Abstract
A principle of regulation of matrix metalloproteinase (MMP) activity has been introduced as the cysteine-switch mechanism of activation (Springman et al. 1990). According to this mechanism, a critical Cys residue found in the auto-inhibitory propeptide domain of latent proenzyme is important to determine whether or not activation is turned on or off. The mechanism further allows for multiple modes of activation. To determine whether or not activation is accomplished proteolytically within a rat test cartilage model, protease analysis by the neoepitope approach, which relies upon a set of antibodies, was applied. One is used to identify the MMP-9 proenzyme bearing the critical cysteine residue, the other to identify any enzyme present bearing a new NH2-terminus 89FQTFD. This is indicative of MMP-9 lacking the cysteine switch. The antibody set has been applied to frozen tissue sections and analyzed by light and electron microscopic methods. Results reveal that activation of the MMP-9 protease involves limited proteolysis resulting in propeptide domain release. Here we report the observed changes of protease form to indigenous cells and extracellular matrix, thereby making it possible to uncover the features of MMP-9 activation within a specified set of tissue circumstances where a cartilage model is transformed into definitive bone. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
M
In the skeleton and elsewhere, MMP-9 is synthesized as a latent proenzyme composed of the following parts (Björklund and Koivunen 2005; Nagase et al. 2006): a propeptide domain that imparts latency, a catalytic domain involved in the hydrolysis of the substrate and, finally, a domain ending as C terminus or hemopexin-like domain that can bind MMP-9 to gelatin (Roeb et al. 2002) or to natural inhibitors of this enzyme [TIMP-1 or TIMP-3 (Murphy and Willenbrock 1995; Olsen et al. 1997)]. In addition, MMP-9 has three fibronectin-like repeats essential for substrate recognition and binding. Distinctive to this enzyme is a collagen type V domain whose role remains unknown.
The “cysteine-switch” principle of activation introduced initially to explain the activation of MMP-1 (interstitial collagenase) was later extended to all MMP members (Van Wart and Birkedal-Hansen 1990). The latency imparted by the propeptide domain is explained by the presence of a cysteine-switch peptide that blocks the active site cleft of the catalytic domain. This peptide consists of a sequential series of amino acids centered about a critical cysteine residue. The principle proposes that the presence of cysteine prevents substrate hydrolysis by coordinating to the active site Zn2+ poised within the catalytic domain. Thus, dissociation of Cys from the zinc ion is viewed as the switch that leads to activation. X-ray crystal structure of the human MMP-9 proenzyme has been resolved at 2.4Å and supports this proposal by showing that the propeptide domain is, in fact, inserted into the active-site cleft, thus blocking access to the catalytic zinc (Elkins et al. 2002).
Whereas the underlying basis for activation does lead to the separation of the critical cysteine residue in the prodomain from the zinc ion and a concomitant exposure of the active site, the cysteine-switch model allows for multiple modes to achieve activation including (a) autolytic activation, (b) activation accomplished proteolytically, (c) organomercurial agents (commonly applied in vitro) that lead to activation and autolysis, and (d) irreversible sulfhydryl group modifiers (oxidants) that expose an autolytic site and facilitate release of the propeptide domain (Van Wart and Birkedal-Hansen 1990). Added to this collection is another group consisting of non-proteolytic agents that like some of the above may have physiologically relevance. These agents activate the proenzyme but retain the cysteine-switch peptide (Fridman et al. 2003).
In considering what is known about the specific features of the MMP-9 protease per se, little is known about the details of how its activation is achieved in a tissue. At least two scenarios for MMP-9 activation may be envisaged based upon in vitro tests. Proteases have the ability to convert the latent 92-kDa proenzyme form of MMP-9 to the active 82-kDa species (Figure 1). Non-proteolytic agents may also activate MMP-9 by an apparent disengagement or conformational change enabling the active site to function.
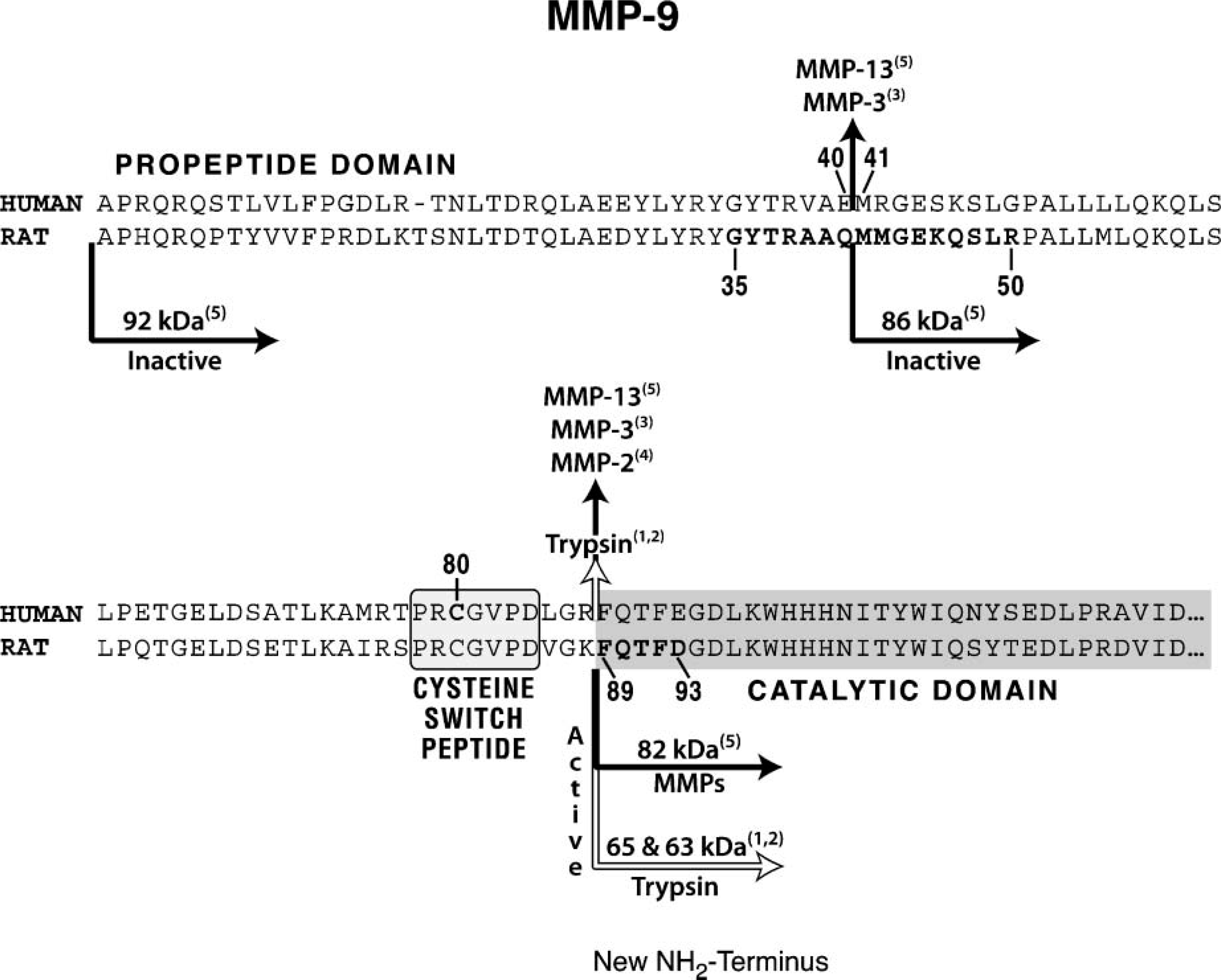
Partial amino acid sequence of human and rat matrix metalloproteinase-9 (MMP-9) proenzyme summarizing salient features about their propeptide and catalytic domains. The cysteine-switch peptide is centered around the critical cysteine residue. Also indicated on the intact protein sequence are the peptide bonds susceptible to protease cleavage and the proteases capable of producing the cuts. Numbers denote experimental sources. (1) Sopataand Dancewicz (1974); (2) Duncan etal. (1998); (3) Ogata etal. (1992); (4) Fridman etal. (1995); and (5) Knauper et al. (1997). Finally, two groups of residues are highlighted in the rat sequence (residues 35-50 and 89-93); these have been used as models to produce the set of anti-peptide antibodies described in this study. (Refer to supplemental material, Table 1S, for the sequence source.)
The basis for non-proteolytic activation is sustained by the following three facts. The first is the finding that oxidative modifications of the cysteine side-chain thiol residue (as might occur during oxidative tissue injury) can result in an active proenzyme with a retained cysteine-switch peptide (Okamoto et al. 2001; Gu et al. 2002). The second relates to the observation that dentin matrix protein 1, which is known to be present in cartilage, can bind to the MMP-9 proenzyme resulting in conformational changes that cause the proenzyme to become catalytically active without prodomain loss (Fedarko et al. 2004). Finally, the mere binding of the MMP-9 proenzyme to gelatin or type IV collagen substrates in vitro is shown to induce enzymatic activity without propeptide domain release, suggesting that the binding of MMP-9 to a ligand or a substrate may be sufficient to activate it (Bannikov et al. 2002).
The purpose of the current work is to document the fate of a MMP-9 proenzyme directly in a tissue where the MMP-9 protease is known to function (Figure 2). We reasoned that, by having antibodies able to bind to sequences poised either NH2 or COOH-terminal to the cysteine-switch peptide, it should be possible to determine the structure of the functional MMP-9 enzyme in this test model. Protease analysis by neoepitope approach has uncovered a proteolytic-based removal of the MMP-9 prodomain inclusive of the cysteine-switch peptide. Details of the conversion are related directly to the involved cells and to the extracellular matrix of cartilage that these cells are preparing to destroy, making this study the first documentation of MMP protease processing observed at high resolution in a tissue.
Materials and Methods
Preparation and Characterization of Anti-peptide Antibodies
Affinity-purified antibodies were prepared using two immunizing peptides. The first is modeled after an internal sequence (16 mer) present in the rat propeptide domain (G35-R50), and it is prodomain distinct and MMP-9 specific (Figure 1). The second antibody is modeled after the first five amino acid residues (F89-D93) indicative of the start of the rat catalytic domain (Figure 1; Supplemental Table 1).
Peptide Synthesis
Immunizing peptides (Figures 3A and 3B) and test-peptides (Figures 3C and 3D) were synthesized on an Applied Biosystems peptide synthesizer (model 431A, solid phase; Applied Biosystems, Foster City, CA). The residues (represented in bold type and bracketed) denote actual protease sequence (Figures 3A and 3B). Those in regular type represent insertions either as a cysteine residue (added for coupling to ovalbumin) or as one or two glycine residues (added as spacers). To assure purity, all peptides were purified by reverse phase chromatography using acetonitrile gradient in 0.1% trifluoroacetic acid.
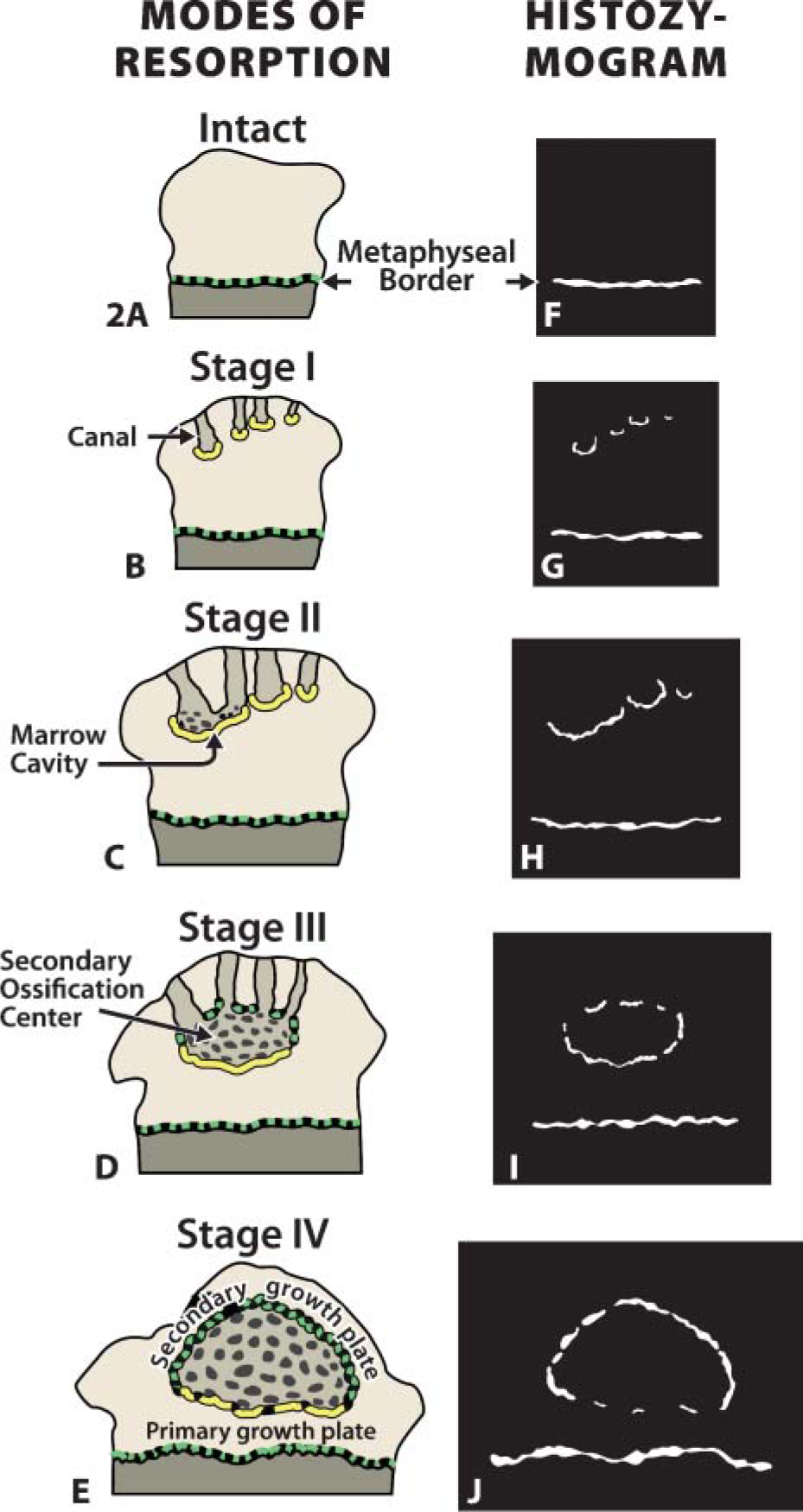
Diagram introducing the test cartilage model. Postnatal remodeling of the rat tibial epiphysis depicted in brown (
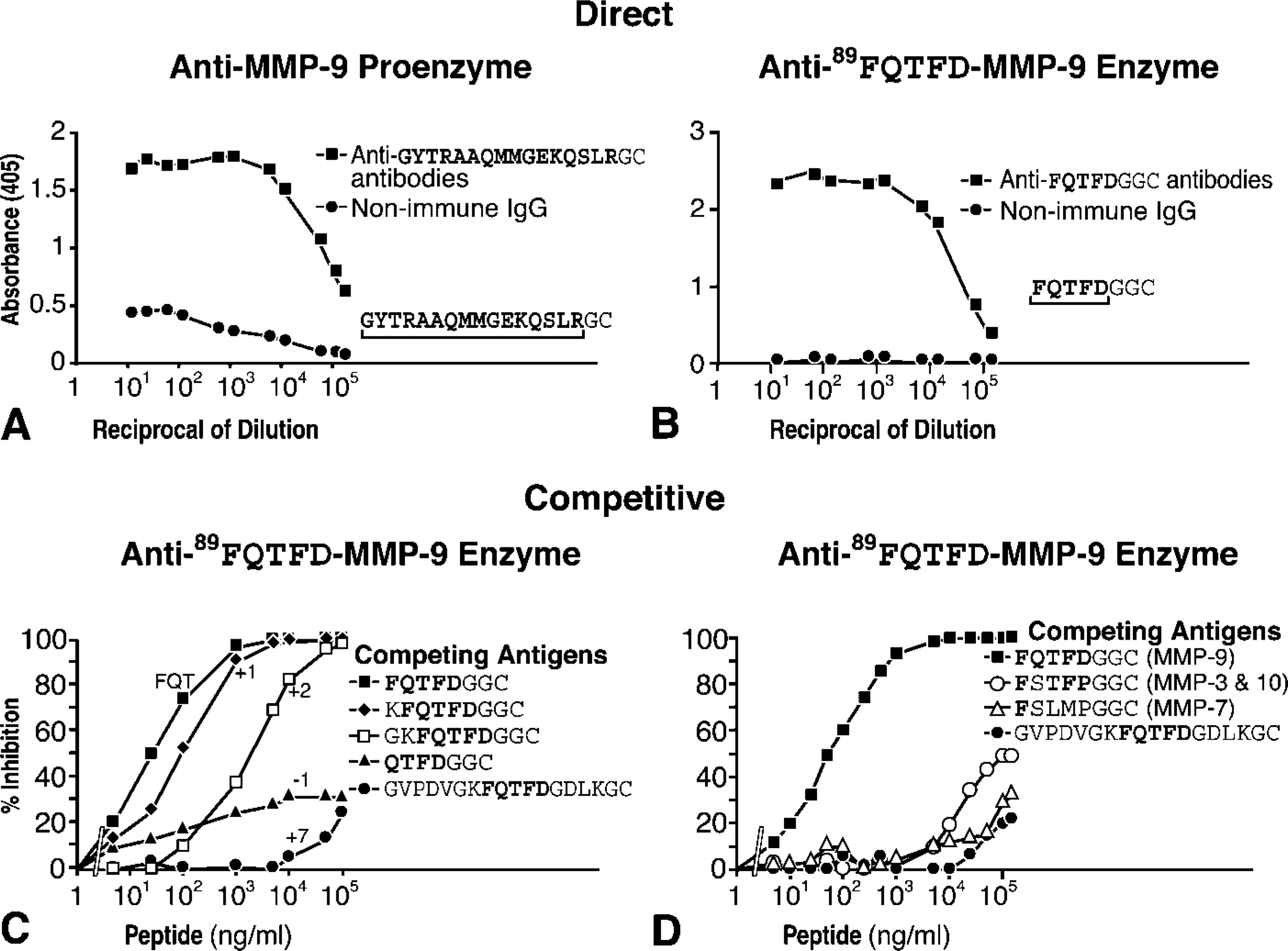
ELISA characterization of antibody set. (
Coupling
Ovalbumin (grade V; Sigma-Aldrich Canada Ltd., Oakville, Ontario) was coupled to the immunizing peptides using the bifunctional reagent N-hydroxysuccinimidyl-bromoacetate (Sigma-Aldrich). Coupling efficiency was assessed by the procedure described by Hughes et al. (1992).
Immunization
For each antibody preparation, a New Zealand White rabbit was immunized according to the guidelines of the Canadian Council on Animal Care, and all procedures were approved by the University Animal Care Committee. The schedule and method of immunization was described previously (Lee et al. 1998). Resulting antisera were affinity purified using Sulfolink Coupling Gel (Pierce; Rockford, IL) following manufacturer's instructions.
Test Assays
All experiments were carried out in triplicate and respective outcomes were plotted as the mean of the three trials (Figure 3).
Direct ELISA
Immunizing peptides were coated (50 ng/well) on Immulon 2, flat-bottomed ELISA plates (VWR Canlab; Ville Mont Royal, Quebec). Both the antibodies and non-immune IgGs were diluted to concentrations ranging from 10 ng/ml to 489 μg/ml and applied to the coated plates. Immunoglobulin binding was estimated colorimetrically with alkaline phosphataseconjugated anti-rabbit IgG, followed by exposure to p-nitrophenyl phosphate (Lee et al. 1998).
Competitive ELISA (Competitive Inhibition Assay)
The procedure was the same as above but the antibodies were first pretreated with one of the test-peptides for 1 hr at room temperature prior to application to the prepared plates. Inhibitory effect of each test-peptide on antibody binding was then assessed by changes in the absorbance readings (Lee et al. 1998) and reported in Figures 3C and 3D.
Western Blots
Antibody specificity was also tested by exposing the antibodies to material conFoining relevant protease. Materials involved in these characterizations are described below.
Recombinant Human MMP-9
Human MMP-9 proenzyme (Figure 4, Lanes 1, 2, 4, and 5) was prepared in stably transfected NSO mouse myeloma cells purified from serum-free conditioned cell culture medium using SP-Sepharose (Knäuper et al. 1997).
Rat MMP-9 Proenzyme
Rat MMP-9 proenzyme (Figure 4, Lane 3) was obtained from the medium produced by NMU rat mammary adenocarcinoma cells (CRL 1743; American Type Culture Collection, Rockville, MD) as described previously (Davoli et al. 2001).
Trypsin-activated Human Recombinant MMP-9 Enzyme (Figure 4, Lanes 2 and 5)
To obtain appropriate amounts of protein for the immunoblots shown, a volume of the human proenzyme (containing 7.2 μg of protein) was precipitated with 95% acetone overnight at −20C, centrifuged, and air dried. Activation was achieved by applying trypsin (bovine pancreas, TPCK treated; Sigma-Aldrich) as a 13.4-μl volume (containing 268 ng of trypsin suspended in 50 mM Tris-HCl, 0.5 M NaCl, 5 mM CaCl2, and 0.05% Brij 35; Sigma-Aldrich), pH 7.5, for 30 min at 37C. The reaction was terminated by the addition of a 1.6-μl volume of 0.0625 mM Pefabloc (Roche Diagnostics; Laval, Quebec).
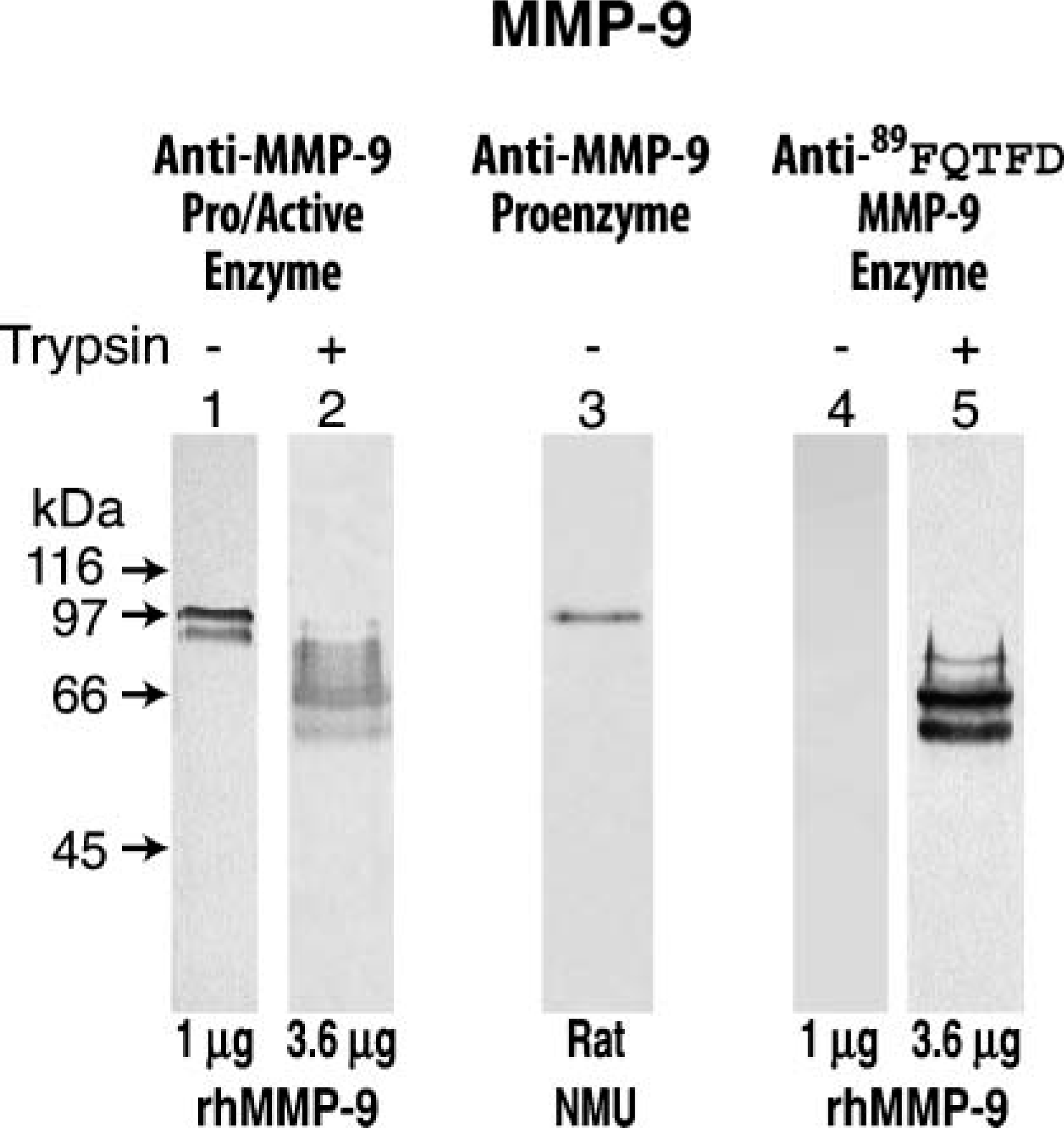
Immunostaining on Western blots of human and rat enzymes. Western blots of two materials are presented, a recombinant human MMP-9 (rhMMP-9) sometimes pretreated with trypsin (+) and an extract prepared from rat cultured carcinoma cells (NMU). Th rhMMP-9 exhibits two bands with the upper band representing the MMP-9 intact proenzyme (standard, Lane 1) and the lower band (representing the 86-kDa species produced by a cut at the Glu40-Met41 bond (Figure 1) Knäuper et al. 1997). Untreated rhMMP-9 yields no reaction following application of the anti-89FQTFD-MMP-9 antibodies (Lane 4). However, a group of associated bands appears after trypsin treatment (Lane 5); all commence with the NH2-terminal sequence, FQTFD (Figure 1) Duncan et al. 1998).
SDS-PAGE and Immunoblotting Techniques
All enzyme materials were resolved under reducing conditions in 8% mini-polyacrylamide gels and visualized by immunostaining after transfer to nitrocellulose paper. Standard immunoblots (Figure 4, Lanes 1 and 2) were prepared for the MMP-9 material (sheep anti-pig MMP-9, 4 μg/ml) (Murphy et al. 1989). Experimental immunoblots were prepared by testing the antibodies against the human material (anti-89FQTFD-MMP-9 antibodies), antiserum diluted 1:200, and treated, respectively, in the presence or absence of trypsin (Lanes 4 and 5). In Figure 4, Lane 3, anti-proenzyme specificity was tested on rat proenzyme material (antiserum diluted 1:200). In all cases, reactivity was visualized by incubating the membrane with alkaline phosphatase-conjugated anti-rabbit or anti-sheep immunoglobulins (Jackson ImmunoResearch Laboratories; West Grove, PA) followed by exposure to nitro-blue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate (BCIP/NBT Color Development substrate; Promega, Madison, WI) according to the manufacturer's directions.
Animals and Tissue Preparation
Sprague Dawley rat pups aged 5, 8, 10, or 15 days, housed and handled as recommended by the Canadian Council on Animal Care, were anesthetized IP with 50 mg/kg sodium pentobarbital (Somnotol; MTC Pharmaceuticals, Cambridge, Ontario). Animals were exsanguinated under anesthesia by perfusion with lactated Ringer's solution into the left ventricle for 20 sec while the right atrium was opened. Perfusion continued for an additional 5 min at room temperature by replacing the Ringer's with a fixative solution (periodatelysine-paraformaldehyde) prepared as described by McLean and Nakane (1974). Proximal tibiae were harvested and processed for immunohistochemistry. Briefly, tibiae were immersed in fixative overnight at 4C and then decalcified for 12 days in 10% EDTA (w/v) in 0.1 M Tris-HCl, pH 7.4. Tissue was rinsed in PBS and infiltrated with and embedded in a 2:1 volume of sucrose (20% w/v in PBS) and optimal cutting temperature compound (Somagen; Edmonton, Canada). Cryostat sections were prepared at a thickness of 8 μm or 30 mm, respectively, mounted on gelatin-coated slides, and stored in a sealed slide box at −20C until further use.
Immunohistochemistry
Light microscopic immunochemistry followed the method of Lee et al. (1998,2001). Antibody reactivity was localized in the sections by an indirect immunoperoxidase technique performed at room temperature following manufacturer's directions (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA).
Immunoelectron Microscopy
Processing and immunostaining of frozen tissue sections for electron microscopy was as described by Lee et al. (1998). Briefly, preparation and immunoprocessing of the tissue for electron microscopy were as described above for light microscopy, but with several modifications. Whereas antibody concentrations remained the same, they were applied to 30-μm-thick tissue sections and binding was visualized by the horseradish peroxidase (HRP)-DAB-H2O2 method as before, but following exposure to the chromagen the sections were osmicated, dehydrated in acetone, and embedded in epoxy resin. Ultrathin sections were prepared and examined in a Philips 400 electron microscope (Philips; FEI Company, Toronto, Ontario, Canada) at 80 kV in the absence of any counterstaining.
Routine Electron Microscopy
Tibiae were dissected from rats that had been perfused through the heart with a mixture containing 2.5% glutaraldehyde (highly purified; J.B. EM Services, Pointe-Claire, Quebec), 2.0% formaldehyde (freshly prepared from paraformaldehyde) in 0.1 M cacodylate buffer, pH 7.3. All samples were postfixed in potassium-reduced osmium tetroxide, dehydrated in acetone, and embedded in JEMbed epoxy resin before examination at 80 kV.
Immunostaining by Various Antibodies of the Cells Rich in MMP-9-Proenzyme
Sections were immunostained with antibodies to detect cell antigens. Relevant details about these antibodies are summarized in Table 1. Finally, processing and immunostaining of the frozen tissue sections for light microscopy were as described above.
Summary of canal cell population responses to common antigenic markers a
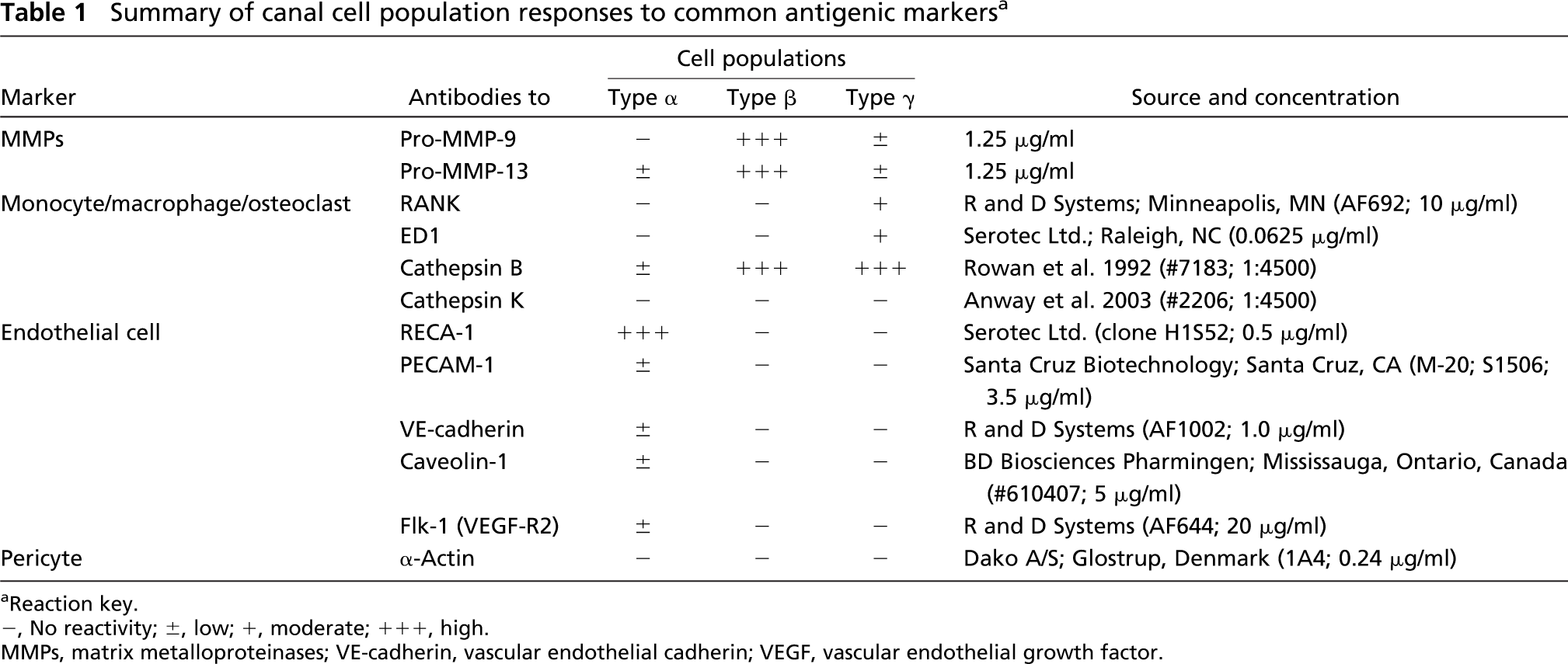
Reaction key.
-, No reactivity; ±, low; +, moderate; +++, high.
MMPs, matrix metalloproteinases; VE-cadherin, vascular endothelial cadherin; VEGF, vascular endothelial growth factor.
Tartrate-Resistant Acid Phosphatase Activity (TRAP)
To determine whether MMP-9 proenzyme-rich cells are TRAP positive, 8-μmm-thick frozen sections were exposed to filtered incubation medium in the presence of 20 mM sodium tartrate (Sigma-Aldrich) for 2 min at 37C (Cole and Walters 1987; Shevde et al. 1994).
Results
Because it was not possible to purchase or acquire the antibodies that were needed, we began by making and characterizing our own. Accordingly, anti-peptide antibodies were prepared using two immunizing peptides: one based on a 16-mer sequence in the rat propeptide domain, the other modeled from the five amino acid residues located at the beginning of the rat catalytic domain (Figure 1).
Specificity of the antibodies was confirmed by routine ELISA (Figures 3A and 3B). Competitive inhibition ELISA was used for definitive specificity studies. The latter test consisted of exposing the anti-89FQTFD-MMP-9 antibodies to test-peptides based on the immunizing peptide sequence (Figure 3C) or to test-peptides whose sequence alternatively mimicked other MMPs (Figure 3D). Both tests involved generating a standard inhibition curve with the immunizing peptide FQTFD (Figures 3C and 3D) against which the inhibition curves of the test-peptides were compared.
Figure 3C revealed the requirements for antibody binding to antigen. Removal of the Phe residue (-1) was shown to diminish the inhibition exerted by this test-peptide showing the Phe residue to be important for antibody binding. On the other hand, addition of one residue at the NH2-terminus had little effect (+1). However, with further additions inhibitory binding was reduced and eventually lost (+2, +7). Significantly, antibodies were shown to be incapable of binding to an extension inclusive of part of the cysteine-switch peptide (+7). Collective findings revealed that the inhibitory binding of the anti-89FQTFD-MMP-9 antibodies was substantial but only when the end sequence of the terminus of the test-peptide was similar to the immunizing one.
Figure 3D reported the response of the anti-89FQTFD antibodies to test-peptides imitating other MMP enzymes (Table 1S, Supplemental Material). The absence of response produced by these test peptides was confirmed because only the immunizing peptide produced inhibition.
The final step of the characterization consisted of demonstrating appropriate binding of the antibody set to the MMP-9 protein by SDS-PAGE and Western blotting techniques. To test the properties of the anti-89FQTFD antibodies toward the protein, these anti-peptide antibodies were exposed to immunoblots containing electrophoretically separated recombinant human MMP-9 proenzyme (Figure 4). The anti-89FQTFD antibodies immunostained the trypsin-treated recombinant enzyme (Figure 4, Lane 5) but not its untreated counterpart, confirming that prodomain cleavage was needed for the antibodies to bind (Figure 4, Lane 4). Additionally, binding by the anti-MMP-9 proenzyme antibodies was accomplished but only when the propeptide domain was present on the enzyme. Binding by the anti-proenzyme antibodies to a single band indicative of the rat proenzyme confirmed them as specific for the proenzyme (Figure 4, Lane 3) (Lyons et al. 1991).
mmunohistochemistry
To achieve the goals initially specified for the test-tissue model, analysis of protease by neoepitope began by assessing antibody binding in frozen sections prepared from animals aged 5-15 days.
Stage 1 Epiphysis
We observed in confirmation of our previous reports that stage I epiphysis is characterized by two to four canals entering from the superior surface and growing toward its center (Lee et al. 2001) (Figure 2B). A vasculature-cell complex is present in the lumen of the canal base. Poised adjacent to the lumen is a modified cartilage layer or preresorptive cartilage layer whose thickness ranges from 3.3 to 14.3 mm (mean 6.3 ± 2.5 μm). Our first objective was to determine how the antibody set responded to the functional MMP-9 protease uncovered previously in this layer (Figure 2B) (Davoli et al. 2001; Lee et al. 2001).
Parallel series of tests were applied to sequential frozen tissue sections to compare the immunoreactions produced by the anti-89FQTFD-MMP-9 or the anti-MMP-9 proenzyme antibodies at the canal blind end (Figures 5E-5F and Figures 6E-6F). Bound immunoglobulin was observed as brown immunoreactions produced by HRP-DAB-H2O2 methods. Controls consisted of substituting the affinity-purified antibodies with non-immune IgG at equivalent concentration (Figures 5A-5D and Figures 6A-6D). In both control cases the tissue was unstained, confirming the specificity of the immunostainings produced by the antibody set. Intriguingly, immunoreactions indicative of the proenzyme were localized mainly in mononuclear, canal cells (Figure 6H), whereas staining indicative of the cleaved protease was found mainly in the preresorptive cartilage layer (Figure 5H). In summary, two molecular forms of MMP-9 were uncovered, each in different elements of the tissue.
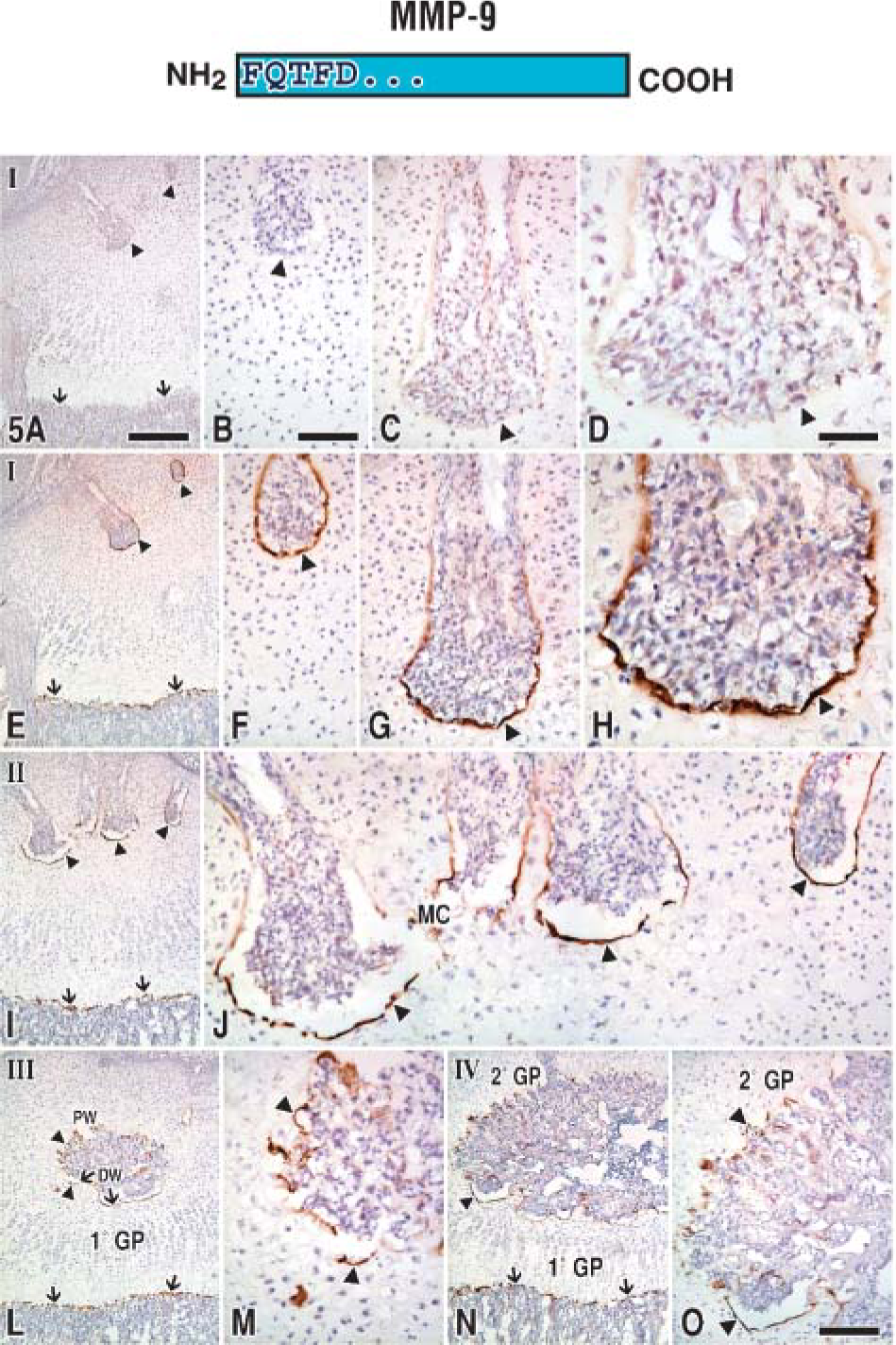
Epiphyses at stages I-IV immunostained with anti-89FQTFD-MMP-9 antibodies. This is the first of two figures (Figure 5 and Figure 6) applying the same layout. Both contain sequential sections and show the results produced by immunostaining epiphyses at various ages identified by antibody set member (top) or with non-immune IgG (presented as a control as a single row in
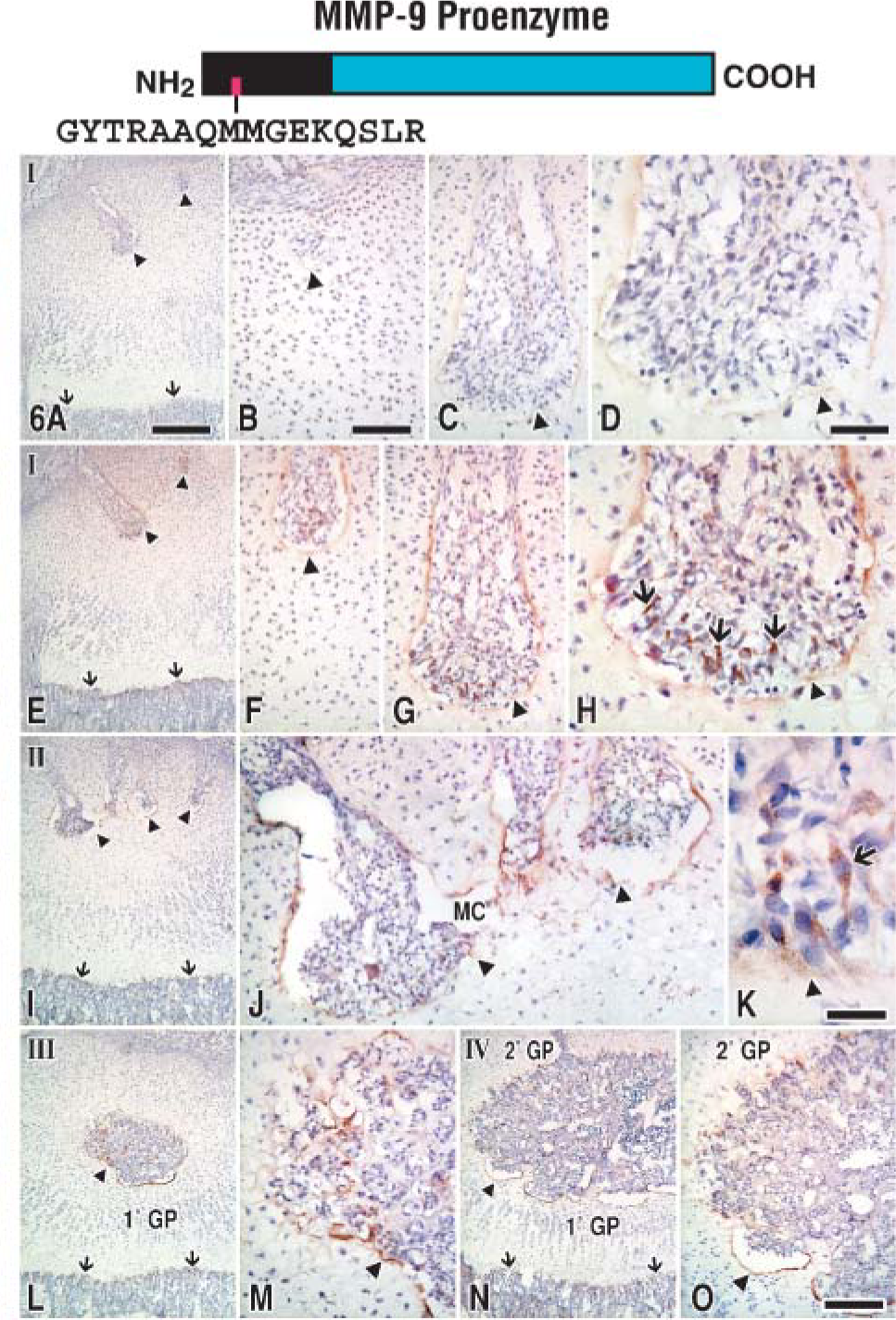
Epiphyses at stages I-IV immunostained with anti-MMP-9 proenzyme antibodies. Arrows pinpoint brown-colored canal cells (
Stages II-IV Epiphyses
To determine the fate of MMP-9 at other cartilage remodeling sites, tissue from older animals was treated with the antibody set (Figures 5I-5O). Other preresorptive layers were found to immunostain with the anti-89FQTFD-MMP-9 antibodies (stage II, Figures 5I and 5J; stage III, Figures 5L and 5M; and stage IV, Figures 5N and 5O) as they had done in the younger specimens. Immunoreactions obtained with the anti-89FQTFD antibodies corresponded with the sites shown to contain active MMP-9 protease by histozymography (Figures 2G-2J). We conclude that the active enzyme involved in cartilage resorption is the form that lacks a prodomain.
We next characterized immunoreactions by high-resolution immunoelectron microscopy by choosing to focus on the best known of the cartilage remodeling sites or the stage I canal base, even though our understanding was not entirely complete. Our first step was to uncover the cell(s) involved in MMP-9 synthesis. However, little was known about the cells in the lumen at the canal base; therefore, we began by examining and characterizing them. Three types of mononucleated cells were identified within the canal lumen by electron microscopy: (a) cells named “type α” were the regular endothelial cells lining canal blood vessels (Figure 7A) and are, as expected, tied to one another by tight junctions. (b) Cells named “type β” cells (Figures 7B and 7C) were characterized in two ways. First, their organelles, Golgi apparatus, rER, and small vesicles were prominent and indicative of high synthetic activity. Second, they were attached by junctional complexes to type a cells next to which they were often located. (c) Cells named “type γ” cells (Figure 7D) bore some resemblance to the cathepsin-B-rich cells resorbing the primary growth plate cartilage and were named “septoclasts” (Lee et al. 1995). Osteoclasts were only occasionally encountered in the canal lumen and, therefore, are unlikely to play a major role in local resorption events as was previously reported by Cole and Wezeman (1987).
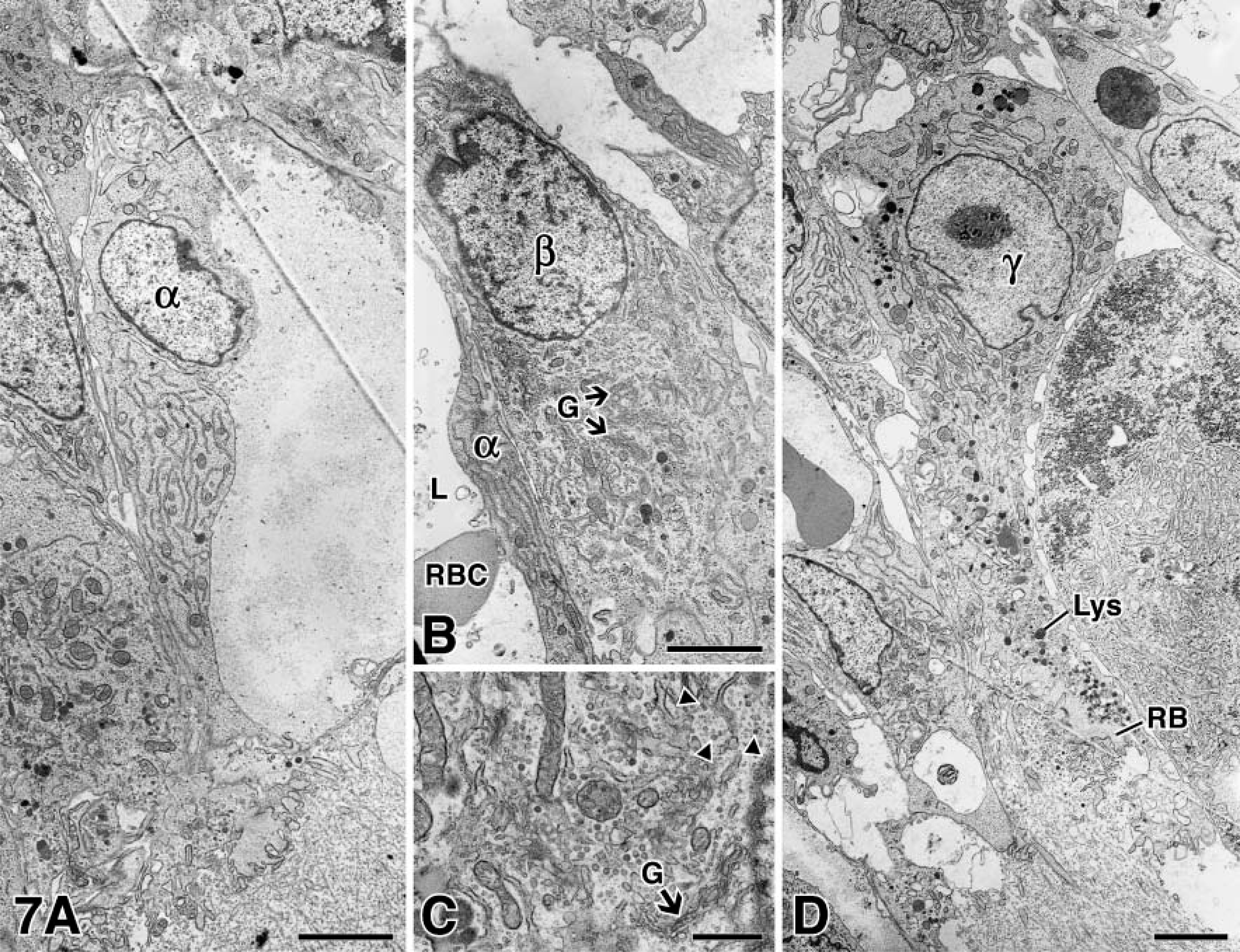
Routine electron microscopy of cells at the canal blind end. (
MMP-9 producer cell was uncovered by immunostaining frozen tissue sections with the anti-MMP-9 proenzyme antibodies and then examining the tissue by electron microscopy. Under these conditions, controls appeared dull and gray (Figure 8B). However, binding of the antibodies visualized by the presence of HRP-DAB-H2O2-OsO4 complexes was imparted with an electron density at immunostained sites (Figure 8A). The β cells were mainly immunostained by the anti-MMP-9 proenzyme antibodies (Figure 8A; Table 1), identifying them as the main producers at the canal blind end. Intriguingly, the preresorptive layer was unstained (data not shown), confirming that the proenzyme form is detectable during its synthesis in the β cell but not thereafter.
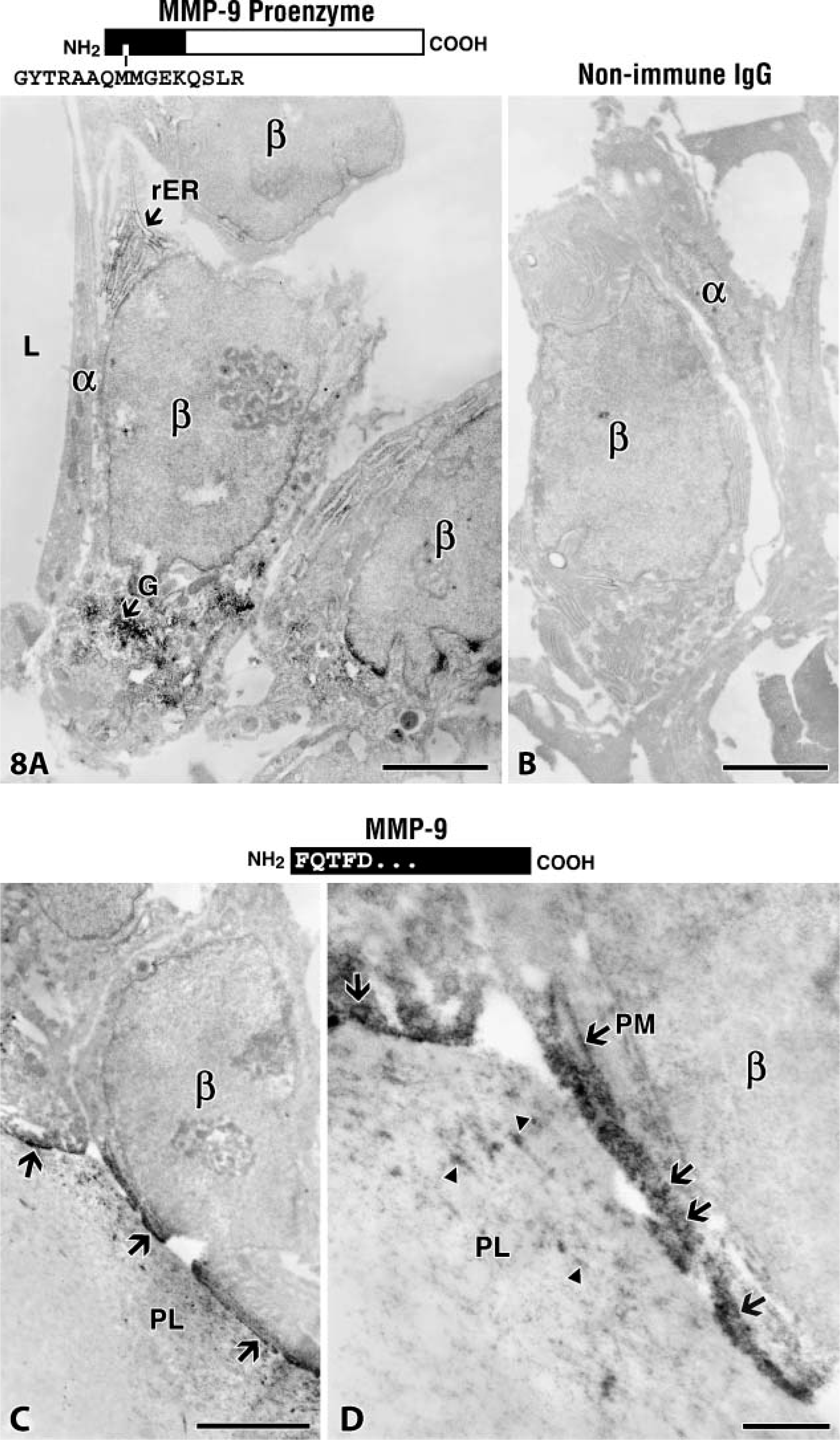
Electron microscopic immunostaining of canal cells. (
Because the β cell plays a key role in the elaboration of the MMP-9 protease, further steps were taken to learn more about it. Accordingly, β cells were examined for their response to other antibodies. The β cells were shown to be unreactive to most cell-related antibodies examined by light microscopy (Figure 9) and as independently confirmed by immunoelectron microscopy, which enabled definitive conclusions to be drawn (Table 1). Additionally, β cells did not exhibit TRAP when tested for acid phosphatase activity (compare Figure 9C to Figure 9A). Combined microscopy approaches confirmed the α cell as endothelial and the γ cell as a preosteoclast (Table 1). However, although antibodies did uncover other proteases present within them (MMP-13 proenzyme) (Figure 9B) and cathepsin B (Figure 9F; Table 1), identity of the β cell still remains distinct.
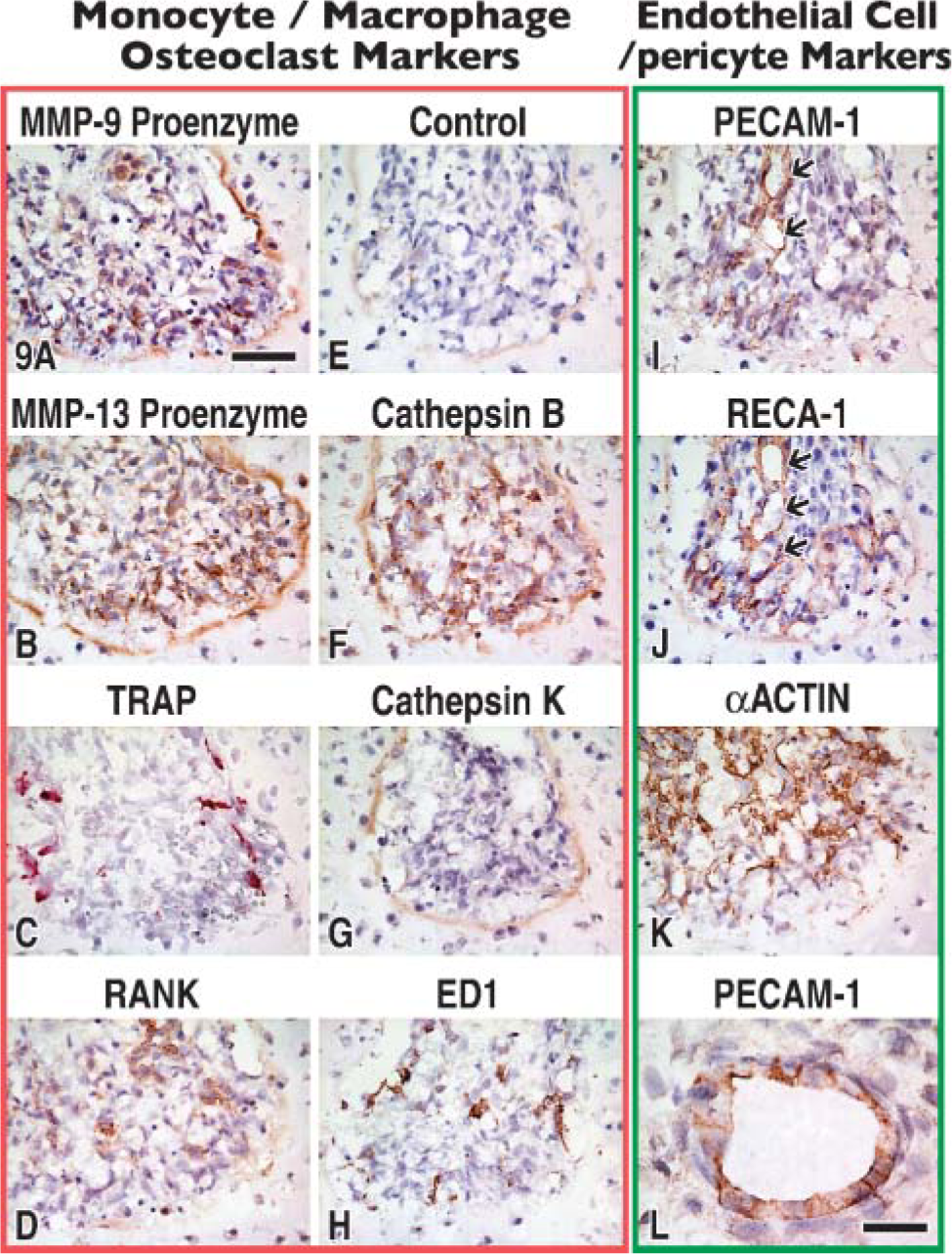
Stage 1 canal blind end summarized with the help of sequential sections immunostained (as indicated) with antibodies directed to macrophage/osteoclast markers or endothelium and associated cells. Immunostaining is detected by a brown color except in
The next line of inquiry was to determine whether or not the anti-89FQTFD-MMP-9 antibodies immunostained β cells. We found that they did (Figures 8C and 8D). Immunostained cells bore vesicles containing reactive material indicative of the activated protease. Vesicles were observed in various stages of fusing with the plasma membrane (Figure 8D). Plasma membrane was also found reactive where it faced the modified cartilage of the preresorptive cartilage layer in which scattered reactivity was accruing (Figure 8D). We conclude that the proteolytically processed protease is transported in vesicles close to the plasma membrane of the cells, but only where the cell opposes the tissue target.
The final step in the assessment involved the preresorptive cartilage layer. This layer was initially defined as a modified cartilage layer that immunostained with antibodies indicative of MMP-generated aggrecan core protein cleavage (Lee et al. 2001). The target revealed by these antibodies was G1-341 fragments. We now know that the anti-89FQTFD-MMP-9 antibodies produce exactly the same outcome in the preresorptive layer, at least when their immunoreactions are viewed by light microscopy (Figure 5H) (Lee 2006). In addition to the cleavage of aggrecan, however, which is believed to occur as a rapid, local event (at the distal limit of the preresorptive cartilage layer) (Figure 10A), more gradual changes involve the second cartilage component or collagen fibril as the fibrils thin and eventually disappear when the layer terminates as a proximal limit (Figures 10A and 10B). With the aid of the electron microscope, we concentrated on the distal layer where it was possible to examine early phases in the break-up of the components. Immunoreactions produced with our anti-89FQTFD-MMP-9 antibodies (Figure 10D) or with the anti-FVDIPEN antibodies revealing the presence of G1-341 fragments (Figure 10E) are shown. Both produced reactions at the layer's distal limit and colocalized there with fibrillar collagen. A complex comprised of fibrillar collagen and aggrecan core protein was the target of the processed MMP-9 protease.
In conclusion, the collective findings have shown protease analysis by the neoepitope approach to impart a new precision in the examination of enzyme behavior and processing because the origin of the MMP-9 proenzyme has been followed in situ from its synthesis in the β cell to its target in the preresorptive cartilage layer.
Discussion
Regulation of MMP activity occurs at various levels including transcription, secretion, activation, and through the operation of proteinase inhibitors (Björkland and Koivunen 2005). The approaches we have used (gelatin histozymography and protease analysis by neoepitope) have allowed MMP-9 activity and its regulation at the level of activation to be probed directly in a tissue where it was known to exert a physiological affect (Stickens et al. 2004).
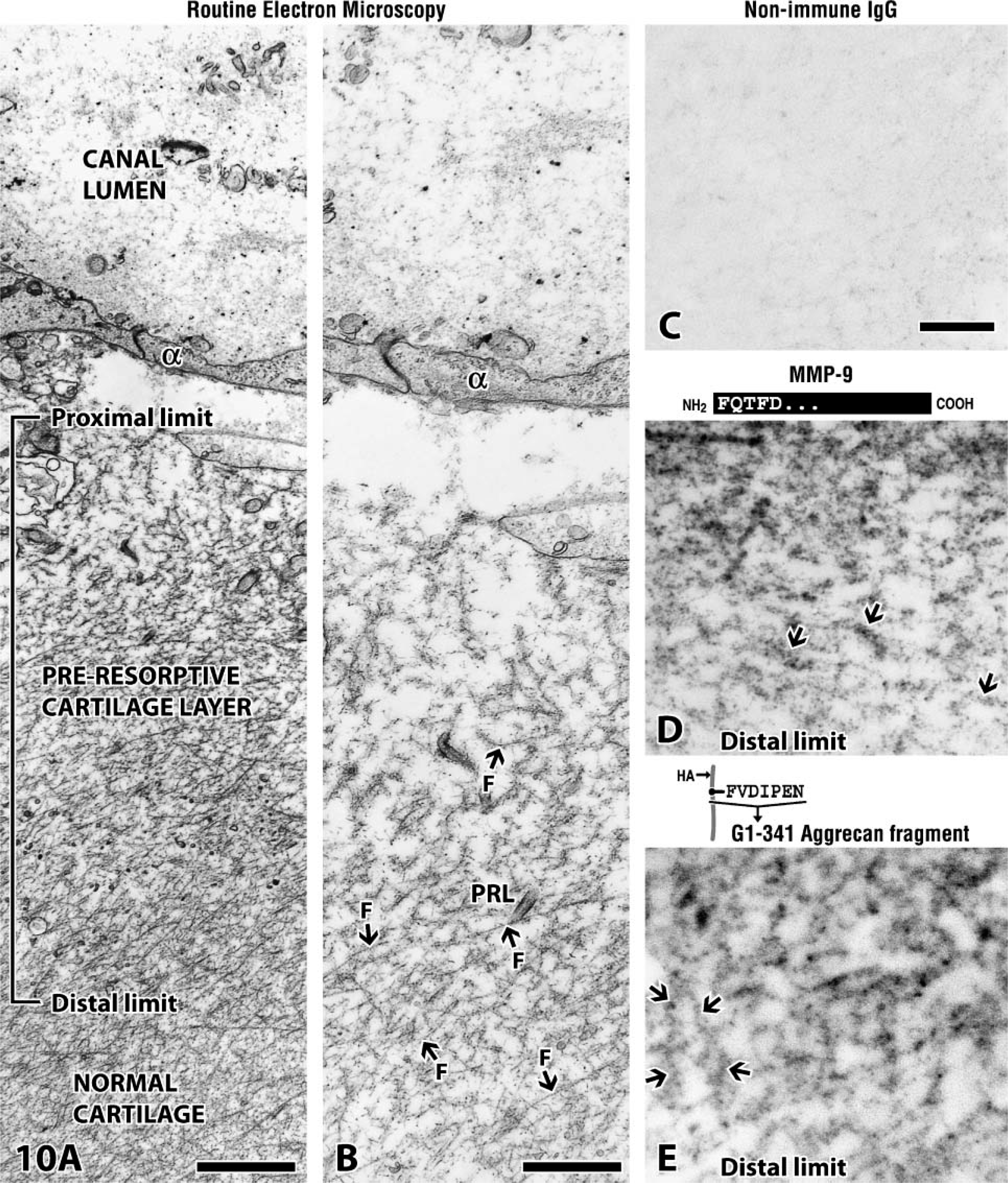
Preresorptive cartilage layer visualized by electron microscopy. (
The gelatin histozymography approach was well known (Figure 2). Protease analysis by neoepitope was not. The latter was similar to a previously described approach and used by others to biochemically monitor the intracellular transition of μ-calpain (cysteine protease) from pre- to postautolysis forms (Saido et al. 1992). The term “protease analysis by neoepitope” was chosen to precisely define the identity of the target under study (Lee 2006). To the best of our knowledge the present article represents the first of its kind to examine the precise transition of MMPs directly in a tissue.
MMP-9 Activation
The presence of two MMP forms was revealed by the approach in the current study: the MMP-9 proenzyme and a MMP-9 species from which the propeptide domain was removed. Because proteolysis is required to generate the neoepitope present on the latter, the MMP-9 proenzyme produced in this tissue must have been subjected to limited proteolysis. Was the described processing autolytic or was it generated by another protease? Data obtained in vitro by others definitively indicate that MMP-9 cannot generate the detected cleavage itself or once activated cannot produce it in other MMP-9 (proenzyme) molecules (Figure 1). We conclude that a protease other than MMP-9 must accomplish the observed cleavage in the tissue.
MMP-9 Transitions at the Canal Blind End
Protease analysis by neoepitope approach permitted us to assess other aspects of MMP-9 activation and, although less definitive, to gain insight into where it might occur. Assessment in both cases was assisted by the fact that we evaluated one site in detail that enabled us to weigh our findings against the mechanisms proposed for MMP-9 activation by others (Van Wart and Birkedal-Hansen 1990; Okamoto et al. 2001; Bannikov et al. 2002; Gu et al. 2002; Fedarko et al. 2004).
We reasoned that if the MMP-9 proenzyme were to bind to a substrate(s) within the preresorptive cartilage layer and either acquire activity as a proenzyme through conformational change or achieve it via localized proteolytic modification, in either case the the proenzyme should be detectable within the layer. However, only trace amounts of MMP-9 proenzyme were found, a finding confirmed by electron microscopic examination. These observations implied that either little of the proenzyme reached the layer or that it was incapable of binding to the target(s), thus explaining why it was not retained in the tissue. Because the MMP-9 protease exhibited the potential to bind to a variety of test substrates including fibrillar collagen and denatured collagen (gelatin) in vitro either as proenzyme or active enzyme (Allan et al. 1994,1995), the latter interpretation was considered unlikely. In the model under study, the possibility was therefore dismissed that the MMP-9 proenzyme acquired activity either by binding to cartilage constituents (Bannikov et al. 2002) or to a binding partner (Fedarko et al. 2004; Ye et al. 2005) or by proteolytic cleavage after binding to sites subjected to lysis. This implies a tight regulation of MMP-9 activation by the cell as will be explained in more detail below.
Surface-associated MMP-9 has been identified in a variety of normal and tumor cells in culture (Fridman et al. 2003). At least four binding mechanisms have been identified (Fridman et al. 2003; Björklund and Koivunen 2005): (1) MMP-9 proenzyme that can associate with the α2 (IV) chain of collagen type IV, (2) the hyaluronan receptor CD44 that is involved in the membrane association of active MMP-9 and is implicated in cancer cell invasion (Yu and Stamenkovic 1999; Karadag et al. 2005), (3) a low-density lipoprotein-related scavenger receptor that can be responsible for the internalization of MMP-9 proenzyme, or (4) proteins like RECK (
In the current article, the active MMP-9 enzyme was revealed in vesicles observed to merge with the plasma membrane of the β cell (Figures 8C and 8D). One or more of the binding mechanisms listed above (2 and 4) could theoretically explain this observation. However, an alternative interpretation, and one that we favor, is MMP-9 protease secretion by the vesicle. Intriguingly, in working with non-malignant kidney epithelial cells it was observed that pro-MMP-9 activation is a highly regulated surface-associated process (Toth et al. 2003). MMP-9 proenzyme activation was examined by the MT1-MMP/MMP-2 axis in culture. Activation is accomplished by a cascade initiated by MT1-MMP and mediated by MMP-2. Activation has been found more efficient in the presence of purified plasma membrane fractions than activation in a soluble phase, suggesting that the concentration of MMP-9 proenzyme around the plasma membrane may favor its activation. We suspect, but cannot prove, that Figures 8C and 8D reveal MMP-9 activation commencing in the vesicle and continuing along the plasma membrane. Careful examination of the immunoreactions of the vesicles reveals them to be concentrated along the inner surface of the vesicle membrane. If present, this is where we would expect the MT1-MMP/MMP-2 axis to function.
Secretion
In contrast to the molecular mechanisms that regulate vascular invasion and the resorption of cartilage during endochondral bone formation (Provot and Schipani 2005; Zelzer and Olsen 2005), the mechanisms that regulate canal invasion in general, and β-cell expression of MMP-9 in particular, were unknown. During endochondral bone development, differentiation of chondrocytes into hypertrophic chondrocytes is key to the regulation and is achieved by the release of vascular endothelial growth factor (VEGF) by hypertrophic chondrocytes (Gerber et al. 1999; Carlevaro et al. 2000; Colnot and Helms 2001; Zelzer et al. 2001). Chondrocyte hypertrophy, however, plays no role in the process under study at the canal blind end because epiphyseal chondrocytes do not synthesize type × collagen protein or express type × collagen mRNA (Lee E, unpublished data; Alvarez et al. 2005) or do they calcify their matrix. However, some evidence does link VEGF to epiphyseal vascularization and to the transcription factor hypoxia-inducible factor-1 (Cramer et al. 2004; Maes et al. 2004; Zelzer and Olsen 2005). We presume that β-cell synthesis of MMP-9 occurs in response to a signal. The signal may be VEGF, although we did not uncover any VEGF receptor when MMP-9 protein was detectable in the β cell. Clearly, the directional migration of the β cell to the epiphyseal center is consistent with the presence of a chemical concentration gradient promoting chemotaxis. However, to get to the center, the β cell was required to destroy cartilage comprised of aggrecan and type II collagen. In an earlier article published by this group, we resolved the MMP-9 protease to the type II collagen-rich fibril, but we were unable to prove whether or not the enzyme was active (Lee et al. 1999). Here we localized a proteolytically processed form of the MMP-9 protease to a perifibrillar locale (Figure 11). Various tests have demonstrated that MMP-9 facilitates MMP-13 digestion of fibrillar collagen and degrades aggrecan core protein, albeit less efficiently than other MMPs (Harris and Krane 1972; Murphy et al. 1982,1991; Lee et al. 1999; Engsig et al. 2000; Lee 2006). The current findings therefore strengthen the case by showing that the activated MMP-9 protease binds to matrix components shown to be undergoing lysis in situ. We acknowledge, however, that other non-extracellular matrix functions are also possible (Ortega et al. 2004).
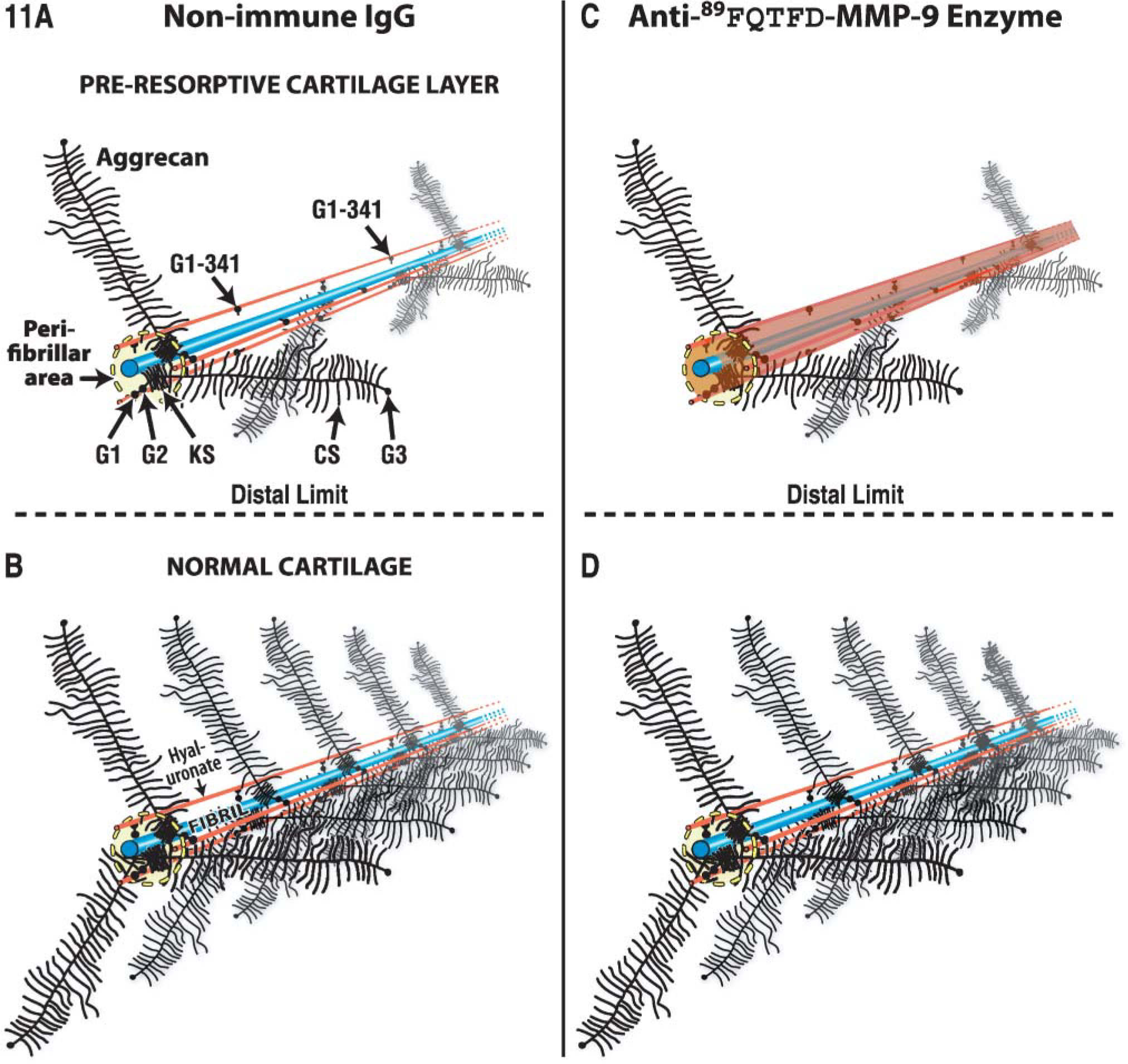
Model proposed for the col 2-aggrecan complex observed at the canal blind end after treatment with non-immune IgG (
In conclusion, involvement of the MMP-9 protease was confirmed in dependent and independent cartilage resorptions (Lee et al. 1999; Davoli et al. 2001). New and definitive information about MMP-9 processing and fate were uncovered as the MMP-9 proenzyme form was subjected to limited proteolysis for both modes of resorption. In the independent mode, transition from proenzyme to processed enzyme likely begins in vesicles involved in the delivery of the proenzyme for secretion. Additionally, the enzyme can be traced from the β cell to a cartilage target to which it was shown to bind. This binding occurs after, rather than before, propeptide domain release. Together the findings imply cell involvement at every turn, the synthesis of the enzyme, its activation and, by a mechanism unknown, its presence in cartilage as a TIMP-free form (Davoli et al. 2001). Finally, the β cell is the only cell appropriately poised, present in sufficient numbers, and enriched in the MMP-9 proenzyme to be assigned the role as MMP-9 producer cell. We suspect but cannot prove that β cells are elements of a vascular mesenchyme recruited to remove “pure cartilage” (via the “independent” mode) and sustain extramedullary (liver or spleen) or bone marrow-derived progenitor cells. We do not know whether or not β cells are related to other cells previously implicated in cartilage resorption and called septoclasts (Lee et al. 1995) or chondroclasts (Vu et al. 1998). Finally, as stated by Fridman et al. (2003), “the relevant mechanism(s) of pro-MMP-9 activation in tumor tissues and in particular at the cell surface remains elusive. Most puzzling of all is the consistent inability to detect active MMP-9 in cultured cells and tissues.” The current work represents an attempt by us to clarify this and other problems. With the help of a neoepitope approach, we show that the information sought can now be acquired in tissue by a method adapted to histology.
Footnotes
Acknowledgements
This work was supported by the Shriners of North America (grant #8530 to ERL).
Dr. Gillian Murphy (Department of Oncology, University of Cambridge, Cambridge, England) generously provided the recombinant human MMP-9 enzyme and the sheep anti-pig MMP-9 antiserum. Peptides used in this study were prepared by Ms. Elisa de Miguel in the Core Biotechnology Facility of the Shriners Hospital for Children. Illustrations were prepared by Ms. Guylaine Bedard.
