Abstract
The aim of this study was to ascertain how perlecan was localized in human fetal cartilaginous joint rudiment tissues. Perlecan was immunolocalized in human fetal (12–14-week-old) toe, finger, knee, elbow, shoulder, and hip joint rudiments using a monoclonal antibody to domain-1 of perlecan (MAb A76). Perlecan had a widespread distribution in the cartilaginous joint rudiments and growth plates and was also prominent in a network of convoluted hairpin loop-type vessels at the presumptive articulating surfaces of joints. Perlecan was also present in small perichondrial venules and arterioles along the shaft of the developing long bones, small blood vessels in the synovial lining and joint capsules, and in distinctive arrangements of cartilage canals in the knee, elbow, shoulder, and hip joint rudiments. Perlecan was notably absent from CD-31-positive metaphyseal vessels in the hip, knee, shoulder, and fingers. These vessels may have a role in the nutrition of the expanding cell populations in these developing joint tissues and in the establishment of the secondary centers of ossification in the long bones, which is essential for endochondral ossification.
Keywords
P
The carboxyl terminal peptide domain of perlecan, endorepellin, displays anti-angiogenic properties (Mongiat et al. 2003b). Perlecan also interacts with type XVIII collagen, and the anti-angiogenic peptide derived from the carboxyl domain of type XVIII collagen, endostatin, which co-localizes with perlecan in a number of human tissues (Miosge et al. 2003). Furthermore, perlecan also interacts with progranulin and promotes angiogenesis during tumor development (Gonzalez et al. 2003). Perlecan can therefore display both pro- and anti-angiogenic properties, depending on its developmental environment, and undoubtably has important regulatory roles in vascular remodeling (Nakahama et al. 2000; Gonzalez et al. 2003; Jiang and Couchman 2003).
Perlecan has previously been immunolocalised in a range of mouse tissues (Handler et al. 1997; Arikawa-Hirasawa et al. 1999; French et al. 1999), including cartilage, bovine nasal cartilage, and chondrosarcoma (Iozzo et al. 1994; Murdoch et al. 1994; SundarRaj et al. 1995; Costell et al. 1999), newborn ovine vertebral growth plate and cartilaginous end plates of the intervertebral disc (Melrose et al. 2002), and in the cartilaginous rudiments of the developing human fetal spine (Melrose et al. 2003). In the present study perlecan was immunolocalized in a number of fetal (12-14-week) human cartilaginous rudiments (fingers, toes, ribs, knee, elbows, shoulder, hips), where it was identified as a component of the joint rudiments. Perlecan was also a prominent component of small vessel networks at the presumptive articulating surface of the developing joint, small blood vessels in the perichon-drium, synovial lining, joint capsules, and cartilage canals deep within the joint rudiment. In contrast, perlecan was notably absent in the metaphyseal vessels of fetal joint rudiments.
Materials and Methods
Reagents and Antibodies
Histochoice was an Amresco product (Solon, OH). The monoclonal antibody (MAb) to perlecan domain I (A76) was prepared, characterized (Knox et al. 2001,2002), and used as described previously (Melrose et al. 2002,2003). Anti-type IV collagen, anti-CD31 (PECAM, platelet endothelial cell adhesion molecule, clone JC/70A) and negative control mouse monoclonal IgG1 (clone DAK-GO1) against Aspergillus niger glucose oxidase were purchased from DAKO (Glostrup, Denmark). Anti-type I collagen (clone 18H5) was obtained from ICN (N. Ryde, Australia).
Preparation of Human Fetal Tissues for Histological Procedures
Six 12-14-week-old human fetuses were obtained at termination of pregnancy after ethical approval by the Human Research Ethics Committee of the Royal North Shore Hospital. The tissue specimens were fixed in Histochoice for 24 hours and briefly decalcified (1-3 days) in 5% formic acid, dehydrated in graded alcohols, and embedded in Paraplast using standard histology procedures. Four-μm sections were cut and mounted on SuperFrost Plus glass slides (Menzel-Glaser; Mannheim, Germany), deparaffinised in xylene (two changes for 2 min), and rehydrated through graded ethanol washes (100-70% v/v) to water.
Histochemistry
Histochoice-fixed tissue sections were routinely stained for 10 min with 0.04% w/v toluidine blue in 0.1 M sodium acetate buffer, pH 4.0, to visualize the tissue PGs. This was followed by a 2-min counterstaining in an aqueous 0.1% w/v fast green FCF stain to differentiate areas stained for PG. Hematoxylin and eosin-stained tissue sections were used to examine general cell morphology and aid in the selection of appropriate sections for detailed immunolocalizations of perlecan and other basal lamina components.
Immunohistochemistry
Incubations with primary antibodies were performed using a Sequenza vertical coverplate immunostaining system (Melrose et al. 2002,2003). Endogenous peroxidase activity was blocked by incubating the tissue sections with 3% H2O2 for 5 min and, after washing in water, nonspecific binding sites were blocked with 10% swine serum for 10 min. In the perlecan immunolocalizations the tissue sections were predigested with bovine testicular hyaluronidase (500 U/ml) for 1 hr at 37C in phosphate buffer, pH 5.0, followed by three washes in 50 mM Tris-HCl buffer, pH 7.6, containing 0.15 M NaCl and 0.05% Tween-20 (TBS-T). The anti-type I, IV, and anti-CD-31 immunolocalizations did not require any predigestion steps. The tissue sections were incubated overnight at 4C with anti-perlecan domain 1 antibody (1.5 μg/ml), anti-type I (1:200), anti-type IV (1:200), and CD-31 primary antibodies (1:50) dilution. After washing, the slides were incubated with appropriate biotinylated secondary antibodies and horseradish peroxidase-conjugated streptavidin for visualization using Nova Red substrate for color development. Control sections were prepared by substituting the authentic primary antibodies with an irrelevant mouse IgG directed against A. niger glucose oxidase, an enzyme that is neither present nor inducible in mammalian tissues.
Results
Perlecan was strongly localized to the pericellular matrix of chondrocytes in the cartilaginous joint rudiment and to the hypertrophic growth plate chondrocytes in joints of the fingers and toes (Figures 1A and 1C). However, its levels were diminished at the margins of the cartilaginous rudiment. Perlecan was also a prominent component of a network of small blood vessels located at the presumptive articulating surface of these joints and to the basal lamina of small blood vessels in the synovial folds of the toe and finger joints and the adjacent perichondrial tissues (Figures 1B and 1D). Type IV collagen was also immunolocalized to the small vessels of the synovial folds and perichon-drium but not to the vessels in the presumptive articulating surfaces of these joints (Figures 1E-1G). However, these did stain positively for CD-31 (data not shown), as did the various arrangements of small vessels throughout the fetal finger (Figure 1H). Type I collagen was ubiquitously expressed throughout the developing finger, including the calcifying cartilage of the metaphysis (Figure 1I, asterisk), but was conspicuously absent from the cartilaginous rudiment itself.
Perlecan also displayed a pericellular immunolocalization pattern in the chondrocytes of the metachromatic PG- and collagen-rich cartilaginous rudiments of the developing ribs (Figures 2A and 2B) but was also a prominent component of a network of convoluted vessels at the margins of the cartilaginous rudiment (Figures 2C and 2D).
Perlecan was also a prominent pericellular component around the chondrocytes within the cartilaginous joint rudiments of the knee and was also present in an intricate network of vessel- like structures at the developing margins of the presumptive articulating surfaces of the femoral condyles of 12-week-old human fetal tissues (Figures 3A and 3B). These surface vessels also stained positively for CD-31 and type IV collagen (Figures 3C and 3D). Red blood cells were discernible in some of these vessels (Figure 3C). However, the distorted shapes of the majority of the surface vessels were more reminiscent of lymphatic vessel networks than of blood vessels.
Perlecan was also a prominent component of the 14-week metachromatic cartilaginous rudiments of the humerus (Figures 4A–4D and 5K), displaying a pericellular distribution in the elbow rudiment proper, and the synovial lining blood vessel and surface vessel networks (Figures 4B–4D). This was reminiscent of the knee rudiments at week 12 (Figures 3A and 3B). Small perlecan-positive vessels were also observed in the perichondrium, extending along the axis of the humeral cartilaginous rudiment (Figure 4B).
The surface vessel networks of the 14-week human fetal knee were somewhat better defined compared with their counterparts in the 12-week specimens (Figures 5A, 5B, 5E, and 5F). Immunolocalization of type IV collagen (Figures 5A and 5F), CD-31 (Figures 5B and 5E) and perlecan (Figure 5C) again confirmed the presence of basal lamina components in these surface vessels. Red blood cells were also observed in some of these vessels using Nomarski interference contrast microscopy (Figure 5E).
Immunolocalization of perlecan in the 14-week cartilaginous knee joint rudiments also delineated a number of larger vessels or canals deep within the rudiment proper, which were lined with perlecan (Figures 5H and 5I). These were also CD-31 (Figure 5G) and type I (Figure 5D) and type IV collagen-positive (data not shown). These cartilage canals had distinctive arrangements of vessels consisting of a single central smaller vessel, possibly a venule/arteriole, surrounded by four to six larger vessels. Collectively, such vessel arrangements were >100 μm in circumference and were located deep within the femoral or humeral rudiment (Figures 5H-5O). Some variation in vessel arrangements was evident in the cartilage rudiments, with the outer canals of such canal arrangements of the shoulder being somewhat larger and more misshapen (Figures 5J and 5K) than those observed in the knee rudiments, which generally had a more regular circular geometry (Figures 5H and 5I). The morphology of the cartilage canals of the hip rudiment also differed markedly from those of the knee and shoulder rudiments (Figures 5L-5O). All of the cartilaginous hip rudiment (Figure 5L) cartilage canals were lined with perlecan (Figure 5M), type IV collagen (data not shown), and were CD-31- (Figure 5N) and type I collagen- (Figure 5O) positive, which contrasted markedly with the rudiment proper, which contained only type II collagen (data not shown). Furthermore, such cartilage canals were not seen in 12-14-week-old human fetal finger and toe rudiments.
Although perlecan was present throughout the cartilaginous hip rudiment (Figures 6A and 6B) and its expression was elevated pericellularly around the hypertrophic growth plate chondrocytes, its distribution did not extend markedly past these cells into the calcifying cartilage and the metaphysis, despite the fact that perlecan was still strongly associated with small vessels in the perichondrium and joint capsule tissues of the femur (Figure 6C). The hip metaphyseal vessels were clearly delineated using the CD-31 (Figure 6D) and type IV collagen (data not shown) MAbs. However, these vessels did not stain for perlecan, whereas some residual deposits of perlecan were still detectable within the calcified cartilage (Figure 6C). Localization of type I collagen also clearly delineated the calcified structures (sinusoids) surrounding the metaphyseal vessels in the hip (Figure 6E). Type I collagen was also strongly expressed pericellularly by the most terminally differentiated hypertrophic growth plate chondrocytes in the hip rudiment (Figure 6E) but was totally absent in the cartilaginous hip rudiment proper, which contained type II collagen (data not shown). This permitted the frank demarcation of the metaphysis from the cartilaginous rudiment. Similar localization patterns were also observed for types I and IV collagen and CD-31 in the fetal fingers and toes (Figures 1A-1I). Perlecan also did not localize with the metaphyseal vessels in the fingers and toes.
Discussion
Endochondral bone formation involves a complex sequence of events that is initiated by condensation of mesenchymal cell aggregates to form the limb rudiment cartilage, which serves as a developmental scaffold for subsequent long bone development. An early stage in this process is the vascularization of the diaphysis perichondrium, transformation of peripheral chondrogenic cells into an osteogenic phenotype, and formation of a subperiosteal bone collar (Haines 1933; Levene 1964; Wilsman and Van Sickle 1972; Cole and Wezeman 1985; Floyd et al. 1987; Skawina et al. 1994; Hirano et al. 1994; Roach et al. 1998). Penetration of perichondrial blood vessels into the diaphysis then results in the establishment of secondary centers of ossification at each pole of the limb rudiment, and the subperiosteal collar progressively thickens and lengthens concomitant with the resorption of the cartilage in the long bone rudiment, leading to axial extension of the long bone (Haines 1933; Levene 1964; Wilsman and Van Sickle 1972; Kugler et al. 1979; Cole and Wezeman 1985; Floyd et al 1987; Skawina et al. 1994; Hirano et al. 1994; Roach et al. 1998). The epiphyseal plates and the ends of the epiphyses, which will be replaced by articular cartilage, are spared these changes. Ossification of the epiphysis occurs from its center, the secondary center of ossification. Blood vessels enter the epiphysis without vascu-larizing the cartilage to form a network around which spongy bone forms. Vascularization of the joint rudiment is a critical step in the elongation of long bones and in joint development. Perlecan has key roles to play in these early vascularization events, underscoring its importance in skeletal development (Hassell et al. 2002; Gustafsson et al. 2003). However, this study has demonstrated that perlecan is not a component of the metaphyseal vessels.
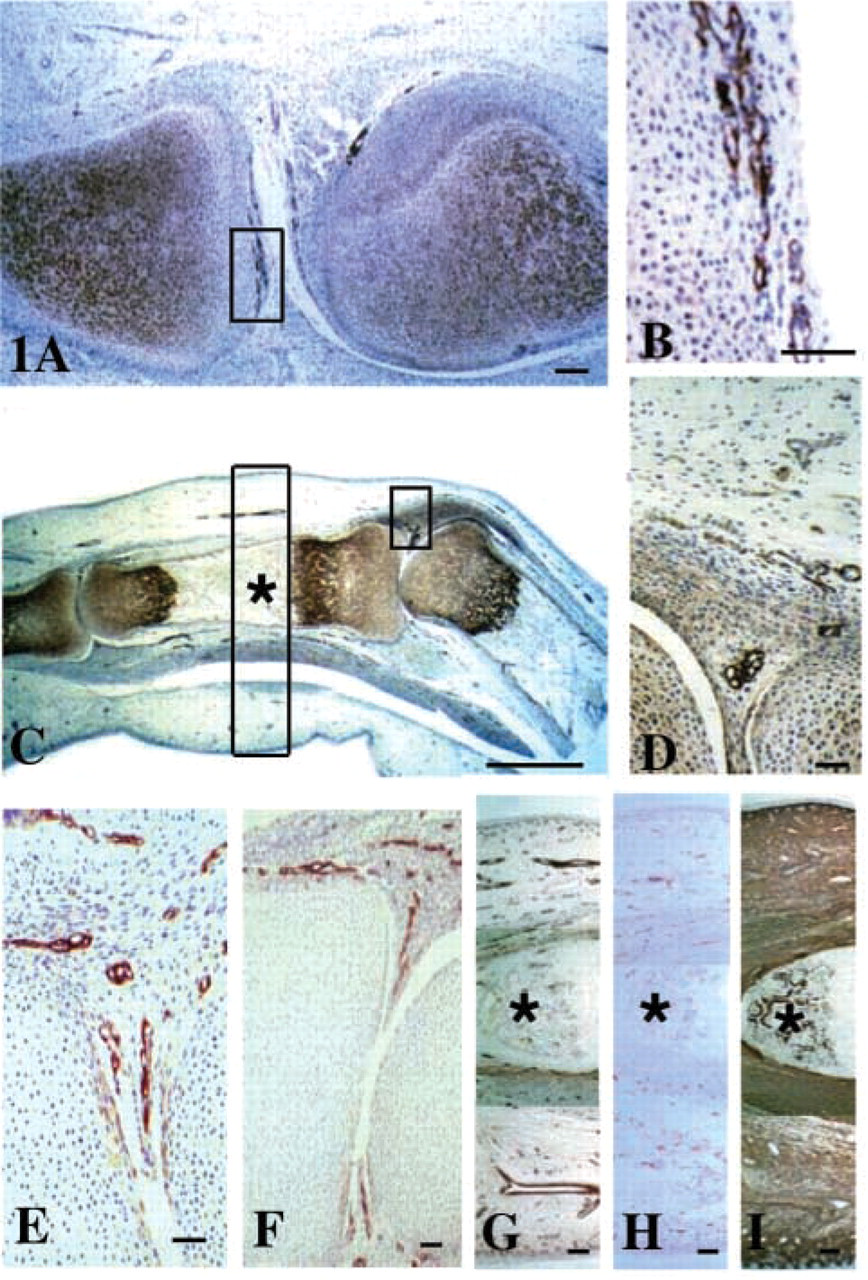
Immunolocalisation of perlecan (
Intrachondral vascularization events have previously been investigated in the talus of the human foot (Cheng et al. 1997), human femoral head (Trueta 1957; Skawina et al. 1994), fetal elbow (Reidenbach and Schmidt 1994), femoral head and tibiae of growing rats (Kai et al. 1992; Hirano et al. 1994; Morini et al 1999; Davoli et al. 2001; Lee et al. 2001) and mice (Cole and Wezeman 1985; Floyd et al. 1987) newborn and postnatal rabbit knee (Ganey et al. 1992; Shapiro 1998; Doschak et al. 2003), and in normal and arthritic human synovial tissue (Haywood and Walsh 2001).
Scanning electron microscopy has been used to examine blood vessel resin casts of human fetal knee joints to describe the sequence of angiogenic events leading up to vascularization of human fetal femoral cartilage and to the development of the femoral secondary center of ossification (Skawina et al. 1994). The process of cartilage vascularization is initiated by the penetration of hairpin loops from the perichondrial vascular supply into the adjacent cartilage. These vessels are similar to the perichondrial capillary networks stained with anti-perlecan antibodies in the present investigation. A capillary glomerulus is then formed at the leading end of the penetrating vessels and the entire vascular unit eventually assumes a mushroom-like shape (Skawina et al. 1994). Further elongation of this capillary network is accompanied by its backward expansion, resulting in the network surrounding a pair of main vessels (arteriole and venule) like a cuff around a sleeve, a feature that Skawina et al. (1994) termed a manchette. This may be similar to the distinctive vessel arrangements observed deep in the femoral and humeral rudiments in the present study. Skawina et al. (1994) noted that such vessels merged to form a precursor to the cartilage canals involved in the establishment of the secondary center of ossification. Such canals have also been examined in the rabbit, sheep, rat, and mouse (Haines 1933; Levene 1964; Kugler et al. 1979; Cole and Wezeman 1985; Floyd et al. 1987; Ganey et al. 1992,1995), and their morphologies and ultrastructures have been described (Stockwell 1971; Wilsman and Van Sickle 1972; Skawina et al. 1994; Ganey et al. 1995). This study has demonstrated a number of different geometries and vessel arrangements for the cartilage canals in the joint rudiments examined.
Vascular supplies similar to the surface vessel networks observed in the present investigation have also been identified in rabbit joint tissues, where long loops of arterioles supply blood to areas of synovium subjected to high levels of mechanical stress in the rabbit knee joint (Lindstrom and Branemark 1962). However, these vessels are susceptible to resorption as a consequence of alterations in joint biomechanics. The surface vessel networks observed in the present study were of a similar size (50-70 μm) and appearance to the convoluted vessels of lymphatic endothelium, particularly in the 12-week fetal tissues (Jackson 2003; Xu et al. 2003). However, they were better defined in the 14-week fetal rudiment samples and more typical of small blood vessels. Occasional groups of entrapped red blood cells were also observed in some of these vessels. Such surface vessel networks may therefore represent a mixture of blood and lymphatic vessels. Future work from our laboratory will aim to explore this possibility using some new lymphatic endothelium markers that have recently become commercially available.
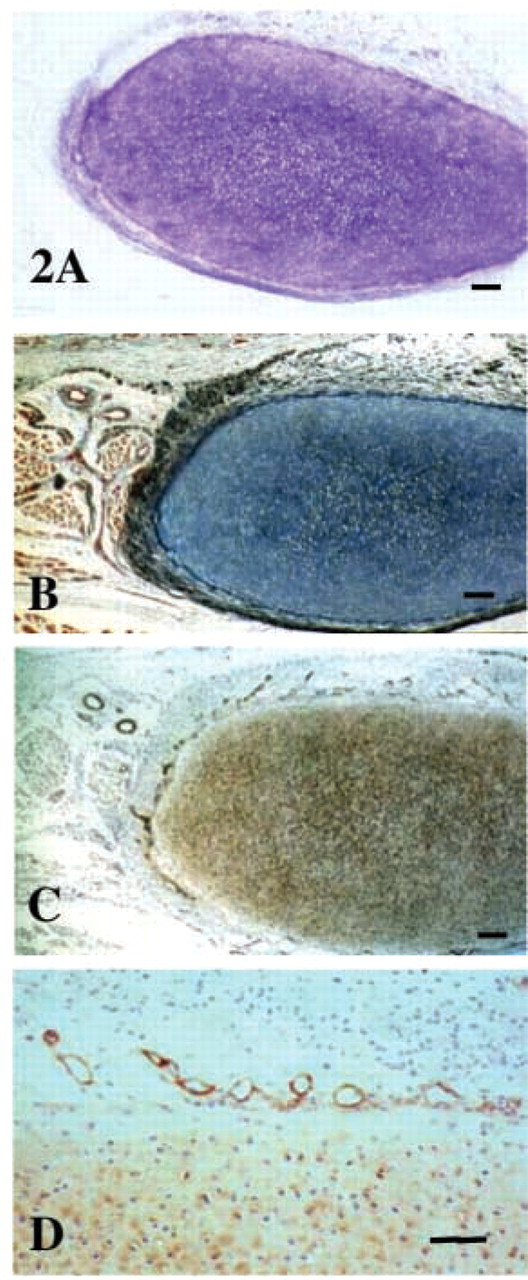
Histochemical localization of anionic proteoglycan (toluidine blue/fast green,
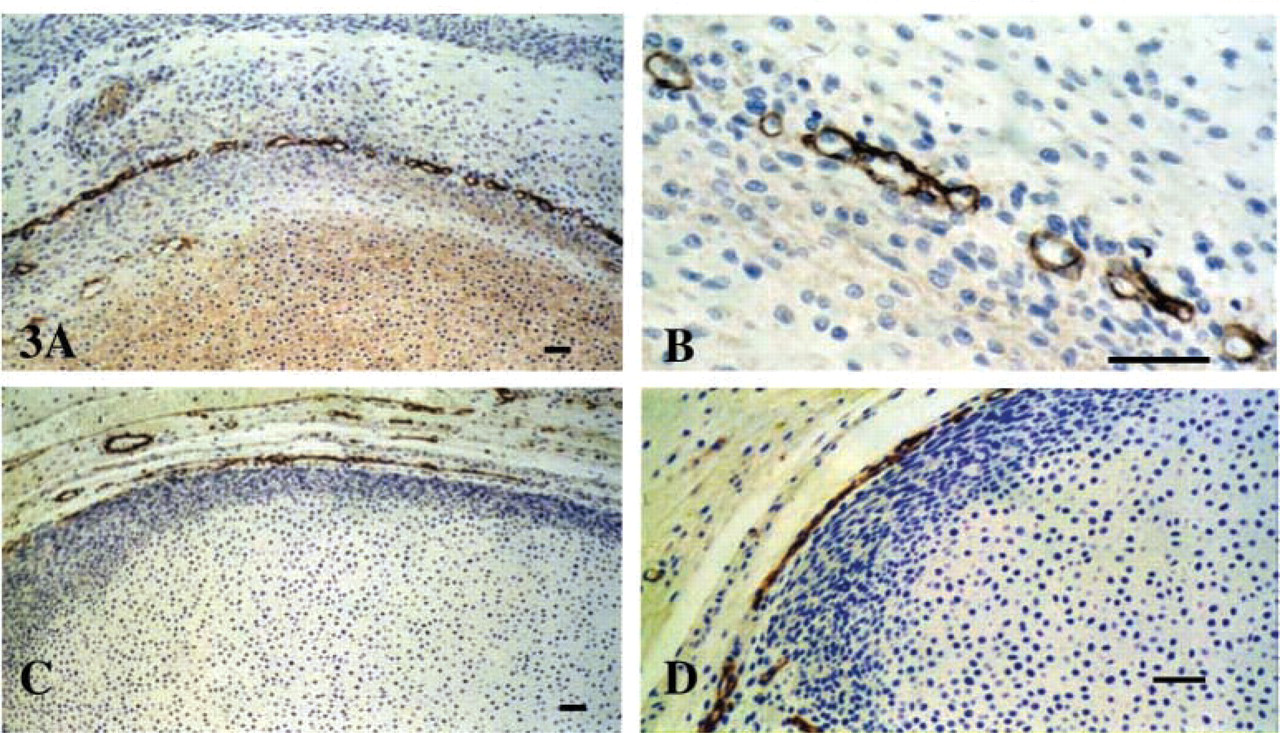
Immunolocalisation of perlecan (MAb A76,
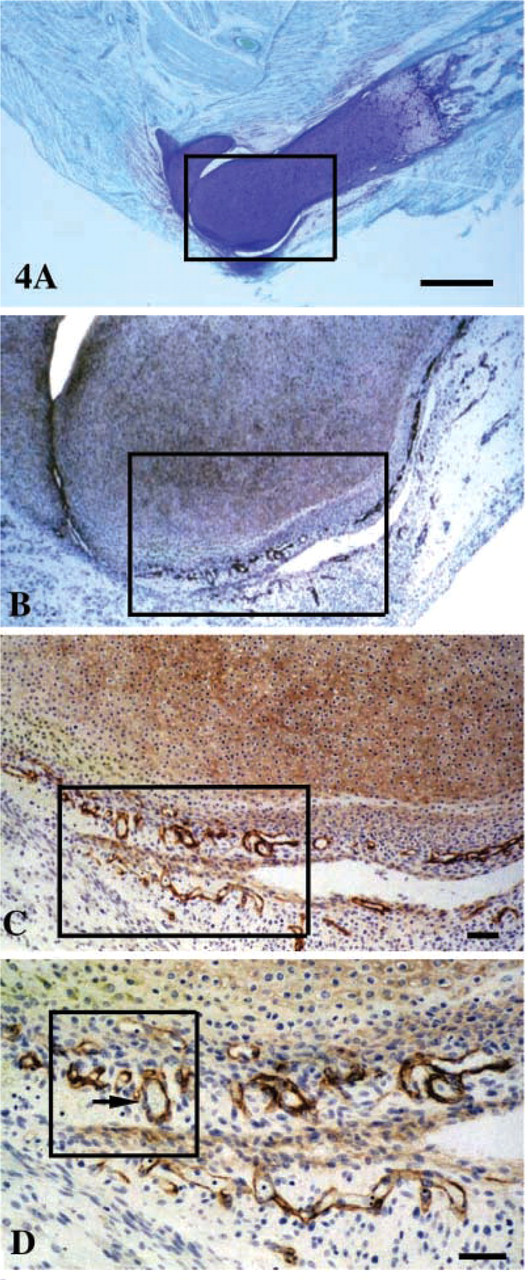
Histochemical localization of anionic proteoglycan using toluidine blue/fast green (
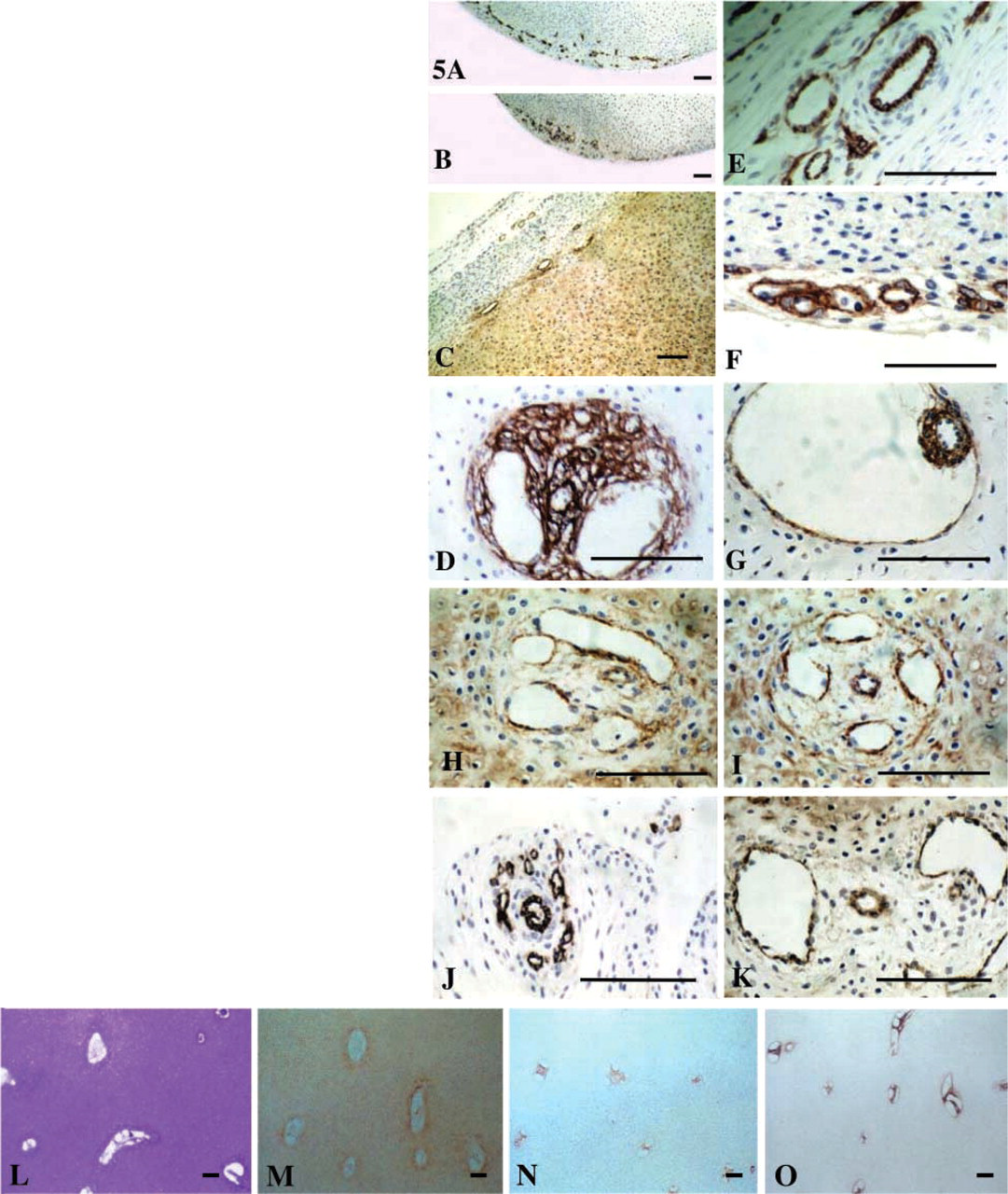
Immunolocalization of perlecan (
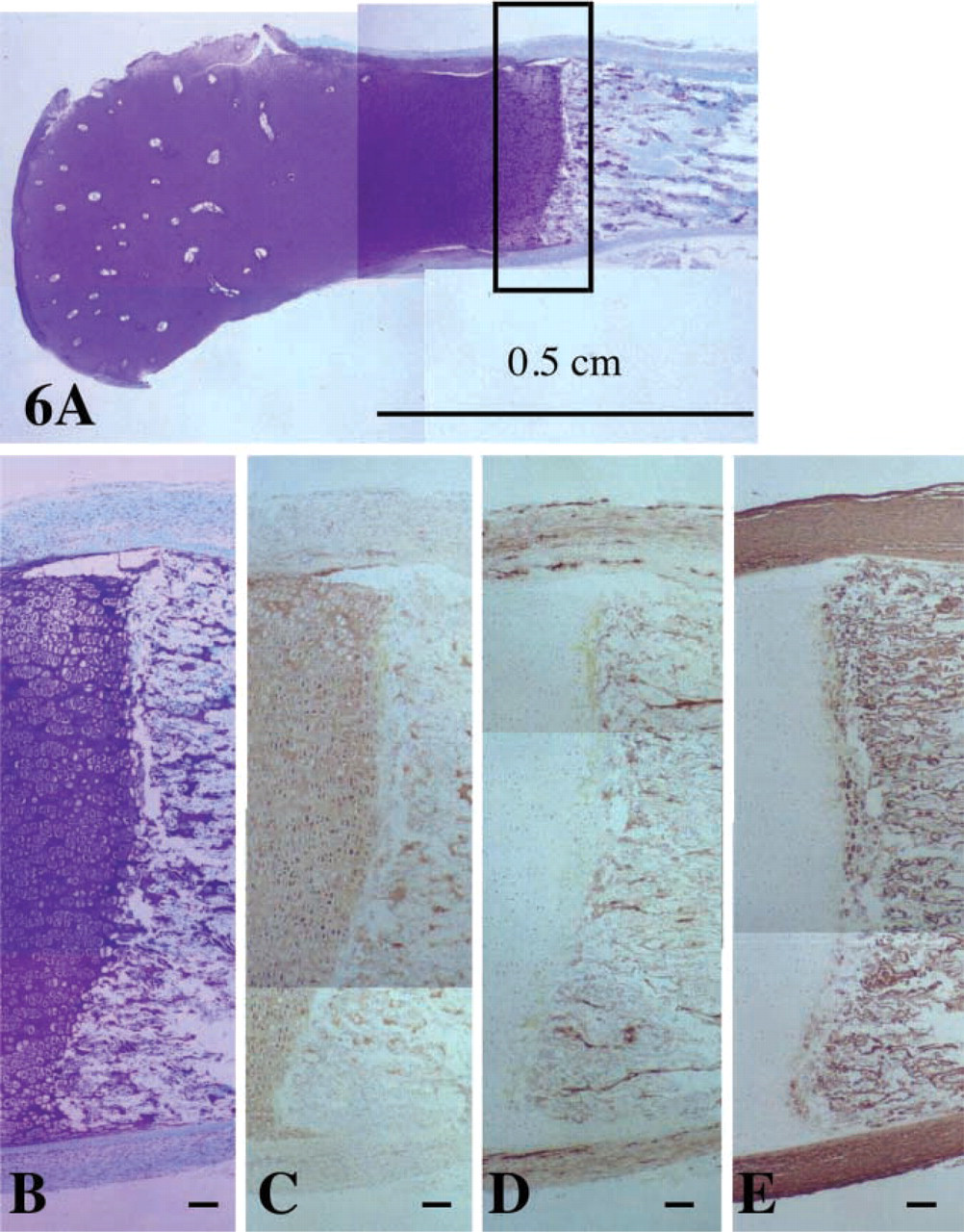
Composite figure depicting the distribution of anionic proteoglycan (toluidine blue/fast green) in low-power (
The major emphasis of the present study was to elucidate early vascular events associated with joint and long bone development. Perlecan was an abundant pericellular component of rudiment and growth plate chondrocytes. Therefore, the distribution of per-lecan observed in the present study was consistent with perlecans' known chondrogenic properties (Iozzo et al. 1994; Arikawa-Hirasawa et al. 1999; Costell et al. 1999; French et al. 1999). A novel finding of the present study was the identification of perlecan as a component of intricate networks of surface vessels located between the presumptive articulating joint surfaces and in the larger manchette- type vessel arrangements located deeper in the joint rudiments. This vasculature is required for the nutrition of the developing joint rudiment and may also have an important role in the establishment of the secondary center of ossification in the epiphyses (Floyd et al. 1987; Ganey et al. 19921995; Hirano et al. 1994; Davoli et al. 2001; Lee et al. 2001; Doschak et al. 2003). Basal lamina components, including laminin and heparan sulfate proteoglycans, have previously been identified lining the cartilage canals of developing joints (Kai et al. 1992; Ganey et al. 1995) and in blood vessels associated with cartilaginous fracture callus repair tissue (Hulth et al. 1990). Despite the fact that cartilage is devoid of a basement membrane, chondrocytes are nevertheless capable of synthesizing basement membrane components such as perlecan (SundarRaj et al. 1995; Handler et al. 1997; Costell et al. 1999; French et al. 1999; Melrose et al. 2002,2003), laminin (Durr et al. 1996; Lee et al. 1997), and nidogen (McArthur et al. 2000), and these adhesive proteins may nevertheless have roles to play in cellular attachment in cartilage.
The function of perlecan in cartilage as a chondrogenic agent and as a regulator of long bone growth is well established (Iozzo et al. 1994). Perlecan is also a well-known component of blood vessels (Iozzo 2001). However, its association with perichondrial capillary networks and cartilage canals has not previously been recognized. Perlecan appears not to be a component of the proximal metaphyseal vessels of the fetal human knee and hip.
The findings of the present investigation raise the possibility that perlecan has additional roles in early vascularization events associated with the development of diarthrodial joints and the secondary centers of ossification.
Footnotes
Acknowledgements
Supported by the National Health and Medical Research Council (project grant no. 211266) and by Research Grants from the Arthritis Foundation of Australia and Rebecca Cooper Medical Research Foundation, whose support is greatly appreciated.
