Abstract
The so-called neurointermediate lobe is composed of the intermediate and neural lobes of the pituitary. The present immunohistochemical study investigated components of the basal lamina (laminin, agrin, and perlecan), the dystrophin–dystroglycan complex (dystrophin, β-dystroglycan, α1-dystrobrevin, β-dystrobrevin, utrophin, and α1-syntrophin), and the aquaporins (aquaporin-4 and −9). Glia markers (GFAP, S100, and glutamine synthetase) and components of connective tissue (collagen type I and fibronectin) were also labeled. In the neurohypophysis, immunostaining of basal lamina delineated meningeal invaginations. In these invaginations, vessels were seen to penetrate the organ without submerging into its parenchyma. On the parenchymal side of the invaginations, β-dystroglycan was detected, whereas utrophin was detected in the walls of vessels. Immunostaining of α1-dystrobrevin and α1-syntrophin did not delineate the vessels. The cells of the intermediate lobe were fully immunoreactive to α1-dystrobrevin and α1-syntrophin, whereas components of the basal lamina delineated the contours of the cells. GFAP-immunoreactive processes surrounded them. Aquaporin-4 localized at the periphery of the neurohypophysis, mainly adjacent to the intermediate lobe but not along the vessels. It colocalized only partially with GFAP and not at all with α1-syntrophin. Aquaporin-9 was not detected. These results emphasize the possibility that the components of the dystrophin–dystroglycan complex localize differently and raise the question about the roles of dystrobrevins, α1-syntrophin, and aquaporin-4 in the functions of the intermediate and neural lobes, respectively.
T
Basal lamina separates brain tissue from the surrounding (meningeal and vascular) connective tissues. This basal lamina is produced by astrocytes (Bernstein et al. 1985) and is fused with the basal lamina of the cerebral vessels. This fused gliovascular basal lamina has essential roles in brain vascularization, gliovascular connections, organization of the blood–brain barrier, and transport processes [for review, see Sixt et al. (2001), Hallmann et al. (2005), and Wolburg et al. (2009)]. Because the neurohypophysis is modified to release neurosecretory products into vessels, it is interesting to note how the cerebral basal lamina is modified here. The basal lamina in the adenohypophysis has been investigated by immunohistochemical methods (Murray et al. 1997) and in the neurohypophysis by electron microscopy (Tweedle and Hatton 1987).
The main adhesive component between the basal lamina and the cells is laminin, a glycoprotein [for its structure, see Dow and Wang (1998) and Colognato and Yurchenko (2000)]. Agrin, another component (a heparansulfate proteoglycan), has been found to be important in the basal lamina of vessels with special barrier properties [e.g., brain, testis, and thymus (Warth et al. 2004)]. Together with another proteoglycan component, perlecan, they have roles in the formation of polarity of the cells (Warth et al. 2004; Mirouse et al. 2009). For the chemical structures see Dow and Wang (1998). The importance of the basal lamina attracted our interest in the dystroglycan–dystrophin laminin receptor complex, which interconnects the astrocytes and the basal lamina.
The dystroglycan was identified first in skeletal muscle and later also in other tissues, including brain [for an early review see Henry and Campbell (1996)]. It is required for stabilization of vascular structure and for the functional integrity of the blood–brain barrier (Tian et al. 1996; Jancsik and Hajós 1999; Nico et al. 2001, 2003, 2004; Zaccaria et al. 2001). The dystroglycan is encoded by a single gene and cleaved by posttranslational processing into two proteins, α- and β-dystroglycan (Ibraghimov-Beskrovnaya et al. 1992; Smalheiser and Kim 1995). The α-dystroglycan is an extracellular protein which binds laminin as well as agrin and perlecan (Gee et al. 1994; Henry and Campbell 1999). The β-dystroglycan is a trans-membrane protein that anchors α-dystroglycan to the cell membrane.
The other end of the β-dystroglycan forms a complex with one of the isoforms of dystrophin or, in some localizations, with utrophin, the autosomal homolog of dystrophin. The dystrophin (or utrophin) molecule connects actin, dystrobrevin (α1, α2, or β), and syntrophin (α1, β1, β2, γ1, or γ2) molecules (dystrophin-associated proteins). It also has connections with intracellular signaling pathways. For a more detailed description of the dystrophin–dystroglycan complex and its associate proteins see Chamberlain (1999), Moukhles and Carbonetto (2001), Culligan and Ohlendieck (2002), Ehmsen et al. (2002), Amiry-Moghaddam et al. (2004), Warth et al. (2004), and Wolburg et al. (2009).
The dystrophin–dystroglycan complex has a role in the distribution of the water channel protein aquaporin-4. The aquaporin-4 protein is the prevalent aquaporin in the mammalian brain (Hasegawa et al. 1994; Jung et al. 1994; Frigeri et al. 1995). It mediates movement of water between the brain parenchyma and the vascular space and therefore has a crucial role in water homeostasis of the brain (Venero et al. 2001; Agre et al. 2002; Vajda et al. 2002; Agre and Kozono 2003). Nico et al. (2001) found that aquaporin-4 was a marker for the maturation and integration of the blood–brain barrier. Aquaporin-4 occurs in astrocytes, predominantly where their perivascular endfeet contact the basal lamina. α1-Syntrophin is supposed to anchor the aquaporin-4 protein to dystrophin and thereby to the dystroglycan and the cell membrane (Neely et al. 2001; Inoue et al. 2002; Amiry-Moghaddam et al. 2003, 2004; Nico et al. 2003; Warth et al. 2004, 2005). Laminin and agrin, via their binding to α-dystroglycan, induce the aggregation of aquaporin-4 to the basal lamina (Guadagno and Moukhles 2004; Warth et al. 2004). Whereas aquaporin-4 occurs mainly in astroglial endfeet, aquaporin-9 was found to localize to astrocyte perikarya and processes (Badaut and Regli 2004). Therefore, we extended the investigations beyond those of aquaporin-4 and −9.
Because astrocytes produce basal lamina in the central nervous system (Liesi et al. 1983; Bernstein et al. 1985) and they contain the dystrophin–dystroglycan complex, as well as aquaporins (Amiry-Moghaddam et al. 2004; Warth et al. 2004) and because modified astrocytes (pituicytes) are the characteristic cells of the neural lobe, visualization of astrocytes could not be omitted from our study. Their widely accepted immunohistochemical marker is the glial fibrillary acidic protein (GFAP) (Eng et al. 1971; Bignami et al. 1980), their intermediate filament protein. However, according to several studies, not all astrocytes can be detected by the immunohistochemical reaction against GFAP (Ludwin et al. 1976; Linser 1985; Patel et al. 1985; Hajós and Kálmán 1989; Kálmán and Hajós 1989). It is useful, therefore, to apply further astroglial markers, i.e., the glutamine synthetase and the S100 proteins (Ludwin et al. 1976; Linser, 1985; Patel et al. 1985). Axons (e.g., of the hypothalamohypophyseal tract) form a considerable part of the mass of the neural lobe. To visualize them, the intermediate filament protein of neurons can be applied.
The questions are how the special function of the neurohypophysis modifies its basal lamina and dystrophin–dystroglycan complex (including the dystrophin-associated proteins and aquaporins) and how these structures differ from their correspondents found in the brain tissue in general. Due to the intermediate lobe's close position to the neurohypophysis, our study extended to the intermediate lobe, and the results have been incorporated in this report.
The present immunohistochemical study, therefore, investigates (Table 1) components of the basal lamina (laminin, agrin, and perlecan) and the dystrophin–dystroglycan complex and its associated proteins α1-dystrobrevin, β-dystrobrevin, α1-syntrophin, and aquaporin-4 and −9. The presence of connective tissue was checked by immunohistochemical reactions to collagen type I and fibronectin. Occurrences of glia and axons were estimated by detection of their markers mentioned above.
The cerebral distribution of the substances investigated in the present study has already been published in several papers (see Discussion). Our observations of brain tissues (Bagyura et al. 2007) are in accordance with those previous studies. The Results section of the present study, therefore, is confined to our new findings concerning the pituitary gland. In the Discussion section, these findings are compared with previously published findings for brain tissue.
Materials and Methods
Animals
Twelve adult Wistar rats of either sex, weighing 250 to 300 g, were used. The animals were supplied with rat food (Charles River Laboratories; Germantown, MD) and water ad libitum and kept in an artificial 12-hr light/12-hr dark period. All experimental procedures were performed in accordance with European Communities Council Directive (86/609/EEC) guidelines.
Fixation and Sectioning
The animals were deeply anesthetized with ketamine and xylazine injections (20 and 80 mg/kg, respectively, IM) and perfused through the aorta with 100 ml of 0.9% sodium chloride followed by 300 ml of 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). After hypophyses were perfused, they were removed and postfixed in the same fixative for 1 day at 4C. Serial sections (100-μm thickness, 4–6 sections per animal) were cut by a vibration microtome (VT 1000S model; Leica Microsystems GmbH, Wetzlar, Germany) in the coronal plane.
Immunohistochemistry
This method has been applied by our group in several similar studies (Szabó et al. 2004; Szabó and Kálmán 2004, 2008; Goren et al. 2006; Adorján and Kálmán 2009; Bagyura et al. 2010). Floating sections were pre-treated with normal goat serum or (in the case of the dystrobrevins) horse serum diluted to 20% in PBS (Sigma; St. Louis, MO) for 90 min to block nonspecific binding of antibodies. This and the following steps were followed by an intensive wash, stirring in abundant PBS (30 min at room temperature). Primary immunoreagents were diluted as shown in Table 1 in PBS containing 0.5% Triton X-100, and 0.01% sodium azide was added as a conserving agent. Sections were incubated for 40 hr at 4C. Fluorescent secondary antibodies (Table 2) were used at room temperature for 3 hr. The sections were finally washed in PBS (1hr at room temperature), mounted onto microscope slides, cover-slipped in a mixture of glycerol and bi-distilled water (1:1), and sealed with lacquer. Control sections were created by omission of the primary antibody in the presence of normal goat or horse serum. No structure-bound fluorescent labeling was observed in these specimens.
The primary antibodies applied in the study
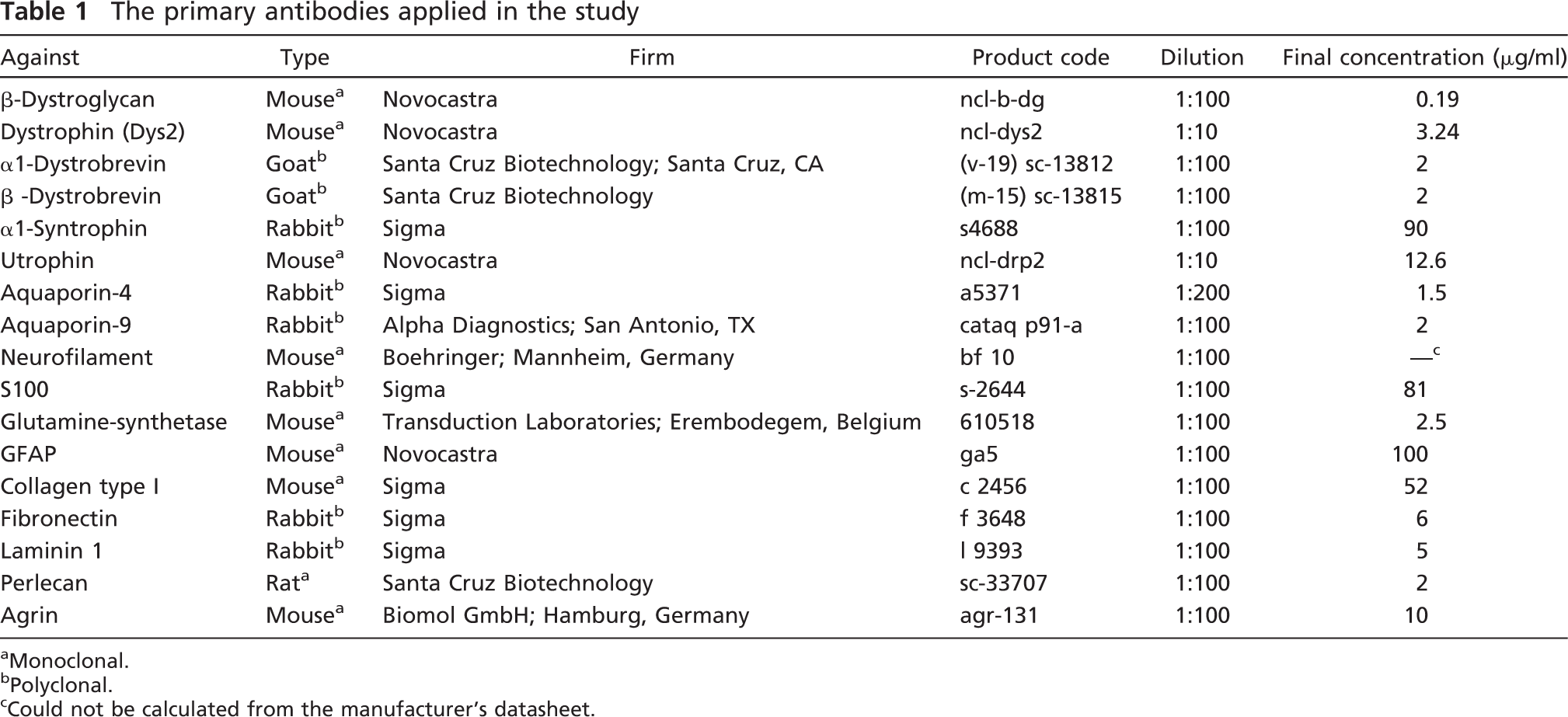
Monoclonal.
Polyclonal.
Could not be calculated from the manufacturer's datasheet.
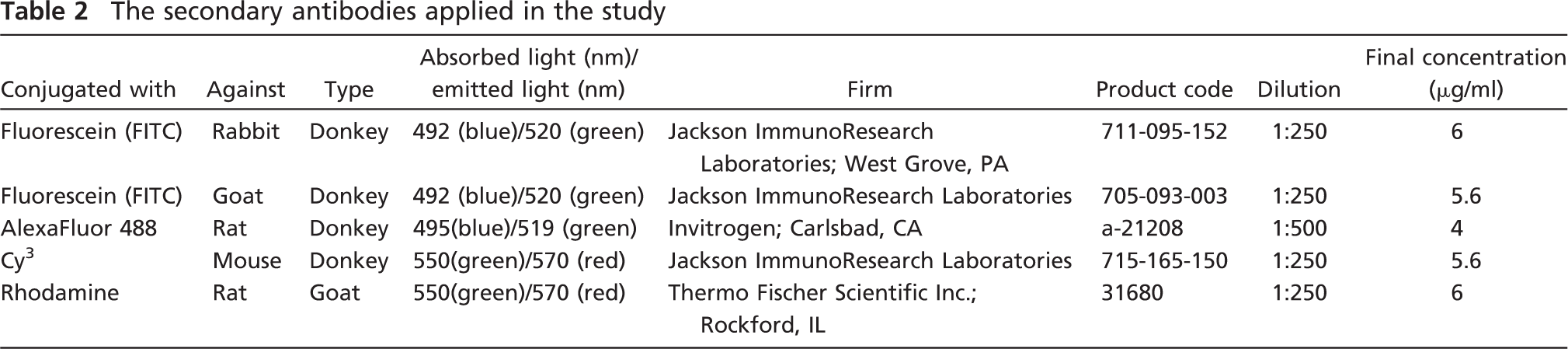
The secondary antibodies applied in the study
Double-labeling Immunofluorescent Reactions
The antigens were detected simultaneously where their antibodies were raised in different species [e.g., mouse versus rabbit (see Table 1)]. The antibodies were applied in parallel in the same incubation medium. Otherwise, the protocol was similar to that described previously. For anti-rat antibodies, green or red fluorochromes were used to enable a combination with Cy3-conjugated anti-mouse (red) or FITC-conjugated anti-rabbit (green) antibodies, respectively. Perlecan, therefore, could be either green or red (Table 2).
Confocal Laser Scanning Microscopy and Digital Imaging
Slide images were photographed with a DP50 model digital camera mounted on an Olympus BX-51 microscope (both, Olympus Optical Co. Ltd.; Tokyo, Japan) or, in the case of double labeling, with a Radiance-2100 model (Bio-Rad; Hercules, CA) confocal laser scanning microscope. Green and red colors on the photomicrographs correspond to the emitted colors of the fluorescent dyes as shown in Table 2. Digital images were processed using Photoshop 9.2 software (Adobe Systems; Mountain View, CA) with minimal adjustments for brightness and contrast.
Results
Distribution of Laminin and β-Dystroglycan Indicates the Position of the Basal Lamina
A survey of specimens following immunohistochemical reactions against laminin and β-dystroglycan showed that the two immunostaining patterns seemed to be identical (Figures 1A and 1B, respectively). In the neurohypophysis, they surround the organ and form a dense network inside, a labyrinth in which the septa proved to be double walled (Figure 1C). The walls of some septa continued on the surface of the organ. The septa, therefore, seemed to be invaginations of the basal lamina that demarcate the neurohypophyseal tissue from the meningeal tissue. Collagen type I- and fibronectin-immunopositive reactions were confined to the meningeal surface of the organ; they did not extend or extended only weakly into the invaginations (not shown in Figure 1C). In the intermediate lobe, β-dystroglycan- and laminin-immunoreactive single lines delineated smaller or larger areas which seemed almost empty, since they contained only very few small immunopositive elements (Figures 1C and 1D). These areas might correspond to the cell groups of the intermediate lobe, as suggested by further immunostaining.
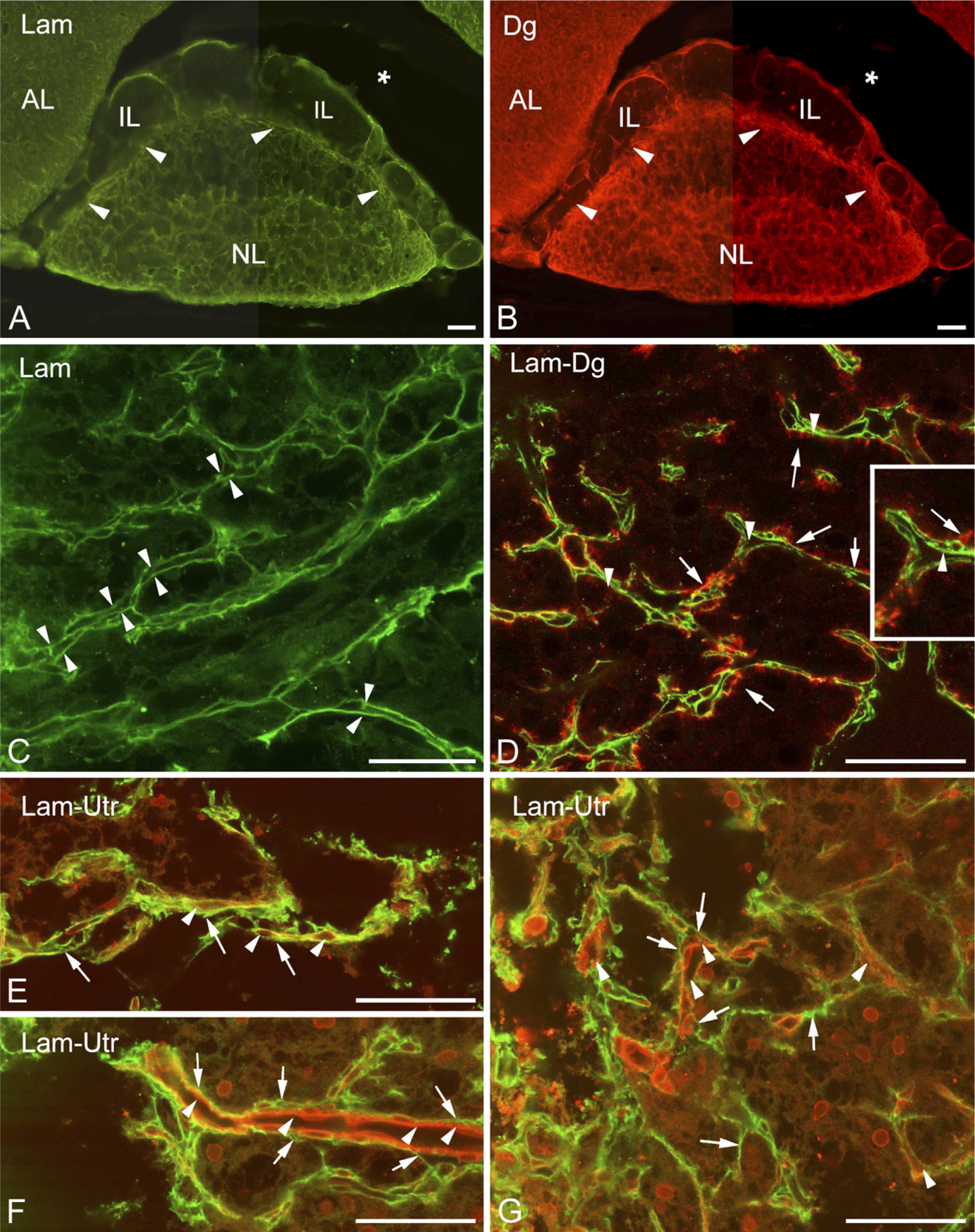
Distribution of laminin and dystroglycan in the neurointermediate lobe.
β-Dystroglycan and Utrophin Localized on Opposite Sides of the Basal Lamina in the Neurohypophysis
Following double labeling, β-dystroglycan proved to be appositional to the outer parenchymal side (i.e., the side of the neurohypophyseal tissue) of the double-contoured (bilaminar) laminin-immunoreactive septa but not in the narrow slits between the two laminin-immunopositive layers (Figure 1D). In this latter position, following double labeling of utrophin and laminin, utrophin immunopositivity was detected, visualizing the apparently vascular lumena there (Figures 1E–1G). The inner surface of the bilaminar septa (i.e., the invaginations of the basal lamina) was therefore regarded as the vascular or meningeal surface because it seemed that within the septa, vessels penetrated the neurohypophysis. No vessel-like structures were seen on the opposite, parenchymal, side of the basal lamina, applying an immunohistochemical reaction against either β-dystroglycan or utrophin.
Relationship of Basal Lamina Components and Dystrophin–Dystroglycan Complex to Cells in the Neurohypophysis
Invaginations of the meningeal basal lamina were also immunoreactive to agrin and perlecan (Figures 2A and 2B), the other basal lamina components. In addition, perlecan formed a fine mesh inside the areas separated by laminin-immunoreactive septa (Figure 2B). When immunostaining was applied against glial markers in the neurohypophysis, the majority of cells proved to be immunopositive to glutamine synthetase or S100 protein (Figures 2C–2F). The two astroglial markers did not display an exact colocalization; actually, they appeared to visualize different cell shapes (Figure 2F, insets). As shown later, GFAP-immunopositive cells were relatively scarce and occurred mainly at the periphery of the organ. Immunopositivity to β-dystroglycan, dystrophin, and perlecan were detected as layers on the surface of the S100-positive cell groups (Figures 2C–2E, enlarged in 2C inset).
Axons and Their Environment in the Neurohypophysis
The positions of the axons were detected by immunohistochemical staining of neurofilament protein. The above-mentioned laminin- and β-dystroglycan-immunopositive labyrinthine invaginations of the neural lobe surrounded and fasciculated the bundles of the neurofilament protein-immunoreactive axons (Figure 3A). S100-immunopositive cells were in close contact with axons (Figure 3B), and perlecan-containing substance enmeshed them (Figure 3B, inset).
Distribution of the Dystrophin-associated Dystrobrevin and α1-Syntrophin Proteins
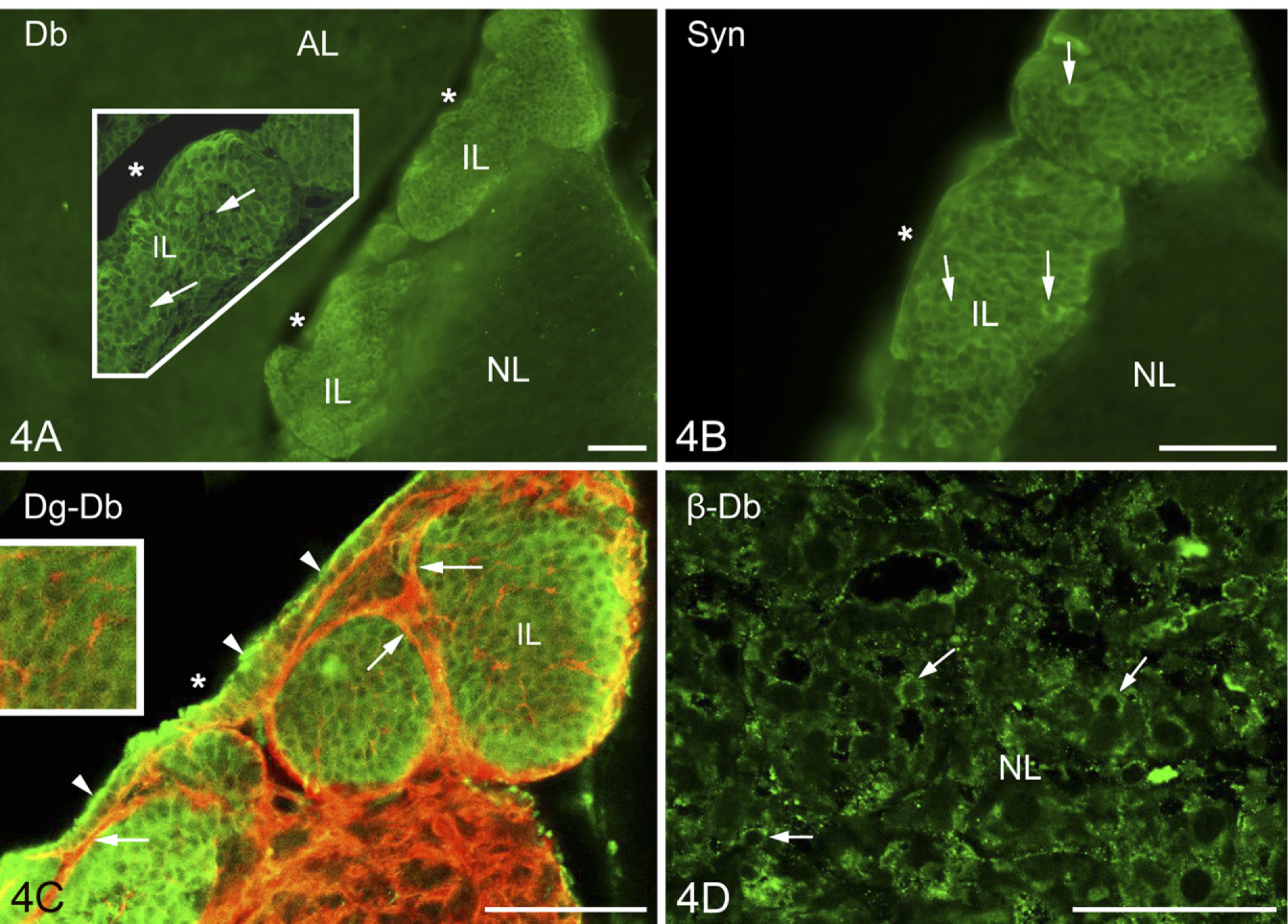
Previously it was mentioned that in the intermediate lobe, the immunostained β-dystroglycan and laminin proteins surrounded round areas which seemed to be empty under low-power magnification. The α1-dystrobrevin and α1-syntrophin proteins (Figures 4A and 4B, respectively), however, proved to be intensely immunoreactive in these areas, labeling round cell-like structures but leaving small unstained spots, which appeared to be places of nuclei. When specimens were double labeled against α1-dystrobrevin and β-dystroglycan (Figure 4C), the latter marked a conspicuous common layer around the cell groups and a delicate mesh inside, between the cells.
The cuboidal epithelium which lined the remnant of Rathke's pouch was also immunopositive to α1-dystrobrevin and α1-syntrophin. In the neural lobe, no immunoreactivity to either α1-dystrobrevin or α1-syntrophin was found, not even around the vessels. Neurohyophysial cells, however, showed β-dystrobrevin immunoreactivity (Figure 4D). Immunoreactivity to β-dystrobrevin was not observed in the cells of the intermediate lobe.
Intercellular Network in the Intermediate Lobe
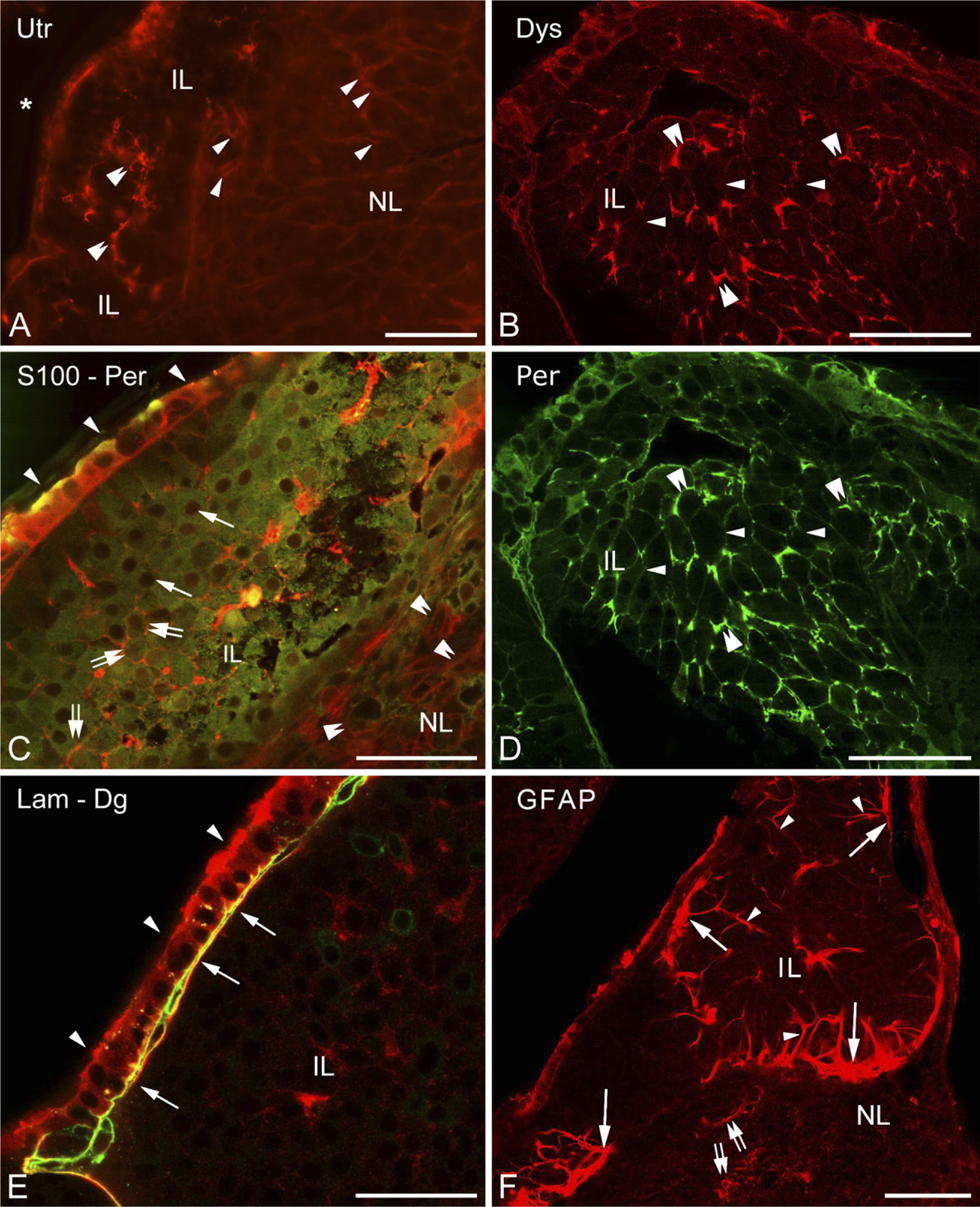
Higher magnification revealed the above-mentioned faintly immunoreactive contours of a loose mesh between the cells of the intermediate lobe not only in the case of β-dystroglycan (Figure 4C) but also following immunostaining to utrophin, dystrophins, perlecan (Figures 5A, 5B, and 5D), and even to laminin (Figure 5E). When double staining was applied, patterns of immunostaining of the components of the dystrophin–dystroglycan complex (here, e.g., dystrophin) and those of the basal lamina (here, perlecan) seemed to be identical (Figures 5B and 5D). When a cellular marker, the S100 protein, was colabeled with perlecan, the latter was found at the borders of the cells (Figure 5C). In the intermediate lobe GFAP (Figure 5F) and another astroglia marker, glutamine synthetase (not shown in Figure 5), visualized only a few, long, radially oriented fibers which formed endfeet on the surface of the cell groups.
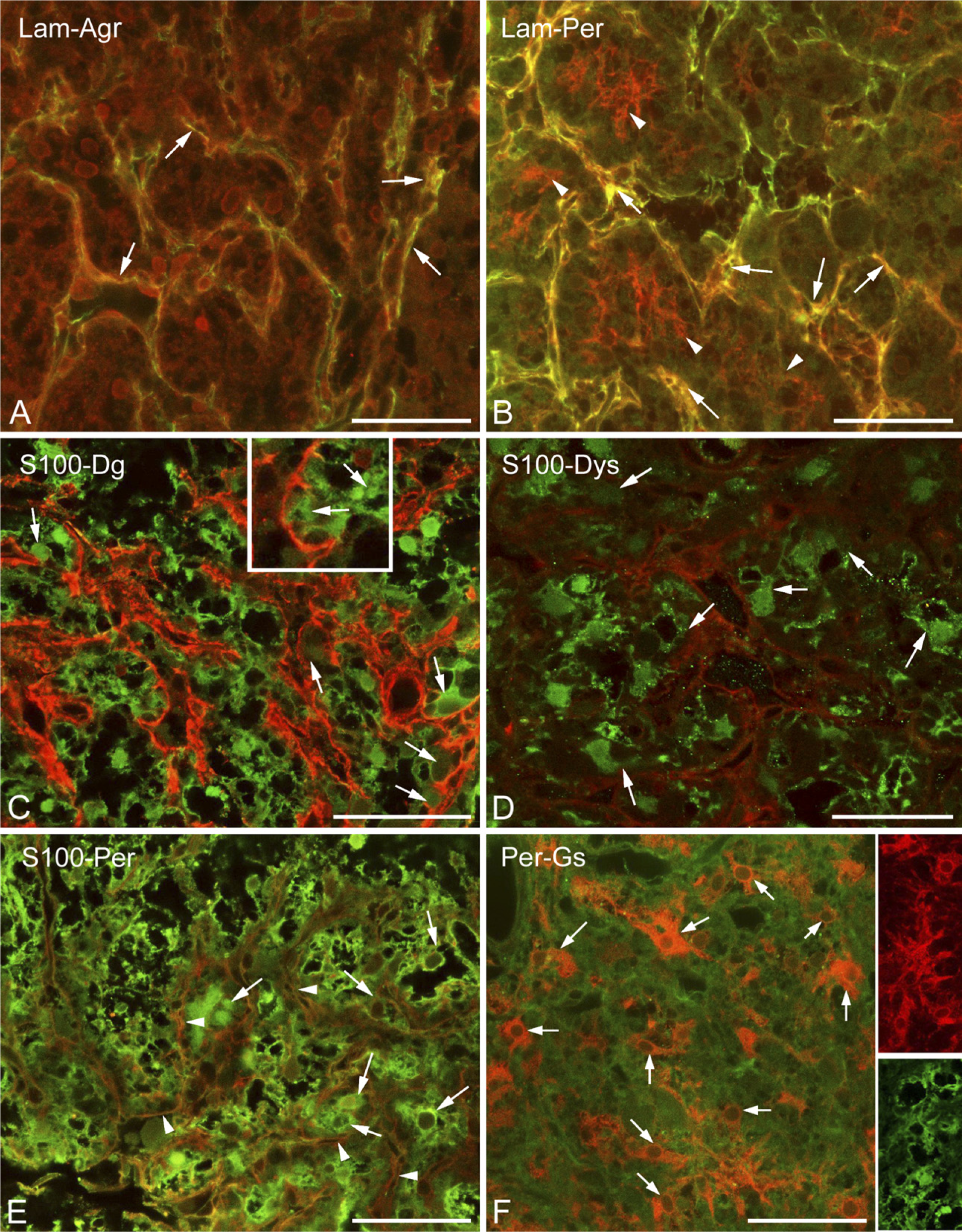
Basal lamina and dystroglycan-dystrophin complex in the neurohypophysis; relationship to cells.
Cuboidal Epithelium of the Rathke's Pouch Remnant
The cuboidal epithelium lining the remnant of Rathke's pouch displayed immunoreactivity against several substances investigated. Besides its immunopositivity to α1-dystrobrevin and α1-syntrophin, it proved to be immunopositive to the S100, perlecan, and β-dystroglycan (at least in the cell contours) proteins (Figures 5C–5E), as well as to glutamine synthetase and aquaporin-4 (see below). This cell layer was separated from the underlying cell groups of the intermediate lobe by basal lamina (Figure 5E).
Distribution of Aquaporin-4 Relative to That of GFAP
In the neurohypophysis, aquaporin-4 was localized along the periphery but in uneven thickness. Whereas, corresponding to the meningeal surface, only a thin rim was formed by aquaporin-4-immunoreactive cells, a thick zone proved to be immunoreactive where the neurohypophysis contacted the intermediate lobe (Figure 6A, inset). GFAP occurred in large but rather scarce astrocytes, which were found mainly at the periphery. When double labeling was applied, aquaporin-4 was found to localize only partially in GFAP-immunopositive astrocytes (Figures 6B and 6C). Some colocalization of aquaporin-4 with β-dystroglycan and glutamine synthetase was also found (Figures 6D and 6E). Aquaporin-9 was not found either here or in the intermediate lobe. The GFAP-immunopositive fibers and their endfeet-like extensions which were found within and around the cell groups of the intermediate lobe (Figure 5F) also proved to be immunoreactive to aquaporin-4 (Figure 6F). Aquaporin-4 also occurred in the epithelium lining the remnant of Rathke's pouch (Figure 6A, see inset, and Figures 6F and 6G). Within its cells, glutamine synthetase was detected, whereas at their bases, GFAP-immunoreactive cell processes were observed.
Discussion
System of Basal Lamina: Invaginations for Vessels Into the Neurohypophysis
In both parts of the neurointermediate lobe the parenchyma was surrounded by layers immunoreactive to laminin and β-dystroglycan, which suggested basal laminae connected to the laminin receptor dystroglycan. The polyclonal antiserum (raised against laminin-1) (Sigma) could detect any type of laminin containing either an α1 or β1 or γ1 chain [i.e., laminin-1 to −12, except for 5 (Colognato and Yurchenko 2000; Libby et al. 2000)], since laminin-1 consists of α1, β1, and γ1 chains. In the basal lamina, agrin and perlecan colocalized with laminin. In the neurohypophysis, the immunostaining of the basal lamina delineated a labyrinth of invaginations from the meningeal surface. In these invaginations, vessels were visualized by immunohistochemical reaction to utrophin [occurs in brain vessels (Khurana et al. 1992; Haenggi et al. 2004; Haenggi and Fritschy 2006)]. In this way, extrinsic vessels penetrate the organ but remain separate from its parenchyma, as electron microscopy observations have also suggested (Tweedle and Hatton 1987).
In cerebral vessels, laminin proved to be immunoreactive only in the vessels’ segments entering at the brain surface (in the Virchow–Robin spaces), in circumventricular organs with a leaky blood–brain barrier, in immature brain, or near lesions (Shigematsu et al. 1989; Zhou 1990; Krum et al. 1991; Sixt et al. 2001; Szabó and Kálmán 2004,2008). In these cases, the two basal laminae, the glial (parenchymal) and the vascular, did not fuse completely together, in contrast to the majority of the mature, intact cerebral vessels (Bär and Wolff 1972; Marin-Padilla 1985; Hallmann et al. 2005). Therefore, the perivascular laminin immunoreactivity of the neurohypophysis seems to be in accordance with its leaky blood–brain barrier (Ermisch et al. 1985) and perivascular spaces (Tweedle and Hatton 1987). Reports of nervous varicosities that extend over the meningeal basal lamina to contact vessels have been published (Tweedle and Hatton 1987), but to visualize such varicosities remained beyond the limits of our study.
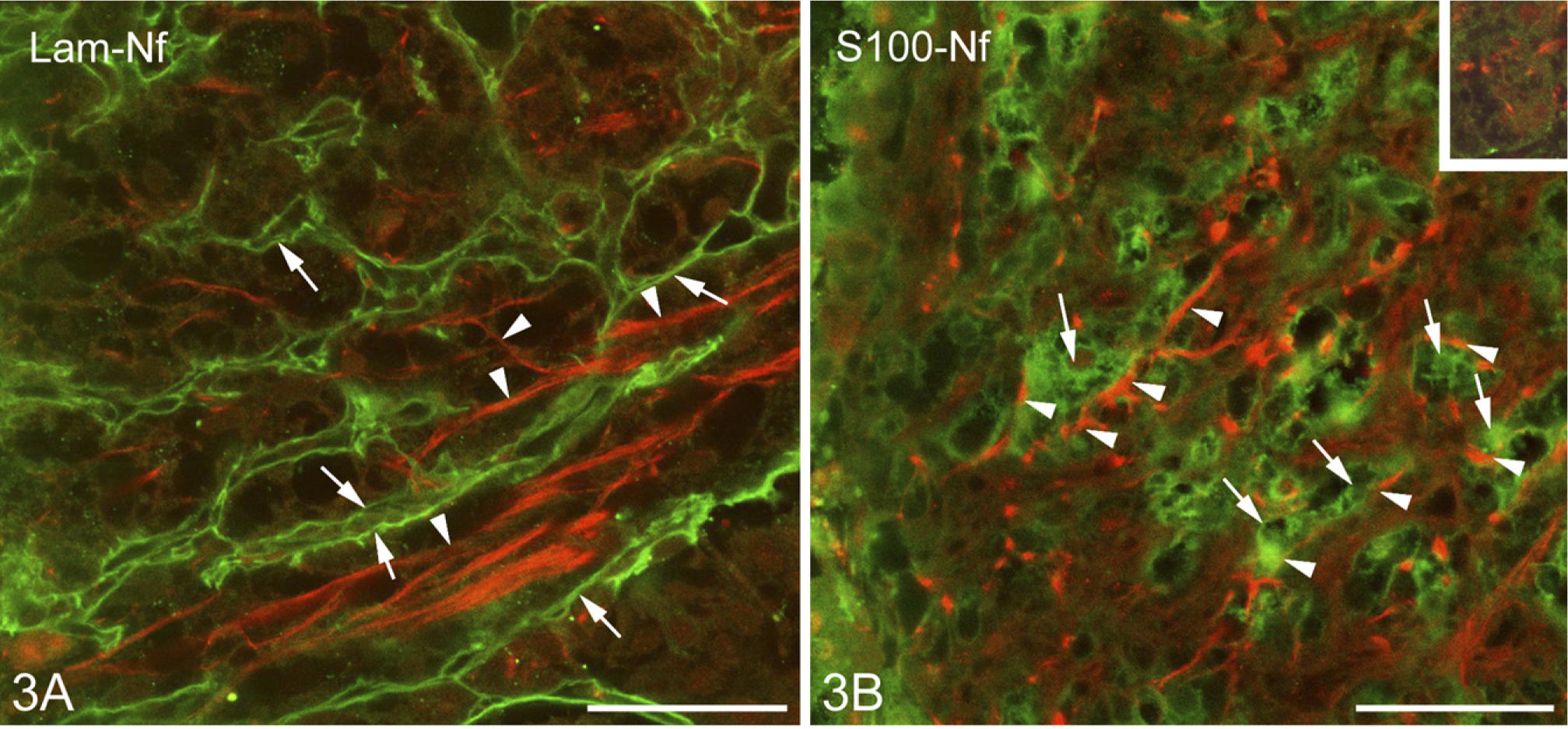
Relationship of axons to the basal lamina components and cells.
Localization of dystrobrevins and syntrophin.
Differences From Cerebral Vessels: Neural Lobe Vessels Are Actually Neither Hypophyseal nor Cerebral
The occurrence of utrophin in brain vessels has been previously published (Khurana et al. 1992; Haenggi et al. 2004; Haenggi and Fritschy 2006). However, whereas we found only utrophin in vessels of the neurohypophysis, β-dystroglycan, dystrophin, α1-dystrobrevin, and α1-syntrophin can also be detected along the cerebral vessels in the perivascular glial endfeet (Khurana et al. 1992; Uchino et al. 1994; Tian et al. 1996; Uchino et al. 1996; Blake et al. 1998, 1999; Jancsik and Hajós, 1999; Ueda et al. 2000; Zaccaria et al. 2001; Haenggi et al. 2004; Milner et al. 2008). Even the β-dystroglycan protein, although it delineates the cerebral vessels, is found in perivascular glial endfeet but not in the vessels themselves (Tian et al. 1996; Hallmann et al. 2005; Haenggi and Fritschy 2006; Milner et al. 2008; Wolburg et al. 2009). This observation is in accordance with our finding in the neurohypophysis, where the β-dystroglycan occurred on the parenchymal side of the basal lamina. In the neurohypophysis, β-dystroglycan did not delineate vessels, in contrast to the immunostaining of utrophin. Immunostaining of α1-dystrobrevin also delineates vessels in the brain (Blake et al. 1998; Ueda et al. 2000) as well as immunostaining of β-dystroglycan does, and α1-syntrophin was also detected around them (Vajda et al. 2002; Bragg et al. 2006). However, α1-dystrobrevin as well as α1-syntrophin were completely missing from the neurohypophysis, although they were detectable in cells of the intermediate lobe. The immunostaining of aquaporin-4, which is also concentrated in the cerebral perivascular glial endfeet, did visualize vessels throughout the brain but not in the neurohypophysis (see the detailed discussion about aquaporins below). These differences are attributed to the fact that “neurohypophyseal” vessels are actually out of the parenchyma (they are not really “hypophyseal”) but remain within the intrusions of the surrounding meningeal tissue and have no gliovascular connections which are found in the cerebral vessels (so they are not “cerebral”). It is to be noted that our investigations did not reveal any vessel-like structure in the intermediate lobe, in accordance with other findings (Murray et al. 1997).
Different Distributions of Dystrophin and Utrophin
Previous studies of the neurohypophysis detected utrophin and β-dystroglycan as well as different dystrophin isoforms, the major isoform Dp71, Dp140, and the full-length Dp427, by immunoblotting methods (Dorbani-Mamine et al. 1998). These isoforms have also been found in brain tissue (Lidov 1996). The anti-dystrophin antibody (Dys2; Novocastra Laboratories Ltd, Newcastle upon Tyne, UK) we applied recognizes C termini, which is uniform in these isoforms (Jancsik and Hajós 1999), i.e., it reacts with either of them. Our study demonstrated for the first time the localization of the β-dystroglycan, utrophin, and dystrophin proteins in the intermediate lobe. Whereas here the occurrences of these substances were similar [i.e., between the cells (see discussion below)], in the neurohypophysis, dystrophin was found around the cell groups of the neurohypophysis, whereas utrophin was found in the vessels. The differences in occurrences of dystrophin and utrophin are characteristic of brain tissue: dystrophin and utrophin have the same actin-binding position in the dystrophin–dystroglycan complex; therefore, they are alternatives of each other. Dystrophin (mainly the Dp71 isoform) occurs in the astrocytes and in the neurons (mainly the Dp427 isoform), whereas endothelium contains utrophin (Khurana et al. 1992; Knuesel et al. 2000; Haenggi et al. 2004; Haenggi and Fritschy 2006; Wolburg et al. 2009).
Further investigations in the intermediate lobe show basal lamina and dystroglycan–dystrophin complex and their relationship to cells.
α1-Dystrobrevin and α1-Syntrophin Occur in the Intermediate Lobe and β-Dystrobrevin in the Neurohypophysis
A fascinating result is the intense immunoreactivity of α1-dystrobrevin and α1-syntrophin in the cells of the intermediate lobe, where, however, their usual anchoring proteins utrophin or dystrophin were confined to the contours of the cells. Lien et al. (2004) detected α1-dystrobrevin in isolated cells of the embryonic Rathke's pouch but only transiently during the first 9 to 11 embryonic days. In the neurohypophysis, dystrobrevin was represented by its β form, which was found previously in neurons by Blake et al. (1998). Note that Górecki et al. (1997) declared that the expression of syntrophins correlates with the expression of dystrophins and dystroglycans. In neurons, the β-dystrobrevin and α1-syntrophin proteins colocalize with dystrophin (Blake et al. 1998, 1999). It must be mentioned, however, that α1-dystrobrevin can be integrated into the sarcolemma in the absence of dystrophin (Crawford et al. 2000). The dystrophin–dystroglycan complex without syntrophin has been found in the brain (Moukhles and Carbonetto 2001), as has syntrophin without dystroglycan in muscles (Cote et al. 2002). In the neurohypophysis, the presence of other syntrophins [β1, β2, γ1, and γ2 (Tinsley et al. 1994; Górecki et al. 1997; Piluso et al. 2000)] has not yet been ruled out; actually, the presence of β2-syntrophin has been indicated by its mRNA (Górecki et al. 1997). Since the different components of the dystrophin–dystroglycan complex have different tasks, it is possible that in some localizations, they occur alone, free of the complex, having special functions. Within the dystrophin–dystroglycan complex, they are supposed to participate in signaling processes (Grady et al. 1999; Culligan and Ohlendieck 2002). In neurons, dystrobrevin may occur without colocalization with utrophin or dystrophin (Hazai et al. 2008), and the authors, together with others (Chen et al. 2008), have suggested its role in neuroendocrine mechanisms. The role of α1-syntrophin is further discussed with that of aquaporins next.
Distribution of Aquaporin Is Not Coincident With Either α1-Syntrophin or Vessels
Along the cerebral vessels in the perivascular glial endfeet, α1-syntrophin anchors the aquaporin-4 molecules to the dystrophin–dystroglycan complex and, thereby, to the cell membrane (Neely et al. 2001; Inoue et al. 2002; Amiry-Moghaddam et al. 2003, 2004; Nico et al. 2003, 2004; Warth et al. 2004, 2005). Laminin and agrin components of the basal lamina also have a role in this arrangement (Guadagno and Moukhles 2004; Warth et al. 2004), since the dystroglycan binds to them (i.e., they also bind the dystrophin–dystroglycan complex). Therefore, it was an unexpected observation that in the neurointermediate lobe, the distribution of aquaporin-4 did not coincide with that of α1-syntrophin. In contrast to brain tissue (Nielsen et al. 1997; Goren et al. 2006), in the neurohypophysis, aquaporin-4 immunopositivity was not found along the vessels, adjacent to the basal lamina, which contained laminin and agrin. An aquaporin-4-immunopositive zone marked the peripheral border of the neurohypophysis, but the major part of the immunoreactivity was found where the neural lobe attached to the intermediate lobe. Although only incompletely colocalized with GFAP and to an even lesser extent with glutamine synthetase, aquaporin-4 immunopositivity visualized astrocyte-like elements. Colocalization with S100 was not investigated because the host animal of the primary antibodies was rabbit at both anti-aquaporin-4 and anti-S100 protein antibodies. It should be noted that in the neurohypophysis, Kuwahara et al. (2007) also found aquaporin-4 in the pituicytes, whereas aquaporin-1 was found at the vessels and aquaporin-5 in the cells of Rathke's pouch, where we detected aquaporin-4.
The unexpected distribution of aquaporin-4 can be attributed to the absence of α1-syntrophin here, because it is responsible for the concentration of aquaporin-4 in the astrocyte endfeet, so in its absence, aquaporin-4 dissipates along the whole cell (Amiry-Moghaddam et al. 2003, 2004; Warth et al. 2004, 2005). The physiological role of the unusual aquaporin-4 localization, however, remains unknown. Note that Warth et al. (2004) and Nicchia et al. (2008) found an α1-syntrophin-independent aquaporin-4 fraction in the cerebellum and in the ependyma.
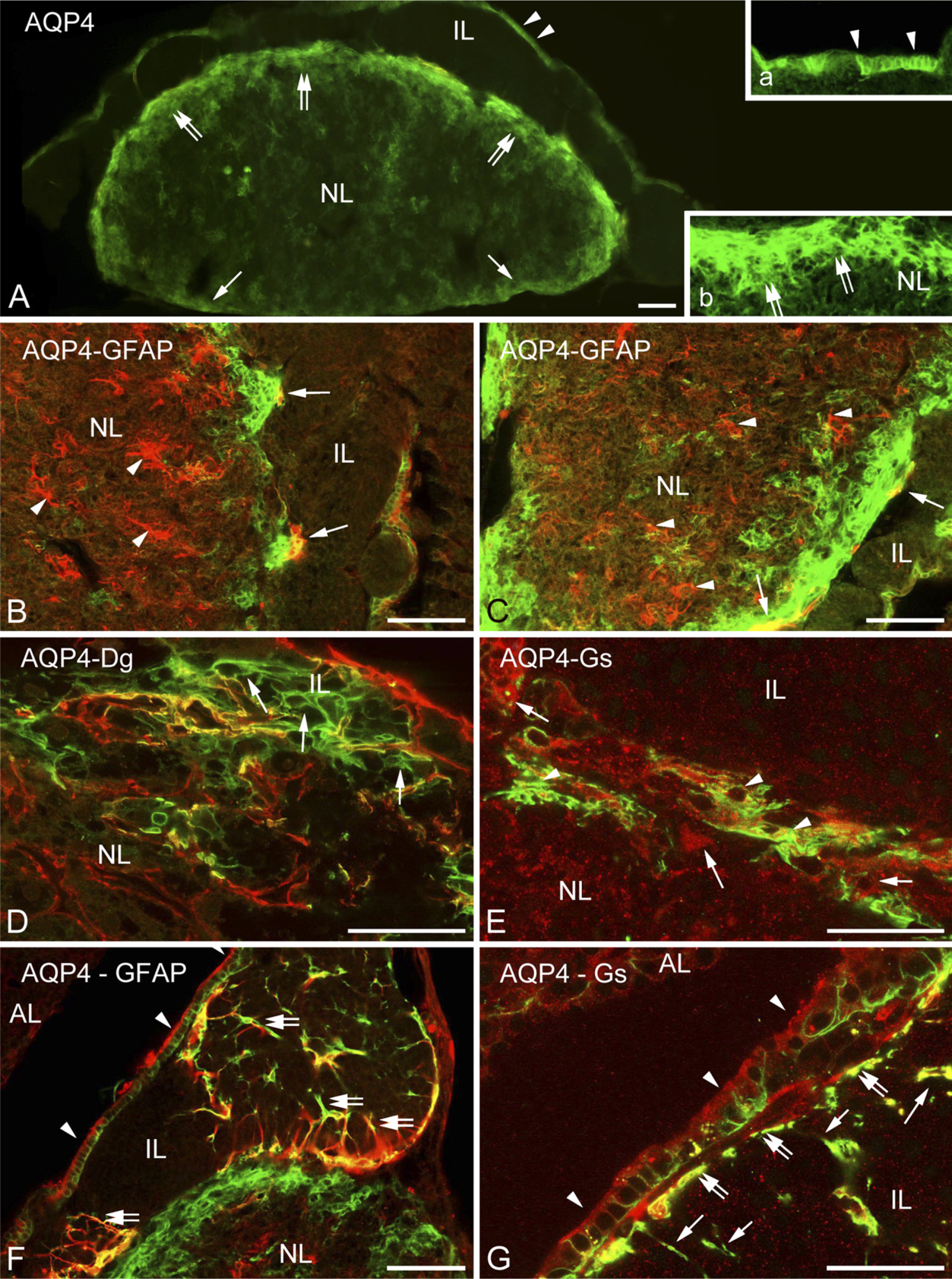
Aquaporin and glial structures.
Glial Markers in the Neurohypophysis
In accordance with findings of previous studies, cellular elements of the neural lobe proved to be immunopositive to astroglial markers, GFAP (Salm et al. 1982; Velasco et al. 1982; Marin et al. 1989; Redecker and Morgenroth 1989), S100 (Hofler et al. 1984; Lauriola et al. 1984; Marin et al. 1989), and glutamine synthetase (Shirasawa and Yamanouchi 1999). According to our results, however, GFAP and glutamine synthetase seemed to be localized in a lesser part of cells than S100 was. Cell shapes delineated by immunostainings to GFAP, S100, and glutamine synthetase were quite different. Differences may be due to the different intra-cellular localizations, but we suppose that the GFAP-immunoreactive cells may correspond to the classically termed pituicytes, which were found to be GFAP immunoreactive (Suess and Pilska 1981).
Pericellular Pattern in the Intermediate Lobe: Epitheloid Cells With Glial Cover?
In the intermediate lobe, S100 immunoreactivity has been described in glia-like cells which spread long, slender processes among the epithelial cells (Sands et al. 1995; Chronwall et al. 2000). Some authors suggest that these cells belong to the folliculostellate cells for which the S100 protein is the best marker (Cocchia and Miani 1980; Nakajima et al. 1980; Allaerts and Vankelecom 2005). GFAP-immunopositive stellate cells have also been mentioned in this area (Stoeckel et al. 1981; Gary and Chronwall 1995; Gary et al. 1995). Berardi et al. (1999) even found laminin immunoreactivity covering the melanotrope cells of the intermediate lobe. Our observations may modify this picture, since S100 occurred in the epithelium-like cells in general, whereas the slender processes between them proved to be immunoreactive to GFAP and aquaporin-4-like astroglial cells of the brain. Their courses were perpendicular to the dystroglycan- and laminin-covered surface of the cell groups, and their endfeet-like formations around these groups resembled the glia limitans of the brain. Inside the intermediate lobe, immunostaining of β-dystroglycan, utrophin, dystrophin, and perlecan was seen only faintly, if at all, as loose mesh delineating cell contours. This suggests the presence of a thin intercellular matrix, or at least a remnant of it, which separated the cells from each other as it happens to epitheloid cells.
Remnant of Rathke's Pouch
The cuboidal epithelium lining the remnant of Rathke's pouch is supposed to be a homolog of ependyma (Correr and Motta, 1985) or even a germinative layer of the intermediate lobe (Carbajo et al. 1992). The wide-ranging spectrum of immunoreactivities supports these proposals. For similar results see Marin et al. (1989) and Ogawa et al. (1990); the latter group also detected keratin in addition to the GFAP and S100 proteins, i.e., glial and epithelial markers occur together, uniquely. Beneath the cuboidal epithelium, we found basal lamina and aquaporin-4-immunoreactive glia, separating the epithelium from the cell groups of the intermediate lobe, which had their own basal lamina around.
In conclusion, the results demonstrate the separation of the vessels from the neurohypophyseal parenchyma and their different immunostaining patterns from those of the cerebral vessels: the lack of α1-syntrophin and α1-dystrobrevin and a different distribution of aquaporin-4. Vessels in the neurohypophysis are neither really hypophyseal nor cerebral, rather they are meningeal vessels. The cell groups of the intermediate lobe seem to be (a) epitheloid, since they are separated by remnants of basal lamina, and (b) surrounded by a glia limitans. The results support the possibility of the separate localizations of the components of the dystrophin–dystroglycan complex and raise questions about the roles of α1-dystrobrevin, α1-syntrophin, and the separately localized aquaporin-4 in the functions of the intermediate and neural lobes, respectively.
Footnotes
Acknowledgements
This study was supported by Hungarian Scientific Research Fund 60930 (to MK).
