Abstract
BRD7 is a novel bromodomain gene. It plays critical role in cell growth, cell cycle progression, and signal-dependent gene expression. Overexpression of the BRD7 gene in nasopharyngeal carcinoma cells is effective to inhibit cell growth and cell cycle progression from G1 to S phase. However, little is known about its bio-functions because of the unavailability of a specific BRD7 antibody. In this study, for the first time, we generated a highly specific BRD7 antibody. It is able to specifically recognize recombinant GST-BRD7N protein with a molecular mass of 65 kDa and recognize BRD7-Myc and endogenously expressed BRD7 protein with an approximate molecular mass of 75 kDa, which corresponds well with the calculated molecular mass of the BRD7 protein. More importantly, with these antisera, we analyzed BRD7 distribution in the human fetus by Western blot and immunohistochemistry assays. Obvious nuclear expression of BRD7 protein presents in human cerebellum, pancreas, intestines, liver, and kidney. Cardiomyocyte shows high cytoplasm expression of the BRD7 protein. Weak nuclear expression of the BRD7 protein is found in human cerebrum, lung, and stomach. These data may help to further study the cellular role of the BRD7 gene. In particular, the prepared BRD7 antibody will be helpful for studying the bio-functions of endogenously expressed BRD7 protein.
BRD7 is a novel bromodomain gene (Yu et al. 2000). Because of a family member of bromodomain genes and the sequence similarity with BP75 and other bromodomain-containing proteins, it has been suggested that BRD7 may be a component of chromatin remodeling complexes and possess histone acetyltransferase activity (Patrizia et al. 2001; Zeng and Zhou 2002). Kzhyshkowska et al. (2003) showed that BRD7, together with E1B-AP5, functions as an inhibitor of basic transcription in several viral and cellular promoters in the nucleus. Staal et al. (2000) reported that BRD7 protein (celtix-1) interacts with interferon regulatory factor 2 in the nucleus and associates with transcriptionally active chromatin in situ. Overexpression of the BRD7 gene in NPC cells is effective to inhibit cell growth and cell cycle progression from G1 to S phase by transcriptional regulation of some cell cycle–related genes (Yu et al. 2001; Zhou et al. 2004; Peng et al. 2006). To better understand the cellular role of the BRD7 gene, in this study, we explored an approach to generate a highly specific BRD7 antibody, and showed that the prepared BRD7 antibody is able to specifically recognize recombinant GST-BRD7N protein and endogenously expressed BRD7 protein. More importantly, with these antisera, we analyzed the distribution of the BRD7 protein in the human fetus and showed that BRD7 protein is expressed in most human fetal tissues and is strongly expressed in human cardiocyte, cerebellum, pancreas, intestine, and liver. These data may help to further study the cellular role of the BRD7 gene. In particular, the prepared BRD7 antibody will be helpful for studying the bio-functions of endogenously expressed BRD7 protein.
Materials and Methods
Construction of Fusion Genes
The sequence [spanning from 54 to 1112 nucleotides (nt)] encoding the N-terminal 353 amino acids of the BRD7 protein (referred to as BRD7N) was amplified by PCR using the primers as follows: BRD7N forward primer, 5′-TTTTGGATCCATGGGCAAGAAGCACAAGAAG-3′; BRD7N reverse primer, 5′-CCTTGTTGCTGCAACCCTGAAACTGAGTCTTTT-3′. PCR conditions were 94C for 2 min; 25 cycles of 94C for 1 min, 55C for 1 min, and 72C for 2 min; and 72C for 5 min. The PCR fragment was purified and ligated into BamHI/XhoI digested pGEX-4T vector (GE Healthcare Biosciences; Piscataway, NJ), yielding the construct pGEX-4T-BRD7N. To identify the positive clones with inserts, plasmid DNA extracted from clones after transformation with recombinant constructs was first subjected to PCR using the same primer pairs mentioned above and confirmed by sequencing.
Expression and Purification of the BRD7N Protein
A single transformed BL21 colony harboring the recombinant construct pGEX-4T-BRD7N was grown in 5 ml Luria-Bertani (LB) broth containing 50 mg/L ampicillin at 37C, 220 rpm overnight. Two ml of this culture was transferred into 1 liter of fresh LB medium containing ampicillin and was grown at 37C to an optical density of 0.6 at 600 nm. Isopropyl-β-D-thiogalactopyranoside (IPTG) was added to the culture at a final concentration of 0.5 mM to induce expression of the GST-BRD7 protein. The culture was harvested at 3.5 hr postinduction and centrifuged at 5000 × g at 4C for 15 min. After washing, the recombinant cell pellet was resuspended in 20 ml of 20mM Tris-HCl buffer (pH 8.0), containing 0.5 mM EDTA and 1 mM PMSF, sonicated at 200 W for 6 min in an ice bath, and centrifuged at 12,000 × g at 4C for 15 min. The supernatant was transferred into a fresh tube. Solution containing 60 mM ATP, 0.3 M MgSO4, and 1.5 M Tris (pH 7.4) was added into the supernatant in a proportion of 1:30, mixed, and incubated at 37C for 10 min before affinity chromatography. Four hundred μl of 50% slurry of glutathione-Sepharose was added to the supernatant and incubated overnight. After several washes of glutathione-Sepharose beads, the GST-BRD7N fusion protein was eluted by adding 600 μl of PBS/T containing 100 mM glutathione, 1% Triton X-100, 58 mM Na2HPO4, 17 mM NaH2PO4, and 68 mM NaCl (pH 8.0). The purity of the GST-BRD7N protein in the supernatant was determined on a Coomassie-stained 10% SDS polyacrylamide gel and verified by Western blot using an anti-GST monoclonal antibody. The concentration of the protein was quantified using Bradford reagent (Bio-Rad; Hercules, CA), using BSA as the protein standard.
Antiserum Preparation Using Purified BRD7N
The purified GST-BRD7N protein was injected into rabbits to produce a polyclonal BRD7 antibody as described by Xiao et al. (2006) and Cao et al. (2007). Two New Zealand White rabbits were immunized intravenously with 200 μg of the GST-BRD7N protein per rabbit, followed by a second immunization of 100 μg per rabbit 4 weeks later. After the second injection, three additional injections (100 μg protein per injection) were performed at 2-week intervals. Three weeks after the last injection, sera were collected and used to test the anti-BRD7N antibody. To purify the prepared antisera specific to BRD7N, IgG was fractionated from the rabbit antiserum by precipitation with 40% saturated ammonium sulfate at 4C. After desalinization, the IgG solution was freed of anti-GST antibody using GST binding agarose (AG-GST) at 4C for 30 min. The flow-through was collected and stored at −20C for the use of ELISA, Western blot, and immunohistochemistry assays.
Embryo Tissues
Three 28- to 30-week gestation human fetuses were collected from termination of pregnancy material, with appropriate written consent and approval from the Central South University Health Authority Joint Ethics Committee and following national guidelines. Fetuses were collected into cold PBS (150 mM sodium chloride and 150 mM sodium phosphate, pH 7.2). Eleven kinds of tissues were collected for Western blot assay and immunohistochemistry. For immunohistochemistry, the tissues were fixed in 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.2) at 4C overnight. Each type of tissue was obtained from three different fetal subjects.
SDS–PAGE and Western Blot
Proteins were separated by SDS-PAGE according to Laemmli (1970), using a 4.5% stacking gel and 10% separating gel. After electrophoresis, the proteins were electronically transferred onto nitrocellulose membranes. Western blotting was performed as described by Towbin et al. (1979). The membranes were incubated with monoclonal anti-GST antibody, or monoclonal anti-c-Myc and the anti-BRD7N primary antibodies, respectively. After three washings, the membrane was incubated with horseradish peroxidase–conjugated goat antimouse or horseradish peroxidase–conjugated goat anti-rabbit secondary antibodies, respectively, and finally visualized using the Lumi-light Western blotting substrate kit (Roche; Indianapolis, IN), according to the manufacturer's instructions.
Immunohistochemistry
The tissues were deparaffinized in xylene and rehydrated through graded alcohols (70%, 80%, and 100% alcohol; 5 min each). Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 10 min. For antigen retrieval, sections were incubated in sodium citrate buffer (0.01 M, pH 6.0) for 15 min in a household microwave oven (600 W). After cooling to room temperature, slides were incubated with normal rabbit serum in TBS (1:5) for 30 min to reduce background nonspecific staining. The anti-BRD7N serum (1:400) was applied at 4C overnight. Secondary antibody (biotinylated goat anti-rabbit IgG) and streptavidinperoxidase conjugate (S-P kit; DAKO, Carpinteria, CA) were added according to the manufacturer's instructions. Color reaction was developed using diaminobenzidine chromogen solution (Liquid DAB; DAKO), and all slides were counterstained with hematoxylin. Negative control slides were included in the experiment.
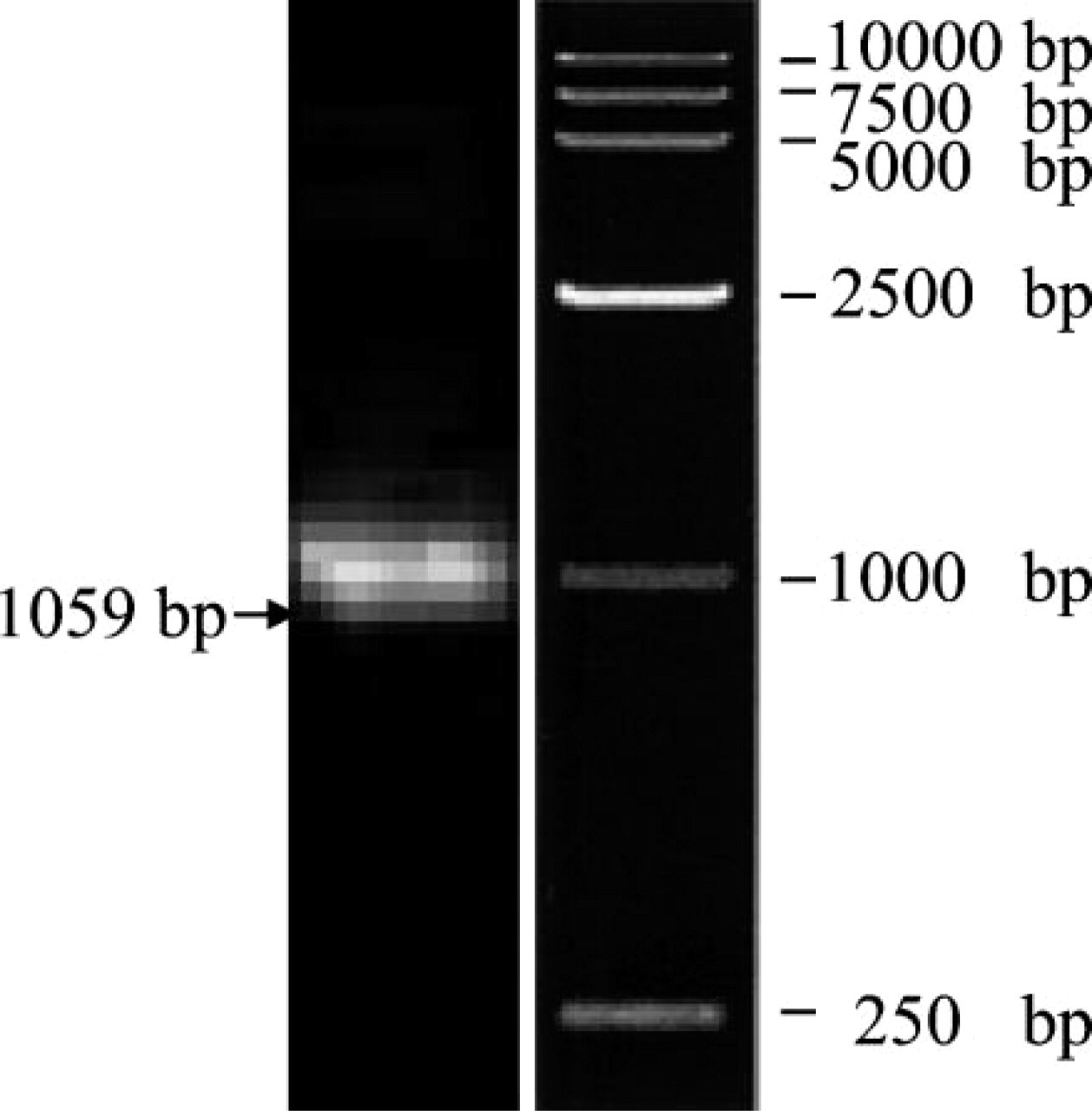
Identification of the positive clones by PCR amplification of the DNA fragment encoding BRD7N by using the same primer as amplification of BRD7N from cDNA library. Lane 1, BRD7N; Lane 2, DNA marker.
Immunoprecipitation Analysis
Protein extraction was performed with a lysis buffer consisting of 50 mM Tris-HCl (pH 7.5), 0.1% SDS, 1% Triton X-100, 150 mM NaCl, 1 mM dithiothreitol, 0.5 mM EDTA, 0.1 mM phenylmethylsufonyl fluoride, 12 mg/ml leupeptin, 20 mg/ml aprotinin, 100 mM sodium vanadate, 100 mM sodium pyrophosphate, and 1 mM sodium fluoride. Equal amounts of protein extraction were subjected to immunoprecipitation (IP) with antibodies against BRD7 or c-Myc. After all steps of co-IP, the immuno-complexes were eluted from the agarose beads by boiling in 2× SDS sample buffer with 0.2 M dithiothreitol and subjected to 12% SDS-PAGE. Proteins were transferred to nitrocellulose (PALL). The membrane was detected with antibody against BRD7 and c-Myc, respectively. The binding of the antibodies was detected using a chemiluminescence detection kit (Pierce Biotechnology; Rockford, IL).
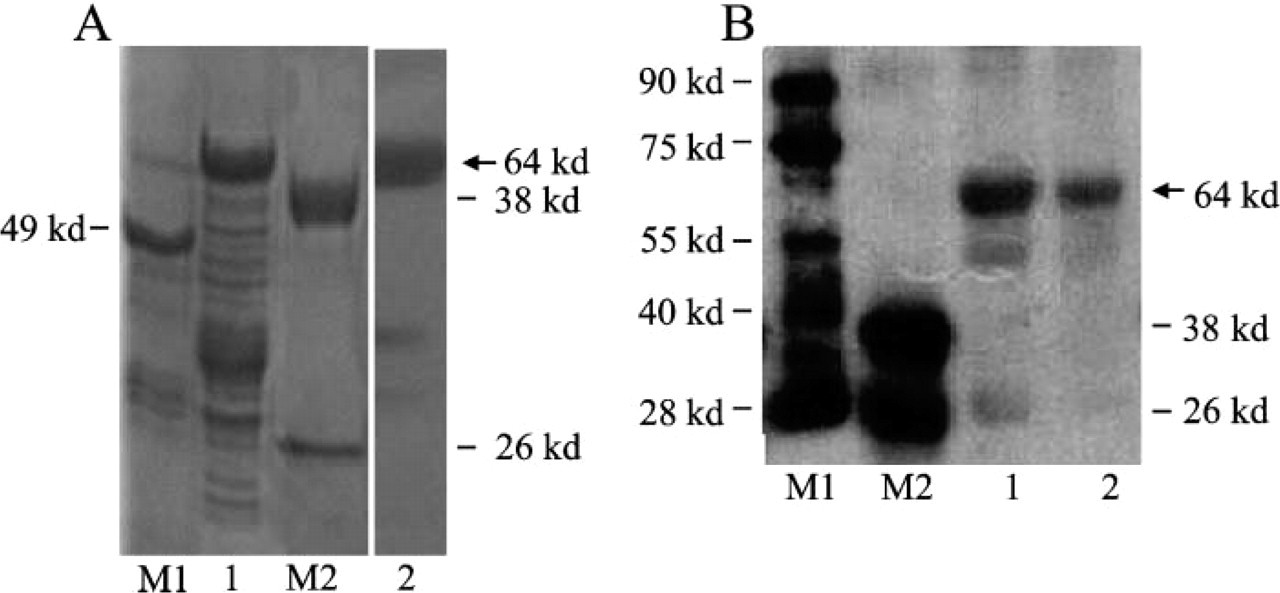
Analysis of the expression and purification of GST-BRD7N fusion protein in bacteria. (
Results
Construction of Expression Vector of pGEX-4T-BRD7N
The sequence (spanning from 54 to 1112 nt) encoding the N-terminal 353 amino acids of the BRD7 protein was amplified by PCR from a cDNA library of human fetal brain and cloned into BamHI/XhoI-digested pGEX-4T vector. The positive clones with recombinant construct were first identified by PCR with the same primer pairs as amplification of BRD7N from the cDNA library. As shown in Figure 1, a 1059-bp fragment (Lane 1) was amplified from a positive clone. DNA sequencing confirmed this fragment to be the coding region of the N-terminal 353 amino acids of the BRD7 protein.
Expression and Purification of the GST-BRD7N Fusion Protein
The prokaryotic expression vector pGEX-4T-BRD7N was introduced into the Escherichia coli BL21 according to standard protocol. The expression of the fusion protein was induced with IPTG at a concentration of 0.5 mM for 3.5 hr. As shown in Lane 2 of Figure 2A, GST-BRD7N was expressed efficiently in bacteria with a molecular mass of 64 kDa. Western blot results confirmed the expressed protein was GST-BRD7N (Figure 2B, Lane 3). After purifying by affinity chromatography, one band of the recombinant GST-BRD7N protein could predominantly be seen in the SDS-PAGE and Western blot assay, indicating that GST-BRD7N was highly purified (Figures 2A, Lane 4, and 2B, Lane 4). The total amount of purified protein was >2 mg in 600 μl. The concentration of purified protein was 1.5 mg/ml, and the protein remained intact with no proteolytic degradation during the purification procedure.
BRD7 Antibody Specifically Recognizes BRD7 Protein
Five immunizations with the GST-BRD7N protein were given to two rabbits. After two booster immunizations, test antisera containing BRD7 antibody was harvested from two rabbits 55 days after the primary immunization day. ELISA results showed that the titer of the antisera was 1:1,000,000 (Figure 3A). Western blot results showed that these antisera were able to recognize recombinant GST-BRD7N protein with a molecular mass of 64 kDa as that recognized by the anti-GST monoclonal antibody (Figure 3B). After two more booster immunizations, the antisera were harvested at 100 days after primary immunization and subjected to affinity purification. A GST-Sepharose 4B affinity chromatography column was used in the purification system. ELISA results showed that the final titer of the purified rabbit anti-BRD7 sera was 1:1,000,000, and the titer of flow through was 1:500 (Figure 4A). SDS-PAGE results showed that the estimated concentration of BRD7 antibody was 300 μg/ml for rabbit 1 and 150 μg/ml for rabbit 2. Preimmunized rabbit serum collected before the day of primary immunization was applied as a negative control. More importantly, the purified BRD7 antibody recognized the full-length recombinant fusion protein Myc-BRD7 with a molecular mass of 75 kDa in COS7 cells transfected with pCMV-Myc-BRD7 (Figure 5A, Lane 2), which corresponds to the molecular mass detected by monoclonal anti-c-Myc in COS7 cells transfected with pCMV-Myc-BRD7 (Figure 5A, Lane 5). As shown in Lanes 1 and 3 of Figure 5A, weak endogenous BRD7 protein was detected in COS7 and COS7 transfected with pCMV-Myc vector, respectively. Results in Lanes 4 and 6 of Figure 5A showed no endogenous c-Myc protein in COS7 and COS7 transfected with pCMV-Myc vector, respectively. To further study the specificity of BRD7 antibody, we carried out IP assays. As shown in Lane 2 of Figure 5B, the ectopically expressed BRD7-Myc protein was detected by c-Myc antibody in the immuno-complex precipitated with anti-BRD7 antibody. Similarly, BRD7-Myc protein was detected by BRD7 antibody in the immuno-complex precipitated with anti-c-Myc antibody. These data indicate (1) BRD7 antibody specifically recognizes BRD7 protein; (2) the ectopically expressed BRD7 protein shows one band in the Western blot assay; and (3) the doublets of bands in Western blot assay of Figure 5A may represent phosphorylation or other posttranslational modifications of endogenously expressed BRD7 protein or splice variants of the BRD7 gene. These results imply that the purified BRD7 antibody has high specificity and can be used for further detection of the BRD7 protein.
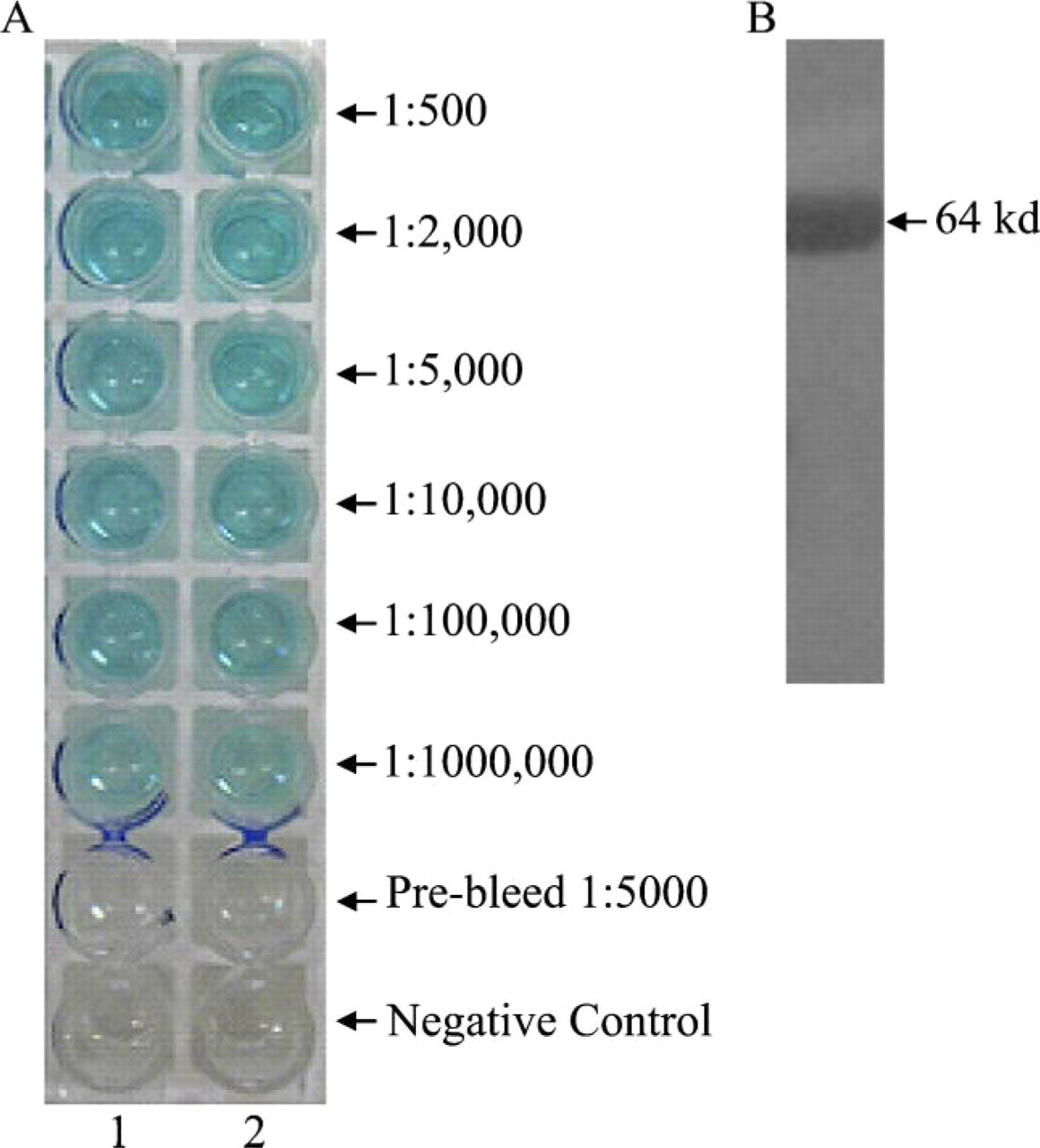
ELISA and Western blot analysis of rabbit anti-BRD7 sera harvested after three boost immunization. (
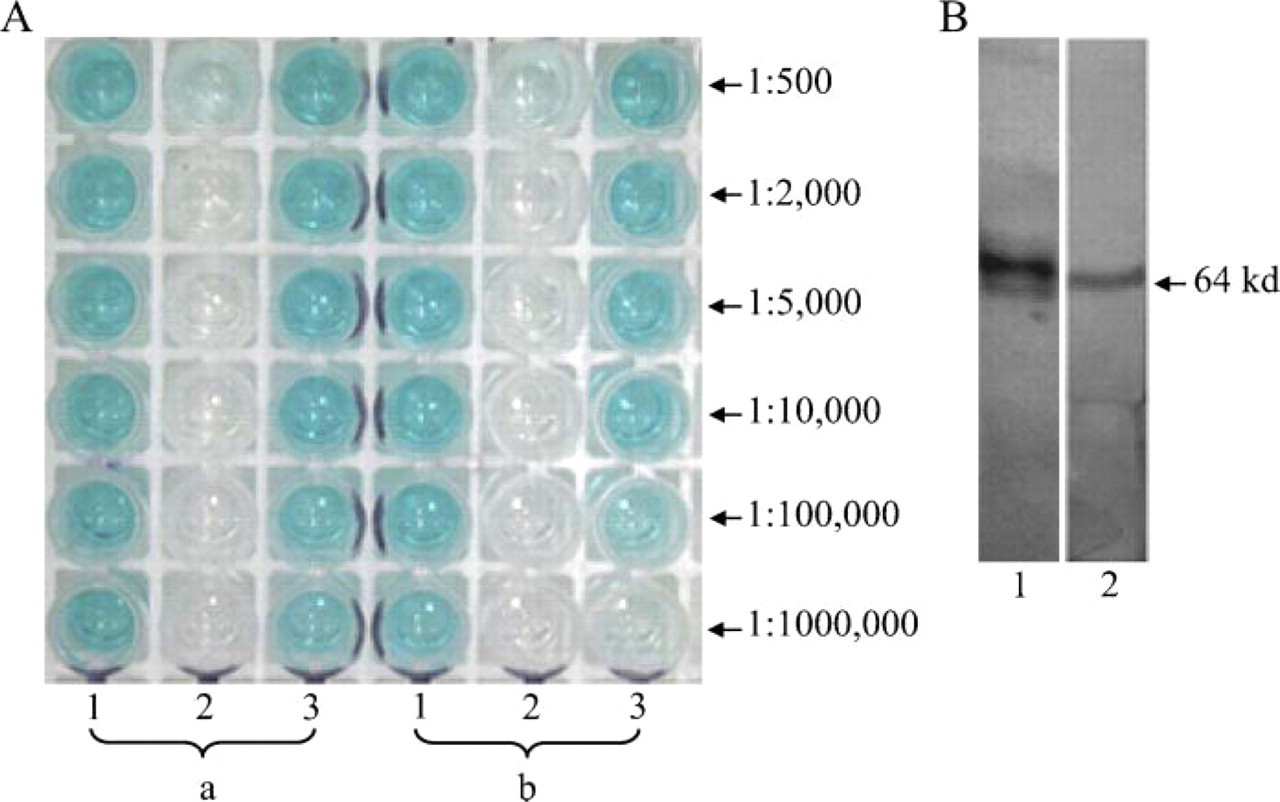
ELISA and SDS-PAGE analysis of the concentration of purified BRD7 antibody. (
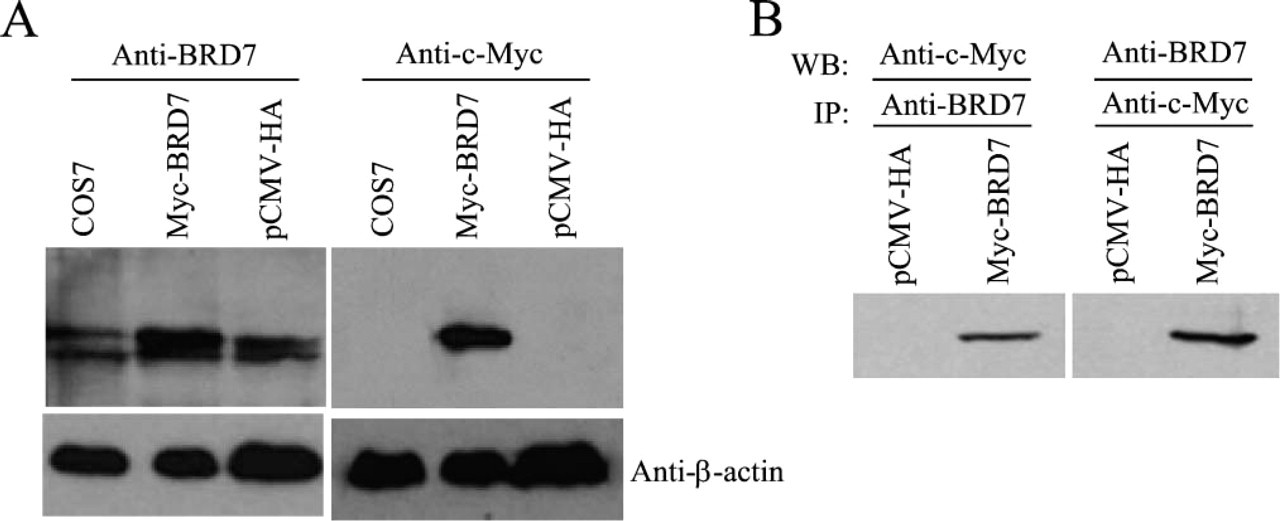
The purified BRD7 antibody specifically recognizes recombinant Myc-BRD7 fusion protein. (
BRD7 Protein Is Expressed in Most Human Fetus Tissues
With the prepared BRD7 antibody, we detected the tissue distribution of BRD7 in human fetus. In the Western blot assay, a high expression level of the BRD7 protein was seen in the cerebellum, cardiocyte, and pancreas (Figure 6, Lanes 3, 6, and 7). A moderate expression level of the BRD7 protein was seen in the intestine, liver, and kidney (Figure 6, Lanes 4, 8, and 11). A very low expression level of the BRD7 protein was shown in the human cerebrum (ectocinerea and alba), lung, spleen, and stomach (Figure 6, Lanes 1, 2, 5, 9, and 10). Immunohistochemistry assays showed similar results. Intensive nuclear staining of BRD7 protein was seen in the human cerebellum, epithelium of pancreas, intestines, liver, and kidney (Figures 7B, 7C, 7F, 7G, and 7J). Cardiomyocyte epithelium showed intensive cytoplasm expression of the BRD7 protein (Figure 7E). Very weak nuclear staining of the BRD7 protein was found in the human cerebrum, lung, spleen, and stomach (Figures 7A, 7D, 7H, and 7I).
Discussion
Bromodomain consists of a motif of 59–63 amino acids, flanked by a structurally conserved sequence to give an extended domain of ∼110 amino acids (Jeanmougin et al. 1997). It is a protein–protein interaction domain and has a specific binding affinity for acetylated lysines on N-terminal tails of histones (Dyson et al. 2001). Previous studies have shown that bromodomain is characteristic of proteins that regulate signal-dependent, but not basal, transcription during active proliferation through modulating chromatin remodeling or acetylation of histones, therefore facilitating accession of transcription factors to chromatin (Lavau et al. 2000; Horn and Peterson 2001). Accumulating evidence has shown that the genetic alterations of bromodomain genes contributed to the development of many human cancers and other disorders (French et al. 2001; Liedman and Zeleznik-Le 2001; Panagopoulos et al. 2001; Wolffe 2001). BRD7 has been confirmed as a new member of the bromodomain family. It is mapped to the human chromosome 16 at q11.1 to q12.2 and contains an open reading frame of 2317 bp encoding a 651 amino acid nuclear protein. eMotif analysis showed that BRD7 contains multiple phosphorated sites. The 129–237 amino acids of BRD7 is a functional bromodomain, which is commonly observed in the bromodomain family proteins (Jeanmougin et al. 1997). To date, very few functional studies on BRD7 have been carried out, which is mainly because of the unavailability of a specific BRD7 antibody. The purpose of this study was to generate the antibody for further study of the bio-functions of the BRD7 gene. To obtain abundant target protein to generate polyclonal antibodies against BRD7, a prokaryotic expression system was used. The GST-BRD7N fusion protein was highly and rapidly expressed in E. coli. Efficient expression of BRD7N could be achieved with 0.5 mM IPTG for 3.5 hr at 37C. The purified recombinant proteins were found to be immunogenic in rabbits and induced the production of polyclonal antibodies. A GST-Sepharose 4B affinity chromatography column was used to purify the polyclonal antibodies to ensure specificity. Western blot and ELISA results showed that this antibody had the advantage of high specificity and sensitivity.
More importantly, with these antisera, we analyzed BRD7 distribution in the human fetus by Western blot and immunohistochemistry assays and found that the BRD7 protein is highly expressed in the human cerebellum, cardiomyocyte, and pancreas, which is in agreement with the results of RT-PCR provided by Yu et al. (2000, 2001). The BRD7 protein is moderately expressed in the human liver, intestines, and kidney and weakly expressed in the human cerebrum (ectocinerea and alba), lung, spleen, and stomach. Severinsen et al. (2006) found widespread expression of BRD1 in mammalian brain tissue, as well as in the nucleus, perikaryal cytosol, and proximal dendrites of neurons in the adult rat, rabbit, and human central nervous system. Quantitative mRNA analysis in developing fetal pig brain showed spatiotemporal differences with high expression at early embryonic stages, with intense nuclear and cytosolic immunohistochemical staining of the neuroepithelial layer and early neuroblasts, whereas more mature neurons at later embryonic stages had less nuclear staining. The results suggest a role of BRD1 in neurodevelopment. LIN-49 is structurally most similar to the human bromodomain protein BR140 and is required for normal development of the mating structures of the adult male tail, for normal morphology and function of hindgut (rectum) cells in both males and hermaphrodites, and for maintenance of structural integrity in the hindgut and egg-laying system in adults (Chamberlin and Thomas 2000). Chamberlin and Thomas (2000) also proposed the bromodomain-containing protein LIN-49 functions in somatic development similar to the Drosophilatrx-G genes. Chua et al. (2005) found that the bromodomain protein GTE6 controls leaf development in Arabidopsis by histone acetylation at ASYMMETRIC LEAVES1. These findings indicate that the bromodomain-containing protein is not only associated with chromatin and in nuclear histone acetyltransferases and functions in numerous cellular processes including chromatin remodeling and transcriptional activation, but also plays an important role in development. The differential expression pattern of the BRD7 protein in human fetal tissues implies that BRD7 may play a critical role in the development of the human nervous system, cardiovascular system, digestive system, and urinary system. Further studies will be needed to clarify this function of the BRD7 protein.
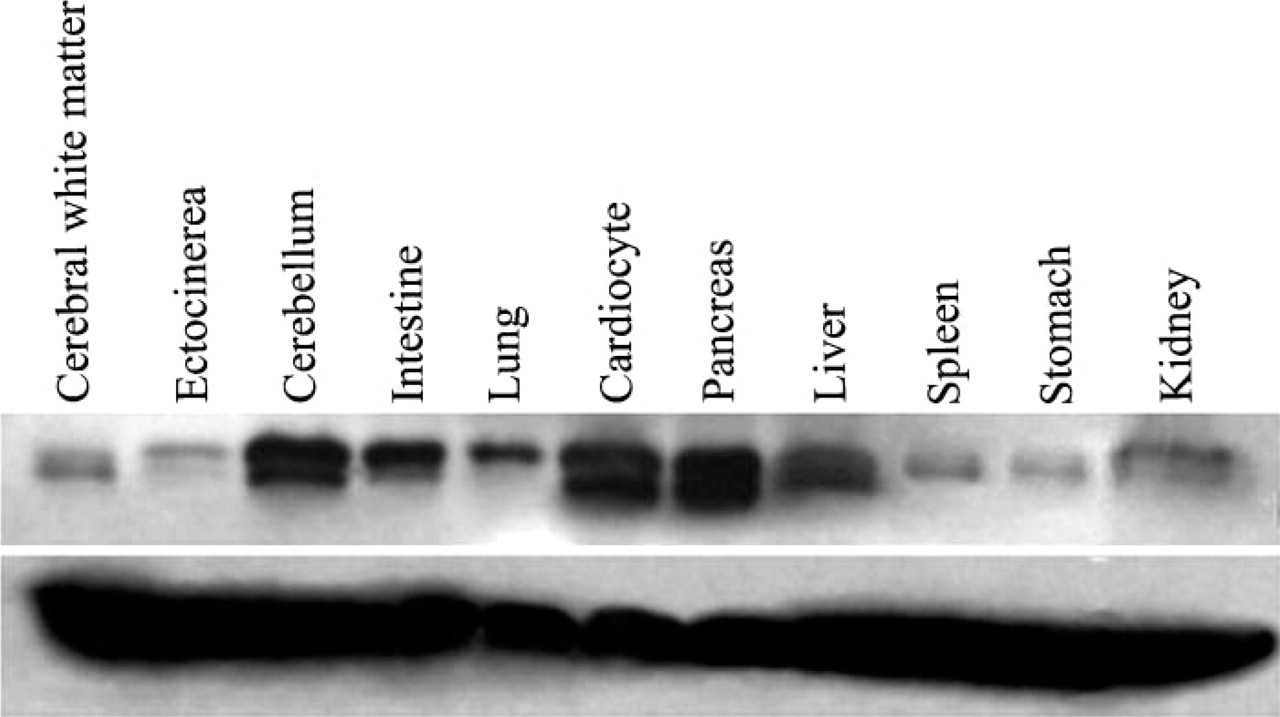
Detection of the endogenous BRD7 protein in various tissues of human fetus with purified BRD7 antibody by Western blot.
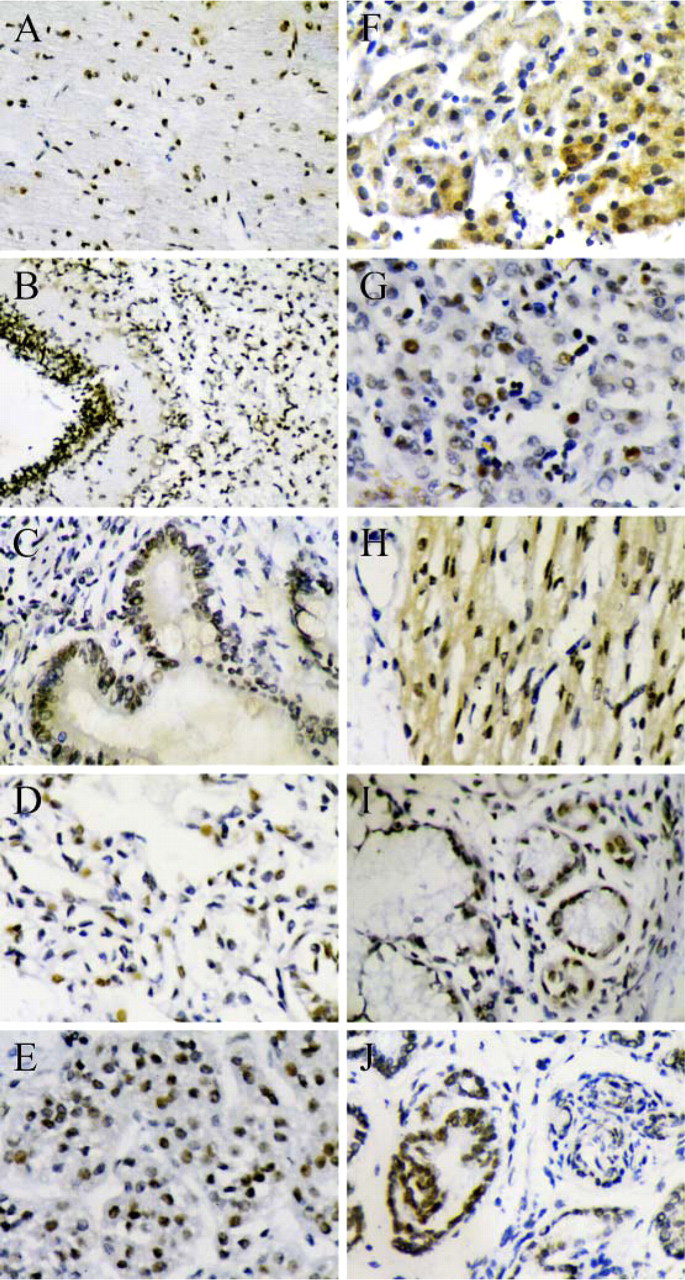
Detection of the endogenous BRD7 protein in various tissues of human fetus with purified BRD7 antibody by immunohistochemistry assay.
Footnotes
Acknowledgements
This work was supported by National Scientific Foundation of China Grants 30400528, 30470367, and 30400238, Natural Science Foundation of Hunan Province Grant 06JJ20013, and Chinese Key Program of National Scientific Foundation Grant 30330560.
