Abstract
Peripheral lymphoid tissues act as important organs of immunological defense. Characteristic of their architecture is the rich reticular fiber meshwork composed of various extracellular matrix (ECM) molecules with which the stationary non-lymphatic cells stay in intimate contact and form channels through which the lymphatic cells travel. Here we studied the distribution of various laminin (Ln) chains and different types of collagens in human spleen, lymph node, and tonsil to clarify their chain-specific distribution. The most widely distributed proteins in all these organs were Ln chains α5, β1, γ1 and collagen types IV and XVIII, which were present in practically all compartments. Conversely, Ln α1, α2, α4, and type VII collagen showed a more restricted expression pattern. A unique feature was that Ln α3-, β3-, and γ2-chains, which normally are not localized to the vascular wall in non-lymphatic tissues, were present also in capillary basement membranes (BMs) of the follicular structures of lymph node and tonsil and in Ln α1-chain and type VII collagen also in the splenic white pulp. We also found that collagen XVII was exclusively present in the ring fibers of the spleen. The results indicate that BMs of lymphatic tissues contain a variety of macromolecules that probably contribute strongly to immunological events. In addition, capillaries of the lymphoid tissue exhibit a specified BM composition resembling that in epithelial BMs of non-lymphoid tissues.
P
Previous immunohistochemical studies have shown the abundant presence of the main extracellular matrix (ECM) components laminin (Ln), type IV collagen, vitronectin, fibronectin, and types I and III collagen in the RFs of spleen, lymph nodes, and tonsil (Liakka et al. 1991; van den Berg et al. 1993; Liakka 1994; Jaspars et al. 1996). During embryonal development, specific changes can be distinguished in the ECM composition (Liakka and Autio-Harmainen 1992), and many in vitro studies have shown that the ECM components may affect lymphocyte migration, adhesion, and proliferation (Li and Cheung 1992; Geberhiwot et al. 2001).
The most typical molecules of basement membranes (BM) are Lns and type IV collagen. They are connected to each other via nidogen-1, to which other BM components are joined as well (Colognato and Yurchenco 2000). We currently know of 11 different Ln chains, and there is evidence of at least 15 Ln heterotrimers and 6 α-chains of type IV collagen forming also several type IV collagen heterotrimers. Immunohistochemical (IHC) studies have shown that BMs may have heterogeneous compositions in different locations. In lymphoid tissues, especially, Ln and type IV collagen are abundant (Karttunen et al. 1989; Liakka et al. 1991; van den Berg et al. 1993). In some of these tissues the BMs are highly discontinuous in areas where the cell traffic is high, e.g., the venous sinuses of the splenic red pulp (RP) and the tonsillar crypt epithelium. In addition to serving as adhesive matrix for the stationary cells, the RFs and BMs rich in Ln and type IV collagen can contribute to the function of non-stationary cell types through signal transduction via integrin-type receptors (Hemler 1990; Ogata et al. 1996; Madri and Graesser 2000).
Many of the previous investigations have been performed by using polyclonal antibodies (PAbs) without knowledge of their chain specificity (Liakka et al. 1991; van den Berg et al. 1993). Now when chain-specific antibodies are available, a more detailed description of the expression of the individual BM components in lymphoid tissues is possible. In this study we used IHC to evaluate the distribution of Ln α1–5, β1–3, and γ1–2 chains as well as collagen types IV, VII, XVII (previous bullous pemphigoid antigen 180; BP-180), and XVIII in adult human spleen, lymph node, and tonsil. The results showed that these molecules have their own characteristic expression patterns, which indicate their specific contribution to the function of the organs.
Materials and Methods
Material
All the material was collected from surgical operations and was obtained from the files of the University Hospital of Oulu. The material comprised six spleens, five tonsils, and five lymph nodes. The tonsils were removed because of chronic tonsillitis and showed follicular hyperplasia. All the lymph nodes were collected from surgical resections of neoplastic disease (two mammary carcinomas, two gastric carcinomas, and one colon carcinoma), and they occasionally showed follicular hyperplasia but otherwise appeared normal and did not contain any neoplastic growth. One of the five spleens was removed after traumatic rupture and four spleens were removed during surgery for abdominal neoplasia (three gastric carcinomas and one colon carcinoma). These spleens contained no tumor deposits. All the material was immediately cooled in liquid nitrogen and stored at –80C until use.
Antibodies and Immunohistochemistry
Antibodies used are listed in Table 1. For IHC, 4-μm frozen sections were air-dried for 15 min, followed by fixation in cold acetone for 10 min, and then incubated with fetal calf serum (FCS) (Hyclone Laboratories; Logan, UT) 1:5 in PBS for 20 min to block nonspecific binding of IgG. The sections were then incubated with the primary antibody at 4C for 2 hr, followed by biotinylated anti-mouse or anti-rabbit IgG secondary antibody (DAKO; Glostrup, Denmark) for 30 min and avidin–peroxidase complex for 30 min. The color was developed with diaminobenzidine tetrahydrochloride–H2O2 (DAB) (Sigma; St Louis, MO) in Tris buffer, pH 7.4. Finally, the sections were lightly counterstained with hematoxylin. As a negative control, the sections were treated as described above but PBS was used instead of the primary antibody.
Antibodies used in the study
The IHC and immunofluorescence staining reactions were analyzed by two observers and the intensity of immunoreactivity was scored as: negative (–) when the staining reaction was indistinguishable from the background, weak (+) when the reaction was weak, and moderate to strong (+ +) when a clear and strong immunoreaction was observed.
Positive Controls
Frozen skin sections were used as positive controls. All antibodies, except for Ln α1-, α2-, and β2-chains, reacted with the epidermal BM. Collagen XVII showed a cell membrane-bound and partially intracytoplasmic immunoreactivity in basal keratinocytes. Ln β2-chain reacted with the endothelial BMs of dermal vascular structures and Ln α2 was present in the stroma of dermis. Because of the negative staining reaction for the α1-chain in skin sections, we used the BM of pharyngeal mucous glands as a positive control for the Ln α1-chain stainings.
Results
Results are summarized in Table 2. According to the IHC stainings, different BM components showed specific distributions in lymphoid tissues. In general, Ln α1-chain and types VII and XVII collagen had the most restricted distribution in all cases studied. Conversely, Ln α5, β1, γ1, and types IV and XVIII collagens were the most widely expressed proteins and were co-expressed in all the BM structures and RF meshwork studied. Interestingly, Ln α1-, α3-, β3-, and 72-chains and type VII collagen were present also in the vascular BMs of follicular structures. This expression pattern clearly differs from that in non-lymphoid tissues, where vascular BMs contain mainly Ln α5-, β1-, β2-, and γ1-chains and types IV (Määttä et al. 2001) and XVIII (Saarela et al. 1998) collagen. However, the RP of the spleen, the paracortical areas of the lymph node, and the interfollicular areas of the tonsil exhibited vascular BM composition similar to that in non-lymphoid tissues.
Distribution of laminin chains and types IV, VII, XVII, and XVIII in human spleen, lymph node, and tonsila
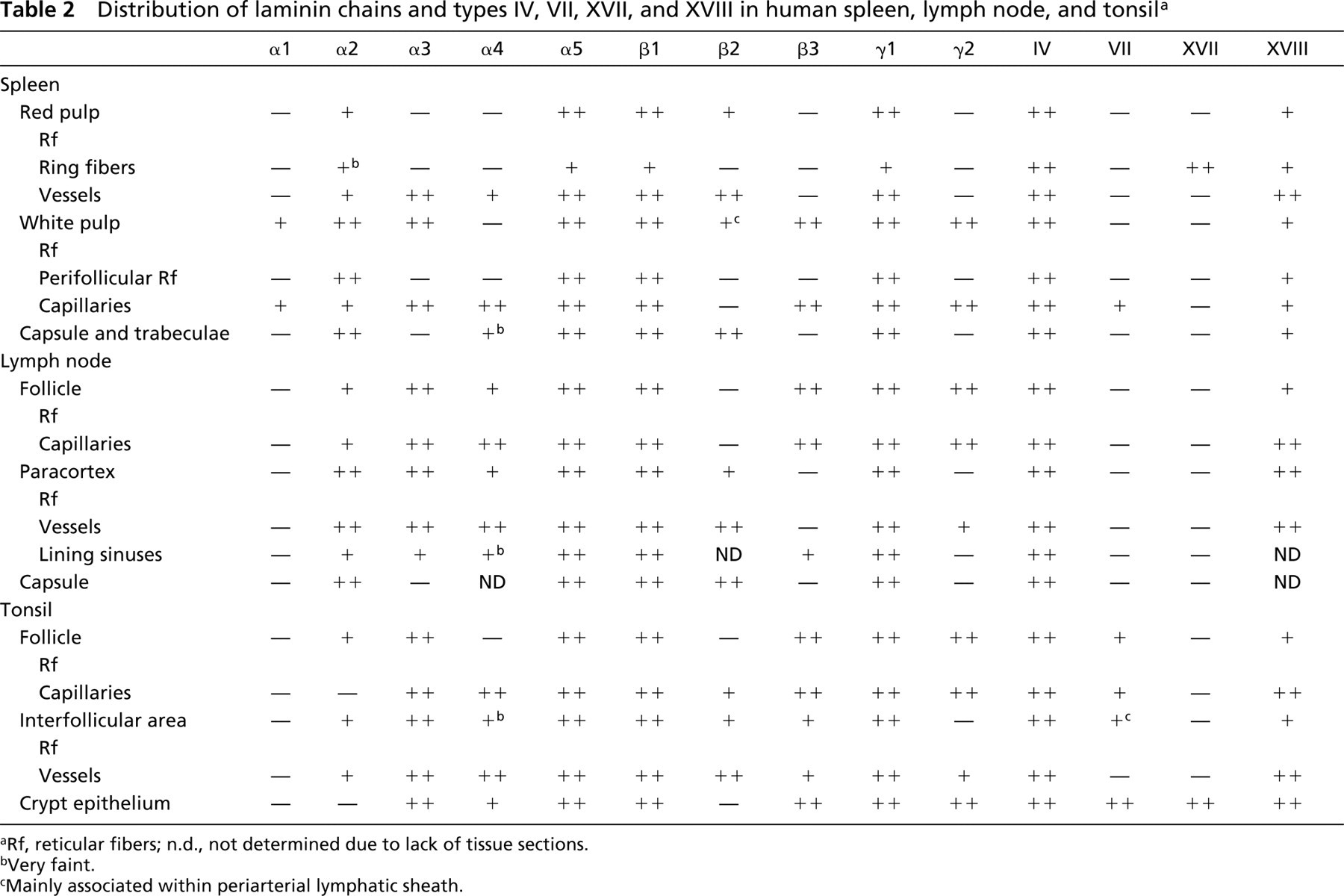
Rf, reticular fibers; n.d., not determined due to lack of tissue sections.
Very faint.
Mainly associated within periarterial lymphatic sheath.
Spleen
RFs of the white pulp (WP) immunoreacted for the Ln chains α1 (Figure 1A), α2, α3, α5, β1, β2, β3 (Figure 1B), γ1, and γ2 (Figure 1C), but the Ln α4-chain showed no immunoreactivity. The staining intensity was moderate or weak for Ln α1-, α2-, and β2-chains, whereas other chains showed strong and clear immunoreactivity. Fibers in the perifollicular area reacted only for Ln α2-, α5-, β1-, and γ1-chains. Capillaries in the WP immunoreacted for all the Ln chains investigated except for the β2-chain. However, vessels outside the WP showed immunoreactivity for Ln α2-, α3-, α4-, α5-, β1-, β2-, and γ1-chains. In the RP, the RFs and the ring fibers of the venous sinuses were stained for Ln α2, α5 (Figure 1D), β1, and γ1 (Figure 1E). Capsule and trabeculae showed immunoreactivity for Ln α2-, α5-, β1-, β2-, and γ1-chains.
The type IV collagen immunoreactivity was comparable to that of type XVIII. Their immunoreactivity was strongly present in all the compartments examined, including RFs and capillaries of the WP and RP, vascular subendothelial BM of the RP and trabeculae, ring fibers around the venous sinuses (Figure 1F), and the tissue of capsule and trabeculae.
Faint immunoreactivity for type VII collagen was seen in capillaries of the WP, but other structures were negative.
The type XVII collagen immunoreacted with the sinusoidal ring fibers of the spleen (Figure 1G) but other structures were negative. It was accumulated in the wall of the venous sinuses and appeared under the endothelial cells in a dot-like manner.
Lymph Node
The RFs of the lymphatic follicles immunoreacted for all the Ln chains except the α1- (Figure 2A) and β2-chains. Follicular capillaries showed a chain composition similar to that of follicular RFs (Figure 2B). In the paracortex, RFs and vascular BMs were stained for Ln α2-, α3- (Figure 2C), α4-, α5-, β1- (Figure 2D), β2-, and γ1- (Figure 2E) chains. In addition, a faint staining for Ln γ2 was seen in the paracortical vessels. Marginal sinuses immunoreacted for Ln α2-, α3-, α5-, β1-, β3-, and γ1-chains. Capsular tissue showed staining reaction for Ln α2-, α5-, β1-, β2-, and γ1-chains.
Types IV and XVIII (Figure 2F) collagen gave an identical staining pattern in all the compartments studied, including RFs of the follicular and paracortical area, BMs of blood vessels and marginal sinuses, and the tissue of capsule and trabeculae.
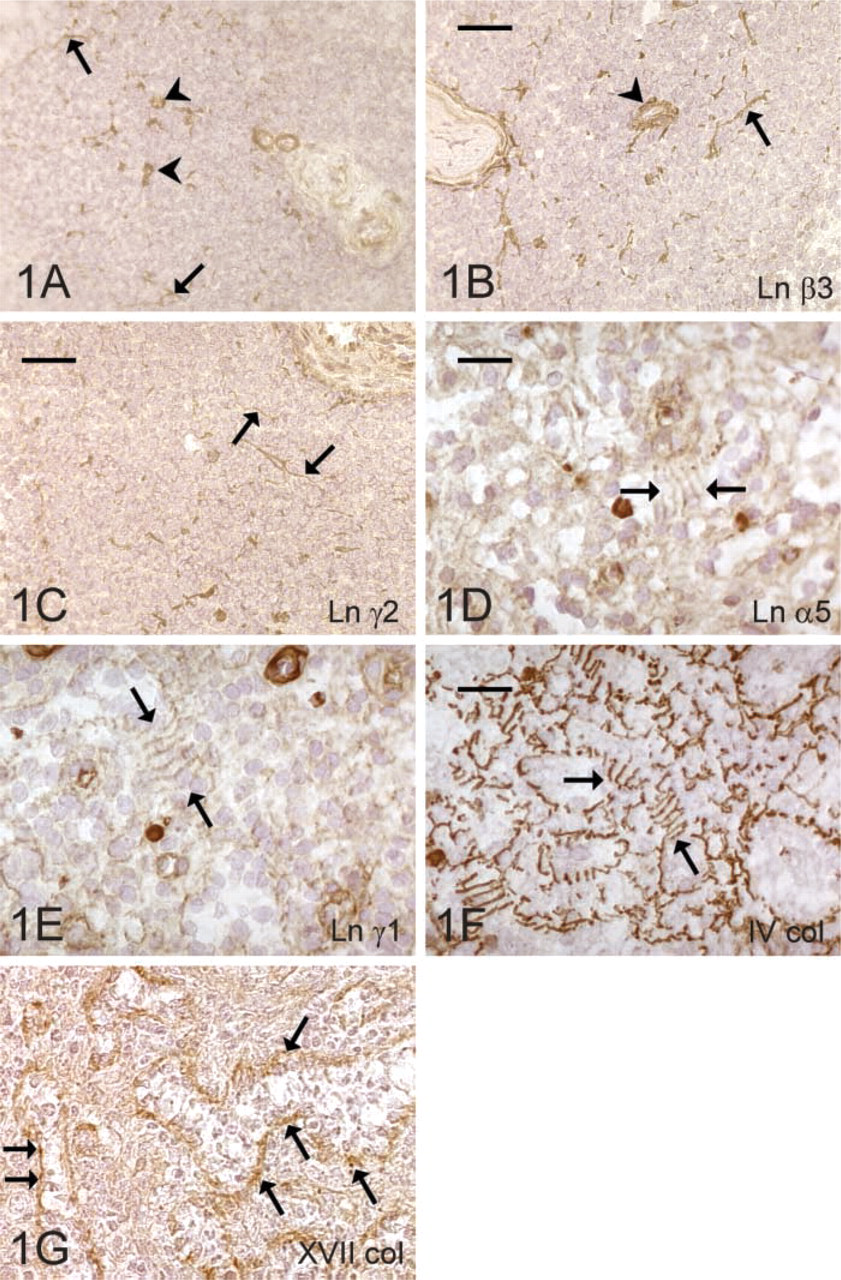
Expression of the ECM proteins in the spleen. Reticular fibers (arrow) and blood vessels (arrowhead) of the white pulp contain laminin α1- (
Types VII and XVII collagen were not detected in the lymph node.
Tonsil
The RFs of the tonsillar lymphatic follicles immunoreacted for Ln α2-, α3-, α5-, β1-, β3-, γ1-, and γ2-chains (Figure 3A). Follicular capillaries showed otherwise similar composition, but they additionally contained Ln α4- and β2-chain immunoreactivity, and the α2- chain staining was weak and sporadic. Interfollicular RFs immunoreacted with Ln α2-, α3- (Figure 3B), α5-, β1- (Figure 3C), β2-, β3-, and γ1-chains and occasionally also with α4-chain. Endothelial BMs of the interfollicular blood vessels (Figure 3D) contained all the other chains except for α1. The BMs of the surface and crypt epithelium showed a similar composition, containing Ln α3-, α4-, α5-, β1-, β3-, γ1-, and γ2-chains. BM staining of the crypt epithelium was, however, more disrupted and irregular deeper in the tonsillar bed.
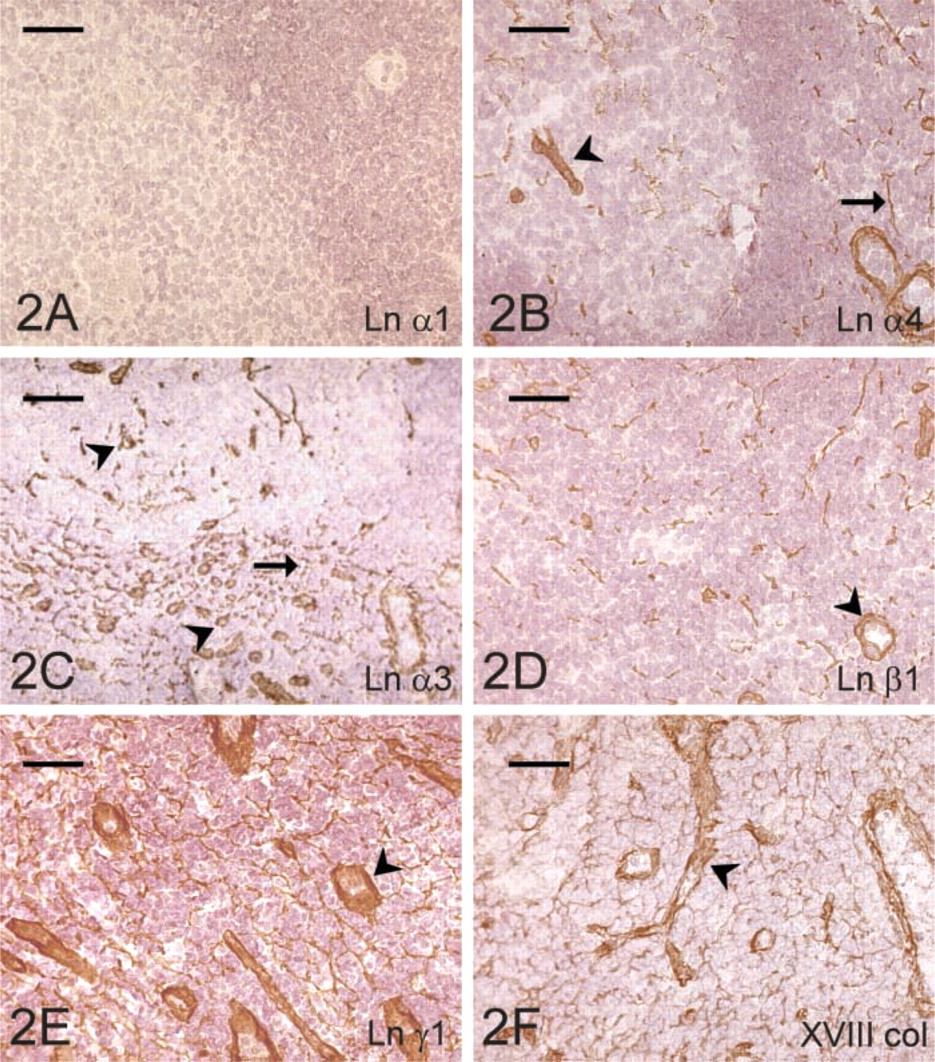
Expression of the ECM proteins in the lymph node. Lack of the laminin α1-chain (
Types IV (Figure 3E) and XVIII collagen showed identical immunoreactivity and stained all the BM compartments of the tonsillar tissue.
Type VII collagen antibody immunoreacted faintly with the follicular RFs and capillaries, but the staining reaction was much fainter and sporadic in the interfollicular area. BMs of the surface and crypt epithelia strongly immunoreacted for type VII collagen (Figure 3F), whereas other compartments were negative.
Immunoreactivity against type XVII collagen was observed in the cell membranes and cytoplasm of the basal cells of the surface and crypt epithelia (Figure 3G). An identical staining pattern was seen in the skin, which served as a positive control.
Discussion
The present study describes the IHC distribution of several Ln chains and the occurrence of types IV, VII, XVII, and XVIII collagen in lymphatic organs that included spleen, lymph node, and tonsil. According to the results, types IV and XVIII collagen were the most widely expressed collagenous proteins in the RFs and vascular BMs of lymphatic tissues. Ln isoforms showed more variable expression patterns and the most widely distributed chains, supporting our previous notion that tissues other than lymphoid (Määttä et al. 2001) were the Ln α5, β1, and γ1 which were present in all the compartments investigated. This indicates that Ln-10 (α5β1γ1) would be the most common laminin heterotrimer in the lymphatic tissues, and the relatively low level of Ln β2-chain gives indirect evidence that most of the α5-chains are bound to Ln-10 rather than Ln-11 (α5β2γ1) molecules. Ln-10 might represent some kind of general Ln skeleton with which other heterotrimers are associated. The heterogeneity of Ln chain composition in various tissue compartments of both lymphoid and non-lymphoid organs could simply reflect functional demands, as has been postulated in in vitro studies (Erickson and Couchman 2000; Quondamatteo 2002).
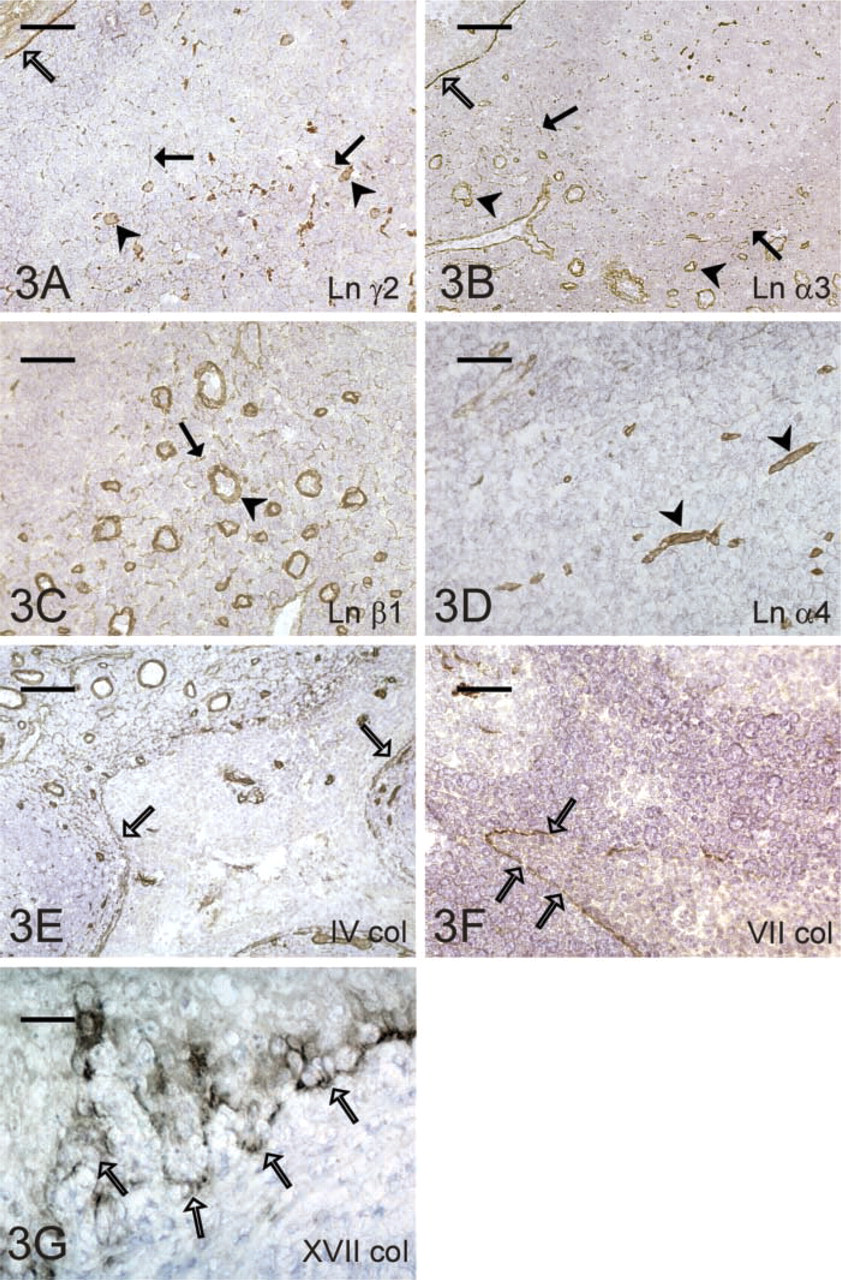
Expression of the ECM proteins in the tonsil. Crypt epithelium (open arrow), as well as blood vessels (arrowhead) and reticular fibers (arrow) of the lymphatic follicle, contain laminin γ2-chain (
Ln α1, a component of Ln-1 (α1β2γ1) and Ln-3 (α1β2γ1), seemed to be the most restricted chain. It was not detected in the tonsil and lymph node and was seen only in the RFs of the splenic WP and, unexpectedly, in the capillary BMs of the follicular structures of the spleen. Ln α1-chain has been shown to be restricted mainly to the subepithelial glandular BMs and to be absent from the endothelial BMs (Virtanen et al. 2000; Määttä et al. 2001). In addition to the presence of Ln α1-chain, another unique feature of the follicular capillaries was the additional presence of chains of Ln 5 (α3β3γ2) in their BMs. Ln 5 is a well-known component of BMs of squamous epithelia, in which it forms a part of the hemidesmosomal complex and usually has been immunolocalized to the epithelium (Marinkovich et al. 1992; Rousselle et al. 1997). However, it has been previously reported in capillaries of the human thymus and follicular capillaries of the lymph node and tonsil (Jaspars et al. 1996). This indicates that Ln 5 can rarely be detected in unusual locations, such as endothelial BMs, and the co-expression of Ln 1 and Ln 5 in the lymphatic tissues may also indicate a unique role for these Lns in the migration of lymphocytes into the follicle parenchyma.
One of the functions of ECM components is the modulation of leukocyte extravasation through the subendothelial Lns (Madri and Graesser 2000). It has been previously shown that especially Ln 8 (α4β1γ1) and Ln 10 have a strong binding capacity for monocytic cells (Pedraza et al. 2000). Moreover, lymphoid progenitor cells actively synthesize and adhere to Ln 8 and Ln 10 (Gu et al. 2003). Geberhiwot et al. (2001) have shown in an in vitro study that lymphoid cells are also able to synthesize Ln 8. Laminin molecules on the cell membranes of lymphocytes may also contribute to the cell-cell interactions between lymphocytes and antigen-presenting cells, e.g., dendritic reticular cells and macrophages, whereby Lns play a role in the immunological function of lymphatic organs. However, it must be remembered that because RFs and BMs of lymphatic tissues are covered by the cytoplasmic processes of fibroblastic reticular cells and the cytoplasm of endothelial cells (Chen and Weiss 1972), the lymphocytes traveling within the lymphatic tissues are usually not in direct contact with them.
Collagen types VII and XVII in the spleen and tonsil, as described in this study, have not been previously reported. They are both mainly expressed by the squamous epithelium and in agreement with this were both localized in this study to the tonsillar crypt epithelium, which is a direct extension of the surface squamous epithelium. Surprisingly, type VII collagen was also detected in the follicular capillaries and RFs of the tonsil and in capillaries of the splenic WP. All the evidence obtained from this study indicates that the capillaries in lymphoid tissues seem to be specified to possess a more “epithelium-like” BM composition than in other tissues. Of the hemidesmosomal components, the integrin α6β4 has also been detected in human lymph node and tonsil (Liakka 1994; Jaspars et al. 1996). As far as we know, however, it is not known whether there are true hemidesmosomes in those locations of lymphatic tissues where hemidesmosomal proteins are found, and this issue needs future investigation.
An interesting finding of this study was that type XVII collagen was specifically present in ring fibers of the spleen. This raises the question of its possible stabilizing role for the sinusoidal structures. Ring fibers are unique BMs of the venous sinuses that surround the endothelial cells like hoops around a barrel. Earlier electron microscopic and IHC studies (Chen and Weiss 1972; Drenckhahn and Wagner 1986) have shown that within the endothelial cells of the splenic venous sinuses there are basally located fine filamentous bands arranged longitudinal to the cell axis. They run from one ring fiber to the other, and appear to be inserted into the plasma membrane on the ring fiber and to connect the endothelial cells to the ring fiber. In fact, in this study the type XVII collagen staining reaction pattern of the ring fibers differs clearly from that of types IV and XVIII collagen and was seen as dots in the walls of the venous sinuses. We suggest that these dots represent the insertion site of the filamentous bands on the ring fiber matrix. In addition to our finding of type XVII collagen in the ring fibers, the integrin subunit α6 has been found previously in sinus endothelial cells (Liakka 1994; van den Berg et al. 1993). This subunit associates with the β4 subunit to form one of the components of epithelial hemidesmosomes. However, according to our results ring fibers lack Ln 5 and type VII collagen, which are normal constituents of epithelial hemidesmosomes. This indicates that the contact of the sinus endothelial cells with the ring fibers is not mediated via conventional hemidesmosomes. But because endothelial cells must resist marked shear forces caused by the fluid and blood cells that travel through the interendothelial slits from the red pulp cords into the sinuses, our findings suggest that this resistance is maintained through the formation of a firm hemidesmosome-like contact site and that the type XVII collagen and the integrin subunit α6 are involved in the connection between the ring fiber matrix and the intermediate filaments of the sinusoidal endothelial cells.
The type XVIII collagen, a member of the non-fibril-forming collagens, is a widely distributed BM component in many tissues (Halfter et al. 1998; Saarela et al. 1998; Erickson and Couchman 2000). A particular interest is concentrated on its 20-kD proteolytic C-terminal peptide fragment, called endostatin (O'Reilly et al. 1997). This fragment has a strong ability to inhibit endothelial cell activity, angiogenesis, and tumor growth. Thus far, type XVIII collagen distribution in lymphatic organs has not been published. We noted here that type XVIII collagen and type IV collagen showed a similar distribution pattern and were expressed in all the tissue compartments analyzed. The wide presence of type XVIII collagen/endostatin indicates its role not only as an important structural protein but also in controlling endothelial cell proliferation in the lymphatic organs.
In conclusion, our results demonstrate that BM-associated proteins show characteristic expression patterns in lymphatic tissues. The distributions of the various Ln chains, as well as types VII and XVII collagen, differ from those of BM proteins in non-lymphoid tissues and suggest a specific role for these proteins in the maintenance of tissue architecture and in the migration and immunological function of lymphocytes.
Footnotes
Acknowledgements
We thank Ms Heli Auno, Ms Annikki Huhtela, Ms Tuulikki Moilanen, Ms Riitta Vuento, and Mr Hannu Wäänänen for their excellent technical help.
Ln α1, α4, β1 and γ1 antibodies were a kind gift from professor Ismo Virtanen, University of Helsinki, Finland, and collagen type XVIII antibodies were provided by Prof Taina Pihlajaniemi, Universty of Oulu, Finland. The antibody to β2 Ln chain purified by Dr Dale Hunter and Dr Joshua Sanes was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, IA.
