Abstract
The aim of this study was to localize perlecan in human fetal spine tissues. Human fetal spines (12–20 weeks; n=6) were fixed in either Histochoice or 10% neutral buffered formalin, routinely processed, paraffin-embedded, and 4-μm sagittal sections were cut and stained with toluidine blue, H&E, and von Kossa. Perlecan, types I, II, IV, and X collagen, CD-31, aggrecan core protein, and native and Δ-HS 4, 5 hexuronate stub epitopes were immunolocalized. Toluidine blue staining visualized the cartilaginous vertebral body (VB) rudiments and annular lamellae encompassing the nucleus pulposus (NP). Von Kossa staining identified the VB primary center of ossification. Immunolocalization of type IV collagen, CD-31, and perlecan delineated small blood vessels in the outer annulus fibrosus (AF) and large canals deep within the VBs. Perlecan and type X collagen were also prominently expressed by the hypertrophic vertebral growth plate chondrocytes. Aggrecan was extracellularly distributed in the intervertebral disk (IVD) with intense staining in the posterior AF. Notochordal tissue stained strongly for aggrecan but negatively for perlecan and types I and II collagen. Type I collagen was prominent in the outer AF and less abundant in the NP, while type II collagen was localized throughout the IVD and VB. The immunolocalization patterns observed indicated key roles for perlecan in vasculogenic, chondrogenic, and endochondral ossification processes associated with spinal development.
Keywords
P
The human perlecan gene encodes a large core protein of 467 kD, which consists of five discrete domains (I–V) with homology to the laminin A-chain, low-density lipoprotein receptor, neural cell adhesion molecule, and epidermal growth factor (Noonan et al. 1991; Murdoch et al. 1992,1994). Domain I is the main acceptor for glycosaminoglycan (GAG) substitution in human perlecan and contains a cluster of three potential attachment sites. Domain V has also been shown to be a potential GAG acceptor region in mouse perlecan (Friedrich et al. 1999; Tapanadechopone et al. 1999). Perlecan interacts with a range of basement membrane and ECM components including nidogen/entactin, type IV collagen, fibronectin, fibulin-2, laminin, PRELP, type XIII collagen, and heparin (Friedrich et al. 1999; Hopf et al. 2001; Iozzo and San Antonio 2001; Bengtsson et al. 2002; Tu et al. 2002). Heparan sulfate substitution in domain I of perlecan promotes binding to laminin I, fibronectin, and type IV collagen. This is important not only in developmental processes involving cellular attachment but also in the structural organization of the developing cartilaginous extracellular matrix (Iozzo et al. 1994; Friedrich et al. 1999; Tapanadechopone et al. 1999; Hopf et al. 2001; Iozzo and San Antonio 2001). The HS side chains of perlecan can also bind and store growth factors such as fibroblast growth factor (FGF)-1, −2, −4, and −9 and can act as low-affinity co-receptors, underscoring its importance in growth and development (Handler et al. 1997; Arikawa–Hirasawa et al. 1999; Costell et al. 1999; French et al. 1999; Klint and Claesson–Welsh 1999; Chang et al. 2000; Clayton et al. 2001; Iozzo 2001, Iozzo and San Antonio 2001; Knox et al. 2001,2002). FGF-7 has also been shown to bind avidly to domain III of the perlecan core protein, and this appears to regulate the activity of this growth factor (Mongiat et al. 2000).
Perlecan has previously been demonstrated in a range of embryonic and newborn mouse and rat tissues including the spine and intervertebral disc (Handler et al. 1997; Arikawa–Hirasawa et al. 1999, 2001a, b,2002; Costell et al. 1999; French et al. 1999; Hayes et al. 2001). Perlecan has been immunolocalized in basement membrane-rich human tissues such as liver and kidney (Murdoch et al. 1994), in the intima of balloon catheter-injured porcine carotid arteries (Nugent et al. 2000), and in intimal atherosclerotic lesions in apolipoprotein E-deficient and low-density lipoprotein receptor-deficient mice (Kunjathoor et al. 2002), in the infarct zone in a mouse myocardial infarction model (Nakahama et al. 2000), and in the follicular basal lamina of bovine reproductive tissues (McArthur et al. 2000).
Perlecan was first localized in cartilage and chondrosarcoma by Iozzo et al. (1994) and was later demonstrated as an important matrix component of bovine nasal cartilage (SundarRaj et al. 1995). Handler et al. (1997) subsequently demonstrated perlecan in murine disk and Hayes et al. (2001) in rat disk tissues. More recently, Melrose et al. (2002) localized perlecan in the vertebral growth plate and cartilaginous end plates of the ovine intervertebral disk. Surprisingly, despite its obvious importance to developmental processes, perlecan has yet to be immunolocalized in human spinal tissues. The present study was therefore initiated to immunolocalize perlecan in human fetal spinal tissues using a recently characterized monoclonal antibody to perlecan domain I, MAb A76 (White-lock et al. 1996, 1999; Melrose et al. 2002). The perlecan localizations were compared and contrasted with the distribution of other matrix components including types I, II, IV, and X collagen, as well as aggrecan, to assess the role of perlecan in the structural organization of the rapidly developing cartilaginous primordia of the human fetal spinal column.
Materials and Methods
All materials and suppliers were as noted previously (Melrose et al. 2002). Details of the antibodies used are provided in Table 1. Histochoice fixative was obtained from Amresco (Solon, OH). MAbs 10-E-4, 3-G-10 (David et al. 1992), and heparitinase were obtained from Seikegaku (Tokyo, Japan). Anti-aggrecan G1 domain (MAb AD11–2A9) was from Bioscience Europe (Nivelles, Belgium). Anti-collagen type I (clone I8H5) and type II (clone II-4CII) antibodies were purchased from ICN (N. Ryde, NSW, Australia); anti type-IV collagen antibody (clone CIV-22), and anti CD-31 (PECAM, platelet endothelial cell adhesion molecule, clone JC/70A) were purchased from DAKO Australia (Botany, NSW, Australia). The anti-type X collagen and perlecan domain 1 antibodies have been described elsewhere (Gibson et al. 1996; Whitelock et al. 1996,1999; Melrose et al. 2002).
Preparation of Human Fetal Tissues for Histology and Histological Procedures
Six 12–20-week-old human fetuses were obtained at termination of pregnancy after ethical approval by The Human Care and Ethics Review Board of The Royal North Shore Hospital. The posterior elements were dissected from the fetal spines and they were longitudinally bisected in a mid-vertical plane. Half of the spines were fixed in Histochoice for 24 hr and the remaining specimens were fixed in 10% (v/v) neutral buffered formalin for 24 hr. Selected specimens were also briefly decalcified in 10% formic acid for up to 24 hr. The tissues were dehydrated in graded alcohols, embedded in Paraplast, and 4-μm sections were cut and mounted on StarfrostPlus glass microscope slides (Menzel-Glaser; Heidelburg, Germany). The tissue sections were then de-par-affinized in xylene before rehydration through graded ethanol washes to water.
Details of the monoclonal antibodies, fixatives, and epitope retrieval procedures used in this study
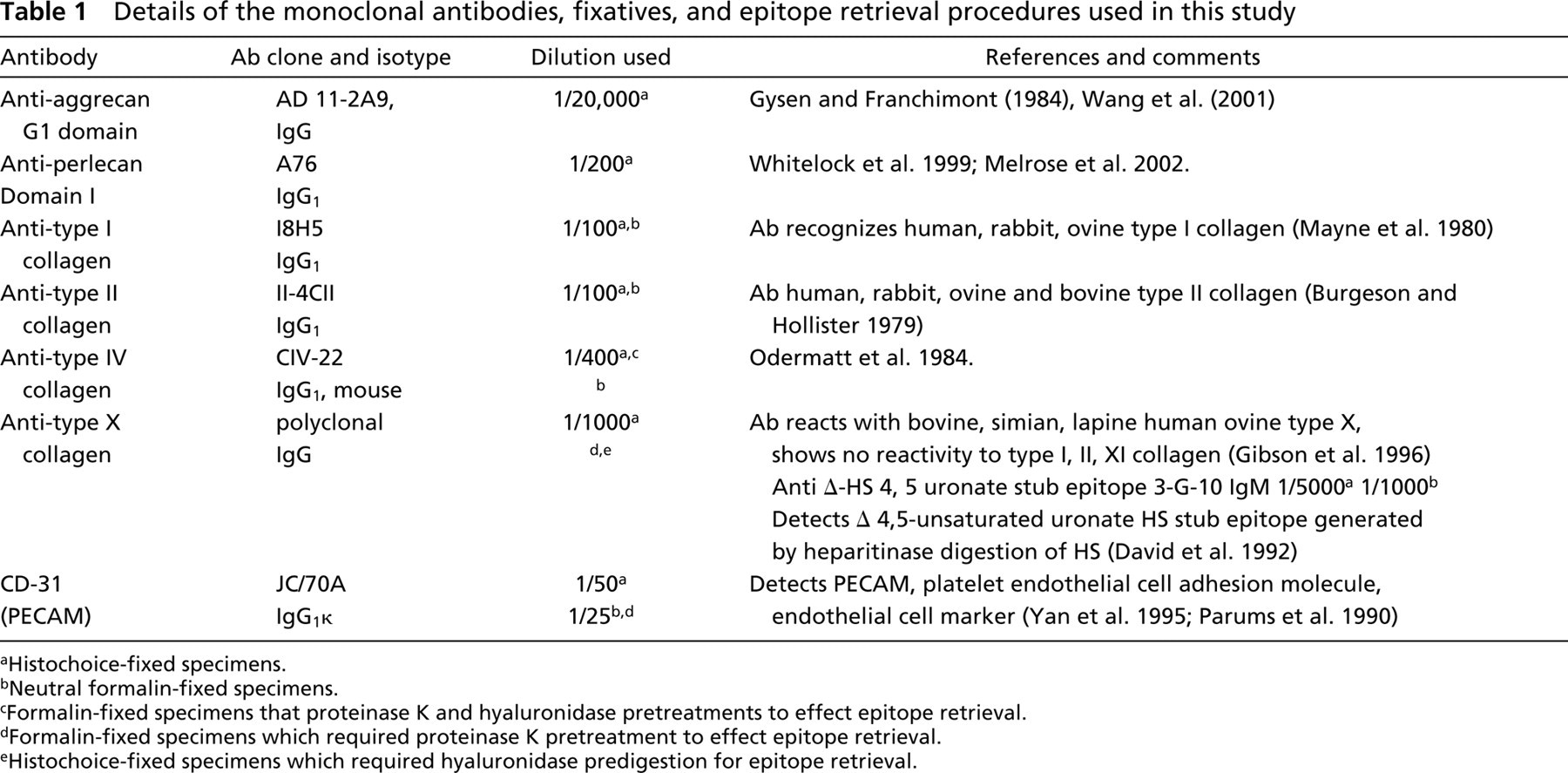
Histochoice-fixed specimens.
Neutral formalin-fixed specimens.
Formalin-fixed specimens that proteinase K and hyaluronidase pretreatments to effect epitope retrieval.
Formalin-fixed specimens which required proteinase K pretreatment to effect epitope retrieval.
Histochoice-fixed specimens which required hyaluronidase predigestion for epitope retrieval.
Histochemistry
Neutral formalin-fixed tissue sections were routinely stained for 10 min with 0.04% (w/v) toluidine blue in 0.1 M sodium acetate buffer, pH 4.0, to visualize the tissue proteoglycans. This was followed by 2-min counterstaining in an aqueous 0.1% (w/v) fast green FCF stain which aids in the differentiation of the areas stained for proteoglycan. Hematoxylin and eosin-stained tissue sections were used to examine cell morphology. Mineral deposition in the vertebral center of ossification was visualized in non-decalcified mid-sagittal sections of human fetal spinal tissues using a further modification developed in house at the Department of Anatomical Pathology, Royal North Shore Hospital (NSW, Australia) of the modification of the von Kossa procedure (von Kossa 1901) described by Drury and Wallington (1967). Tissue sections were briefly stained with silver nitrate (1.5% w/v) in the dark for 90 sec to 2 min. The sections were then incubated with hydroquinone (1% w/v) for 1–2 min and finally with sodium thiosulfate (5% w/v) for 5 min. The sections were then counterstained with H&E and mounted in Eukitt.
Immunohistochemistry
The immunolocalizations were undertaken using a Sequenza vertical cover-plate immunostaining system (Immunon) as described earlier (Melrose et al. 2002). Details of the antibodies used in this study and of the epitope retrieval steps required by specific antibodies and fixation conditions are provided in Table 1. Endogenous peroxidase activity was initially blocked by incubating the tissue sections with 3% H2O2 for 5 min. For the perlecan and aggrecan immunolocalizations, Histochoice-fixed specimens were used. The tissue sections were pre-digested with bovine testicular hyaluronidase 500 U/ml for 1 hr at 37C in phosphate buffer, pH 5.0, followed by three washes in 50 mM Tris-HCl buffer, pH 7.6, containing 0.15 M NaCl and 0.05% Tween-20 (TBS-T). For MAb 3-G-10, a 2-hr digestion with heparitinase (2.5 mU/ml) in 50 mM HEPES buffer, pH 7.0, containing 100 mM NaCl, 3 mM CaCl2, and 1 mg/ml bovine serum albumin was used to generate the Δ-4,5 uronate HS stub epitopes identified by this antibody. MAb 10-E-4, in contrast, identifies native HS chains, and heparitinase digestion destroys 10-E-4 reactivity (David et al. 1992). Some of the antibodies used on formalin-fixed tissue in this study also required enzymatic pre-digestion steps to effect epitope retrieval (see Table 1 for details). This included proteinase K digestion for 6 min at room temperature (RT) using a proprietary proteinase K reagent (DAKO cat # S03020) according to the manufacturer' instructions. This was followed in some cases by a hyaluronidase digestion step using the conditions outlined above but employing a hyaluronidase concentration of 1000 U/ml (see Table 1 for details). Histochoice-and neutral formalin-fixed tissue specimens were both suitable for the immunolocalization of types I, II, IV, and X collagen, native HS, Δ-HS stub epitope, and CD-31. After the pre-digestion procedures the sections were rinsed twice in TBS-T and blocked with 10% swine serum for 10 min at RT before addition of primary antibody. Incubation with primary antibodies was performed overnight at 4C using the dilutions specified in Table 1. Appropriate biotinylated secondary antibodies and horseradish peroxidase-conjugated streptavidin were used for visualization with Nova Red substrate for color development. Control sections were also prepared in which an irrelevant isotype-matched primary antibody was substituted for the primary antibody of interest. Commercial (DAKO) isotype-matched mouse IgG (DAKO code X931) or IgM (DAKO code X942) control antibodies (as appropriate) were used for this step. The DAKO products X931 and X942 are mouse monoclonal IgG1 (clone DAK-GO1) and monoclonal IgM (clone DAK-GO8) antibodies directed against Aspergillus niger glucose oxidase, an enzyme that is neither present nor inducible in mammalian tissues. In the case of the 3-G-10 antibody, the heparitinase digestion step was omitted in the control sample, and for the 10-E-4 antibody the control sample was treated with heparitinase.
Results
Immunolocalization of aggrecan, perlecan, and types I and II collagen in longitudinal mid-sagittal sections of human fetal spinal tissues showed some interesting differences in the spatial distributions of these matrix components (Figures 1A–1H). Toluidine blue and von Kossa staining clearly showed the cartilaginous nature of the primordial vertebral bodies and the extent of mineralization in the ossifying center at the 12–20-week stage of spinal development (Figures 1A and 2C). Type I collagen was confined mainly to the intervertebral disk, with diminished staining evident in the NP (Figure 1B). Strong staining of the anterior and posterior longitudinal ligaments was also observed but no staining of the cartilaginous vertebral body rudiment (Figure 1B). This contrasted with type II collagen, which was more extensively distributed throughout the cartilaginous vertebral body rudiment and intervertebral disk (Figure 1C). However, the outer AF, together with the anterior and posterior longitudinal ligaments, showed no significant staining for type II collagen (Figure 1C). Aggrecan was detected primarily in the intervertebral disk and very little staining was evident for this proteoglycan in the cartilaginous vertebral rudiment (Figure 1D). Staining for aggrecan was generally stronger in the posterior AF. Some cell-associated aggrecan staining in the hypertrophic vertebral growth plate chondrocytes was also evident (Figure 1D). Further examination of the outer AF confirmed the spatial localization patterns for the aforementioned matrix components (Figures 1E–1H). Heparan sulfate was diffusely localized throughout the outer AF and displayed somewhat higher levels evident at the cranial and caudal margins of the AF, where it merged with the vertebral growth plates. The extreme periphery of the outer AF lamellae stained negatively for type I collagen (Figure 1F). However, the outer AF stained positively for type I collagen. Type II collagen staining, in contrast, was negative throughout the outer AF except at its margins, where it merged with the vertebral growth plate. Type II collagen staining intensity increased steadily in moving through the inner AF towards the NP (Figure 1G). Aggrecan was again strongly localized in the outer AF tissues examined (Figure 1G).
Perlecan was produced abundantly by the hypertrophic chondrocytes of the vertebral growth plate (Figure 2A). The most terminally differentiated hypertrophic chondrocytes of the vertebral growth plate located adjacent to the vertebral center of ossification also strongly expressed type X collagen, confirming their differential state (Figure 2B). On closer inspection of the vertebral growth plate, a pericellular localization of HS and perlecan around small columns of hypertrophic vertebral growth plate chondrocytes was apparent, although their distributions were not identical (Figures 2D and 2E). Perlecan was also localized diffusely throughout the cartilaginous vertebral body rudiment and was associated pericellularly with intervertebral disk cells in the NP and in the inner and outer AF (Figure 1F). Perlecan displayed a similar distribution to that previously documented for type IIA collagen in developing intervertebral disk tissues (Zhu et al. 2001).
Type IV collagen was also extracellularly localized to the outer AF and to many of the basal laminae of blood vessels in the outermost annular lamellae (Figures 3A and 3C). The CD31, perlecan, and Δ-HS MAbs used in the present study also were localized to the blood vessels in the outer AF (Figures 3B, 3D and 3E). Small arterioles and venules were also discernable in many cases in the outer AF of these developing spinal tissues (Figures 3A–3E).
Cartilage canals in the cartilaginous vertebral body rudiments were also prominently featured in longitudinal parasagittal sections of the human fetal spines examined. Several basal lamina components were also immunolocalized to the lining tissues of these cartilage canals, including native and Δ-HS hexuronate stub epitope (Figures 4A and 4C), perlecan (Figure 4E), and type IV collagen (Figure 4F). Higher-power assessment of individual cartilage canals demonstrated the presence of endothelium-like cells on the basal lamina of the canal. These cells also stained positively with the MAb to CD-31 (Figure 4D). Occasional clumps of red blood cells were also observed entrapped within these cartilage canals, suggesting that these structures may have some important nutritional role in these rapidly developing cartilaginous primordial spinal tissues.
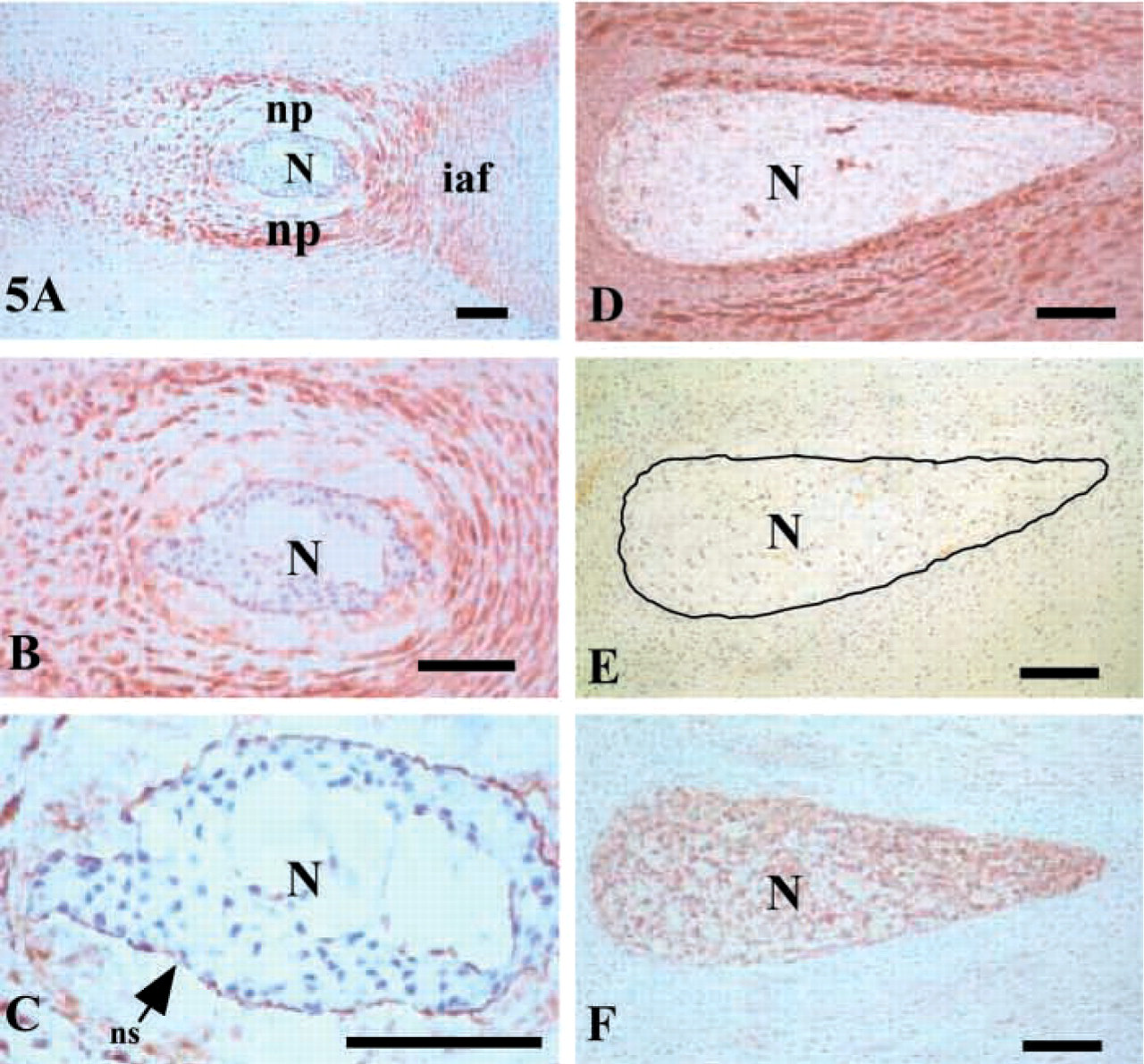
Further immunolocalization of perlecan, types I and II collagen, and aggrecan in parasagittal sections of additional 12–14-week-old human fetal spinal tissues confirmed that perlecan was a prominent component of the NP and inner AF. However, perlecan was not present in the notochordal tissue, but some staining of the notochordal sheath for perlecan was noted (Figures 5A–5C). Type II collagen was also localized to both the inner AF and NP but, like type I collagen, it was not identified in the notochordal tissue (Figures 5D and 5E). Interestingly, this same tissue stained strongly with the anti-aggrecan antibody (Figure 5 F).
Discussion
The formation of the primordial spinal cartilages that will act as developmental scaffolds and eventually become the ossified vertebral bodies is a complex process. Initially, this involves the migration of precursor cells from remote places to form mesenchymal cell aggregates. Sclerotome cells located lateral to the neural tube also migrate ventrally towards the notochord. This eventually leads to the formation of the primordial cartilage around the notochord, which will become the vertebral body (Götz et al. 1995; Christ et al. 2000; Fleming et al. 2001). This cell-migration process is guided by morphogens such as Sonic Hedgehog protein secreted by the notochord and by Pax-1, Pax-9, and Hox gene expression (Smith and Tuan 1994; Burke et al. 1995; Peters et al. 1999). Growth differentiation factor-11 (Gdf-11), also known as bone morphogenetic protein (BMP)-11 also determines the anterior–posterior patterning of the axial skeleton (McPherron et al. 1999). This is a complex process involving the interplay of core-binding transcription factor alpha-1 (Cbfa-1), other local BMPs and their receptors (Monsoro–Burq et al. 1996; Ducy and Karsenty 2000; Yamaguchi et al. 2000), and cartilage-derived morphogenetic proteins (Tsumaki et al. 1999). Sonic and Indian Hedgehog are also important bone-inductive factors that critically direct the assembly of the axial skeleton (Iwasaki et al. 1997; Nakamura et al. 1997; Yuasa et al. 2002). Perlecan is detectable as early as the two-cell blastocyst stage of embryonic development (Carson et al. 1993; Smith et al. 1997) and can bind FGF-1, −2, −4, −7, and −9 (Aviezer 1994; Klint and Claesson–Welsh 1999; Chang et al. 2000; Mongiat et al. 2000; Clayton et al. 2001; Knox et al. 2001,2002). The ability of perlecan to act as a low-affinity co-receptor for the FGFs and its involvement in subsequent cell signaling events indicate that it has a fundamental role in cell proliferation and differentiation events critical to early spinal development. HS proteoglycans have also recently been demonstrated to sequester noggin in the ECM of costal cartilage (Paine–Saunders et al. 2002). Therefore, perlecan may also have the potential to regulate the bioavailability of BMPs that regulate the development of the rudimentary cartilage of the spinal column. The widespread distribution of perlecan in the cartilaginous vertebral body rudiments observed in the present study supports the proposal that perlecan has an important regulatory role in these early developmental processes in the spine.
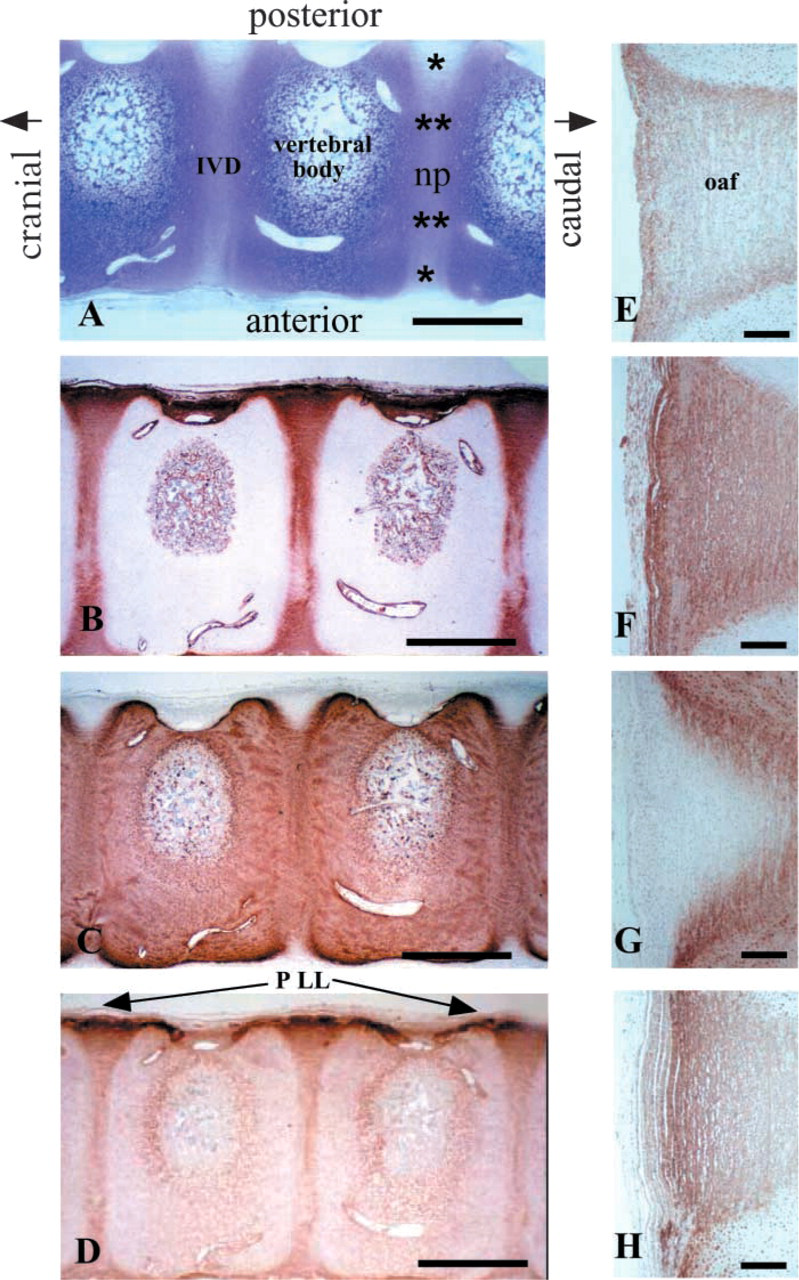
Immunohistochemical and histochemical localization of ECM components in longitudinal mid-sagittal sections of a 14-week-old human fetal spine. (
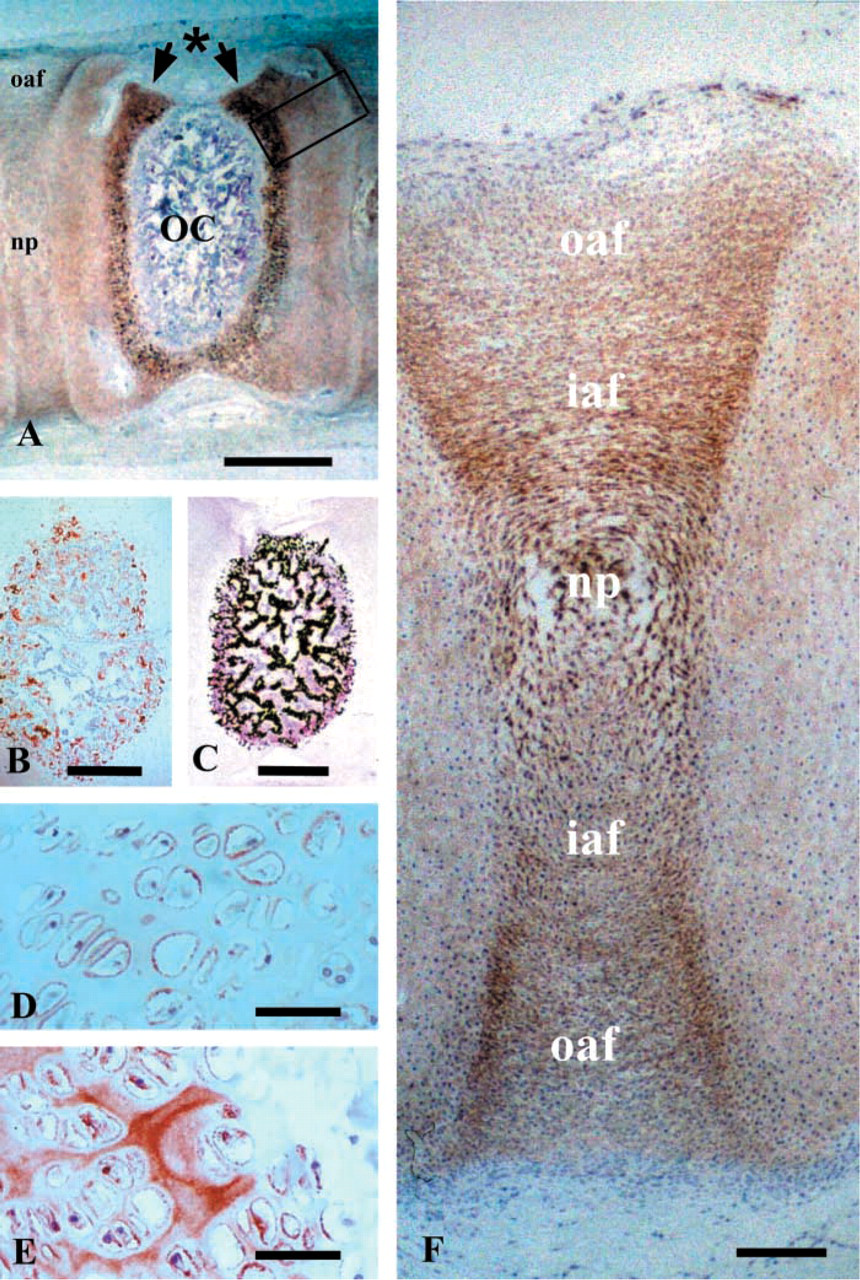
Immunolocalization and histochemical localization of intervertebral disk and vertebral components in 12–14-week-old human fetal spinal specimens. (
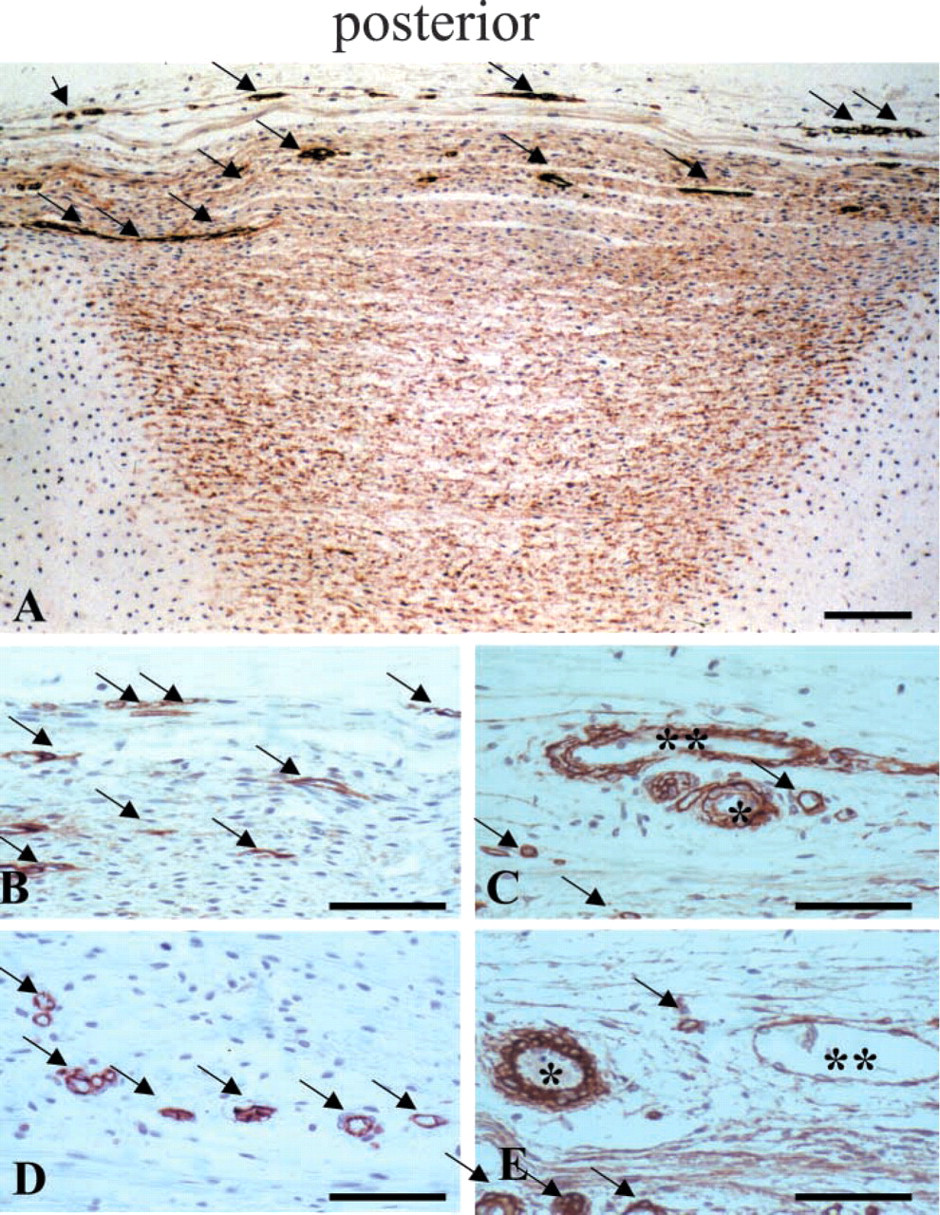
Immunolocalization of basal lamina components and endothelial cells associated with blood vessels in the outer AF (arrows) of an L4–L5 intervertebral disk from a 14-week human fetal spine. (
Handler et al. (1997) and Hayes et al. (2001) also examined perlecan expression in embryonic to newborn mouse and in rat spinal tissues and demonstrated its widespread distribution in the cartilaginous spinal primordia, suggesting potential roles for perlecan in chondrogenesis, endochondral ossification, and vasculogenic processes related to spinal development. The present study has extended these earlier observations in human tissues and has also compared the distribution of perlecan with other well-characterized ECM components such as types I, II, IV, and X collagen and aggrecan. It was somewhat surprising that aggrecan, the major proteoglycan of weight-bearing connective tissues (Knudson and Knudson 2001) and considered to be a primary chondrogenic phenotypic marker, was not as extensively distributed in the cartilaginous vertebral rudimentary spinal tissues as was perlecan. However, perlecan did display a similar pericellular distribution pattern in the present investigation to that previously documented for type IIA collagen in the developing intervertebral disk (Zhu et al. 2001). This may indicate that perlecan also has an important role in processes relating to spinal primordial cartilage remodeling, vertebral bone formation, and spinal development.
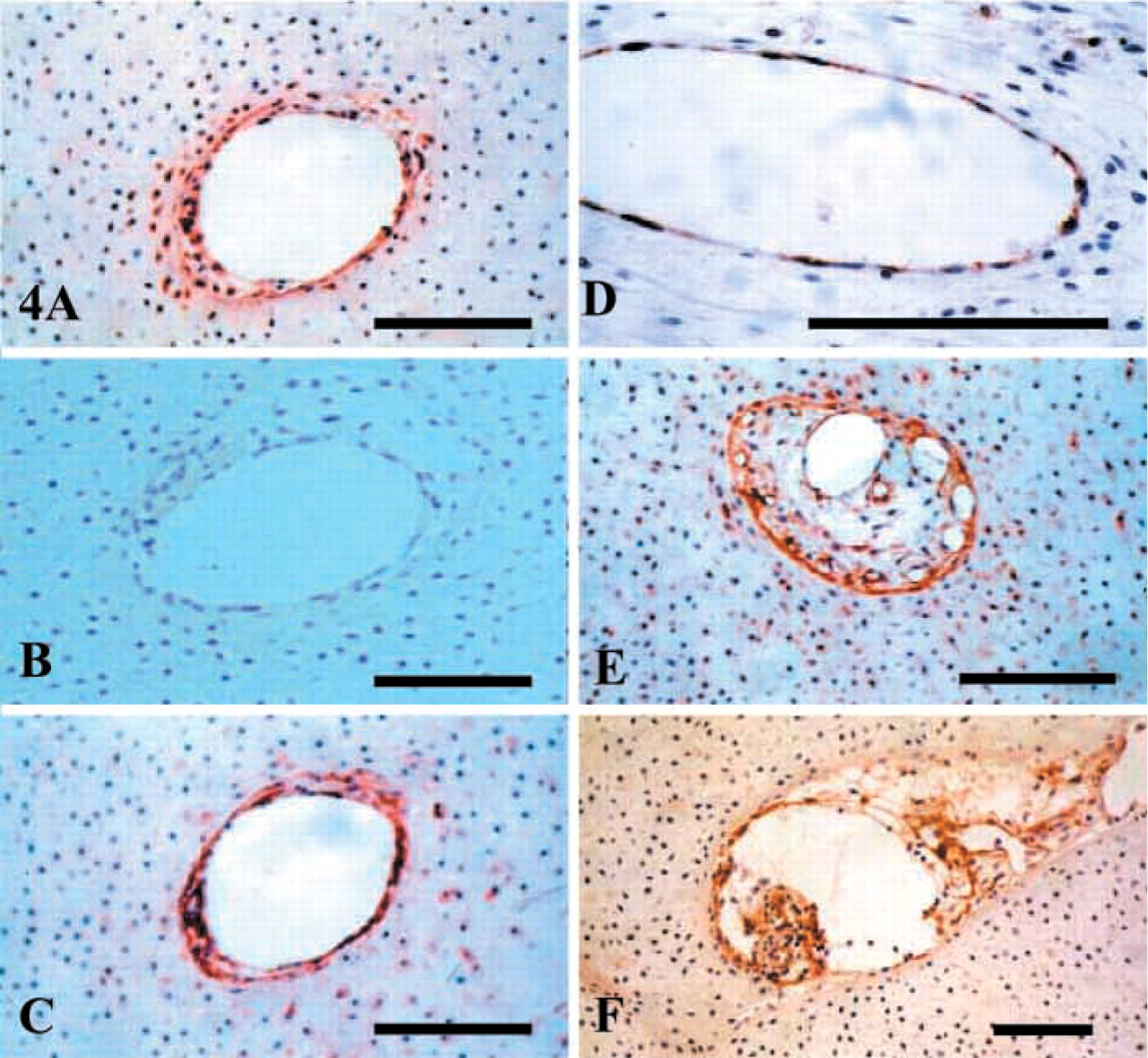
Composite figure depicting the presence of cartilage canals in the cartilaginous vertebral body rudiments in vertical parasagittal longitudinal sections of a 12-week-old human fetal spine. (
Immunolocalization of perlecan (
An earlier study from our laboratory also examined the distribution of perlecan and other major ECM components (types I, II, VI, and X collagen, aggrecan) in the newborn ovine intervertebral disk (Melrose et al. 2002). Perlecan was also strongly associated peri-cellularly with hypertrophic growth plate chondrocytes in the cartilaginous end plate and the vertebral growth plates of the newborn ovine disk (Melrose et al. 2002), which is consistent with its proposed role in the present investigation in spinal development. However, vascular channels and small blood vessels were less evident in the newborn ovine disk. Therefore, direct comparisons with the present study are not possible. Moreover, it should also be noted that the vertebral growth plate in the ovine spine (as in other quadrupeds) is dissimilar to its counterpart in humans. The newborn ovine vertebral growth plate is particularly well defined and is separated from the cartilaginous endplate (which demarcates the intervertebral disk proper) by a bony vertebral epiphysis. In humans the vertebral growth plate is contiguous with the cartilaginous endplates of the intervertebral disk. Furthermore, in humans the vertebral body is not a “short” long bone. It has a primary ossification center. The centrum and its polar endplates, the vertebral growth plates, act as modified growth zones in a similar manner to the polar growth plate cartilages in a carpal or tarsal bone. However, in contrast to a long-bone physis, the cartilage columns in the human vertebral growth plate in man are short and irregularly arranged. Despite these organizational differences between the newborn ovine and fetal spinal cartilages, the immunolocalization of perlecan to the hypertrophic vertebral growth plate chondrocytes in both studies is consistent with an important role for perle-can in cartilage development, terminal differentiation of growth plate chondrocytes, vertebral bone formation, and spinal development.
The vascularization of the human spine has been the subject of anatomic and microanatomic studies for almost a century, and most attention has focused on the external segmental blood supply and drainage of the vertebral bodies (Skawina et al. 1997, and references cited therein). Only a few articles have described the intraosseous arteries of the vertebral bodies in detail. Among the more recent studies, a comprehensive description of the extra- and intraosseous vessels of the vertebral bodies was presented by Crock and Yoshizawa (1976) and Ratcliffe (1982). Skawina et al. (1997) studied the vasculature of the human fetal (17–24-week) vertebral bodies using an acrylic dye injection technique, light microscopy, and corrosion casting methods that were evaluated by scanning electron microscopy. This permitted complete visualization of the vascular system and three-dimensional images of the vertebral microvascular architecture. Three vascularization zones were identified: (a) a peripheral zone containing perichondrial vessels, (b) an intermediate zone of radial vessels, and (c) a central zone of vessels in the ossification center. This is consistent with observations made in the present study. However, the same authors noted a complete absence of blood vessels in the intervertebral spaces, which is surprising considering the findings of the present study. Rudert and Tillmann (1993), however, used histological and immunohistological methods to demonstrate blood and lymph vessels in the adult human IVD. Therefore, their apparent absence in the Skawina et al. (1997) study may stem from technical limitations evident in the respective methodologies employed.
Skawina et al. (1997) also observed capillary cuffs, which they termed manchettes, surrounding pairs of larger vessels, and considered these as typical vascular arrangements in the developing vertebral cartilage canals. Such vascular arrangements were also observed in the present study (Figure 4). Widened and irregular segments of metaphyseal vessels directed towards the cartilaginous endplates from the ossification center of the intervertebral disk were also observed, which are also consistent with the arrangement of cartilage canals observed in the present study. Cartilage canals have also been observed in human fetal and rabbit vertebral bodies, where they are an important source of nutrition to the avascular disk regions during fetal development (Whalen et al. 1985). Such cartilage canals were also observed in spine specimens aged 12–20 weeks in the present investigation. This is therefore in agreement with observations made by Skawina et al. (1997), who noted that the metaphyseal vessels had started to penetrate the vertebral body cartilages by the second trimester but became incorporated into the ossification center only at 36 weeks of fetal life.
The present study has demonstrated perlecan as a prominent basal lamina component of small peripheral blood vessels in the outer AF and of cartilage canals within the vertebral body rudiment. Basal lamina components have also been immunolocalized to developing epiphyseal cartilage canals (Kai et al. 1992) and are considered to have important roles in the vascularization of developing limb bud cartilages (Rayan et al. 2000). Furthermore, not only do such vessels have important roles in the nutrition of the rapidly developing cartilaginous spinal tissues but they also have roles in the establishment of the primary centers of ossification in the vertebral bodies. The present study has therefore demonstrated potentially important regulatory roles for perlecan, not only in the initiation but also in the promotion and maintenance of chondrogenesis and osteogenesis, as well as early vasculogenic events, which are critical for normal spinal development.
Footnotes
Acknowledgements
Supported in part by the National Health and Medical Research Council of Australia (project grant 211266) and by a Seeding Grant from The Lincoln Centre for Research into Bone and Joint Disorders (Royal North Shore Private Hospital; St Leonards, NSW, Australia).
Dr G. Gibson (Henry Ford Hospital, Detroit, MI) is thanked for his kind gift of antibodies to type X collagen.
