Abstract
Caveolin-1 is a member of a subset of intracellular proteins that regulate endothelial nitric oxide synthase (eNOS) activity. In caveolae, caveolin-1 inhibits eNOS activity via a direct interaction with the enzyme. Previous work has indicated that both eNOS and caveolin-1 are also localized at the perinuclear Golgi complex. Whether caveolin-1 is involved in eNOS regulation in this cell compartment is unknown. Here we studied the localization of eNOS and caveolin-1 in the perinuclear region of primary bovine aortic endothelial cells. By immunofluorescence microscopy we show that both eNOS and caveolin-1 co-localize with Golgi markers. On treatment of the cells with the microtubule-depolymerizing drug nocodazole, the Golgi complex is scattered and caveolin-1 is found in vesicles at the periphery of the cell, while eNOS is localized at large structures near the nucleus. The nocodazole-induced redistribution of eNOS is similar to that of
T
At present it is not clear whether eNOS that is localized at the Golgi complex is active or might be activated by specific stimuli. If Golgi-resident eNOS can be activated, it may also be associated with caveolin-1. Caveolin-1 is reported to be present at the Golgi complex and is believed to recycle between this organelle and the caveolae at the plasma membrane (Conrad et al. 1995). In addition, caveolin-1 may also be present at the ER (Smart et al. 1996; Pol et al. 2001). The hypothesis that eNOS is already associated with caveolin-1 at the Golgi has been postulated (Garcia-Cardena et al. 1996a). If this were the case, eNOS and caveolin-1 need to be localized at the same subcompartment of the Golgi complex.
In this study we used the microtubule-depolymerizing agent nocodazole to determine whether eNOS and caveolin-1 are localized at the same subcompartment of the Golgi. By depolymerizing the microtubules, nocodazole induces fragmentation of the Golgi complex (Cole et al. 1996). Nocodazole treatment is a well-established method to localize proteins to distinct Golgi subcompartments (Chavrier et al. 1990; Rojo et al. 1997; Yang and Storrie 1998; Zhang et al. 1999). Here we show that treatment of bovine aortic endothelial cells (BAECs) with nocodazole causes segregation of eNOS- and caveolin-1-containing vesicular structures, indicating that these proteins are not present at the same subcompartment of the Golgi and that caveolin-1 has no regulatory role in eNOS function at this compartment.
Materials and Methods
Materials
BAECs were purchased from Clonetics (San Diego, CA). Normal goat serum and secondary fluorescent antibodies were obtained from Jackson Immunochemicals (West Grove, PA). Monoclonal anti-eNOS antibody (immunofluorescence) was purchased from BioMol (Plymouth Meeting, PA) and monoclonal anti-eNOS (immuno-EM), anti-caveolin-1 (#C37120), and anti-GM130 from Transduction Laboratories (San Diego, CA). Polyclonal anti-caveolin-1 (#C13630), anti-myc, and anti-mannosidase II antibodies were obtained from Transduction Laboratories (San Diego, CA), Upstate Biotechnologies (Lake Placid, NY), and K.W. Moremen (University of Georgia; Athens, GA), respectively. Polyclonal anti-GOS-28 antibody was raised in rabbits using the His6-tagged cytoplasmic portion of GOS-28 (His6-GOS-28-ATM) as immunogen (Gerez et al. 2000). Anti-sec22 antibody was a generous gift from Richard Scheller (Stanford University School of Medicine; Stanford, CA). Myc-α-2,6-sialyltransferase cDNA in SMH4 vector was kindly provided by Sean Munro (MRC, Cambridge, UK). Nocodazole was obtained from Sigma (St Louis, MO).
Cell Culture
Primary BAECs were cultured according to the supplier's instructions. BAECs were cultured in EGM BulletKit medium (Clonetics), split 1:6 on reaching confluence, and used between passages 3 to 7. For immunofluorescence, cells were grown on glass coverslips in 24-well dishes. The different passage number of the cells used for the experiments did not affect the localization of any of the studied proteins. All experiments were performed at 37C. Because of a better morphology, cells were used at a subconfluent stage. Cell confluence did not affect the Golgi localization of any of the proteins described in this study.
For transfection of BAECs with the eukaryotic expression vector pcDNA3.1 (Invitrogen; Carlsbad, CA) containing myc-α-2,6-sialyltransferase cDNA (cloned in HindIII/XbaI sites), cells were incubated with DNA-Lipofectamine complexes according to the supplier's instructions (Life Technologies; Rockville, MD).
Immunofluorescence Microscopy
BAECs were immunostained by indirect fluorescent labeling. Cells were incubated with 20 μM nocodazole for the time periods indicated. To study the reversibility of the nocodazole-induced Golgi scattering, cell cultures were incubated with nocodazole for 60 min, washed once with preconditioned medium without nocodazole, and incubated with medium for the indicated periods of time.
After nocodazole incubation, cells were fixed with 3% paraformaldehyde, permeabilized, blocked with normal goat serum, incubated with primary antibodies for 60 min, washed three times with PBS, incubated with secondary antibodies (goat anti-rabbit-FITC and goat anti-mouse-Texas Red) for 30 min, washed three times with PBS, and embedded in Mowiol. Confocal laser scanning microscopy was performed using a Leica TCS 4D system. FITC and Texas Red label were scanned sequentially and overlaid using Adobe Photoshop software. Control cells that were labeled with the primary or secondary antibody only showed no fluorescence (not shown).
Immunoelectron Microscopy
BAECs were fixed by mixing the culture medium 1:1 with double-strength fixative (4% paraformaldehyde, 2% acrolein in 0.1 M sodium phosphate buffer, pH 7.4) at ambient temperature. After 4 min the mixture was replaced by single-strength fixative (2% paraformaldehyde, 1% acrolein in phosphate buffer) and fixation proceeded for 2.5 hr at room temperature. Then cells were scraped and processed for ultracryotomy and immuno-EM as described (Raposo et al. 1997). Briefly, 60-nm-thick cryosections were thawed and immunolabeled for eNOS by incubating them with a monoclonal anti-eNOS antibody, rabbit anti-mouse IgG, and protein A-gold successively. Sections were then stained with uranyl acetate and dried in methylcellulose. Control cryosections that were labeled with secondary antibody and protein A-gold only showed no labeling (not shown).
Results
Immunofluorescence observations revealed a typical perinuclear localization for eNOS (Figure 1A). By immuno-EM these eNOS-positive areas were recognized as the Golgi apparatus, in which eNOS appeared to be distributed in a rather diffuse fashion over all recognizable Golgi elements, including the cisternae and vesicular/tubular membranes in their surroundings (Figure 1C). Anti-caveolin-1 antibody gave identical perinuclear labeling patterns by immunofluorescence as eNOS (Figure 1B). However, immuno-EM was not successful for caveolin-1 localization, which is probably due to differences in the processing of the cells. Therefore, nocodazole in combination with immunofluorescence was used to study the localization of eNOS and caveolin-1 in detail and to see whether these proteins are actually present at the same sub-compartment of the Golgi. BAECs were treated with nocodazole for various periods of time, after which the cells were fixed, labeled with monoclonal antibodies against eNOS and caveolin-1, and analyzed by confocal immunofluorescence microscopy (Figure 2). An antibody against the Golgi SNARE [soluble
The reversibility of the drug treatment was also studied. BAECs were incubated for 1 hr with nocodazole, washed, and incubated for various time periods in the absence of nocodazole (Figure 3). eNOS and GOS-28 returned to their original perinuclear Golgi-like distribution between 15 and 60 min after drug removal. Most of the caveolin-1 co-localized with GOS-28 (and therefore also with eNOS) within 10 minutes after the nocodazole was removed from the cells. This co-localization was complete after 15 min. This clearly demonstrates that on nocodazole removal, the caveolin-1-positive vesicles quickly redistribute from the cell periphery towards the large nocodazole-induced eNOS- and GOS-28-containing Golgi fragments before these structures regain their original perinuclear morphology.
To define the Golgi subcompartments in which eNOS and caveolin-1 are localized, BAECs were incubated for 1 hr with nocodazole and labeled with monoclonal antibodies directed against eNOS, caveolin-1, and Golgi matrix protein GM130 (Nakamura et al. 1995) in combination with polyclonal antibodies directed against caveolin-1, mannosidase II (Moremen and Robbins 1991), myc-tag (for the detection of transfected myc-tagged sialyltransferase) (Taatjes et al. 1988), and Golgi SNAREs sec22 (Hay et al. 1998), and GOS-28 (Figure 4). In the absence of nocodazole, both eNOS and caveolin-1 co-localized with the Golgi protein GOS-28 in the perinuclear Golgi complex, which was also positive for all of the other studied proteins (Figure 4, left panels). In immunofluorescence microscopy, the monoclonal anti-caveolin-1 antibody recognized only the caveolin-1 present in the Golgi and did not react with cell surface caveolin-1, as has been described for other anti-caveolin-1 antibodies directed against the COOH-terminus of caveolin-1 (Luetterforst et al. 1999). To ascertain the validity of the monoclonal anti-caveolin-1 antibody, a double labeling with a polyclonal anti-caveolin-1 antibody was performed. The polyclonal antibody predominantly stained caveolae at the plasma membrane. In addition, the antibody immunolabeled Golgi and Golgi fragments that were also labeled by the monoclonal anti-caveolin-1 antibody (Figure 4C). As for the monoclonal antibody, the polyclonal anti-caveolin-1 antibody did not label the nocodazole-induced Golgi fragments that were positive for eNOS. In nocodazole-treated cells, eNOS co-localized with the
Because our data indicated that caveolin-1 was not present in Golgi stacks or TGN, BAECs were double labeled with antibodies against caveolin-1 and sec22 (Figure 4G). Sec22 is a marker for the intermediate compartment (IC). The sec22 antibody labeled the Golgi region as well as peripheral vesicular structures, most likely representing vesicular tubular clusters (VTCs) near the Golgi and peripheral VTCs adjacent to ER exit sites, respectively (Hay et al. 1998; Chao et al. 1999). In nocodazole-treated BAECs, caveolin-1-positive vesicles were also labeled with anti-sec22 antibody. Strikingly, in these cells a minor part of the sec22-positive structures was not labeled by the anti-caveolin-1 antibody. This may account for the presence of a relatively small amount of sec22 in the
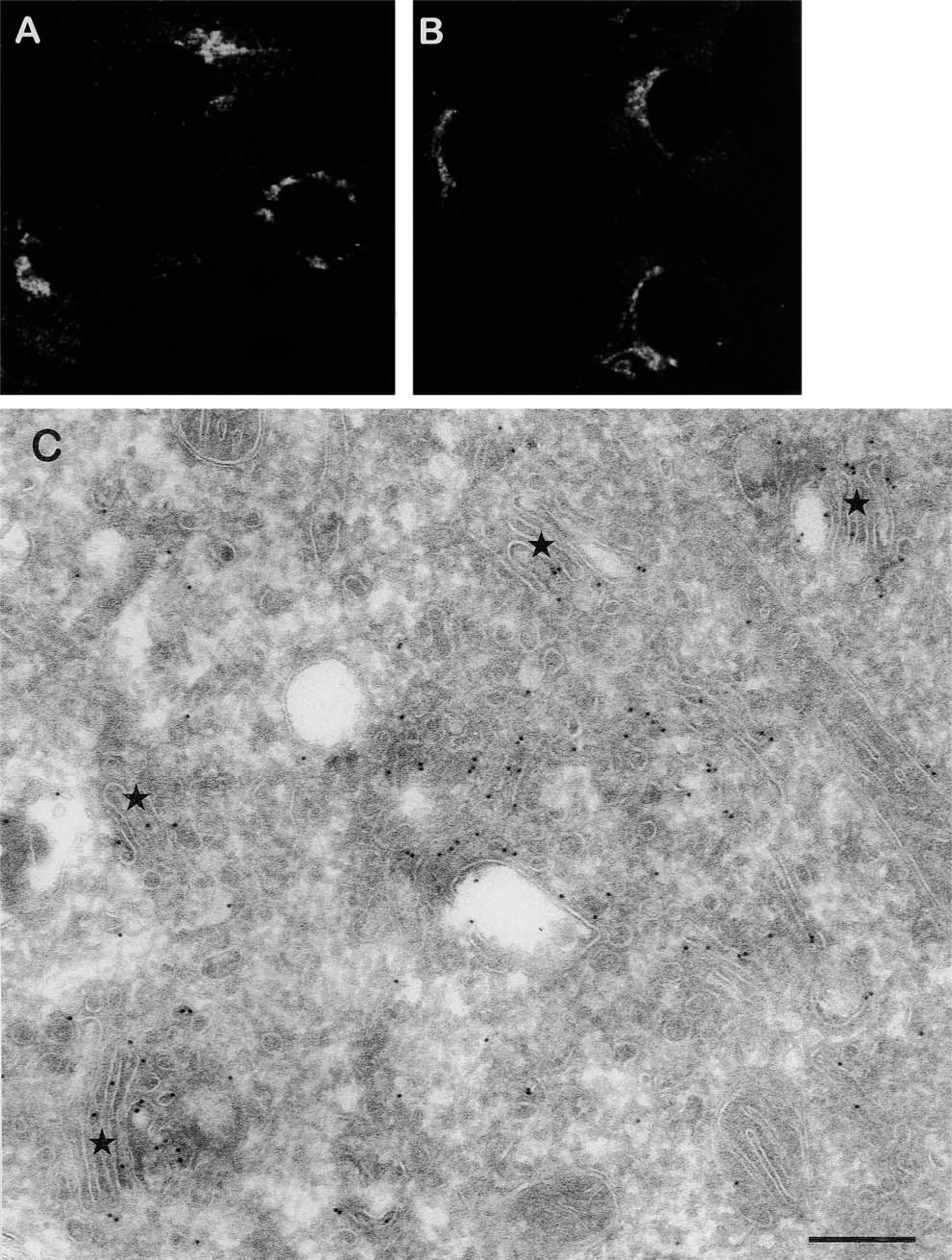
Localization of eNOS and caveolin-1 in a perinuclear region. (
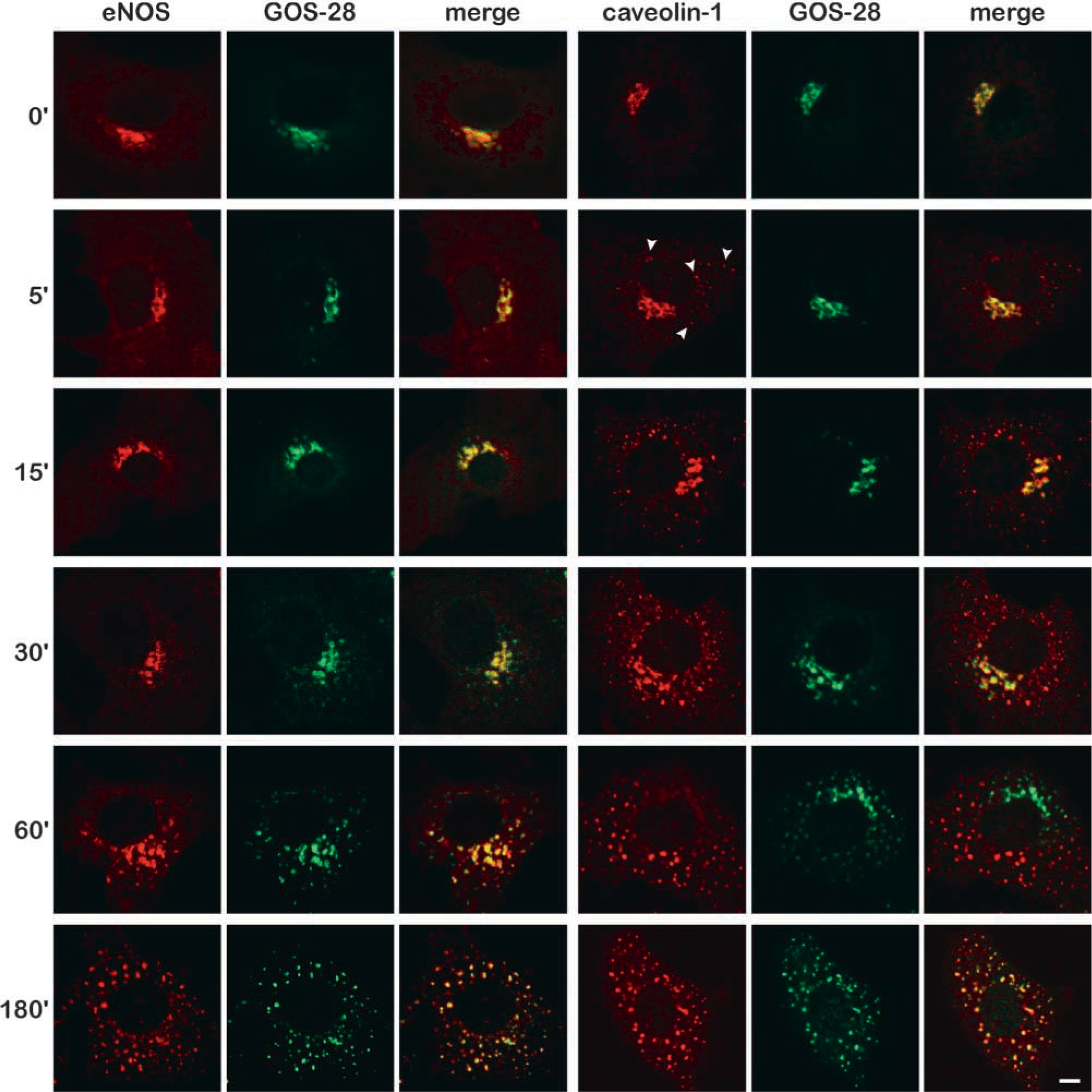
Kinetics of nocodazole-induced redistribution of eNOS and caveolin-1. BAECs were incubated for the indicated periods of time (0–180 min) with 20 μM nocodazole, fixed, and immunostained with antibodies against eNOS and GOS-28 (left panels) or antibodies against caveolin-1 and GOS-28 (right panels) and subsequently with fluorescent secondary antibodies. Third image in each panel shows an overlay of the images of the indicated antibodies (yellow staining indicates co-localization). Arrowheads in caveolin-1 image at 5 min of nocodazole treatment indicate peripheral vesicles positive for caveolin-1 that are rapidly formed in nocodazole-treated cells. Bar = 5 μm.
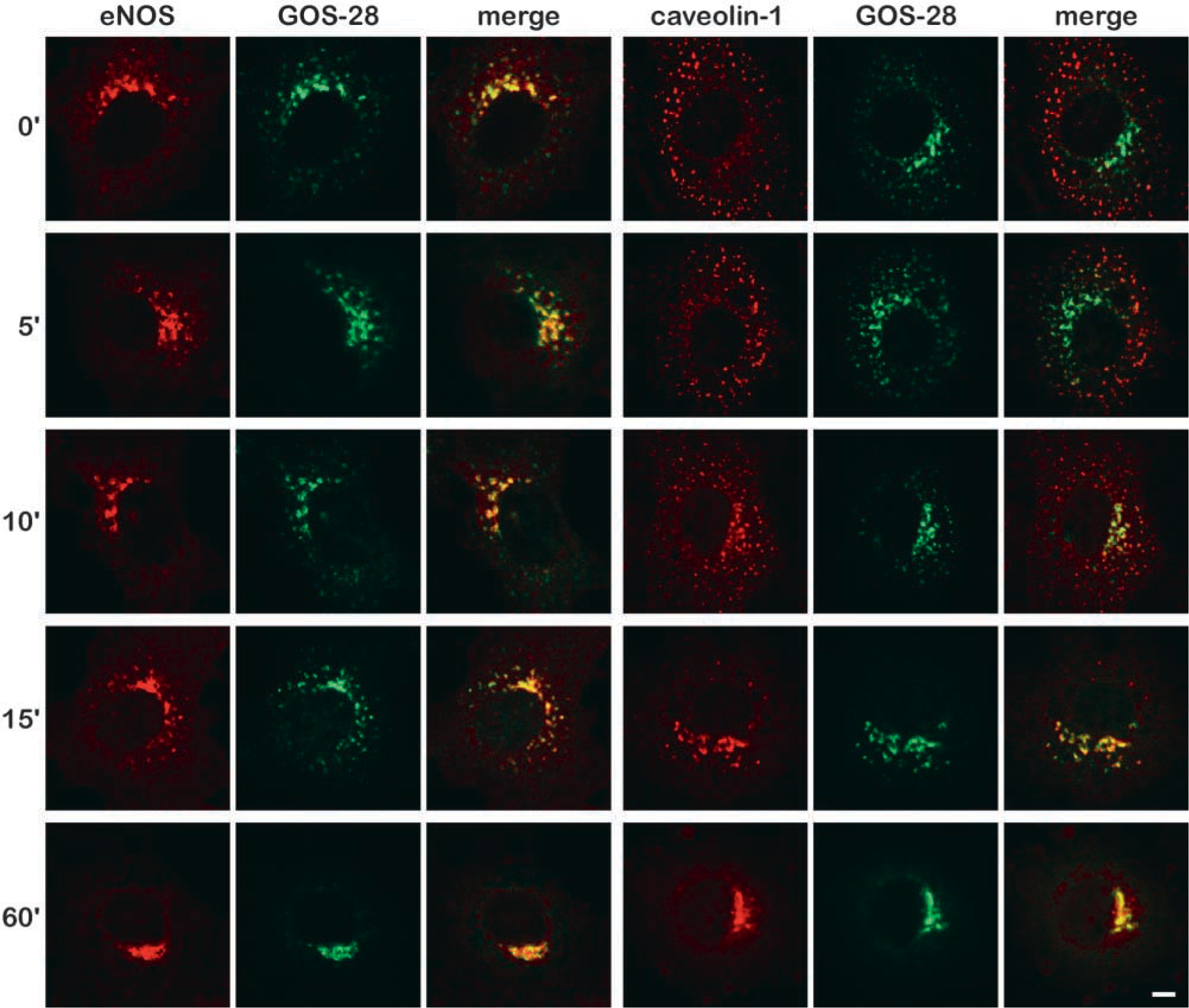
Kinetics of redistribution of eNOS and caveolin-1 in nocodazole-treated cells on removal of nocodazole. BAECs were incubated for 60 min with 20 μM nocodazole and for the indicated periods of time (0–60 min) without nocodazole, fixed, and immunostained with antibodies against eNOS and GOS-28 (left panels) or antibodies against caveolin-1 and GOS-28 (right panels) and subsequently with fluorescent secondary antibodies. Third image in each panel shows an overlay of the images of the indicated antibodies (yellow staining indicates co-localization). Bar = 5 μm.
Discussion
The data presented here demonstrate that eNOS and caveolin-1 are localized at distinct perinuclear cell compartments that behave differently when exposed to the Golgi-disrupting drug nocodazole. A 1-hr incubation of BAECs with nocodazole results in a shift in the cellular distribution of eNOS and caveolin-1 from a dense perinuclear Golgi-like structure into a vesicular staining pattern, with virtually no overlap between eNOS and caveolin-1. The caveolin-1-positive vesicles, in contrast to the structures containing eNOS, are highly dynamic. Their formation is initiated after 5 min of drug treatment and within 10 min after nocodazole removal most of these vesicles can no longer be distinguished from eNOS-containing structures. Unfortunately, it was impossible to confirm these data by immuno-EM because in these cells none of the caveolin-1 antibodies that were tested labeled caveolin-1 in or near the Golgi complex using immuno-EM techniques.
The Golgi markers GOS-28, GM130, mannosidase II, α-2,6-sialyltransferase, and the IC marker sec22 were studied to determine the perinuclear (sub)compartments where eNOS and caveolin-1 are localized. The Golgi v-SNARE GOS-28 is localized predominantly at vesicles at the terminal rims of the Golgi stacks, where it is involved in intra-Golgi transport (Nagahama et al. 1996). GM130 is a Golgi matrix protein and plays a role in maintaining the
Yang and Storrie (1998) have demonstrated that, in nocodazole-treated cells,
The Golgi localization of eNOS is dependent on its first 35 amino acid residues (Liu et al. 1997). Its presence at the Golgi is mediated by myristoylation at its amino-terminus (Gly-2), whereas eNOS palmitoylation (at Cys-15 and −26) is not required for eNOS localization at the Golgi but for targeting of eNOS to caveolae (Sessa et al. 1995; Garcia-Cardena et al. 1996b). Because palmitoyl-deficient eNOS shows a marked decrease in NO-generating capacity in intact cells, although the activity of purified mutant eNOS is not impaired, the localization of eNOS at the Golgi complex is required but not sufficient for proper NO synthesis (Liu et al. 1996). Therefore, it is unlikely that Golgi-localized eNOS produces significant amounts of NO. The lack of co-localization of eNOS and caveolin-1 within the Golgi apparatus suggests that within this compartment eNOS is kept inactive by another interacting protein. Other potential mechanisms that may be responsible for this include phosphorylation (Fleming et al. 2001) and the (local) absence of factors that are required for its enzymatic activity (e.g., calcium, L-arginine, and tetrahydrobiopterin). eNOS that is present in the Golgi complex does not represent a silent eNOS pool. Using kinetic fluorescence techniques on eNOS-GFP chimeric proteins in living cells, it was shown that eNOS is rapidly transported to and from the Golgi complex (Sowa et al. 1999). In addition, nocodazole treatment causes a 35% decrease in eNOS activity (data not shown), which might indicate that the localization of eNOS at the Golgi complex is important for its cellular regulation.
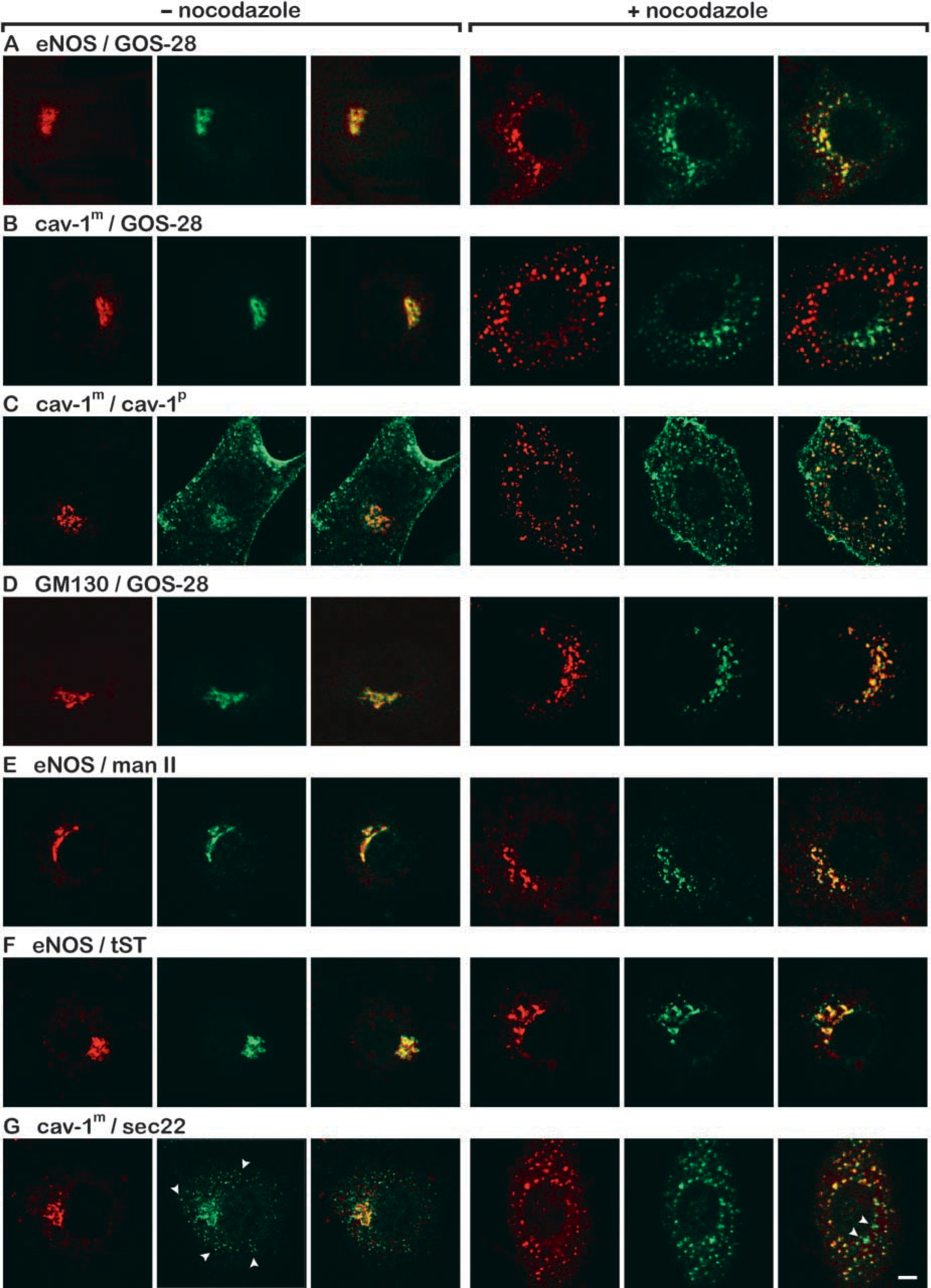
Nocodazole-induced redistribution of Golgi proteins. BAECs were incubated for 60 min in the absence (left panels) or presence (right panels) of 20 μM nocodazole, fixed, and indirectly immunolabeled with the indicated antibodies. Red staining represents immunolabeling with antibodies against eNOS (
Previous studies have shown that in BAECs eNOS is activated by shear stress (Buga et al. 1991), estradiol (Goetz et al. 1999), and VEGF (He et al. 1999). Because eNOS in the Golgi complex is not likely to be involved in the production of NO, eNOS will also be localized at caveolae at the cell surface. However, in our studies we were unable to detect much eNOS at the plasma membrane of BAECs using immunofluoresence techniques. This is most probably caused by a relatively low amount of eNOS at the plasma membrane compared to the Golgi complex where, in addition, the high density of membranes accentuates its presence. That BAECs do express eNOS at the plasma membrane is evident from other studies (Garcia-Cardena et al. 1996b; Kim et al. 1999).
Our studies indicate that eNOS and its inhibitor caveolin-1 do not co-localize within the Golgi complex. Because Golgi-resident eNOS is probably not involved in NO production, this may imply the presence of other regulatory proteins in the Golgi complex that reduce eNOS activity. An important step in understanding the complex cellular regulation of eNOS will now be to reveal the mechanism by which eNOS activity is regulated in this cell compartment.
Footnotes
Acknowledgments
Supported by grants to R. Govers from the Netherlands Organization for Scientific Research (NWO; 902-26-224) and from the Netherlands Heart Foundation (99.041).
We thank Dr Richard Scheller for the anti-sec22 antibody, Dr Sean Munro for the myc-α-2,6-sialyltransferase cDNA-containing SMH4 plasmid, and Dr Judith Klumper-man for stimulating discussions.
