Abstract
Aquaporins (AQPs) are membrane water channel proteins expressed in various tissues in the body. We surveyed the immunolocalization of AQP3, an isoform of the AQP family, in rat epithelial tissues. AQP3 was localized to many epithelial cells in the urinary, digestive, and respiratory tracts and in the skin. In the urinary tract, AQP3 was present at transitional epithelia. In the digestive tract, abundant AQP3 was found in the stratified epithelia in the upper part, from the oral cavity to the forestomach, and in the simple and stratified epithelia in the lower part, from the distal colon to the anal canal. In the respiratory tract, AQP3 was present in the pseudostratified ciliated epithelia from the nasal cavity to the intrapulmonary bronchi. In the skin, AQP3 was present in the epidermis. Interestingly, AQP3 was present at the basal aspects of the epithelia: in the basolateral membranes in the simple epithelia and in the multilayered epithelia at plasma membranes of the basal to intermediate cells. During development of the skin, AQP3 expression commenced late in fetal life. Because these AQP3-positive epithelia have a common feature, i.e., they are exposed to an environment of possible water loss, we propose that AQP3 could serve as a water channel to provide these epithelial cells with water from the subepithelial side to protect them against dehydration.
R
In the kidney collecting duct principal cells, AQP3 is localized at the basolateral plasma membranes (Ecelbarger et al. 1995; Frigeri et al. 1995a; Ishibashi et al. 1994; Ma et al. 1994). AQP2 resides in intracellular vesicles and moves to the apical plasma membranes when stimulated by vasopressin (Nielsen et al. 1995; Yamamoto et al. 1995). A combined action of AQP2 and AQP3 appears to be important in water reabsorption (Fushimi et al. 1993; Ishibashi et al. 1994). In addition, AQP3 is expressed in several extrarenal organs, whereas AQP2 is specifically expressed in the kidney (Fushimi et al. 1993; Umenishi et al. 1996). To elucidate a possible role of AQP3 in these organs, we systematically surveyed the localization of AQP3 in extrarenal organs.
Although localization of AQP3 has been described sporadically (Frigeri et al. 1995a,b; Nielsen et al. 1997; Hamann et al. 1998), its functional importance has not been discussed in depth. In this study we examined the comprehensive and detailed immunolocalization of AQP3 in rat epithelial tissues and the developmental pattern of its expression in the skin, and propose a possible role of AQP3. We found that AQP3 is present at the epithelial cells exposed to an environment of possible water loss. We speculate that AQP3 serves as a water channel to provide these epithelial cells with water from the subepithelial side to protect them from dehydration.
Materials and Methods
Anti-AQP3 Antibody
Oligopeptides corresponding to the COOH terminal amino acids of rat AQP1 (257-269), AQP2 (257-271), AQP3 (264-279), AQP4 (311-323), and AQP5 (251-265) were synthesized with a Model 431A peptide synthesizer (Applied Biosystems; Foster, CA). Anti-AQP3 antibody was raised in a rabbit using the AQP3 peptide conjugated to keyhole limpet hemocyanin (Pierce; Rockford, IL).
Animals
Adult male Wistar rats 6 weeks of age, fetal Wistar rats at embryonic Days 15 and 18, and 4-day postnatal rats were used. All animal experiments were in compliance with the NIH Guide for the Care and Use of Laboratory Animals.
Immunoblotting
In adult rats, specimens of kidney inner medulla, mucosal layer of the forestomach, and palmar skin of the hind leg were removed. In the 4-day postnatal rats, specimens of palmar skin of the hind leg were removed. In the fetuses at embryonic Days 15 and 18, specimens of hind legs were removed. These specimens were cut into small pieces and quickly frozen with liquid nitrogen until used. Specimens were homogenized with a glass homogenizer in 10 volumes of ice-cold homogenization buffer consisting of PBS with 2 μg/ml pepstatin A, 2 μg/ml leupeptin, 100 KIE/ml aprotinin, and 2 mM phenylmethylsulfonyl fluoride. Proteins were determined with a BCA Protein Assay Kit (Pierce). Samples were denatured at 37 C for 10 min or at 70 C for 10 min in denaturation buffer composed of 2% SDS, 25 mM Tris-HCl, pH 7.5, 25% glycerol, 0.005% bromophenol blue, 23 mg/ml dithiothreitol, 300 μg/ml DNase I, and 4 mM MgCl2. SDS-PAGE and immunoblotting were carried out by a standard method using [125 I]-protein A or [125 I]-protein G (NEN; Wilmington, DE) (Ezaki et al. 1986; Takata et al. 1990). As a primary antibody, anti-AQP3 antibody (1:5000 dilution) was used. As controls, the primary antibody was replaced with anti-AQP3 antibody (1:5000 dilution) preincubated with 50 μg/ml COOH terminal peptide of AQP3. The signals were scanned with a BAS2000 bioimaging analyzer (Fuji Film; Tokyo, Japan).
Immunofluorescence Microscopy
From adult rats, specimens of (a) the urinary system: kidney, ureter, urinary bladder, and urethra; (b) body surface: skin from palm, ear, lower lip, anus, abdomen, back, and eyelid; (c) the digestive tract: lower lip, tongue, esophagus, stomach, duodenum, jejunum, ileum, cecum, proximal colon, distal colon, rectum, and anus; and (d) the respiratory tract: nasal cavity wall, trachea, bronchus, and lung, were removed. From rats at postnatal Day 4 and fetuses at embryonic Days 15 and 18, specimens of hind legs and skin from the abdomen were removed. These specimens were cut into small pieces, fixed in 3% formaldehyde in 0.1 M sodium phosphate buffer, pH 7.4, for 3 hr on ice. For preparation of cryostat sections, formaldehyde-fixed specimens were infused with 20% sucrose in PBS overnight, embedded in Tissue-Tek OCT compound (Sakura Finetechnical; Tokyo, Japan), and rapidly frozen with liquid nitrogen. Cryostat sections 3-6 μm thick were cut, mounted on poly-L-lysine-coated glass slides, immediately immersed in ethanol at −20C for 30 min, and rinsed with PBS. For semithin frozen sections, formaldehyde-fixed specimens were infused with 2.3 M sucrose in 0.1 M sodium phosphate buffer, pH 7.4, overnight (Takata and Singer 1988). Semithin frozen sections 1 μ thick were cut, mounted on poly-L-lysine-coated glass slides, washed with PBS, immersed in ethanol at room temperature for 30 min, and rinsed with PBS. Indirect immunofluorescence staining was carried out basically as described previously (Takata et al. 1991). In short, rabbit anti-AQP3 antibody (1:500 dilution) was used as a primary antibody. Lissamine-rhodamine sulfonyl chloride (LRSC)-conjugated donkey anti-rabbit IgG (Jackson Immunoresearch; West Grove, PA) was used as a secondary antibody. Both antibodies were diluted with 5% normal goat serum-PBS. For nuclear counterstaining, 2 μg/ml 4′,6-diamidino-2-phenyl-indole (DAPI) was included in the secondary antibody solution. To check the specificity of the immunostaining, we performed the following controls: (a) incubation with normal rabbit serum (1:500 dilution) instead of anti-AQP3 antibody; (b) absorption controls by preincubating the anti-AQP3 antibody (1:500 dilution) with COOH terminal peptide of AQP3 protein (10 μg/ml) used as immunogen; (c) absorption controls by preincubating the anti-AQP3 antibody (1:500 dilution) with a mixture of COOH terminal peptides of AQP1, −2, −4, and −5 proteins (10 µg/ml each). Specimens were examined with an AX-70 microscope equipped with Nomarski differential interference contrast and epifluorescence optics (Olympus; Tokyo, Japan).
Immunoelectron Microscopy
Cryostat sections 10-16 μm thick from formaldehyde-fixed specimens of the urinary bladder and lower lip were cut, mounted on poly-L-lysine-coated glass slides, immediately immersed in ethanol at −20 C for 30 min, and rinsed with PBS. After incubation with the rabbit anti-AQP3 antibody (1:500 dilution), they were then incubated with Nanogoldconjugated anti-rabbit IgG (1:50 dilution; Nanoprobe, Stony Brook, NY), washed with PBS, and fixed with 1% glutaraldehyde in 0.1 M sodium phosphate buffer, pH 7.4, for 10 min (Shin et al. 1996). Labeled sections were rinsed with water and then incubated in silver acetate solution for an appropriate time (Murata et al. 1992). They were next rinsed quickly with water and immersed in 0.05% sodium acetate for 1 min, rinsed extensively with water, treated with 0.05% gold chloride for 2 min, and again rinsed extensively with water (Sawada and Esaki 1994). Thereafter, specimens were fixed with 1% osmium tetroxide in 0.1 M sodium phosphate buffer, pH 7.4, for 30 min, dehydrated with ethanol, and embedded in Epon. Ultrathin sections were cut, stained with uranyl acetate and lead citrate, and examined with a JEM-1010 electron microscope (JEOL; Tokyo, Japan). To check the specificity of the immunostaining, we performed the following controls: (a) incubation with normal rabbit serum (1:500 dilution) instead of anti-AQP3 antibody; (b) absorption controls by preincubating the anti-AQP3 antibody (1:500 dilution) with COOH terminal peptide of AQP3 protein (10 μg/ml) used as immunogen.
Results
Antibody Specificity
By immunoblotting with anti-AQP3 antibody, two bands were observed in the homogenates of the kidney inner medulla as previously reported (Ecelbarger et al. 1995; Ishibashi et al. 1997c), i.e., a sharp band at 26 kD of the nonglycosylated form and a broad band at 32-40 kD of glycosylated forms (Figure 1A). These 26-kD and 32-40-kD proteins were not detected with the anti-AQP3 antibody in the presence of COOH terminal peptide of the AQP3 protein used as immunogen (Figure 1B).
Immunohistochemistry and immunoblotting in the kidney with rabbit anti-AQP3 antibody gave the same results as those in previous studies (Ecelbarger et al. 1995), i.e., the labels were localized at the basolateral membranes of the collecting duct cells (Figures 2A and 2B). To further confirm the specificity of the staining, we performed control experiments. Replacement of anti-AQP3 antibody with normal rabbit serum gave no specific labeling (not shown). Replacement of the anti-AQP3 antibody with a mixture of anti-AQP3 antibody preincubated with the COOH terminal peptide of AQP3 protein gave no specific labeling (Figures 2C and 2D). Addition of a mixture of COOH terminal peptides of AQP1, −2, −4, and −5 proteins, on the other hand, did not affect AQP3 staining (Figures 2E and 2F). These results, along with the results of the immunoblotting, confirmed that the rabbit anti-AQP3 antibody specifically detected the AQP3 protein.
Immunoblotting
By immunoblotting with this anti-AQP3 antibody, one sharp band at 26 kD and one broad glycosylated band at 32-40 kD were detected in the homogenates of both forestomach and palmar skin, similar to those in the kidney (Figure 1A). These bands disappeared in the presence of COOH terminal peptide of the AQP3 protein (Figure 1B).
Immunofluorescence Microscopy
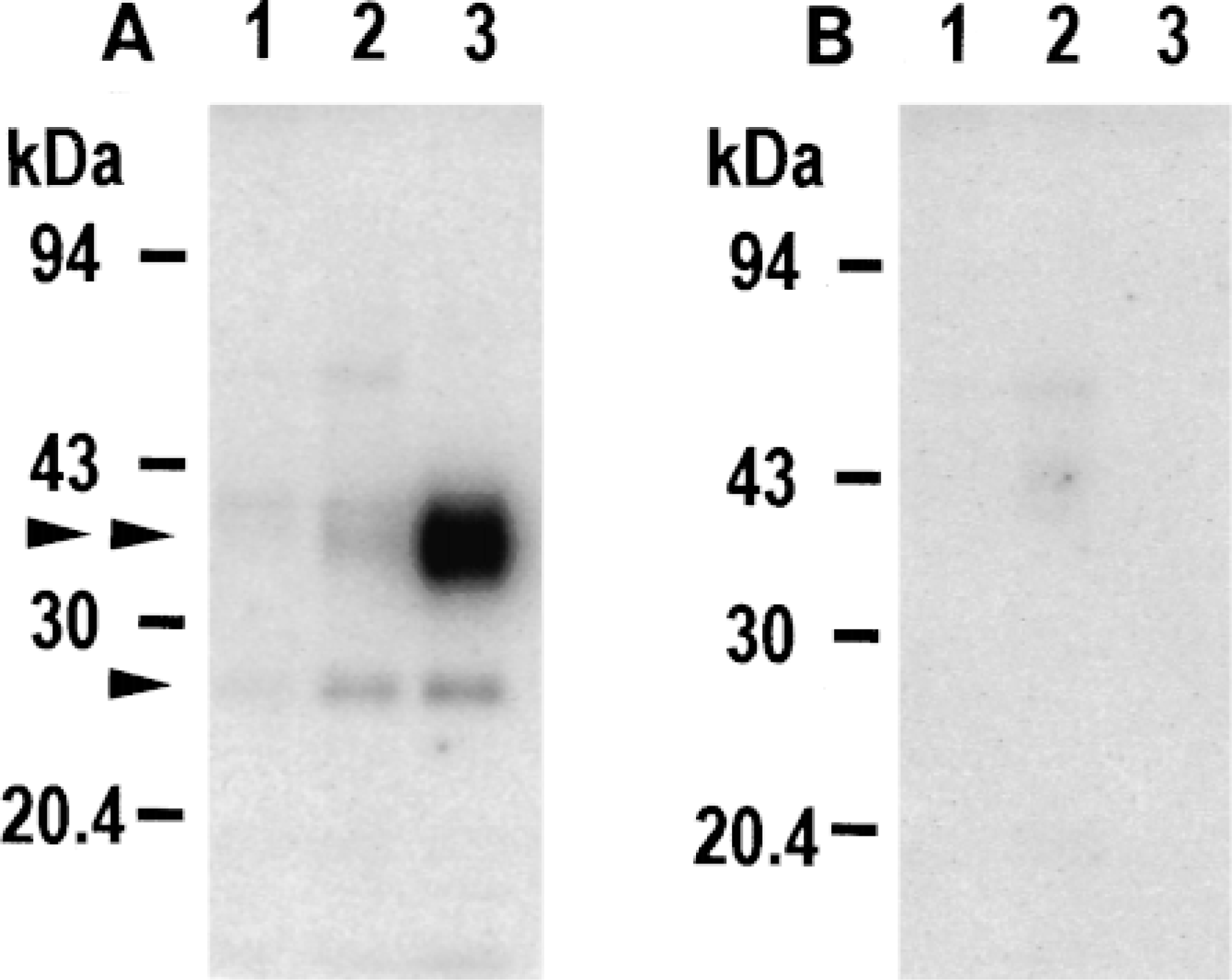
Immunoblotting of rat tissues with antibody against AQP3. Ten micrograms of protein of tissue homogenate prepared from mucosal layer of forestomach (Lane 1), palmar skin (Lane 2), and 5 μg of homogenate from kidney inner medulla (Lane 3) were applied to SDS-PAGE (13% acrylamide), electrophoresed, and subjected to immunoblotting with anti-AQP3 antibody (A) or with anti-AQP3 antibody preincubated with COOH terminal peptide of AQP3 protein used as immunogen (B). Arrowhead and double arrowhead indicate 26-kD nonglycosylated AQP3 and 32-40-kD glycosylated AQP3, respectively.
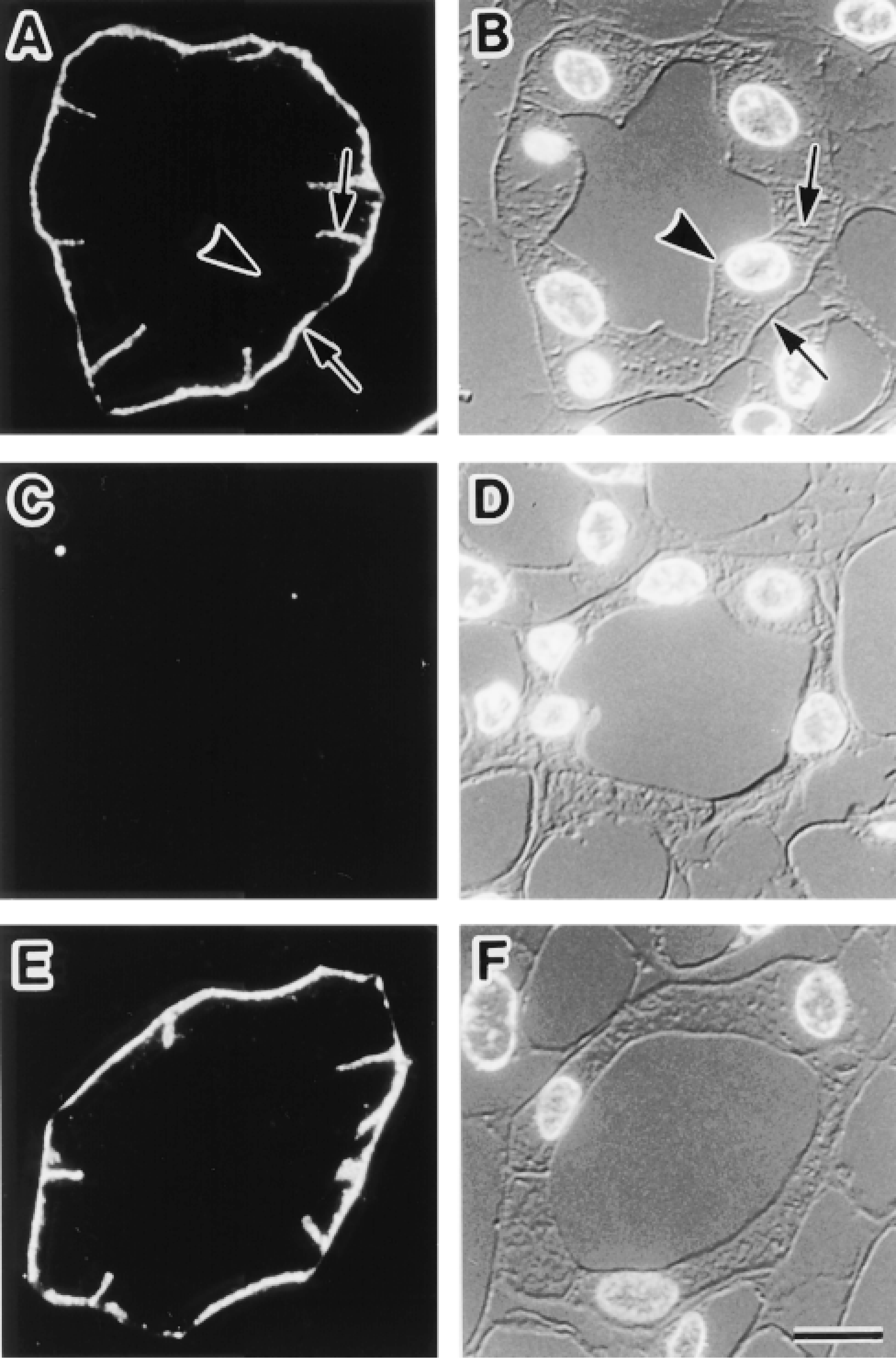
Immunofluorescence localization of AQP3 in the kidney collecting duct. Semithin frozen sections from kidney inner medulla were stained. Fluorescence images for AQP3 (A,C,E) and corresponding Nomarski differential interference contrast images counterstained with DAPI for nuclei (B,D,F) are shown. (A,B) AQP3 is localized at basolateral membranes (arrows), whereas it is absent from apical membranes (arrowhead). (C-F) Histochemical controls. (C,D) A section was incubated with anti-AQP3 antibody in the presence of COOH terminal peptide of AQP3 protein used as immunogen. No positive labeling is seen. (E,F) A section was incubated with anti-AQP3 antibody in the presence of a mixture of COOH terminal peptides of AQP1, −2, −4, and −5 proteins. The labeling pattern is the same as in A. Bar = 10 μm.
Immunofluorescence localization of AQP3 in the urinary tract. Cryostat sections were stained. Fluorescence images for AQP3 (A,C,E) and corresponding Nomarski differential interference contrast images counterstained with DAPI for nuclei (B,D,F) are shown. (A-D) Ureter (A,B) and urinary bladder (C,D). Label for AQP3 is present at the basal and intermediate cell layers of the transitional epithelia, where the label is seen along the plasma membranes (arrows). The labeling intensity gradually decreases towards the surface and no label for AQP3 is seen in large superficial cells (arrowheads). (E,F) Urethra. Labels for AQP3 are seen in the epithelium. Bars: A,B,E,F = 20 μm; C,D = 10 μm.
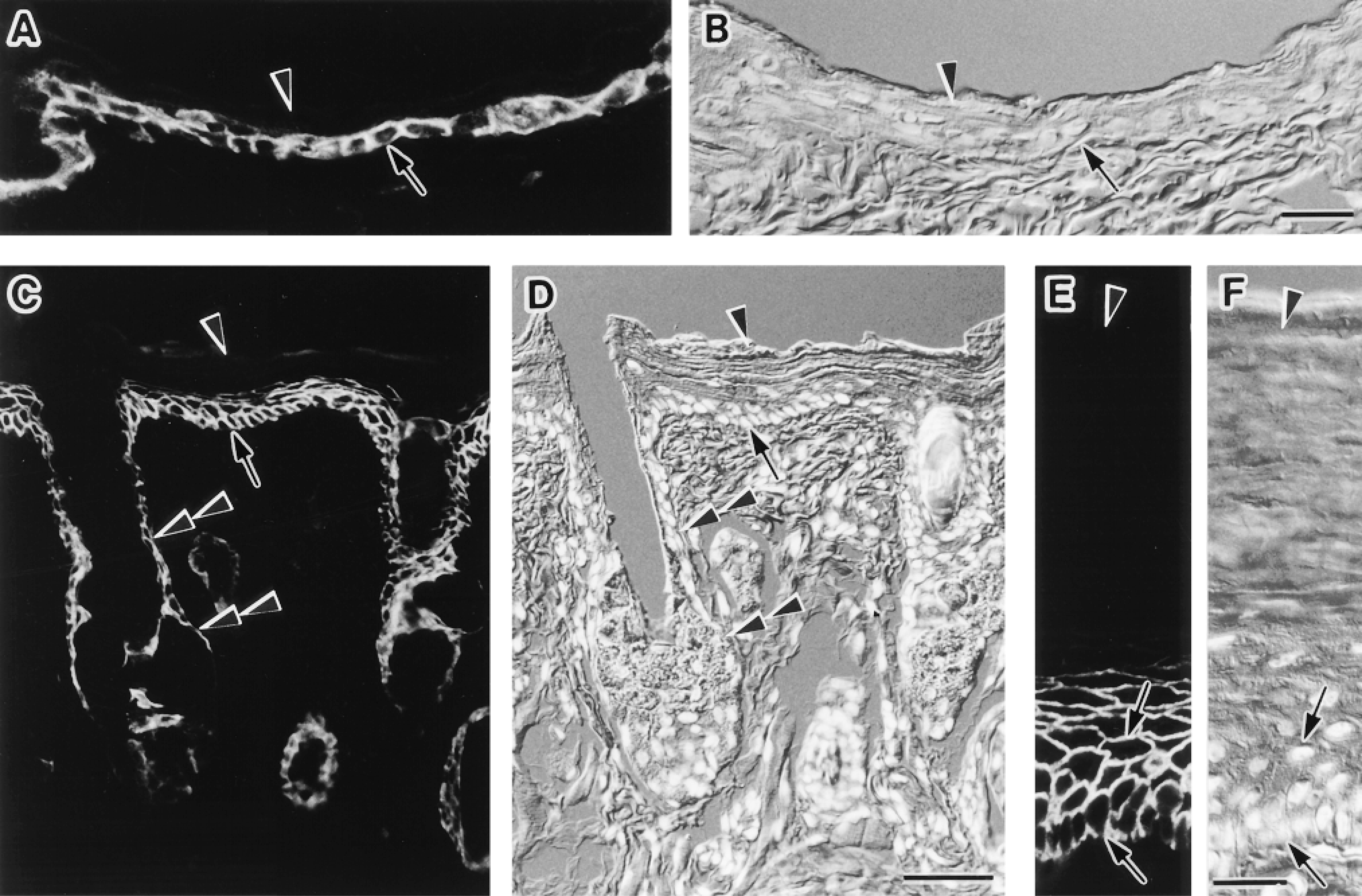
Immunofluorescence localization of AQP3 in the body surface. Cryostat sections were stained. Fluorescence images for AQP3 (A,C,E) and corresponding Nomarski differential interference contrast images counterstained with DAPI for nuclei (B,D,F) are shown. (A,B) Abdominal skin. The epidermis is thin, and AQP3 is present in a few layers from the basal layer (arrow). No label is seen in the stratum corneum (arrowhead). Bar = 20 μm. (C,D) Cutaneous surface of the lower lip. The epidermis is of intermediate thickness. AQP3 is present in the basal (arrow) and intermediate cell layers but is absent in the stratum corneum (arrowhead). AQP3 is also present along the external root sheath of the hair follicle and in the sebaceous gland (double arrowheads). Bar = 50 μm. (E,F) Palmar skin. The epidermis is thick. Label for AQP3 is intense in the basal and intermediate layers (arrows). Labeling intensity gradually decreases towards the surface from the basal cells, and no label for AQP3 is seen in the thick stratum corneum (arrowhead). Bar = 20 μm.
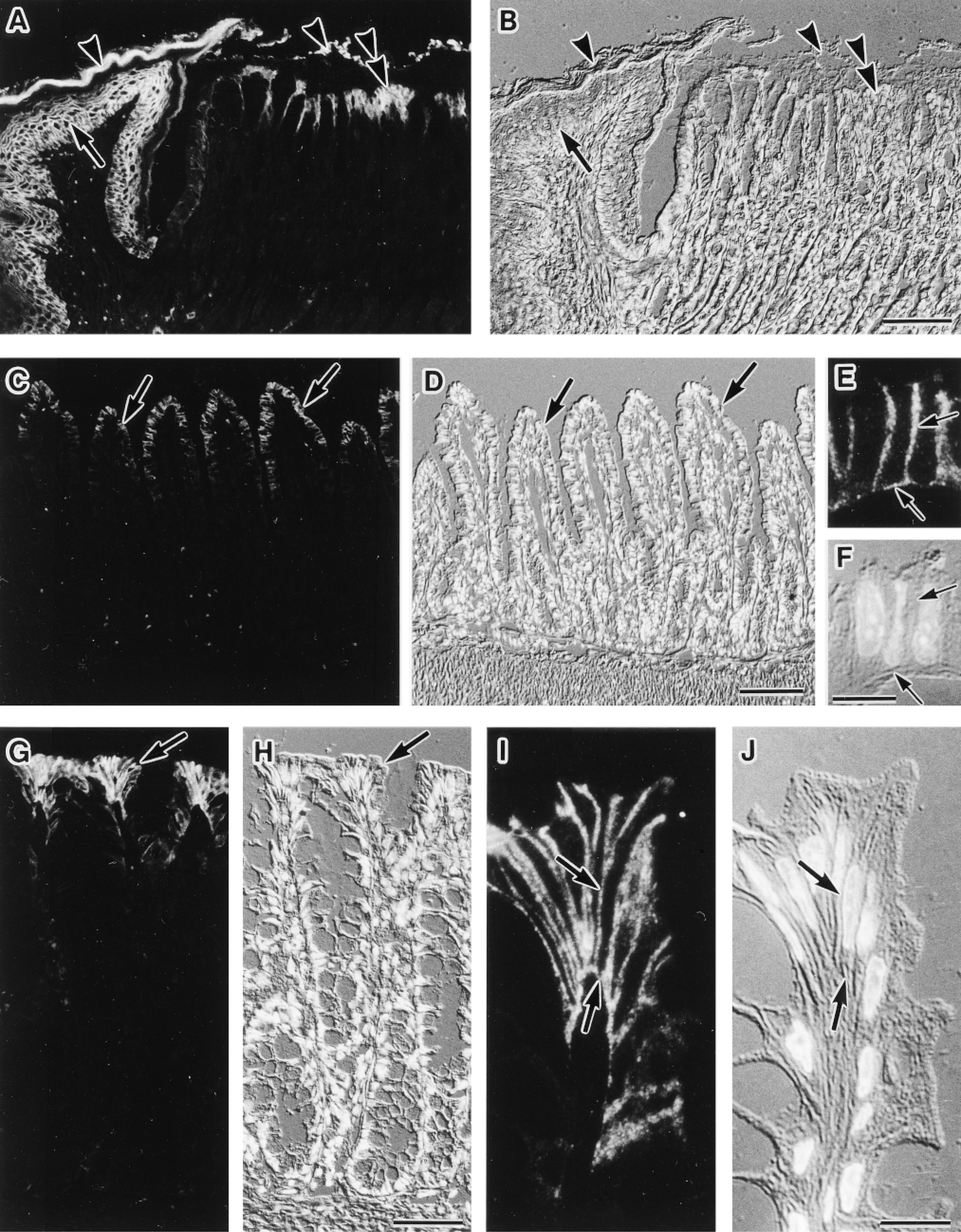
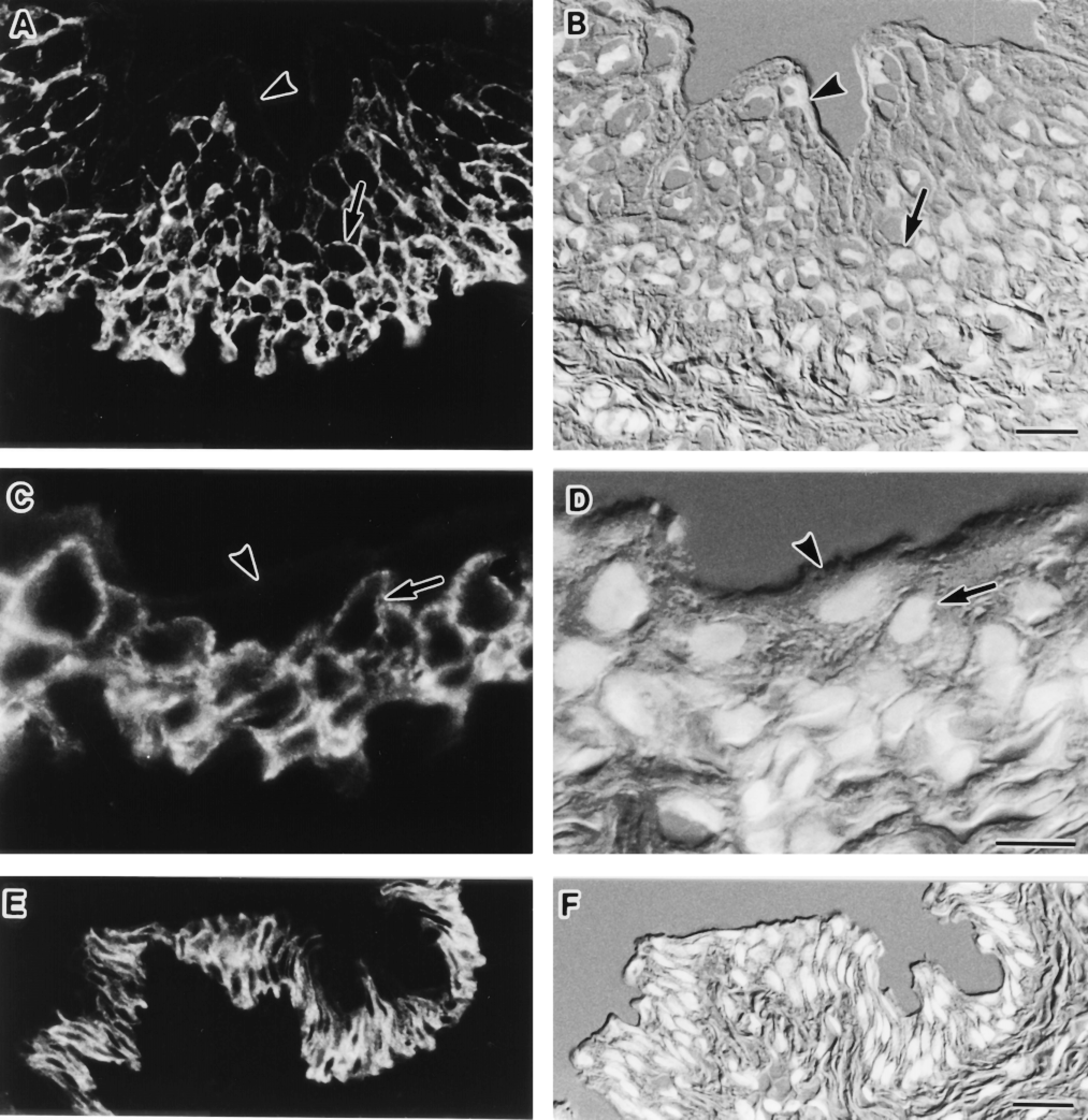
Immunofluorescence localization of AQP3 in the digestive tract. Cryostat sections (A-D,G,H) and semithin frozen sections (E,F,I,J) were stained. Fluorescence images for AQP3 (A,C,E,G,I) and corresponding Nomarski differential interference contrast images counter-stained with DAPI for nuclei (B,D,F,H,J) are shown. (A,B) Junctional zone between forestomach (nonglandular portion) and fundic stomach (glandular portion). The luminal surface of forestomach is covered with cornified stratified epithelium in which AQP3 is present in the basal and intermediate cell layers (arrow). The luminal surface of the fundic stomach is covered with simple columnar epithelium, in which AQP3 is present in surface mucous cells (double arrowhead). In the gastric pit, the labeling intensity gradually decreases according to the depth. No label is seen in cells deep in the gastric pits and in the gastric glands. The signal in the lumen (arrowheads) is also observed in the histochemical controls, hence is nonspecific. Bar = 100 μm. (C,D) Ileum. Only weak label for AQP3 is seen in absorptive epithelial cells (arrows) in the villous tip. Bar = 100 μm. (E,F) Enlarged view of AQP3-positive absorptive cells in the ileum. AQP3 is restricted to basolateral membranes (arrows). Bar = 10 μm. (G,H) Distal colon. AQP3 is present at absorptive epithelial cells (arrow) covering the lumen. In the crypt, positive label for AQP3 is restricted to the upper region and no label is seen in cells deep in the crypt. Bar = 50 μm. (I,J) Enlarged view of the AQP3-positive absorptive cells in the distal colon. AQP3 is restricted to basolateral membranes (arrows). Bar = 10 μm.
Immunoelectron Microscopy
We examined the ultrastructural localization of AQP3 with Nanogold probes in the transitional epithelium of the urinary bladder and the epidermis of the cutaneous surface of the lower lip. Label for AQP3 was present along plasma membranes of both transitional epithelial cells and epidermal keratinocytes (Figures 7A and 7C). No positive label for AQP3 was seen in the cytoplasmic organelles. Immunohistochemical controls gave no positive staining, showing the specificity of the stain (Figure 7B).
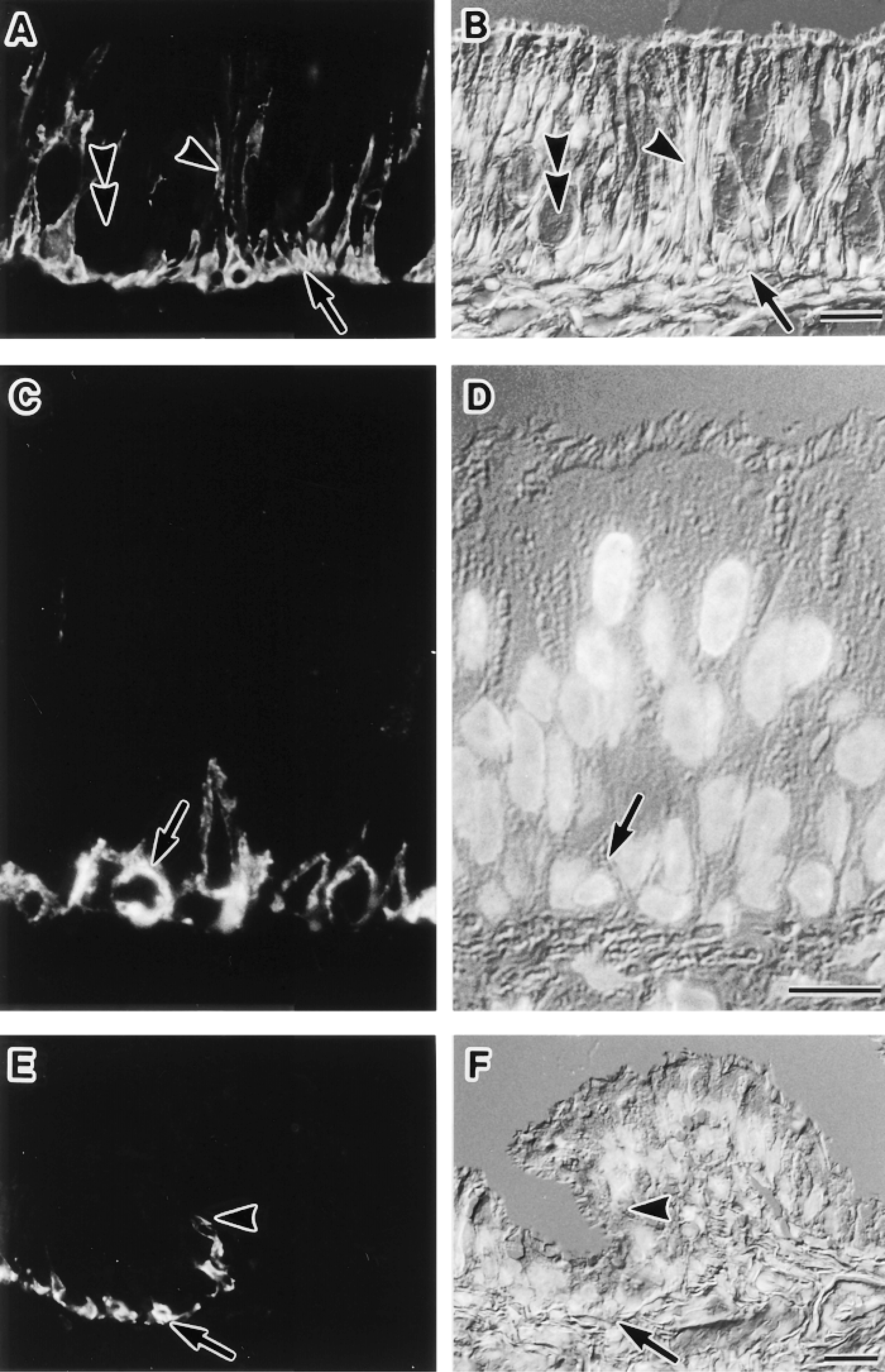
Immunofluorescence localization of AQP3 in the respiratory tract. Cryostat sections (A,B,E,F), and a semithin frozen section (C,D) were stained. Fluorescence images for AQP3 (A,C,E) and corresponding Nomarski differential interference contrast images counterstained with DAPI for nuclei (B,D,F) are shown. (A,B) Nasal cavity wall. AQP3 is seen in basal cells (arrow) and relatively large numbers of ciliated cells (arrowhead) but not in mucous cells (double arrowhead) in the pseudostratified ciliated epithelium. Bar = 20 μm. (C,D) Trachea. AQP3 is localized at plasma membranes of basal cells in the pseudostratified epithelium (arrow). Bar = 10 μm. (E,F) Proximal portion of the intrapulmonary bronchus. AQP3 is present in basal cells in the pseudostratified ciliated epithelium (arrow). The label for AQP3 disappears from the point indicated by the arrowhead. Bar = 20 μm.
Developmental Changes of AQP3 Expression in the Skin
By immunoblotting of the hind leg, AQP3 was not detected at embryonic Day 15. AQP3 was found in the hind leg at embryonic Day 18 as well as in the hind leg palmar skin at postnatal Day 4 (Figure 8). Next, we examined the immunolocalization of AQP3 in the hind leg and abdominal skin. No label for AQP3 was detected at embryonic Day 15 (Figures 9A and 9B), whereas at embryonic Day 18 AQP3 was present at the epidermis (Figures 9C and 9D). In the skin at postnatal Day 4, AQP3 was localized to the epidermal basal and intermediate cell layers but not in the stratum corneum, similar to that in the adult rat (Figures 9E and 9F).
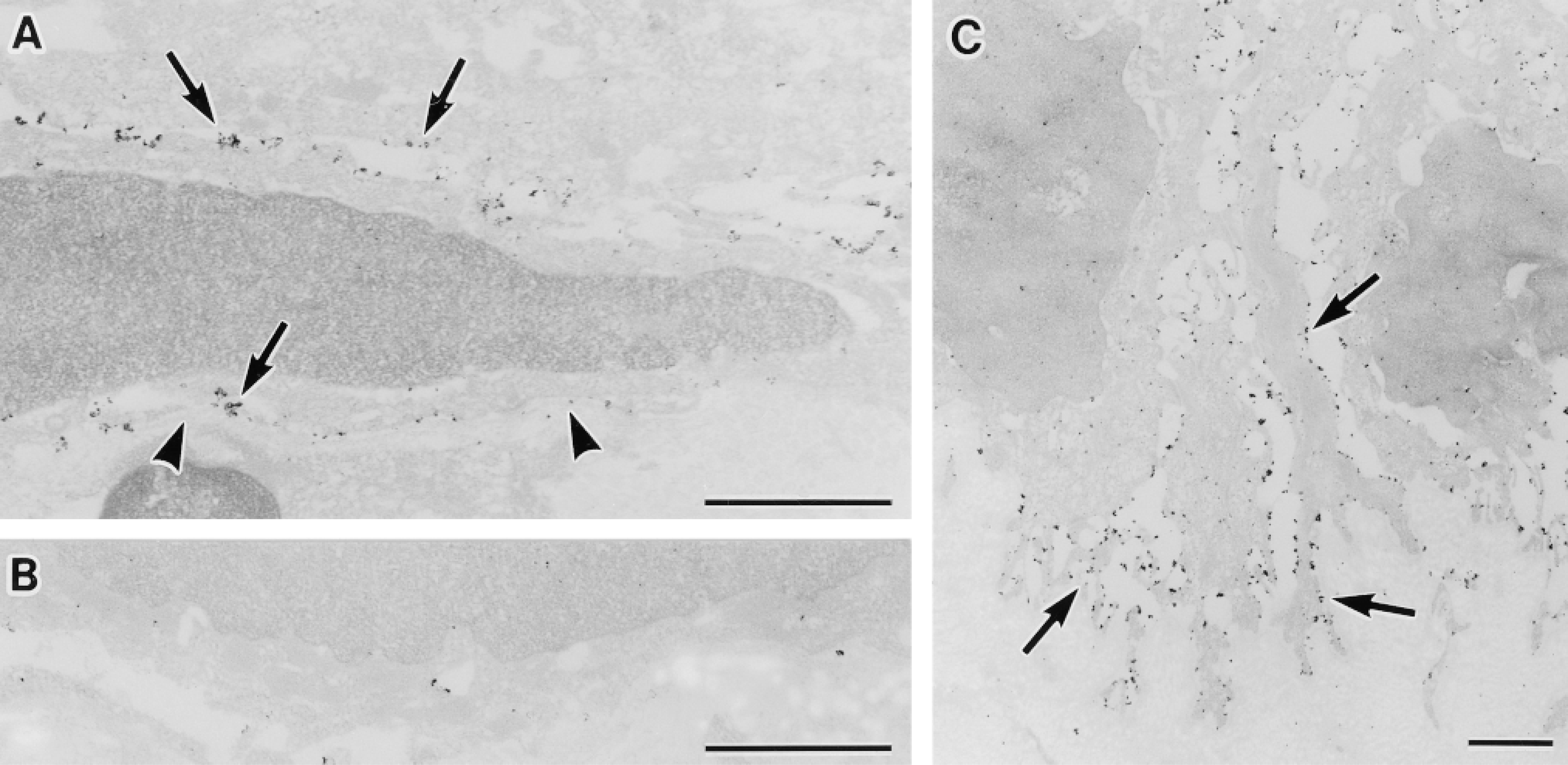
Ultrastructural localization of AQP3 in the urinary bladder (A,B) and skin (C). (A) Transitional epithelium of the urinary bladder. AQP3 is localized along the plasma membrane of the basal and intermediate cells (arrows). Arrowheads indicate basal lamina. (B) Histochemical control. A urinary bladder specimen was incubated with anti-AQP3 antibody in the presence of COOH terminal peptide of AQP3 protein used as immunogen. No positive labeling is seen. (C) Epidermis of the lower lip skin. AQP3 is localized along the plasma membrane of basal cells of the epidermis (arrows). Bars = 1 μm.
Discussion
We showed in this work that the water channel protein AQP3 was present in the epithelia covering the body surface and its continuities invaginated into the body, i.e., the urinary tract, digestive tract, and respiratory tract. AQP3 is expressed in the principal cells of the renal collecting ducts and is localized at the basolateral membrane (Ecelbarger et al. 1995). In combination with AQP2 at the apical membrane, the function of AQP3 is postulated to be transcellular water reabsorption, i.e., water transfer from the lumen to the interstitium transcellularly according to the osmotic gradient (Fushimi et al. 1993; Ishibashi et al. 1994). Frigeri et al. (1995b) showed previously at lower magnification that AQP3 is also present in the urinary bladder transitional epithelium. In the urinary tract downstream from the collecting ducts, it appears unlikely that water moves from the lumen to the subepithelial side. The luminal surface of the urinary tract is covered with transitional epithelium. Morphological and physiological studies revealed that the apical plasma membrane of mammalian transitional epithelium of the urinary tract is unique in its structure and exhibits low permeability to water (Hicks 1965,1966; Staehelin et al. 1972; Chang et al. 1994). In fact, AQP3 was not seen in superficial cells, whereas abundant AQP3 was present along plasma membranes of the cells beneath them. These observations indicate that, unlike renal collecting duct cells, it is unlikely that AQP3 in the transitional epithelium serves for the transcellular transfer of water.
On the body surface, AQP3 was present in the stratified epithelium covering the body surface, i.e., the epidermis of the skin. The epidermis, like the transitional epithelium of the urinary tract, serves as a barrier against water loss. The outermost layer of the epidermis is composed of keratinized stratum corneum (Ebling 1992). AQP3 was localized to the basal and intermediate cells in the epidermis, and therefore it is unlikely that AQP3 serves for transepithelial water absorption.
Moreover, we examined AQP3 distribution throughout the digestive tract from the oral cavity to the anal canal. AQP3 was present at the epithelium of the upper part (from the lower lip to the forestomach) and the lower part (from the distal colon to the anal canal). AQP3 was present in the stratified squamous epithelia of the oral cavity, esophagus, and forestomach, whose ability to absorb water from the luminal contents is minimal. Much more AQP3 was present in the large intestine than in the small intestine, whereas 80-90% of total water absorption throughout the digestive tract is carried out in the small intestine (Selub 1995). Judging from the AQP3 distribution and localization, it appears unlikely that AQP3 provides the major route for water absorption from the luminal contents in the digestive tract.
What, therefore, is the role of AQP3 in these epithelia? We suggest that AQP3 in these epithelia could provide a route of water entry to water-deprived cells to maintain intracellular osmolality and cell volume. The epithelia of the body surface and its invaginated continuities appear to exist in an environment of potential water loss for mammals that live on land. The mammalian integument is directly exposed to the air and is subject to possible loss of water through evaporation after birth. AQP3 was present in these covering epithelia. Upper digestive epithelia (from the oral cavity to the forestomach), lower digestive epithelia (from the distal colon to the anal canal), and upper respiratory epithelia, in all of which AQP3 is present, may be exposed to an environment similar to that of the body surface, i.e., potential water loss from the epithelial cells. In addition, AQP3 was present in the epithelia of the urinary tract directly facing the hypertonic urine, where the epithelial cells would encounter possible osmotic water loss due to the difference in tonicity between the luminal urine and the intracellular fluid. The above-mentioned epithelia in an environment of potential water loss have barrier properties on their surfaces, i.e., the stratum corneum in the cornified stratified epithelium and the unique apical membranes of the superficial epithelial cells in the transitional epithelium. AQP3 present in the basal (or abluminal) aspects of these epithelia may provide an additional protective system against water loss. Although AQP3 in the epithelia of the digestive tract may function in absorption of water in case of low osmolality in the lumen, the principal role of AQP3 at their plasma membranes may provide a route of water entry into epithelial cells from the subepithelial side to maintain intracellular osmolality and cell volume if cells undergo water loss.
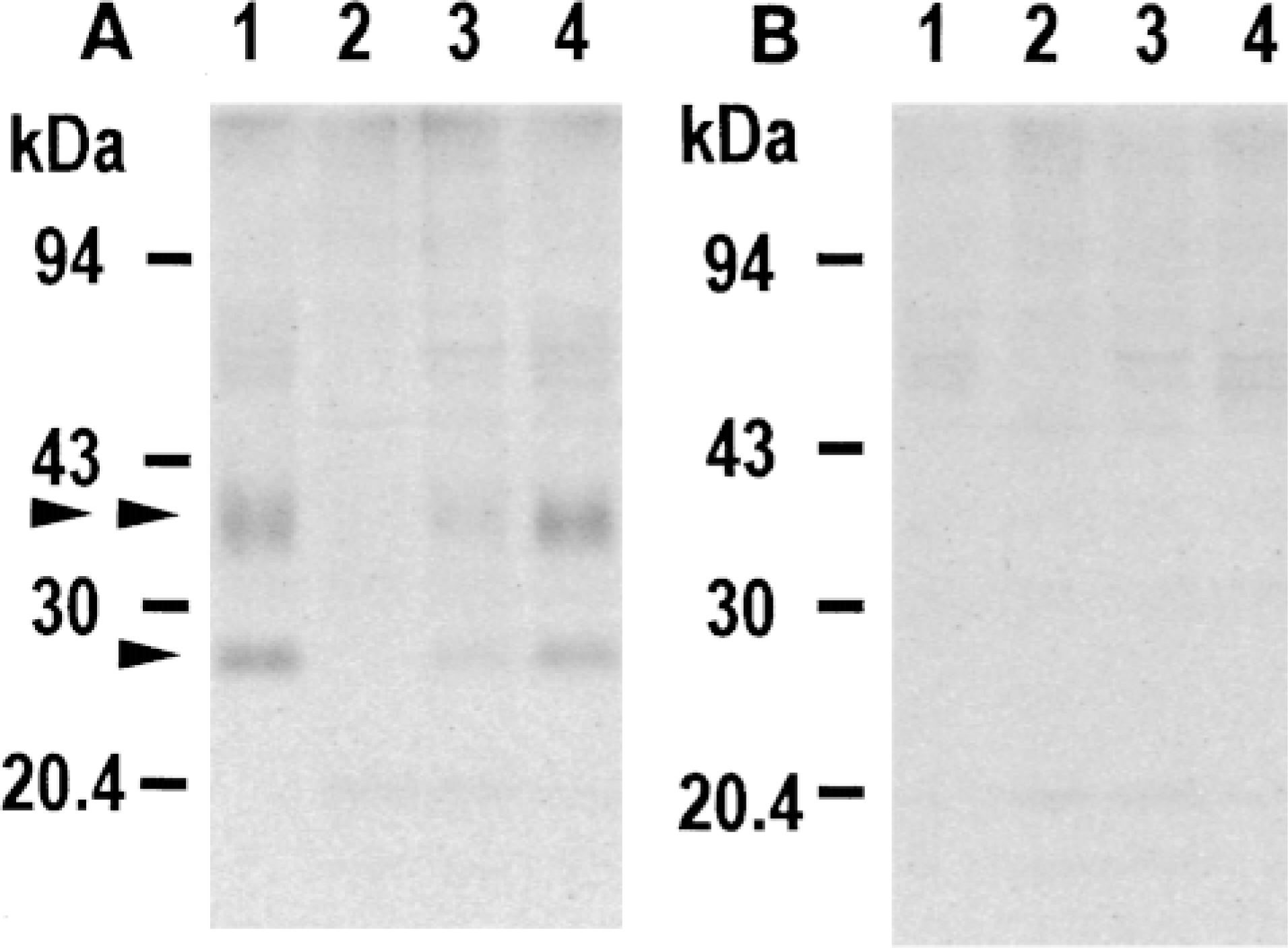
Immunoblotting of fetal, newborn, and adult rat skin with antibody against AQP3. Ten micrograms of protein of tissue homogenates prepared from palmar skin from adult rat (Lane 1) and from total hind leg of fetus at embryonic Day 15 (Lane 2), 18 (Lane 3), and newborn at postnatal Day 4 (Lane 4) were applied to SDS-PAGE (13% acrylamide), electrophoresed, and subjected to immunoblotting with anti-AQP3 antibody (A) or with anti-AQP3 antibody preincubated with COOH terminal peptide of AQP3 protein used as immunogen (B). Nonglycosylated and glycosylated AQP3 are indicated by arrowhead and double arrowhead, respectively.
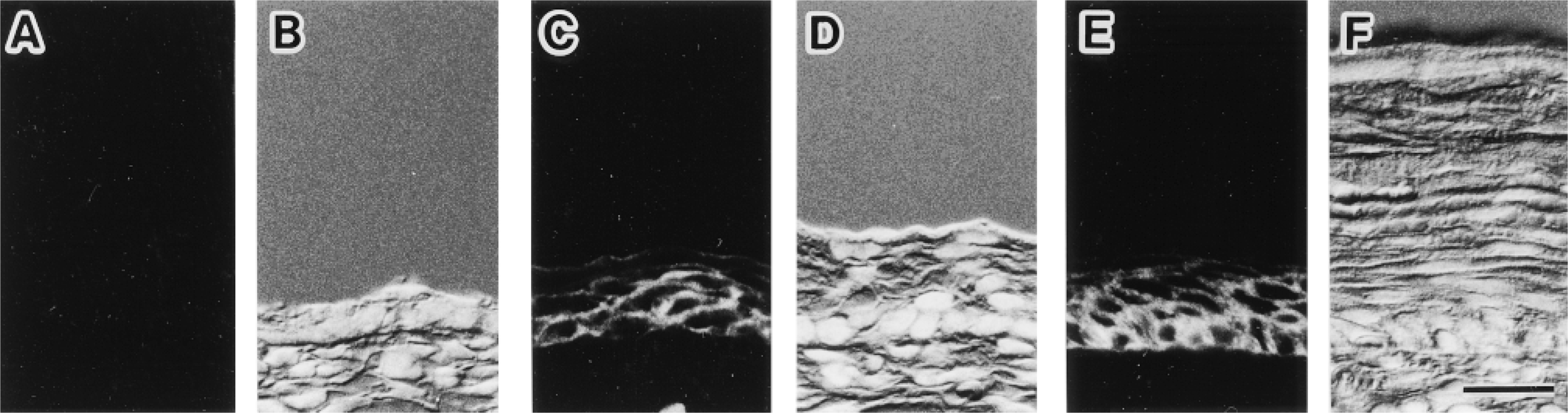
Immunofluorescence localization of AQP3 in the fetal and newborn rat skin. Fluorescence images for AQP3 (A,C,E) and corresponding Nomarski differential interference contrast images counterstained with DAPI for nuclei (B,D,F) are shown. Cryostat sections from hind leg of fetus at embryonic Days 15 (A,B), 18 (C,D), and newborn at postnatal Day 4 (E,F) are shown. (A,B) No label for AQP3 is seen in the epidermis at embryonic Day 15. (C,D) Label for AQP3 is present at epidermis at embryonic Day 18. (E,F) Label for AQP3 is present in epidermal basal and intermediate cell layers at postnatal Day 4. Bar = 20 μm.
In a simple epithelium sealed by tight junctions, AQP3 localized at the basolateral membranes may function directly in the entry of water into the cell. In the stratified epithelia, uppermost epithelial cells may be highest risk for dehydration, whereas AQP3 is localized in the intermediate and basal cells. Because epithelial cells are connected by gap junctions (Bruzzone et al. 1996), water can easily pass through from cell to cell. Therefore, AQP3 in basal cells could participate in the supply of water to the cells located above.
To provide insight into the roles of AQP3, including the above possibility in the epithelial cells, we examined the ontogenic changes of AQP3 expression in rat skin. The fetus has a low risk for osmotic change because it is surrounded by the amniotic fluid. After delivery, the body surface of the neonate is abruptly exposed to the air, and the risk for dehydration through the body surface arises. AQP3 was expressed in the epidermis of the body surfaces on embryonic Day 18 and postnatal Day 4, whereas it was not on embryonic Day 15. Aszterbaum et al. (1992) previously demonstrated that the epidermal barrier to water loss in the rat is rapidly established during embryonic Day 20. The appearance of AQP3 before birth suggests that AQP3 might be among the protective mechanisms that prevent water loss from the body surface in newborn animals.
In summary, we showed the comprehensive and detailed immunolocalization of AQP3 in rat epithelial tissues. Developmentally, expression of AQP3 in the epidermis begins in the late stage of fetal development. These observations suggest that AQP3 in these epithelia may play a role in providing water to water-deprived cells to maintain intracellular osmolality and cell volume. Further examination of the relationship between AQP3 expression and osmolality is needed to establish the functional role of AQP3 in the epithelia.
Footnotes
Acknowledgements
Supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan. T. Matsuzaki is a JSPS Research Fellow.
We thank S. Matsuzaki for assistance.
