Abstract
Thirteen mammalian aquaporin (AQPs) isoforms with a unique tissue-specific pattern of expression have been identified. To date, 11 isoforms of AQP have been reported to be expressed in female and male reproductive systems. The purpose of our study was to determine the localization and quantitative changes in the expression of AQP1, 5 and 9 within the pig oviduct during different stages of the estrous cycle and early pregnancy. The results demonstrated that AQP1, 5, and 9 were clearly detected in all studied stages of the estrous cycle and pregnancy. AQP1 was localized within oviductal blood vessels. In cyclic gilts, the expression of AQP1 protein did not change significantly between days 10–12 and 14–16 but increased on days 2–4 and 18–20. AQP5 was localized in smooth muscle cells and oviductal epithelial cells. The expression of AQP5 protein did not change significantly between days 10–12 and 14–16 of the estrous cycle but increased on days 2–4 and 18–20. The anti-AQP9 antibody labeled epithelial cells of the oviduct. The expression of AQP9 did not change significantly between days 10–12 and 14–16 of the estrous cycle but increased on days 2–4 and 18–20. In pregnant gilts, expression of AQP1, 5, and 9 did not change significantly in comparison with the estrous cycle. Therefore, a functional and distinctive collaboration seems to exist among diverse AQPs in water handling during the different oviductal phases in the estrous cycle and early pregnancy.
During late follicular development and the first 4 days of the estrous cycle, the oviduct occupies a central role in the establishment of pregnancy. The ovarian steroids induce marked morphological, physiological, and biochemical changes in the oviduct microenvironment. Alterations include changes in the biosynthetic activity and the release of macromolecules by the oviductal epithelium, which becomes part of the luminal microenvironment. Furthermore, both regional and temporal differences in activity and protein production occur through hormonal changes during the estrous cycle and early pregnancy (Buhi et al. 1997). Moreover, the oviduct, as a dynamic reproductive organ, enables reception, transport, and maturation of male and female gametes, as well as their fusion, and supports early embryo development (Brüssow et al. 2008).
Aquaporins (AQPs) are integral plasma membrane proteins that primarily transport water across the plasma membrane. These proteins were identified more than a decade ago (Preston and Agre 1991). There are 13 members (AQP0-12) in humans, and many other AQPs have also been found in plants, yeast, bacteria, amphibians, and various lower organisms (Verkman and Mitra 2000).
Aquaporins, based on their structural and functional properties, are divided into three subgroups: classical aquaporins (AQP0, 1, 2, 4, 5, 6, 8), aquaglyceroporins (AQP3, 7, 9, 10), and recently identified superaquaporins (AQP11, 12) (reviewed by Ishikawa et al. 2006). Based on the protein expression, so far at least nine AQP isoforms have been confirmed to be present in the female reproductive system of humans, rats, and mice (reviewed by Huang et al. 2006). The first confirmation of AQP in the female reproductive system was obtained by isolating and sequencing the complementary DNA (cDNA) encoding water channels from the human uterus (Li et al. 1994). Afterwards, Li et al. (1997) found AQP1 mRNA in the rat uterus.
In literature, there have been three reports pertaining to AQP localization in oviductal tissues. Branes et al. (2005) showed by immunohistochemistry the expression of AQP5, 8, and 9 in the epithelial cells of the rat oviduct. In turn, Gannon et al. (2000) found AQP1 labeling in the innermost and the inner cells of the circular muscular layer of the rat oviduct. Our recent work demonstrates the localization of AQP1 in the pig oviductal vessels, AQP5 in muscle cells, and AQP5 and AQP9 in oviductal luminal epithelium (Skowronski et al. 2009).
There is also evidence indicating that ovarian steroids can affect the expression of several AQPs (Branes et al. 2005; Jablonski et al. 2003; Lindsay and Murphy 2006, 2007). It suggests that marked fluctuations in AQP expression in target tissues for steroids, including the oviduct, may exist. In our previous study, immunohistochemistry and Western blot analyses using antibodies against nine AQPs (AQP1, 2, 3, 4, 5, 7, 8, 9, and 11) were performed to examine which of these water channel proteins are expressed in the pig oviduct (on days 17–19 of the estrous cycle). The analysis confirmed the expression of AQP1, 5, and 9 (Skowronski et al. 2009).
Nevertheless, data concerning the quantitative expression of AQPs in the oviduct are still very limited and not available in relation to the pig. Therefore, the aim of this study was to examine the cellular localization as well as changes in protein expression of AQP1, 5, and 9 in the oviduct of gilts during the estrous cycle and early pregnancy.
Materials and Methods
Experimental Animals
All experiments were performed in accordance with the principles and procedures of the Animal Ethics Committee of the University of Warmia and Mazury in Olsztyn. Tissue samples were recovered from mature cross-bred gilts (Large White × Polish Landrace) at the early (days 2–4; n = 5), middle (days 10–12; n = 5), and late (days 14–16; n = 5) stage of the luteal phase and the late (days 18–20; n = 5) stage of the follicular phase of the estrous cycle as well as on days 14 to 16 (n = 5) and 30 to 32 (n = 5) of gestation (the onset and the end of implantation process, respectively). Gilts were observed daily for estrus behavior in the estrous cycle, and they were used during their third consecutive normal estrous cycle. In addition, stage of the cycle was confirmed according to Akins and Morrissette (1968). Gilts assigned to the pregnant group were naturally bred on the second day of estrus. Pregnancy was detected by ultrasound scanning before killing of the gilts (days 30–32). In addition, pregnancy was confirmed by the presence of embryos/fetuses after flushing of uterine horns with 20 ml sterile saline (days 14–16 and 30–32). Within a few minutes after slaughter, oviduct was separated from each gilt, and a 10-g weight fragment of the tissue was frozen in liquid nitrogen immediately after dissection and then stored at −70C until Western blot analysis. For immunohistochemistry, tissues were fixed by immersion in 4% paraformaldehyde for 24 hr (Skowronski et al. 2007).
Primary Antibodies
In the present study, affinity-purified polyclonal antibodies to AQPs were used (SulfoLink Kit; Pierce, Rockford, IL). The antibodies to AQP1, 5, and 9 were previously characterized, respectively, by Terris et al. (1996), Nielsen et al. (1997), and Carbrey et al. (2003). Moreover, the anti-β-actin antibody was used (cat. no. A2066; Sigma-Aldrich, St Louis, MO). In addition, immunoglobulins from non-immunized rabbit were used as a negative control.
SDS-PAGE and Western blot
Following isolation, the tissues were immediately placed in ice-cold dissection buffer (0.3 M sucrose, 25 mM imidazol, 1 mM EDTA in ddH2O, pH 7.2) containing 8.4 µM leupeptin and 0.4 mM pefabloc (Skowronski et al. 2007). The tissue samples were homogenized using an ultra Turrax T8 homogenizer (IKA Labortechnik, Staufen, Germany) and centrifuged at 4000 × g for 15 min at 4C. The supernatant diluted in SDS buffer contained a final concentration of 62 mM Tris (hydroxymethyl)-aminomethane, 0.1 M sodium dodecyl sulfate (SDS), 8.7% glycerol, 0.09 mM bromophenol blue, and 0.04 M dithiothreitol (DTT), pH 6.8. The protein samples were heated for 5 min at 90C and stored in a refrigerator for further analysis.
The samples warmed to 37C were loaded into 12.5% polyacrylamide gels, and proteins were separated by electrophoresis. The total protein amount in each sample was adjusted by staining with Gelcode Coomassie Blue Stain Reagent (Bie and Berntsen A/S, Åbyhøj, Denmark) to provide equal loading. The proteins of studied gels were then electro-transferred onto nitrocellulose membranes (Hybond ECL RPN3032D; Amersham Pharmacia Biotech, Little Chalfont, UK) for 1 hr at 100 V. The membranes were blocked with 5% milk in PBS-T (80 mM Na2HPO4, 20 mM NaH2PO4, 100 mM NaCl [pH 7.5], and 0.1% vol/vol Tween 20) for 1 hr. After washing, the membranes were incubated overnight at 5C with anti-AQPs or β-actin antibodies.
The membranes were washed and incubated with horseradish peroxidase–conjugated goat anti-rabbit IgG secondary antibody (P448, diluted 1:3000; Dako A/S, Glostrup, Denmark) in PBS-T for 1 hr. After washing with PBS-T, the sites of antibody–antigen reaction were visualized with an enhanced chemiluminescence (ECL) system (Amersham Pharmacia Biotech) and exposure to photographic film (Hyperfilm ECL, RPN3103K; Amersham Pharmacia Biotech). The results of Western blotting were quantified by densitometric scanning of immunoblots with GelScan for Windows version 1.45 software (Kucharczyk, Poland). Data were expressed as a ratio of AQP proteins relative to actin protein in OD.
Immunohistochemistry
For preparation of paraffin-embedded tissue sections (4-µm thickness), the tissues were dehydrated in ethanol followed by xylene and finally embedded in paraffin (Skowronski et al. 2009). The staining was carried out using indirect immunoperoxidase. The sections were dewaxed and rehydrated. For immunoperoxidase labeling, endogenous peroxidase was blocked by 0.5% H2O2 in absolute methanol for 10 min at room temperature. To reveal antigens, the sections were submerged in 1 mM Tris solution (pH 9.0) supplemented with 0.5 mM EGTA and heated in a microwave oven at 650 W for 6 min and then at 350 W for 10 min as described previously (Skowronski et al. 2007).
After the treatment, the sections were left for 30 min in the buffer for cooling. Nonspecific binding of IgG was eliminated by incubating the sections in 50 mM NH4Cl for 30 min, followed by blocking in PBS supplemented with 1% BSA, 0.05% saponin, and 0.2% gelatin. The sections were incubated overnight at 4C with primary antibodies diluted in PBS supplemented with 0.1% BSA and 0.3% Triton X-100. The sections were rinsed with PBS supplemented with 0.1% BSA, 0.05% saponin, and 0.2% gelatin and then incubated with horseradish peroxidase–conjugated secondary antibody (Dako A/S). Labeling was visualized by 0.05% 3,3 diaminobenzidine tetrahydrochloride (DAB). The microscopy was carried out using a Leica DMRE light microscope (Leica, Heidelberg, Germany).
Statistical Analysis
All data were analyzed by one-way ANOVA and least significant difference (LSD) post hoc test and were reported as the means ± SEM from five independent observations. Statistical analyses were performed using the Statistica program (StatSoft, Inc., Tulsa, OK). Values for p < 0.05 were considered statistically significant.
Results
Western Blot
The expression of AQP1 protein in the porcine oviduct at different stages of the estrous cycle and pregnancy (see Materials and Methods) is shown and summarized in Figure 1A–C. A band of AQP1 protein product of the expected size (29 kDa) was clearly detected in all stages of the estrous cycle and pregnancy (Fig. 1A,B). In cyclic gilts, the expression of AQP1 protein did not change significantly between days 10–12 and 14–16 but increased (p < 0.05) on days 2–4 and 18–20 when compared with days 10–12 and 14–16 of the cycle (Fig. 1A). In pregnant gilts, the expression of AQP1 did not change significantly during the onset and the end of implantation process (Fig. 1B) and also when compared to mid- and late-luteal phases of the estrous cycle (Fig. 1C). Using immunoblotting with the AQP1 antibody, we detected AQP1 in a protein fraction isolated from the porcine kidney, which served as a positive control for AQP1 protein expression (Fig. 1D).
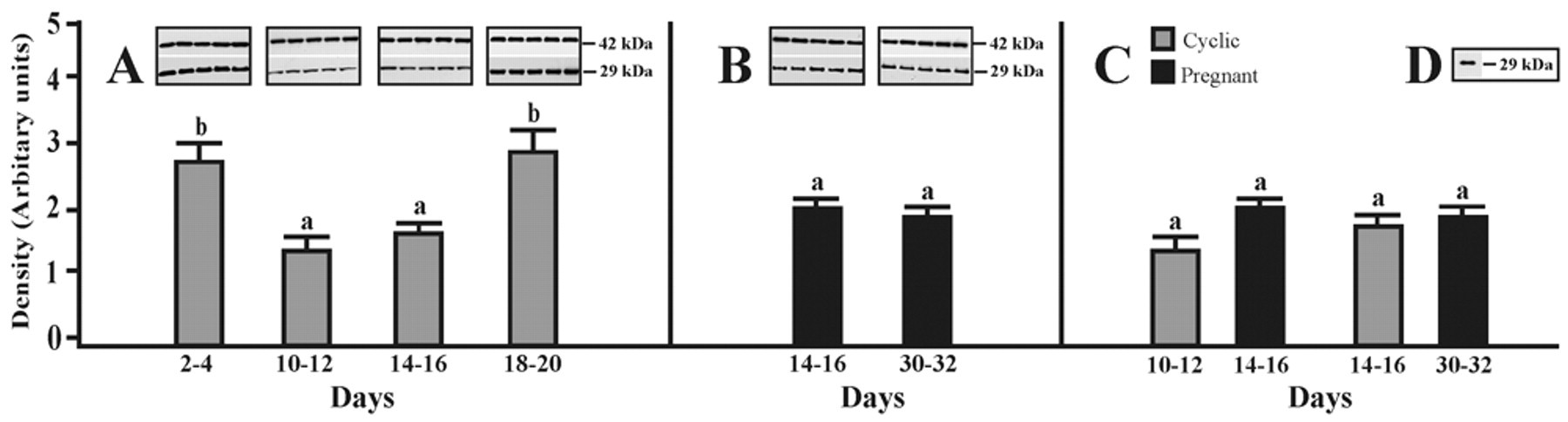
Western blot analysis of AQP1 expression. Expression of AQP1 assessed by Western blotting in the porcine oviduct on days 2–4, 10–12, 14–16, and 18–20 of the estrous cycle (A) as well as on days 14–16 and 30–32 of pregnancy (B). Densitometric analyses of AQP1 protein levels normalized against β-actin (A, B). In addition, comparison of the AQP1 expression during two stages of the cycle and two periods of pregnancy is shown in panel C. Positive control for Western blot analysis of the porcine kidney (D). Values are the mean ± SEM (n = 5). a,bMeans with different superscripts are significantly different.
The expression of AQP5 in the porcine oviduct at different stages of the estrous cycle and pregnancy is shown and summarized in Figure 2A–C. A band of AQP5 protein product of the expected size (28 kDa) was clearly detected in all stages of the estrous cycle and pregnancy (Fig. 2A,B). The expression of AQP5 in cyclic gilts increased from days 10–12 to 14–16, but this difference was insignificant (p > 0.05) (Fig. 2A). In turn, the expression of AQP5 was significantly higher on days 2–4 and 18–20 of the cycle (p < 0.05) when compared to middle and late stages of the luteal phase (Fig. 2A). In pregnant gilts, the expression of AQP5 did not change significantly during the onset and the end of the implantation process (Fig. 2B) and also when compared to mid- and late-luteal phases of the estrous cycle (Fig. 2C). Immunoblotting with the anti-AQP5 antibody revealed AQP5 in porcine lung samples, which served as a positive control for AQP5 (Fig. 2D).
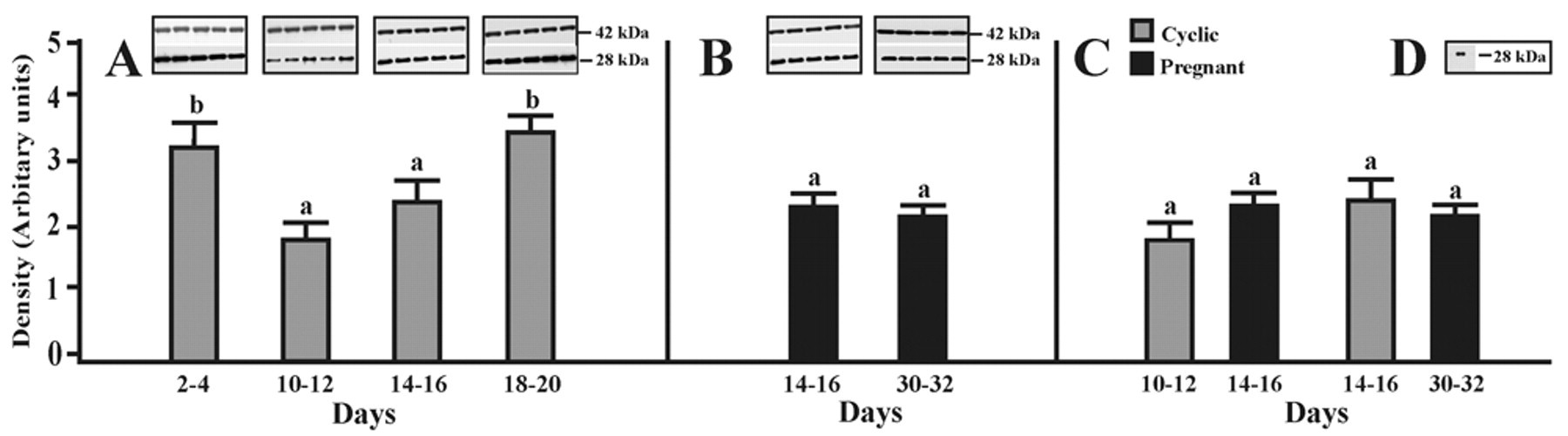
Western blot analysis of AQP5 expression. Expression of AQP5 assessed by Western blotting in the porcine oviduct on days 2–4, 10–12, 14–16, and 18–20 of the estrous cycle (A) as well as on days 14–16 and 30–32 of pregnancy (B). Densitometric analyses of AQP5 protein levels normalized against β-actin (A, B). In addition, comparison of the AQP5 expression during two stages of the cycle and two periods of pregnancy is shown in panel C. Positive control for Western blot analysis of the porcine lung (D). Values are the mean ± SEM (n = 5). a,bMeans with different superscripts are significantly different.
The expression of AQP9 in the porcine oviduct at different stages of the estrous cycle and pregnancy is shown and summarized in Figure 3A–C. A band of AQP9 protein product of the expected size (32 kDa) was clearly detected in all stages of the estrous cycle and pregnancy (Fig. 3A,B). In cyclic gilts, the expression of AQP9 did not change significantly between mid- and late-luteal phases but increased (p < 0.05) on days 2–4 and 18–20 when compared with days 10–12 and 14–16 of the cycle (Fig. 3A). In pregnant gilts, the expression of AQP9 did not change significantly during the onset and the end of implantation process (Fig. 3B) and also when compared to mid- and late-luteal phases of the estrous cycle (Fig. 3C). Immunoblotting with the anti-AQP9 antibody revealed AQP9 in porcine liver samples, which served as a positive control for AQP9 (Fig. 3D). Negative controls for immunoblot analyses were performed by omitting or replacing the primary antibodies against AQP1, 5, and 9 with non-immunized rabbit immunoglobulins; no immunostaining was observed (data not shown).
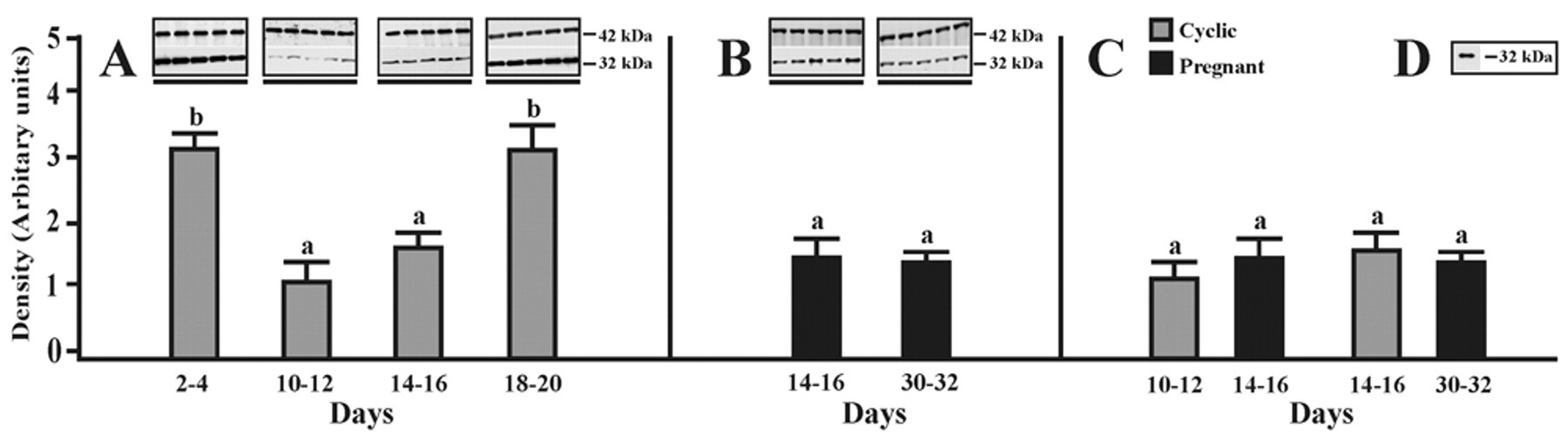
Western blot analysis of AQP9 expression. Expression of AQP9 assessed by Western blotting in the porcine oviduct on days 2–4, 10–12, 14–16, and 18–20 of the estrous cycle (A) as well as on days 14–16 and 30–32 of pregnancy (B). Densitometric analyses of AQP9 protein levels normalized against β-actin (A, B). In addition, comparison of the AQP9 expression during two stages of the cycle and two periods of pregnancy is shown in panel C. Positive control for Western blot analysis of the porcine liver (D). Values are the mean ± SEM (n = 5). a,bMeans with different superscripts are significantly different.
Immunohistochemistry
Figure 4 shows the expression of AQP protein in the porcine oviduct at different stages of the estrous cycle and pregnancy (see Materials and Methods). Immuno-histochemical analysis confirmed expressions of AQP1, 5, and 9 in the porcine oviduct throughout all examined reproductive stages. AQP1 immunoreactivity was detected in the capillary endothelium of the oviduct (Fig. 4A–F). As a positive control, AQP1 labeling was seen in the apical and basolateral plasma membranes of the proximal tubule cells in the pig kidney (Fig. 4S), which is consistent with the previous findings in the kidneys of mice, rats, pigs, and humans (Nielsen et al. 1995; Maunsbach et al. 1997; Skowronski 2010; Ma et al. 1998). AQP5 was localized in epithelial cells (Fig. 4G1–L1) and smooth muscle cells (Fig. 4G2–L2) of the pig oviduct. Consistent with previous findings seen in the lung tissues of mice, rats, pigs, and humans (Nielsen et al. 1997; Krane et al. 2001; Skowronski 2010; Kreda et al. 2001), the AQP5 antibody noticeably stained (Fig. 4U) the apical membrane of type I porcine pulmonary epithelial cells as a positive control. The anti-AQP9 antibody, similar to AQP5, labeled the epithelial cells of the oviduct (Fig. 4M–R). As a positive control, AQP9 staining was seen on the sinusoidal surfaces of hepatocyte plates in the pig liver (Fig. 4X), which is in agreement with a previous observation in the liver tissues of mice, rats, and pigs (Rojek et al. 2007; Carbrey et al. 2003; Skowronski 2010).
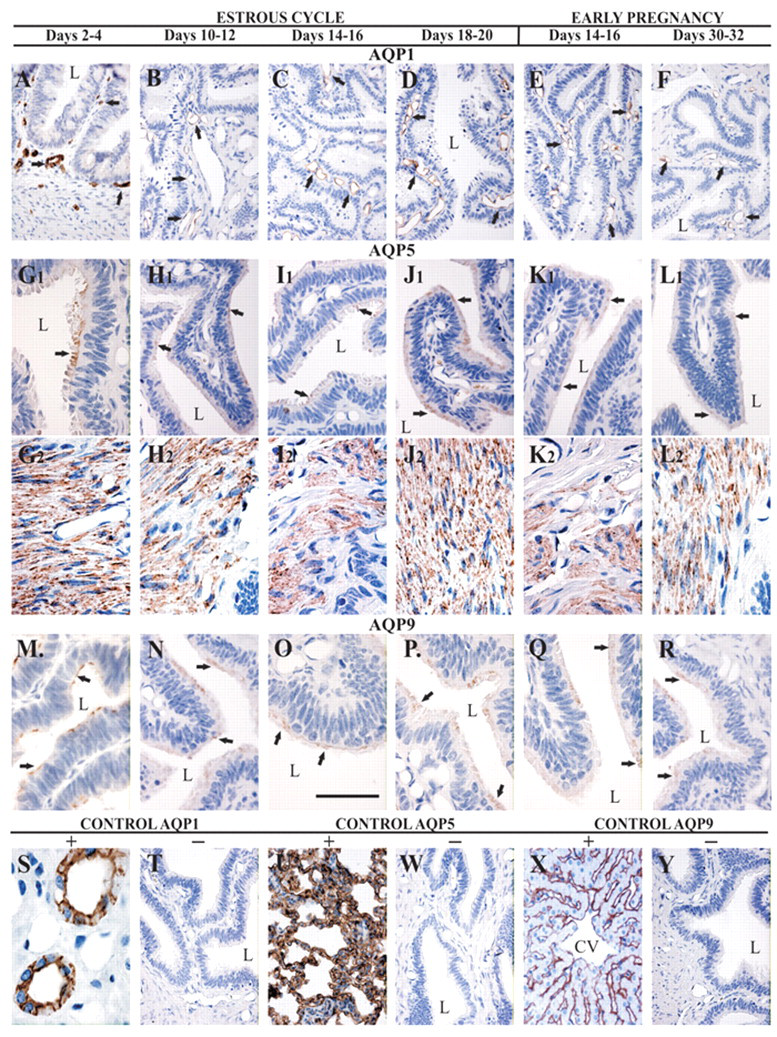
Immunolocalization of AQP1, 5, and 9. Immunohistochemical staining of AQP1 in paraffin-embedded sections of the oviduct from pigs. Anti-AQP1 antibody labels capillary endothelium of the oviduct (A–F). Immunoperoxidase labeling of AQP1 from the pig kidney cortex (positive control) (S). The labeling is seen in both the apical cell membrane and the basolateral cell membrane in proximal tubule cells. AQP5 antibody stains epithelial cells (G1-L1) and smooth muscle cells (G2-L2). The anti-AQP5 labels apical membrane of type I pulmonary epithelial cells of the pig (positive control) (U). AQP9 labels epithelial cells of the oviduct (M–R). Immunoperoxidase labeling of AQP9 from the pig liver (positive control) (X). No staining was observed with using non-immune immunoglobulins (negative controls) (T, W, and Y). L, lumen; CV, central vein. Bar = 50 µm.
Negative controls for all immunohistochemical analyses were performed by omitting (data not shown) or replacing the primary antibodies against AQPs with non-immunized rabbit immunoglobulins; no immunostaining was observed (Fig. 4T, W, Y).
Discussion
In this study, immunohistochemistry expression of AQP1, 5, and 9 in porcine oviduct during the estrous cycle and early pregnancy showed AQP1 in the oviductal capillary, AQP5 in the muscle layer, and both AQP5 and AQP9 in the epithelial cells. Our previous study (Skowronski et al. 2009) showed the same pattern of labeling of the above-mentioned AQPs in the pig oviduct on days 17 to 19 of the estrous cycle. Branes et al. (2005) demonstrated that the oviduct of cycling rats expresses AQP5, 8, and 9 in epithelial cells. Gannon et al. (2000) found AQP1 labeling in the cell membrane of smooth muscle cells of the rat oviduct. These studies suggest that increased water movement into oviductal muscles through AQP1 could lead to muscle swelling that shuts down the lumen. These changes might be implicated in ovum transport toward the uterus. One hypothesis (“tube locking”) concerning the mechanism of ovum transport suggests that the ovum is delayed at the ampullar–isthemic junction, probably as a result of muscle contraction, ischemic edema, and vascular distention (reviewed by Huang et al. 2006). In contrast, the pig oviductal muscles exhibited AQP5 expression but not AQP1. However, the expression of AQP1 was present in the pig oviductal vessels. The localization of AQP1 in the oviductal capillaries is a reasonable result, whereas the presence of AQP5 in the smooth muscle is unusual in view of its distribution in other organs. Recently, Skowronski (2010) confirmed AQP5 protein expression in the smooth muscle cells of the pig uterus. In addition, the author showed AQP5 mRNA expression in these cells by RT-PCR. Helguera et al. (2009) and Girotti and Zingg (2003) reported AQP5 and AQP8 expression in rat myometrium by microarray or quantitative RT-PCR (qRT-PCR) and by microarray, respectively. Immunohistochemical experiments performed by Gawronska et al. (1999, 2000) revealed luteinizing hormone/human chorionic gonadotropin (LH/hCG) receptor protein expression in the epithelium of tubal mucosa, smooth muscle cells, and blood vessel endothelium in pigs. However, LH/hCG receptors in the mouse were detected in the serosa and subepithelial cells but not in the epithelium of the oviduct (Zhang et al. 2001). This discrepancy may reflect the species differences or results from different experimental techniques. For AQP localization in the oviductal cells, a similar discrepancy may occur between species.
Our results clearly demonstrated that AQP1, 5, and 9 protein expression in the porcine oviduct is influenced by estrous cycle and pregnancy. The expression of AQP1, 5, and 9 protein did not change significantly between days 10–12 and 14–16 but increased on days 2–4 and 18–20 when compared with days 10–12 and 14–16 of the cycle. In pregnant gilts, the expression of AQP1, 5, and 9 did not change significantly during the onset and the end of the implantation process and also when compared to mid- and late-luteal phases of the estrous cycle. Branes et al. (2005) detected that the oviductal expression of epithelial AQP5, 8, and 9 was lost in ovariectomized rats. However, only AQP9 expression was restored after a specific combination and timing of estradiol (E2) and/or progesterone (P4) administrations, further supporting a role for AQP9 in fluid formation. The authors suggest that sex steroid hormones control AQP9 expression and that other molecules produced by the ovary or by other organs in an ovary-dependent manner could control AQP5 and AQP8 expression. Moreover, Branes et al. (2005) found that the recovery of AQP9 reactivity after ovariectomy correlated with increased mRNA and protein levels after treatment with E2 alone or P4 administration after E2 priming. Interestingly, P4 administration after P4 priming also induced AQP9 expression but without a change in mRNA levels. Levels of AQP9 varied along the estrous cycle, with their highest levels during proestrus and estrus (Branes et al. 2005). This is in agreement with our present data where AQP1, 5, and 9 expression increased on days 2–4 and 18–20 of the estrous cycle.
The recent study by Skowronski (2010) confirms our present findings that AQP5 is expressed in smooth muscle cells of the female pig reproductive system. The myometrial expression of AQP5 protein did not differ significantly during the estrous cycle but increased in pregnancy (Skowronski 2010). A recent study by Helguera et al. (2009) showed in rat myometrium a significant downregulation of AQP5 and AQP8 during delivery, underscoring their potential role in parturition. Moreover, both AQP5 and AQP8 were upregulated during the first 20 days of pregnancy. However, both proteins were downregulated from day 20 to day 23 of pregnancy (Helguera et al. 2009; Girotti and Zingg 2003). There was no change in the expression of AQP5 when comparing term versus labor, whereas AQP8 was further downregulated in labor (Girotti and Zingg 2003). In the present study, AQP1, 5, and 9 protein expression in the pig oviduct did not differ when comparing periods of pregnancy versus mid- and late-luteal phases. However, the expression of the above-mentioned AQPs significantly increased in the pig uterus during early pregnancy when compared to the estrous cycle (Skowronski 2010).
It is well known that both muscular contractions and cilia activity are involved in the transportation of gametes and embryos. Wånggren et al. (2008) showed that muscular contractions of the human oviduct increased after treatment with prostaglandin F2α (PGF2α) and prostaglandin E2 (PGE2). The contractions decreased after PGE1, P4, levonorgestrel, mifepristone, oxytocin, and hCG. Recently, Marino et al. (2010) found that hCG may increase AQP9 protein expression and functionality in human preeclamptic placenta via cAMP pathways. They speculate that as a neutral solute channel, AQP9 may be implicated in energy metabolism or may participate in the clearance of excess lactate in the extracellular space during placental ischemia in preeclamptic placentas. The hormonal status of the pig may also influence the LH/hCG receptor expression levels in the porcine oviduct (Gawronska et al. 1999). It has been shown that spontaneous contractions of the porcine oviduct could be modulated by LH during various stages of the estrous cycle (Gawronska et al. 1999). In vitro experiments showed that LH can modulate the oviduct contractility in gilts primed with both E2 and P4 but not in gilts primed only with P4, E2, or corn oil (Gawronska et al. 1999, 2000). However, further studies are required to substantiate the hypotheses of LH role in regulation of AQPs in the oviduct.
Hara-Chikuma and Verkman (2008) showed that AQP3 facilitated water transport in epidermal cell migration and glycerol transport in epidermal cell proliferation. Steffl et al. (2004) found that estrogen may induce the initial proliferation of secretory cells and promote the differentiation into ciliated cells in the pig oviduct. Estradiol may play a key role during the proliferation and differentiation of porcine oviduct tissues by activating the important paracrine or autocrine epidermal growth factor (EGF) system through its receptor (Wollenhaupt et al. 2001). Moreover, the vascular endothelial growth factor (VEGF) system and fibroblast growth factor (FGF) system were detected in porcine oviductal and endometrial tissue during the estrous cycle and the time of implantation (Wollenhaupt et al. 2004). The authors suggest that these growth factor systems seem to be involved in a supposed paracrine network to successfully establish and maintain pregnancy in pigs.
During the period of late follicular development and the first 4 days of the estrous cycle, the oviduct occupies a central role in the establishment of pregnancy (Buhi et al. 1997). In the present study, AQP1, 5, and 9 expression was the highest on days 2–4 and 18–20 of the estrous cycle.
Taken together, the results of our study provide an anatomical basis for AQP1, 5, and 9 expression in the porcine oviduct during different stages of the estrous cycle and pregnancy. As clearly demonstrated, AQP1, 5, and 9 protein expression is influenced by estrus cycle and pregnancy. Furthermore, localization of AQPs in the porcine oviduct may provide an environment that sustains and enhances fertilization and early cleavage-stage embryonic development. Therefore, our results could be useful for future studies investigating the establishment of pregnancy and implantation of porcine embryos.
Footnotes
Acknowledgements
The authors thank Prof. Stanislaw Okrasa for comments and suggestions.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This research was supported by the Polish Ministry of Science and Higher Education (grant numbers N N308 0042 33 and 0206.0805).
