Abstract
α1-M
α1m has a strong tendency to form complexes with other proteins. In human and rat, approximately half of the plasma α1m exists as high molecular weight forms, which are too large to escape from the circulation via glomerular filtration. Curiously, the α1m complexes that have been found thus far in human plasma are not found in rat plasma, and vice versa. α1m has been shown to be complexed with IgA, prothrombin, and albumin in humans (Tejler et al. 1976; Grubb et al. 1983; Berggård et al. 1997) and with fibronectin and the α2-macroglobulin homologue α1-inhibitor-3 in rats (Falkenberg et al. 1990,1994). The sites of formation and metabolism of these high molecular weight α1m complexes are unknown, but the α-inhibitor-3-α1m complex was shown to be rapidly cleared from the circulation (Falkenberg et al. 1995).
The gene for α1m has been isolated and characterized (Diarra-Mehrpour et al. 1990; Vetr and Gebhard 1990) and encodes two tandemly arranged proteins, α1m and bikunin (Kaumeyer et al. 1986). Bikunin is the light chain found in the plasma proteins inter-α-inhibitor, pre-α-inhibitor, and heavy chain 2-bikunin (Enghild et al. 1989). The bikunin subunit is responsible for the proteinase inhibitory activities of these proteins. Bikunin also apparently stimulates the growth of normal endothelial cells (McKeehan et al. 1986) and fibroblasts (Perry et al. 1994). Moreover, inter-α-inhibitor has been shown to be an important component for the formation of the extracellular matrix (Chen et al. 1992; Blom et al. 1995).
When synthesized, a precursor protein is translated in which α1 m and bikunin are connected by a short peptide. The precursor is cleaved into the two mature proteins before secretion (Bratt et al. 1993,1994; Thøgersen and Enghild 1995). The co-expression of α1m and bikunin is conserved in all species examined to date (from fish to human) (Hanley and Powell 1994; Leaver et al. 1994). This suggests that the two proteins are involved in common processes. However, thus far no functional relation has been shown between α1m and bikunin in plasma. α1m has been extensively studied in blood, but although different immunoregulatory properties have been reported for α1m (for reviews see Åkerström and Lögdberg 1990; Åkerström 1992), the physiological function of the protein is still unclear.
α1m is generally considered to be a plasma protein, but the result reported in this study reveals that α1m is present in the extracellular matrix of many tissues. Moreover, α1m is not generally synthesized in the tissues but mainly in the liver, and is probably transported to the tissues across the endothelial cell membrane by an unknown mechanism. In addition, both high and low molecular weight forms of α1m are widely distributed in the matrix of tissues, suggesting a possible function in these interstitial tissue domains.
Materials and Methods
Materials
The general serine-proteinase inhibitor 3,4-dichloroisocoumarin (DCI) and the general cysteine-proteinase inhibitor N-[[[N[-[(-3-trans-carboxyl-oxiran-2-yl)-carbonyl]
SDS-polyacrylamide Gel Electrophoresis
Aliquots of the supernatant from each of the homogenized tissue samples were subjected to SDS-PAGE under nonreducing conditions. This was performed in 5-15% linear gradient gels using the glycine-2-amino-2-methyl-1,3-propanediol-HCl system described by Bury (1981). Molecular mass standards were rat α1-inhibitor-3 (180 kD) mixed with Bio-Rad (Hercules, CA) low molecular weight mass standard, visualized by staining with Coomassie Brilliant Blue.
Western Blot Analysis
Proteins were separated by SDS-PAGE and transferred to polyvinylidene diflouride membrane (PVDF) as described by Matsudaira (1987). All subsequent steps were performed on a rotating table at 25C. The membrane was equilibrated in 20 ml of 10 mM Tris-HCl, 0.15 mM NaCl, and 0.05% NP-40 (TSN buffer) containing 1% bovine albumin for 30 min before the addition of 10 μl α1m antiserum (1:2000 dilution). After 1 hr the membrane was washed twice in TSN buffer and twice in 10 mM Tris-HCl, 10 mM NaCl, pH 7.5 (TS buffer) for 5 min each. Then 20 ml of TSN buffer containing 10 μl anti-rabbit IgG-alkaline phosphatase conjugate (1:2000 dilution) was allowed to react for 30 min before the membrane was washed as above. The substrates bromochloroindolyl phosphate and nitroblue tetrazolium were added and color was allowed to develop in the dark for up to 30 min. The reaction was stopped with 5 mM EDTA in TS buffer.
Perfusion of Rat Tissues
Sprague-Dawley rats were anesthetized by IP injection with pentobarbital. After sedation they were opened and one lung was removed. A blood sample was taken from the left ventricle of the heart, and the animal perfused via the same needle, using 500 ml of 0.9% normal saline. Perfusion of the lungs was performed by infusion of 100 ml of 0.9% saline through the right ventricle of the heart after cutting the abdominal aorta. The organs were removed, weighed, and homogenized in a high-speed homogenizer (Cole-Parmer Instruments; Niles, IL) in 100 mM NaCl, 50 mM (N-[2-hydroxyethyl]piperazine-N'-[2-ethanesulfonic acid] (HEPES), 100 mM 3,4-dichloroisocoumarin (DCI), 500 μM 1,10-phenanthroline, and 50 μM N-[[[N[-[(-3-trans-carboxyloxiran-2-yl)-carbonyl]
Immunohistochemistry
Human tissues were fixed in 10% formalin followed by routine processing and paraffin embedding. Sections were then labeled for antigen using an indirect immunoperoxidase method (Milde et al. 1989; Randell et al. 1991) with a biotinylated goat anti-rabbit IgG and streptavidin-horseradish peroxidase. To reduce background staining, the sections were incubated in 1% H2O2 in methanol to inactivate endogenous peroxidases and 10 mM borohydride to block aldehydes. Nonspecific binding was blocked by incubation with 5% normal goat serum (NGS), 5% milk, and 1% BSA in PBS. Primary and secondary antibody dilutions were determined empirically and made in PBS with 1% milk plus 1% BSA (milk was not included in the streptavidin solution). The slides were developed using diaminobenzidine (10 mg diaminobenzidine, 50 ml 0.05 M Tris-Cl, pH 7.6, 100 ml 3% H2O2) and counterstained with hematoxylin.
As a control, one serial section on each slide was incubated with preimmune rabbit serum or with anti-α1m anti-bodies absorbed by immunosorbent affinity chromatography. The latter was done by immobilizing 1 mg of purified α1m to 1 ml CNBr-activated Sepharose according to the manufacturer's instructions (Sigma). The affinity gel was then incubated for 24 hr with 1 ml of primary antibodies at two times the concentration used for immunolabeling. The supernatant was then used for immunohistochemistry.
Reverse Transcriptase PCR
The presence of mRNA in the tissue was examined by extracting RNA essentially as described previously (Chomczynski and Sacchi 1987) followed by RT-PCR. This method involves a single acid guanidium thiocyanate-phenol-chloroform RNA extraction followed by an acid phenol extraction and incubation with RNase-free DNase. The sample is subsequently extracted with acid phenol and α1m-bikunin RNA is amplified with specific primers using a thermocycler (Perkin-Elmer Cetus GeneAmp PCR system 9600; Norwalk, CT) and appropriate reagents (Gene Amp, EZ rTth RNA PCR kit). The α1m-specific primers were 5'tgctgggagagggcgctaca3' (forward) and 5'gcccatgcagccgccgtact 3' (reverse). The expected 593-bp product was generated by first incubating at 30 min at 60C and 1 min at 94C followed by 40 cycles at 94C for 15 sec and 70C at 30 sec. As a positive control, RTPCR was performed on all tissues using primers specific for glyceraldehyde 3-phosphate dehydrogenase. The product was generated by first incubating at 30 min at 60C and 100 sec at 94C followed by 40 cycles at 94C for 15 sec and 69C at 30 sec. Presence of the predicted 336-bp product ensured that negative results for α1m-bikunin RNA were not due to RNA degradation in the tissue.
Results
Identification of α1m in Perfused Rat Tissue
Perfused rat tissues were homogenized and insoluble debris was removed by centrifugation. Aliquots of the supernatants were analyzed by SDS-PAGE, followed by Western blotting using anti-rat α1m antiserum. The distributions of the different α1m species in rat plasma and in the perfused rat tissues were compared (Figure 1A). In plasma, several α1m-immunoreactive bands with apparent molecular masses of 28, 43, 75, 110, 180-210, and 240 kD were evident. These bands specifically reacted with rat α1m antisera (Figure 1B). The 28-kD band is free uncomplexed rat plasma α1m and the 240-kD and 180-210 kD bands represent the fibronectin-α1m and α1-inhibitor-3-α1m complexes, respectively (Falkenberg et al. 1990,1994). The 43-kD band has not been characterized. The 240- and 180-210 kD bands were seen in most tissues. It is likely that these also represent fibronectin-α1m and α-inhibitor-3-α1m. The 180-210 kD α-inhibitor-3-α1m complex was absent from the liver, and the 240-kD fibronectin-α1m complex was absent from the lung and pancreas. As discussed previously, the 110- and 75-kD bands are most likely degradation products of the two complexes (Falkenberg et al. 1990,1994).
Some tissues contained forms of α1m that were not found in plasma. The liver contained five α1m isoforms with molecular masses between 40 and 50 kD. The 50-kD α1m isoform was also present in the kidney, and the heart contained a slightly smaller 45-kD isoform. The pancreas and the eye contained more free α1m relative to the larger complexes than the other tissues. The 18-kD band observed in the eye homogenate represented a nonspecific reaction because it was present even when the primary antiserum was omitted (not illustrated). The brain contained only the 110-kD band. Western blotting of the same samples using an antibody against a control plasma protein yielded the expected band in plasma, whereas other tissue samples were negative (not shown). This shows that the α1m bands in the tissues did not originate from plasma contamination.
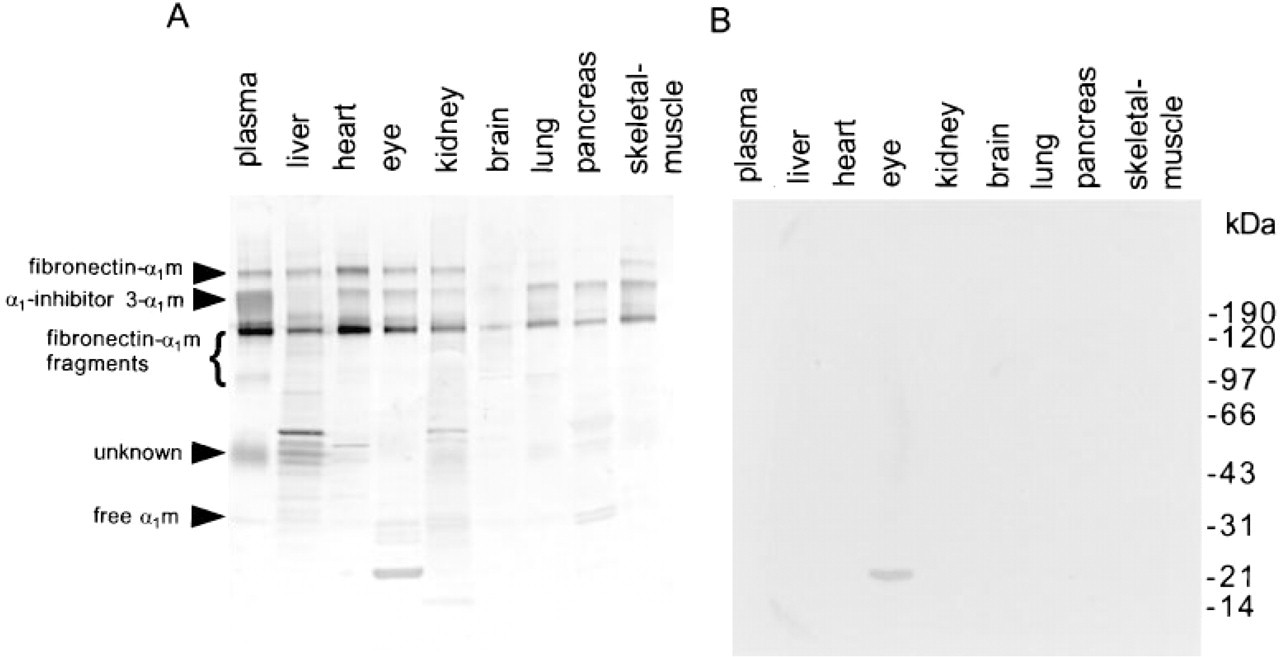
Identification of α1m in rat tissues. A rat was perfused and the organs were removed and homogenized in 50 ml of a buffer (100 mM NaCl, 50 mM HEPES, containing 100 mM DCI, 500 mM 1,10-phenanthroline, and 50 mM E-64). The homogenized organs were centrifuged and portions of the supernatants were subjected to nonreducing SDS-PAGE followed by immunoblotting with anti-rat α1m antiserum
Immunochemical Localization of α1m Proteins in Human Tissues
To investigate the tissue distribution of the α1m proteins, we obtained paraffin-embedded sections of human tissue, including heart, liver, lung, and kidney.
These tissue sections were labeled with antisera to α1m and detected using a biotin-streptavidin-horseradish peroxidase technique. The studies revealed a granular, cytoplasmic labeling for α1m in the liver (Figure 2A). This is expected because hepatocytes have been shown to be the primary site of α1m synthesis (Salier et al. 1987,1993; Lindqvist et al. 1992; Chan et al. 1995). In addition, labeling for α1m was present in the proximal tubules of the kidney (Figure 2G). This result is also expected because α1m is known to be filtered by the glomeruli and reabsorbed in the tubules. Immunochemical staining of the heart and lung also demonstrated positive labeling for α1m, supporting the results of Western blot analysis of these tissues (Figure 1). Specifically, labeling for α1m was found in the ex-tracellular matrix of blood vessels in the heart (Figure 2B), lung (Figure 2F), liver, and kidney (not illustrated). Furthermore, α1m was also present on the cell surface of cardiocytes in the heart (Figure 2C) and within the matrix of alveolar septa and airways in the lung (Figures 2E and 2F). No labeling was observed when nonimmune serum was substituted for the primary antibody or when antibodies were absorbed with purified α1m (Figures 2D and 2H). These results indicate that the labeling was specific for α1m.
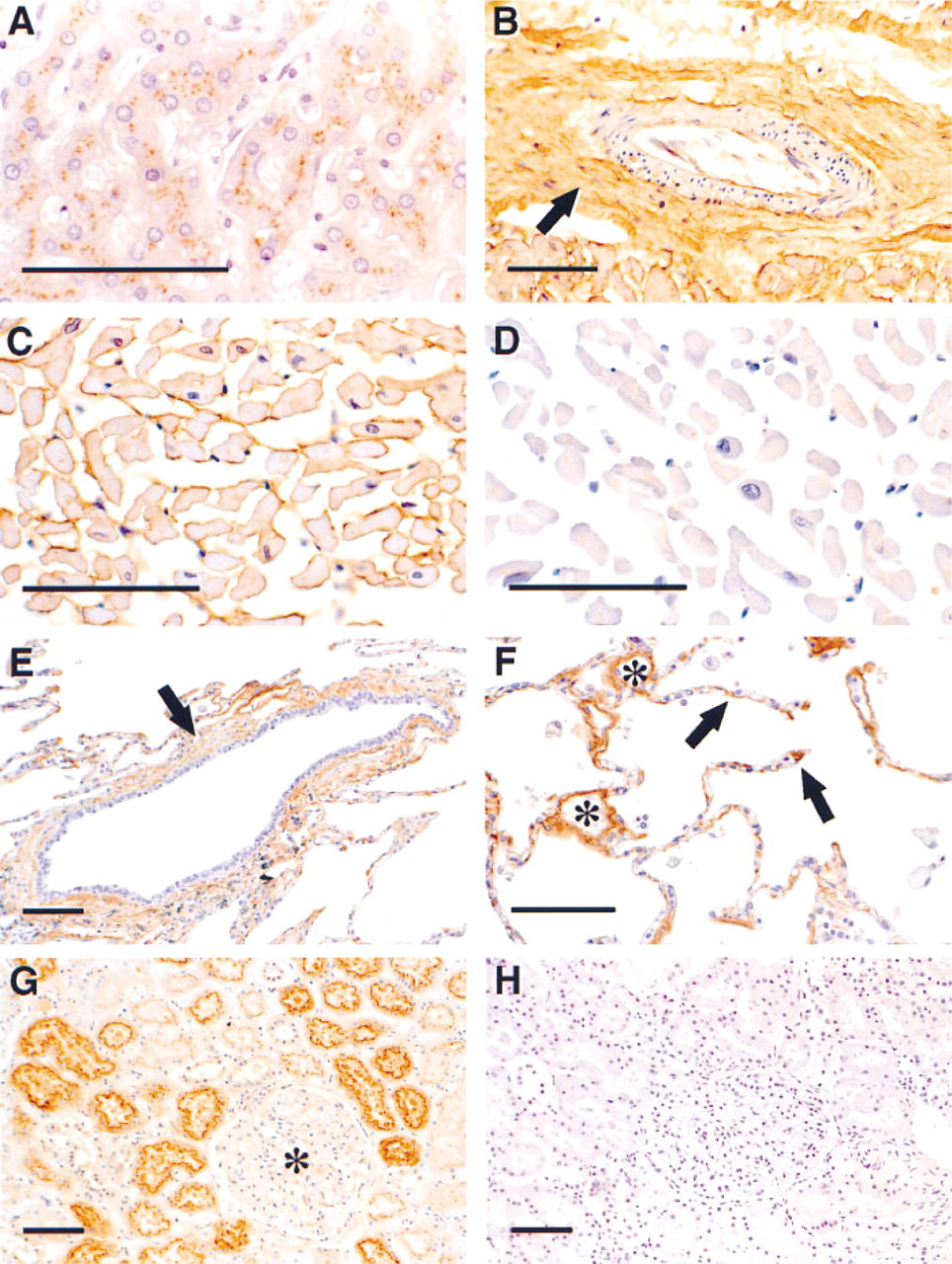
Immunochemical localization of α1m in human tissues. Sections of human kidney, heart, liver, and lung were labeled with an antibody to α1m or with nonimmune serum, and the antibody was detected using a biotin-streptavidin-horseradish peroxidase technique.
Reverse Transcriptase PCR
The results described above show that α1m is distributed throughout the body. To determine if the protein was made locally, we examined tissue samples for the presence of α1m mRNA. Appropriate primers were designed as described above and reverse transcriptase PCR was performed on mRNA extracted from the following human tissues: liver, kidney, lung, heart, spleen, muscle, thyroid, pancreas, and adrenal gland. Figure 3 shows that only the liver and the pancreas produced α1m RNA. The 0.6-
Discussion
Previous studies have demonstrated α1m immunoreactivity in skin, colon, testis, and ovary (Bouic et al. 1984; Odum and Nielsen 1994,1997). We have shown that α1m is also present in many other tissues, including liver, heart, eye, kidney, brain, lung, pancreas, and skeletal muscle. An intense granular intracellular labeling was observed in hepatocytes, consistent with production and secretion by these cells. Intracellular α1m labeling was also observed in the proximal tubule cells of the kidney. This was expected because the small size of α1m promotes uptake, reabsorbance and degradation by the kidneys (Strober and Waldmann 1974; Tejler et al. 1978; Salier et al. 1987,1993; Lindqvist et al. 1992; Chan et al. 1995). Other α1mcontaining tissues did not show any intracellular labeling but instead demonstrated widespread staining in the interstitial connective tissue matrix. For example, the matrix of blood vessels and the matrix of alveolar septa and airways in the lung were labeled by anti-α1m. Most of the known plasma proteins, except the immunoglobulins, are synthesized by hepatocytes. In some cases, additional synthesis of plasma proteins takes place in other cell types (Lamontagne et al. 1985; Campbell and Law 1992). The synthesis by these cells is often minor compared to the synthesis by the hepatocytes. α1m has been reported to be synthesized by the liver (Åkerström and Landin 1985; Salier et al. 1987,1993; Lindqvist et al. 1992; Chan et al. 1995). Other organs, such as kidney in rat and plaice (Karsten et al. 1986; Leaver et al. 1994), stomach in pig (Tavakkol 1991), blood cells in plaice (Leaver et al. 1994), and pancreas in humans (Itoh et al. 1996), have also been suggested to produce α1m. In the present study, done on human tissue, α1m mRNA was detected in the liver and, to a lesser extent, in the pancreas, suggesting that human α1m is mainly produced in the liver, but with a minor contribution from the pancreas. The presence of α1m in pancreas is presently being investigated by immunohistochemical and biochemical analyses (Lögdberg et al., manuscript in preparation). All other organs in which staining by anti-α1m was demonstrated lacked α1m mRNA. The α1m present in the interstitial connective tissue matrix of different organs is therefore most likely not synthesized locally but is transported to these compartments from blood. This is supported by the fact that although the α1m-bikunin mRNA content is elevated during inflammation, the total plasma concentrations and urinary excretion of bikunin and α1m remain unchanged (Falkenberg et al. 1997).
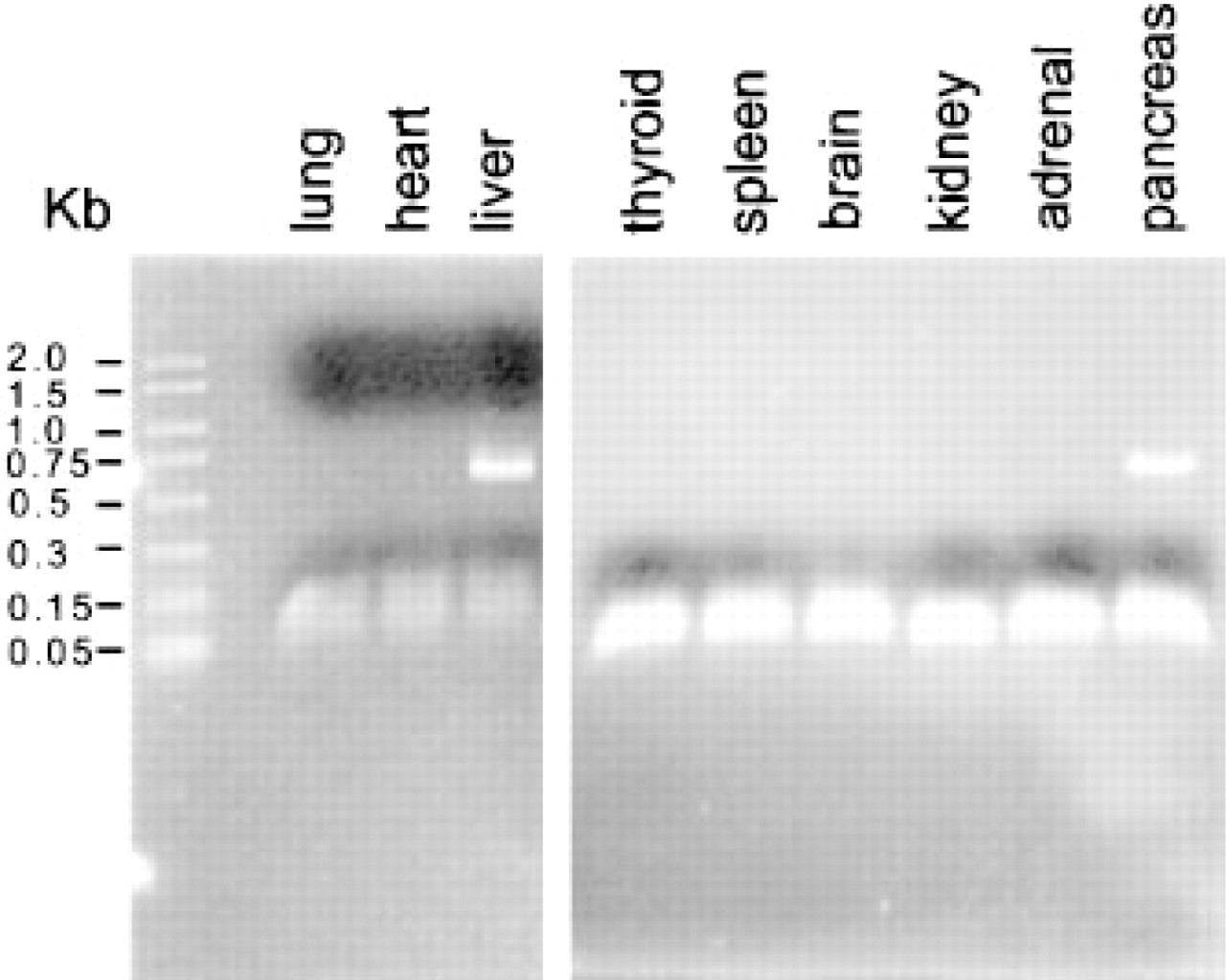
Expression of α1m. Total RNAs were extracted from the indicated tissues. The extracted RNA was reverse-transcribed and amplified by RT-PCR using primers specific for α1m mRNA. The PCR products were electrophoresed in a 2% agarose gel and stained with the ethidium bromide. α1m is secreted by the liver and the 593-bp product was detected as expected. The mRNA was also detected in the pancreas, but all other tissues examined did not appear to contain α1m mRNA.
α1m is found in plasma both as a monomer and in complexes with other plasma proteins. These plasma complexes are mainly covalent and have previously been analyzed (Grubb et al. 1983; Falkenberg et al. 1990,1994; Berggård et al. 1997). The biosynthesis and the site of formation of the high molecular weight α1m complexes are not known. Many low molecular mass plasma proteins are rapidly eliminated from plasma by glomerular filtration. In some cases, the low molecular weight proteins are complex-bound to larger proteins in plasma, resulting in a slower elimination time (Cowan et al. 1990). In accordance with this, it has been speculated that one purpose of the complex formation between α1m and other plasma proteins may be to prevent loss of α1m by filtration through the kidney glomeruli (Berggård et al. 1997). The molecular mass of each protein also determines the extent of passive exchange between the intercellular space and plasma. Although larger proteins are less likely to passively penetrate the endothelium of blood vessels, we detected higher levels of high molecular weight α1m, relative to free α1m in the tissues. In addition, the brain contained high molecular weight α1m, although plasma proteins are unable to cross the blood-brain barrier. These findings suggest that the transport from the blood to the tissues is receptormediated.
The results reported here indicate that the biodistribution of α1m is not restricted to blood. In fact, α1m is widely distributed in the interstitial matrices and appears to form tissue-specific complexes with other proteins that are not found in plasma. Therefore, α1m could be regarded as a matrix protein in addition to its previous categorization as a plasma protein. Several possible functions for tissue α1m can be envisioned. As speculated previously (Odum and Nielsen 1994,1997; Falkenberg et al. 1997), it is possible that the immunosuppressive effects of α1m, i. e., the inhibition of lymphocyte proliferation and granulocyte chemotaxis and migration, protect “bystander” interstitial tissue from the immune and inflammatory reactions. Furthermore, α1m is a member of the lipocalin superfamily. Most of the lipocalins are carriers of small hydrophobic prosthetic groups such as retinol, pheromones, odorants, bilirubin, and steroids (Flower 1996). It can be speculated that α1m is a transporter of such small prosthetic groups from blood to tissues. This would be in agreement with α1m being synthesized mainly in the liver but found in most organs. Finally, the specific presence of α1m around blood vessels and in lung airways and alveolar septa resembles that of extracellular superoxide dismutase (EC-SOD), a scavenger of the superoxide anion (Oury et al. 1994). This protein is believed to be important in the defense against superoxide-mediated tissue damage. A similar protective role for α1m in these domains is possible, as an immunoprotective agent, as a potential scavenger of small toxic substances, or both.
Footnotes
Acknowledgements
Supported by grants from the Swedish Medical Research Council (project no. 7144), King Gustav Vs 80-year Foundation, the Medical Faculty at Lund University, the Swedish Society for Medical Research, the Royal Physiographic Society in Lund, the Foundations of Crafoord, Greta Johan Kock, and Alfred Österlund, the Swedish Rheumatism Association, and National Institutes of Health grant HL-49542.
