult aorta in mouse and rat (Figures 4A and 4C). In addition, we observe Scpep1 in the endothelial cells of the rat aorta (Figure 4C) but not in mouse aorta (Figure 4A). Strong staining is seen in the PCT epithelia of the kidney but not in the distal convoluted tubular epithelia or glomeruli (Figure 4E). Consistent with the Western data above (Figure 1B), immunostaining of Scpep1 is specific to the indicated cell types because preabsorption with the Scpep1 peptide abolishes staining (Figures 4B, 4D, and 4F).
To further clarify cell localization of Scpep1 across tissues, we performed extensive immunostaining analysis. As shown in Figure 5, Scpep1 is widely evident in vascular, nervous, digestive, respiratory, immune, and urogenital systems. In adult mouse aorta, Scpep1 is localized in the SMCs of the tunica media (Figure 5A). Consistent with decaying expression of Scpep1 in developing myocardium in Figure 2 and low expression by Western blotting in Figure 3, no Scpep1 immunoreactivity is detected in adult cardiac muscle (unpublished data). In the brain, numerous neuronal fibers are Scpep1 positive in discrete regions of the cerebral cortex (Figure 5B) but not in cerebellum and hippocampus (unpublished data). In the urinary system, PCT and macula densa of the kidney are strongly positive for Scpep1 (Figures 5C, 5C′, and 5C"). The epithelium of the bladder (Figure 5D) is also Scpep1 positive, consistent with previous mRNA studies (Chen et al. 2001). In the digestive system, epithelial cells of the esophagus are intensively stained for Scpep1 (Figure 5E). In stomach, Scpep1 is highly expressed in oxyntic glands of epithelium; surface mucous cells are only weakly positive for Scpep1 (Figures 5F and 5F′). Villi and crypt of Lieberkuhn of the intestine (Figures 5G, 5G′, and 5G") also stain strongly for Scpep1. In the accessory organs of the alimentary canal, Scpep1 is observed within hepatocytes of the liver (Figures 5H and 5H′) and islets of Langerhans in the pancreas (Figure 5I). Type II alveolar cells (Figure 5J) and macrophages (Figure 5J) in the lung also stain positive for Scpep1. Many Scpep1-positive macrophage-like cells are noted in the spleen (Figures 5K, 5K′, and 5K") and lymph nodes (Figures 5L and 5L′). Of note is the immunolocalization of Scpep1 in testis. Here, Scpep1 is most abundant in the interstitial space of the testis (Figure 5M). Higher magnification reveals abundant Scpep1 protein in Leydig cells of the connective tissue interstitium, whereas low levels of Scpep1 expression are seen in Sertoli cells and spermatogonia within the seminiferous tubules (Figure 5M′). Table 1 summarizes expression of Scpep1 in mouse embryonic and adult tissues.
Subcellular Localization of Scpep1 in PCTs of the Adult Kidney
High-powered immunohistochemistry reveals punctate staining of the epithelial cells of PCTs in kidney (Figure 6A). To further investigate the subcellular localization of Scpep1 in vivo, kidney sections were stained for Scpep1 with an immunoperoxidase technique and then processed for electron microscopy (see Materials and Methods). This assay reveals concentrated immunoreactive Scpep1 in the lysosomes of the PCTs (Figure 6B) with low or undetectable levels in other organelles. No immunoreactive Scpep1 is observed when sections are incubated with control IgG instead of αScpep1 (unpublished data).
Colocalization of Scpep1 with LAMP2 in Lysosomes
To further substantiate the subcellular location of Scpep1 in lysosomes, we costained cells for Scpep1 and LAMP2 (Chen et al. 1985). Indirect immunofluorescent double staining was performed in adenoviral Scpep1-transduced HeLa cells with anti-FLAG and anti-LAMP2 antibodies (Figures 7A–7C). Punctate expression of C-terminal FLAG-tagged Scpep1 (Figure 7A) and LAMP2 (Figure 7B) in HeLa cells reveal colocalization in the lysosomal compartment of these cells (Figure 7C). To evaluate whether such a colocalization also exists with respect to endogenous Scpep1, we stained mouse NIH 3T3 cells with αScpep1 and anti-LAMP2 antibodies for confocal microscopic analysis (Figures 7D-7F). Consistent with the adenoviral transduction experiments, punctate immunofluorescent Scpep1 is observed mainly in the cytoplasm of 3T3 cells (Figure 7D). The staining pattern of LAMP2 also reveals a punctate pattern that appears to be similar to Scpep1 (Figure 7E). Merging images reveal colocalization of the immunoreactivities of Scpep1 and LAMP2 in the lysosomes (Figure 7F). Together with the immunoelectron microscopy data, the results provide compelling support for an endosomal/lysosomal concentration of Scpep1.
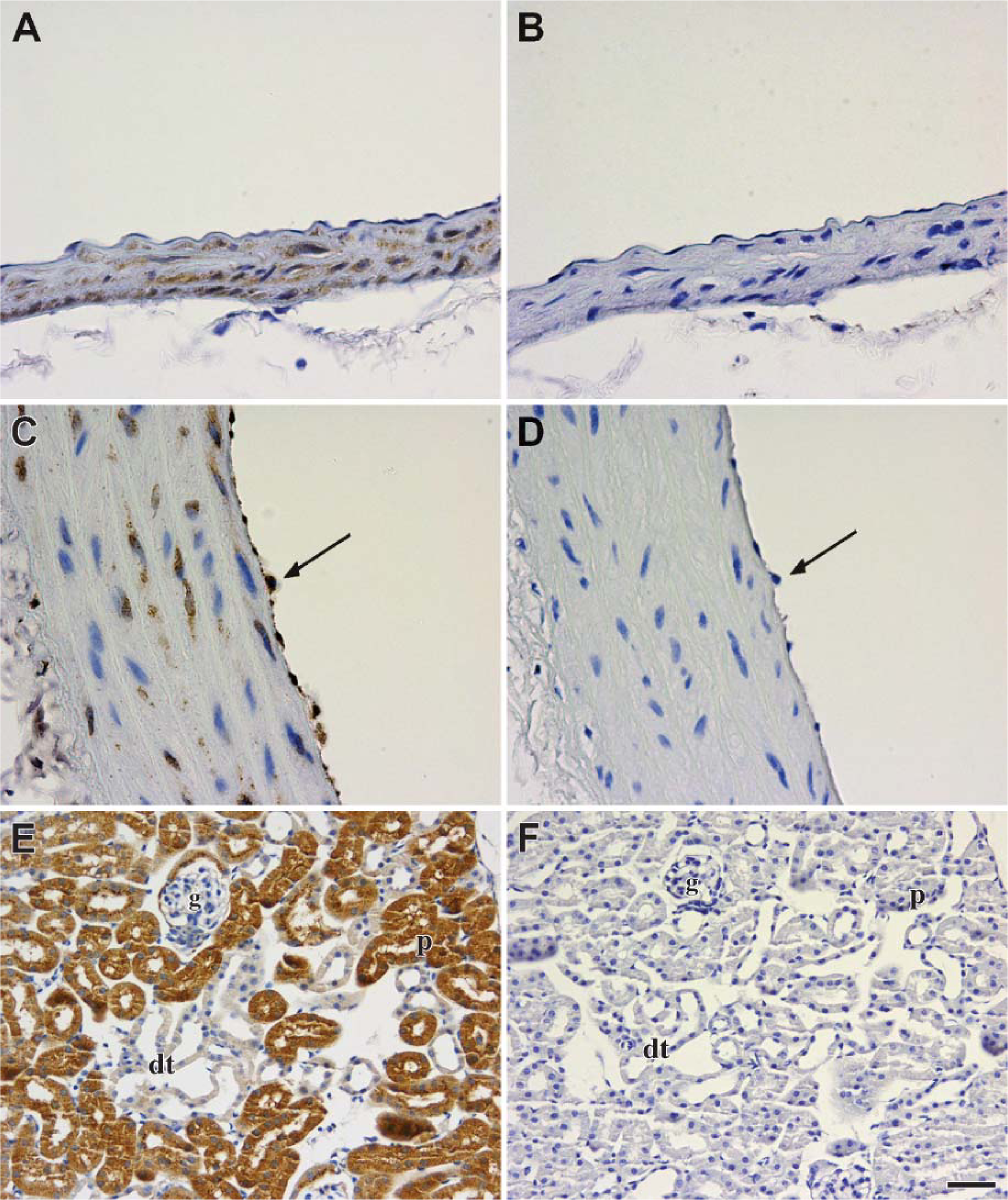
Immunospecificity of αScpep1 in adult mouse tissues. Sections of tissues were immunostained with αScpep1 (
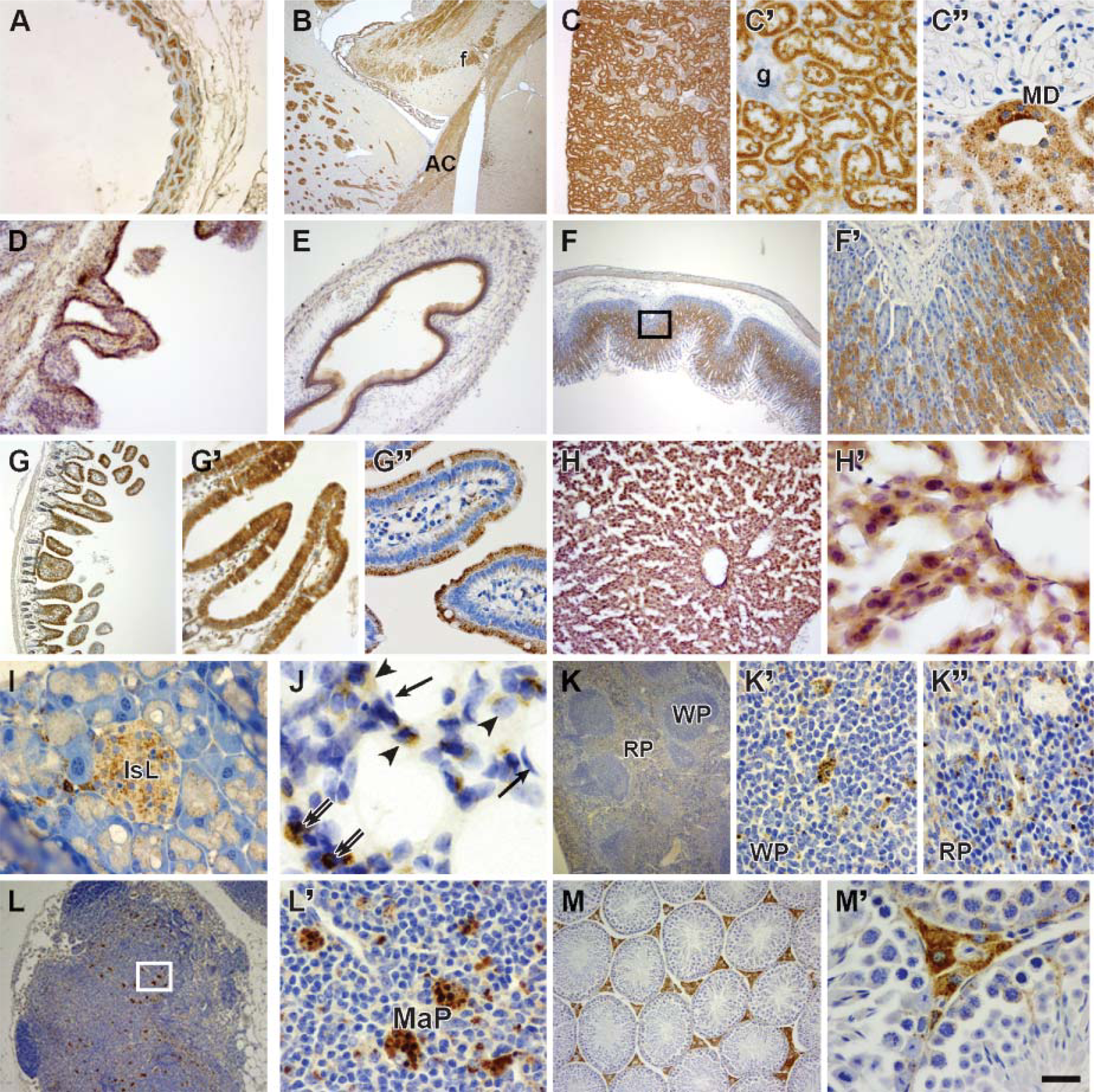
Immunolocalization of Scpep1 across adult mouse tissues. (
Discussion
In the present study, we have generated a polyclonal antibody that specifically detects rodent Scpep1. Western blot analysis reveals Scpep1 (predicted MW ∼51 kDa) to be cleaved into a mature protein of ∼35 kDa in essentially all adult tissues. Immunohistochemistry of staged embryos shows a somewhat less broad distribution of Scpep1 with detectable protein in cells of the cardiovascular, nervous, and digestive systems. Although previous studies have defined protein expression of other mammalian SCs in adulthood (Tan et al. 1993; Rottier et al. 1998; Mahoney et al. 2001), this is the first report of expression of a SC during embryogenesis.
Summary of retinoid-inducible serine carboxypeptidase (Scpep1) expression in mouse embryonic and adult tissues
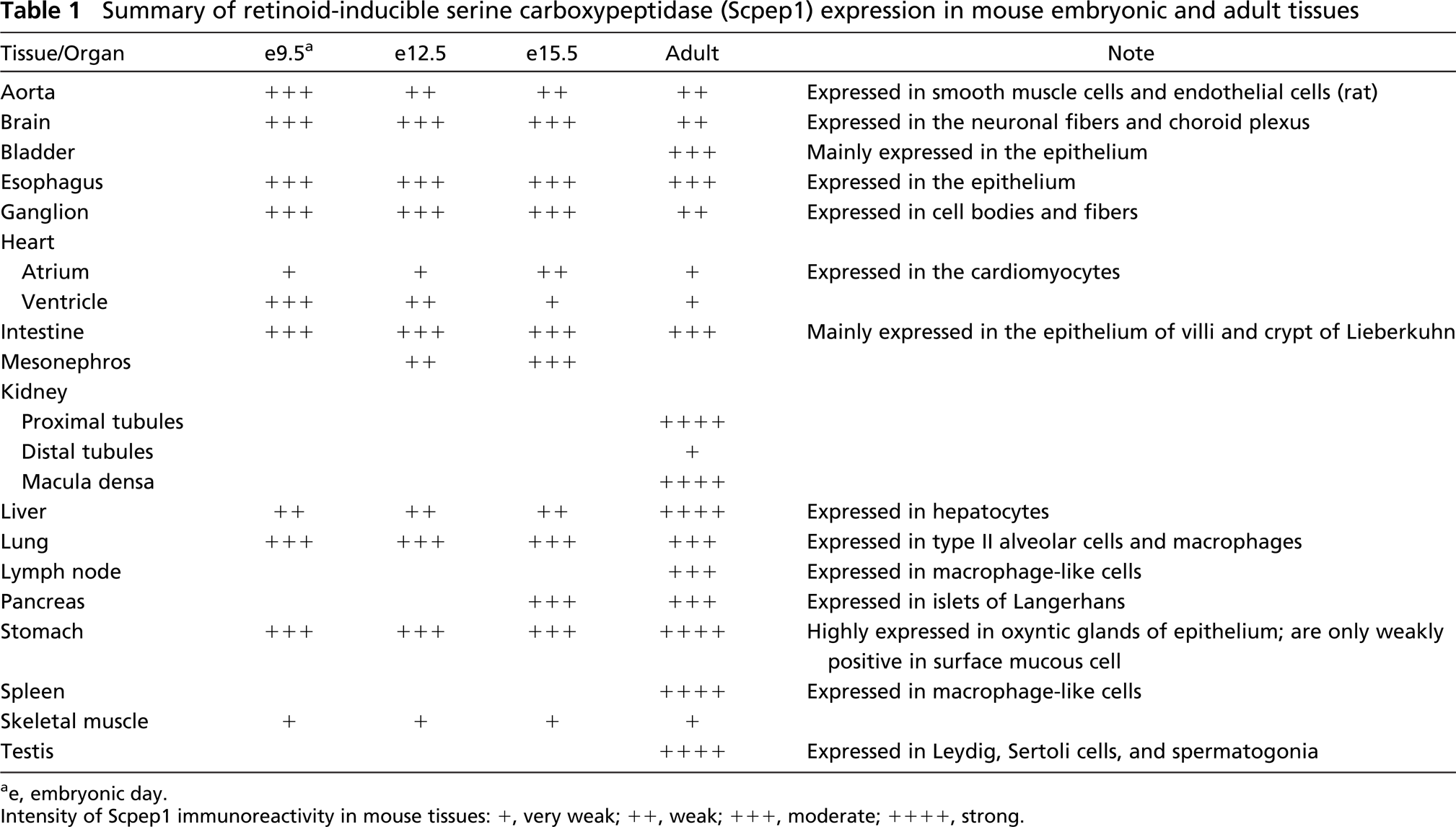
ae, embryonic day.
Intensity of Scpep1 immunoreactivity in mouse tissues: +, very weak; ++, weak; +++, moderate; ++++, strong.
Immunohistochemistry further localizes Scpep1 to specific cell types within each adult tissue such as the PCT epithelium of the kidney and macrophages of the lymph nodes and spleen, as well as endothelial and SMC of the aorta. Protein expression of Scpep1 in kidney, vessel wall, and bladder are concordant with its mRNA expression in these tissues (Chen et al. 2001). In addition to its cellular distribution, Scpep1 is concentrated ultrastructurally within lysosomes of the PCT epithelium, and confocal immunofluorescent studies show colocalization of Scpep1 with LAMP2, a lysosome-specific marker (Chen et al. 1985). Collectively, these data extend our previous mRNA studies by showing the tissue distribution of Scpep1 protein as well as its enrichment within the lysosomal compartment of the cell.
Several carboxypeptidases have been reported to be involved in the vascular system and kidney, where they function to either activate or inactivate proteins related to vasomotion and growth (Layne et al. 1998; Takai et al. 2001; Dominguez et al. 2002). For example, aortic carboxypeptidase-like protein is upregulated dramatically during vascular SMC differentiation in vitro (Layne et al. 1998). In plants, serine carboxypeptidase III plays a role in programmed cell death during the development of vascular tissue in wheat (Dominguez et al. 2002). Recent studies indicate Prcp, an angiotensin II-inactivating enzyme, functions as an endothelial cell prekallikrein activator in the renin-angiotensin system (Moreira et al. 2002; Shariat-Madar et al. 2002). Ppgb has been shown to inactivate endothelin 1, a potent SMC vasoconstrictor and mitogen, suggesting Ppgb may also play an important role in the reninangiotensin system (Jackman et al. 1992; Itoh et al. 1995; Skidgel and Erdos 1998). Ppgb, Cpvl, and Prcp have not been observed in endothelium and SMCs of aorta but are detectable in the heart (Tan et al. 1993; Rottier et al. 1998; Mahoney et al. 2001). We have shown that Scpep1 decreases in myocardium during embryogenesis but is continuously present in aorta from embryo to adult. Moreover, Scpep1 is observed in the macula densa of kidney in adult, suggesting a possible function related to the renin-angiotension system. These expression data suggest that Scpep1 may exert its function to regulate activities associated with vascular SMC during development and adulthood.
The distribution pattern of Scpep1 in metabolically active and phagocytic cells is similar to that of other SCs in mammals. For example, Scpep1 is expressed intensely in secreted and/or absorptive cells (epithelial cells of choroid plexus, intestine, and stomach; PCTs of kidney; hepatocytes; type II alveolar cells of lungs; islets of Langerhans of pancreas; and Leydig cells of testis) and phagocytic cells in immune organs (lymph nodes and spleen). Similarly, Ppgb significantly distributes in brain, choroid plexus, liver, spleen, kidney, small intestines, testis, and epididymis (Rottier et al. 1998; Luedtke et al. 2000; Korah et al. 2003a,b). Immunohistochemistry of Ppgb reveals expression in PCTs of kidney as well as in Sertoli and Leydig cells of the testis, the latter of which are highly endocytic and involved in the production, differentiation, and nourishment of sperm (Luedtke et al. 2000). In this study we show that Scpep1 is also present in PCTs and Sertoli and Leydig cells of the testis. The presence of Scpep1 in renal epithelial cells and the supporting/germ cells of testis suggest a possible role in homeostasis of the renal and reproductive systems. As with Scpep1, expression of Cpvl and Ppgb is abundantly present in macrophages (Mahoney et al. 2001; Korah et al. 2003a). We speculate that Scpep1 has similar functions with Cpvl and Ppgb, which may be involved in the process of digesting phagocytosed particles in the lysosomes of macrophages (Mahoney et al. 2001; Korah et al. 2003a).
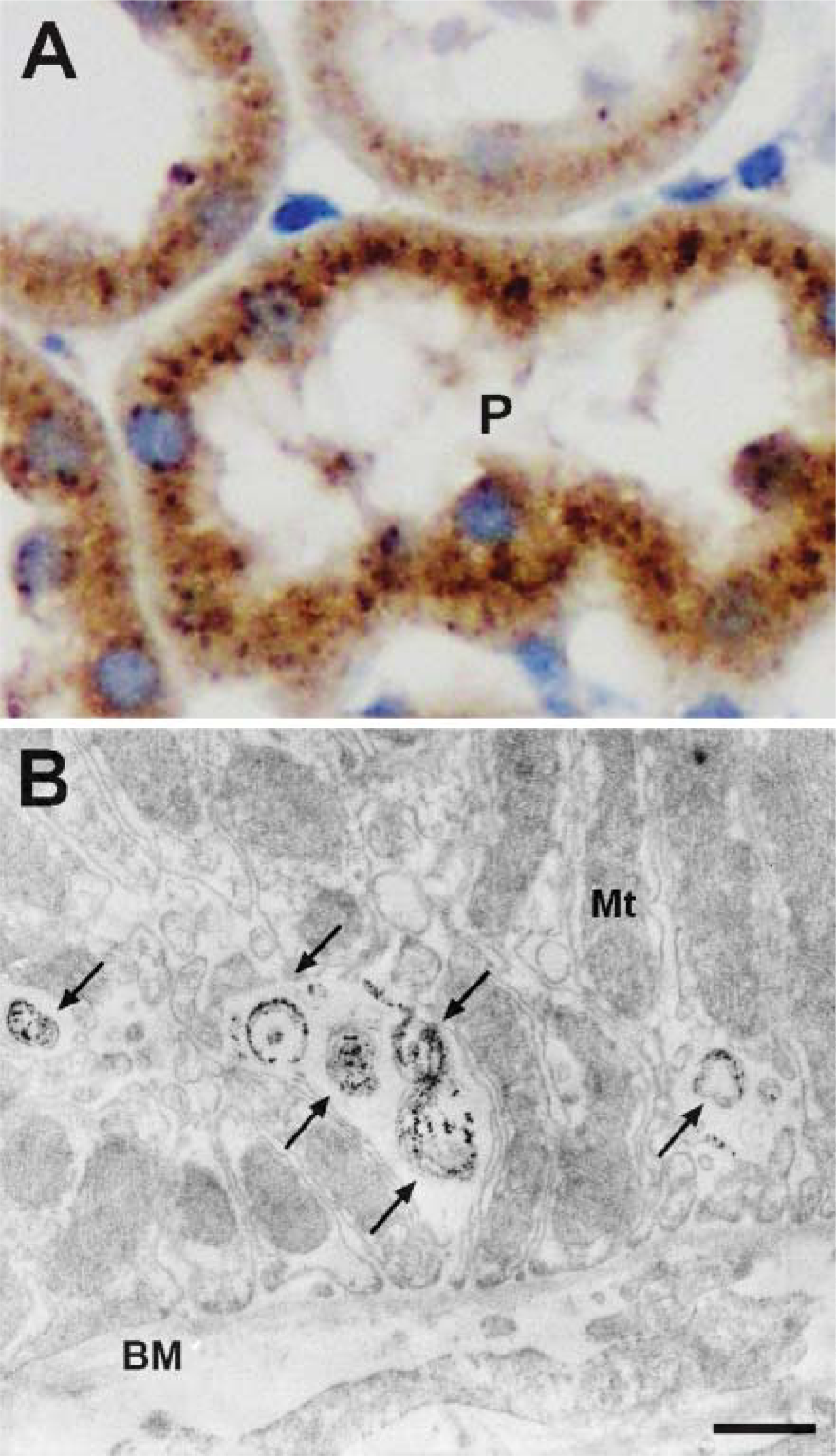
Immunolocalization of Scpep1 in kidney. (
We previously showed that exogenously expressed Scpep1 (formerly RISC) exists as an ∼51-kDa protein (Chen et al. 2001). Here, however, we report the appearance of a mature, ostensibly proteolytically cleaved ∼35-kDa protein in all tissues analyzed. Secreted Scpep1 in the concentrated conditioned medium is also cleaved to a similarly sized peptide (unpublished data). Moreover, adenoviral overexpression of a C-terminal FLAG-tagged Scpep1 yields an ∼35-kDa protein in Western blots (unpublished data). Because our Scpep1 antisera was raised to the C terminus of Scpep1 (see Figure 1), we do not see the ∼16-kDa N-terminal peptide. These data support the notion that Scpep1, like other SCs (Galjart et al. 1988; Jung et al. 1999; Parussini et al. 2003), undergoes proteolytic cleavage to a mature active protease. Detailed mechanisms underlying Scpep1 cleavage will require further study.
Ppgb and Prcp have been shown to be lysosomal glycoproteins (Morreau et al. 1992; Sleat et al. 2005), and Cpvl is also a possible lysosomal protein (Mahoney et al 2001). In recent studies, a series of experiments including MPR affinity purification, two-dimensional gel electrophoresis, and mass spectrometry have demonstrated Scpep1 to be associated with the MPR (Kollmann et al. 2005; Sleat et al. 2005). Exogenous V5-tagged Scpep1 is colocalized in lysosomes with antibodies against the V5-tag and LAMP1, a lysosomal membrane protein (Kollmann et al. 2005). In the present report, we have direct evidence demonstrating endogenous Scpep1 colocalizes with LAMP2 in lysosomes, a finding that is further supported by our immunoelectron microscopic data. Together, Scpep1 appears to be a novel lysosomal SC requiring MPR for targeting to lysosomes.
To date, >50 lysosomal proteins have been found including many acid hydrolases and associated proteins for the degradation of macromolecules in the endolysosomal system. More than 40 lysosomal storage diseases have been identified with virtually all of them associated with a mutated or deficient lysosomal-associated gene, at least one of which is a SC. For example, Ppgb deficiency in humans causes the degradation of β-galactosidase and neuraminidase in galactosialidosis (OMIM #256540) (Galjart et al. 1988), a disease associated with Ppgb's protective function of these lysosomal proteases. Although Scpep1 shares homology to Ppgb and is localized to lysosomes in similar cells (e.g., PCT epithelium of kidney), it apparently cannot compensate for loss in Ppgb's stabilization of β-galactosidase/neuraminidase. In addition, we have been unable to demonstrate proteolytic activity of Scpep1 for any of Ppgb's substrates (e.g., endothelin-1), and exhaustive yeast two-hybrid screening assays have failed to reveal any evidence for substrate binding to Scpep1 (unpublished data). Together these findings suggest that the function of Scpep1 is separate and distinct from that of Ppgb and perhaps other SCs as well. Inactivating or attenuating the expression of Scpep1 is the focus of ongoing work in this laboratory as a means to define the role of Scpep1 in development and homeostatic processes.
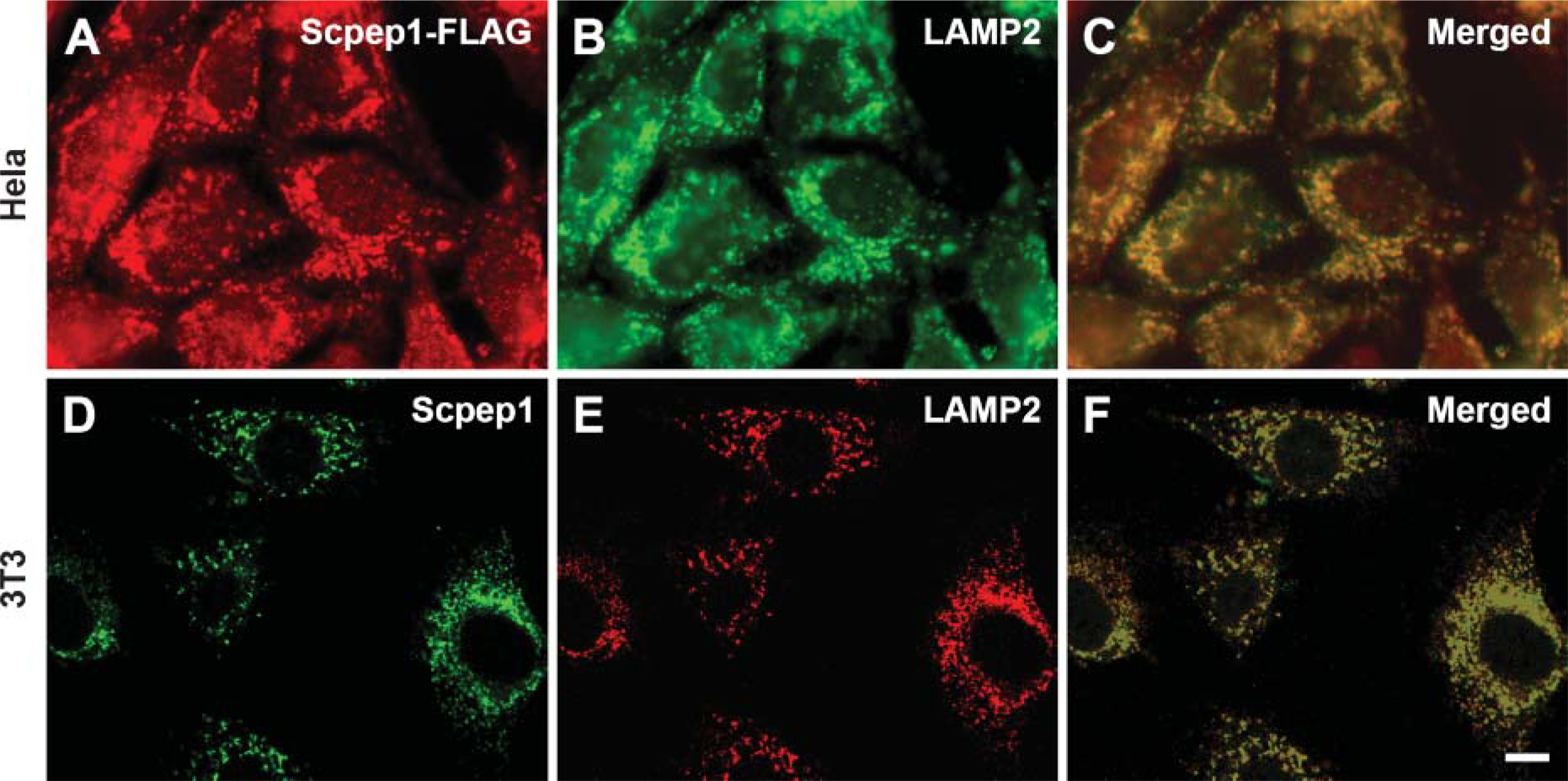
Localization of Scpepl in lysosomal compartment. Indirect immunofluorescent double labelings were performed in Ad-Scpep1-transduced HeLa cells with anti-FLAG and anti-lysosome-associated membrane peptide (LAMP2) antibodies (
In summary, we report the characterization of a new antibody to Scpep1. By Western blot and immunohistochemistry, Scpep1 is located to a variety of organ systems from the embryonic stage to adulthood. In addition, Scpep1 appears to be cleaved to an ∼35-kDa mature form and expressed in the endosomal/lysosomal compartments. The precise functions of Scpep1 will require a multifaceted approach including genetic inactivation, atomic structure, and substrate screening assays. The antibody to Scpep1 described here will be a vital reagent in these future studies.
Footnotes
Acknowledgements
This work was supported by the National Institutes of Health (Grants HL-62572 and HL-070077). J.M.M is an Established Investigator of the American Heart Association (Award 0340075N).
The authors thank Ms. Hou-Yu Chiang for performing the immunoelectron microscopy study and Dr. Jane Sottile for assisting with the confocal microscopy.
