Abstract
The heat shock proteins are a family of stress-inducible proteins that act as molecular chaperones for nascent proteins and assist in protection and repair of proteins whose conformation is altered by stress. HSP72 and HSP73 are two major cytosolic/nuclear stress proteins of mammalian cells, with extensive sequence homology. HSP73 is constitutively expressed, whereas HSP72 is highly stress-inducible. However, it is unclear why two isoforms are expressed and whether these two proteins have different functions in the cell. To assist in the delineation of function, we have completed a detailed study of the localization of HSP72 and HSP73 in the cell before and after heat stress, using two different methods of detection. By indirect immunohistochemistry, the localization of these two proteins is similar, cytoplasmic and nuclear in nonstressed cells with a translocation to nucleoli immediately after heat. By the more sensitive immunogold electron microscopy technique, differences in localization were noted. In nonstressed cells, HSP72 was primarily nuclear, localized in heterochromatic regions and in nucleoli. HSP73 was distributed throughout the cell, with most cytoplasmic label associated with mitochondria. Mitotic chromosomes were also heavily labeled. After stress, HSP72 concentrated in nuclei and nucleoli and HSP73 localized to nuclei, nucleoli, and cytoplasm, with increased label over mitochondria. These differences in localization suggest that the HSP72 and HSP73 may associate with different proteins or complexes and hence have different but overlapping functions in the cell.
H
The functional specificity of the two cytosolic isoforms, HSP72 and HSP73, has not yet been resolved in mammalian cells. HSP70 genes contain an ATPase domain, a peptide binding domain, a nuclear localization signal (NLS), and a carboxy-terminal domain of unknown but essential function (Zhu et al. 1996).
Some differences in peptide binding specificity have been found among different isoforms, e.g., between HSP73, DnaK (the bacterial homologue of HSP73), and BiP/GRP78 (Fourie et al. 1994) but, to our knowledge, no comparison has been made between mammalian HSP73 and HSP72. In the yeast
The observation that the development of thermotolerance, a transient resistance to heat shock induced by prior mild heating, correlates with the induction of HSP72 suggests that this protein targets stress-damaged proteins to a greater extent than the constitutively expressed HSP73 (Li and Werb 1982; Lee and Lai 1995). Furthermore, Cheng and Lai (1994) report that HSP73 is associated with intermediate filaments whereas HSP72 is associated with tubulins. However, in other studies we have shown that HSP73 appears to be as effective as HSP72 in reactivating heat-damaged luciferase expressed in mammalian cells (Tavaria et al. unpublished data).
Differences in function between the cytosolic HSP70 isoforms may be reflected in differences in intracellular localization before and after stress. HSP72 is a very minor protein not usually detected in rodent cells before stress, but in human cells it shows basal expression and is further induced by stress. Previous studies in human HeLa cells using indirect immunofluorescence have shown that HSP72 and HSP73, which share over 95% sequence identity, are present in both the cytosol and nucleus in resting cells and become predominantly nuclear during and soon after heat stress (Welch and Feramisco 1984; Ohtsuka et al. 1986; Wang et al. 1998; Xu et al. 1998). With time after stress, these proteins return to the cytosol. In Chinese hamster ovary cells, HSP73 follows a similar pattern after heat stress (Ohtsuka and Laszlo 1992). Other reports have shown that HSP72/73 is located on the outer mitochondrial membrane (Lithgow et al. 1993) and on the plasma membrane of some cells after heat shock (Multhoff et al. 1995).
In this study, we have analyzed in detail, using both indirect immunohistochemistry and immunogold electron microscopy, the localization of HSP72 and HSP73 before and after stress, as a means of addressing possible differences in function between the two proteins. We have utilized antibodies with specificity for one isoform only and a third antibody that recognizes both isoforms. To find the conditions that provide the best results in terms of antigen preservation, antibody binding, and sensitivity, we have used both freeze-fixation and chemical fixation before immunogold staining.
Materials and Methods
Cell Culture
A single cell clone of Chinese hamster ovary cells (CHO-K1) was used in all experiments. The cells were grown in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum and antibiotics. For indirect immunohistochemistry, cells were grown on sterile 13-mm coverslips inserted into flasks. Cells in exponential growth phase were used throughout these studies.
Heat Treatments
Cells were heated by submersion of sealed flasks in a water-bath set at 44 ± 0.1C. Fresh medium was always added before heating the cells. Two heat treatment protocols were followed. Cells were heated for 30 min at 44C or were first made thermotolerant by exposure to an initial heat treatment of 10 min at 44C followed by a recovery phase of 16 hr at 37C before a heat shock treatment of 30 min at 44C. After the latter treatment, cells were either processed directly or allowed a further recovery period at 37C before being processed.
Antibodies
Three different antibodies that recognize HSP72/73 were used in these studies. N6, a murine monoclonal antibody (MAb) (a kind gift from Dr. W. Welch), recognizes both HSP73 and HSP72 in CHO-K1 cells. IB5 (SPA-815; Stress-Gen Biotechnologies, Vancouver, BC, Canada) is a rat MAb that recognizes only HSP73 in CHO-K1 cells. C92 (SPA-810; StressGen Biotechnologies) is a murine MAb that recognizes only HSP72.
Western Analysis
Cells from control or heated flasks were harvested by rinsing in PBS, then scraping directly into gel sample buffer. Samples of equal protein loading were run on 10% SDS-PAGE and transferred to nitrocellulose. The membranes were probed for reactivity to N6, C92, or IB5 and detected using a secondary alkaline phosphatase-conjugated antibody.
Indirect Immunofluorescence
The procedure described by Ohtsuka and Laszlo (1992) was used in these studies. Briefly, cells were washed three times with PBS, fixed for 10 min in 3.7% paraformaldehyde in PBS containing 0.2% Triton X-100, washed three times with PBS, all at room temperature (RT), and then extracted with absolute acetone at −20C for 10 min, followed by three washes with PBS at RT. A blocking step of 30 min in 5% Carnation skim milk powder in PBS at RT was included. Cells were then incubated with N6, IB5, or C92 at a 1:60 dilution. Cells labeled with either N6 or C92 were detected with affinity-purified FITC-conjugated goat antimouse antibody (Boehringer Mannheim; Mannheim, Germany) at a 1:100 dilution. IB5-labeled cells were detected using affinity-purified FITC-conjugated goat anti-rat antibody (ICN Pharmaceuticals; Costa Mesa, CA) at a 1:100 dilution. The coverslips were mounted in Citifluor mountant solution (Agar Scientific; Stanstead, UK) and observed with a Bio-Rad (Bio-Rad Laboratories; Hercules, CA) confocal scanning microscope (model MRC1000). Confocal images of cells were obtained with a Leica microscope using a X63 (NA 1.4) plan-apo infinity-corrected objective (Leica Microscopy and Scientific Instruments Group; Heerbrugg, Switzerland). The 488-nm line from a krypton/argon laser was used to excite the FITC-tagged secondary antibody and emission was detected through the 522-nm filter. Optical sections were recorded at 0.4-μm vertical steps with each image averaged for seven scans. All images were processed using the Comos program (Bio-Rad) and Cas 4.02 (confocal assistant software, copyright Todd Clark Brelje). For all antibodies and for all heat regimens, the specificity of the fluorescence was constantly checked by omitting the primary antibody from some samples.
Electron Microscopy
Cells were gently scraped off flasks using a cell scraper (Costar; Cambridge, MA) before fixation. Two different methods of fixation, aldehyde and freeze fixation, were used. Cells in Group A were fixed in 2% paraformaldehyde, 0.05% glutaraldehyde in PBS, pH 7.4. Cells were then cryoprotected by immersion for 15 min in 1.2 M sucrose in PBS, pH 7.4, followed by 30 min in 2.3 M sucrose in PBS, pH 7.4, before being plunged into liquid nitrogen. Cells in Group B were centrifuged in 1% ultra-low geling temperature agarose (Sigma; St Louis, MO) and 2.3 M sucrose in PBS, pH 7.4. Approximately 5 μl of cells was placed inside a polypropylene ring and slammed onto a polished nitrogen-cooled copper mirror using the MM80 head attached to the KF80 (Leica). Settings were thickness = 2, speed = 8, force = 8. Cells from both Group A and Group B were transferred to a Reichert AFS (Leica) and substituted at −91C in methanol for 4-5 days. Substituted samples were warmed to −50C and embedded in Lowicryl HM20, which was polymerized at −50C by UV polymerization. Sections 90 nm thick were cut on a Reichert-Jung Ultracut E (Leica) and mounted on formvar-coated nickel grids. A wide range of fixation and embedding protocols were tested and the procedures presented here gave the best retention of antigenicity of HSP72 and HSP73.
Immunocytochemistry
Samples from Group A were placed on drops of 0.05 M glycine in PBS, pH 7.6, to quench free aldehydes. Groups A and B were then processed identically, as follows. Sections were floated on drops of blocking buffer [5% bovine serum albumin, 5% normal goat serum in incubation buffer (IB): 0.8% bovine serum albumin, 0.1% coldwater fish skin gelatin in PBS, pH 7.4] for 60 min. They were transferred to 8-μl droplets of either N6 at a dilution of 1:7, IB5 at a dilution of 1:20, or C92 at a dilution of 1:5, and incubated overnight in a moist chamber at 4C. After six 5-min washes in IB, N6, and C92 were detected by incubation for 60 min with goat anti-mouse IgG+M conjugated to 10-nm colloidal gold (Biocell Research Laboratories; Cardiff, UK). IB5 was detected by incubation for 60 min with goat anti-rat IgG+ M conjugated to 10 nm colloidal gold (ICN) or with a goat anti-rat IgG and IgM conjugated to ultrasmall gold (Aurion; Wageringen, The Netherlands). Ultrasmall gold was silver-enhanced according to Danscher (1981). A further six 5-min washes were followed by two 5-min washes in PBS and the antibody was “fixed” in place with 2% glutaraldehyde. Grids underwent a further six 5-min washes in ultrapure (18 Mil) water before drying. Some grids were double stained with methanolic uranyl acetate and lead citrate to improve contrast. Control sections were incubated in the absence of primary antibody. A positive control of a section known to react well with the antibodies used was also included. Examination was in a Hitachi H-600 electron microscope.
Results
Antibody Specificity
The intracellular localization of the two major isoforms of HSP70, i.e., HSP72 and HSP73, was investigated in CHO-K1 cells before and after a heat shock at 44C. Antibodies specific to these two isoforms or an antibody that recognizes both isoforms were used, and localization was determined both by indirect immunofluorescence and immunogold electron microscopy. The specificity of the antibodies is shown by Western analysis of the cells before and after heat shock (Figure 1). Only HSP73 is present in lysates of control cells, whereas a mild heat treatment of 10 min at 44C followed by 16 hr at 37C induces expression of HSP72. Thus, in thermotolerant cells, N6 detects both proteins and C92 now detects HSP72. IB5 staining, which detects only the constitutively expressed HSP73, remains unchanged between control and heated samples. No other proteins were reactive with these antibodies (Figure 1).
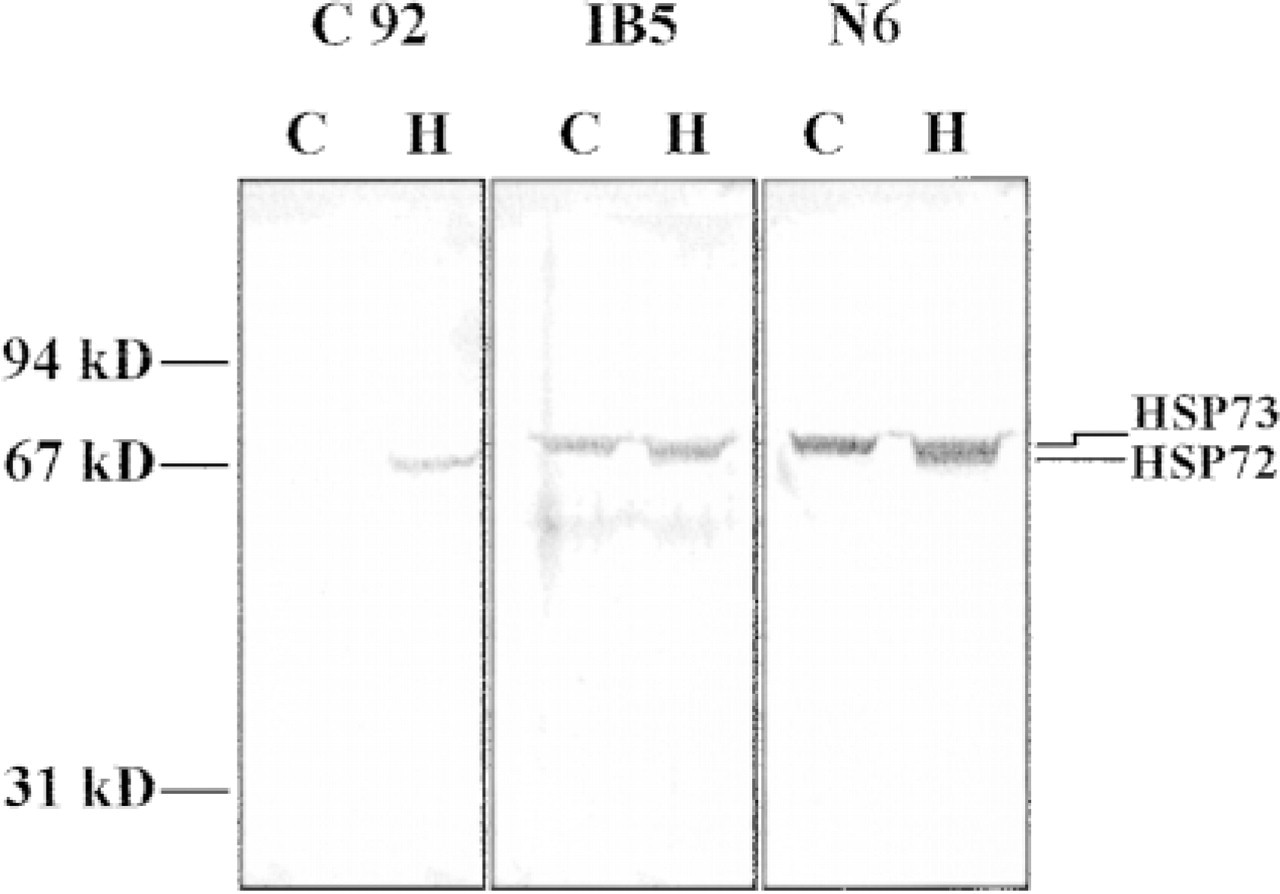
Specificity of the anti-HSP70 antibodies. CHO cells were heated for 10 min at 44C, then returned to 37C for 8 hr to allow expression of HSP72. Samples of control or heated CHO cells were subjected to SDS-PAGE and transferred to nitrocellulose. The membranes were probed with either C92, IB5, or N6 at 1:1000 dilution (1 μg/ml).
Localization of HSP72 and HSP73 by Indirect Immunofluorescence
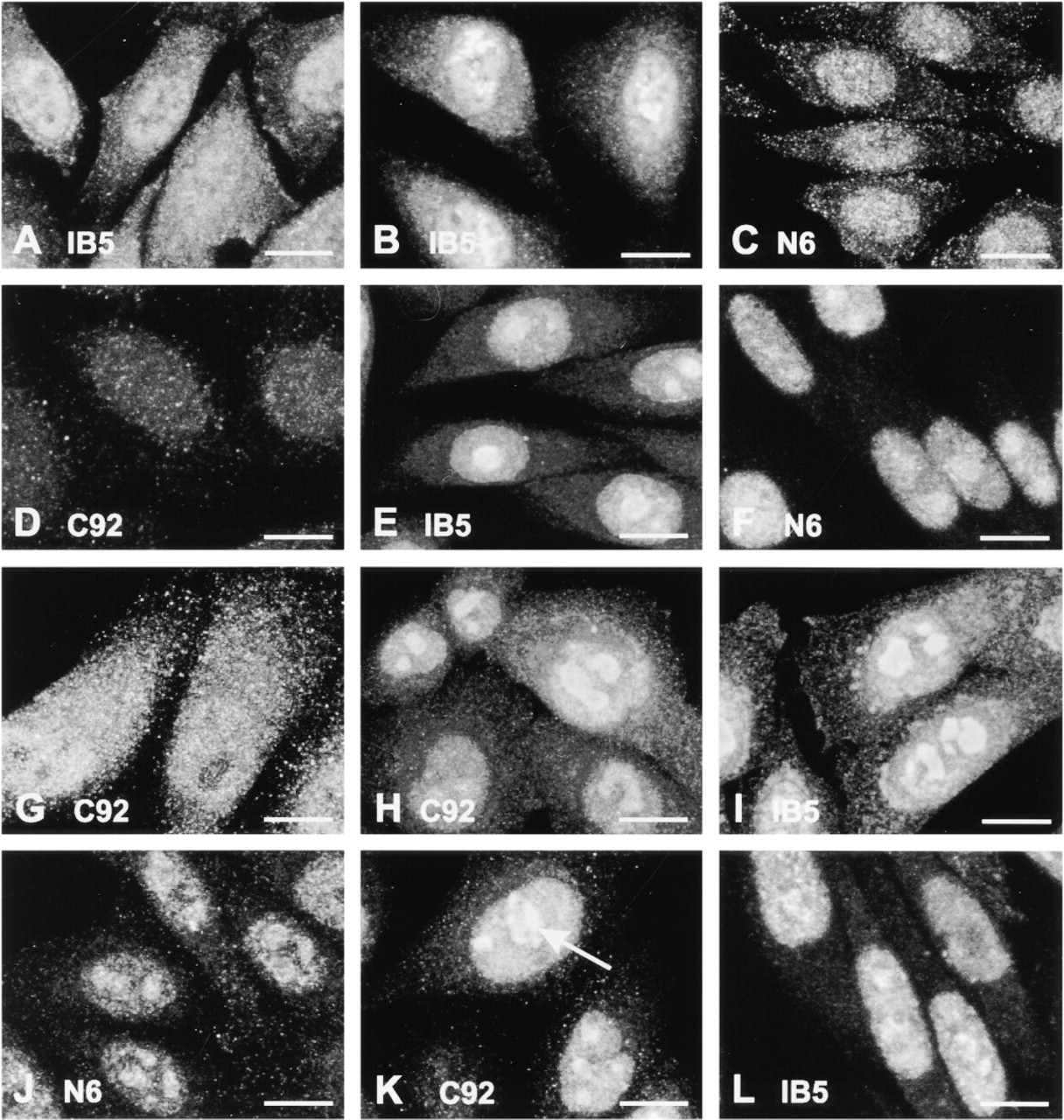
Distribution of HSP72 and HSP73 in CHO cells as revealed by indirect immunofluorescence. (
In an attempt to simplify the results, we have summarized them in Table 1, giving an indication of fluorescence intensity in the cytoplasm, nuclei, and nucleoli for each of the three antibodies after each heat regimen. Some caution should be exercised in reading this table because it does not reveal differences in appearance of fluorescence patterns but merely indicates fluorescence intensity.
Summary of levels of expression of HSP72 and HSP73 in different compartments of the cell after different heat regimens, as measured by indirect immunofluorescence
Localization of HSP72 and HSP73 by Immunogold Staining and Electron Microscopy
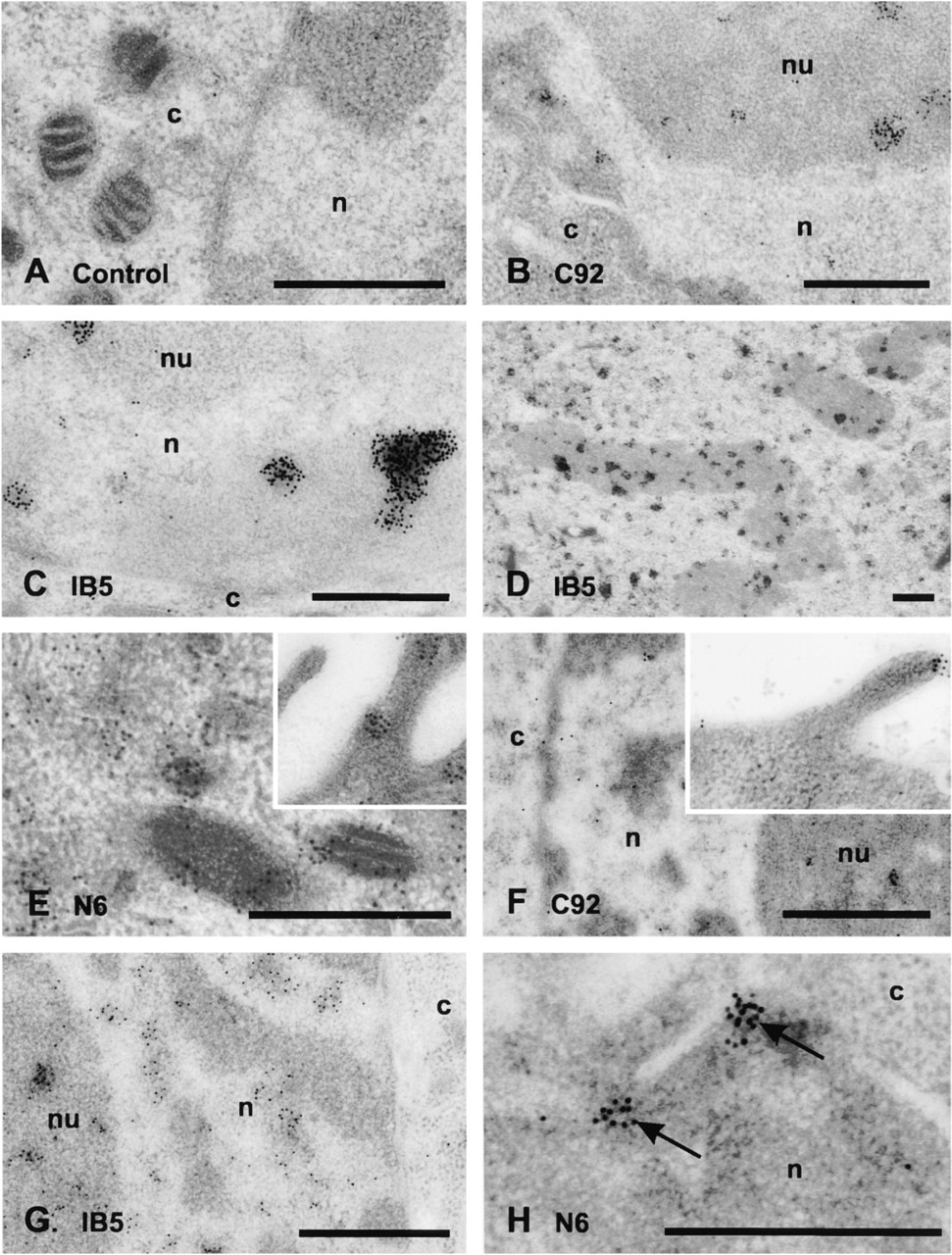
The method of fixation of cells before immunolocalization of proteins has variable effects on proteins and can destroy the antigenic determinants. Therefore, we initially compared two methods—chemical fixation and freeze fixation—for the ability of the three anti-HSP70 antibodies to detect their epitopes. We found that the staining intensity was greater in freeze-fixed cells for the antibodies IB5 and N6, suggesting better preservation of their antigens in their native state. No difference was observed in the labeling intensity or specificity when the antibody C92 was used. Another notable difference in staining patterns was seen when IB5 was used to detect HSP73. Substantial mitochondrial labeling was seen after freeze-fixing (Figure 3A), whereas little was detected in chemically fixed cells (Figure 3B). All further analyses with IB5 and N6 were completed using the freeze-fixing technique, whereas analysis with C92 was drawn from both fixation procedures. A minimum of five separate experiments utilizing freeze-fixation was completed for each control and heat shock regimen. Multiple blocks were cut for each experiment and labeling with the three different antibodies was completed on sections cut from these blocks. There were eight to 13 repeat labeling runs for each antibody and heat treatment.
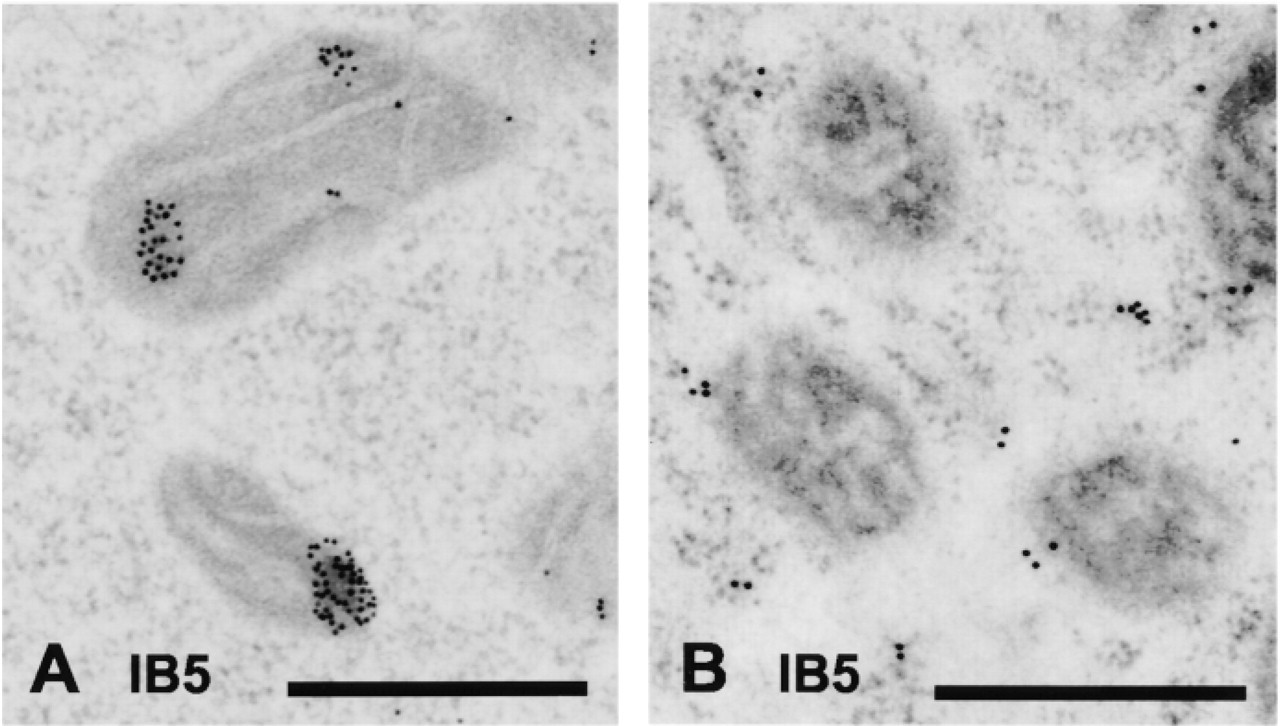
Transmission electron micrographs of mitochondrial labeling by IB5 in freeze-fixed (
HSP73, as detected by IB5, was distributed primarily in the nucleus, including the nucleoli (Figure 4G). Clusters of gold occasionally decorated the heterochromatin, and mitochondria were positive for HSP73. The localization of total HSP72/73 as detected with N6 was again different from that seen with IB5 or C92. The cytoplasm generally remained the most heavily stained compartment after heat exposure, although clusters of gold were often observed either surrounding the nucleolar periphery or lying over the nucleolus itself. HSP72/73 could be seen crossing the nuclear membrane (Figure 4H).
A tabular summary of the immunogold electron microscopic data, as was presented above for the fluorescence data, is not given because it is difficult to quantitate the intensity of gold particles in the thin sections used for antibody binding.
Discussion
As shown previously by others (Welch and Feramisco 1984; Ohtsuka et al. 1986; Wang et al. 1998; Xu et al. 1998), HSP72 and HSP73 are both cytoplasmic and nuclear before heat shock and move from the cytoplasm to the nucleus and nucleoli during heat shock. In this study, we set out to determine whether there are any differences in the localization of HSP72 and HSP73 before and after heat stress that could indicate different functions of the two isoforms. To compare the distribution of these two proteins, it was necessary to induce expression of HSP72 by exposing the cells to a mild heat treatment. The localization of the two proteins was then compared before and after a second more severe heat treatment. We did observe some differences in distribution of HSP72 and HSP73 before heat exposure, but these differences were less pronounced in stressed cells. The differences in localization were more evident with the more sensitive technique of immunogold staining after freeze-fixation of the cells rather than with indirect immunofluorescence.
A Comparison of Chemical vs Freeze-Fixation Before Immunogold Electron Microscopy
Freeze-fixation improved the intensity of immunological staining of N6 and IB5. Using IB5, we found that labeling of mitochondria was much more evident after freeze-fixation compared to chemical fixation, although labeling of other cell organelles by IB5 was not affected. These differences may result from the masking of the HSP73 epitope after chemical fixation due to crosslinking of the surrounding proteins. Sensitivity of HSP to chemical fixation by small amounts of glutaraldehyde has been reported previously (Soltys and Gupta 1996). Given the poor binding of N6 and, to a lesser extent, IB5 after aldehyde fixation, caution should be exercised in interpreting localization patterns from aldehyde-fixed cells.
Differences in HSP72/73 Localization Using Two Different Detection Methods
Confocal immunofluorescence and immunogold electron microscopy are two complementary techniques for determination of protein localization. The fluorescence approach allows the reconstruction of a 3D image of the cell and the overall localization of HSP72/ 73. For a more detailed examination for the presence of HSP72/73 in individual organelles, electron microscopy is used. This latter technique, when applied after freeze-fixation, has the added advantage of avoiding antigen damage and/or masking during chemical crosslinking and leaching of proteins from the cell by detergent or acetone. We believe that the immunogold labeling is more sensitive because it is done on thin sections that provide easy access of the antibody and because chemical crosslinking, detergent, and acetone are avoided.
Transmission electron micrographs of CHO cells before and after different heat regimens. (
In general, the fluorescence patterns of HSP72/73 distribution did reflect those seen at the EM level, but some differences were observed. First, HSP72 was not detected by fluorescence in the cells before induction of thermotolerance, whereas it does appear to be present in control cells as assayed by the immunogold technique. It is possible that, during the fixation and permeabilization process used for fluorescence, proteins may leach from the cells (Ohtsuka and Laszlo 1992). Alternatively, the levels of HSP72 may be too low to be detected above the background fluorescence.
We also noted a marked difference in HSP72/73 distribution between the fluorescence and EM experiments when the antibody N6 was used. After heat shock, most fluorescence staining was associated with the nucleoli, whereas the EM immunogold staining revealed the cytoplasm to be the most heavily labeled compartment. These differences could be due to a loss or masking of cytoplasmic HSP72/73 during the fixation and Triton-X/acetone permeabilization, as suggested previously (Ohtsuka and Laszlo 1992). It is evident that N6 binding is sensitive to aldehyde fixation. The association of HSPs with other cellular proteins has previously been implicated in preventing recognition of the antigenic epitope by the antibody (Milarski et al. 1989). On the other hand, care must be exercised in comparing results obtained from the thin sections (90-nm) used for EM to those obtained by immunofluorescence, which reveals whole-cell localization.
Localization of HSP72 and HSP73 in Control and Heated Cells
In nonheated cells, most HSP72 was located in the nucleus, especially in nucleoli and in regions of heterochromatin. In contrast, HSP73 revealed by IB5 was distributed throughout the cell, with much of the label found in the nucleolus or aggregated over the mitochondria. Heterochromatin and mitotic chromosomes were also positive for HSP73. A striking difference in localization was observed when N6 was used. This antibody recognizes both isoforms, but in resting cells the predominant isoform is HSP73. N6 detected HSP73 predominantly in the cytoplasm, with less label associated with the nucleus and generally no label associated with nucleoli. Mitotic chromosomes were not labeled by N6. The large immunogold clusters of IB5 contrasted with the diffuse gold labeling pattern of N6, which was mostly in the form of single grains.
In contrast to resting cells, differences in localization between HSP72 and HSP73 were not evident after heat stress. Immediately after heating, HSP72/73 tended to localize more strongly in the nucleus, especially in nucleoli. By 1 hr after heating, HSP72/73 had moved from the nucleoli to regions of heterochromatin, and the mitochondrial signal was reduced. Clusters of gold particles indicating HSP72/73 passing through nuclear pores into the cytoplasm could be observed.
The antibodies used here do not recognize the HSP70 isoform in the mitochondrial matrix, but there are reports of an HSP70 isoform associated with the outer mitochondrial membrane (Carbajal et al. 1993; Lithgow et al. 1993). From our results, it appears that HSP73 is the predominant isoform associated with mitochondria, but we did detect small amounts of mitochondrial HSP72 in control cells.
Labeling of HSP72/73 in the cytoplasm was both in clusters and in single grains, suggesting association both with individual proteins and with protein complexes. Nascent proteins are kept translocation-competent by association with cytosolic HSP73 (Rassow and Pfanner 1996). Soltys and Gupta (1996) found labeling of HSP60 in the cytoplasm over discrete sites of non-membrane-bound amorphous material similar to the clusters we observed. They suggest that these sites represent aggregates of protein.
Labeling of nucleoli is believed to occur in granular regions rather than in fibrillar regions (Pelham 1984; Welch and Feramisco 1984; Welch and Suhan 1986). It was not possible to discern the fibrillar and granular regions of the nucleoli after heat shock by their morphology alone. IB5 labeled nucleoli of nonheated cells in what appeared to be the granular region.
When the antibody C92 was used to detect HSP72 by immunofluorescence, we noted some variability of staining among cells, as has been reported previously. Henriksson et al. (1992) reported variable staining of HSP72 in myc-transfected cells. Milarski et al. (1989) reported heterogeneity in fluorescence staining of HeLa cells both before and after heat shock. They were able to show that the variability in fluorescence patterns and intensity of fluorescence after labeling with C92 was a cell-cycle phenomenon; once cells were synchronized, uniformity in fluorescence was observed. However, depending on which antibody was used to detect HSP72 after heat shock, different results for the nuclear distribution of HSP72 were observed. The differences were proposed to be due to the association of HSP72 with various proteins (including HSP73) throughout the cell cycle, resulting in masking of the antibody epitope. C92 was least able to recognize its epitope throughout the cell cycle, whereas N15, another antibody that recognizes human HSP72, always recognized its epitope regardless of the cell-cycle phase (Milarski et al. 1989).
We have noted that the distribution of HSP73 depends on the antibody being used. N6 and IB5, which by definition must recognize different epitopes, suggest different distributions of HSP73 in nonstressed cells. Most marked was the inability to detect significant amounts of HSP72/73 in the nucleoli and mitochondria when N6 was used compared to IB5. We hypothesize that the epitope for N6 is masked by binding to components of these organelles, whereas the epitope for IB5 remains free to bind the antibody. This highlights the importance of using at least two antibodies directed against different epitopes in determining localization of a protein.
In conclusion, we have shown that, in resting cells, the distribution of HSP72 and HSP73, although overlapping, reveals distinct variations that may reflect different chaperone functions. After heat stress these differences in localization are minimal, suggesting that both proteins are associating with similar damaged complexes or molecules. These results suggest that, in nonstressed cells, HSP72 and HSP73 may associate with different although overlapping proteins or complexes but that, during stress, both proteins are mobilized to protect essential sites, especially in the nucleoli and mitochondria.
Footnotes
Acknowledgments
We wish to thank Dr W. Welch for his gift of the N6 antibody and Manuela Palatsides for valuable assistance with experiments.
