Abstract
Heterotrimeric extracellular matrix proteins laminins are mostly deposited at basal membranes and are important in repair and neoplasia. Here, we localize laminin beta 2 (LAMB2) at the sites of blood–brain barrier (BBB). Microvasculature (MV) of normal brain is endowed with complete LAMB2 coverage. In contrast, its cognate protein laminin beta 1 (LAMB1) is absent in MV of normal brain but emerges at the sprouting tip of a growing vessels. Similarly, vascular proliferation in high-grade gliomas (HGG) is accompanied by marked overexpression of LAMB1, whereas LAMB2 shows deficient deposition. We find that many brain pathologies with presence of post-gadolinium enhancement (PGE) on magnetic resonance imaging (MRI) show disruption of LAMB2 vascular ensheathment. Inhibition of vascular endothelial growth factor signaling in HGG blocks angiogenesis, suppresses PGE in HGG, prevents expression of LAMB1, and restores LAMB2 vascular coverage. Analysis of single-cell RNA sequencing (scRNA-seq) databases shows that in quiescent brain LAMB2 is predominantly expressed by BBB-associated pericytes (PCs) and endothelial cells (ECs), whereas neither cell types produce LAMB1. In contrast, in HGG, both LAMB1 and 2 are overexpressed by endothelial precursor cells, a phenotypically unique immature group, specific to proliferating hyperplastic MV.
Introduction
Human brain consumes about 20% of glucose and oxygen. 1 Being enclosed by rigid calvarium and supplied by relatively thin caliber arteries, it has developed a set of mechanisms to precisely redistribute a limited quota of blood to the neuronal populations immediately responsible for a given task. Neoplastic processes affecting brain greatly challenge this already strained equilibrium.
In fact, striking alterations of microvasculature (MV) play a pivotal role in various cerebral pathologies. Such high-grade gliomas (HGG) display profound changes in density, shape, and permeability of their MV. 2 To overcome a hypoxic environment, tumor cells of densely cellular gliomas potently overexpress vascular endothelial growth factor (VEGF).3,4 This pluripotent molecule induces proliferation of endothelial cells (ECs) with a formation of thin-walled, and pericyte (PC) poor vasculature 5 with increased permeability. Such leaky vessels lead to an elevation in interstitial fluid pressure which is partly responsible for a significant decline in a patient’s quality of life. An inhibition of VEGF signaling inhibits vascular proliferation and decreases permeability and thus proved to be an important adjunct in treatment of gliomas.6,7 Predictably, cessation of anti-VEGF therapy allows a robust regrowth of abnormal tumor MV. 8
Arterioles, capillaries, and venules are the most plastic parts of the vasculature. ECs, PCs, and smooth muscles cells (SMCs) of small vessels play a pivotal role in regulation of both perfusion and permeability. There appears to be a primary role of ECs in mediation of directionality, speed, and shape of sprouting blood vessels. ECs, in turn, secrete platelet-derived growth factor (PDGF), which activates recruitment of PC. 9 MV of PDGF B–deficient mice shows loss of PC coverage and microaneurysms. 9 Similarly, in kidney, a genetic ablation of PDGF B leads to a loss of mesangial cells of renal glomeruli, a specialized type of kidney cells analogous to PC. 10 Surprisingly, a block of PDGF signaling not only inhibits PC recruitment and proliferation but also leads to a loss of MV in non-neoplastic brain. 11 It is thought that PC can stabilize nascent MV and even promote endothelial sprouting through mechanisms involving secretion of VEGF and angiopoietin 1.12–15 PC and glioma-derived PDGF can also stimulate EC growth and enhance angiogenesis. 16
Many aspects of physiology of MV rely heavily on signaling originating from basal membranes (BMs). 17 Extracellular matrix proteins (ECMPs) which are in mature form appear as defined BMs and are produced and secreted by EC, PC, and SMC. Both PC and SMC are enclosed by BM, whereas BM of EC abuts its abluminal surface. ECMPs of BM provide a structural support, play a direct role in signalizing through an interaction with integrin complexes, as well as retain and release in controlled fashion some growth factors. Deposition and stability of BM often rely on contribution from more than one cell type. For example, in vascular cell culture models, ECs alone fail to express BM, whereas in EC-PC co-culture, BM is formed efficiently. 18
Studies from our group as well as many others have shown striking alterations in expression of ECMPs in gliomas.19–21 In particular, differential deregulations in a family of trimeric proteins laminins (LAM), which represent one of the major constituents of brain MV, were identified. Laminins constitute a family of proteins composed of stoichiometric 1:1:1 ratio of three separate chains: α, β, and γ. In total, human genome contains 12 LAM genes, each with different chromosomal localizations (https://www.genenames.org/data/genegroup/#!/group/626). There are five separately encoded α (LAMA), four β (LAMB), and three γ (LAMC) chains, which form 15 known combinations. 22 Current nomenclature sequentially names specific α, β, and γ chains to come up with a specific name of a trimer. 23
By immunohistochemistry (IHC), in a quiescent brain, LAMs are seen along the lining of an arachnoid (LAMA1), glia limitans (LAMA1 and 2), capillary endothelium (LAMA5 and LAMA4), vascular (non-capillary) endothelium (LAMA4 and 5).24,25 LAMA1, LAMB2, and LAMC1 are predominantly expressed in quiescent brain MV. 19 In gliomas, the MV loses LAMA1 and gains LAMA2, 3, 4, 5; LAMB1, 2; and LAMC1. 26 Our group has also demonstrated that HGG, such as glioblastoma multiforme (GBM), strongly overexpress LAMB1, 21 which was not detectable in quiescent normal MV. 19 This is often accompanied by a loss of LAMB2. Downregulation of LAMB2 was associated with more aggressive clinical course of GBM and predicted a shorter patients’ survival. 27 However, precise mechanisms responsible for this association have not been elucidated.
A clue to a possible involvement of laminins in a control of vascular permeability was provided by transgenic animal studies. Knockout of LAMB2 expression in kidney was shown to drastically increase glomerular BM permeability reproducing a nephrotic phenotype.28,29 In fact, mutations in LAMB2 are responsible for Pierson syndrome with its characteristic congenital nephrosis. 30
Since our previous studies revealed that LAMB2 is strongly expressed in normal brain, wherein the vascular permeability is tightly controlled, but is often lost in GBM with its leaky MV, we sought to re-evaluate the expression of LAMB2 in various brain pathologies to explore a possible correlation between LAMB2 expression and vascular permeability. To this end, we chose to evaluate LAMB expression in conditions with increased permeability of blood–brain barrier (BBB) and compare it with normal and pathologic conditions with retained BBB function. We used a state of post-gadolinium enhancement (PGE) (presence or absence), on brain imaging [computed tomography (CT) or magnetic resonance imaging (MRI)], which served as an approximation measure of vascular permeability. 31 We observed that quiescent MV of normal brain or vasculature in low-grade infiltrative gliomas, both of which are characterized by no PGE, shows uninterrupted microvascular coverage by LAMB2. In contrast, significant breaks in LAMB2 were detected in brain conditions with avid PGE. We also observed that LAMB2 is deposited at all sites of BBB and proposes that LAMB2 plays a cell-independent critical role in a control of cerebral microvascular permeability. Since vascular integrity/permeability is generally believed to be associated with PCs, we therefore evaluated whether LAMB2 is PC-derived. We performed IHC, in situ hybridization (ISH), as well as bioinformatics analysis of scRNA-seq databases for the (1) cell of origin and (2) magnitudes of expression of LAMB1 and LAMB2. Our findings confirmed that in quiescent brain and in low-grade glioma (LGG), LAMB2 was indeed predominantly expressed by PCs but also ECs. In contrast, no LAMB1 was detectable. In contrast, in HGG, both LAMB1 and 2 were expressed by a phenotypically unique endothelial precursor cells (EPCs).
Initial detection of elevated LAM411 expression in GBM by whole tissue extract–based RNA profiling suggested the origin of the protein from the tumor cells. Indeed, dissociated glioma cells can express various ECMPs in culture.26,27,32,33 Moreover, inhibition of expression LAMB1 in glioma can suppress tumor growth possibly through a block in Notch pathway. 34 In contrary, IHC localizes LAM to BMs, which are not produced by typical astrocyte (except for the pial one), and therefore it has been uncertain to what extent glial cells contribute to LAM expression and deposition. In this study, we use IHC and ISH to evaluate a magnitude of contribution of microvascular versus glioma cells in respect to LAMB expression.
IHC, ISH, and also bioinformatics analysis of scRNA-seq databases showed that the predominant cell types expressing both LAMB1 and 2 are microvascular cells as well as normal glia limitans. Except for cases of gliosarcoma there was only minimal expression by cells of glioma.
To gain further insight into the regulation of LAMB expression in gliomas, we took an advantage of a clinical use of VEGF signaling inhibitor bevacizumab. The drug is widely used to block endothelial proliferation in intracranial tumor to normalize vascular permeability and relieve brain edema. Pretreatment of GBMs with bevacizumab lead to striking halt in LAMB RNA expression, normalization of the vasculature, with loss of staining for LAMB1 and restoration of LAMB2 coverage in BMs.
In silico analysis revealed a differential expression of transcription factors (TFs) in LAMB-producing cells. LAMB1-expressing EPCs and PCs were enriched for TF: FOSB, TFAP2A, TFAP2B, and TFAP2C, whereas LAMB2 (+) cells showed expression of TFs: E2F3, E2F4, and FOXF2, indicating that signaling pathways controlling LAMB1 and 2 are distinct and non-overlapping.
Since VEGF and PDGF play critical roles in EC and PC, we also analyzed a differential expression of molecules associated with VEGF and PDGF signaling cascades in normal brain, LGG and HGG, wherein we identified highly distinct sets of differentially regulated genes.
Collectively, our data indicate that LAMB1 and 2 have different roles in MV, with LAMB1 being involved in microvascular expansion, whereas LAMB2 in integrity of BBB. Increased microvascular permeability as detected by PGE during subacute phase of stroke, in radiation necrosis, GBMs, and in glial tumors with degenerative changes is tightly associated with an extensive loss of LAMB2 coverage of MV.
Materials and Methods
Human Tissue Collection
Human brain tumor samples were obtained in 2006–2024 via open neurosurgical resections in compliance with Cedars-Sinai Medical Center Institutional Review Board–approved human tissue collection protocol. Each case was independently evaluated and graded according to World Health Organization (WHO) criteria, as of 2021 and 2016 editions, 35 by two neuropathologists (SIB and Xuemo Fan). Since the goal of our study was to correlate LAMB1 and 2 expression and endothelial hyperplasia, we restricted our final analysis to tumors which met both WHO 2021 and WHO 2016 criteria. Therefore, cases of isocitrate dehydrogenase 1/2-negative glioblastomas which had no PGE on MRI and were also lacking endothelial hyperplasia were excluded.
IHC data on 1131 neuroectodermal tumors/controls (Table 1) were included in this study, which predominantly focused on infiltrative gliomas. Some parameters in various controls and other neuroectodermal tumors were evaluated for comparison. Expression of LAMB1 and LAMB2 was analyzed for all cases. Only a small fraction (29 total) of tumors and controls was tested by ISH. This selection was based on the availability of freshly submitted (less than 30 days in paraffin blocks) excisional biopsies, given our empirically determined tendency of ISH signal to greatly diminish upon prolonged storage. Clinical MRI and CT images of all the cases were assessed and a presence or an absence of PGE was recorded and correlated with expression patterns of LAMB1 and 2 by IHC (Table 1).
Summary of Cases evaluated by in vivo imaging, immunohistochemistry (IHC), in situ hybridization (ISH), and morphometric analysis.

IHC and ISH
Paraffin sections were stained on a BenchMark ULTRA platform using high pH antigen retrieval and the UltraView Universal diaminobenzidine (DAB) detection kit (Ventana Medical Systems, Tucson, AZ). A mouse monoclonal antibody to LAMB1 (DG10) was from Abcam (Cambridge, MA) and a mouse monoclonal antibody to LAMB2 (clone sc-59980) was from Santa Cruz Biotechnology (Dallas, TX). We used antibody to ERG (ETS-related gene) (Rabbit Monoclonal EPR3864 supplied by Roche, Tucson, AZ 85755, prediluted) as a marker of EC, and RGS5 (Santa Cruz, sc-390245, at 1:400 dilution) as a marker of PC. Negative control was performed by omitting the primary antibody and this revealed no immunoreactivity.
ISH was performed with custom-designed probes purchased from Advanced Cell Diagnostics (Newark, CA). For human LAMB1, a probe Hs-LAMB1 (11ZZ) was targeting 2040-5684 of NM_002291.2, and for human LAMB2, a probe Hs-LAMB2 (15ZZ) was targeting 2056-4667 of NM_002292.3. RNA probes were detected using RNAscope 2.5 HD Reagent as per manufacturer’s instruction (https://acdbio.com/rnascope%C2%AE-25-ls-assay-brown). Standard incubation times were used to obtain a quantifiable density of puncta corresponding to abundance of intracellular RNA. We observed that labeling for both LAMB1 and 2 was no longer detectable on tissue cut from older blocks. Therefore, only the tissue samples embedded in paraffin not earlier than 30 days prior to hybridization were used in this semiquantitative study.
Morphometric Analysis of IHC Stains
Evaluation of immunostaining was performed by two neuropathologists (SIB and Xuemo Fan, MD, PhD), blinded in respect to the diagnosis. At least three separate fields of the tumor, each containing not less than 10 vascular profiles, were evaluated. Staining intensity (grades: 0, 1, 2, and 3) was assigned by comparison with the LAMB2 intensity of entrapped or adjacent normal vasculature (grade 1). Lack of staining was given grade 0, staining intensity above the normal was assigned grade 2, while very intense diffuse staining was designated as grade 3. Non-paired Student’s t-test was used to evaluate the difference between expression index among the tumors.
To measure the vascular density, the whole slide images were acquired using Aperio GT450 scanner (Leica Biosystems, Deer Park, IL) and evaluated with a help of Aperio software (v12.4.2.7000). Randomly selected sites of normal white matter (control) and white matter invaded by glioma were projected at ×10 and the density of MV was determined using point counting method. 36 The exterior outline of the vasculature was defined as a layer of LAMB (+) vascular cells. Staining of glia limitans was disregarded. Staining for LAMB2 was used for quantification of microvascular density in LGG and for LAMB1 in HGG, given the partial loss of LAMB2 in the latter.
To measure a “matrix porosity index (MPI)” as a marker of LAMB2 abluminal surface coverage loss, we used Aperio-scanned images taken at magnification of ×400. The vascular profiles in the extracted fields were manually traced. The length of vascular outline was measured with ImageJ software (National Institutes of Health, Bethesda, MD, USA). MPI was calculated and defined as a percentage of LAMB2 (+) stretches of BM divided by the full length of vascular outline. At least 10 vascular profiles per tumor case were randomly selected. Average expression index per group was expressed as mean ± standard deviation (M ± SD). Non-paired Student’s t-test was used to evaluate the difference between expression index among the tumors. P<0.05 was considered statistically significant.
Quantification of ISH signal was performed on Aperio-scanned images taken at magnification of ×400. Subsequently, the extracted images were imported to ImageJ. Circumference of vessel along the outer surface was directly measured, followed by quantification of nuclei associated with LAMB staining. The total number of distinct quanta (dots) of staining was quantified to determine two parameters: quanta per length of a vessel and quanta per nucleus. Given the variable thickness of “glomeruloid” vasculature, we restricted our analysis to the more reproducible quanta/nucleus ratio. Our standard incubation protocol produced grains with on average surface area of 0.4 µ 2 . We applied this value to semiquantitative evaluation of labeling in the case, wherein there was an overlap/fusion of ISH signals. First, the total area of signal was measured using ImageJ and then the area was divided by 0.4. We acknowledge that thus obtained values were an underestimation, wherein some of dots were “overrun” and obscured by neighboring dots. At least three separate fields of the tumor, each containing not less than 10 vascular profiles, were evaluated. Average expression index per group was expressed as mean ± standard deviation (M ± SD). Non-paired Student’s t-test was used to evaluate the difference between expression index among the tumors.
Bioinformatic Analysis
Bioinformatic analyses were performed using 47 external single-cell data sets of three adult brains as normal samples 37 and 44 brain tumor samples (4 LGG, 22 newly diagnosed GBMs, and 18 recurrent GBMs)38,39—total cell count: 319,152. Scanpy v1.9 was used to merge, filter, and analyze the single-cell data. Samples were individually filtered by percentage of mitochondria genes (≤0.05), read counts (≤17,500), number of genes (4500≤ ≥750), and doublet score (≤0.04). After the filtering, the total cell count was 65,079 and the total gene count was 3193. Harmony was used to normalize all the data previous to the analyses to avoid bias between samples and data sets. Leiden algorithm and UMAP were used to establish the clusters of the samples. To characterize the clusters, marker genes (pval_cutoff = 0.0001, log2fc_min = 1.5) were ranked using t-tests and Wilcoxon rank-sum tests (see Table 1S for marker genes used for the clustering). This selection of lineage-specific markers was done in concordance with previously reported studies.39,40 Plotly (v5.18.0) was used to plot the data generated in these analyses.
Results
Expression of LAMB1 and LAMB2 in Normal Human Central Nervous System and Its Coverings
IHC detected both LAMB1 and LAMB2 in large caliber cerebral arteries and veins. LAMB1 showed higher levels around SMC of veins with very little staining of arteries, whereas LAMB2 was strikingly abundant in BM of SMC of large arteries (Fig. 1).
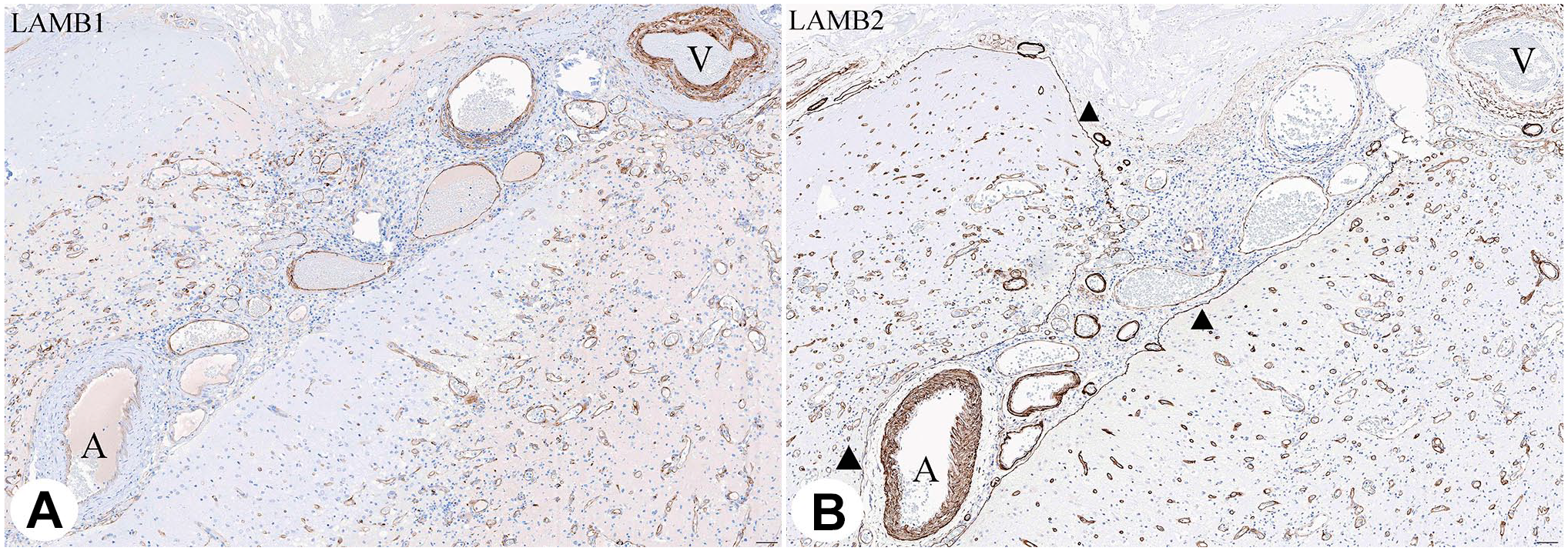
Consecutive sections of neocortex and leptomeninges (subacute infarct). Note predominance of LAMB1 (A) in leptomeningeal veins (V) and LAMB2 (B) in arteries (A). Glia limitans expresses LAMB2 only (triangles). Activated microvessels show weak expression of LAMB1 and strong of LAMB2. Scale bar: 100 µ.
No LAMB1 was detectable in normal cerebral MV, indicating that neither ECs nor PCs deposit appreciable quantities of LAMB1. In contrast, there was a continuous uniform single-layer marking of LAMB2 in quiescent brain capillaries. In addition, there was a complete coverage of pial membrane by LAMB2 (Fig. 1). From pia it extended in a sleeve-like configuration tracking along the arteries and veins toward the sites of commencement of MV. Such, LAMB2 outlines Virchow–Robin spaces in a distribution strikingly reminiscent to astroglia-derived BM of glia limitans. 41 Importantly, this is also the site of brain–cerebrospinal fluid (CSF) barrier. Thus, LAMB2 is specifically deposited within two distinct gates of permeability: BBB (microvascular outline) and brain–CSF barrier. Even though LAMB2 in BBB is likely to be EC/PC-derived, whereas in brain–CSF barrier is likely deposited by glia, its localization at critical junctions strongly suggests a unifying cell type–independent role of LAMB2 as a possible gate keeper in brain-fluid permeability.
Expression of LAMB1 and LAMB2 During Post-Infarct Vascular Remodeling
Next, we analyzed dynamics and a distribution of LAMB1 and 2 during non-neoplastic vascular remodeling. Five surgical pathology cases of subacute ischemic neocortical infarct of 7 to 12 days duration were identified in our surgical and autopsy files and were stained with antibodies to LAMB1 and 2. By IHC, we detected that the immature vascular sprouts at the leading edge of the infarct predominantly expressed LAMB1 with significantly less of LAMB2, whereas the trailing side of neovasculature tended to show less expression of LAMB1 but higher expression of LAMB2 (Fig. 2). This suggests that perhaps a sprouting edge of neovasculature requires transient expression of LAMB1, whereas subsequent maturation is accompanied by buildup of LAMB2 with a loss of LAMB1.
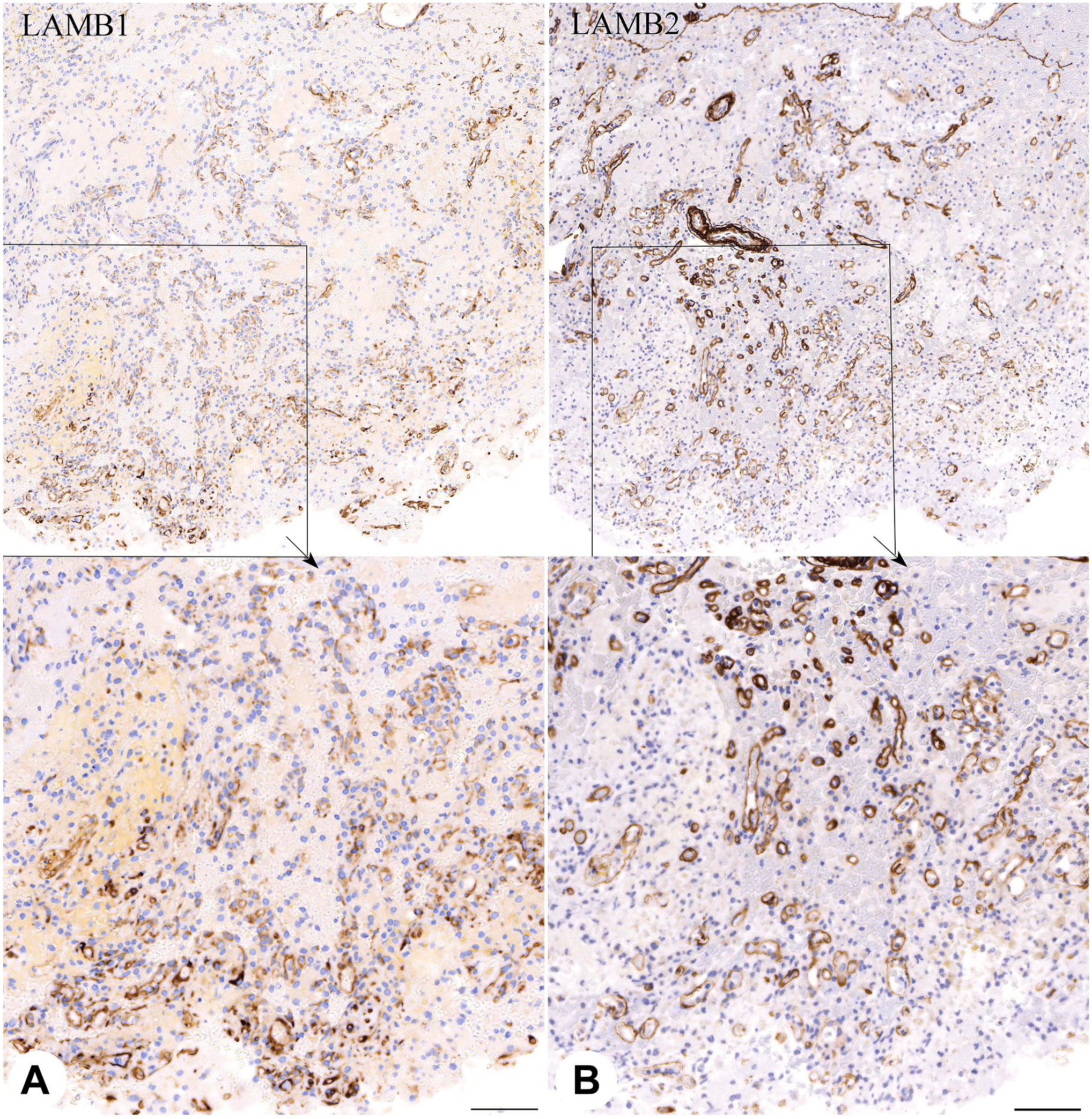
Consecutive sections of neocortex showing proliferating microvasculature in cerebral infarct of 7 days duration. Leading front (indicated with an arrow pointing toward necrosis) shows predominance of LAMB1 (A), whereas trailing edge shows more LAMB2 expression (B). Lower panel shows magnified portions of the upper images as highlighted by black rectangles. * Indicates pial membrane better seen with LAMB2. Scale bar on the lower panels: 100 µ.
Increased Density of MV in Low-Grade Diffuse Astrocytomas Is Not Accompanied by a Loss of LAMB2
Glioblastomas belong to a group of tumors with particularly striking hypervascularity and extensive areas of PGE. In contrast, diffuse astrocytomas, 2016 WHO grades II and III, and oligodendrogliomas, WHO grade II, show no PGE suggesting of intact state of BBB. To gain insight into an involvement of LAMB1 and LAMB2 in gliomas, we set to compare different grades of diffuse gliomas in respect to microvascular density and types of LAMB in their BM. Low-grade diffuse astrocytomas, 2016 WHO grades II and III, as well as oligodendrogliomas, WHO grade II, are characterized by tumor edema [detected as hyperintensity of T2 and fluid-attenuated inversion recovery (FLAIR) images], but no PGE (Fig. 3A). In contrast, HGG (glioblastoma, AKA diffuse astrocytoma, 2016 WHO grade IV, and anaplastic oligodendroglioma, 2016 WHO grade III) both show varying degrees of PGE (Fig. 3B). We used antibodies to LAMB1 and LAMB2 to measure white matter microvascular density (WMVD) in diffuse astrocytomas, 2016 WHO grades II, III, and IV, and oligodendrogliomas, 2016 WHO grades II and III (Fig. 3C to F). We found a slight but statistically significant increase in WMVD in low-grade (WHO grades II and III) astrocytomas (LGAs) as compared to a normal cerebral white matter: from 0.5±0.1 in normal white matter, to 2.0±0.2 and 3.08±0.37 in WHO grade II and III astrocytomas (Fig. 3G and H). Similar to prior reports, 42 WMVD in oligodendrogliomas was higher than in astrocytomas: 6.8±2.2 and 6.1±1.5 in WHO grade II and III lesions respectively) (Fig. 3B). This argues that even in LGG the MV undergoes expansion. In contrast to a normal brain, wherein there was no LAMB1 expression, WMVD in some of LGG showed appreciable, albeit weak, LAMB1 expression (Fig. 3C). LAMB2 showed strong and contiguous uniform expression in WMVD, which appeared somewhat stronger than in normal white matter (Fig. 3D).
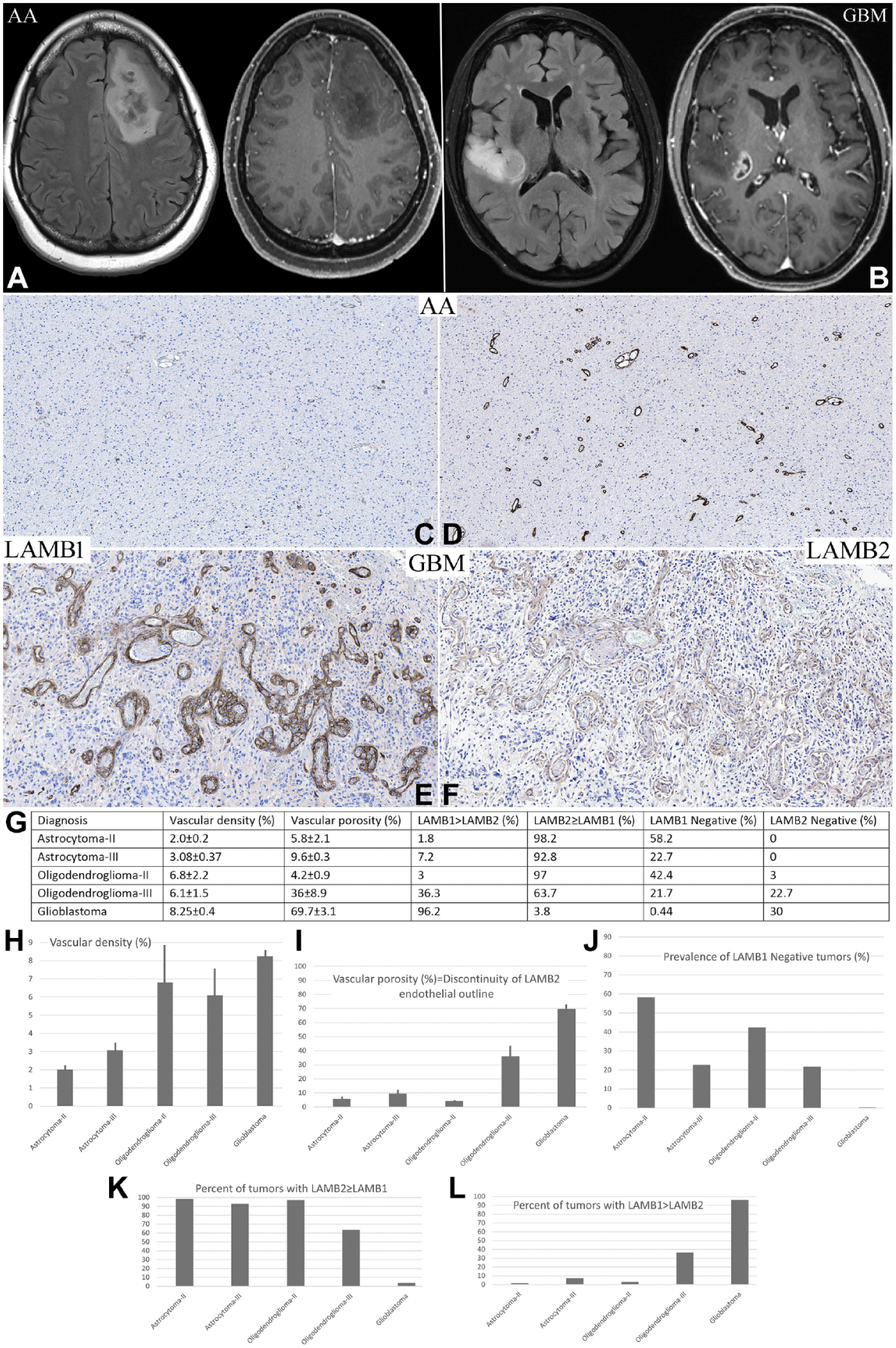
MRI, IHC, chart, and graph showing a summary of contrasting findings of low- and high-grade gliomas. A and B are representative MRI FLAIR (left) and T1 PGE (right in a pair) of IDH1 mutant CNS WHO grade 3 (anaplastic astrocytoma [AA] or WHO 2016) and IDH1 wild-type CNS WHO grade 4 (GBM). C-F: Corresponding IHC images from the above patients stained with antibodies against LAMB1 (C, E) & LAMB2 (D, F). The IHC images depict serial sections. G-L: Charts and Graphic representations of the morphometric analysis of CNS WHO grade 2-4 astrocytomas and CNS WHO grade 2-3 oligodendrogliomas. Scale bars: 200 µ.
Of note, in most of WHO grade II oligodendrogliomas, LAMB2 expression did not show a glia limitans-like outer layer, suggesting that the expanding vasculature of oligodendroglioma (so-called “chicken-wire” vessels) is a part of capillary network rather than arterioles or venules. 41
Expression of LAMB1 and LAMB2 in Glioblastomas
GBMs showed marked increase in WMVD as compared to 2016 WHO grade II and III astrocytomas (Fig. 3G). Similar to our previous studies, we detected a striking overexpression of LAMB1 in MV (Fig. 3E). In contrast, many vessels in HGG showed either a complete loss or extensive discontinuity of LAMB2 coverage (Fig. 3F, Fig. A1 A to D)
To assesses the extent of coverage of abluminal surface of MV by LAMB2, we designed an IHC-based “MPI,” which quantified the extent of LAMB2 staining discontinuity along the abluminal outline of MV (not inclusive of glia limitans, see “Materials and Methods”). A striking increase in MPI was observed in proliferating vasculature of GBM as compared to LGG and of normal brain (Fig. 3I). On average, LGG showed predominance of LAMB2 over LAMB1 expression, whereas this ratio was reversed in HGG (Fig. 3J to L).
In addition, LAMB2 loss in MV was also accompanied by discontinuity and extensive losses in LAMB2 coverage of glia limitans, similar to phenomenon previously reported for general laminin staining. 43 Thus, both sites with normally complete coverage of LAMB2 in the brains with intact BBB showed a failure of LAMB2 ensheathing. We posit that such loss might be at least partially responsible for increased BB permeability in HGG and characteristic PGE on radiologic imaging.
Of note, one can also appreciate a potent dissociation of vascular and glia limitans layer of LAMB in GBMs. This space is absent in capillaries and usually poorly discernible around arterioles and venules on regular histologic preparation of normal brain. 41 As described above for the LGG, the majority of vessels are thin and showed no duplication of LAMB2 lining in the outer space of a vessels, suggesting that angiogenic stimuli in the LGG drive predominant expansion of capillaries. In contrast, the vast majority of vessels in GBM showed a clear duplication of perivascular LAMB layers. This not only indicates fluid retention in perivascular spaces, but also shows “de-capillarization” of vascular bed with replacement of capillaries by arterioles and venules. One can speculate that vascular growth pathways in LGG and especially oligodendroglioma have a propensity to specifically target EC of capillaries, whereas those in GBM effect cells constituting pre- and post-capillary vascular beds.
Next, we examined the distribution of LAMB1 and LAMB2 in respect to luminal versus abluminal location within vasculature and also performed double labeling for LAMB and markers of EC or PC. We found that in LGG and HGG, both LAMB1 and LAMB2 were distributed throughout the entire thickness of the wall, both along the luminal/endothelial and also external surfaces (Fig. 4). Staining with antibody to EC marker ERG (ETS-related gene) showed encircling of ERG (+) nuclei by LAMB1 (+) matrix. In addition, some of ERG (−) nuclei (arrows on Fig. 4) tended to be predominantly excluded from the exterior of glomeruloid MV. No conclusive differential staining with markers of PCs (smooth muscle actin and RGS5) was obtained (not shown). We conclude that at least in HGG, one cannot reliably associate LAMB1 or LAMB2 with specific origin form either EC or PC, using IHC.
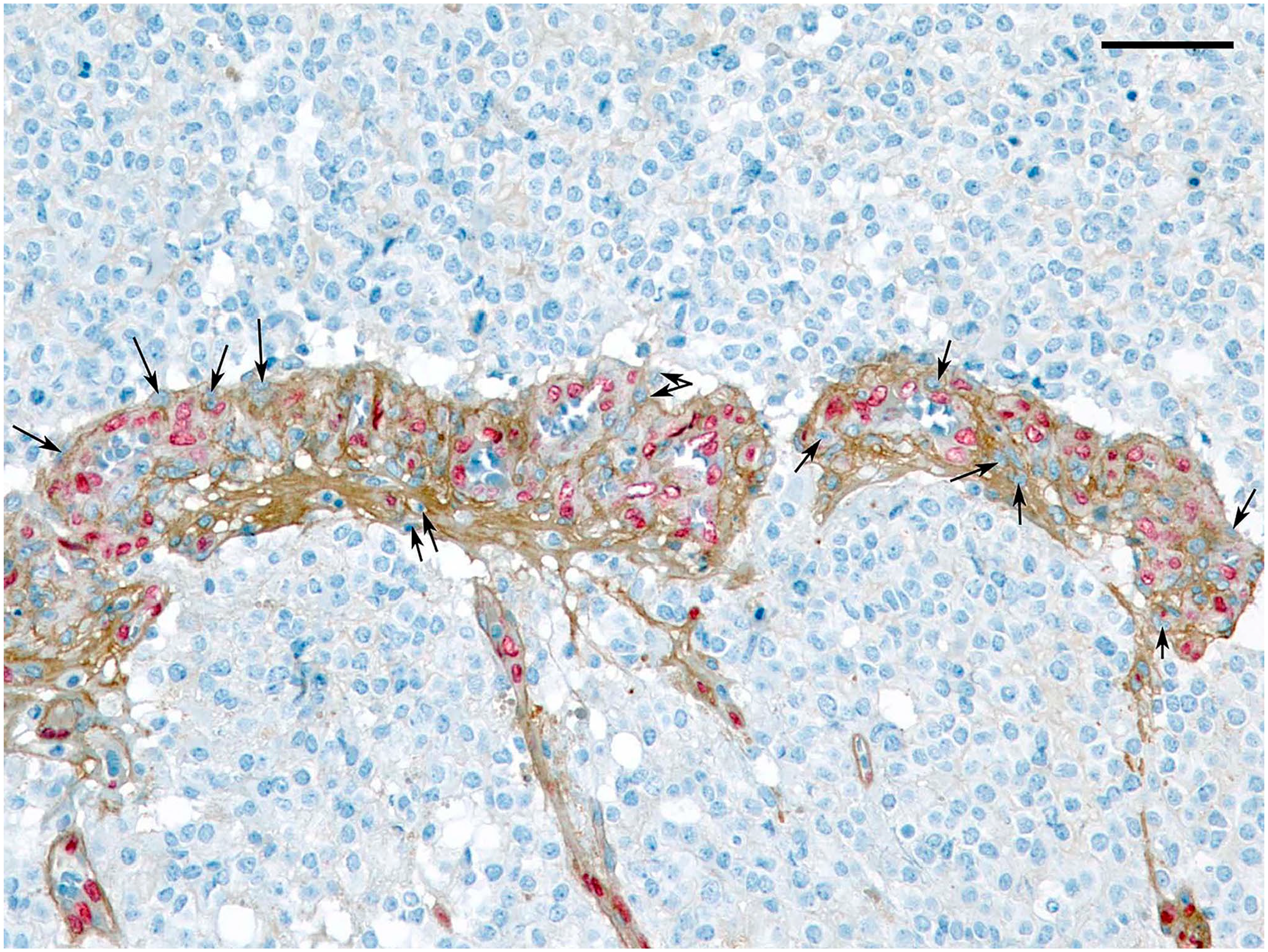
Double labeling of glioblastoma with antibodies to LAMB1 (brown) and endothelial marker ERG (red nuclear stain). Arrows indicate ERG-negative nuclei enclosed by LAMB1 (+) matrix. Scale bar: 100 µ.
The increased porosity of LAMB2 in MV can potentially result from a range of processes, such as failure of gene expression/RNA processing and altered protein secretion, or be a result of local dysregulation of proteases with protein-altered rate of degradation. IHC detects a steady-state presence of proteins and reflects a relative rate of protein secretion, proper assembly, and degradation. To alternatively estimate a rate of LAMB formation as well as to address the cell of origin of LAMBs, we performed an ISH studies with a set of RNAscope probes complementary to either LAMB1 or LAMB2. Both probes were produced by a commercial vendor (Advanced Cell Diagnostics), see “Material and Methods.” To assess probes’ specificity and lack of cross-reactivity, we took advantage of a differential expression of LAMB1 and 2 in human kidneys. 28 Wherein the BMs of immature glomeruli express LAMB1, whereas mature ones completely replace it with LAMB2. In contrast, BMs of renal tubules contain LAMB1 but not LAMB2. We confirmed that in human adult kidney, LAMB2, but not LAMB1, is detected by IHC in glomeruli. Our RNAscope probes showed exclusive expression of LAMB2 by cells of glomeruli and LAMB1 in tubular epithelium, thus validating usefulness of these probes to address a nature of cells of origin of LAMBs in the brain.
In agreement with our IHC data, diffuse astrocytomas of 2016 WHO grades II and III showed low to no mRNA signal for LAMB1 in the MV. LAMB2 mRNA levels in these tumors were also low. In contrast, GBMs showed striking upregulation of both LAMB1 (Fig. 5) and LAMB2 by ISH. We did not detect any appreciable difference in distribution of LAMB1 and LAMB2 in respect to luminal versus abluminal cells. No appreciable difference in ratios of overexpression of LAMB1 or LAMB2 between high-grade astrocytomas to LGAs was detected likely attributable to a near-threshold low expression of LAMB1 in LGAs. Although more signals of LAMB1 over LAMB2 were detected in GBMs, these were tested with different probes and therefore no reliable comparison could be drawn. In addition, based on technical limitations of ISH, we could not reveal any unequivocal foci of loss of LAMB2 ISH signal as was readily appreciable on IHC stains (Fig. A1).
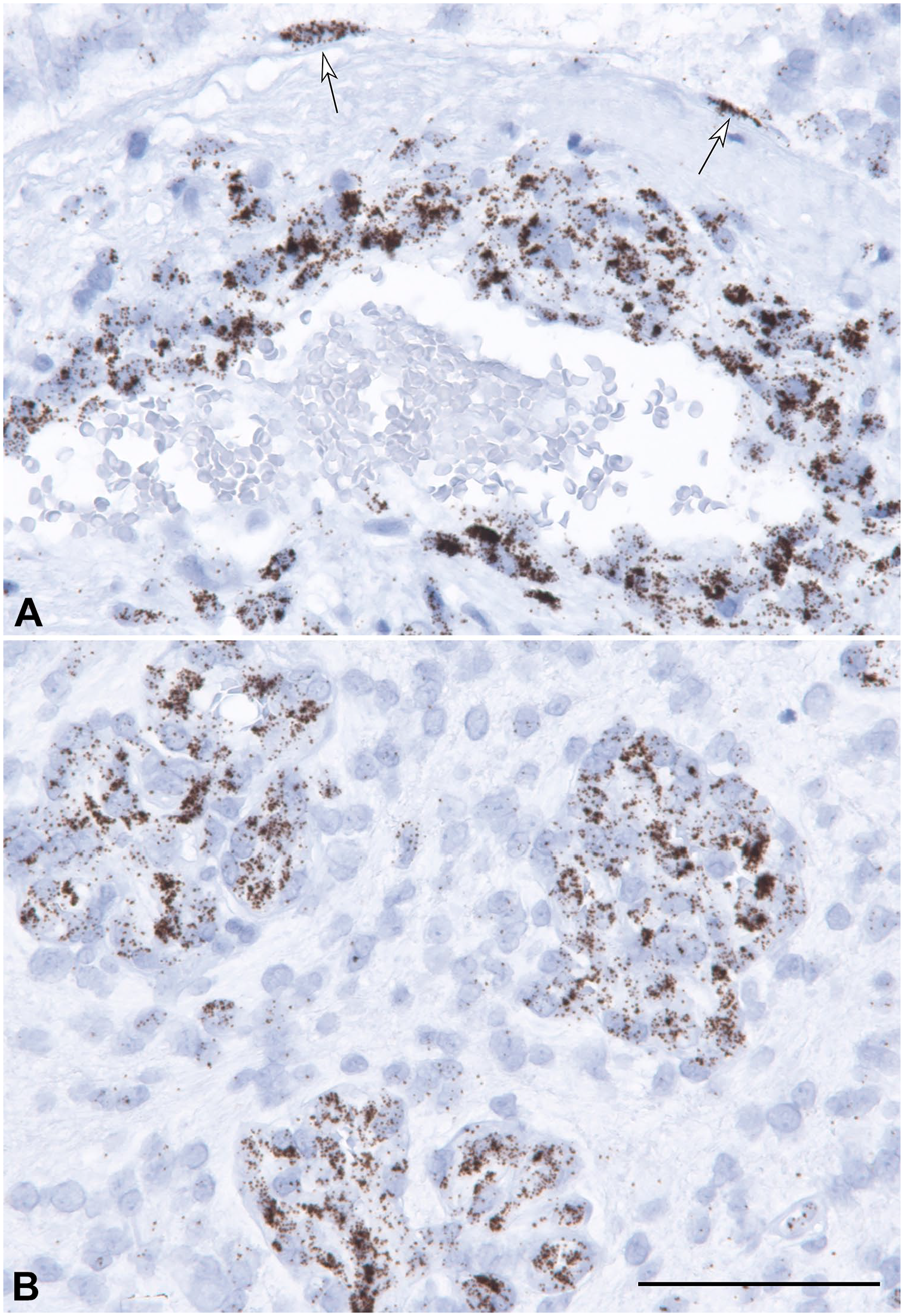
In situ hybridization labeling of GBM with a probe to LAMB1. Note much stronger staining of vascular cells over that of glial tumor cells. Also, remarkably strong staining of cells outlining Virchow–Robin space (arrows on A), suggesting a derivation of the vessels from artery or a vein, rather than from a capillary bed (B). Scale bar: 100 µ.
Many vessels contained distinct glia limitans-like layer of LAMB1 or 2 ISH-positive glial cells with high number of signals (Fig. 5).
We also observed that glial cells, but not neurons, were associated with a few ISH signals for both LAMB1 and 2. However, their density was much lower (1–5/nucleus versus 40–80/nucleus in microvascular cells, Fig. 6).
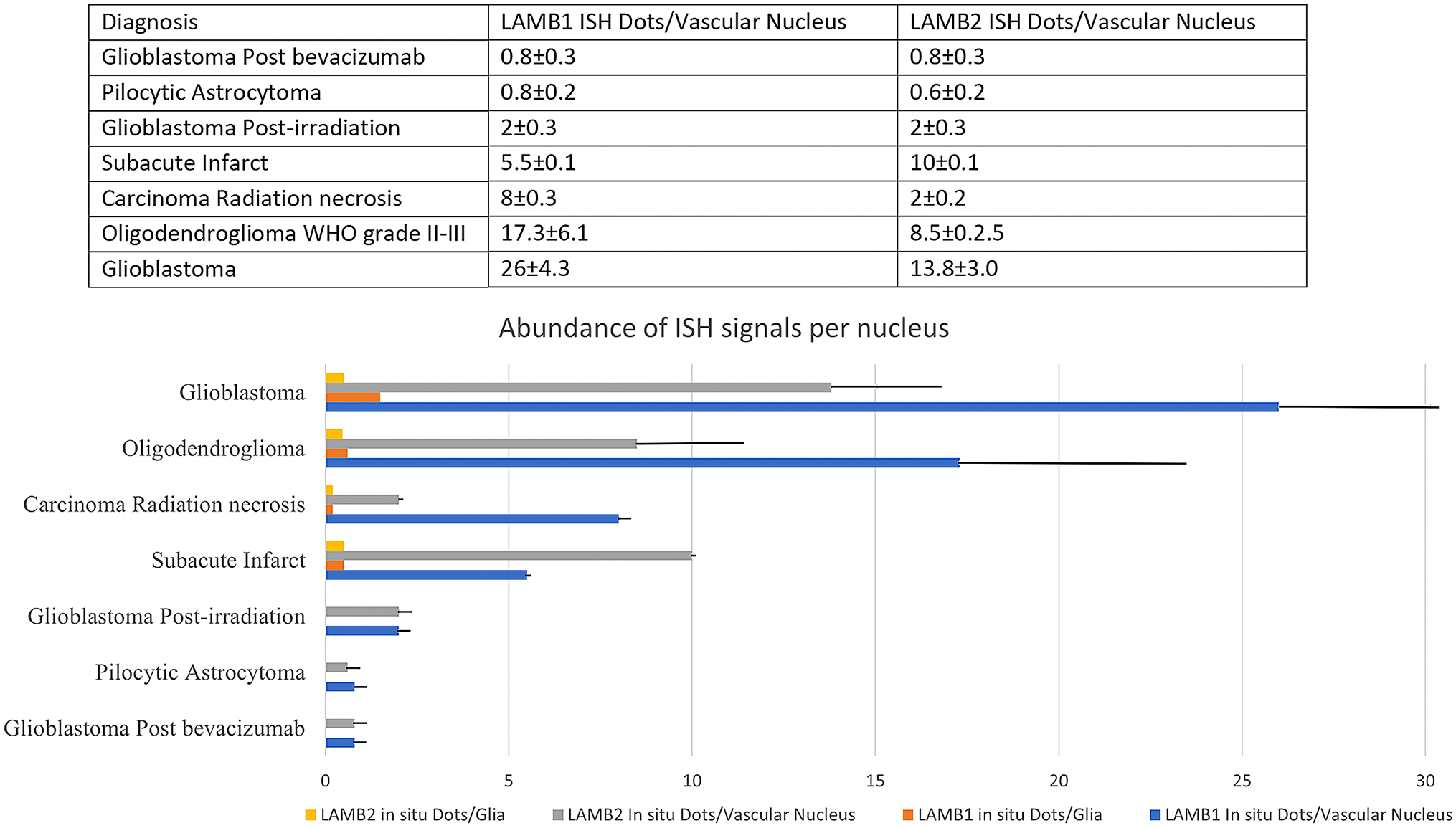
Chart and graph representations of morphometric analysis of LAMB1 and 2 in gliomas evaluated by in situ hybridization. Note a dramatic downregulation of LAMB1 and 2 in bevacizumab (Avastin)-treated glioblastomas to a level seen in benign pilocytic astrocytomas.
Normalization of Histology and Loss of Expression of LAMB in GBMs Treated With Bevacizumab
Next, we examined the effect of abrogation of VEGF signaling in GBM in patients who recently received bevacizumab (Avastin) treatment. We identified five patients with recurrent GBMs who underwent a repeat tumor biopsy in a setting of recent bevacizumab infusion. Specifically, after initial surgery with gross total resection (of the contrast-enhancing tumor), the treatment proceeded with concurrent temozolomide (TMZ) (75 mg/m2/day for 6 weeks) and radiation therapy (60 Gy in 30 fractions) and followed by six maintenance cycles of TMZ (150–200 mg/m2/day for the first 5 days of a 28-day cycle), according to the results of the phase III EORTC 26981. The patients were followed with serial MRI. Upon new emergence of PGE, they have received several dozes of bevacizumab. Subsequent MRI showed minimal to no PGE. All patients received a last infusion of the drug 4–5 weeks prior to biopsy. All samples demonstrated recurrent gliomas with no endothelial proliferation. IHC for LAMB1 showed weak to no staining corresponding to levels seen in diffuse astrocytomas, WHO grades II and III, whereas LAMB2 was seen fully encircling the MV. ISH for LAMB1 showed less than one signal per nucleus (Fig. 6) and was similar to ISH labeling for LAMB2. Our data indicate that a block in VEGF signaling results in normalization of LAMB1 and 2 expression.
Failed Deposition of LAMB2 in Post-Irradiated MV
Radiation injury (RI) is a major concern in oncology given its intrinsic morbidity as well as clinical and radiologic mimicry to tumor recurrence, for example, PGE. RI clinically manifests months to many years after treatment and there are no established biomarkers, predictive of individual susceptibility to the pathology. Histologic appearance of radiation effect on a brain predominantly manifests in vascular injury (ectasia, increased permeability, fibrin exudation, vascular hyalinization, endothelial nuclear atypia) and additional cytopathology of oligodendroglia. Because of its association with PGE, we predicted a loss of LAMB2 coverage in patients with histologic evidence of RI developed after treatment of GBM or metastases. Indeed, for LAMB1, its expression was reminiscent to subacute infarction with sustained deposits along the sprouting MV, whereas for LAMB2, we observed extensive zones of LAMB2-negative microvascular buds (Fig. 7A and B, respectively). ISH labeling mirrored IHC stains (Fig. 7C, D for LAMB1 and E for LAMB2, Fig. 6). This suggests that either proliferating EC failed to switch LAMB1 to LAMB2 expression or that radiation more selectively inhibited proliferation or maturation of PC precursors. The LAMB phenotypes were similar between patients with GBM, who have been maintained on temozolomide and our patients with metastases, which were not on maintenance chemotherapy. This argues for a dominant role of RI in respect to a damage to vascular BM.
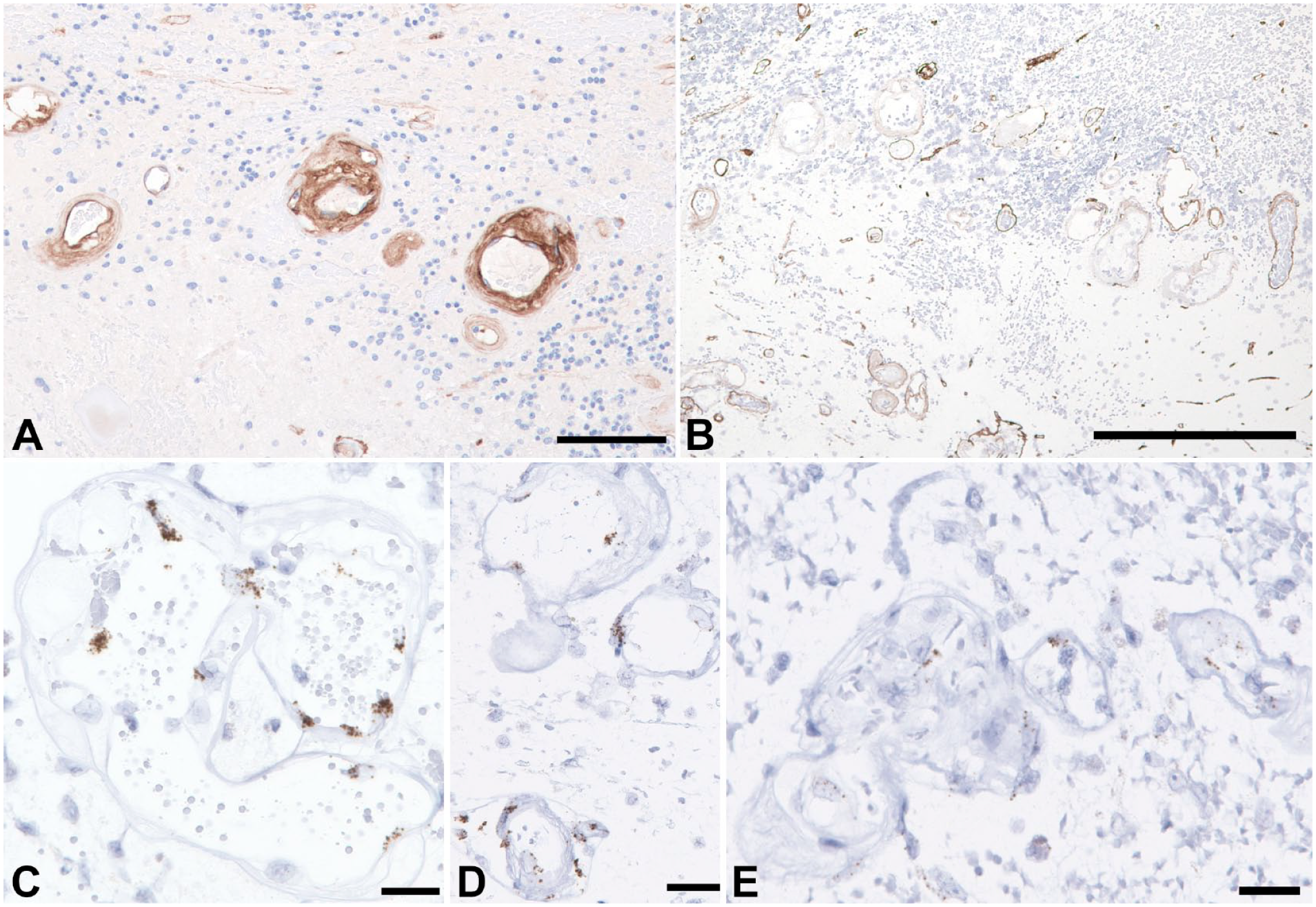
Representative images of IHC (A & B) and ISH (C-E) of post-radiation necrosis in patient with a metastatic carcinoma. Damaged vessels overexpress LAMB1 (A) and show extensive loss of LAMB2 with apparent devitalization (B). ISH shows rather strong expression of LAMB1 in endothelium (C & D) corresponding to IHC staining and comparative diminution of LAMB2 (E). Scale bars: 200 µ.
LAMB1 and LAMB2 Expression at Single-Cell Level in the Brain MV
To elucidate the extent of LAMB1 and LAMB2 expression in individual cells of healthy brain MV versus tumor MV, we performed in silico analysis of 3 control and 44 tumor samples (see “Materials and Methods” for accessed databases). Specifically, we used in silico isolation of the following five different types of samples: (1) adult brain endothelial (AdultBrainEndo), (2) adult brain perivascular (AdultBrainPeri), (3) LGG, (4) newly diagnosed glioblastoma (ndGBM), and (5) and recurrent glioblastoma (rGBM); (Fig. 8A, right panel).
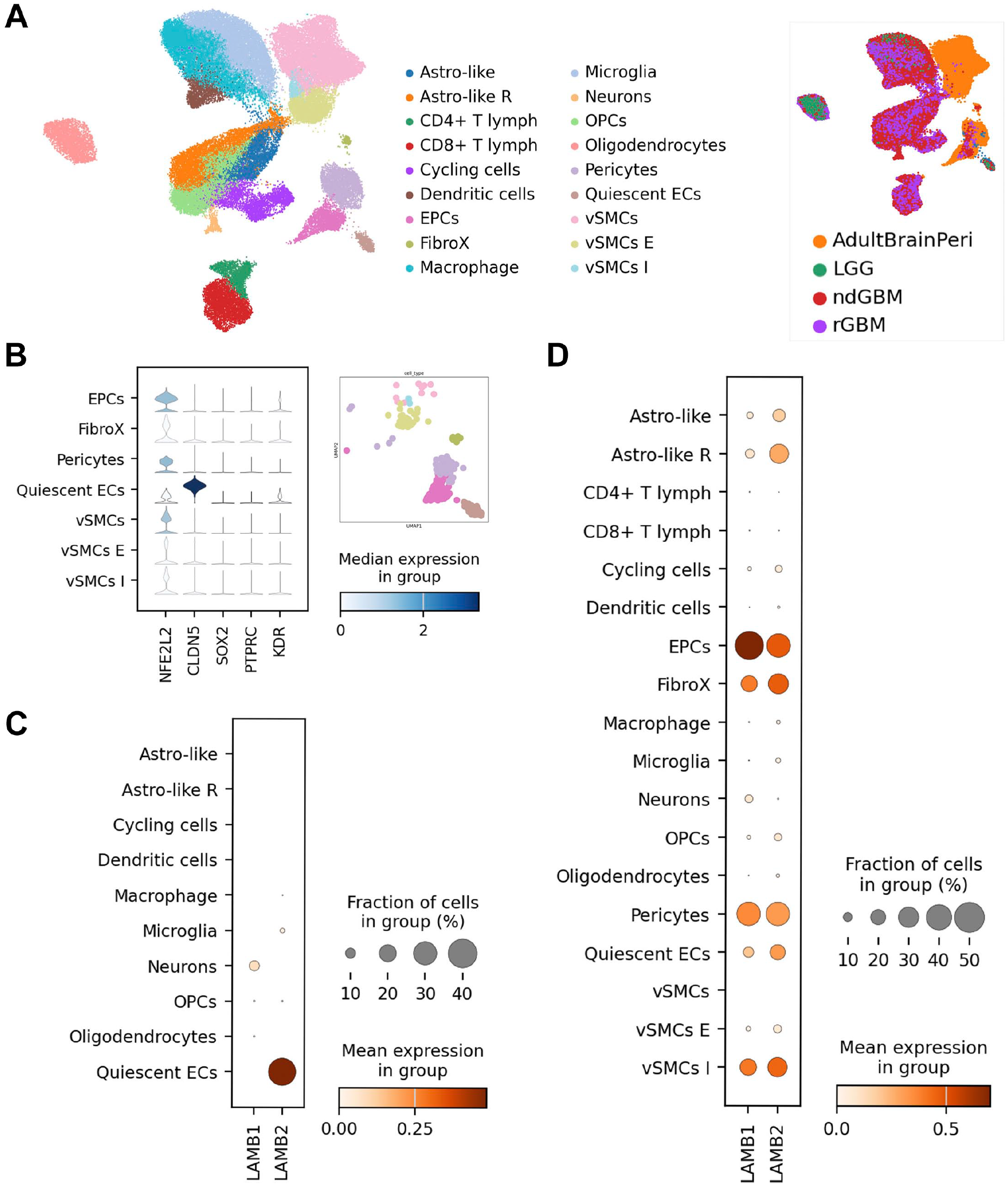
(A) Left panel: UMAP visualization depicting normalized cell clustering with the different cell types characterized based on the differential expression genes on Table 1S. Right panel: equal UMAP visualization showing the five distinct human single-cell data sets included in this study: (adult brain endothelial [AdultBrainEndo], adult brain perivascular [AdultBrainPeri], low-grade gliomas [LGG], newly diagnosed glioblastoma [ndGBM], and recurrent glioblastoma [rGBM]). (B) Top five markers employed for characterization of microvasculature-related cells. Differential gene expression analysis of the expression patterns of LAMB1 and LAMB2 in low-grade tumor data sets (C) and high-grade tumor data sets (D).
In total, 18 clusters were identified, 7 of which were significantly MV-related (Fig. 8A, left panel). PCs and FibroX (fibromyoblast and fibroblast) were clearly identified by the top-ranked marker genes (Table 1S). Based on the UMAP plot (Fig. 8A), together with the expression of five marker genes (NFE2L2, CLDN5, SOX2, PTPRC, and KDR; Fig. 8B), we found two clusters of ECs that were divided into EPCs and quiescent endothelial cells (quiescent ECs). Adding ACTA2, TAGLN, and ABCC9 as markers, we divided the cluster of vascular smooth muscle cells (vSMCs) into three different sub-clusters: mature vSMCs (vSMCs; no ACTA2, TAGLN, ABCC9 expression), intermediate-stage vSMCs (vSMCs I; TAGLN-ABCC9 expression), and early-stage vSMCs (vSMCs E; ACTA2-ABCC9 expression). To evaluate for a potential contribution of tumor cells in production of LAMB, we also separated four clusters enriched in the neoplastic cells. These include oligodendroglial precursor cells (OPC), astrocyte-like cells, astrocyte-like reactive cells, and cycling cells. Differential overexpression of the following markers was used for cluster designation: OLIG1, OLIG2, NKX2-2 for OPCs; MCM9, CNDs, CDKs for cycling cells; ALDHL1, SOX9, NFIA for the astrocyte-like cells; and GFAP, S100B, AQP3 for astrocyte-like reactive clusters.
Next, we characterized LAMB1 and LAMB2 expression in the above-described cell type–specific samples (Fig. A2 A to D). Consistent with histochemical data, LAMB1 exhibits low expression in normal adult vascular or glial cells. It showed no expression in LGG, except for a minor group of EPCs and a modest expression associated with neurons (Fig. 8C and D; Fig. A2 C and D).
In contrast, we observed that LAMB1 was widely expressed in GBM samples being enriched in EPCs, PCs, vSMCs I, FibroX, and quiescent ECs (Fig. 8D; Fig. A2 D). Minimal expression of LAMB1 was detected in cell clusters associated with neoplastic glial cells (Fig. 8D; Fig. A2 B and C).
LAMB2 was identified in all clusters related to MV, except for vSMCs.
In addition, contrasting patterns of LAMB2 expression were observed in LGG versus GBM (Fig. 8C and D; Fig. A2 C and D). Low but noticeable expression of LAMB2 was detected in a fraction of presumably neoplastic astrocyte-like cells and astrocyte-like reactive cells (Fig. 8D; Fig. A2 B and D).
Thus, bioinformatic analysis confirmed our IHC and ISH findings of predominant association of LAMB1 with proliferating microvascular cells of HGG with minimal expression by tumor cells. It also validated a substantial contribution of PCs to expression of LAMB2 both in quiescent brain and in HGG (Fig. 8D; Fig. A2 B and D).
In Silico Identification of LAMB TFs and Relevant Pathways in the MV
To gain additional insights into the regulation of LAMB genes, we next analyzed patterns of co-expression of their potential TFs using genecard database by QIAGEN determinations.
They predicted that the LAMB1 gene promoter can potentially bind the following TFs: AP-1, AP-2alpha, AP-2alphaA, AP-2beta, AP-2gamma, Brachyury, c-Fos, c-Jun, FosB, Pbx1a, while for the LAMB2 gene, the TFs were E2F, E2F-1, E2F-2, E2F-3a, E2F-4, FOXF2.
Given that prediction, we next applied in silico approach to evaluate for involvement of the above TFs in respect to expression of LAMB1 and 2 in MV (Fig. A3).
For LAMB1, we found that while certain TFs showed high gene expression, LAMB1 itself is not highly expressed in some microvascular cells, as we can see in tumoral quiescent cells, whereas JUN and FOS are highly expressed. However, a co-expression of the FOSB, TFAP2A, TFAP2B, and TFAP2C TFs appears to be correlated with increased LAMB1 expression in EPCs and PCs (Fig. A3 A).
For LAMB2, the TFs previously associated with the LAMB2 gene promoter did not seem to directly correlate with the expression of LAMB2, revealing the intricate nature of gene regulation for both genes (Fig. A2 B).
There is a key role of VEGF in the biology of EC, whereas PDGF is critical for PC. Therefore, we next analyzed the pattern of genes regulated by the VEGF and PDGF pathways in MV-related cells using KEGG database (Figs. 9 and 10).
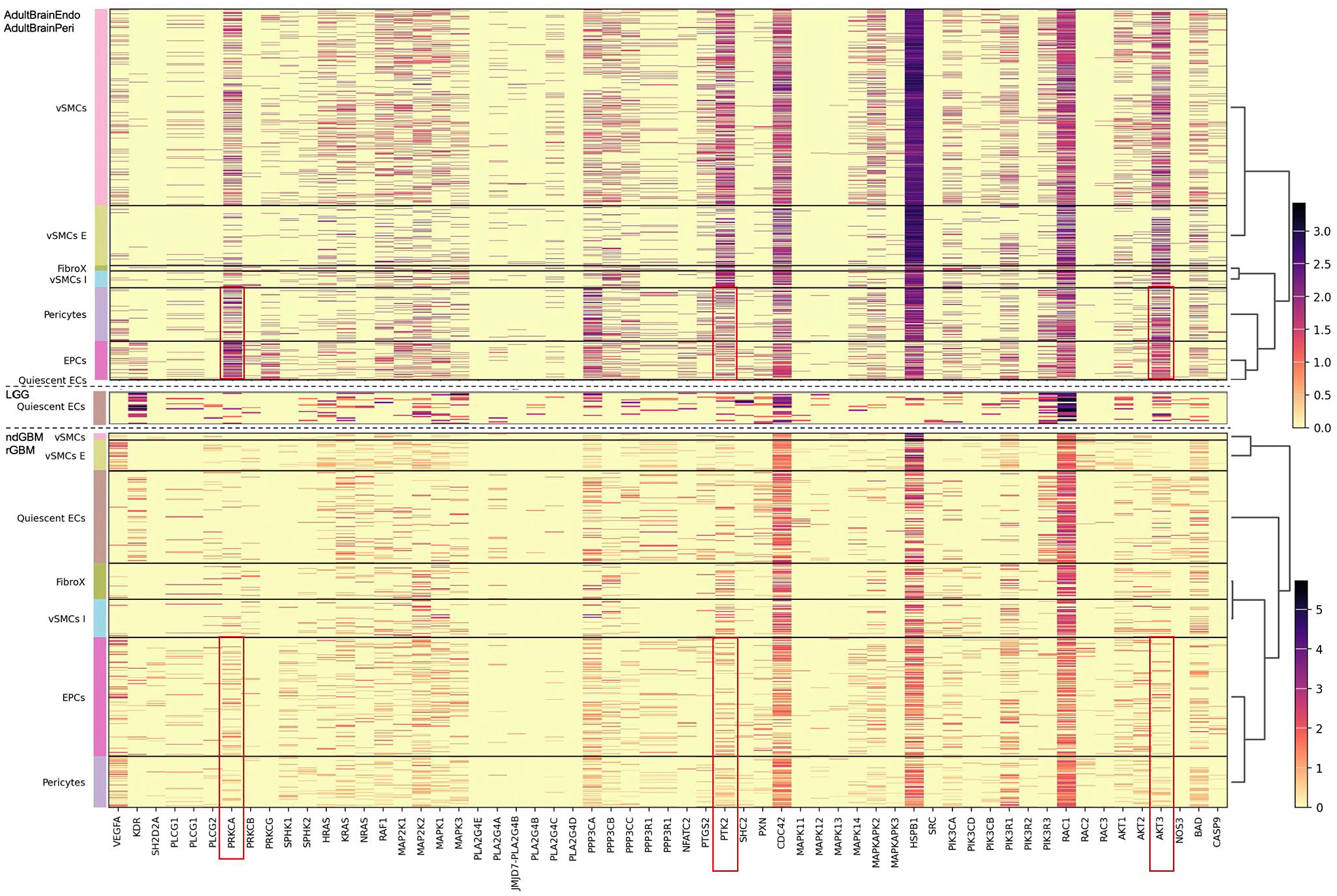
Expression of VEGF pathway-associated genes in the microvasculature-related cells analyzed in adult brain endothelial (AdultBrainEndo), adult brain perivascular (AdultBrainPeri), low-grade gliomas (LGG), newly diagnosed glioblastoma (ndGBM), and recurrent glioblastoma (rGBM). Note the striking downregulation of PRKCA, AKT3 and to a lesser degree PTK2 in EPC and PC of GBM as compared to adult brain (highlighted with red frames).
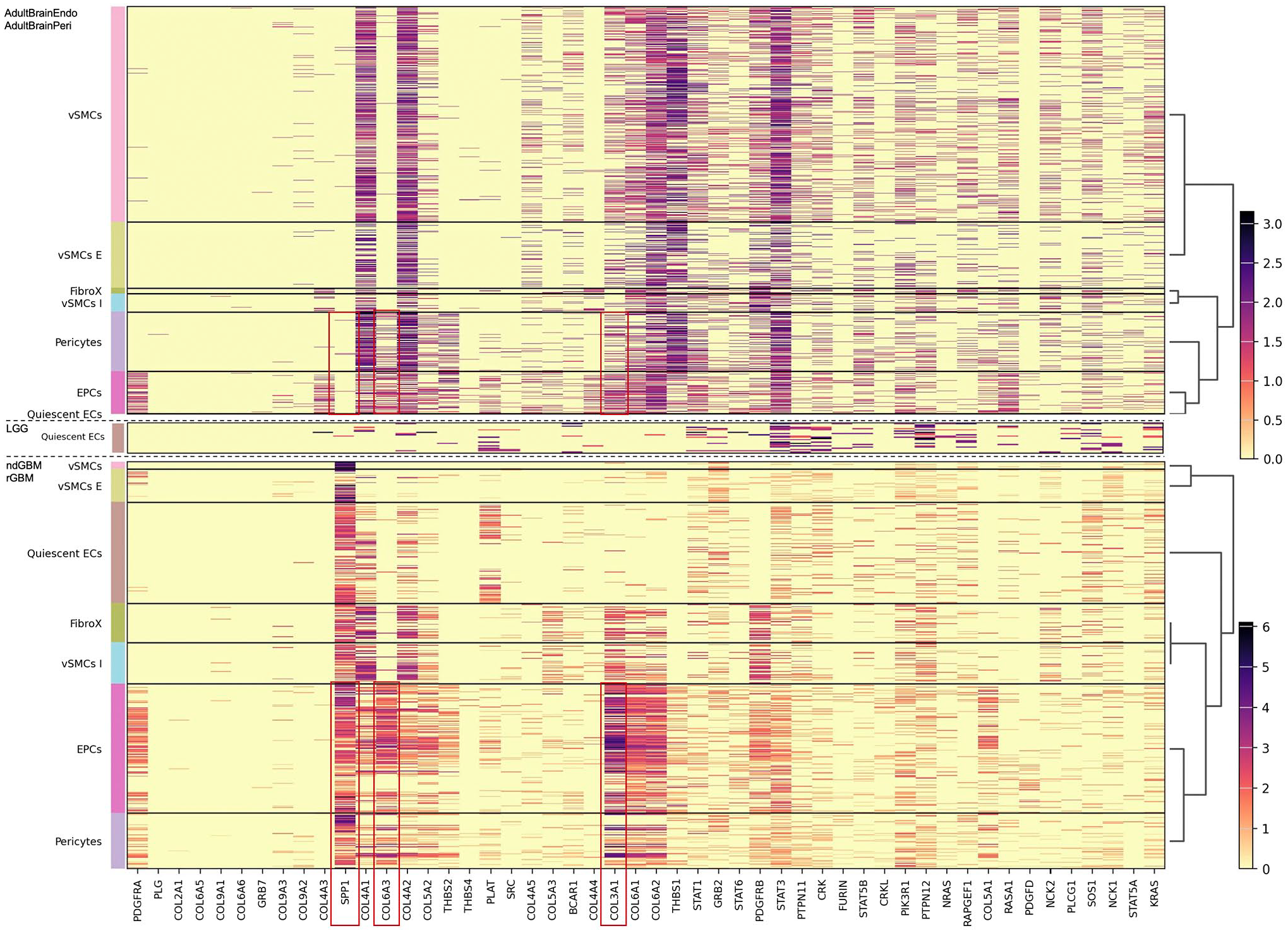
Expression of PDGF pathway-associated genes in the microvasculature-related cells analyzed in adult brain endothelial (AdultBrainEndo), adult brain perivascular (AdultBrainPeri), low-grade gliomas (LGG), newly diagnosed glioblastoma (ndGBM), and recurrent glioblastoma (rGBM). Note the upregulation of SPP1COL6A3, COL3A1 in EPC and PC of GBM as compared to adult brain (highlighted with red frames).
The gene expression of VEGF pathway–associated genes showed a similar pattern when we compared adult brain with LGG and HGG (Fig. 9). However, we detected a strong downregulation of PRKCA, AKT3 and to a lesser degree PTK2 in EPC and PC of GBM as compared to adult brain (Fig. 9).
In respect to PDGF signaling pathway–associated genes, the differences were even more profound. Whereas the expression of PDGF pathway–associated genes did not appear to be cluster-specific in the adult normal brain samples (Fig. 10), there was a cluster-specific expression pattern of PDGF-related genes in GBM, with a significantly higher expression of collagen family/related genes, particularly in EPCs (Fig. 10). Last, it is noteworthy that there was a marked overexpression of SPP1 in GBM as compared to adult brain samples.
Discussion
Neoplasia invokes expansion and remodeling of tumor’s vascular supply. In glioma, it is driven predominantly by hypoxia, which induces secretion of VEGF by the tumor cells. This potent mitogen crowds EC in MV and is often associated with vascular thrombosis. The mechanics of vascular growth are only partially understood, but require an integration of sprouting, branching, transient focal destabilization of BMs, and remodeling of pericytic sheaths.
In this study, we extended our prior observations on the involvement of laminins 411 and 421 in the biology of brain tumors. We observed that presence of LAMB2 is characteristic of MV of quiescent brain with intact BBB. This feature is maintained in low-grade, gadolinium non-enhancing gliomas with intact deposition of microvascular LAMB2.
PCs are thought to be the primary cell type responsible for integrity and permeability of MV. PC coverage of MV is much higher in tissues with tight microvascular barrier functions, such as central nervous system. PC to EC ratio is very high in the retina (1:1), lower in brain (1:3), being still much higher as compared to a myocardium (1:100). 44
Because many aspects of microvascular integrity are conferred by PC, we hypothesized that LAMB2 is also predominantly secreted by PC. However, presence of a continuous coverage of MV by LAMB2 argues for an alternative origin from the EC. Our in silico analysis of scRNA-seq data set of vascular cells in rodent brain has unequivocally demonstrated that LAMB2 expression is detected predominantly in SMC, PC, and perivascular mural cells, but not in EC. 45 Similar findings were also detected in human brain MV. 37
Our IHC and ISH studies were inconclusive in respect to the cell of origin for LAMB2, which is likely due to intrinsic limits of technologies. The steady-state rate of LAMB2 expression is too low in the quiescent brain MV and is also transient in normal repair. In contrast, in HGG there is a very high level of both LAMB1 and 2 expression. scRNA-seq data analysis in HGG shows striking enrichment of both LAMB1 and 2 in a distinct population of EPC. This suggests that the microvascular cells of GBMs are locked in an immature EPC phenotype, perhaps by an overwhelming stimulatory onslaught of tumor-derived VEGF.
Localization of LAMB2 along the sites of BBB and CSF–brain barrier strongly suggests its contribution to a control of permeability. PGE is a crude but clinically useful tool to assess BBB permeability in vivo. 46 Low-grade infiltrative gliomas (grade II and III as per 2016 iteration of WHO classification) show no PGE and maintain tight BBB. When we analyze a distribution of LAMB2 in low-grade infiltrative gliomas, we observe a contiguous, uninterrupted sleeve of the protein in MV. This contrasts with extensive disruption of both microvascular and also parenchymal (glia limitans) sleeves of LAMB2 in HGGs, which are known for their extensive PGE and leaky BBB. Therefore, irrespective of cell of origin of LAMB2 (which based in our single-cell data could be tumor grade dependent), a complete coverage of both BBB and parenchymal glia limitans barriers by LAMB2 appears to correlate with restricted permeability.
In contrast to LAMB2, LAMB1 is not readily detectable in normal brain MV. Its expression is upregulated during physiologic remodeling and strikingly so in neoplastic neoangiogenesis. Although the levels of LAMB1 drastically increase in HGG and we observe a complete coverage of vasculature by LAMB1, this does not correlate with intact BBB permeability as proven by PGE on MRI. This points toward a functional non-redundancy between growth-associated LAMB1 and impermeability-associated LAMB2. We therefore assessed structural differences between LAMB1 and LAMB2 proteins using in silico approaches.
The respective genes for LAMB1 and LAMB2 are located on 7q22 and 3p21 and encode for proteins of 1786 and 1798 aminoacids, respectively. 23 Most of the differences between two proteins are within a stretch of extra 12 aminoacids in N-terminus in LAMB2. Despite this rather subtle difference in N-terminus, in silico analysis predicts a completely different set of interactive proteins, which might account for their non-overlapping functions.
Interestingly, α4 chain is a component of both laminins 411 and 421. In glioblastomas, α4 chain is strongly overexpressed 19 and replaces predominant α1 chain of normal MV. 26 In contrast to α1 (LAMA1), the LAMA4 has a truncation of N-terminal tail, which is normally responsible for an association with other laminin trimers. The laminins with full LAMA N-terminus, such as LAMA1, form a regular planar hexagonal mesh in BM. 47 Therefore, one can predict that replacement of LAMA1 to LAMA4 confers higher porosity BMs, which might become more conducive to EC sprouting. An additional replacement of laminin β2 to β1 is predicted to further increase the permeability.
In contrast to another group of ECMPs, collagens, which are pre-assembled into protofibrils in endoplasmic reticulum (ER), little is known about the site of and control of assembly of the hexagonal scaffolding mesh of laminins. Our data show that both LAMB1 and LAMB2 are co-expressed in the same microvascular cells and perhaps an assembly of a mixed laminin 411/421 mesh already commences in ER or Golgi complex. It is presently unclear how much mobility persists in the trimers of laminins within the plane of BM laminin layer and if there is any mechanism for individual removal and replacement of α, ß, and γ chains in trimers.
We determined that abrogation of VEGF signaling in vivo by bevacizumab inhibits expression of LAMB1 at both RNA and protein levels. LAMB2 responded to bevacizumab by a lesser decline in RNA expression (Fig. 7). By IHC, we detected an increase of LAMB2 vascular coverage which correlated with a loss of PGE on MRI. While we hypothesized that the normalization of vasculature by bevacizumab might be associated with a direct increase in LAMB2 RNA levels, its regulation by VEGF signaling appears to be more complex or indirect. Mechanistically, it could be mediated by increased resistance to RNA/protein degradation or improved extracellular retention, but the exact mechanisms clearly need further exploration.
scRNA-seq study of normal rodent brain demonstrated production of LAMB2 by PC and vascular mural cells. 45 This agrees with our in silico data in human cells. It has been previously shown that ECs recruit PCs, and its renal analogue, mesangial cells, induces their proliferation and maturation through the secretion of PDGF and its receptor on PC. 15 This also involves angiopoietin (Ang)-TIE (Tyr kinase with Ig and EGF homology domains) signaling. 48 Disruption of PDGF signaling leads to a loss of PC/mesangial cells, formation of microaneurysms, and proteinuria.9–11 Importantly, in vitro experiments show that EC alone cannot produce BM, whereas in a co-culture of EC and PC, BM is formed, in which secretion of LAMB1 is attributable to EC only, whereas LAMB2 is produced by PC. 18
VEGF plays a direct role of an inhibitor of PC and vessel maturation. 49 Therefore, we posit that the origin of LAMB1 and LAMB2 throughout the full thickness of vascular wall in GBMs is likely to be from a population of in silico-defined EPCs and fibroblasts (FibroX), which coincidentally is the predominant cell population in this setting (Fig. A1). A blockade of VEGF signaling in GBMs shifts a balance of growth signals and promotes a maturation of EPCs and FibroX toward PC, rather than maintaining them in more pluripotent LAMB1+/LAMB2+ state.
Scarce attention was paid to the the regulation of transcriptional control of LAM expression. One study determined that LAMB1 promoter in human is under direct control of c-JUN transcriptional factor and works downstream of tyrosine kinase signaling pathway. 50 In addition, AP-1 complex also promotes LAMB1 expression in human colon carcinoma.51,52 Our analysis has confirmed that at transcriptomic level there is a co-expression of LAMB1 together with JUN, FOS, and FOSB (part of the AP-1 complex) in microvascular cells of normal adult brain as well as in LGG and HGG. In addition, we unveiled that TFAP2 family genes (TFAP2A, TFAP2B, and TFAP2C) seem to be more specific in terms of co-expression with LAMB1 in EPCs and PCs in GBMs (Fig. A2 A).
Regarding LAMB2, there has not been extensive investigation of its genetic regulation. Here, by drawing insights from prior findings and our recent single-cell analysis, it becomes evident that the regulatory mechanisms governing LAMB2 show high complexity. Despite the very similar primary sequence of LAMB1 and LAMB2 genes, molecular and single-cell expression analysis shows a little overlap in direct mechanisms of their transcriptional control (Fig. A2 B). It is therefore likely that a fine-tuning of specific programs requires more complex expression hubs and suggests diverse ways of regulation.
Our group has previously demonstrated that a downregulation of expression of LAMB1 slows down a growth of GBM in a rodent model, 34 while small-interfering RNA (siRNA) is also arrested at G1 phase, a progression of cell cycle of glioma cells in vitro. 21 As a possible mechanism, a direct targeting of LAMB1 on glioma cells was proposed. Since glioma cells can express LAMB1 both in vivo and ex vivo, their targeting by inhibitory siRNA and by nano-conjugates can be of a potential benefit. However, a specific mechanistic explanation on how LAMB1 can directly promote growth of individual tumor cells is uncertain. In this study, we detect an IHC evidence of LAMB1 and 2 expression by glioma cells only in a small fraction of GBMs, most of which histologically meet the criteria of GBM with mesenchymal phenotype. Moreover, somewhat unexpectedly the analysis of LAMB1 overexpression across the variants of GBM showed a preferencing enrichment of LAMB1 in proneural subtype. 21 In addition, LAMB1 and 2 were also expressed in glial cells of pleomorphic xanthoastrocytoma, a tumor initially thought to be derived from pial astrocytes. In the vast majority of gliomas, LAMB was detected only around vasculature by IHC. By ISH we did not detect significant LAMB ISH signals in the most common parenchymal cells of the normal brain (except for glia limitans), in LGGs, stroke, after bevacizumab, or postradiation treatment. However, glial cells of GBM showed substantial, albeit 8–40 times lower compared with the adjacent vasculature, labeling. No labeling was seen with scrambled probes. The origin of this phenomenon is uncertain. It might be related to exocytosis of mRNA from vasculature to gliomas or non-specific event at perhaps amplification step of the labeling protocol.
As expected for trimeric proteins, the laminins are assembled intracellularly, first as ab dimers, to which g chains are added, allowing extracellular export. 53 It is therefore likely that the expression of a trimer is controlled at intracellular (transcription/translation) rather than extracellular (such as chain-specific degradation) mechanisms. However, the precise mechanisms that control transcription, translation, intracellular trafficking, and deposition of laminins are insufficiently studied.
There are at least two possible explanations for the inhibitory effect of anti-LAMB1 siRNA and nano-conjugates on the gliomas. The first resembles effect of bevacizumab, wherein a suppression of LAMB1 expression inhibits angiogenesis. Better survival of the animals thus can be due to a difference in MV. Perhaps, new vessels formed in the absence of LAMB1 express relatively more LAMB2, which helps to decrease vascular permeability and diminish noxious effects of cerebral edema. An alternative possibility, wherein nano-conjugates directly deplete LAMB1 in the glioma cells, could involve more indirect mechanisms, such as an inhibition of extracellular matrix–dependent survival or decreased tumor cells migration.
Differential roles of laminin chains have been previously established in respect to normal development (for review, see Hallmann et al. 24 and Miner and Yurchenco 54 ), inflammatory responses, and tumorigenesis. The most common heritable condition involving LAM is Pierson syndrome, characterized by nephrosis (with proteinuria) and eye abnormalities (microcoria). 55 It is caused by pathogenic mutations in LAMB2. 30 Furthermore, mapping of Pierson syndrome–associated mutations identified their clustering in N-terminal domain (reviewed in Matejas et al. 56 and Shaw et al. 57 ), which is responsible for formation of hexagonal laminin lattice in BMs. 58 Kidney glomeruli in LAMB2 knock-out animal model show no apparent morphologic defects but are highly permeable to proteins. Importantly, the proteinuria occurs despite a replacement of a genetically missing LAMB2 to LAMB1, indicating that LAMB2 confers to the glomerular BM a much better barrier function. 28
Mice homozygous null for the laminin β2 and γ3 chains exhibit brain cortical laminar disorganization with a phenotype reconstituting cobblestone lissencephaly. This is mediated through a failed deposition of LAM in the pial layer with subsequent failure of attachment for Cajal-Retzius and radial glial cells. 59 In retina, LAMB2 helps to recruit and wrap astrocyte around MV. 60 LAMB2 and/or LAMC3-deficient retinas showed leaky capillaries. 60 Retinal astrocytes can themselves secrete LAMB2 in vitro. Interestingly, a typical star-burst appearance of reactive astrocytes was lost in LAMB2-ablated retinas resulting in collapsed disorganized processes. 60 Intriguingly, similarly appearing flag-shaped astrocytes is a feature of glioblastoma. Depletion of LAMB2 also causes a profound dysmorphogenic effect on the development of synapses in the retina. 61 In kidney, the glomerular mesangial cells also require laminin 421 but not 411 for their migration. 62 LAMB2 containing pericellular matrix can profoundly change expression pattern of pancreatic islet cells promoting maturation toward insulin-expressing beta cells. 63
In the animal model of multiple sclerosis, the experimental autoimmune encephalomyelitis, an inflamed MV replaces LAMA5 to LAMA4. This is associated with acquisition of perivascular lymphoid cuffs. 25 Similar role of LAMA4 was described in renal cell carcinomas, wherein the protein increased migration and inhibited cohesion. 64
In addition to gliomas, other neoplastic processes selectively engage various laminins to promote tumorigenesis. In meta-analysis of gene expression of carcinomas of stomach, an expression of LAMB1 was associated with increased proliferation, invasion, and migration with resulting poor clinical outcome. Correspondingly, LAMB1 knockdown decreased aggressiveness of gastric cancer cells in vitro. 50
Our data give a glimpse to a novel facet of extracellular matrix in dynamic processes of neoangiogenesis. It is well established that ECM creates a structural niche, which restricts boundaries of cell, retains growth factors, thus nourishing the cell and providing a tonic support to its basic functions, but also limiting the cell’s exposure to only a specific set of growth signals. This can be summarized as “segregate and nourish.” Our data indicate that modulation of the composition of ECM modifies permeability and perhaps cell motility. Moreover, modulation of ECM composition produces lasting functional effect, wherein ECM alone can modify a tissue function irrespective of which specific cell type was involved in its secretion. As an example, LAMB2 is normally expressed by perivascular cells and appears to increase microvascular stability/impermeability. After PC or mural cell deposits LAMB2 in BM, the focal presence of PC is no longer needed, and vessel maintains a non-leaky state until LAMB2 is degraded.
Regarding angiogenesis, our data lend a support to the following simplified model, which incorporates the roles LAM in neovascularization and subsequent maturation. Initially, VEGF (secreted by glioma) drives proliferation of ECs with coinciding activation of stromalysis. Expression of LAMA4 and LAMB1 creates a porous BM conducive for ECs budding. Following an initial burst of proliferation, the ECs start to secrete PDGF, which recruits PCs and activates their proliferation. PCs secrete LAMB2, which restricts vascular permeability. It is possible that LAMB2 containing matrix promotes PC differentiation, improves contraction, and helps to prune endothelial buds with subsequent smoothening of outlines of neovasculature. Relatively impermeable LAMB2-containing BM might also restrict an access of EC to glioma-derived VEGF, thus limiting ECs proliferation, “cooling off” their environment and promoting maturation. In conditions of either excess of VEGF or failure to deposit LAMB2, the vasculature becomes hyperplastic with multiple layers of EC.
It is unclear as to why LAMB2 deposition is inhibited in a large fraction the GBMs. One can envision a possibility of a physical interference of proliferating ECs on a presumably limited number of PCs capable of LAMB2 production. ISH and single-cell data show that both LAMB1 and LAMB2 are expressed in the vast majority if not all microvascular proliferating cells in GBM. Alternatively, perhaps an assembly of LAM trimer containing LAMB1 is more effective/stable as compared to the one with LAMB2. LAMB2 might be more vulnerable to intracellular or extracellular degradation since our data show a relative abundance of LAMB2 mRNA in GBMs in comparison with depletion of extracellular LAMB2 protein in BM of GBM.
In conclusion, here we present a correlative evidence of an involvement of LAMB1 in the development of microvascular bed. In addition, we find a correlation between a loss or disruption of microvascular LAMB2 in various brain conditions with increased BBB permeability or increased PGE. We recognize limitations of our work and that functional studies are needed for more direct estimation of a magnitude of involvement of LAMB2 in BBB. In particular, no specific brain-related structural or physiologic abnormalities are found in patients with Pierson syndrome or in LAMB2 knock-out mice model. Given a critical role of CNS in survival of vertebrates, we reason that loss of LAMB2 function in the brain is substituted by upregulation of alternative mechanisms involved in BBB maintenance.
All mice were used in accordance with the Cedars-Sinai Institutional Animal Care and Use Committee. Embryonic day (E) 0.5 was established as the day of vaginal plug. Wild-type CD1 mice were provided by Charles River Laboratories. Gt(ROSA)26Sortm4(ACTB-tdTomato,-EGFP)Luo/J and Gt(ROSA)26Sortm1.1(CAG-EGFP)Fsh/Mmjax mice (JAX Mice) were bred with wild-type CD1 mice (Charles River) or C57BL/6J mice to generate heterozygous mice. Male and female embryos between E12.5 and E15.5 were used for the in utero electroporations and pups between postnatal day (P) 0 and P21 for the postnatal experiments. Pregnant dams were kept in single cages and pups were kept with their mothers until P21, in the institutional animal facility under standard 12:12 h light/dark cycles.
Further information and requests for resources and reagents should be directed to and will be fulfilled by the Lead Contact, Serguei I. Bannykh (serguei.bannykh@cshs.org).
Supplemental Material
sj-xlsx-1-jhc-10.1369_00221554241281896 – Supplemental material for Laminin Beta 2 Is Localized at the Sites of Blood–Brain Barrier and Its Disruption Is Associated With Increased Vascular Permeability, Histochemical, and Transcriptomic Study
Supplemental material, sj-xlsx-1-jhc-10.1369_00221554241281896 for Laminin Beta 2 Is Localized at the Sites of Blood–Brain Barrier and Its Disruption Is Associated With Increased Vascular Permeability, Histochemical, and Transcriptomic Study by Katherine S. Bannykh, Antonio C. Fuentes-Fayos, Paul W. Linesch, Joshua J. Breunig and Serguei I. Bannykh in Journal of Histochemistry & Cytochemistry
Footnotes
Appendix
Acknowledgements
The authors would like to thank Dr. Xuemo Fan, Department of Pathology and Laboratory Medicine, Cedars-Sinai Medical Center for his contribution in evaluation of histology and IHC staining, Dr. Alexander Ljubimov and Dr. Julia Ljubimova for help with IHC protocol and selection of antibodies, Fai Chung and Evelyn Kim for validation and supervision of IHC staining. The authors also acknowledge Neurosurgical and Neuro-Oncologic teams for patient recruitment and follow-up. They are grateful to the patients and to the support staff at the Cedars-Sinai Medical Center.
Authors’ Note
The study was presented in part at the 34th European Congress of Pathology, September 6, 2022, Basel, Switzerland.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
The authors have contributed to this article as follows: conceptualization (SIB), methodology (SIB, JJB), formal analysis (SIB, KSB), single-cell analyses (ACFF), investigation (SIB, KSB), technical assistance (KSB), writing—original draft (SIB), writing—review and editing (SIB, KSB, JJB, ACFF, PL), critical review of the final manuscript (SIB, KSB, JJB, ACFF, PL), resources (SIB, JJB), funding acquisition (SIB, JJB). All authors have read and agreed to the order of appearance and of the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article from a mini-grant by the Department of Pathology and Laboratory Medicine, Cedars-Sinai Medical Center. The authors were also supported by DoD grant HT9425-23-1-0269, NIH grant R33CA236687, American Cancer Society grant RSG-16-217-01-TBG. They acknowledge support from the Board of Governors RMI of Cedars-Sinai (to JJB).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
