Abstract
Immunohistochemistry was used to study the distribution of laminin (Ln) chains, collagen types IV (α 1/2), VII, and XVIII and Lutheran antigen (Lu) in 36 frozen ovarian carcinoma samples. Surface epithelial basement membrane (BM) of the normal ovary showed immunoreactivity for Ln α1, α3-α5, β1-3, γ1, and γ2 chains and type IV and XVIII collagens. Chains of Ln-5 (α3β3γ2) and Ln-10 (α5β1γ1) as well as type IV and XVIII collagens were found in most tumor BMs, but Ln α2 chain and type VII collagen were detected only in few tumors. Contrary to serous tumors, BMs of mucinous carcinomas showed Ln α4 chain, but not Ln α1 and β2 chains. Ln α1 chain was found in most endometrioid carcinomas, whereas chains of Ln-5 were only moderately detectable in comparison with serous and mucinous carcinomas. In the normal ovary, Lu immunoreactivity was confined to basal aspect in the ovarian epithelial cells, but in tumor specimens Lu immunostainings showed variable polarized and nonpolarized patterns. The results suggest that the three types of ovarian carcinoma have distinct differences in their Ln distribution and can be grouped based on their expression pattern. This suggests that they may have histogenetically different precursors and may help to distinguish these tumors from each other.
It has been considered that the ovarian surface epithelial (OSE) cell layer plays a central role in vitro and in vivo as a precursor for development of ovarian carcinomas (Auersperg et al. 2001; Feeley and Wells 2001). It is not fully clear which factors take part in the tumor development clinically seen as a four main histological tumor types—namely serous, mucinous, endometrioid, and clear cell adenocarcinomas. OSE presents many typical epithelial features, such as cytokeratin filaments, mucin, desmosomes, and apical microvilli, but also simultaneously mesenchymal properties including vimentin filaments and N-cadherin (Auersperg et al. 2002). Based on multipotent features, OSE has been suggested to represent an immature precursor and therefore would be susceptible to mutations (Feeley and Wells 2001; Auersperg et al. 2002). It has also been suggested that, during tumor development, these features allow OSE to express properties related to fallopian tube epithelium (serous tumors), endocervix or colonic epithelium (mucinous tumors), endometrium (endometrioid tumors), or urogenital tract (clear cell tumors) (Van Niekerk et al. 1993, Auersperg et al. 2002). However, it has also suggested that OSE may not fully correspond to all premalignant lesions of the ovary and that the secondary Müllerian system would also have contribution (Dubeau 1999).
In the beginning of the neoplastic process, surface epithelium or inclusion cyst becomes stratified and adenomas are formed (Resta et al. 1993; Scully 1995). At this stage, BM is usually still continuous (Visser et al. 1995). BMs form natural barriers for tumor cell invasion, and, after malignant change, ovarian epithelial BMs often show variable disruptions or even total loss (Kuwashima et al. 1995; Skubitz et al. 1996; Capo-Chichi et al. 2002).
Specific receptors, such as integrins and the dystroglycan glycoprotein complex, mediate cell adhesion to the BM macromolecules (Belkin and Stepp 2000). Previously, it has been shown that α6β4 integrin, which normally mediates adhesion of basal epithelial cells to the underlying BM, is decreased or simultaneously lost with Ln and type IV collagen in tumor nests of invasive ovarian carcinomas (Skubitz et al. 1996). Lutheran blood group glycoprotein (Lu), a member of immunoglobulin superfamily, is a recently described receptor for Ln α5 chain-containing heterotrimers, Ln-10 (α5β1γ1) and Ln-11 (α5β2γ1) (Parsons et al. 1995; Udani et al. 1998). Lu binds to LG3 module of G-domain of the long arm of Ln α5 chain (Parsons et al. 2001; Kikkawa et al. 2002), is widely distributed in normal tissues, and localizes to the basal aspect of the cells. However, a solely nonpolarized distribution has previously been reported in ovarian tumors (Gabin-Chesa et al. 1994; Campbell et al. 1994).
In this study, we wanted to clarify the immunohistochemical profile of BM proteins in different types of ovarian adenocarcinomas and to relate the results to the distribution of these components in the normal OSE. The results show that different ovarian carcinomas differ from each other, which may serve as diagnostic help in distinguishing serous, mucinous, and endometrioid adenocarcinomas from each other.
Materials and Methods
Materials
All the tissue samples were collected from the files of the Department of Pathology, Oulu University Hospital and Department of Obstetrics and Gynecology, Helsinki University Central Hospital between 1990 and 1997. Two frozen sections representing OSE were included. Tumor material included 36 tumor cases: 18 serous cystadenocarcinomas (6 grade I, 7 grade II, and 5 grade III tumors); 8 mucinous cystadenocarcinomas (3 grade I, 3 grade II, and 2 grade III tumors); and 10 endometrioid adenocarcinomas (6 grade I, 2 grade II, and 2 grade III tumors). The diagnoses were based on light microscopy of routine hematoxylin-eosin-stained sections and classified and graded according to the World Health Organization classification. For the study, there was permission from the Ethical Committees of the Hospitals, and the research was in compliance with the tenets of the Declaration of Helsinki for experiments involving human tissue.
Antibodies and Immunohistochemistry
The antibodies employed for this study are presented in Table 1. Frozen samples were fixed in cold acetone for 10 min, and all the steps in the immunoreactivity procedures were followed by washes with PBS. Sections were incubated with fetal calf serum (1:5; Hyclone Laboratories, Inc., Logan, UT) in PBS for 20 min; with primary antibody or, for negative controls, with PBS for 1 hr. Two methods were used: avidin-biotin-peroxidase and alkaline-phosphatase-anti-alkaline-phosphatase. In avidin-biotin method biotinylated secondary antibody (Dako; Glostrup, Denmark) was used for 30 min; avidin-peroxidase complex was used for 30 min. The color was developed with diaminobenzidine tetrahydrochloride-H2O2 (Sigma; St Louis, MO). In the alkaline-phosphatase-anti-alkaline-phosphatase method, the reaction was visualized with a substrate solution containing naphthol-AS-BI-phosphate, levamisole, and new fuchsin (all from Sigma). Finally, all sections were counterstained with hematoxylin.
Evaluation of the Results
The continuity of the BMs around tumor islands was analyzed independently by two observers and, in case of differences in scoring, discussion occurred to reach common conclusion. Scoring was as follows: continuous BMs with minimal breaks when more than 80% of the tumor islands were surrounded by visible BM structures (score 3); semi-continuous BMs, when BMs were seen to surround the tumor islands by 30–79% (score 2); highly discontinuous BMs, when the staining positivity was seen around 5–29% of the tumor clusters (score 1); and focal BMs (score 0), when only short fragmented BM strips were seen in the tumor growth and comprised less than 5% of the growth or the result was totally negative. In scoring, the whole tumor area was examined and, when the amount of BM structures varied, the average result was taken into account.
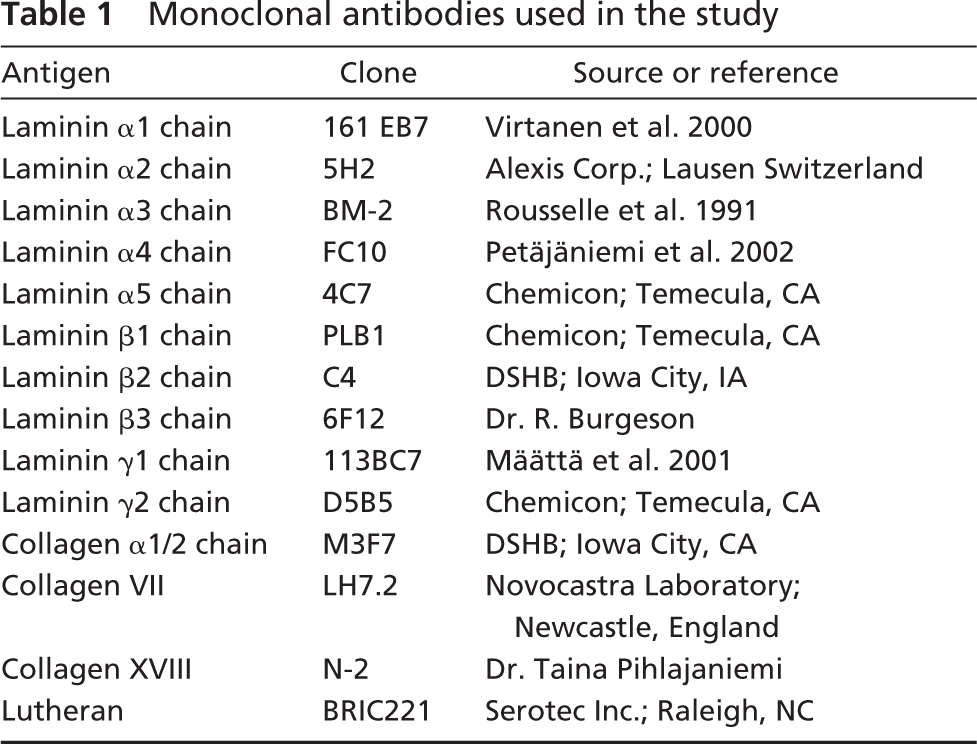
Monoclonal antibodies used in the study
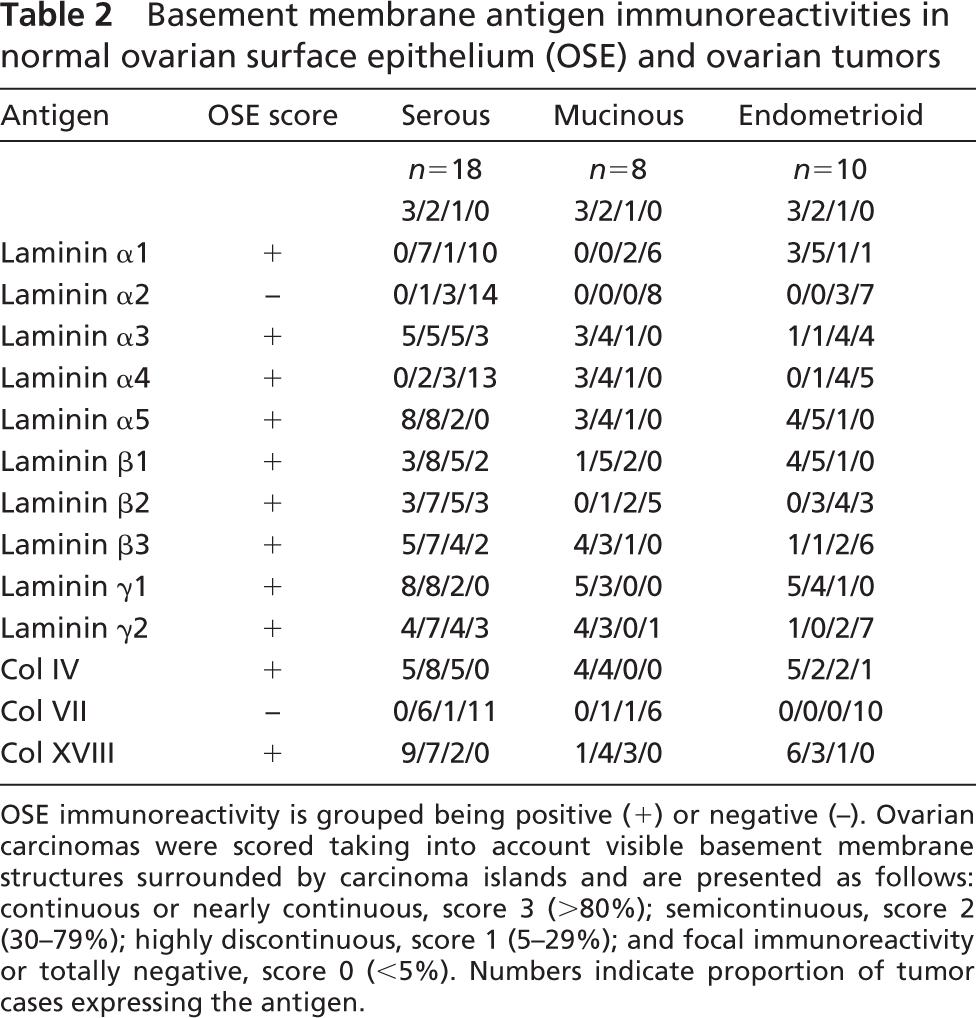
Basement membrane antigen immunoreactivities in normal ovarian surface epithelium (OSE) and ovarian tumors
Results
Normal Ovary
The results are summarized in Table 2. Surface BM zone showed constant immunoreactivity for Ln α1, α3, α4, α5 (Figure 1A), β1-β3, γ1, and γ2 chains (Figure 1B) and for type IV and XVIII collagens, but not for Ln α2 chain or type VII collagen. Lu showed a polarized immunoreactivity in the basal aspect of OSE (Figure 1C). A diffuse stromal reaction was seen for Ln α4 and β1 and β2 chains and for type XVIII collagen, lesser extent for Ln α2 and γ1 chains, but not for Ln α1, α3, and α5, β3 and γ2 chains.
Ovarian Carcinomas
The results are presented in Table 2. Ovarian tumors expressed all the BM antigens, including Ln chains α1-5, β1-3, γ1, 2; and collagen types IV, VII, and XVIII; and Lu glycoprotein. (The number of cases expressing score 1–3 is reported in parentheses in the text.)
Serous Carcinomas
Chains of Lns-10 α5 (Figure 2A), β1 (Figure 2B), and γ1 (Figure 2C); chains of Ln-5 α3 (Figure 2D), β3, and γ2 (Figure 2E); and collagen types IV and XVIII (Figure 2F) were found in BMs of practically all tumors (15/18–18/18). Their immunoreactivities were more constant and linear in grade I tumors than in grade II-III tumors; all except one grade I tumor showed a moderate or high BM linearity according to the scoring, but none of the grade III tumors did. A distinct immunoreactivity in the stroma was occasionally found for Ln β1, β2, and γ2 chains and collagen types IV and XVIII. Interestingly, immunoreactivities for Ln β1 and β2 chains appeared to be reciprocally expressed in the specimens. Ln chains α2 (4/18 cases), α4 (Figure 2G) (5/18 cases), α1 (Figure 2H) (8/18 cases), and collagen type VII (Figure 2I) (7/18 cases) were found only in some of the tumors. In some of the tumors, Ln α2 chain was usually found only as a short, faint strip around tumor cell islands and was also expressed in the stroma. Similarly, Ln α4 chain immunoreactivity was detected, together with tumor BMs, exclusively and diffusely in the tumor stroma per se and endothelial BMs (Figure 2G). Immunoreactivities for Ln α1 chain (Figure 2H) and type VII collagen (Figure 2I) were solely confined to epithelial BM. Type VII collagen immunoreactivity was mainly found in grade I tumors, because only two of the tumors represented grade III tumors.
Mucinous Carcinomas
Ln chains α3, α5, β1 (Figure 3A), β3 (Figure 3B), γ1, and γ2 (Figure 3C) and collagen types IV (Figure 2D) and XVIII were found in BMs of most or all mucinous tumors. Contrary to serous tumors, immunoreactivity for Ln β2 chain (Figure 3E) was detected in BMs of two tumors. Ln α4 chain (Figure 3F) showed a constant immunoreactivity in tumor epithelial BMs (8/8 cases) and was also abundantly found in the tumor stroma per se and in vascular endothelium BMs. Immunoreactivity for Ln α1 (2/8 cases) and α2 (0/8 cases) chains and collagen type VII (2/8 cases) could not be detected in most mucinous carcinomas.
Endometrioid Carcinomas
Ln α5, β1 and γ1 (Figure 4A) chains and collagen types IV and XVIII were found in BMs of most or all tumors. Contrary to serous and mucinous carcinomas the chains of Ln-5 α3 (6/10 cases), β3 (4/10 cases), γ2 (3/10 cases) (Figure 4B) were less present in BMs of this tumor type. Ln α1 (9/10 cases) (Figure 4C) usually showed a linear immunoreactivity in BMs around all tumor islands. Only limited amounts of Ln α2 (3/10 cases) (Figure 4D) and α4 (5/10 cases) chains were found, and immunoreactivity for collagen type VII was practically lacking (0/10) in this tumor type.
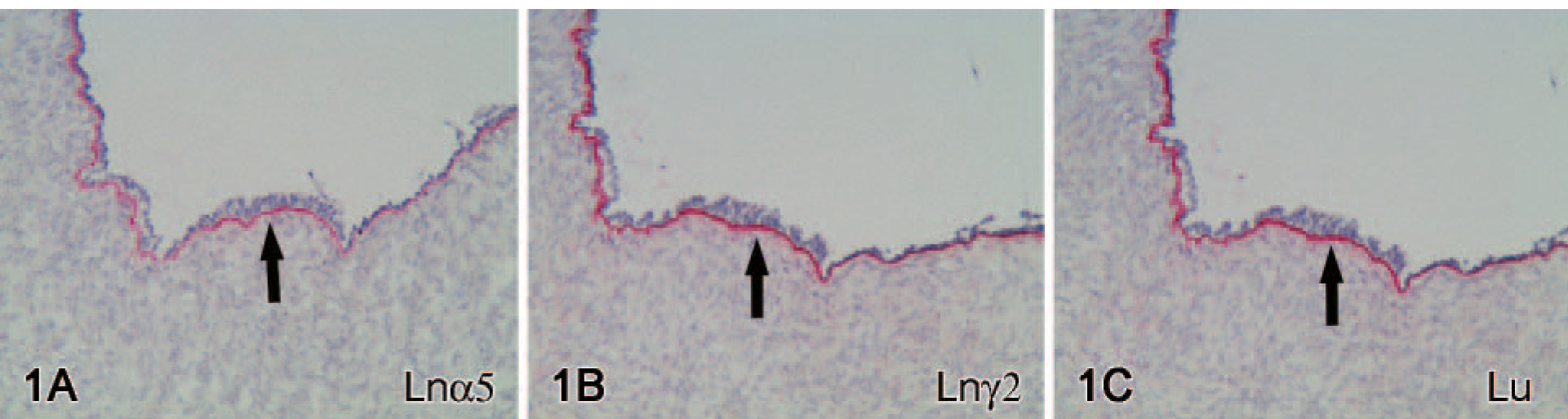
Normal ovarian surface epithelium. Normal ovarian surface epithelial basement membrane (BM) contains immunoreactivity for laminin (Ln) α5
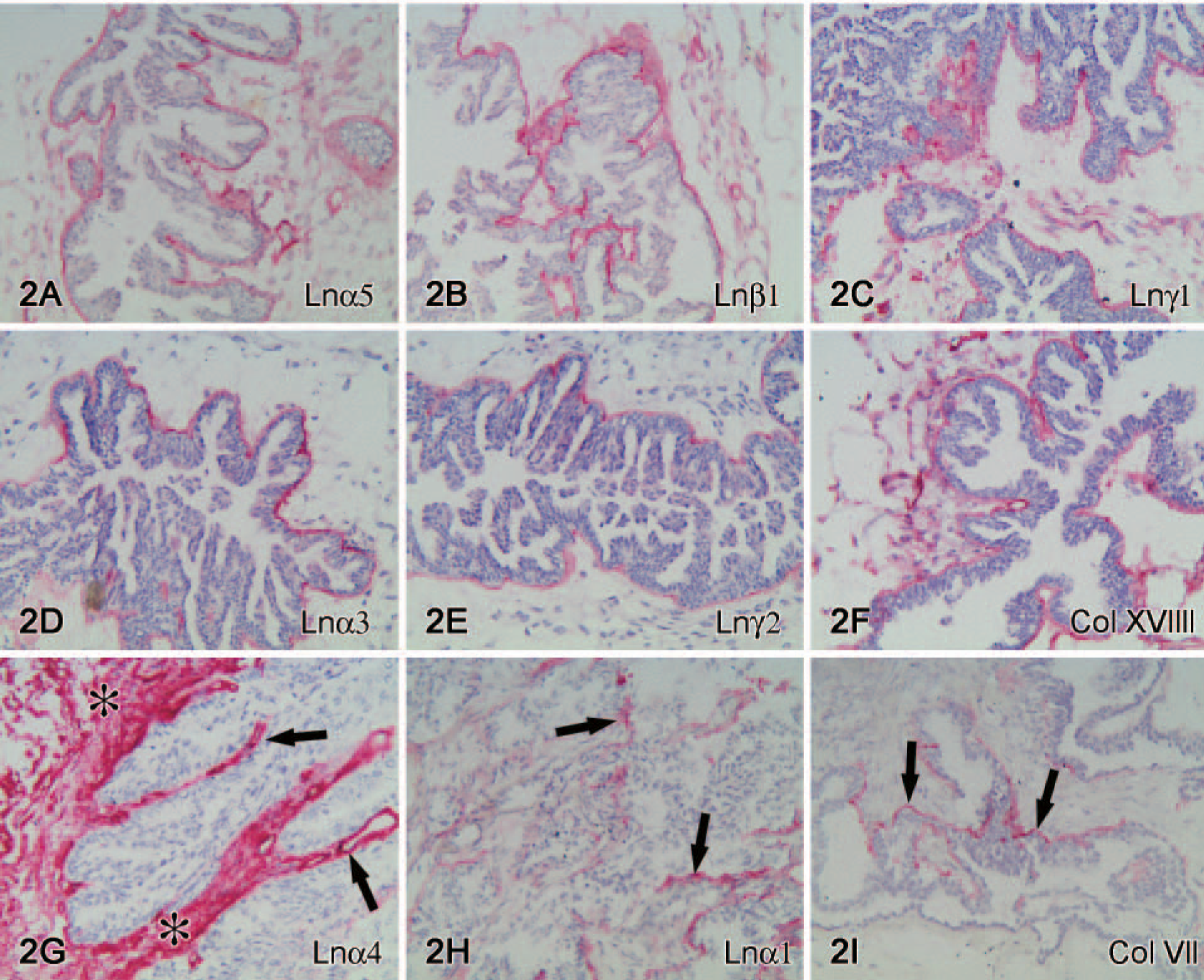
Serous carcinomas. Well-preserved basement membrane (BM) organization (arrows) is seen around tumor islands in grade I serous carcinomas and shows immunoreactivities for Ln α5
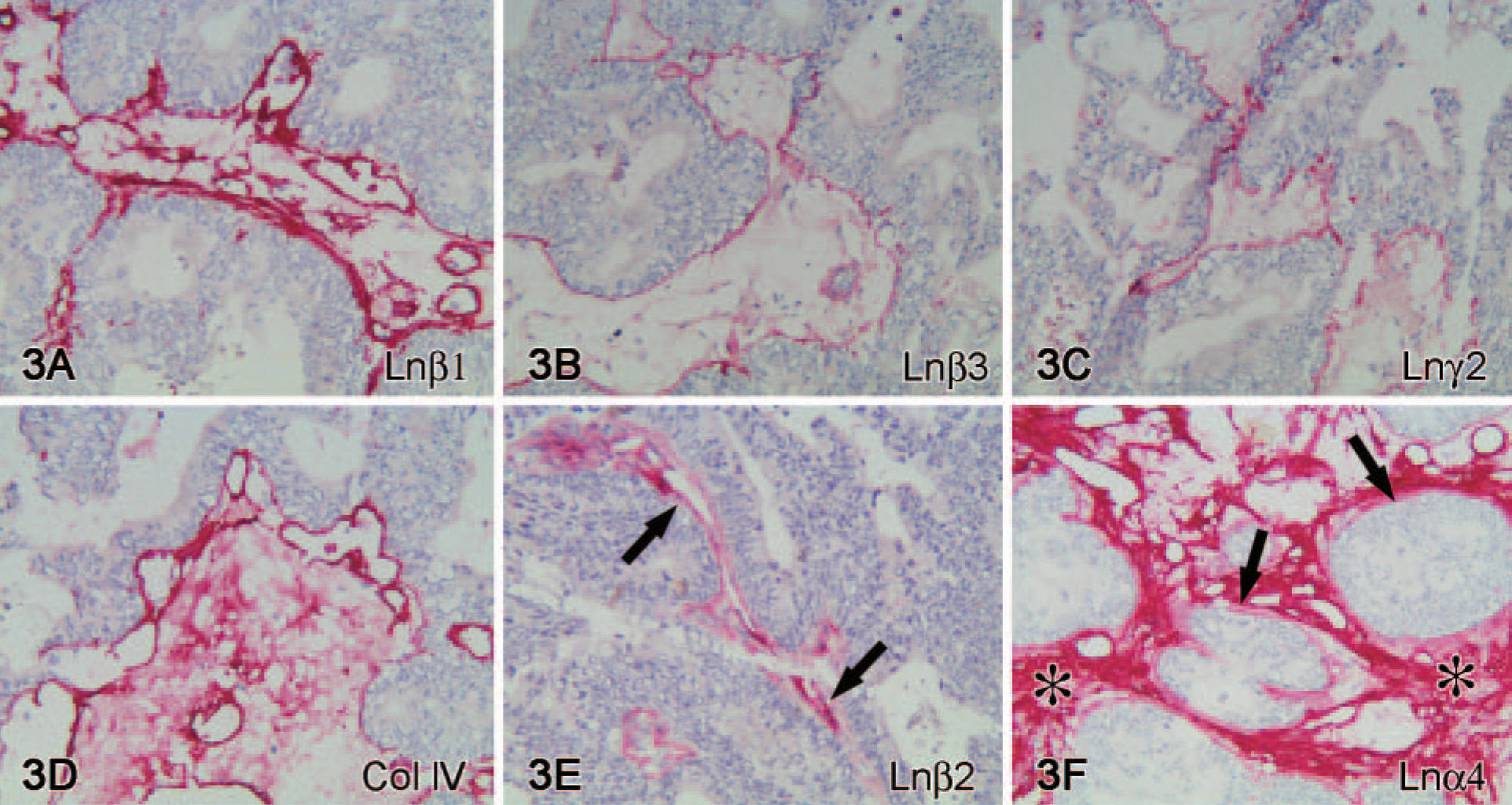
Mucinous carcinomas. Linear immunoreactivity for Ln β1
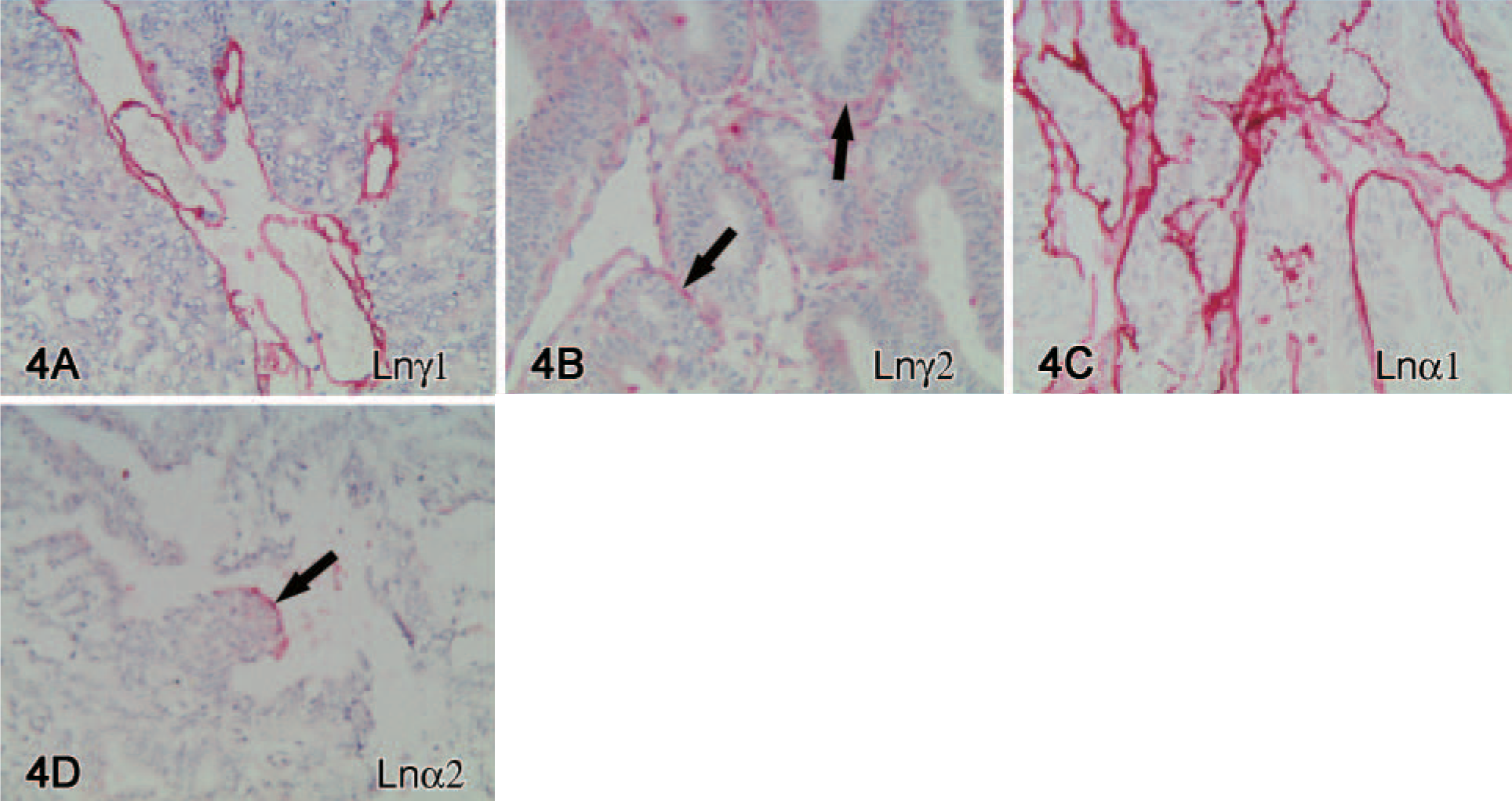
Endometrioid carcinoma. Ln γ1 chain
Lutheran (Lu) immunoreactivity. Lu shows two types of immunoreactivity in ovarian carcinomas. In a grade I serous carcinoma case, immunoreactivity is mainly present in basal tumor epithelial cell in a polarized manner
Lutheran
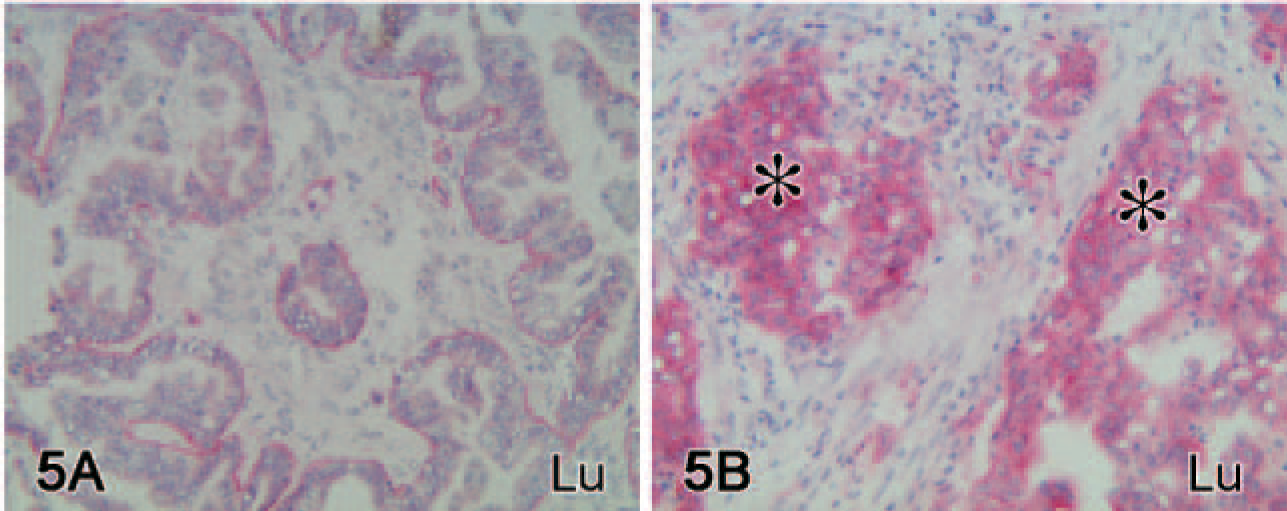
Most of the cases 33/36 (92%) showed immunoreactivity for Lu, being found either in a polarized manner localizing to the basal aspect of the tumor cells (Figure 5A) or in a more diffuse manner on cell surfaces (Figure 5B). These two patterns of immunoreactivities were usually simultaneously present. A solely polarized immunoreactivity was found in 7 tumors and a mainly diffuse cell surface distribution was detected in 20 tumors. The central parts of tumor islands lacked Lu immunoreactivity. Well-differentiated serous carcinomas tended to show the most intense overall Lu expression, followed by endometrioid carcinomas, but mucinous tumors showed only an occasional immunoreactivity. Three tumors lacked Lu-immunoreactivity when Ln α5 chain was present, but no Lu lacking cases when Ln α5 chain was not present. Lu immunoreactivity was constantly present in the vascular endothelium and on stromal fibroblasts.
Discussion
In this study, we have used chain-specific antibodies against Ln chains and antibodies against type IV, VII, and XVIII collagens to study their distribution in the normal ovary and in the three major types of ovarian carcinomas. Previous studies have been performed by using polyclonal antibodies to mouse EHS-Ln (Stenback and Wasenius 1985; Kuwashima et al. 1995), which, however, probably detect Ln heterotrimers containing γ1 chain in man only (Erickson and Couchman 2000). Previous studies of the role of BM in ovarian carcinomas have resulted in conflicting results. It has been reported that BM proteins are highly diminished or even lost in premalignant lesions, whereas, in other studies, fewer discontinuities were found (Barsky et al. 1983; Kuwashima et al. 1995; Skubitz et al. 1996). Capo-Chichi et al. (2002) reported that of 50 primary ovarian tumors, only 6% were positive for the type IV collagen and 24% for Ln. Our study, however, shows that ovarian carcinomas express significant amounts of BM proteins around tumor islands. However, there was a general tendency for the grade I-II tumors to contain more BM immunoreactivity than did grade III tumors. These results suggest that the ability to express and deposit BM proteins in general is not lost during malignant transformation. Differences between the earlier results may be due to differences in tissue sampling, subjective scoring, and antibodies and methods used.
The present study shows immunoreactivity for several different Ln α-chains in BMs of ovarian carcinomas, suggesting the presence of more than one Ln heterotrimer in the tumor BMs. We have previously shown that Ln isoforms found in tumors usually reflect the BM composition of their normal tissue of origin (Määttä et al. 2001; Rissanen et al. 2003); when comparing the results obtained from tumors and OSE, this is likely true also in the case of ovary (Table 2). However, there were some differences; for instance, Ln α2 chain and collagen type VII were not detected in BM of OSE, whereas they were present in variable amounts in BM of ovarian carcinomas. This may represent a true neoexpression, as has been reported for some other tumor types (Lohi et al. 1998; Catusse et al. 2000), or to be related to the origin of the tumor (as previously discussed). Collagen type VII normally localizes to the anchoring fibrils together with Ln-5, but in ovarian carcinomas, such a colocalization was not observed and it was mainly confined to the well-differentiated serous carcinomas. Lack of type VII collagen in tumor BMs has earlier been reported by Tani et al. (1996) and Catusse et al. (2000). This might reduce adhesion of the carcinoma cells to the BMs and hence enhance the ability of Ln-5 to promote cell migration (Katayama and Sekiguchi 2004).
Chains of laminin-10 and collagens type IV and XVIII were found in BMs of most tumors in all three ovarian carcinomas. Chains of Ln-5 were detected in BMs of serous and mucinous tumors, but only inconsistently in endometrioid tumors. We have previously reported that chains of Ln-10 can be found in most carcinomas (Määttä et al. 2001). In serous carcinomas, the presence of a pronounced Ln β2 chain immunoreactivity instead of Ln β1 chain suggested the presence of Ln-11 in these tumors.
Our results show that there are differences in the laminin composition of BMs among the three ovarian tumor types. The BMs of mucinous carcinomas were characterized by the constant presence of Ln α4 chain, suggesting the presence of mostly Ln-8 (α4β1γ1), because only occasional immunoreactivity for Ln β2 chain was found. Ln α4 chain was also found in the tumor stroma and vascular BMs, in line with the previous studies (Miner et al. 1997; Petäjäniemi et al. 2002). Inflammatory cells may also be a significant source for this chain (Geberhiwot et al. 2001). Interestingly, in vitro Ln α4 chain is poorly adhesive, but effectively stimulates cell migration (Fujiwara et al. 2001), a feature that may affect the metastatic potential of carcinomas of the ovary. In this respect, it is of interest that Ljubimova et al. (2001) recently showed that α4 chain is upregulated in human gliomas and antisense inhibition of Ln α4 chain resulted in effective invasion block by glioma cells (Khazenzon et al. 2003).
We also found that Ln α1 chain was constantly present in BMs of endometrioid carcinomas in contrast to findings on serous and mucinous carcinomas. Endometrioid tumors are characterized by endometriosis in significant numbers of cases, and deep endometriotic deposits induce metaplastic Müllerian transformation of adjacent mesenchymal cells (Van Niekerk et al. 1993; Leyendecker et al. 1998). We found that normal OSE contains Ln α1 chain, which suggests that endometrioid adenocarcinoma might be derived from OSE. However, it is also known that endometrial surface epithelium and glandular BM contain Ln α1 chain (Virtanen et al. 2000), which would therefore also support the implantation theory (for review, see Leyendecker et al. 1998). Additionally, it has been noted that endometrioid carcinomas of the ovary and endometrium are often positive for vimentin and carcinoembryonic antigen, contrary to endometrioid carcinomas of endocervix, which are usually negative (Dabbs et al. 1996). Together, these differences may help clinically to detect the origin of the tumor.
We decided to include Lu glycoprotein in the study because it is known to effectively bind to Ln α5 chain-containing heterotrimers, which appeared to be ubiquitously present in normal and neoplastic ovary. BM of normal OSE expressed Ln α5 chain and showed a basally confined immunoreactivity for Lu. In carcinomas, however, Lu showed either a basally confined immunoreactivity or a more diffuse cell surface distribution. There were also three cases that totally lacked Lu immunoreactivity, but presented immunoreactivity for Ln α5 chain. Together our results implicate that during tumorigenesis Lu loses its normal polarized expression in significant amounts of ovarian carcinomas, which indirectly suggests that it also simultaneously may lose its function as a receptor for Ln α5 chain-containing heterotrimers. When comparing our results with those previously reported by Gabin-Chesa et al. (1994), the overall expression was similar (87% vs our 92%), but we found Lu to be mostly localized to basal epithelial cells, whereas they reported that ovarian carcinomas show mainly uniform staining pattern over the tumor area. Many ECM receptor alterations have been observed in ovarian carcinomas, of which some are reported to have decreased expression, whereas others are upregulated in tumor tissues (Skubitz et al. 1996; Strobel and Cannistra 1999; Goldberg et al. 2001; Skubitz 2002). Nonpolarized Lu expression, together with changes in other ECM receptors, may modulate tumor cell adhesion to BMs and influence on migration of tumor cells.
In conclusion, the present results show that ovarian carcinomas present BMs around tumor cell nests consisting mostly of Ln-5 and Ln-10 and collagen types IV and XVIII. Mucinous tumors were characterized by immunoreactivity for Ln α4 chain, whereas BMs of endometrioid tumors were found to have, on the other hand, distinct immunoreactivity for Ln α1 chain. These differences suggest that the latter two tumor types have distinct features that may have a different histogenetic origin or differentiation pattern compared with serous tumors. These features may serve a diagnostic tool to distinguish these carcinoma types from each other.
Footnotes
Acknowledgements
Ms. Heli Auno, Ms. Annikki Huhtela, Ms. Tuulikki Moilanen, Ms. Marja-Leena Piironen, Ms. Riitta Vuento, and Mr. Hannu Wäänänen are acknowledged for their excellent technical assistance.
The antibody to the Ln β2 chain developed by Dr. Dale Hunter and Dr. Joshua Sanes was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, Iowa.
