Abstract
During tooth development, the growth and differentiation of ameloblast lineage (AL) cells are regulated by epithelial–mesenchymal interactions. To examine the dynamic effects of components of the basement membrane, which is the extracellular matrix (ECM) lying between the epithelium and mesenchyme, we prepared AL cells from the epithelial layer sheet of mandibular incisors of postnatal day 7 rats and cultured them on plates coated with type IV collagen, laminin-1, or fibronectin. The growth of AL cells was supported by type IV collagen and fibronectin but not by laminin-1 in comparison with that on type I collagen as a reference. Clustering and differentiation of AL cells were observed on all matrices examined. AL cells showed normal growth and differentiation at low cell density on fibronectin but not on type I collagen. Furthermore, the population of cytokeratin 14-positive cells on fibronectin was lower than that on other ECM components, suggesting that fibronectin may be a modulator to accelerate the differentiation of AL cells. After the cells had been cultured for 9 days on fibronectin, crystal-like structures were observed. These structures overlaid the cell clusters and were positive for von Kossa staining. These findings indicate that each matrix component has a regulative role in the proliferation and differentiation of AL cells and that fibronectin causes the greatest acceleration of AL cell differentiation.
Keywords
T
AL cells used in this study were prepared from the mandibular incisors of newborn rats and were cultured in complete MCDB medium without serum. These cells show three stages of differentiation under these culture conditions. (a) In the early period, almost all cells are scattered and proliferate well (Matsumura et al. 1998), and these scattered cells express c-Met (HGF receptor), a marker molecule for the oral epithelium (Tabata et al. 1996a, b). (b) In the middle period, the cells gather together to assume a paving stone-like appearance. These clustered cells cease proliferation and express cytokeratin 14 (K14), a marker for the tooth germ epithelium (Tabata et al. 1996b). (c) In the last period, the shape of cells in the center of the clusters changes, with the cells becoming tall (Tabata et al. 1996b). These tall cells are positive for En3 antibody, which recognizes amelogenin, an enamel protein secreted from ameloblasts (Inai et al. 1991; Kukita et al. 1992; Tabata et al. 1996b). The characteristics of each stage are summarized in Table 1. Although this is a primary culture system, when we mixed AL cells with dental papilla cells on purpose, such as 10:0, 9:1, 7:3, 5:5, and 0:10, there was no proliferation of dental papilla cells (Tabata et al. 2000). Therefore, we used AL cells in this culture system for the current study.
Materials and Methods
Primary Culture of AL Cells
Sprague-Dawley (SD) rats were purchased from Nihon Dobutsu (Osaka, Japan). The ameloblast-rich cell layer was prepared from the labial side of mandibular incisors of SD rats of postnatal day 7 (P7) according to Kukita et al. (1992). The cell layer was treated twice with 0.25% collagenase in Dulbecco's modified Eagle's medium at 37C for 20 min each time and then with 0.25% trypsin in phosphate-buffered saline for 10 min at 37C (Tabata et al. 1996a, b). Almost all the cells could be dispersed by this method, and the total cell viability was usually 97.5–99.8%. The cells were suspended in “complete” MCDB 153 medium, which is MCDB 153 (Kyokuto; Tokyo, Japan) supplemented with 5 μg/ml insulin, 0.1 mM phosphoetanolamine, 0.1 mM ethanolamine, 1.4 μM hydrocortisone, 0.07 mM CaCl2, and 140 μg/ml bovine pituitary extract (Kyokuto). The cells were inoculated into wells of a 96-well plate at a concentration of 1 × 104 cells/100 μl per well for normal density cultures, 1.2 × 104 cells/100 μl per well for high cell density cultures, and 1.5 × 103 cells/100 μl per well for low cell density cultures. The day of inoculation was designated day 0. The cultures were maintained by changing the medium every 2 days and were incubated in an atmosphere of 5% CO2 in air at 37C.
Preparation of Matrix-coated Plates
Type I collagen (derived from bovine dermis, pH 3.0, #1PC-05; Koken, Tokyo, Japan) and type IV collagen (derived from bovine dermis, pH 3.0, #CL-04; Koken) were individually diluted in 1 N HCl to a concentration of 100 μg/ml. These collagen solutions were put into separate wells of 96-well plates (#430247; Corning Science Laboratories, Corning, NY) at 50 μl per well, and the plates were then incubated for 30 min at room temperature (RT). After removal of the solution, the plates were dried, rinsed with sterilized PBS, and used to culture AL cells. Fibronectin (derived from bovine plasma, pH 7.4, #FN-01; Koken) and laminin-1 (derived from mouse EHS sarcoma, #LAM-00; Koken) were individually diluted with PBS to 20 μg/ml and put into separate wells of 96-well plates (#430247; Corning) at 50 μl per well. After an overnight incubation at 4C, the solution was removed and the plates were dried, rinsed with sterilized PBS, and used to culture AL cells.
Summary of the differentiation of ameloblast lineage cells in culture

a Tabata et al. (1996b) for the marker expression; Matsumura et al. (1998) for the BrdU index.
bObserved in later period of culture.
Crystal Violet Staining and Densitophotometry
For convenient measurement of cell numbers in multiple experiments, densitophotometry after crystal violet staining was performed instead of manual cell counting or cell labeling with a radioisotope. Cells in 96-well plates were rinsed with PBS and treated with 50 μl/well of 0.4% crystal violet (CV) in methanol for 30 min at RT (Saotome et al. 1989), after which the plates were rinsed with water to remove the excess dye and then dried. The absorbance of each well of the 96-well plate was measured at 550 nm at once with a microplate reader (model 450, Bio-Rad Laboratories, Richmond, CA). The number of cells in an area of 100 mm2 that were positive for CV staining was also counted with the aid of a micrometer grid attached to a microscope, and the cell number (n cells/well) was estimated. The relationship between CV staining (A550) and cell number (n) of AL cells was linear in 96 trials. The regression equation calculated by Excel software (Microsoft Japan; Tokyo, Japan) was as follows:
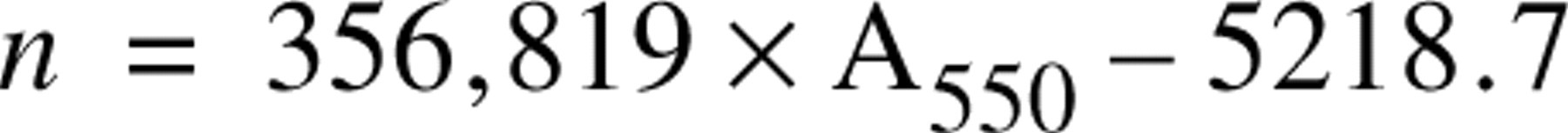
Immunostaining and Densitophotometry
To measure conveniently the cell differentiation rate in multiple experiments, we performed densitophotometry after immunostaining for each marker instead of manual cell counting after immunostaining. The cells in 96-well plates were fixed with 50 μl of neutralized buffered 10% formalin per well for 30 min and used for immunohistochemical analysis. Immunostaining using the biotin-streptavidin system and β-galactosidase reaction was performed as described previously (Tabata et al. 1996a). Color was generated with 5-bromo-4-chloro-3-indolyl-β-
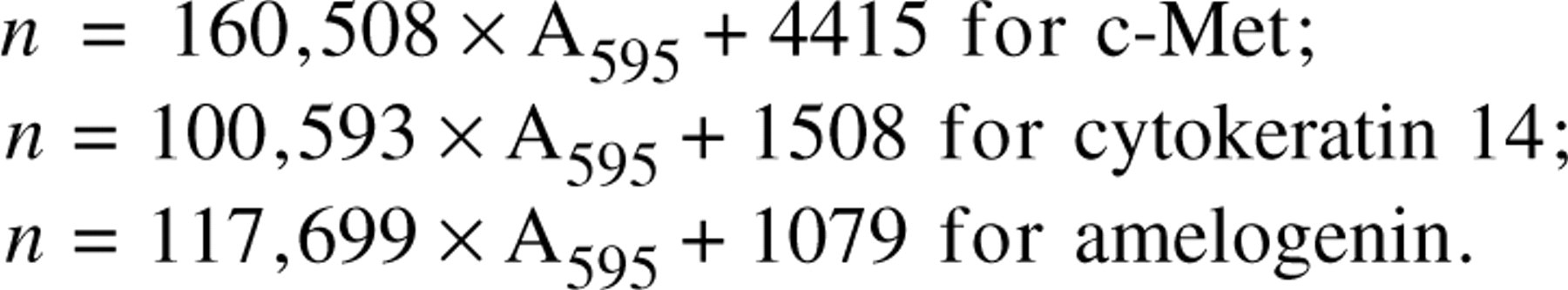
Antibodies used for immunocytochemical staining of AL cells a
aData from Tabata et al. (1996b) and Matsumura et al. (1998).
Results
Cell Morphology and Differentiation
To examine the effect of the ECM components on cell differentiation, we cultured AL cells on type I collagen, fibronectin, type IV collagen, or laminin-1 and examined them microscopically after immunostaining. AL cells prepared from mandibular incisors of P7 rats were inoculated at a cell density of 1.2 × 104 cells/100 μl/well into 96-well plates and cultured for 5 days. On type I collagen, which was used as the control ECM component, AL cells scattered well, starting at day 0. These scattered cells were immunopositive for c-Met (Figure 1A), and some of them began to cluster together to make small colonies from day 1. These clustered cells were immunopositive for cytokeratin 14 (Figure 1B). These colonies grew as sheets with a paving stone-like appearance during the culture period. Finally, the cells at the center of the colonies showed differentiation to cells with a tall shape. These tall cells were immunopositive for amelogenin (Figure 1C). Thus, the AL cells showed three differentiation states: scattering, clustering, and tall.
In the case of the other ECM components, the AL cells differentiated as they did on type I collagen except that they showed some individual properties. On fibronectin, AL cells seemed to proliferate well and to scatter better than on type I collagen (Figure 1D). The cell clustering and tall characteristics appeared earlier on fibronectin than on the other ECMs (Figures 1E and 1F). On type IV collagen, the activity of cell scattering seemed to be slightly lower than in the case of type I collagen (Figure 1G), and the size of the clusters was smaller than on type I collagen (Figure 1H). Tall cells were fewer on type IV collagen than on the other ECM components (Figure 1I). On laminin-1, scattering, clusters, and tall cells were observed, but the cell number for each was obviously low (Figures 1J–1L).
Cell Growth and Differentiation
To examine the cell growth on the various ECM components, we determined the total number of AL cells by A550 after CV-staining (solid lines in Figure 2) and also the number of AL cells in each differentiation stage from the A595 after immunostaining for c-Met, cytokeratin 14, and amelogenin (columns in Figure 2). The cells could proliferate on type I collagen, type IV collagen, or fibronectin, but not on laminin-1 (solid lines in Figure 2). In each stage of differentiation, AL cells on type I collagen and type IV collagen showed a similar pattern of cell growth and cell differentiation (Figure 2A and 2C). On fibronectin, the AL cells positive for c-Met were more numerous than those on type I/IV collagens at day 5 (white columns in Figures 2A–2C), and the positivity for cytokeratin 14 was lower than that on type I/IV collagens (shaded columns in Figures 2A–2C). On laminin-1, the total number of AL cells and the number at each stage were lower than on the other substrates (Figure 2D).
Effect of Low Cell Density
Cells generally show their dependency on their microenviroment when cultured at a low cell density. Therefore, AL cells prepared from mandibular incisors of P7 rats were inoculated at a density of 1.5 × 103 cells/100 μl/well and cultured for 11 days. This cell density is one-eighth of that in the first experiment in this study (1.2 × 104 cells/100 μl/well). On type I collagen, AL cells did not show any obvious cell growth for the first 7 days (solid lines in Figure 3A) but they did proceed to differentiate (columns in Figure 3A). On fibronectin, the AL cells showed obvious proliferation after day 3 in culture, and the populations of c-Met-positive and amelogenin-positive cells were larger than those on type I collagen. Interestingly, the number of cytokeratin 14-positive cells was lower than that of amelogenin-positive cells, and this tendency was common on both ECM components (cf. shaded columns and black columns in Figure 3).
Crystal-like Structures
AL cells prepared from mandibular incisors of P7 rats were inoculated at a density of 1.0 × 104 cells/100 μl/well into 96-well plates and cultured for 9 days. Only on the plates that had been coated with fibronectin did extracellular structures positive for von Kossa staining appear (Figure 4). These structures, which were first seen on day 7 and seemed to grow, overlaid the cell clusters and appeared as transparent yellowish crystals under the microscope. The position of the structure was near the center of the cell clusters and was also close to the tall cells positive for amelogenin.
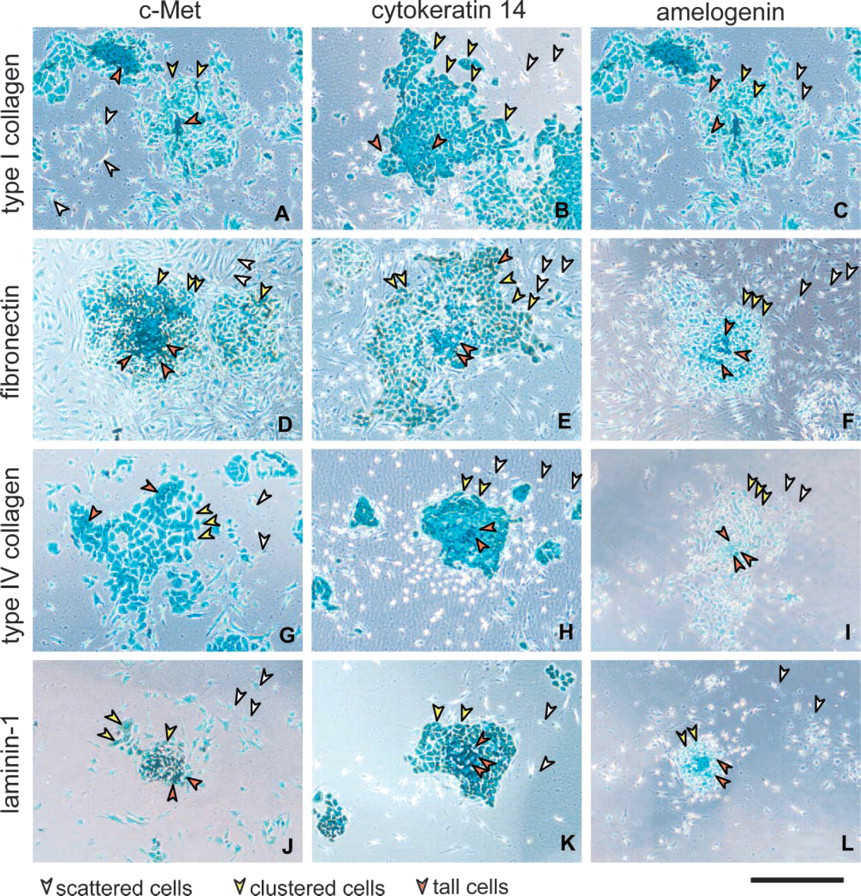
Typical morphology of AL cells on each matrix at day 5 after inoculation for a high cell density (inoculation density 1.2 × 104 cells/well). AL cells cultured on plates coated with type I collagen (
Discussion
Type IV collagen, laminin-1, and fibronectin are components of the BM in tooth germs. Type IV collagen is produced by the tooth germ epithelium and the latter two molecules are synthesized by the tooth germ mesenchyme. In this study, the effect of each of these ECM molecules on growth and differentiation of AL cells was examined. The examination was carried out on primary cultures of AL cells prepared from mandibular incisors of P7 rats, and the results were compared with those obtained for the cells on type I collagen.
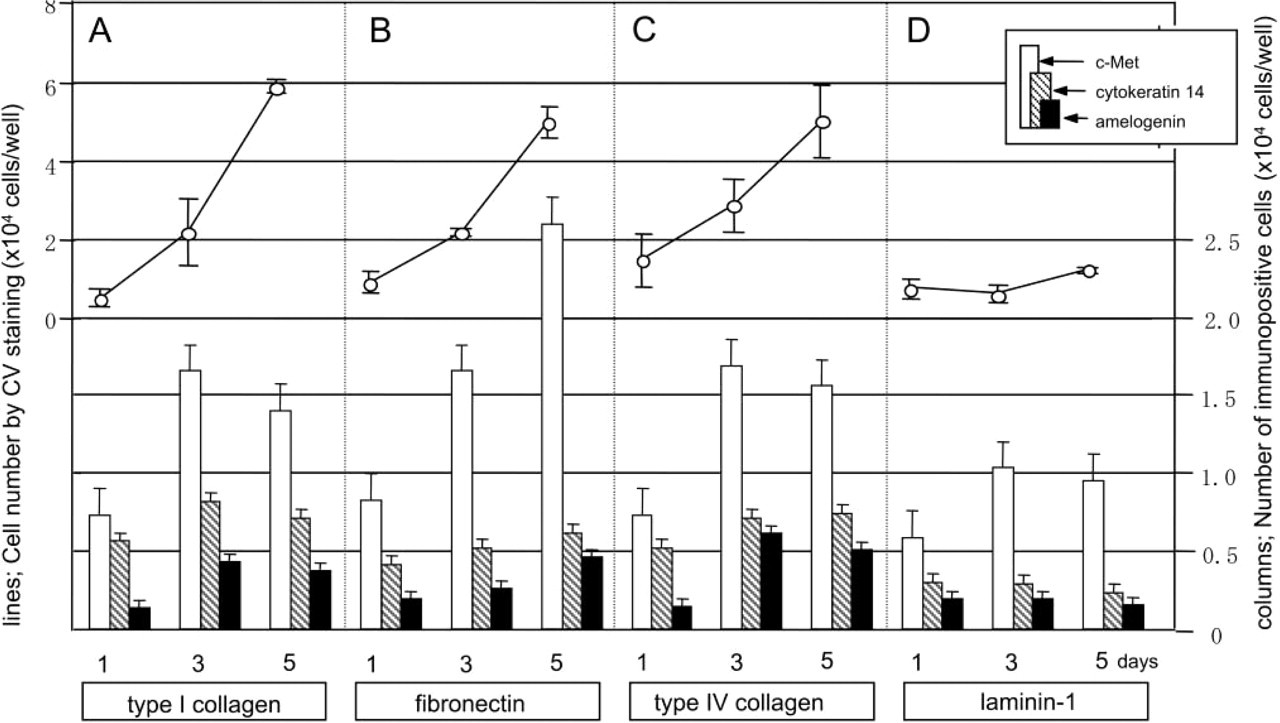
Effect of ECM components on growth (solid lines) and composition (columns) of AL cell population at high cell density (inoculation density 1.2 × 104 cells/well) on each matrix component. Culture plates coated with type I collagen (
Type IV Collagen
Type IV collagen is a BM-specific, network-forming collagen synthesized exclusively by epithelial cells and localized in the BM of tooth germs (Thesleff et al. 1981), whereas type I collagen is the ubiquitous fibrillar collagen synthesized by fibroblasts and is distributed throughout the entire dental papillae and BM of the tooth germ (Lukinmaa et al. 1993; MacNeil et al. 1996; Webb et al. 1998). Because each of these collagens has its individual origin and form, they might be expected to have different functions in tooth morphogenesis. However, AL cells proliferated and differentiated on type IV collagen the same as on type I collagen (Figures 1 and 2). Possible reasons for this similarity of effect are that type IV collagen has a specific function in tooth morphogenesis but at a stage later than the one we examined in this study or that it has the same function as type I collagen in the tooth germ.
Laminin-1
At the onset of cusp formation during tooth development, laminin-1, as well as type IV collagen and BM proteoglycan, is found in the BM (Thesleff et al. 1981). After the first layer of predentin has been secreted by the odontoblasts at the epithelial-mesenchymal interface, laminin-1 remains in close association with the epithelium. Laminin-1 is constructed from sub-units α1β1γ1, and the subunit α1 gene is expressed in the dental papilla during tooth development (Salmivirta et al. 1997). The candidate receptors for laminin-1 are integrins α1β1 (VLA-1), α3β1 (VLA-3), α6β1 (VLA-6), and α6β4. Integrin subunits α6 and β4 are synthesized in the enamel epithelial cells and accumulate mainly in the BM (Salmivirta et al. 1996). These reports indicate that laminin-1 makes some contribution to the growth or differentiation of AL cells before dentin formation. Our result indicates that laminin-1 could have a role in the detachment of ameloblasts from the BM and an inhibitory effect on the growth of AL cells (Figure 1J–L and Figure 2D).
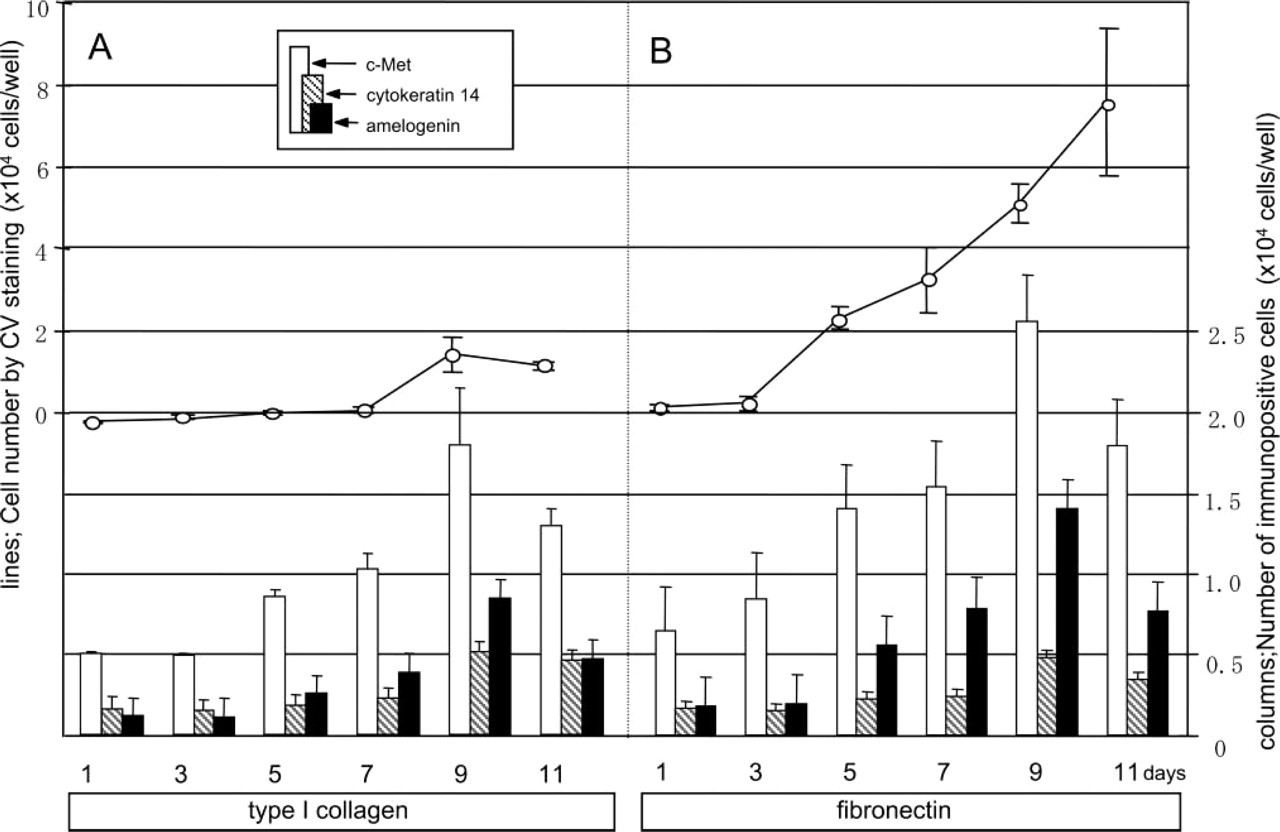
Effect of ECM components on growth (solid lines) and composition (columns) of AL cells at a low cell density (inoculation density 1.5 × 103 cells/well). AL cells were cultured in wells coated with type I collagen (
Fibronectin
In tooth morphogenesis, fibronectin is synthesized in the dental papilla (Thesleff et al. 1987), and this molecule is abundant in the BM and dental papilla (Linde et al. 1982; Laurie et al. 1983). After the beginning of calcification, fibronectin is abundant in the predentin adjacent to the inner enamel epithelium at the region of differentiating odontoblasts (Thesleff et al. 1981, 1991). This region coincides with that in which pre-ameloblasts differentiate into ameloblasts. According to the progress of dentin matrix formation, fibronectin is distributed uniformly throughout this area but is absent from the mineralized dentin (Thesleff et al. 1979). Fibronectin has been also shown a spatial and temporal co-distribution with the first enamel protein, amelogenin (Sawada and Nanci 1995). As the major adhesion receptors for fibronectin, integrin α3β1 (VLA-3), α4β1 (VLA-4), and α5β1 (VLA-5) have been identified. Subunit α4 is localized in both dental papilla cells and inner enamel epithelial cells of the murine incisor tooth germ (Jaspers et al. 1995) and subunit β1 is localized in the BM and dental papilla cells (MacNeil et al. 1996).
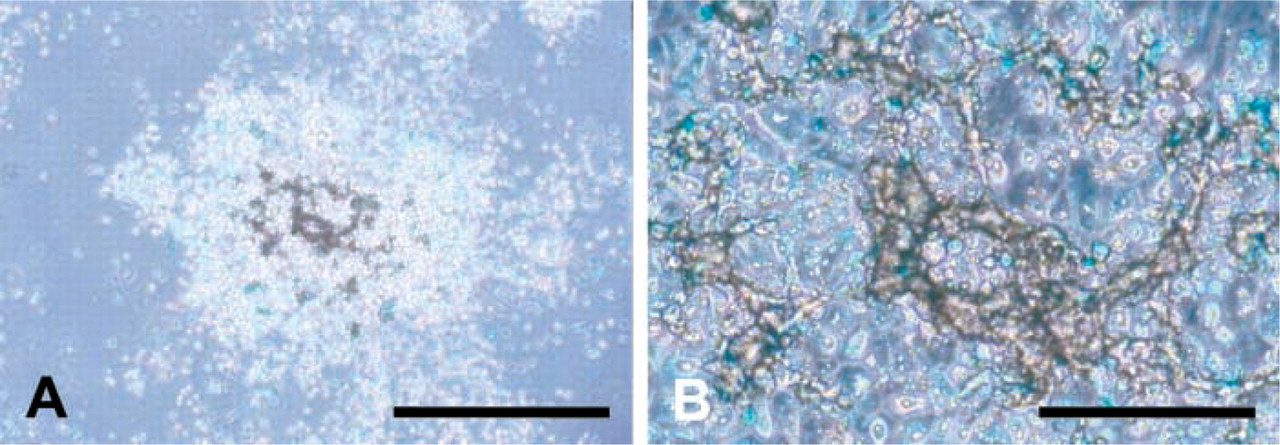
AL cells cultured on fibronectin-coated plates at day 9. Crystal-like structures were positive for von Kossa staining (brown). AL cells were immunostained for amelogenin (light blue). (
Our results show that the proliferation and differentiation of AL cells were accelerated on fibronectin compared with these activities on type I/IV collagens (Figures 1 and 2). This tendency was especially demonstrated by the experiment using low cell density cultures (Figure 3). Above the cell clusters on fibronectin, we found crystal-like structures that were positive for von Kossa staining (Figure 4), which indicates calcification. These structures appeared on the top of the amelogenin-positive cells, suggesting them to be the product of AL cells in vitro, i.e., enamel. However, the structures themselves did not show any obvious immunoreaction for amelogenin (Figure 4B). Therefore, we do not have sufficient evidence to judge whether or not the structure is enamel. Further careful examination is needed to identify it.
The role of fibronectin in tooth development has been proposed to be that of a mediator in ameloblast-mediated terminal differentiation of odontoblasts (Ruch et al. 1995). However, the effects of fibronectin on the attachment, proliferation, and differentiation of AL cells themselves have not been defined. This study has provided the first direct evidence concerning the effect of fibronectin on the differentiation of ameloblasts in murine tooth development.
In conclusion, each ECM component has a regulative role in the proliferation and differentiation of AL cells. The AL cells grew well on fibronectin in high or low cell density cultures, and their differentiation was accelerated on fibronectin. On collagens, AL cells could grow, but not under the low cell density condition. On laminin-1, the cells showed no obvious growth. On fibronectin, we found crystal-like structures overlying the cell clusters at day 9, indicating that AL cells on a fibronectin substratum may be able to produce enamel in our culture system. These results enable us to conclude that AL cells depend on each component of BM during tooth morphogenesis, i.e., on laminin-1 for detachment from the BM, on type IV collagen for their proliferation (but only when the cell density is evenly high), and on fibronectin for acceleration of their differentiation.
Footnotes
Acknowledgements
We thank Dr Sawako Kohara-Takeshita (Seikagaku Kogyo, Japan) and Dr Hidemitsu Harada (Osaka University, Japan) for technical advice.
