Abstract
Cyclic nucleotide phosphodiesterase 11A (PDE11A) is the newest member in the PDE family. Although the tissue distribution of PDE11A mRNA has been shown, its protein expression pattern has not been well studied. The goal of this report is to investigate the distribution of PDE11A proteins in a wide range of normal and malignant human tissues. We utilized a polyclonal antibody that recognized all four PDE11A isoforms. Its specificity was demonstrated by Western blot analysis on a recombinant human PDE11A protein and native PDE11A proteins in various human tissues. Immunohistochemistry showed that PDE11A is widely expressed. Various degrees of immunoreactivity were observed in the epithelial cells, endothelial cells, and smooth muscle cells of all tissues examined. The highest expression was in the epithelial, endothelial, and smooth muscle cells of the prostate, Leydig, and spermatogenic cells of the testis, the tubule epithelial cells in the kidney, the epithelial and endothelial cells in the adrenal, the epithelial cells and macrophages in the colon, and the epidermis in the skin. Furthermore, PDE11A expression was also detected in several human carcinomas. Our results suggest that PDE11A might be involved in multiple physiological processes in various organs via its ability to modulate intracellular cAMP and cGMP levels.
C
PDE11A is the most recent PDE to be cloned. It is most similar to PDE5, based on sequence similarity, but unlike PDE5 it is a dual-substrate enzyme, hydrolyzing both cAMP and cGMP (Fawcett et al. 2000). Four splice variants have been identified—PDE11A1, A2, A3, and A4—that encode proteins of 490, 576, 684, and 943 amino acids, respectively (Fawcett et al. 2000; Yuasa et al. 2000; Hetman et al. 2000). These predict molecular mass of 56, 66, 78, and 105 kDa for PDE11A1–A4, respectively. The four isoforms contain the same carboxyl termini, whereas PDE11A2, A3, and A4 possess unique amino termini (Hetman et al. 2000; Yuasa et al. 2000). Analysis of the steady-state RNA levels have indicated that human PDE11A is most abundantly expressed in the prostate, followed by the testis, liver, skeletal muscle, pituitary gland, pancreas, and kidney (Fawcett et al. 2000; Yuasa et al. 2000). Although recent studies have provided Western blot analyses in some tissues (Fawcett et al. 2000), the cellular localization of PDE11A is not known. We used immunohistochemistry (IHC) to detect PDE11A (all four splice variants) in a variety of normal and malignant human tissues showing its primary cellular localization in normal epithelial, smooth muscle, and endothelial cells, as well as in a variety of carcinoma cells.
Materials and Methods
PDE11A4 Recombinant Protein Generation
Human PDE11A4 open reading frame was isolated from human prostate poly A+ RNA obtained from BD Biosciences (Palo Alto, CA). The forward primer used to amplify the sequence was 5′-TGAGCCAGAGAAGGAAGGGG-3′ and the reverse primer was 5′-TGACCTGGAGGTTTAGTTCCTGTC-3′ according to the publicly available sequence (Genebank accession number AB 036,704). The reverse transcription reaction was carried out using Advantage RT for PCR kit (BD Biosciences), and the PCR reaction was carried out using Advantage HF PCR kit (BD Biosciences) according to manufacturer's instructions. A single product of 3.0 kb was amplified and cloned into pGEM-EASY vector (Promega; Madison, WI). The cDNA was sequence verified and cloned into pBlueBacHis2B (Invitrogen; Carlsbad, CA) for protein expression in Hi5 insect cells (Invitrogen) using baculovirus. The expressed protein was partially purified by batch binding to Ni-NTA resin. The eluted recombinant protein was aliquoted and stored at −80C.
PDE Assay
The effects of several known PDE inhibitors on PDE11A4 were studied using PDE assays described previously (Qiu et al. 2003). Briefly, the assays were conducted in 5 mM MgCl2, 15 mM Tris HCl (pH 7.4), 0.5 mg/ml bovine serum albumin, 30 nM [3H] cGMP (Amersham Pharmacia Biotech; Piscataway, NJ). The total volume of the assay was 100 μl. The reaction mixture was incubated at 30C for 90 min and then stopped by boiling for 1 min, followed by cooling on ice. The resulting [3H] 5′-mononucleotides were further converted to uncharged [3H] nucleosides by adding 25 μl of 1 mg/ml snake venom (Ophiophagus hannah) and incubating at 30C for 10 min. This reaction was stopped by the addition of 1 ml AG1-X2 resin slurry (Bio-Rad; Hercules, CA). After centrifugation at 3000 × g, the [3H] in a 200-μl aliquot of the supernatant solution was quantified by liquid scintillation counting. The amount of PDE11A4 used was such that the hydrolysis of substrates in the peak fractions did not exceed 15% of the total, ensuring that the amount of product increased linearly with respect to time. Percent inhibition was used to generate IC50 values by fitting the data to a non-linear regression model in GraphPad Prism (Graphpad Software Inc.; San Diego, CA). Sildenafil was extracted from Viagra tablets (Pfizer; Groton, CT) and used as the citrate salt. All other PDE inhibitors were purchased from Sigma-Aldrich (St Louis, MO).
Western Blot Analysis
An affinity-purified rabbit polyclonal antibody (PD11A-112AP) was purchased from FabGennix, Inc. (Shreveport, LA). This antibody was generated against a sequence located at the C terminus (VATNRSKWEELHQKR) of human PDE11A that was common to all known splice variants. Human cytosolic proteins from various tissues processed as described by Boolell et al. (1996) were obtained from Analytical Biochemical Services, Inc. (Wilmington, DE). Hi5 cell lysates, human cytosolic proteins (100 μg/lane), and recombinant PDE11A4 were fractionated on 4–12% Bis-Tris SDS mini-gel (Invitrogen). Proteins were transferred to nitrocellulose membranes (0.45 μm; Invitrogen) using a mini-transblot system (Bio-Rad). The membrane was blocked with 5% blocking agent (Amersham Pharmacia Biotech) in TBS at 4C for 2.5 hr and probed PD11A-112AP at 1:500 dilutions in 1% blocking agent in TBS at 4C for overnight. The excess antibody was removed by washing the membrane in 1% blocking solution. Following the washes, alkaline phosphatase-conjugated sheep anti-rabbit IgG antibody diluted 1:10,000 in 1% blocking solution was added to the membrane and incubated for 2.5 hr at 4C. After washing off the excess secondary antibody, the blot was developed with the BCIP/NBT kit from Calbiochem (La Jolla, CA).
Immunohistochemistry
Normal (NormalGrid) and malignant (TumorGrid) human tissue slides were purchased from Biomeda (Foster City, CA). Table 2 lists all of the tissues present on this slide. Methods for IHC have been previously described (D'Andrea et al. 2003). Briefly, tissue sections on microscopic slides were dewaxed and re-hydrated and then processed for IHC. Slides were microwaved in Target buffer (DAKO; Carpinteria, CA), cooled, placed in phosphate-buffered saline (pH 7.4, PBS; Zymed Labs, South San Francisco, CA), and treated with 3.0% H2O2 for 10 min at room temperature. All incubations (30 min) and washes were performed at room temperature. Normal blocking serum (Vector Labs; Burlingame, CA) was placed on the slides for 10 min. After a rinse in PBS, sections were incubated with the rabbit anti-PDE11A antibodies (1:100; FabGennix, Inc.) We also applied the monoclonal anti-vimentin antibodies (DAKO) as a positive control for tissue antigenicity as well as the replacement of the primary antibody with the antibody dilution buffer as a negative control. Furthermore, we preabsorbed the anti-PDE11A with recombinant antigen (35X titer excess) overnight at 4C before placement on the slide as an additional control. Slides were then washed in PBS and treated with biotinylated secondary antibodies (Vector Labs). After washing in PBS, the avidin-biotin-horseradish peroxidase complex reagent (Vector Labs) was added. Slides were washed and were treated with 3,3′-diaminobenzidine (Biomeda) two times each for 5 min, rinsed in dH2O, and counterstained with hematoxylin.
Results
PDE11A4 Characterization
PDE11A4 cDNA was cloned from human prostate RNA using RT-PCR. The protein was His-tagged, expressed in insect cells using baculovirus, and affinity purified on nickel columns. The recombinant PDE11A4 was characterized using a range of selective and nonselective well-known PDE inhibitors. As shown in Table 1, PDE11A4 was insensitive to vinpocetine, EHNA, milrinone, and rolipram, selective inhibitors for PDE1, 2, 3, and 4, respectively. It was moderately sensitive to sildenafil and zaprinast, inhibitors for PDE5 and PDE5/6, respectively. The non-selective PDE inhibitor IBMX displayed little inhibitory effect. Of the inhibitors examined, dipyridamole (an inhibitor for PDE1/5/6) was most potent in inhibiting PDE11A4. These observations were in good agreement with data in the literature (Fawcett et al. 2000; Yuasa et al. 2000), suggesting the recombinant enzyme was indeed PDE11A4.
Western Analysis
The specificity of antibody PD11A-112AP was studied using Hi5 cell lysates infected with pBlueBacHis2B vector containing PDE11A4 cDNA or an unrelated cDNA. The proteins in Hi5 cell lysates, supernatant solutions, and affinity-purified PDE11A4 were separated by SDS-PAGE, transferred onto membranes, and blotted with PD11A-112AP antibody. An immunoreactive band of ∼100 kDa was observed in cell lysate and supernatant of PDE11A4 cDNA infected Hi5 cells (Figure 1A, Lanes 2 and 3) but not in cell lysate of Hi5 cells infected with an unrelated cDNA (Figure 1A, Lane 1). As expected, the affinity-purified PDE11A4 was also immunoreactive (Figure 1A, Lane 4). These observations demonstrated that PD11A-112AP was specific for PDE11A. There was a faint immunoreactive band of ∼30 kDa visible in all cell lysates and supernatant. This protein may be specific to Hi5 cells, as a corresponding band was not observed in any human samples (see below). Western analysis of several human tissue homogenates including prostate, bladder, skeletal muscle, corpus cavernosum, and testis showed that a band migrated close to the size of recombinant PDE11A4 was observed in all the tissue examined (Figure 1B). A second band, migrated around 70 kDa, was visible in all tissues except the bladder. This band was most prominent in prostate (Figure 1B, Lane 1). The intensity ratio of the two bands was different in each tissue. Immediately below the second band there were one or two very faint bands in most of the tissues.
Inhibition of recombinant PDE11A4 by various PDE inhibitors a
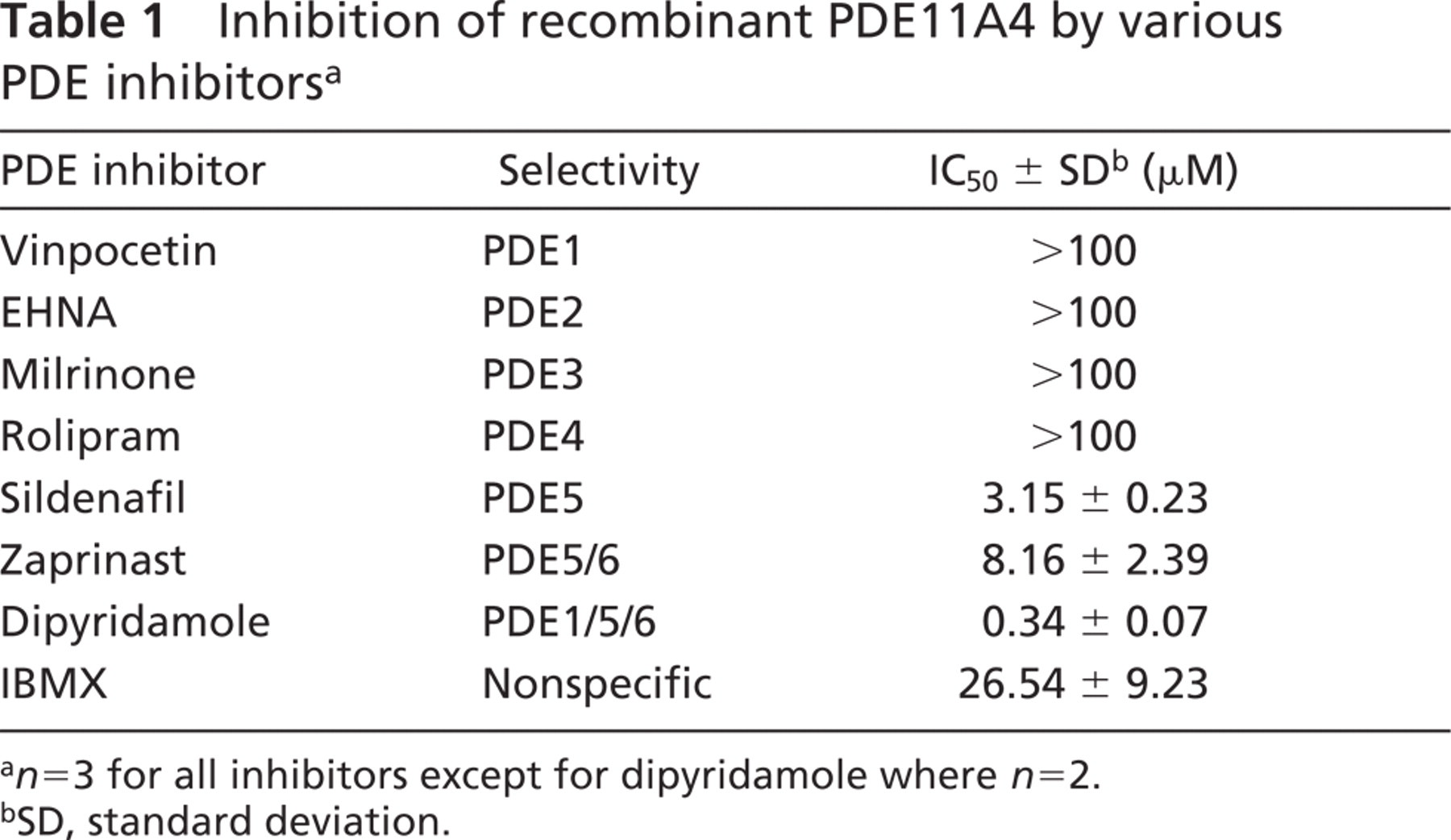

a n=3 for all inhibitors except for dipyridamole where n=2.
bSD, standard deviation.

Immunoblots probed with anti-PDE11A antibody. (
Immunohistochemistry
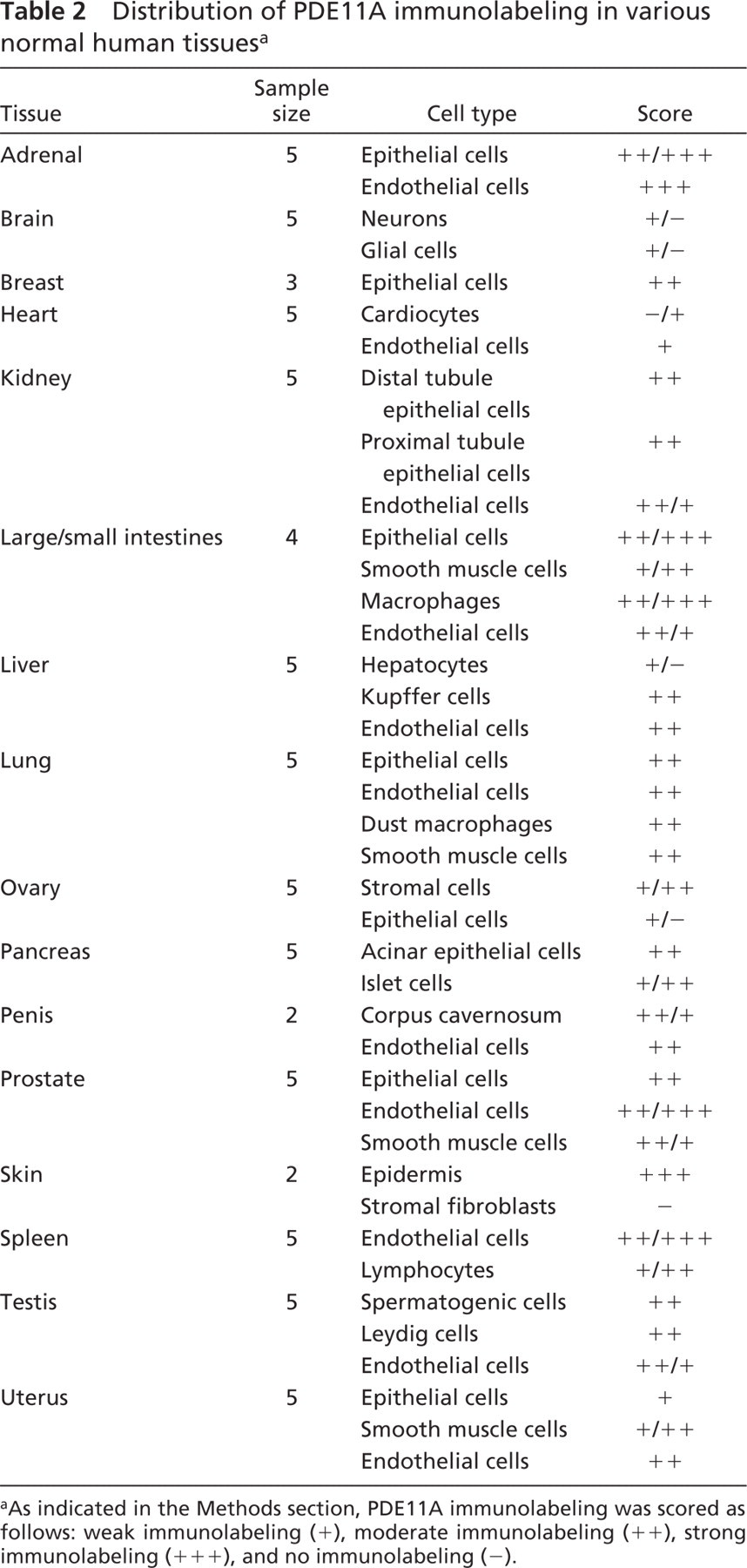
PDE11A was detected in a wide range of normal human tissues (Table 2). Brown labeling represented the presence of PDE11A. The relative intensity was scored as weak, light brown labeling (+), moderate, brown labeling (++), strong, intense, dark brown labeling (+++) and no labeling (−). No detectable labeling was observed in the skin when the antiserum was preabsorbed with the specific antigen (Figure 2A) or in the absence of primary antibody (data not shown). In contrast, when the primary antibody was included, prominent PDE11A labeling was evident in the epidermal cells (larger arrowheads) of the skin (Figure 2B), but not in the stromal fibroblasts (small arrowheads). Generally, PDE11A was detected in the epithelial, smooth muscle, and endothelial cells in such tissues as the prostate, kidney, and small and large intestines. Representative photomicrographs are presented in Figure 2 of normal human tissues. Figure 2C shows the presence of PDE11A in many cell types of the normal human colon. Epithelial cells (large arrowheads) and surrounding submucosal macrophages (small arrows) express strong PDE11A immunoreactivity, which is also present in the vascular endothelial cells (not present) and in the smooth muscle cells (large arrow). PDE11A was also detected in the apical areas of some proximal and distal tubule epithelial cells (large arrowheads) and in the endothelial cells (small arrowheads) of the kidney (Figure 2D). A representative image of the normal human lung (Figure 2E) showed prominent PDE11A immunolabeling in the dust macrophage cells (small arrows) and vascular endothelial cells (small arrowheads). Although not presented in the figure, prominent bronchial epithelial cells also expressed PDE11A immunoreactivity. The male reproductive tissues also expressed prominent PDE11A immunoreactivity in the prostate (Figure 2F) basal areas of the epithelial (large arrowheads) and endothelial cells (small arrowheads) with some diffuse smooth muscle cell staining, in the corpus cavernosal smooth muscle cells (large arrows) and endothelium (small arrowheads) of the penis (Figure 2G) and in the Leydig cells (small arrows), spermatogenic cells (large arrows) and endothelial cells (arrowheads) of the testis (Figure 2H).
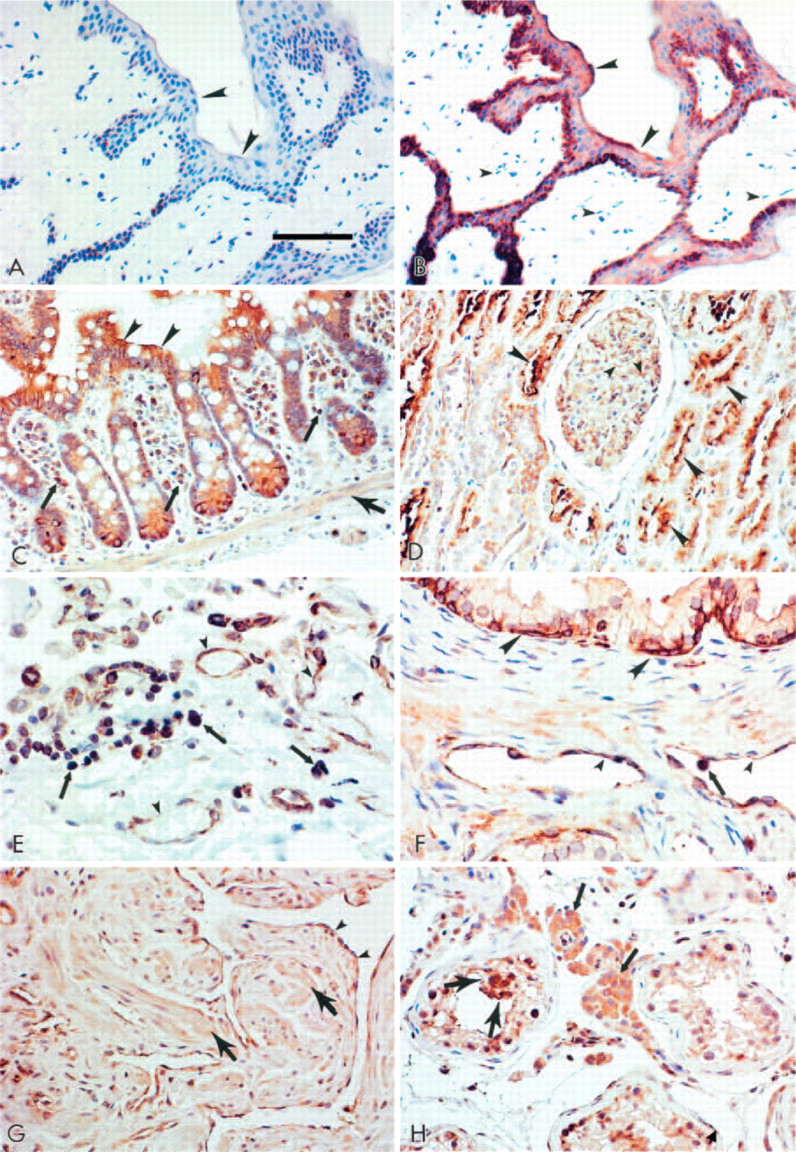
Presence of PDE11A in normal human tissues. Serial sections on the normal human skin show the absence of detectable PDE11A immunolabeling in the antigen competition negative control (
We also detected prominent PDE11A immunolabeling in several malignant human tissues (Table 3). Representative photomicrographs are presented in Figure 3. Serial sections of renal carcinoma show the lack of detectable PDE11A immunolabeling in the preabsorption control (Figure 3A) and the signal that was detected using the PDE11A primary antibody in the carcinoma cells (large arrowheads) of the kidney (Figure 3B). PDE11A immunolabeling was also detected in the carcinoma cells (large arrowheads) of the prostate (Figure 3C), in the colon (Figure 3D), lung (Figure 3E), and in the breast (Figure 3F) carcinoma cells.
Distribution of PDE11A immunolabeling in various normal human tissues a
aAs indicated in the Methods section, PDE11A immunolabeling was scored as follows: weak immunolabeling (+), moderate immunolabeling (++), strong immunolabeling (+++), and no immunolabeling (−).
Discussion
PDE11A is the most recent PDE to be cloned (Fawcett et al. 2000). Its expression at mRNA level, the genomic organization, and the splicing variants have been well studied (Fawcett et al. 2000; Hetman et al. 2000; Yuasa et al. 2000, 2001). However, little was reported on its expression at protein level. In this study, we investigated the cellular distribution of PDE11A proteins.
Distribution of PDE11A immunolabeling in several malignant human tissues
aAs indicated in the Methods section, PDE11A immunolabeling was scored as follows: weak immunolabeling (+), moderate immunolabeling (++), strong immunolabeling (+++), and no immunolabeling (−).
The antibody used here was raised against a peptide located at the C terminus. Thus, it should recognize all four splice variants. The identity of the recombinant protein was confirmed by enzyme activity in PDE assays and by its sensitivity to known PDE inhibitors. PDE11A4 has 943 amino acids with a predicted molecular mass of 105 kDa. The recombinant protein migrated at the expected location. The specificity of the antibody was confirmed using Western blot analysis on Hi5 cell lysates infected with expression vectors containing PDE11A4 cDNA or an unrelated cDNA. All human tissues on the Western blot displayed a band that migrated similarly as the recombinant PDE11A4, with different intensity in different tissues. This suggested that PDE11A4 was present in the tissues examined. The second band on the Western blot migrated at ∼70 kDa, suggesting it most likely represented PDE11A3. This band was most prominent in prostate, much weaker in skeletal muscle, corpus cavernosum, and testis, and totally absent in bladder. The very faint bands of ∼50 kDa seen in many tissues are believed to be PDE11A1. The sizes of these major bands recognized by the antibody were in good agreement with the predicted molecular mass of PDE11A splice variants (Fawcett et al. 2000; Hetman et al. 2000; Yuasa et al. 2000). No other prominent bands were detected.
There are some discrepancies between our observation and the data in the literature. Disagreements also exist among the published reports regarding the distribution of the PDE11A splice variants. Yuasa et al. (2000) showed that human PDE11A4 mRNA was abundantly and specifically expressed in prostate. They reported that it was not expressed in skeletal muscle, testis, and 12 other tissues surveyed. The authors also showed that within the same panel of tissues, PDE11A3 mRNA was only weakly expressed in testis and not in skeletal muscle or the other tissues. Also in contrast to our finding, Fawcett et al. (2000) reported that in Western blots of human prostate, only a 56-kDa protein representing PDE11A1 was detected. They also reported that skeletal muscle had one major band possibly representing PDE11A3 and two minor bands for PDE11A2 and 11A1. Further study with antibodies specific to different splicing variants are needed to clarify the discrepancies.
The IHC study reported here showed that the distribution of PDE11A protein is ubiquitous. Positive signals were found in smooth muscle cells, epithelial cells, and endothelial cells from many tissues. It is well known that alterations of cyclic nucleotide levels in smooth muscle cells modulate multiple physiological processes. A rapid increase of cAMP or cGMP levels decreases the contractile tone of smooth muscle cells (Hofmann et al. 2000). The functional consequence of this has been beautifully demonstrated through the use of PDE5 inhibitors in the treatment of erectile dysfunction (Boolell et al. 1996). PDE5 is the major cGMP hydrolyzing PDE in human corpus cavernosal smooth muscle cells. In cases when erectile dysfunction is caused by insufficient cGMP buildup, inhibition of PDE5 facilitates cGMP accumulation initiated by nitric oxide activation of soluble guanylyl cyclase. The elevated cGMP activates protein kinase G and eventually triggers the lowering of intracellular calcium concentration and the relaxation of corpus cavernosal smooth muscle. This results in an increase of blood flow into the sinusoids and, thus, a more rigid erection (Burnett et al. 1992; Eardley 1997). The same end point could also be achieved via elevation of cAMP levels (Maggi et al. 2000). In addition to responding to rapid changes in cyclic nucleotide concentrations, smooth muscle cells have also been shown to alter their rate of proliferation when continuously exposed to elevated cAMP or cGMP levels. Chronic elevation of cAMP or cGMP levels inhibits human arterial smooth muscle cell proliferation, though to different extents and via distinct mechanisms (Rybalkin and Bornfeldt 1999; Rybalkin et al. 2003). It has also been shown that agents increasing cGMP levels, such as nitric oxide or nitric oxide donors, exhibit antiproliferative effects on human prostatic smooth muscle cells (Guh et al. 1998). In most smooth muscle cells, PDE1, 2, 3, 4, and 5 have been identified (Torphy et al. 1993; Wallis et al. 1999; Maurice et al. 2003; Qiu et al. 2003; Rybalkin et al. 2003). Now PDE11A can be added to the list. Unlike most of the PDEs found in smooth muscle cells, PDE11A hydrolyzes cAMP and cGMP equally well (Fawcett et al. 2000; Hetman et al. 2000). Conceivably, PDE11A could be involved in any of the above-mentioned physiological processes via its ability to modulate levels of both types of cyclic nucleotides.
PDEs also modulate a wide range of cellular functions in various epithelial cells. It has been shown that increasing cAMP concentrations via PDE4 inhibition decreases TNF-α stimulated granulocyte-macrophage colony-stimulating factor (GM-CSF) secretion in a human bronchial epithelial cell line (Sasaki and Manabe 2004). GM-CSF is an important pro-inflammatory cytokine, and bronchial epithelial cells play critical roles in regulating inflammatory responses in the airway. In colonic epithelial cells, increases in cAMP levels through various PDE inhibitor treatments result in increases in transepithelial chloride secretion (O'Grady et al. 2002). It has been observed that in the rat intestinal epithelial cell line IEC18, phosphodiesterase inhibitor inhibits cell proliferation through modulation of TGF-α expression (Diab-Assef et al. 2002). The prominent expression of PDE11A in different types of epithelial cells and its ability to regulate the level and duration of intracellular cAMP indicate it may participate in many of these functions.
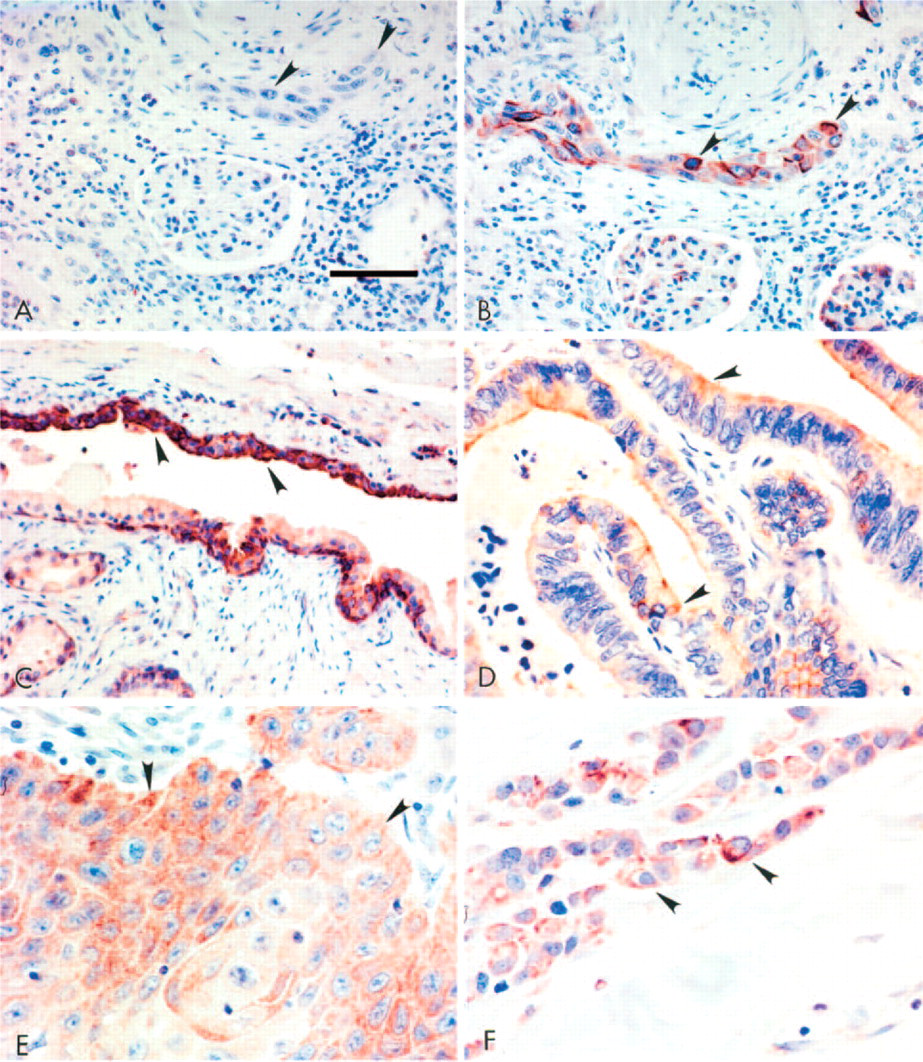
Presence of PDE11A in malignant human tissues. Serial sections of a renal cell carcinoma show the lack of detectable PDE11A immunolabeling in the preabsorption negative control (
Although relatively few studies have investigated the expression and function of PDEs in endothelial cells, PDE1–5 have been identified in various types of endothelial cells and PDE3 and PDE4 inhibitors have been shown to decrease endothelial cell proliferation and the expression of adhesion molecules (reviewed in Maurice et al. 2003). Here we show that PDE11A is expressed in the endothelial cells of most of the tissues examined. Its role in endothelium function awaits further study.
It is well recognized that high levels of cAMP can destroy various types of cancer cells in culture. However, most of the agents that can mimic cAMP action or produce the dramatic increase on cAMP concentrations are highly cytotoxic, precluding their use clinically. In comparison, PDE inhibitors that induce moderate cAMP accumulation also inhibit cancer cell proliferation. For example, elevation of cAMP by nonspecific PDE inhibitors hampers growth in several human prostatic cancer cell lines and induces terminal differentiation in some of the lines (Bang et al. 1994; Goto et al. 1999). Treatments with specific or nonspecific PDE inhibitors result in growth inhibition in small-cell lung carcinoma (Shafer et al. 1998), acute promyelocytic leukemia (Guillemin et al. 2002), and malignant glioma cells (Chen et al. 2002). Although PDE inhibitors might not kill enough cancer cells when used alone, a nonspecific PDE inhibitor could reduce the effective doses of other anti-cancer drugs, thus maintaining the efficacy and decreasing the toxicity (Hirsh et al. 2004). Other and newer PDE inhibitors could provide better specificity and efficacy and less side effects in cancer therapy. As PDE11A is expressed abundantly in multiple human carcinomas, it could be a potential target.
In summary, we have studied the tissue distribution of PDE11A using IHC and found that it is expressed in smooth muscle cells, epithelial cells, endothelial cells, and several other types of cells in most of the tissues surveyed. Our results suggest that PDE11A could be involved in multiple physiological processes in various tissues through their ability to modulate cAMP and cGMP levels. A PDE11A-specific inhibitor could greatly facilitate the elucidation of its functional roles.
Footnotes
Acknowledgements
The authors would like to thank Brenda Hertzog and Danielle Lawrence for expert technical assistance.
