Abstract
Basement membranes (BMs) are a dense meshwork of extracellular matrix (ECM) molecules such as Type IV collagen, laminins, Type VII collagen, and heparan sulfate proteoglycans. Type IV collagen is a major structural component of BMs, forming a network that allows the binding of other BM components. The specific biological functions of the diverse Type IV collagen isoforms are not yet well understood. The major form of Type IV collagen is formed by two α1(IV) chains and one α2(IV) chain and is expressed in most BMs. α3-α6(IV) chains have a more restricted tissue distribution. α3(IV) and α4(IV) are co-localized in glomerular BMs, in some tubular BMs, in kidney BMs, in alveolar BMs, and in the synaptic BM at the neuromuscular junction (Mariyama et al. 1994; Lohi et al. 1997). α5 and α6(IV) chains are also co-expressed in skin BMs, in some renal tubular BMs, in smooth muscle cells, and in adipocytes (Ninomiya et al. 1995). Laminins exist in a variety of isoforms, whose biological roles are not yet known. Some laminins are localized in all BMs, whereas others are tissue-specific. For example, laminin 5 (LN5), a component of anchoring filaments, is found in BMs of human lung, skin, mucosa, and intestine (Carter et al. 1991). LN5 and Type VII collagen have been recently characterized as components of the hemidesmosome structures linking epithelial cells and BMs to the underlying connective tissue (Jones et al. 1994).
Many studies have demonstrated that a loss of some BM components due to degradation by proteolytic enzymes and/or a lack of biosynthesis is correlated with tumor progression. For example, the α1 chain of Type IV collagen displays a continuous labeling of the BMs lining the normal tissue, whereas this chain has an irregular distribution around tumor clusters (Polette et al. 1997). In the same way, Type VII collagen is lost early in the development of malignant melanoma (Kirkham et al. 1989), in breast cancers (Wetzels et al. 1991), and in prostate carcinomas (Nagle et al. 1995). The presence of Type VII collagen and α1(IV) chain in tissues may be correlated with a non-invasive status. On the other hand, recent data have shown a de novo synthesis and/or deposition of some BM components, suggesting an active role for BM molecules in tumor invasion. Immunohistochemical studies have reported the presence of α3(IV) chain around some well-differentiated tumor clusters in bronchopulmonary cancers, whereas it is never found in normal bronchi (Polette et al. 1997). Recently, Dehan et al. (1997) have suggested that, in prostate carcinomas, malignant BMs could be characterized by an abnormal deposition of BM components with a possible association of the α5 chain of Type IV collagen with hemidesmosomal structures. In gastric, liver, and pancreatic carcinomas, laminins are produced and deposited at the invasive front of the tumor clusters. The expression of LN1 in gastric carcinoma has been proposed to be a risk factor for liver metastasis. LN5 is expressed by invasive malignant cells in human carcinomas and can be considered a marker of invasiveness (Pyke et al. 1994, 1995; Tani et al. 1996, 1997). All these results suggest that modifications of the BM composition could influence the invasive behavior of tumor cells. On the basis of these data, it appears that the particular distribution and association of some BM components could be an indicator of cancer progression.
To validate this, we examined the expression pattern and localization of the α1, α3, and α5 chains of Type IV collagen, LN5, and Type VII collagen in different types of bronchopulmonary tumors and normal tissues.
Materials and Methods
Sources of Tissues
Tissues were obtained from surgical resection of lung tumors. We used five normal adult bronchi, three normal lungs at a distance from tumors, and 22 primary lung tumors, among which were 11 adenocarcinomas and 11 squamous cell carcinomas (1). In each sample, TNM (tumor node metastasis) staging was done using the international Staging for Lung Cancer of the Union Internationale Contre le Cancer. Histological typing was according to the classification system of the World Health Organization.
Primary Antibodies
In this study we used three monoclonal antibodies (MAbs) against the globular non-collagenous domain (NC1) of α1(IV) (MAb1), α3(PV) (MAb3), and α5(IV) (MAb5) (Wieslab; Lund, Sweden), an MAb (BMS 4040) against laminin 5 (Boehringer Ingelheim; Heidelberg, Germany), and a rabbit polyclonal antibody against Type VII collagen (a kind gift from Drs. M. Chen and D.T. Woodley).
Western Blotting Analyses
The specificity of the antibodies was evaluated by immunoblotting analyses with normal and tumor tissues. Tissue samples were homogenized with a turrax mixer in a lysis buffer (3.4 M NaCl, 50 mM Tris, 20 mM EDTA, 500 ± NEM, 100 ± PMSF). The extracts were centrifuged at 14,000 × g for 20 min at 4C. The pellets were incubated overnight with solution containing 2 M urea and 50 mM Tris and then were centrifuged at 18,000 × g for 20 min at 4C. The supernatants were collected and stored at -20C until use. Amounts of protein were evaluated with a DC Protein Assay kit (Biorad; Hercules, CA). Ten μg of protein from tissue extracts was mixed with Laemmli buffer (Biorad) containing 5% β-mercaptoethanol (v/v), boiled for 5 min, separated on a 12% SDS-PAGE gel, and transferred to a nitrocellulose filter (Hybond-C Extra; Amersham, Aylesbury, UK). Transfer was monitored with Ponceau Red reversible staining. The membrane was blocked with 5% nonfat milk overnight at 4C before exposure with the primary antibodies [anti-α 1(IV), 1 μg/ml; anti-α3(IV), 1 μg/ml; anti-α5(IV), 1 μg/ml; anti-laminin 5, 2 μg/ml; anti-collagen VII, 2 μg/ml] for 2 hr at room temperature (RT). The membranes were incubated with a secondary biotinylated sheep anti-mouse or donkey anti-rabbit antibody (1:1500; Amersham International) for 1 hr at RT and then with the streptavidin-peroxidase complex (1:1500; Amersham) for 30 min. Immunoreactive protein bands were detected with ECL Western blotting reagents (Amersham).
Immunohistochemistry
We performed an immunohistochemical study on different tissues in which localization of these proteins has already been described (Sakai et al. 1986; Rousselle et al. 1991; Dehan et al. 1997; Sado et al. 1998). We found identical staining patterns to those described in the reports cited above: α1(IV) chain (normal bronchi BM), α3(IV) chain (normal pulmonary alveoli), α5(IV) chain (normal prostate BM), laminin 5 (dermal-epidermal junction), and Type VII collagen (dermal-epidermal junction) (not shown).
Fresh samples were frozen in liquid nitrogen, sectioned at 5-8 ± on a cryostat, and collected on gelatin-coated slides. At the first step, slides were incubated with a PBS buffer containing 3% blocking reagent bovine serum albumin (BSA) for 30 min at RT to block nonspecific binding. The sections were incubated for 60 min in a moist chamber with the primary antibodies directed against the α1, α3, and α5(IV) chains (at a concentration of 10 (μg/ml), against laminin 5 (at a concentration of 10 μg/ml), and against Type VII collagen (at a concentration of 10 μg/ml). The sections were incubated with biotinylated anti-mouse or anti-rabbit Ig complex (Amersham) at a 1:50 dilution in a PBS-BSA solution for 60 min and then incubated with streptavidin-fluorescein isothiocyanate (FITC) (Amersham) at a 1:50 dilution in PBS-BSA for 30 min.
The slides were counterstained with Harris's hematoxylin for 10 sec, mounted in Citifluor anti-fading solution (Agar; Stamsted, UK), and observed with an Axiophot microscope (Zeiss; Oberkochen, Germany) using epifluorescence.
Results
Immunobloting Analysis
To characterize the monoclonal and polyclonal antibodies used in this study, we performed Western blotting analyses on well-differentiated carcinomas and on bronchial tissues. We showed that the antibodies directed against the collagens and the laminin recognized only one band at the correct molecular weight, at 180 kD for the chains of Type IV collagen, 200 kD corresponding to the α3 chain of laminin 5, and 240 ∗kD for the Type VII collagen chain (Figure 1). The α1(IV) chain was present in both normal and tumor bronchial tissues, whereas the α3(IV) chain was detected only in the tumor sample. The α5(IV) chain was not detected in tumor tissue extract. These results confirm that the antibodies against NC1 domains of α1(IV), α3(IV), and α5(IV) are specific.
Immunohistochemistry of Normal Bronchi and Lung
All the BMs (epithelial and glandular) of the normal bronchi were strongly stained with MAbs against α1(IV) chain, LN 5, and Type VII collagen, whereas they were totally devoid of labeling for α3(IV) and α5(IV) chains (1) (Figures 2a-2d). LN 5 and α1(IV) and α3(IV) chains were also found in alveolar BMs.α1(IV) and α5(IV) chains were localized both in vascular BMs and in smooth muscle cell BM. None of these molecules was found in the connective tissue underlying bronchial epithelia.
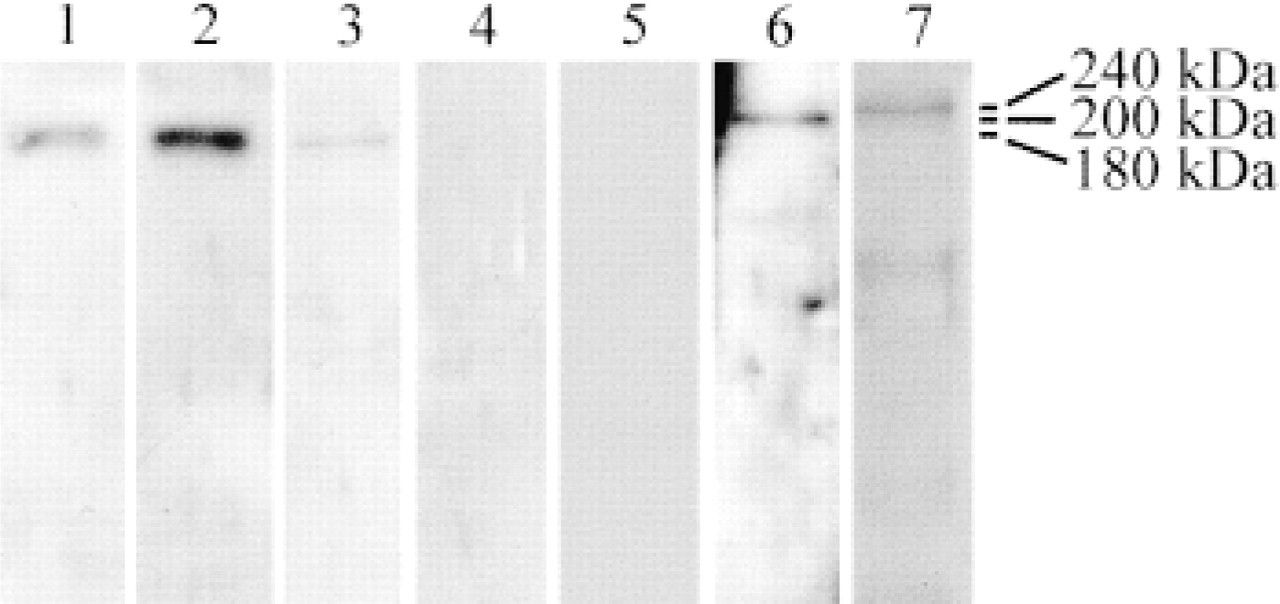
Western blotting analyses of α1(IV), α3(IV), and α5(IV) chains, laminin 5, and Type VII collagen on various tissue extracts. The anti-α1(IV) antibody detects one band at 180 kD both in tumor (Lane 1) and normal (Lane 2) tissue extracts. The α3(IV) antibody recognizes one band at 180 kD in a tumor sample (Lane 3). Normal bronchi (Lane 4) and the tumor tissue extract (Lane 5) are devoid of α3(IV) and α5(IV) chains, respectively. The anti-LN5 (Lane 6) and the anti-Type VII collagen (Lane 7) antibodies detect, respectively, one band at 200 kD and one band at 240 kD in tumor tissue extracts.
Immunohistochemistry of Bronchopulmonary Carcinomas
In this study we found the same expression and distribution of BM molecules in squamous cell carcinomas and adenocarcinomas (1). In these carcinomas, α1(IV) and α5(IV) chains were found in vascular BMs. α1(IV) and α3(IV) chains, and LN 5 antibodies also stained residual alveolar BMs at the periphery of tumors. The tumor BM showed reactivities with α1(IV) chain (in 22/22 tumors) and LN 5 (in 19/22 tumors) antibodies. The α1(IV) chain and LN 5 labelings were co-localized at the interface between tumor cells and stroma. In relation to the degree of tumor differentiation, the linear immunolabeling of BMs was either thin and continuous in well-differentiated carcinoma (Figures 2e and 2g) and disrupted or absent in poorly differentiated carcinoma (Figures 2i and 2k) (1). The α3(IV) chain and Type VII collagen were detected in all well-differentiated carcinomas (Figures 2f and 2h), whereas they were not present in poorly differentiated tumor clusters (Figures 2j and 2l; Table 1). They were localized around well-differentiated tumor cell clusters as a thin continuous labeling (Figures 2f and 2h).
In contrast to their absence in normal bronchial connective tissues, α1(IV) chain (22/22 cancers) and LN 5 (9/22 tumors) were the only BM molecules present in the stromal compartment with a fiber network all around tumor clusters (1).
Concerning the cell labeling, only LN 5 (5/22) and Type VII collagen (8/22) labelings were detected in the cytoplasm of invasive tumor cells close to the BM staining.
Discussion
In this study we examined the particular distribution of some of these macromolecules in relation to tumor progression. We clearly demonstrated here that the redistribution of α1(IV) chain and LN5 and the loss of Type VII collagen and the α3(IV) chain are correlated with invasiveness of tumor cells.
We showed that α1(IV) chain displayed a thin and regular pattern of distribution around well-differentiated tumor clusters, whereas it appeared disrupted or undetectable around poorly differentiated tumor clusters. Such a distribution could reflect the degradation of BM components which has been extensively described during tumor invasion process (Barsky et al. 1983). Of particular interest is the detection of α1(IV) in the stromal compartment. Soini et al. (1993) have demonstrated by in situ hybridization that stromal cells are the principal source of production of α1(IV) chain in lung tumors. The particular distribution of α1(IV) chain could reflect a part of the stromal reaction. Previous studies have demonstrated that interactions between the tumor cells and the ECM could modulate their invasive capacity. More particularly, it has been shown that the α1(IV) 531-543 sequence promotes melanoma cell adhesion and enhances migratory activities through matrigel (Li et al. 1997). In the same way, Chelberg et al. (1990) reported that a Type IV collagen-derived sequence designated as peptide IV-H1 (residues 1269-1277) localized in the triple helical region of α1(IV) chain facilitates adhesion and motility of the M4 melanoma cell line. Therefore, it is possible that after the enzyme-mediated degradation of Type IV collagen BMs, α1(IV) chain sequences in the stroma trigger cell motility and enhance tumor progression.
Distribution of α1(IV), α3(IV), laminin 5, and Type VII collagen in BM and stromal compartment of bronchopulmonary lesions a
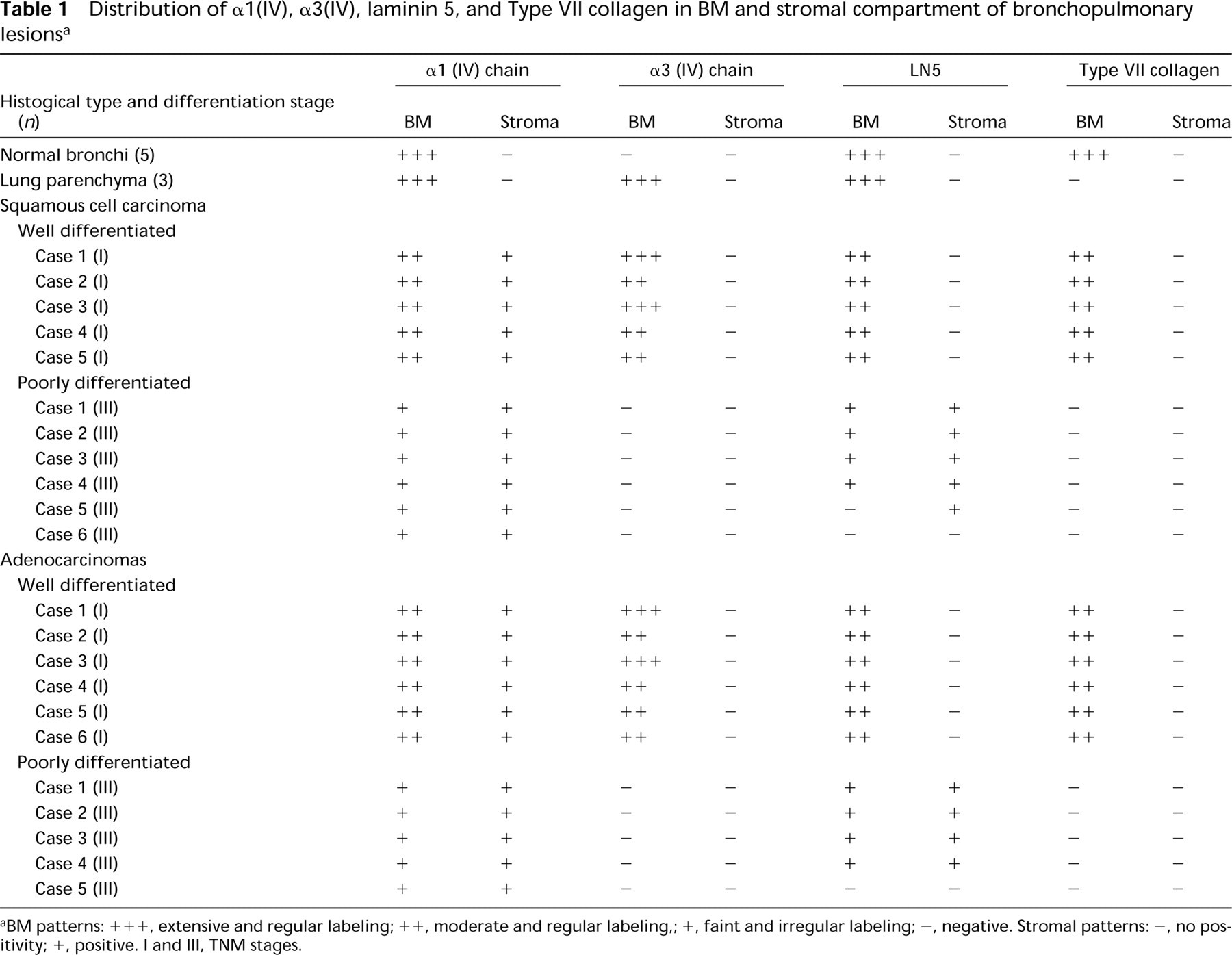
aBM patterns: +++, extensive and regular labeling; ++, moderate and regular labeling,; +, faint and irregular labeling; -, negative. Stromal patterns: , no positivity; +, positive. I and III, TNM stages.
In the same way, LN5 displayed a thin, regular pattern in well-differentiated tumor clusters and a disrupted pattern in poorly differentiated clusters. Likewise, we observed cytoplasmic positivity in cells at the invasive front. Similar results have also shown the overexpression of LN5 in tumor cells at the invasive front, emphasizing its implication in tumor cell invasion of colon carcinomas (Pyke et al. 1994). Moreover, it has been shown that LN5 α2 cleavage by a specific protease (MMP-2) exposes cryptic sites that stimulate cell motility in vitro (Giannelli et al. 1997), suggesting that LN5 may play an important role in tumor progression by increasing the invasive properties of tumor cells. These results suggest a dual function of LN5, which could play a role in cell adhesion as a part of hemidesmosomes but which could also, in contrast, facilitate tumor invasion once cryptic sites are exposed.
These hypotheses strengthened our observations about Type VII collagen distribution because we showed that, in normal epithelia, Type VII collagen staining closely co-distributed with that of LN5. These findings are in agreement with the presence of many hemidesmosomes in bronchial tissue. The anchoring complexes have been well characterized by Rousselle et al. (1997), who have demonstrated that LN5 binds the NC1 domain of Type VII collagen bridging the cell surface to the underlying connective tissue through the hemidesmosomal integrin α6β4. In the same way, Baker et al. (1996) have reported that the α3-subunit of LN5 played a crucial role in maintenance and nucleation of hemidesmosome structural integrity. Therefore, a co-localization of LN5 with Type VII collagen would characterize a cohesive noninvasive state. In contrast, in invasive cancers, we observed that Type VII collagen staining did not co-express with LN5. Similar results have reported that the hemidesmosomal integrin α6β4 and Type VII collagen are not co-localized in bladder cancer compared to normal tissue (Liebert et al. 1994). These redistributions of anchoring complex components are likely to fragilize hemidesmosomal structures and could therefore facilitate the migration of cancer cells in the stromal compartment. In addition, we observed Type VII collagen reactivity in the cytoplasm of some cancer cells, and precisely in tumor cells that edge neoplastic BMs. These results are in agreement with those of Kainulainen et al. (1997), who reported that malignant cells synthesize Type VII collagen at the invasive front. These results suggest that the loss of Type VII collagen around tumor clusters, and consequently the disorganization of hemidesmosomal complexes, therefore reflect an invasive state of bronchopulmonary cancers.
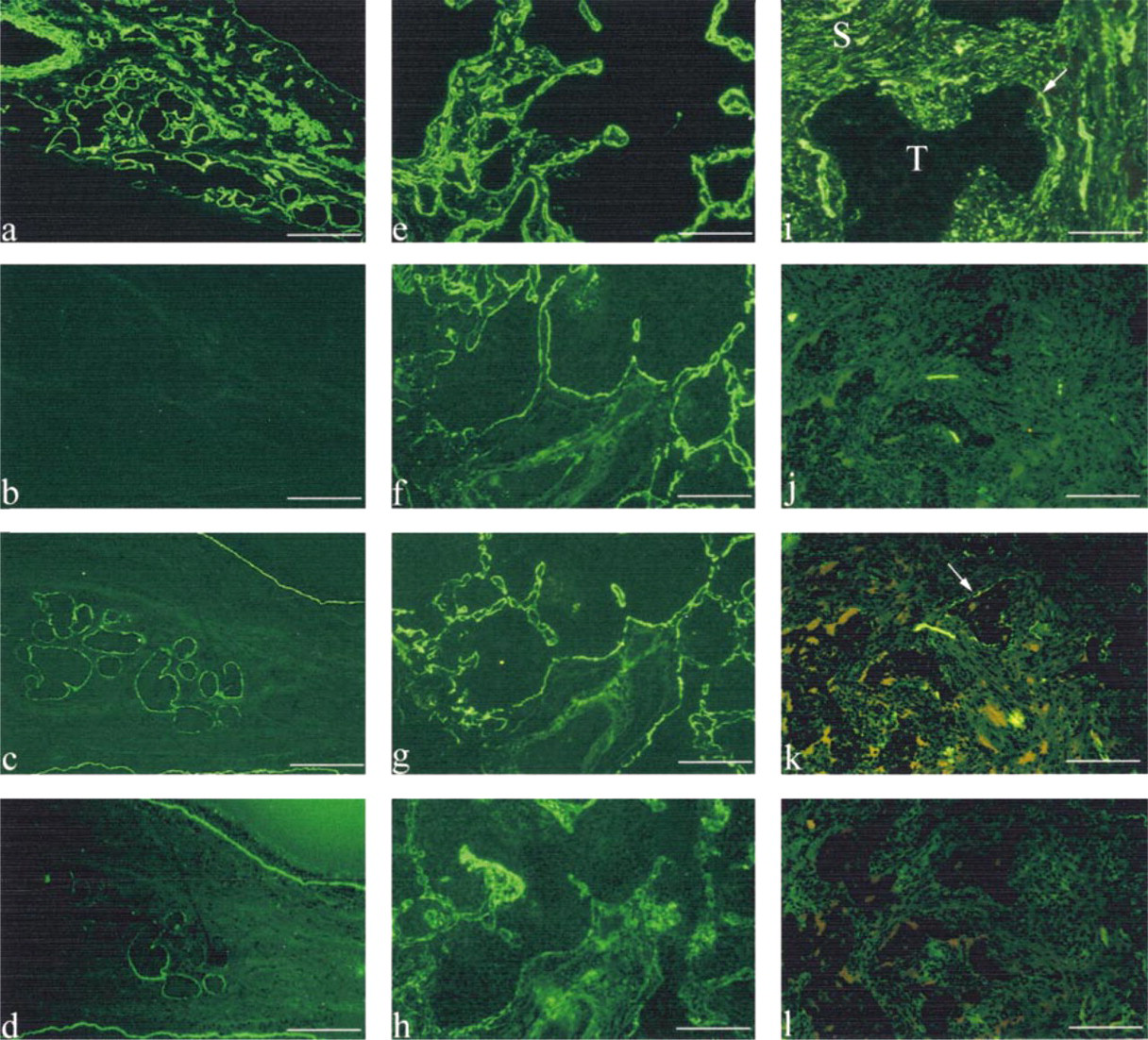
Distribution of α1(IV) and α3(IV) chains, laminin 5, and Type VII collagen in normal bronchi (
Concerning the α3(IV) chain, it was present in some BMs at the interface between tumor clusters and the stromal compartment in bronchopulmonary cancers. Normal bronchial tissues were totally devoid of α3(IV) chain. This observation suggests that this chain is neosynthesized in tumor tissues. Because the α3(IV) chain was detected around well-differentiated tumor clusters, it is likely that the neobiosynthesis of α3(IV) would be implicated in the preinvasive stage of tumor progression. However, at present little is known about the α3(IV) chain function in malignant processes. In vitro experiments suggest that the Type IV collagen α3 chain downregulates activation of human polynuclear leukocytes and thus decreases their capacity to migrate through the capillary walls (Monboisse et al. 1994). More recently, Han et al. (1997) found that the sequence 185-203 within the region of the α3(IV) chain NC1 domain inhibits proliferation of melanoma cells. It can therefore be speculated that α3(IV) chain deposition could be a part of the desmoplastic reaction protecting normal tissues from tumor expansion by inhibiting malignant cell proliferation. Thus, the α3(IV) chain deposited around tumor clusters would limit tumor progression in bronchopulmonary cancers.
In conclusion, we showed an extensive remodeling of BM structures in bronchopulmonary carcinomas, with a progressive disruption of α1(IV) chain and the two hemidesmosomal components laminin 5 and Type VII collagen and the appearance of the α3(IV) chain around well-differentiated tumor clusters. Together, these results emphasize that the composition of BMs reflect different stages of tumor progression and may have prognostic significance in bronchopulmonary cancers.
Footnotes
Acknowledgements
Acknowledgments
Supported by a grant from the ARERS (Association RéAgionale pour l'Enseignement et la Recherche Scientifique) and by the Lions Club of Soissons.
We thank Drs M. Chen and D.T. Woodley (Northwestern University School of Medicine, Chicago, IL) for the generous gift of polyclonal antibody to human Type VII collagen.
