Abstract
Nasopharyngeal carcinoma (NPC)-associated gene 6 (NGX6) is a novel candidate metastasis suppressor gene that can significantly decrease the growth, motility, and invasion of NPC cells. In this study, we generated a highly specific NGX6 polyclonal antibody and analyzed its distribution in the human fetus by Western blot and immunohistochemistry. The result of the Western blot showed the protein of NGX6 had two types of isoforms, isoform a (NGX6a) and isoform b (NGX6b). Isoform a is composed of 472 amino acids with a calculated molecular mass of 52 kDa, whereas isoform b is composed of 338 amino acids with a calculated molecular mass of 37 kDa. It is predicated that there is an epidermal growth factor domain in the N terminal of both a and b isoforms, and seven transmembrane domains in NGX6a, but only two transmembrane domains in NGX6b. The expression level of NGX6a was higher than that of NGX6b in human fetal tissue. Obvious high expression of NGX6a protein presents in the nervous system and epithelial tissues of the human fetus, but the NGX6b protein (37 kDa) is mainly expressed in the nervous system. We further analyzed the tissue microarray, which contained 154 NPC biopsies and 70 non-NPC biopsies, and found that NGX6a was significantly downregulated in NPC and associated with tumor metastasis.
N
Materials and Methods
Construction of Fusion Genes
NGX6ΔTM2 is the second transmembrane (aa 269–291) region-deleted mutant of NGX6b (GenBank accession no. NM_016446). NGX6a (GenBank accession no. NM_001042589 or NM_001042590) is the a isoform of the NGX6 protein. The sequences of NGX6ΔTM2 and NGX6a were derived from template plasmid pcmv-NGX6ΔTM2 constructed by Dr. Shu-Ping Peng (Peng et al. 2006) and the human fetal brain library by PCR using the primers as follows: NGX6ΔTM2: forward, 5'-CGTCATATGAACATGCCCCAGTCCCTG-3'and reverse, 5'-GCCCAAGCTTTCATTTACTCCAATGTG-TCTTACAA-3'; NGX6a: forward, 5'-GAT GAATT-CCCATGAACATGCCCCAGTCC-3' and reverse, 5'-AAGCTCGAGTCAGCTGGCACAGATGCT-3'. PCR conditions were 94C for 5 min, 35 cycles of 94C for 30 sec, 55C for 30s or 70C for 30s, 72C for 1 min or 1 min 40s, and 72C for 5 min. The PCR fragments were purified and ligated into NdeI/HindIII-digested pET28b vector (Novagen; Gibbstown, NJ) and EcoRI/XhoI-digested pCMV-Myc vector (Clontech; Mountain View, CA) respectively, yielding the constructs pET28b-NGX6ΔTM2 and pcmv-myc-NGX6a. To identify the positive clones with inserts, plasmid DNA extracted from clones after transformation with recombinant constructs was first subjected to PCR using the same primer pairs mentioned above and confirmed by sequencing.
Preparation of Polyclonal Antibody
The immunogen used in this study was the recombinant His-NGX6ΔTM2 protein obtained from prokaryotic expression in Escherichia coli. NGX6ΔTM2 is the second transmembrane (aa 269–291) region-deleted mutant of NGX6b. The expression plasmid pET28b-NGX6ΔTM2 and empty vector pET28b were transformed into E. coli and Rosetta Blue (DE3) (Novagen) strains, respectively, and induced at 1 mM isopropyl-b-D-thiogalactopyranoside, 37C for 5 hr. The recombinant His-NGX6ΔTM2 protein was purified with Ni-IDE chromatography resin (Novagen) under denatured conditions. All of the denatured substance was removed by dialysis in PBS (150 mM sodium chloride, 150 mM sodium phosphate, pH 7.2) at 4C overnight. Purified His-NGX6ΔTM2 was analyzed by SDS-PAGE and Western blot (see below). Two 5-month-old New Zealand White rabbits were immunized subcutaneously with 200 mg of the His-NGX6ΔTM2 protein per rabbit, followed by a second immunization of 100 mg per rabbit 4 weeks later. After the second injection, three additional injections (100 mg protein per injection) were performed at 2-week intervals. Three weeks after the last injection, sera were collected and purified using the caprylic acid-ammonium sulfate method of McKinney and Parkinson (1987). The concentration of NGX6 antibody was analyzed by the bicinchoninic acid method. Preimmunized rabbit serum collected before the day of primary immunization was applied as a negative control.
Tissue Specimens and Tissue Microarray (TMA) Construction
Nasopharyngeal biopsy specimens including 158 NPC and 74 non-cancerous nasopharyngeal epithelia (NCNPE) were collected in the Ear, Nose, and Throat Department at Xiangya Hospital (Changsha, China). For laser micro-dissection and Western blot, four NPC and four NCNPE biopsy tissues were snap frozen in liquid nitrogen. For TMA, 154 NPC and 70 NCNPE biopsy tissues were fixed immediately in 4% buffered paraformaldehyde, routinely processed, and embedded with paraffin. The TMA was assembled with a tissue array instrument (Beecher Instruments; Silver Springs, MD). Three 0.6-mm-diameter tissue cores were taken from each NPC, and two 0.6-mm-diameter tissue cores were taken from each NCNPE. The sections were covered with thin paraffin and stored at 4C before immunohistochemistry assay (Fan et al. 2006). Three 28–30-week-gestation human fetuses were collected from termination of pregnancy material by Liu et al. (2008), with appropriate written consent and approval from the Central South University Health Authority Joint Ethics Committee and following national guidelines. Twenty-four types of tissue were collected and frozen in liquid nitrogen for long-term storage (Table 1). For immunohistochemistry, the tissues were fixed in 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.2) at 4C overnight and embedded in paraffin.
NGX6 expression in human tissues
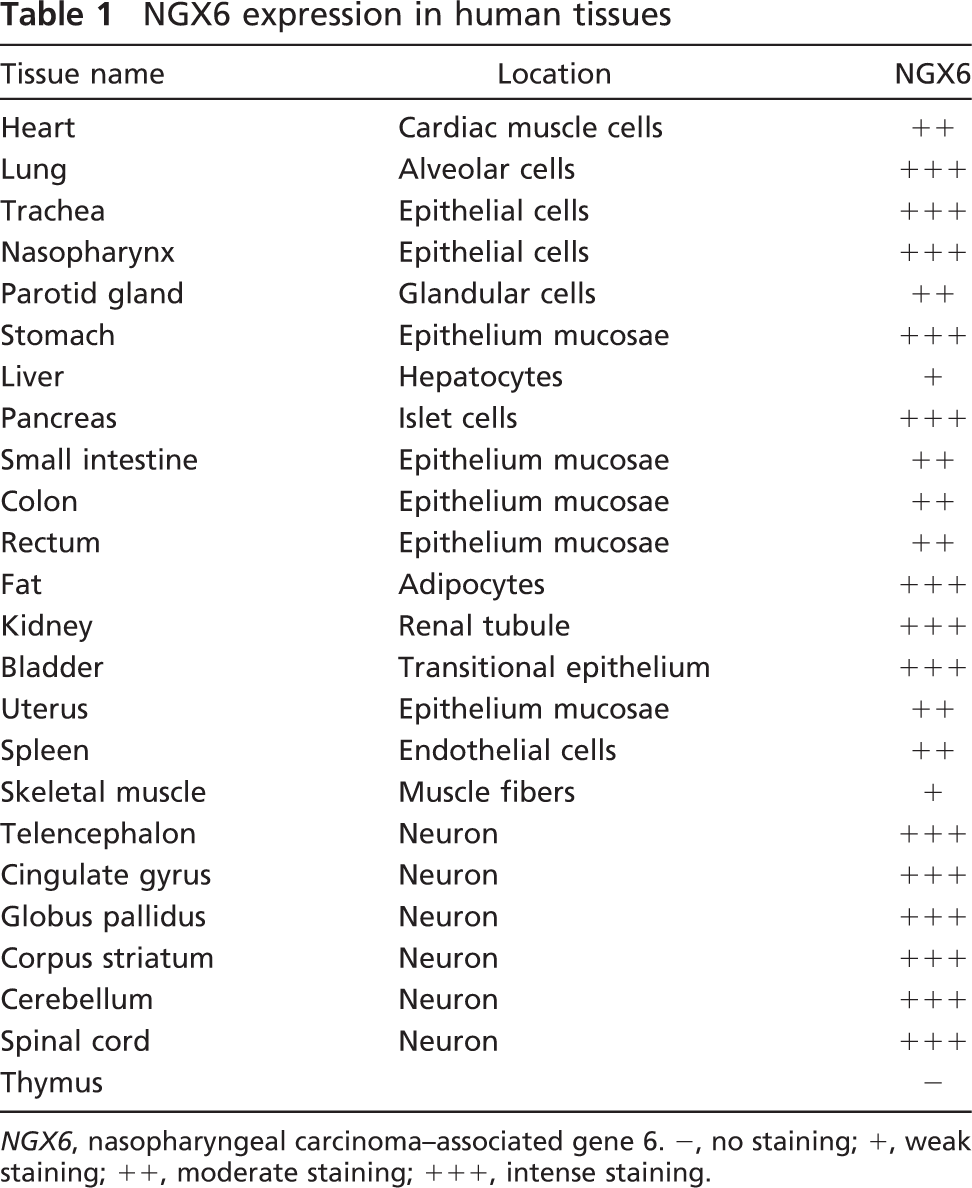
NGX6, nasopharyngeal carcinoma-associated gene 6. -, no staining; +, weak staining; ++, moderate staining; +++, intense staining.
Laser Microdissection (LMD)
LMD was performed using the Leica ASLMD (Leica Microsystems Ltd.; Wetzlar, Germany). The Leica system uses a maintenance-free, pulsed, nitrogen laser at a wavelength of 337 nm. To excise the structure of interest (nasopharyngeal epithelia and NPC tissues), the pulsed laser follows a predrawn line, ablating the material only in the region of the defined line. This method of dissection ensures that the specimen is not heated and that it endures no mechanical contact, therefore eliminating the risk of contamination. We used four NPC and four NCNPE fresh samples to be processed using LMD. This system uses glass slides that are covered in one area with polyethylene naphthalate foil. Slides for the LMD were first treated with ultraviolet light for 30 min to reduce electrostatic charges. Five-μm sections from either NPC or NCNPE tissues were then cut onto a slide by using a freezing microtome. Sections were fixed in 70% ethanol for 1 min and stained with methyl green. Xylene was omitted due to its effects on the morphology of the tissue on the LMD slides. After dissection, the sample fell by gravity into a precisely positioned PCR tube cap. Following collection of the microdissected cells, the cap could be automatically examined to visualize the tissue excised.
SDS-PAGE and Western Blot
Cells and tissues were lysed with radioimmunopre-cipitation assay lysis buffer [50 mM Tris, 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, protease inhibitor cocktail (Complete Mini; Roche, Mannheim, Germany), pH 7.4]. The extracts were clarified by centrifugation at 12,000 rpm for 10 min. One μg recombinant His-NGX6ΔTM2 protein and 50 μg cells or tissues total proteins were separated by SDS-PAGE according to the method of Laemmli (1970), using a 4.5% stacking gel and 10% separating gel. After electrophoresis, the proteins were stained with Coomassie Blue R250 (Sigma; St. Louis, MO) or electronically transferred onto polyvinylidene difluoride membranes. Western blotting was performed as described by Towbin et al. (1979). The membranes were incubated with monoclonal mouse anti-HIS antibody (Sigma) for detecting recombinant His-NGX6ΔTM2 protein, monoclonal mouse anti-c-Myc (Clontech) for detecting myc-NGX6 protein, and the anti-NGX6 primary antibodies, respectively. After three washings, the membrane was incubated with horseradish peroxidase (HRP)-conjugated goat anti-mouse or HRP-conjugated goat anti-rabbit secondary antibodies (KPL; Gaithersburg, MD), respectively, and finally visualized using the ECL PLUS Western blotting substrate kit (GE Healthcare; Buckinghamshire, England) according to the manufacturer's instructions.
Immunohistochemistry and Immunostaining Evaluation
The tissues were deparaffinized in xylene and rehydrated through graded alcohols (100, 90, 70 and 50% alcohol; 5 min each). Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 10 min. For antigen retrieval, sections were incubated in sodium citrate buffer (0.01 M, pH 6.0) for 20 min in a household microwave oven (600 W). After cooling to room temperature, slides were washed in PBS (150 mM sodium chloride, 150 mM sodium phosphate, pH 7.2) and immersed in normal goat blocking serum (Maixin; Fuzhou, China) for 30 min. The anti-NGX6 serum (1:1500) was applied at 4C overnight. Polymerized HRP and anti-rabbit IgG purchased from Maixin were added according to the manufacturer's instructions. Color reaction was developed using diaminobenzidine chromogen solution (Maixin), and all slides were counterstained with hematoxylin. Negative control slides were included in the experiment. Immunohistochemical staining of these sections was scored microscopically (Olympus; Tokyo, Japan) at ×400 magnification in all available tumor cells or epithelial cells meeting the typical morphological criteria, according to two pathologists using the qualitative scale described in the literature (Shanmugaratnam 1991). The number of cells stained was scored as 0 (no staining), 1+ (less than one of three positive cells), 2+ (more than one of three and less than two of three positive cells), and 3+ (more than two of three positive cells). The intensity of staining was scored from 1+ (weak) to 3+ (strong). The immunoreactive score was categorized in three groups by comprehensive evaluation of the percentage of positive cells and staining intensity as reported previously. No staining was considered negative (0 score), weakly, moderately, and strongly staining were considered positive (1 score, 2 scores, and 3 scores, respectively).
Double-fluorescent Staining
HEK 293 cells (6 × 105) were seeded on coverslips in a 6-well plate the day before transfection. On the second day, HEK 293 cells were transfected with pCMV-Myc-NGX6a and pCMV-Myc-NGX6b, respectively, by Lipofectamine 2000 (Invitrogen; Carlsbad, CA) according to the manufacturer's protocol. After incubation for 48 hr at 37C (5% CO2 in a humidified atmosphere), cells were fixed in methanol and acetone (v/v, 1:1) for 30 min, permeabilized using 0.25% Triton X-100, and blocked using normal goat serum as recommended (Molecular Probes; Eugene, OR). The primary antibodies, mouse anti-myc 1:1000 (Clontech) and rabbit anti-NGX6 1:800, were added and incubated at 4C overnight and subsequently washed with PBS. The secondary antibodies, FITC-conjugated sheep anti-rabbit IgG 1:200 and Cy3-conjugated sheep anti-mouse IgG 1:200 (Boster; Wuhan, China) were then added and incubated at 37C for 1 hr. The coverslips were washed four times with PBS. Control experiments were carried out without primary or secondary antibodies. The staining was examined under a 100× objective using an Olympus laser confocal microscope.
Statistical Analysis
Statistical analysis was performed using SPSS 15.0 software (SPSS; Chicago, IL). The Pearson χ2 test was performed to compare the differences in NGX6 protein expression in the selected pairs of the sample groups (NPC-NCNPE, NPC without node metastasis, NPC with node metastasis); p<0.05 was considered statistically significant.
Results
Antibody Specificity
Purified His-NGX6ΔTM2 was analyzed by SDS-PAGE and Western blot. As shown by staining with Coomassie Blue R250 (Figure 1A), His-NGX6ΔTM2 protein was highly purified. The predicted band of 37-kDa recombinant His-NGX6ΔTM2 protein was specifically recognized by the anti-HIS monoclonal antibody (Figure 1B), and no additional bands could be visualized. Western blot results also showed that the new purified NGX6 antibody used in this study was able to recognize recombinant His-NGX6ΔTM2 protein with a molecular mass of 37 kDa (Figure 2A, Lane 2) as that recognized by the anti-HIS monoclonal antibody (Figure 1B, Lane 2). More importantly, this purified NGX6 antibody recognized the full-length recombinant fusion protein Myc-NGX6b with a molecular mass of 38 kDa in HEK 293 cells trans-fected with pCMV-Myc-NGX6b (Figure 2B, Lane 4), which corresponds to the molecular mass detected by monoclonal anti-c-Myc (Figure 2B, Lane 2).
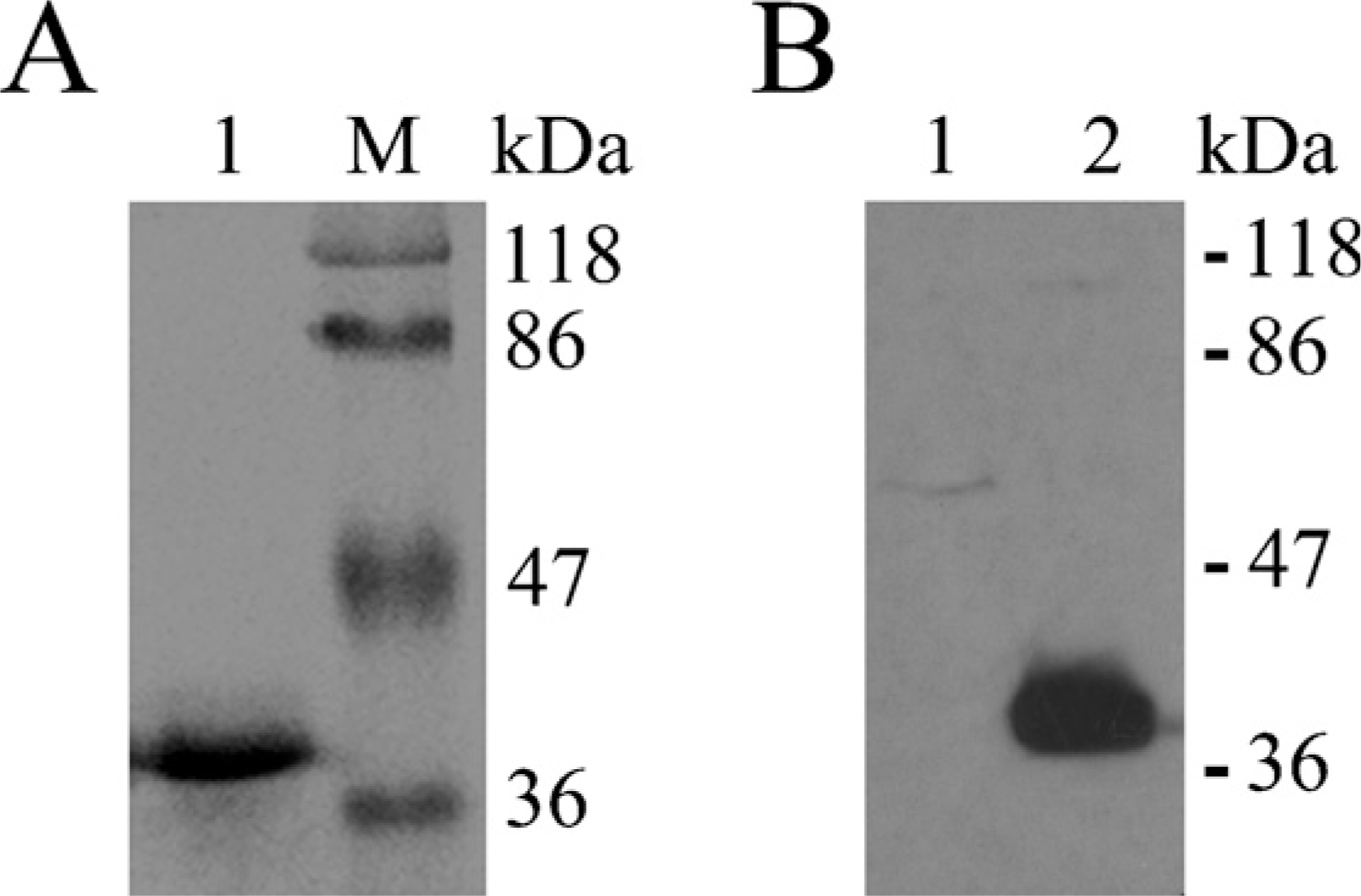
Analysis of the purification of HIS-NGX6ΔTM2 fusion protein in bacteria. (
Identification of Two Types of Isoforms of NGX6 Protein
Our previous work was focused on 37-kDa NGX6 protein (GenBank accession no. NM_016446) before this antibody had been generated. To our surprise, when we used this new antibody to detect the NGX6 protein in brain tissue from human fetus by Western blot, it detected two clear bands on the blot membranes. One was ~37 kDa, and the other was ~50–51 kDa (Figure 2C), which also had been detected in the HEK 293 cell lysate (Figure 2B). By searching for database and bioinformatics analyses (see below), we believed that there were two subtypes of NGX6 protein: isoform a (NGX6a), and isoform b (NGX6b). Subsequently, the cDNA of NGX6a (1419 bp) was cloned by PCR from the human fetal brain cDNA library, and a eukaryotic expression plasmid, pCMV-myc-NGX6a, was trans-fected into HEK 293 cells. The full-length recombinant fusion protein Myc-NGX6a, with a molecular mass of 51 kDa, can be recognized by the purified NGX6 antibody in HEK 293 cells (Figure 2D, Lane 4), which corresponds to the molecular mass detected by monoclonal anti-c-Myc in HEK 293 cells transfected with pCMV-Myc-NGX6a (Figure 2D, Lane 2). As shown in Lane 3 of Figure 2D, the endogenous NGX6a protein was detected in HEK 293 cells transfected with pCMV-Myc vector. To further confirm the antibody specificity and the subcellular localization of NGX6 protein, pCMV-Myc-NGX6a and pCMV-Myc-NGX6b vectors were transfected into HEK 293 cells, respectively. By using mouse anti-myc and highly diluted rabbit anti-NGX6 (1:800) as the primary antibodies, the signals of the two channels were observed to colocalize in the positive transfected cells in double-fluorescent staining (Figures 2E–2G and 2H–2J). These results confirmed that the myc antibody and the NGX6 antibody recognized the same protein, Myc-NGX6a or Myc-NGX6b. The predominant subcellular localization of the NGX6a and -b isoforms was found to be in the cytoplasm and perinuclear.
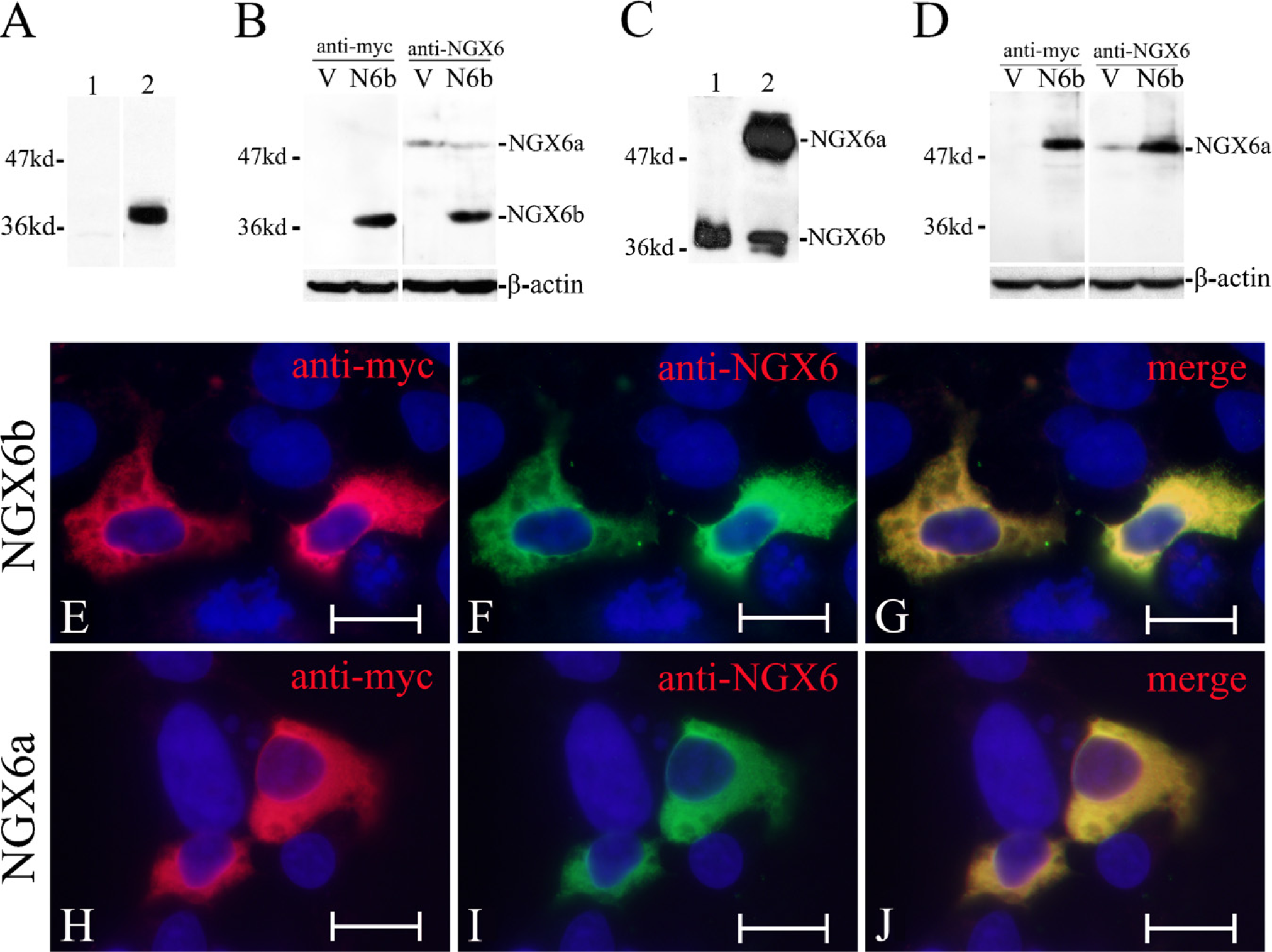
Nasopharyngeal carcinoma (NPC)-associated gene 6 (NGX6) antibody recognized both NGX6a and NGX6b. (
Bioinformatics Analysis
In the NCBI nucleic acid and protein database, there are three mRNA transcript variants of NGX6. Transcript variant 1 (NM_001042590) and variant 2 (NM_001042589) encode the same NGX6 protein isoform (NP_001036055 or NP_001036054), which is composed of 472 amino acids with a calculated molecular mass of 52 kDa, and transcript variant 3 (NM_016446) encodes isoform b (NP_057530), which is composed of 338 amino acids with a calculated molecular mass of 37 kDa. Using SMART software (http://smart.embl-heidelberg.de/) or prosite of ExPASy (http://cn.expasy.org/prosite/), bioinformatics analysis of NGX6a and -b isoforms reveals that there is an EGF domain in the N terminal of both a and b isoforms, and there are seven transmembrane domains in NGX6a, but only two trans-membrane domains in NGX6b. The two isoforms are homologous from the N terminal to aa 273 (Figure 3). The recombinant fusion protein, His-NGX6ΔTM2, purified from E. coli, is a mutant of NGX6b, with a deleted domain from aa 269 to 291, with the same 268 amino acids as in the N terminal of the NGX6a and -b isoforms; therefore, the antibody generated using His-NGX6ΔTM2 as the antigen could recognize both NGX6a and -b (Figure 3).
NGX6 Protein Is Expressed in Most Human Fetal Tissues, Especially in Human Nervous System and Epithelial Tissues
With the prepared NGX6 antibody, we detected the tissue distribution of NGX6 in the human fetus. In the Western blot assay, the NGX6a protein (51 kDa) was detected in most human fetal tissues, especially in the central nervous system (Figure 4). The NGX6b protein (37 kDa) was mainly expressed in the nervous system, and expressed very weakly in heart, lung, nasopharynx, kidney, and fat tissues (Figure 4). The expression level of NGX6a was significantly higher than that of NGX6b in these tissues, particularly in the brain. The expression level of NGX6a is higher (~5.6–10-fold), as measured by gray-scale scanning. Immunohistochemistry assays also showed that NGX6 protein was distributed in the nervous system and epithelial tissues. In the respiratory system, the epithelia of nasopharynx and trachea showed high NGX6 protein expression levels. Both stratified squamous epithelium from nasopharynx (Figure 5A) and the pseudostratified columnar epithelium from trachea (Figure 5B) had positive staining. Lung alveolar epithelium was also highly immunostained (data not shown). In the digestive system, NGX6 was expressed in the stomach mucosa, in which the cytoplasm of the glandular cells near the basal membrane (Figure 5C) and the enterocytes (surface absorptive cells) of the ileum villi (Figure 5D) stained positive. In the pancreas, islet cells were intensely stained (Figure 5I), whereas liver cells had very weak staining (data not shown). In the urogenital system, high expression of NGX6 was found in the transitional epithelium and the renal tubular epithelium (Figure 5J). The uterine mucosa also had intensely positive staining (data not shown). In the nervous system, intense staining was present in neurons (Figures 5K and 5L). In the cerebellum, the expression of NGX6 was localized in the purkinje cells and nerve fibers in the molecular layer (Figure 5L). In conclusion, NGX6 was widely distributed in the nervous system as well as in a variety of epithelial tissues (Table 1).
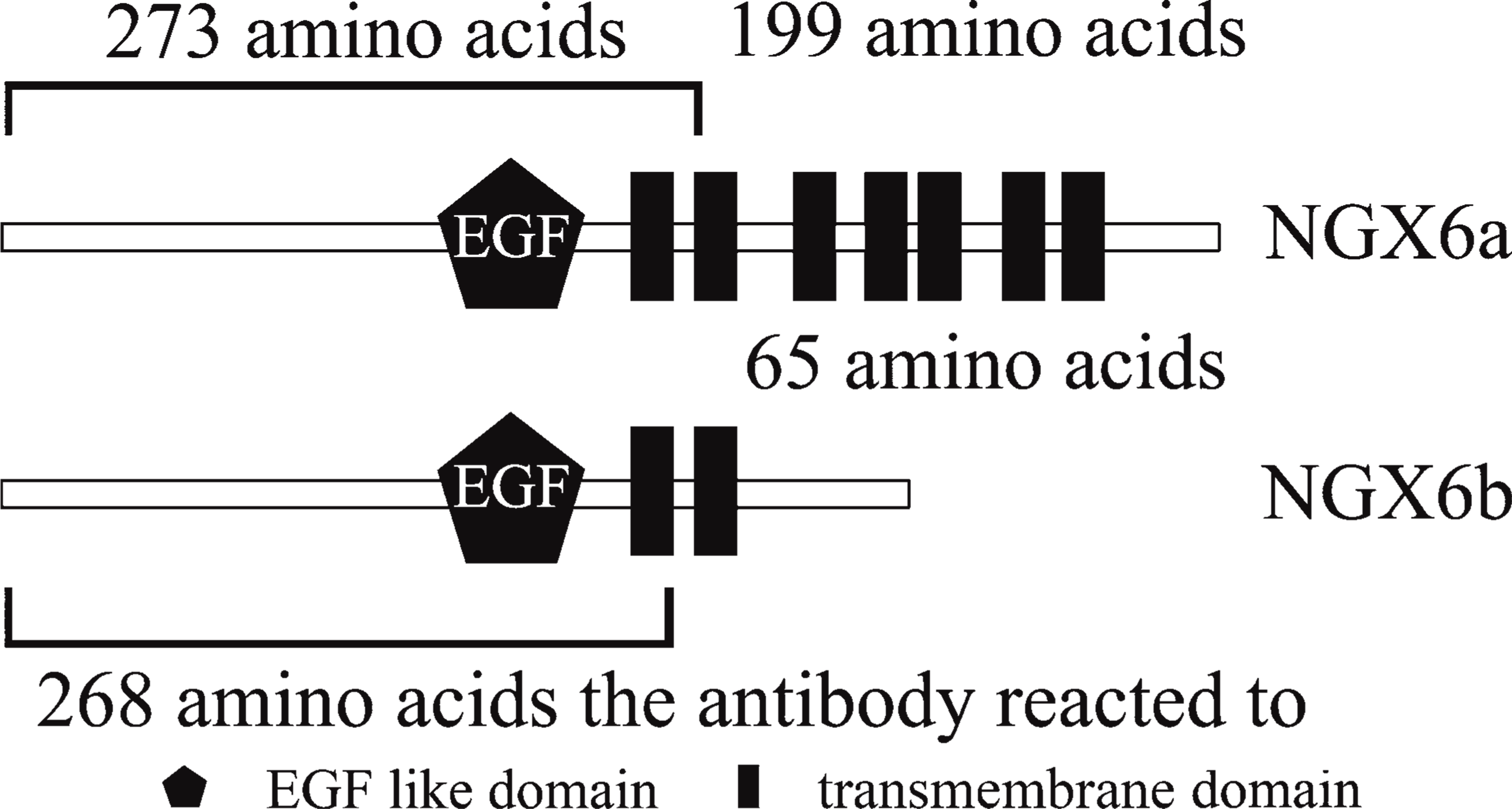
Using SMART software or prosite of ExPASy, bioinformatics analysis of a and b isoforms. There is an epidermal growth factor domain in both a and b isoform N terminals; there are seven transmembrane domains in NGX6a, but only two transmembrane domains in NGX6b. The two isoforms are homologous from the N terminal to aa 273. The antibody reacted to the N-terminal 268 amino acids.
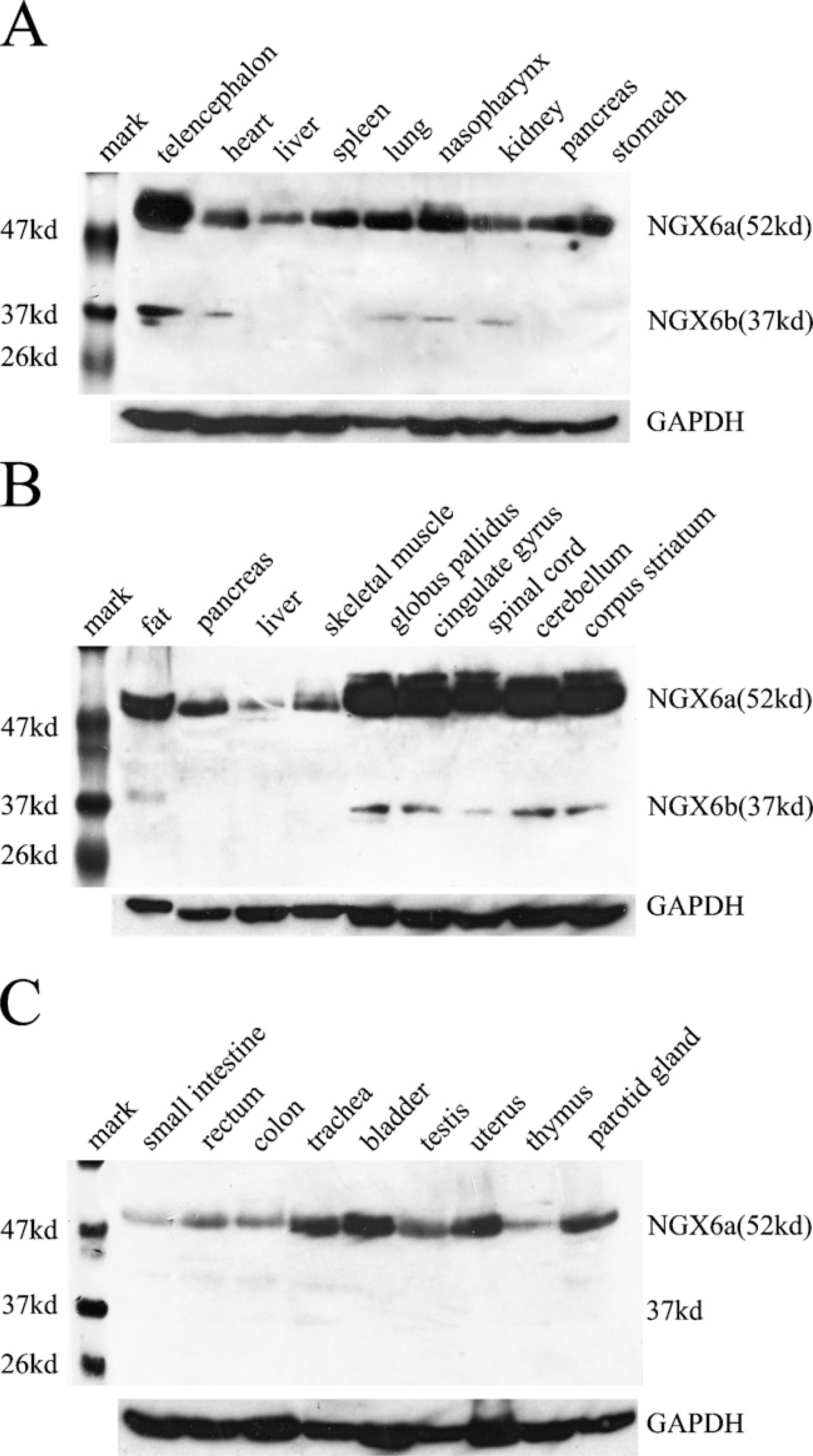
Detection of the endogenous NGX6 protein in various tissues of the human fetus with purified NGX6 antibody by Western blot. (
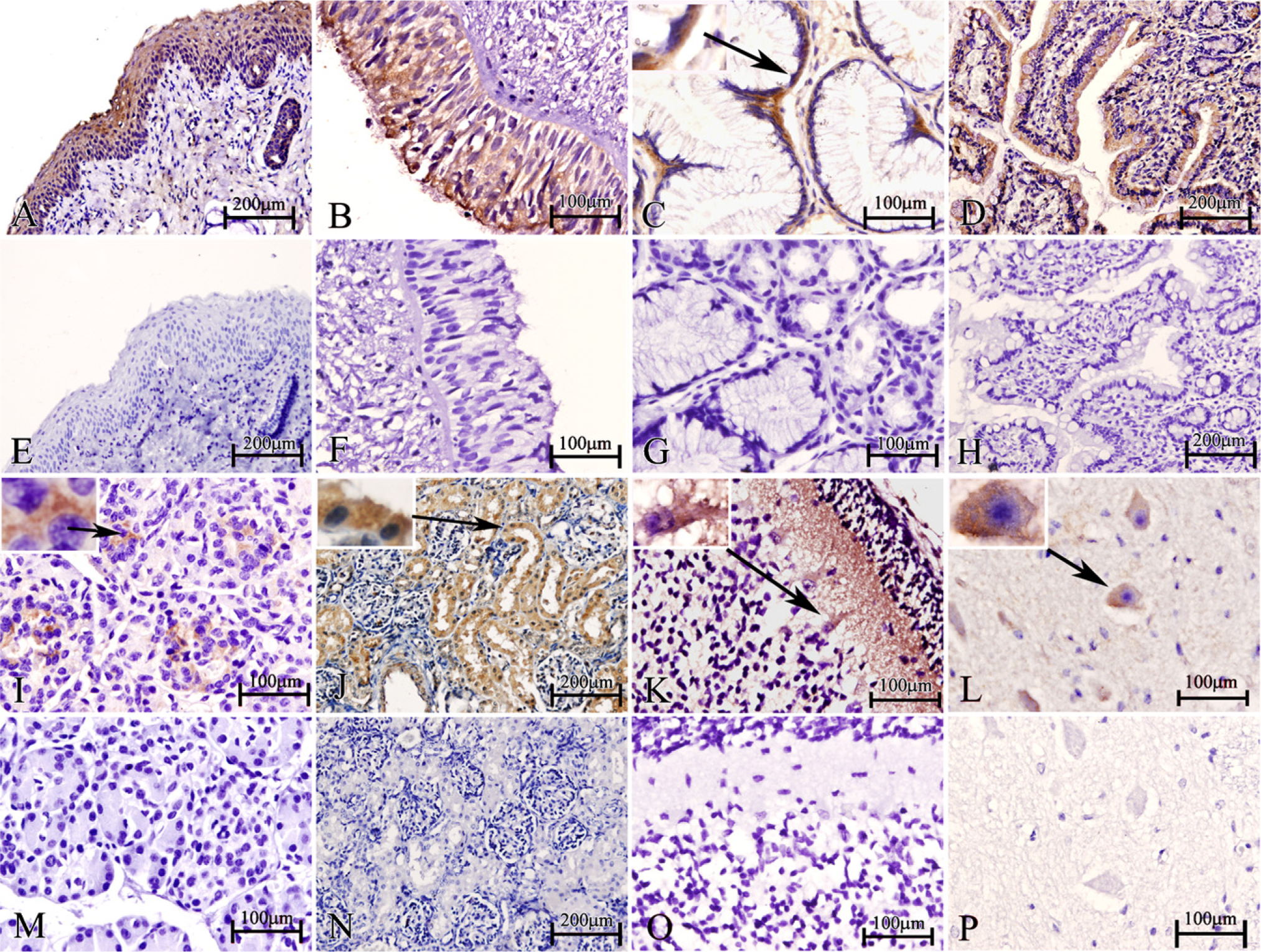
Detection of the endogenous NGX6 protein in various tissues of human fetus with purified NGX6 antibody by immunohistochemistry assay. (
NGX6a is Downregulated in NPC and Associated With Tumor Metastasis
The nasopharyngeal epithelial tissues and NPC tissues from biopsies of four patients were captured by laser microdissection, respectively (Figures 6A–6D). Western blot results showed NGX6a was downregulated in the NPC tissues compared with the nasopharyngeal epithelial tissues, whereas NGX6b cannot be detected by Western blot in adult human nasopharyngeal epithelial tissues (Figure 6E). Moreover, we investigated the association between NGX6a protein expression and the clinical pathological factors (lymph node metastasis) of NPC patients using immunohistochemistry with TMA in 154 NPC biopsies and 70 non-NPC biopsies. The positive expression rate of NGX6a was 28.6% (28/98) in the group of 98 NPC patients with lymph node metastasis, whereas it was 44.6% (25/56) in the group of 56 NPC patients without lymph node metastasis, and 87.14% (61/70) in the group of 70 non-NPC patients (Figures 6F–6K; Tables 2 and 3). Statistical analysis revealed that the positive ratio of NGX6a expression was higher in the non-metastasis group than that in the metastasis group. Similarly, the positive ratio of NGX6a expression was much higher in the non-NPC group than in the NPC groups (Tables 2 and 3).
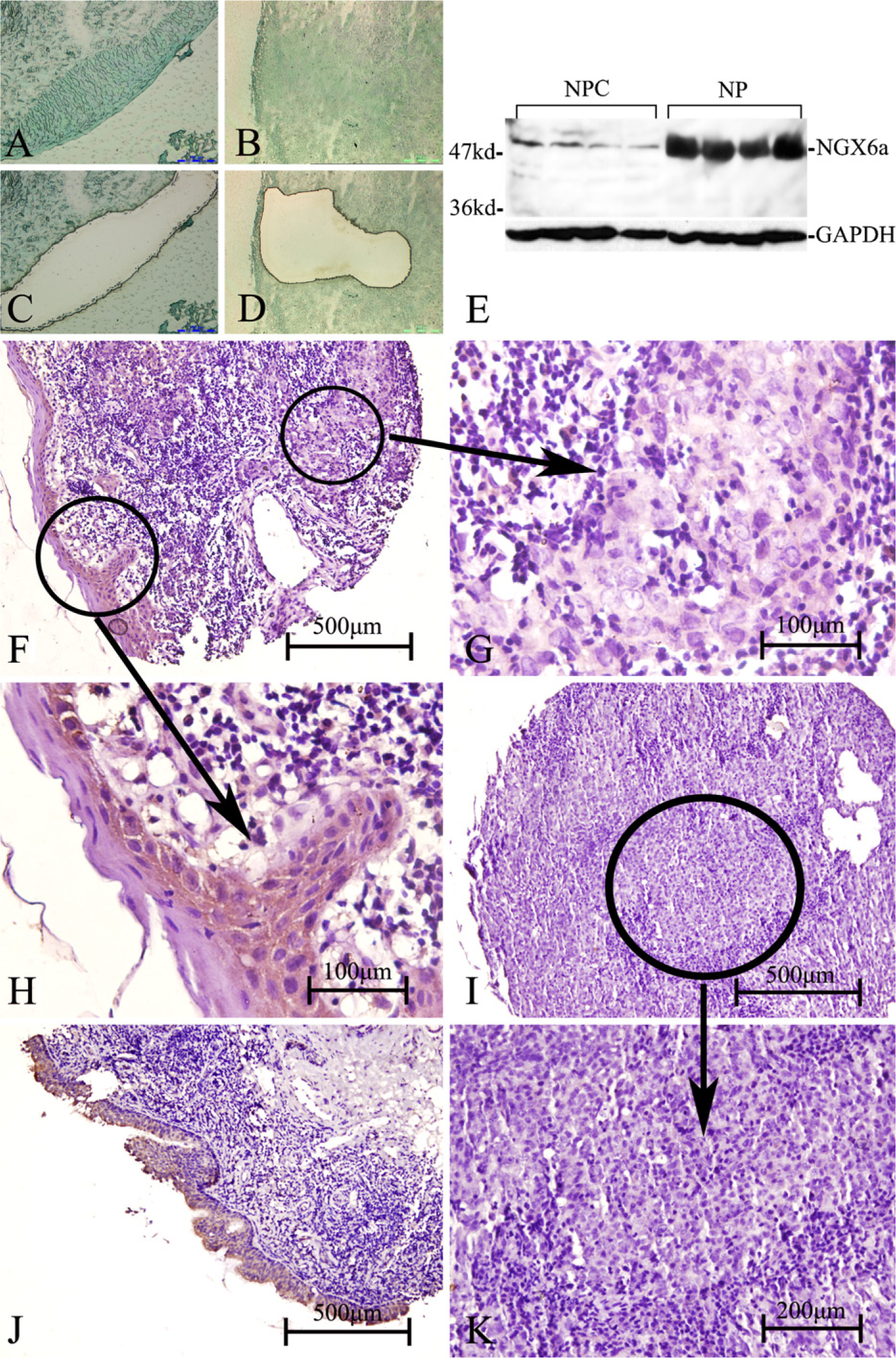
NGX6a was significantly downregulated in NPC. (
Discussion
Multiple factors contribute to NPC initiation and development, although the molecular basis of these is not well understood; gain-of-function of oncogenes and/or loss of expression of tumor suppressor genes are believed to play a critical role in NPC progression. Our previous studies have shown that NGX6, a novel tumor suppressor gene isolated from the high-frequency loss of heterozygosity region at 9p21–22 in NPC patients, is downregulated in NPC cell lines and tumor tissue mRNA levels (Yang et al. 2000a,2000b; Ma et al. 2005). Re-expression of NGX6 can downregulate the EGFR/Ras/Mek/MAPK signaling pathway and inhibit NPC cell growth in vitro and tumor formation in a xenograft model in vivo (Wang et al. 2005), implicating an important role for the NGX6 gene in the progression of NPC. To better understand the function of the NGX6 gene, in the current study, we generated a highly specific NGX6 antibody that detects a new, predominant isoform of NGX6 proteins (NGX6a, 52 kDa), which is broadly and highly expressed in the majority of human fetal tissues, compared with the 37-kDa isoform, NGX6b; importantly, analysis of the NPC tissue microarray implicates the level of NGX6a protein as an important prognostic indicator associated with tumor metastasis.
NGX6a expression in NPC and the nasopharyngeal epithelium

∗ p<0.01.
Note: The χ2 test revealed the higher negative rate of NGX6a expression in NPC compared with chronically inflammatory nasopharyngeal epithelium. NPC, nasopharyngeal carcinoma; NCNPE, non-cancerous nasopharyngeal epithelium.
NGX6a and NGX6b are transmembrane proteins. In our previous studies, the predicted product of the NGX6b gene was a 37-kDa protein composed of 338 amino acids and containing two transmembrane domains, whereas the new isoform, NGX6a, is composed of 472 amino acids with seven transmembrane domains, raising the possibility that NGX6a may be a new member of the TM7 receptors. The TM7 receptors are commonly referred to as G protein-coupled receptors, and constitute the most ubiquitous and versatile family of membrane receptors for many hormones, neurotransmitters, chemokines, calcium ions, tastes, sensory and chemical stimuli (Ji et al. 1998; Bockaert and Pin 1999). On the basis of sequence similarity, the TM7 receptors are divided into the rhodopsin super-family (class A), the secretin receptor superfamily (class B), and the metabotropic glutamate receptor superfamily (class C) (Pierce et al. 2002). Within the class B TM7 receptors, members of a subfamily containing variable numbers of EGF-like domains in the N-terminal extracellular region are referred to as EGF-TM7 receptors (McKnight and Gordon 1998). Currently, six membersofthe EGF-TM7 receptor family have been found in the human: CD97, EGF-like module-containing mucin-like receptor protein 1 (EMR1), EMR2, EMR3, EMR4, and EGF-TM7-latrophilin-related protein (ETL) (Kwakkenbos et al. 2004). With the structural characteristics of one EGF-like domain and seven transmembrane domains, NGX6a probably belongs to the EGF-TM7 subfamily; however, some distinct characteristics shared by the EGF-TM7 family and NGX6a argue for the secluded position of NGX6a within the EGF-TM7 family: (1) all known EGF-TM7 receptors possess at least two tandem arranged EGF-like domains and a conserved cleavage motif in extracellular regions, but only one EGF-like domain was found in NGX6a; (2) the NGX6a gene is located in 9p13.1, and except for ETL at Chr1p32-p33, all other human EGF-TM7 receptor genes are within clusters in 19p13.1 (CD97, EMR2, and EMR3) and 19p13.3 (EMR1 and EMR4). On current evidence, EGF-TM7 receptors are restrictedly expressed by hematopoietic cells and smooth-muscle cells; in contrast, NGX6a is broadly expressed in the majority of human fetal tissues, especially in the central nervous system and epithelial tissues. Whether the NGX6a is a novel EGF-TM7 receptor or represents a totally new subfamily of TM7 receptors, further evaluation is required to define its cellular ligands, mouse orthologs, and evolved family members in other vertebrate genomes.
Relationship between expression of NGX6a protein and NPC metastasis

∗ p<0.05.
Note: The χ2 test revealed that the non-metastasis group had a higher positive NGX6a expression ratio than the metastasis group.
The EGF-like domain, generally including six conserved cysteines, is frequently found in a large number of membrane-bound and extracellular proteins, which is believed to be crucial for disulphide bond formation, correct three-dimensional protein structure, and protein-protein interaction in diverse cellular activities such as blood coagulation, fibrinolysis, cell adhesion, and normal neural and vertebrate development (Campbell and Bork 1993). Recently, several new EGF-containing transmembrane proteins such as BET, M7365 (tomoregulin-1), and MAEG (MAM- and EGF-containing gene) have been shown to be related to human and murine neural system developmental disorders (Buchner et al. 2000; Eib et al. 2000; Nishizumi et al. 2002). In our study, NGX6a protein was found to be ubiquitously expressed in the fetal neural system, in globus pallidus, cingulated gyrus, corpus striatum, and spinal cord, significantly higher (10-fold) than NGX6b (Figure 4), implicating NGX6 in a critical physiological role in CNS development and maturation for NGX6a. Utilizing mouse model and mutant technology to further define the transient and/or developmental expression profiles of NGX6a proteins will provide more insight into the physiological functions of NGX6a. In the current study, an important finding is that NGX6a protein, but not NGX6b protein, was decreased in NPC tumor tissues and associated with lymph node metastasis. Although NGX6b was easily found in human fetal brain, heart, lung, kidney, and nasopharynx tissues, it could not be detected in normal adult human nasopharyngeal tissue or NPC tumor tissue by Western blot. The mechanism underlying this finding is worth further investigation. In previous studies, NGX6 as a candidate tumor suppressor gene was also downregulated in colorectal carcinomas and hepatocellular carcinoma in mRNA levels and associated with tumor-node-metastasis stages and lymph node metastasis (Xiao and Shen 2008; Peng et al. 2009). The NGX6 mRNA levels of these tumor tissues detected by Xiao and Peng were total levels, including both a and b, and therefore, the protein expression levels of NGX6a and NGX6b should be examined in these and other tumor tissues.
Ubiquitous distribution in epithelial tissues is another distinguishing characteristic of NGX6 proteins, which include those in epithelium of nasopharynx and trachea and lung alveolar epithelial cells in the respiratory system, epithelium of stomach mucosa, enterocytes of the ileum villi in the digestive system, and transitional epithelium and renal tubular epithelium in the urogenital system (Figure 5). In epithelial cells, the EGF-like domain-containing proteins have been implicated in a dual adhesion and signaling function by interaction with other cell surfaces or extracellular matrix proteins; e.g., the EGF-TM7 receptor EMR2 mediates myeloid cell attachment through binding with chondroitin sulfate glycosaminoglycans on the surface of adherent cell lines (Stacey et al. 2003). Similarly, in our previous studies, ectopic expression of the NGX6b gene could enhance the NPC cell attachment on extracellular matrix fibronectin or laminin, increase gap junctional intercellular communication, and inhibit cell migration and invasion (Ma et al. 2005). In in vitro and in vivo studies, NPC cells transfected with the NGX6b gene had a marked decrease in cell proliferation, delaying the G0-G1 cell cycle progression, and reducing tumor formation and tumor size in nude mice, concomitant with downregulation of the EGFR/Ras/Mek/MAPK signaling pathway (Wang et al. 2005). Although the exact underlying mechanism(s) is unknown, one hypothesis is that with the EGF-like domain, NGX6b may act as an antagonist receptor to interfere with EGFR distribution and aggregation on the plasma membrane, or to deplete EGFR ligand binding/attenuate the sequential intracellular signaling transduction. It is thought that NGX6a has a similar function in NPC cells, but this will need to be confirmed in further experiments.
We have previously demonstrated that NGX6b is associated with ezrin by its cytoplasmic tail (Ma et al. 2005; Peng et al. 2007). Ezrin plays an important role in cellular cytoskeleton reorganization, cell adhesion and migration, extracellular matrix interactions, and cell-cell communication as a crosslinker between the plasma membrane proteins and the cortical cytoskeleton (Yao et al. 1996). Upregulation of ezrin has been shown to be related to tumor metastasis and progression through the integration of cell surface molecules and signal transduction molecules. For example, in a mouse model of osteosarcoma, ezrin was found to be necessary for metastasis to reach the lung, and ezrin-mediated early metastatic survival was partially dependent on activation of MAPK (Khanna et al. 2004). The high expression level of ezrin was confirmed in NPC cell lines and tumor tissues, and knockdown of ezrin expression in 5–8F NPC cells could reduce invasive ability; more importantly, re-expression of NGX6b in 5–8F NPC cells could downregulate the expression of ezrin (Peng et al. 2007). Thus, the potential mechanism through which NGX6b downregulates the MAPK signaling pathway may be through decreasing the expression level of ezrin, although the precise involvement of ezrin in these processes needs be elucidated. Based on the similarity between NGX6a and NGX6b in the cytoplasm region, we believe that NGX6a can also bind with ezrin; actually, our recent coimmunoprecipitation experiment confirmed that NGX6a indeed interacts with ezrin (data not shown).
In conclusion, we have generated a highly specific NGX6 antibody and identified NGX6a as a predominant new isoform of the NGX6 proteins that is most prevalent in the human nervous system and epithelial tissues, whereas NGX6b was mainly localized in the nervous system. Furthermore, NGX6a was significantly downregulated in NPC and metastatic tumors. These findings provide important clues for further study of the biofunctions of the NGX6 gene and evidence of NGX6a as a potential prognostic marker for metastatic tumors.
Footnotes
Acknowledgements
This research was supported by grants from the National Key Project of Scientific Research Program of China (2006CB910502), the National 863 Projects (2007AA0–2Z170), the National Natural Scientific Foundation of China (30971147), the Programme of Introducing Talents of Discipline to Universities (111–2–12), and the Province Natural Scientific Foundation (06JJ2042, 08JJ3051).
We sincerely appreciate the instruction of Li Liu (SKD Biotech) in antibody preparation.
