Abstract
Thirteen mammalian aquaporin (AQP) isoforms have been identified, and they have a unique tissue-specific pattern of expression. AQPs have been found in the reproductive system of both male and female humans, rats, and mice. However, tissue expression and cellular and subcellular localization of AQPs have been poorly investigated in the male reproductive system of birds. The localization of AQP subtypes (AQP1, 2, 3, 4, 5, 7, 8, 9, and 11) in the goose testis and vas deferens has been studied through immunohistochemistry and immunobloting. Interestingly, the testicular and deferential tissues were positive for AQP1, −5, and −7 but not the others. AQP1 immunoreactivity was detected in the capillary endothelial cells of testis and vas deferens. AQP5 was localized in the interstitial tissue of the testis, including Leydig cells, as well as in the basal cells of vas deferens. Double-labeling confocal microscopy revealed coexpression of AQP5 with capillary AQP1 in the testis. AQP7 was expressed in elongated spermatid and spermatozoa tails in the testis, as well as spermatozoa tails in the vas deferens. These results suggest that several subtypes of AQPs are involved in the regulation of water homeostasis in the goose male reproductive system.
A
The first confirmation of expression, by in situ hybridization, of AQP (AQP7 and AQP8) in the male reproductive system (rat testis) was reported by Ishibashi et al. (1997a, b). To date, multiple AQPs have been found in the testis (AQP0, AQP1, AQP7, AQP8, and AQP9), efferent ducts (AQP1, AQP9, and AQP10), epididymis (AQP1, AQP3, AQP9, and AQP10), vas deferens (AQP1, AQP2, and AQP9), and accessory glands (AQP1 and AQP9) of adult mammals (reviewed by Huang et al. 2006). AQPs are known to be involved in the early stage of spermatogenesis, in the secretion of tubule liquid, and in the concentration and storage of spermatozoa. Fluid reabsorption in some regions of the male reproductive tract is under steroid hormone control and could be mediated by various AQPs (reviewed by Da Silva et al. 2006a). Alterations in the expression and function or regulation of AQPs have already been demonstrated in disorders of the male reproductive system, such as abnormal sperm motility, abnormal epididymis and infertility (seen in cystic fibrosis), and varicocele (reviewed by Huang et al. 2006). Nevertheless, data concerning the expression and role of AQPs in the bird male reproductive system are still very limited (Zaniboni et al. 2004).
The purpose of this study was to identify the tissue expression as well as cellular and subcellular localization of AQPs in avian testis and vas deferens.
Materials and Methods
Experimental Animals
All experiments were performed in accordance with the principles and procedures of the Animal Ethics Committee, University of Warmia and Mazury in Olsztyn. Tissues were obtained at the peak of reproductive activity (March) from male White Koluda Geese. The animals were kept in a 10/14-hr light-dark cycle with feed and water provided ad libitum. Animals were euthanized with sodium thiopental and transcardically perfused with 4% paraformaldehyde/phosphate-buffered saline buffer at 5C. For preparation of tissue sections (5-μm thickness), tissues were dehydrated in ethanol followed by buthanol and finally embedded in paraffin.
Primary Antibodies
In the present study, affinity-purified polyclonal antibodies (SulfoLink Kit, Pierce; Rockford, IL) to the following AQPs were utilized: AQP1 (Terris et al. 1996), AQP2 (Nielsen et al. 2006), AQP3 (Kim et al. 2005), AQP4 (Vajda et al. 2002), AQP5 (Nielsen et al. 1997), AQP7 (Nejsum et al. 2000), AQP8 (Elkjaer et al. 2001), AQP9 (Carbrey et al. 2003), and AQP11 (Gorelick et al. 2006).
SDS-PAGE and Immunoblotting
Following isolation, the tissues were immediately placed in an ice-cold dissection buffer (0.3 M sucrose, 25 mM imidazol, 1 mM EDTA in ddH2O, pH 7.2) containing 8.4 μM leupeptin and 0.4 mM pefabloc (Skowronski et al. 2009). The tissue samples were homogenized using an ultra Turrax T8 homogenizer (IKA Labortechnik; Staufen, Germany) and centrifuged at 4000 × g for 15 min at 4C. The supernatant diluted in SDS buffer contained a final concentration of 62 mM Tris (hydroxymethyl)-aminomethane, 0.1 M SDS, 8.7% glycerol, 0.09 mM bromophenol blue, and 0.04 M dithiothreitol (DTT), pH 6.8. The protein samples were heated for 5 min at 90C and stored in a refrigerator for further analysis.
The samples warmed up to 37C were loaded into 12.5% polyacrylamide gels, and proteins were separated by electrophoresis. The total protein amount in each sample was adjusted by staining with Gelcode Coomassie Blue stain reagent (Bie and Berntsen A/S; & Aslash;byhøj, Denmark) to calculate equal loading. The proteins of studied gels were then electrotransferred onto nitrocellulose membranes (Hybond ECL RPN3032D, Amersham Biosciences; Little Chalfont, UK) for 1 hr at 100 V. The membranes were blocked for 1 hr with 5% milk in PBS-T (80 mM Na2HPO4, 20 mMNaH2PO4, 100 mM NaCl, pH 7.5, and 0.1% v/v Tween 20). After washing, the membranes were incubated overnight at 5C with anti-AQPs antibody.
The membranes were washed and then incubated for 1 hr with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG secondary antibody (Dako A/S; Glostrup, Denmark) in PBS-T. After washing with PBS-T, the sites of antibody-antigen reaction were visualized with HRP-conjugated secondary antibodies (P448, diluted 1:3000; DAKO) with an enhanced chemiluminescence system (Amersham Biosciences) and exposure to photographic film (Hyperfilm ECL, RPN3103K; Amersham Biosciences).
IHC
The staining was carried out using indirect immunoperoxidase as described previously (Skowronski et al. 2007). The sections were dewaxed and rehydrated. For immunoperoxidase labeling, endogenous peroxidase was blocked by 0.5% H2O2 in absolute methanol for 10 min at room temperature. To reveal antigens, the sections were submerged in 1 mM Tris solution (pH 9.0) supplemented with 0.5 mM EGTA and heated in a microwave oven. After the treatment, the sections were left for 30 min in the buffer for cooling. Nonspecific binding of IgG was eliminated by incubating the sections in 50 mM NH4Cl for 30 min, followed by blocking in PBS supplemented with 1% BSA, 0.05% saponin, and 0.2% gelatin. The sections were incubated overnight at 4C with primary antibodies diluted in PBS supplemented with 0.1% BSA and 0.3% Triton X-100. The sections were rinsed with PBS supplemented with 0.1% BSA, 0.05% saponin, and 0.2% gelatin, and then incubated with HRP-conjugated secondary antibody (Dako). Labeling was visualized by 0.05% 3,3 diaminobenzidine tetrahydrochloride.
Fluorescence techniques were employed for double labeling tissues as described previously (Skowronski et al. 2007). After overnight incubation with anti-AQP5 antibody as described above, the sections were incubated with fluorescent secondary antibody (goat anti-rabbit antibodies; AlexaFluor 546, Molecular Probes). After being washed (PBS for 3 × 10 min), the sections were incubated overnight with a biotinylated anti-AQP1 antibody, and the immunoreactivity was visualized using streptavidine FITC (Dako). After being rinsed with PBS for 3 × 10 min, the sections were mounted in glycerol supplemented with antifade reagent (
Results
IHC analysis using antibodies against nine AQPs (AQP1, −2, −3, −4, −5, −7, −8, −9, and −11) was performed to examine whether these water channel proteins are expressed in the male goose reproductive system. The analysis confirmed the expression of AQP1, AQP5, and AQP7. In contrast, no staining for other examined AQPs (AQP2, −3, −4, −8, −9, and −11) was observed in tested tissues (data not shown). The data obtained are presented in Figures 1–3.
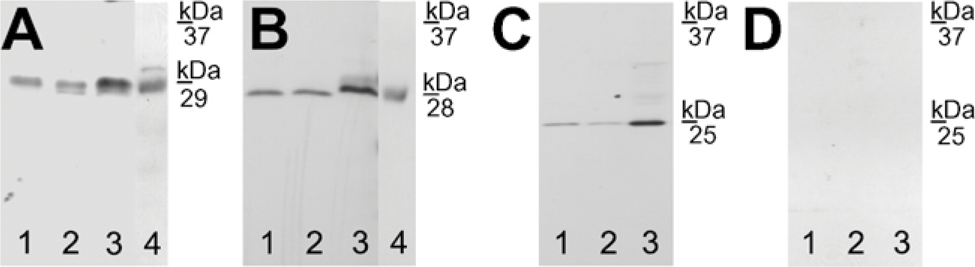
AQP1 immunoreactivity was detected in the capillary endothelium of the testis (Figure 1A). An identical labeling pattern was observed in the vas deferens (Figure 1B). Moreover, immunoblotting revealed that anti-AQP1 antibody recognized a 29-kDa band in testes (Figure 2A, Lane 1) and vas deferens (Figure 2A, Lane 2) of the goose and kidney of the mouse (positive control; Figure 2A, Lane 3), as well as in intestine of the goose (positive control; Figure 2A, Lane 4), as previously reported for kidney of mice, rats, and humans and intestine of chicken (Nielsen et al. 1995; Maunsbach et al. 1997; Ma et al. 1998; Müller et al. 2006
AQP5 was localized in the interstitial tissue of the testis, including Leydig cells (Figure 1C), as well as in the basal cells of vas deferens (Figure 1D). Immunoblotting demonstrated that anti-AQP5 antibody recognized a 28-kDa band in avian testes (Figure 2B, Lane 1) and vas deferens (Figure 2B, Lane 2). As a positive control, in agreement with previous observations in the lung tissues of mice, rats, and humans (Nielsen et al. 1997; Krane et al. 2001; Kreda et al. 2001), the antibody against AQP5 recognized a 28-kDa band in the lung of the mouse (Figure 2B, Lane 3), as well as in the goose small intestine (Figure 2B, Lane 4), as previously reported for the intestine of chicken (Ramírez-Lorca et al. 2006).
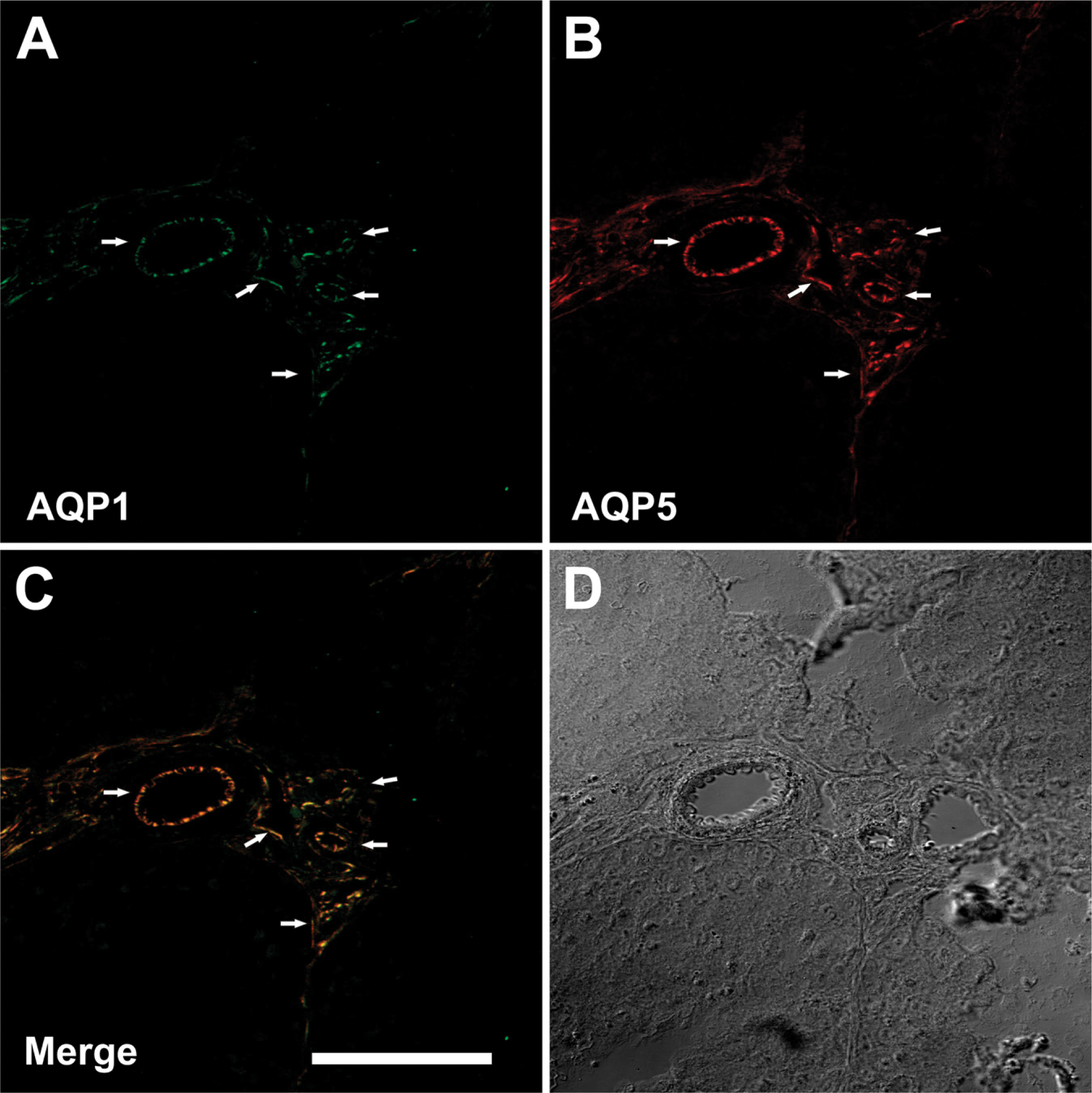
Further microscopic analyses were performed to verify the capillary expression of AQP5 in the testis. AQP1 is a suitable marker of the plasma membranes of capillaries (Mobasheri and Marples 2004). Thus, fluorescent double labeling for AQP1 and AQP5 was employed to determine the localization of AQP5 in these vessels. As shown in Figure 3A, the AQP1 antibody strongly labeled the small vessels of testes (green). The anti-AQP5 antibody showed a similar localization pattern within testes (red; Figure 3B). As shown in Figure 3C, the double immunolabeling revealed a high degree of colocalization of AQP1 and AQP5 proteins in the capillaries of goose testes.
AQP7 immunoreactivity was detected in the tails of elongated spermatids and spermatozoa in the testis (Figure 1E), as well as tails of spermatozoa in the vas deferens (Figure 1F). Immunoblotting also revealed that anti-AQP7 antibody recognized a 25-kDa band in the testes (Figure 2C, Lane 1) and vas deferens (Figure 2C, Lane 2) of the goose and, as positive control, in the kidneys of the mouse (Figure 2C, Lane 3), as previously described by Skowronski et al. (2007).
Negative controls for all immunohistochemical and immunoblotting analyses were performed by omitting the primary antibodies against AQP (not shown) and using non-immune IgG (Figures 1G and 1H; Figure 2D, Lanes 1–3), and no specific immunostaining was observed. As a positive control, AQP1 labeling was seen in the goose small intestine (Figure 1I), consistent with the previous findings in ducks (Müller et al. 2006) and sparrows (Casotti et al. 2007). AQP5 antibody noticeably stained the goose small intestine (positive control; Figure 1J), consistent with previous findings seen in the intestine of chicken (Ramírez-Lorca et al. 2006).
Discussion
In the present study, IHC and immunoblotting revealed that three isoforms of water channel proteins (AQP1, −5, and −7) are expressed in the goose testis and vas deferens. In contrast, the above-mentioned methods demonstrated no immunostaining for other examined AQPs (AQP2, −3, −4, −7, −8, and −11).
AQPs in Testis
The current study showed expression of AQP1, −5, and −7 in the goose testis: both AQP1 and AQP5 in the capillary endothelium, AQP5 in the interstitial tissue of the testis, including Leydig cells, and AQP7 in the tails of elongated spermatids and spermatozoa. The present study provides the first evidence of AQP1, AQP5, and AQP7 expression in the bird testicular tissues. The expression of AQP1 has been previously shown in human endothelial cell membranes of testicular microvessels (Nicòtina et al. 2005) and on the plasma membrane of epithelial cells of mouse rete testis (Danyu et al. 2008). In the literature, there are several reports pertaining to AQP expression in Leydig cells in rats. Tsukaguchi et al. (1998) detected AQP9 mRNA in the interstitial Leydig cells, and AQP9 protein was conspicuously expressed in the plasma and intracellular membranes of this cell (Elkjaer et al. 2000). AQP0 was also expressed in Leydig cells (Hermo et al. 2004). Our findings add AQP5 to this list. In birds, androgens [e.g., testosterone (T)] produced by testicular Leydig cells contribute to rapid gonadal maturation in response to photostimulation. Plasma T peak coincides with territorial behavior of males and the highest quality of sperm production. In both sexes, T stimulates sexual behavior, and its annual maximal level is observed during the mating period (reviewed by Leska and Dusza 2007). It appears to be of interest whether AQP5 is involved in endocrine functions.
The current study also demonstrates that AQP5 is expressed in the capillary endothelium of the testis. A similar finding was reported previously in capillaries of mouse white adipose tissue, where AQP7 colocalized in the vessels with AQP1 (Skowronski et al. 2007).
AQP7 and AQP8 have been identified in rat testes and have been shown by in situ hybridization to be abundantly expressed in germ cells (Ishibashi et al. 1997a,b). This intriguing finding suggests that AQP7 and AQP8 may play an important role in spermatogenesis. Although they share a similar transmembrane structure, AQP7 and AQP8 are different in function and distribution pattern in testes. AQP7 mRNA is transiently expressed at a late phase of spermatogenesis, and AQP7 protein cellular and subcellular localization varies, depending on the stage of spermatogenesis (Suzuki-Toyota et al. 1999; Calamita et al. 2001a, b). However, AQP8 mRNA was found to be present uniformly and constantly in every seminiferous tubule, consistently with the expression of AQP8 protein (Elkjaer et al. 2001), which was found intracellularly as well as over the plasma membrane of all germ cells (Calamita et al. 2001b). Further studies of the two AQPs in the developing rat testis, concerning the maturation of germ cells, support the roles of AQPs in spermatogenesis (Calamita et al. 2001a). Interestingly, in contrast to the abundant AQP8 in rat testes, AQP8 mRNA was absent in human testes (Koyama et al. 1998). AQP7 was observed in epididymal spermatozoa (Calamita et al. 2001a), thus indicating its possible role in sperm maturation and storage as well as its function in spermatogenesis (Suzuki-Toyota et al. 1999). Saito et al. (2004) demonstrated by IHC a similar labeling pattern in human ejaculated sperm. They suggested that a lack of AQP7 expression in sperm may be an underlying mechanism of male infertility.
Immunoblotting of AQP1, −5, and −7 in goose tissues. (
However, a recent study on phenotype analysis of AQP8 knockout (KO) mice showed few and only mild phenotype differences between wild-type and AQP8 KO mice (Yang et al. 2005). Although the weight and size of the testes in AQP8 KO mice were increased, no impaired fertility or abnormalities in sperm count or morphology were found. Moreover, male mice lacking AQP7 were fertile, and their sperm cells showed normal motility (Skowronski et al. 2007). The authors suggested that the presence of AQP7 in spermatozoa cells has no role, or an insignificant role, in sperm-egg interactions, inasmuch as the AQP7 KO female and male mice have been fertile. Collectively, the expression of AQPs found in avian testes is confined to various compartments that are likely to be connected with their specific functions.
AQPs in Vas Deferens
In this study, we demonstrated AQP1, −5, and −7 expression in the goose vas deferens. AQP1 expression was present in the endothelial cells of the blood vessels, AQP5 in the basal cells, and AQP7 in the tails of deferential spermatozoa. To our knowledge, this is the first evidence of AQP expression in the bird deferential tissues.
AQP1 has been reported to be present at plasma membranes of epithelial cells in the ampulla of the vas deferens, but not the cells in proximal parts in rats (Brown et al. 1993). AQP2 expression in vas deferens was also confirmed in rats (Nelson et al. 1998). AQP2 (Stevens et al. 2000) began to appear in some epithelial cells of the middle vas deferens and was concentrated on the apical plasma membrane of principal cells in the distal vas deferens (ampulla), where AQP1 (Brown et al. 1993) was highly expressed. No expression of AQP1 was detected in the proximal region, and very little was observed on intracellular vesicles. The variable expression of AQP1 and AQP2 in different regions of the vas deferens may be due to the marked structural differences of principal cells along the vas deferens (Andonian and Hermo 1999), reflective of diverse functional activities. In addition to AQP1 and AQP2, AQP9 is present in the rat vas deferens, where it is detected throughout the entire length of the duct (Pastor-Soler et al. 2001). Thus, three aquaporins have been identified to date in the vas deferens; these three water channels are present in the same membrane domain (apical) of epithelial cells from the most distal region, suggesting that the composition of the luminal environment, in which spermatozoa terminate their maturation and are stored, involves a complex regulation of transepithelial water and solute transport (reviewed by Da Silva et al. 2006b).
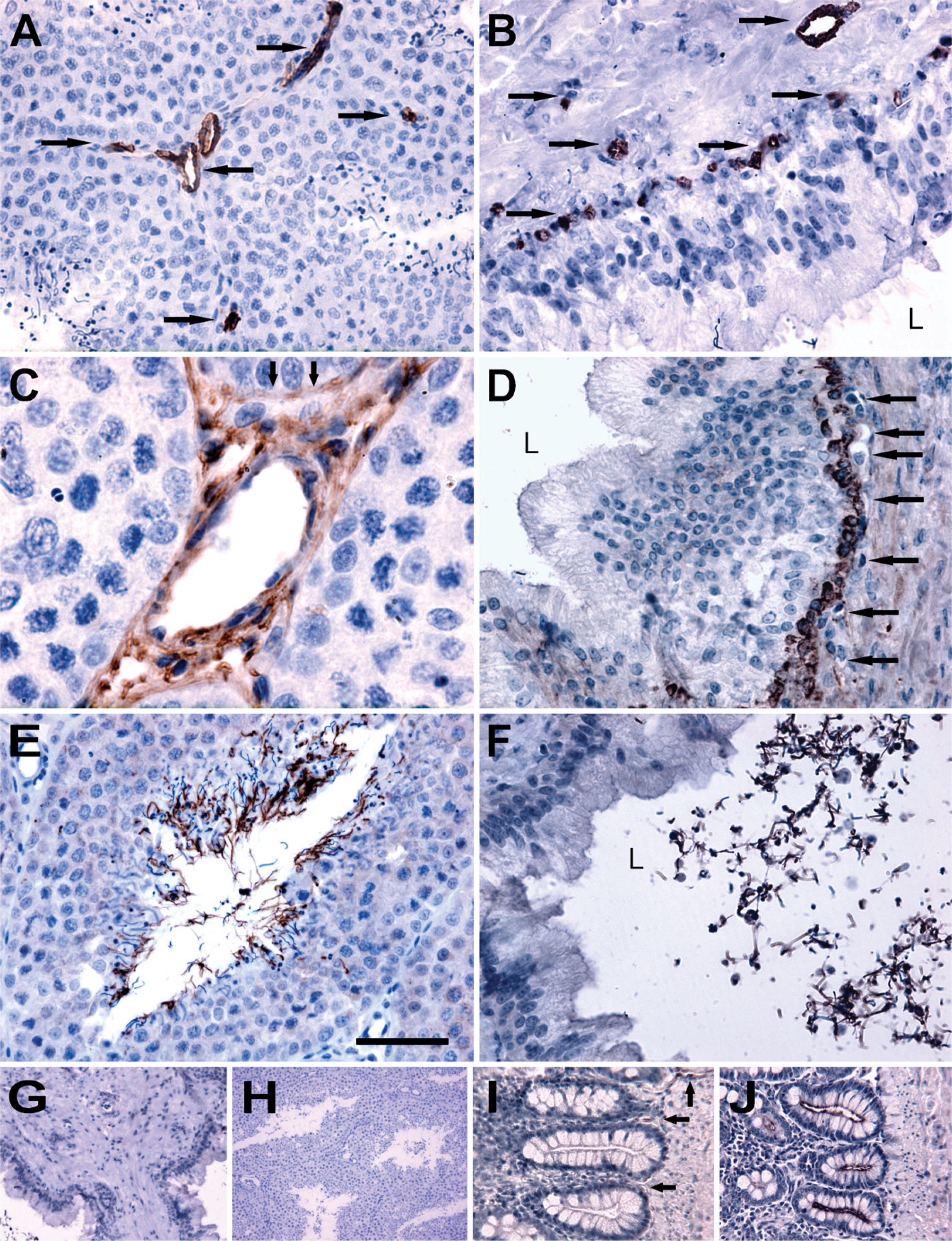
Immunohistochemical staining of aquaporin-1 (AQP1), −5, and −7 in paraffin-embedded sections of the testis and vas deferens from goose. Anti-AQP1 antibody labels capillary endothelium of the testis (
(
The presence of AQP5 in basal cells, found in this study, may have physiological implications. These cells have been proposed as mature, tissue-fixed macrophages playing a role in local immune defense within the epithelium (Yeung et al. 1994; Seiler et al. 1998). Recent morphological studies indicate that basal cells are actively involved in secretion of humoral factors that may serve as paracrine regulators of the functions of principal cells and other cells (Hermo and Robaire 2002). Several aquaporins (AQP3, AQP7, AQP8, and AQP11) have been detected in basal cells of the epididymis (Elkjaer et al. 2001; Hermo et al. 2004, 2008). Nevertheless, participation of AQP5 in this regulation remains to be established.
To our knowledge, this is the first immunohistochemical study pertaining to the expression of AQPs in the male reproductive system in the goose. The present results suggest the role of AQPs (AQP1, AQP5, and AQP7) in water homeostasis within tissues of avian testis and vas deferens. Further investigations are necessary to describe the physiological role of these proteins in relation to reproductive processes in the goose.
Footnotes
Acknowledgements
This research was supported by the Polish Ministry of Science and Higher Education (grant number 0206.0805) and the European Union within the European Social Fund.
