Abstract
Netrins are a family of proteins that mediate axonal guidance in the central nervous system (CNS). In addition to the CNS, netrins are involved in cell adhesion, motility, proliferation, differentiation, and survival. Because these processes occur in the placenta, we raised the question of whether netrin-1 is expressed by placental cells during development. In the present study, we analyzed the spatial and temporal distribution of netrin-1 and its two receptors, DCC (deleted in colorectal cancer) and UNC5B (uncoordinated-5 homolog) in human placenta using RT-PCR, Western blotting, and immunohistochemistry analysis. We demonstrated the presence of the proteins and transcripts of netrin-1 and its receptors in placenta and cytotrophoblasts. Furthermore, using immunohistochemistry, we localized endogenous netrin-1 protein staining to villous and extravillous cytotrophoblasts, and secreted netrin-1 outside the syncytiotrophoblasts. The DCC receptor was localized to syncytiotrophoblasts and invasive extravillous cytotrophoblasts during the first trimester and at term. On the other hand, the UNC5B receptor was localized to villous and extravillous cytotrophoblasts proximal to anchoring areas during the first trimester. At term, UNC5B was observed in decidual cells and weakly in extravillous cells. The discrete pattern of netrin-1 and netrin-1 receptor distribution suggested that netrin-1 protein functions might vary with its localization in the placenta and probably with time of gestation.
N
During development, various placental cells, such as the villous and extravillous cells, are subjected to proliferation, differentiation, migration, and survival. These changes are necessary to allow nutritional and gas exchanges between the fetus and the maternal organism to ensure a successful pregnancy and fetal growth. Considering the importance of netrin-1 in the growth control of various non-placental tissues, we raised the question of whether netrin-1 could play any role in human placenta development.
Except for one study showing at term a slight decrease in netrin-1 mRNA and protein in patients with preeclampsia irrespective of the cell types involved (Yang et al. 2006), there are no available data concerning expression and evolution of netrin-1 and its receptors during the course of pregnancy and in particular during first-trimester gestation. The aim of the present study was to characterize netrin-1 and its receptors in human placenta during first-trimester gestation and at term by using RT-PCR, Western blotting, and immunohistochemistry techniques.
Materials and Methods
Sample Collection
All subjects gave written informed consent. Placental villous tissues were collected on an anonymous basis from 16 healthy women with viable singleton ultrasound-dated pregnancies undergoing elective social termination of pregnancy between 6 and 14 postmenstrual weeks of gestation. Four term placentas were obtained at Caesarian section.
For immunohistochemistry, placental tissues were fixed in 4% paraformaldehyde at room temperature for 24 hr. For protein and RNA extraction, placental tissues were immersed in liquid nitrogen and stored at −80C. For cytotrophoblast purification, placentas were immersed in Hank's balanced salt solution (Ca2+-Mg2+-HBSS).
The transient UNC5B-transfected HEK 293 cells were kindly provided by Dr. Patrick Mehlen; the human colon and endometrial biopsies were provided by the surgical staff of Centre Hospitalier Intercommunal, Hôpital de Poissy-St-Germain.
Isolation and Treatment of Cytotrophoblasts
Placental cytotrophoblasts were isolated from first-trimester human placentas (6–14 weeks of gestation, n=16) and cultured using a combination of two previously described techniques (Kliman et al. 1986; Hoffmann et al. 2006). Briefly, placenta (10 g) was thoroughly washed in 50 ml cold sterile Ca2+-Mg2+-HBSS until the supernatant was nearly free of blood. Areas rich in chorionic villi were selected and minced with scalpels. The resulting fragments were subjected to three 30-min sequential digestions in 100 ml HBSS containing 2000 U/ml trypsin (Sigma; St. Louis, MO), 4.2 mM MgSO4, 25 mM HEPES, and 50,000 U/ml type IV DNase I (Sigma). The dispersed placental cells were filtered through 100-μm nylon gauze and loaded onto a discontinuous Percoll gradient (5–70% in 5% steps of 3 ml each) and then centrifuged at 1200 × g for 20 min at room temperature to separate the different cell types. Cytotrophoblast cells that sedimented between the density markers of 1.049 and 1.062 g/ml were collected and washed with DMEM. Cell viability, assessedbytrypanblue exclusion, was above 95% before starting cell culture.
Cytotrophoblasts were seeded at a density of 150,000 cells/cm2 in DMEM, 10% fetal bovine serum, 100 IU penicillin, 5 μg/ml gentamycin, and 10 μg/ml streptomycin. The dispersed cytotrophoblasts were cultured for 24–72 hr at 37C in 5% CO2, 95% air to allow attachment. Purity of the cell preparation was assessed at the end of the experiment by immunocytochemistry staining for cytokeratin 7 as an epithelial cell lineage marker (Frank et al. 2001), using a mouse monoclonal anti-human cytokeratin 7 antibody (Dako Cytomaton; Glostrup, Denmark). After 48 hr of culture, both mononucleated cytotrophoblasts and aggregates of placental cytotrophoblasts forming syncytial clumps were observed; 90% of the cultured cells were cytokeratin 7-positive, indicating the presence of a majority of cytotrophoblasts.
RNA Extraction and Quantification
RNAs were extracted from cytotrophoblasts, human endometrium biopsies were removed at the proliferative phase by using Qiagen RNeasy Mini Kits (Qiagen GmbH; Hilden, Germany) according to the manufacturer's protocol, and RNA quantification was performed by measuring absorbance at 260 nm.
RT-PCR
After RT-PCR using Super Script III Reverse Transcriptase (Invitrogen; Carlsbad, CA) from 100 ng mRNA in 10 μl final, 4 μl of cDNA dilution (1:10) was added to 0.5 μM of each specific primer pair (see Table 1) and 5 μl of PCR mix (Qiagen; Master SYBR Green I). The mixtures were amplified by PCR on a Light Cycler instrument (Roche Diagnostics; Mannheim, Germany) under the following conditions: step 1, 94C for 15 min; and step 2, 35 cycles consisting of 95C for 15 sec, 60C for 20 sec, and 72C for 10 s.
The specificity of the PCR products was verified by checking their respective melting temperature curves, their sizes by electrophoresis on a 2% agarose gel, and their sequence by dideoxysequencing using the Big Dye Terminator Kit (Applera; Courtaboeuf, France). Sequencing products were loaded onto a MegaBACE sequence analyzer (Amersham Pharmacia Biotech; Little Chalfont, UK).
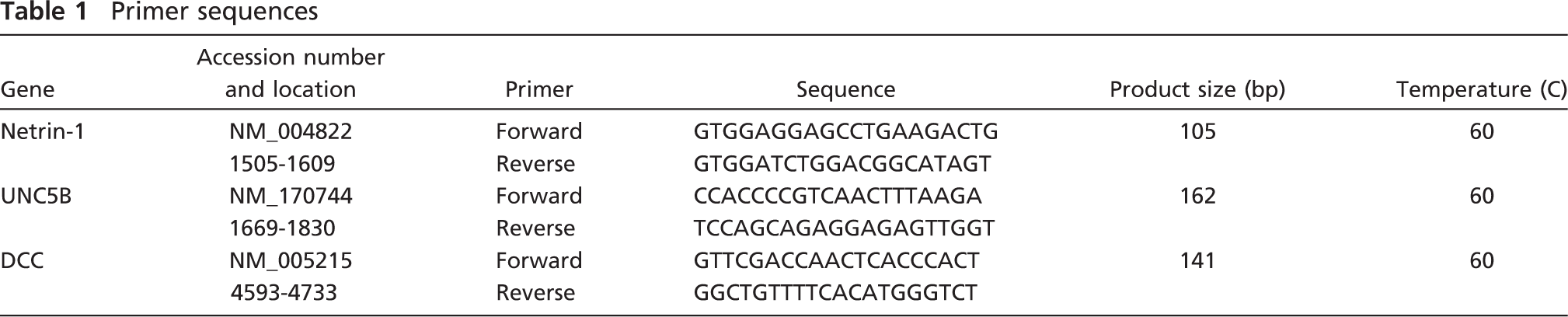
Primer sequences
Western Blotting Analysis
Protein, including netrin-1 receptors membrane and netrin-1 were solubilized as follows: frozen placental samples or cultured cytotrophoblasts were homogenized on ice for 1 min in lysis buffer [50 mM Tris-HCl, pH 7.5, 120 mM NaCl, 1 mM EDTA, 0.1% sodium dodecyl sulfate (SDS), 1% Nonidet 40, 0.5% (w/v) sodium deoxycholate, 1 mM sodium orthovanadate, 30 mM β-glycerophosphate, 12.5 μg/ml leupeptin, 5 μg/ml aprotinin, 10 mM NaF, and 0.1 mg/ml 4-(2-aminoethyl) benzenesulfonyl fluoride hydrochloride]. The homogenates were centrifuged as previously described (Benaitreau et al. 2009). The supernatants (soluble proteins) were collected and diluted in Laemmli buffer (v/v), and the pellets were discarded. Protein concentrations were determined using the Bradford assay (Bradford 1976). In preliminary setup experiments, 2 μg to 100 μg were used before 100 μg was chosen for routine procedures unless stated in the figure legend. Proteins extracted from placenta or trophoblast cells were electrophoretically separated on 0.1% SDS-PAGE (10% polyacrylamide) and electrically transferred onto nitrocellulose membranes. The nitrocellulose membranes were washed with 0.1% TBS-Tween 20 (TBS-T) and incubated for 2 hr in blocking solution (2.5% gelatin in TBS-T). Next, membranes were immunoblotted with goat anti-UNC5B (dilution 1:200) or with mouse anti-DCC (dilution 1:100) overnight or with rabbit anti-netrin-1 (H104, dilution 1:200; Santa Cruz Biotechnology, Santa Cruz, CA). Detailed antibody information is given in Table 2. Blots were then rinsed three times with TBS-T and incubated with the appropriate anti-IgG (horseradish peroxidase-linked anti-rabbit IgG for netrin-1 diluted 1:5000, horseradish peroxidase-linked anti-goat IgG for anti-UNC5B diluted 1:10,000, or horseradish peroxidase-linked anti-mouse IgG for anti-DCC diluted 1:2500) in TBS for 1 hr. Finally, blots were washed three times with TBS-T, and the antibody- antigen complexes were detected using the enhanced chemiluminescence detection system (Amersham Pharmacia Biotech). Membranes were then exposed to radiographic films (Kodak Scientific Imaging Products; Rochester, NY).
Antibodies used in immunohistochemistry and Western blot experiments
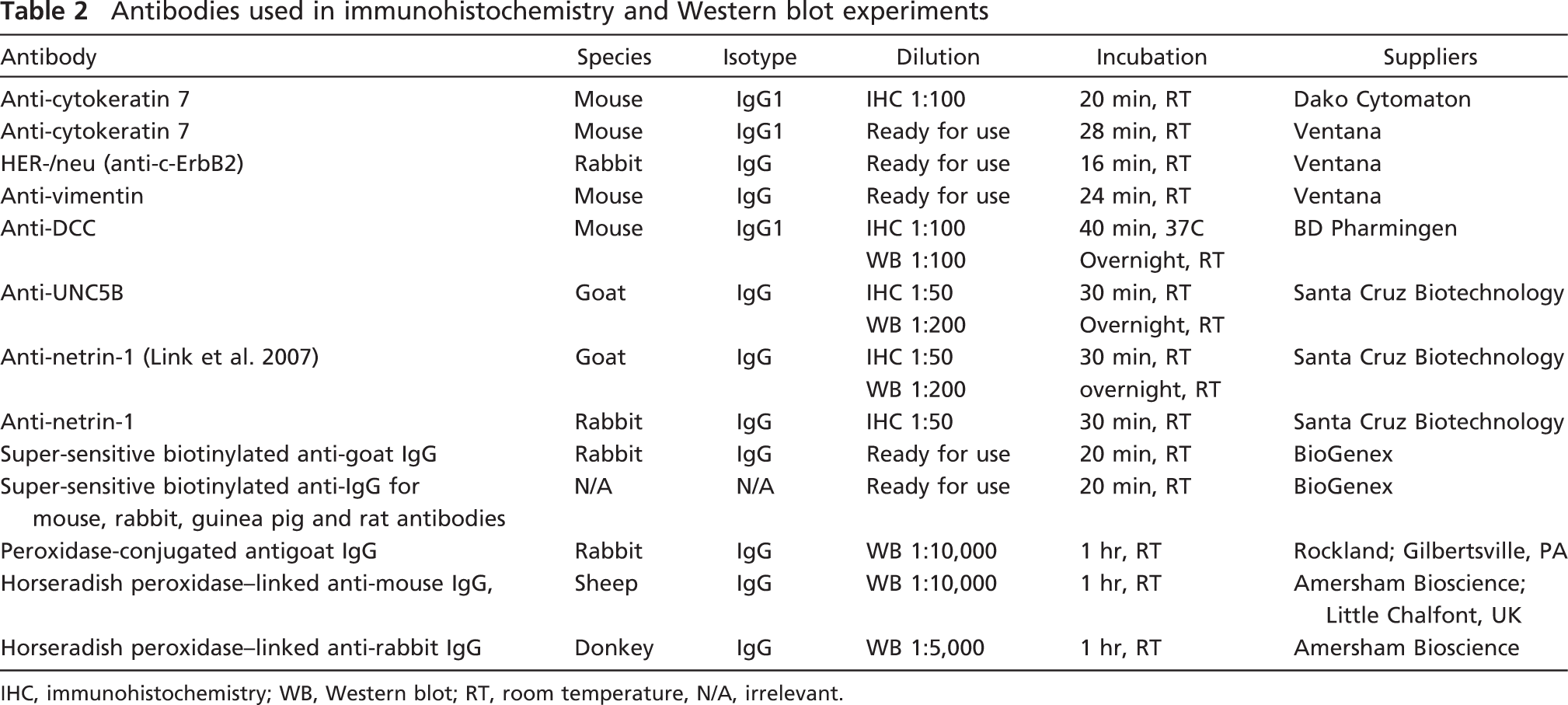
IHC, immunohistochemistry; WB, Western blot; RT, room temperature, N/A, irrelevant.
Immunohistochemistry
Placental tissues collected at 4–12 weeks of gestation and term placenta were fixed for 24 hr at room temperature in 4% (v/v) paraformaldehyde and embedded in paraffin, and serial histological sections (measuring 4.0 μm in thickness) were deparaffinized in xylene, hydrated in graded alcohols, and placed in buffered saline (Ventana; Tucson, AZ). Subsequently, antigen retrieval was achieved by heating the slides in a microwave oven in 10 mM citrate buffer. The slides were rinsed in the buffer supplied by the manufacturer (Ventana). After antigen retrieval, sections were incubated with an aqueous solution of 3% hydrogen peroxide followed by incubation with 5% non-fat milk, which served as a blocking agent for nonspecific binding, for 20 min at 37C. After rinsing the slides in buffer for 5 min, slides were incubated with the commercial primary antibodies (see Table 2) polyclonal goat anti-N-terminal netrin-1 (dilution 1:50), rabbit antiC-terminal netrin-1 (dilution 1:50), goat anti-UNC5B (dilution 1:50) for 30 min, or monoclonal mouse anti-human DCC antibody (BD Pharmingen; San Diego, CA, diluted 1:100) for 40 min at 37C. Mouse monoclonal anti-cytokeratin 7 (Ventana) was used for 28 min at room temperature for villous and extravillous cytotrophoblast characterization (Frank et al. 2001). For extravillous cell characterization, rabbit monoclonal anti-HER-2/neu (anti-c-ErbB2; Ventana) was used for 16 min. For mesenchymal cell characterization, anti-vimentin (Ventana) was used for 24 min. After washing, slides were incubated with secondary antibodies. Biotinyated anti-IgG specific for either mouse, rabbit, or goat was used for 20 min at room temperature (see Table 2). Then, peroxidase-conjugated streptavidin was used for 30 min at room temperature (Super Sensitive Link-Label IHC Detection System; BioGenex, San Ramon, CA). Slides were rinsed in buffer supplied by the manufacturer (Ventana), and the immunoreactive proteins were visualized after the addition of 3–3' diaminobenzidine (BioGenex) for 3 min. Slides were counterstained with hematoxylin for 3 min. After a final rinsing step in 3% ammonia and water, slides were dehydrated in graded alcohols, then in xylene, and mounted with non-aqueous mounting medium and coverslips. As negative controls, sections were processed in the absence of primary antibody.
For first-trimester samples, each labeling experiment was performed at least six times for each antibody. At least three 4–9 weeks gestation samples and three 10–14 weeks gestation samples were used.
Results
RT-PCR
RT-PCR showed that netrin-1, UNC5B, and DCC mRNAs were present in purified cytotrophoblasts (Figure 1). Reverse transcription was performed as described in the Materials and Methods section. Four ml 1:10 cDNA obtained from 100 ng total RNA was used for amplification with the primers described in Table 1. Reverse and forward primers were chosen in different exons to ensure that no genomic DNA was amplified in the PCR reaction. PCR was performed for 35 cycles. PCR products were loaded onto a 2% agarose gel, and bands consistent with the expected size (141, 105, and 162 bp for DCC, netrin-1, and UNC5B, respectively, see Figure 1) were visualized by ethidium bromide staining. Positive controls (4 μl 1:10 cDNA from normal endometrium biopsy removed at the proliferative phase) and negative controls (no cDNA) were also included. In addition to size control, fusion curves and sequence analysis were also used for specificity control (data not shown).
Western Blotting
UNC5B protein expression was analyzed by Western blot on solubilized protein extracts from placental tissue, from cultured cytotrophoblasts, and from UNC5B-transfected HEK 293 cells used as positive control. A band of ~90 kDa (expected size 103 kDa) that was completely abolished in the presence of an excess of the antigenic peptide (UNC5H2 N19; Santa Cruz Biotechnology) was found in placental tissue as well as in cultured cytotrophoblasts and UNC5B-transfected HEK 293 cells (Figure 2).
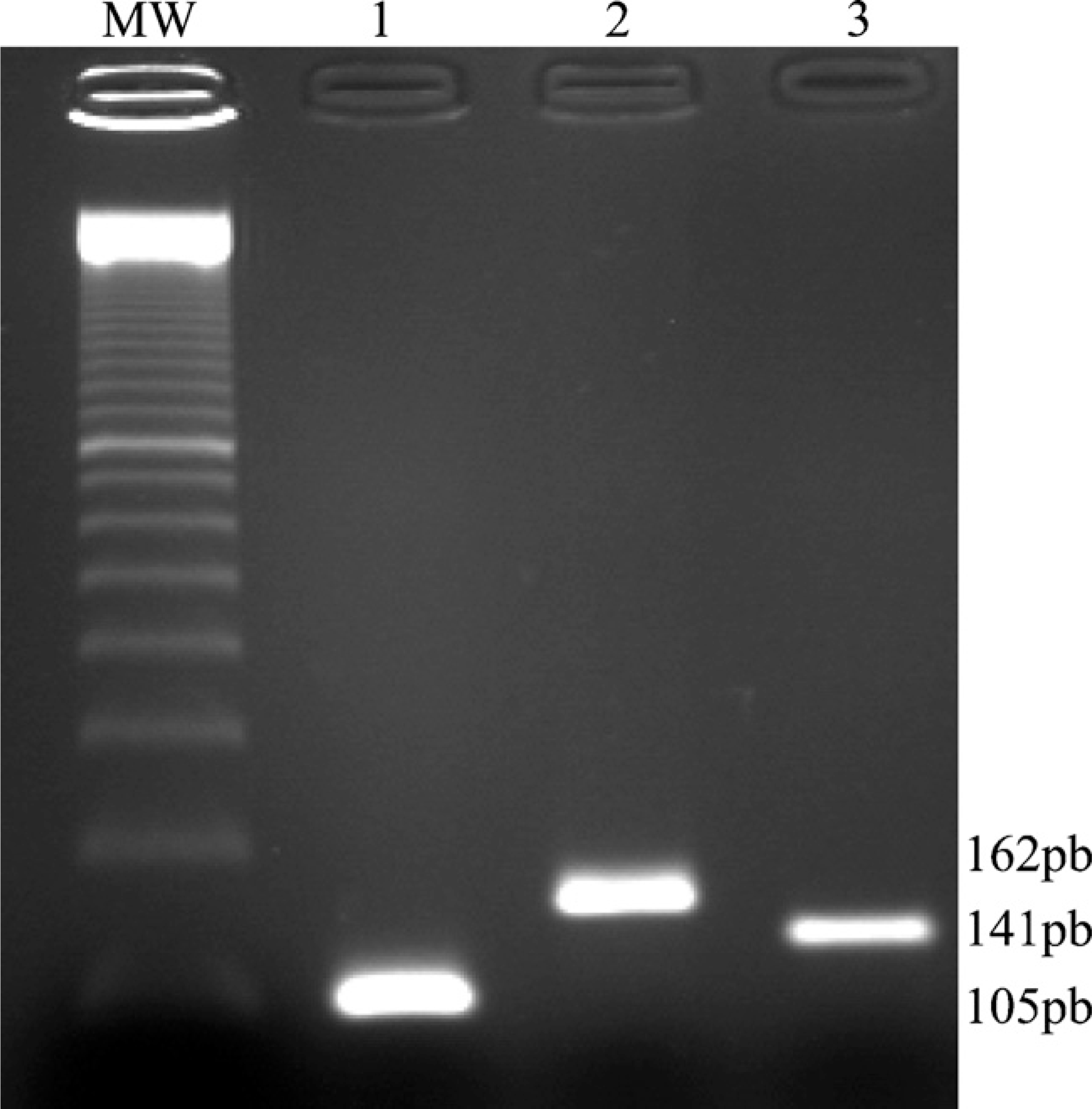
Amplification of netrin-1 and its receptor cDNAs. Gel electrophoresis and ethidium bromide staining of netrin-1 (Lane 1), UNC5B (Lane 2), and DCC (Lane 3) RT-PCR products from purified cytotrophoblast cells. MW, molecular weight marker.
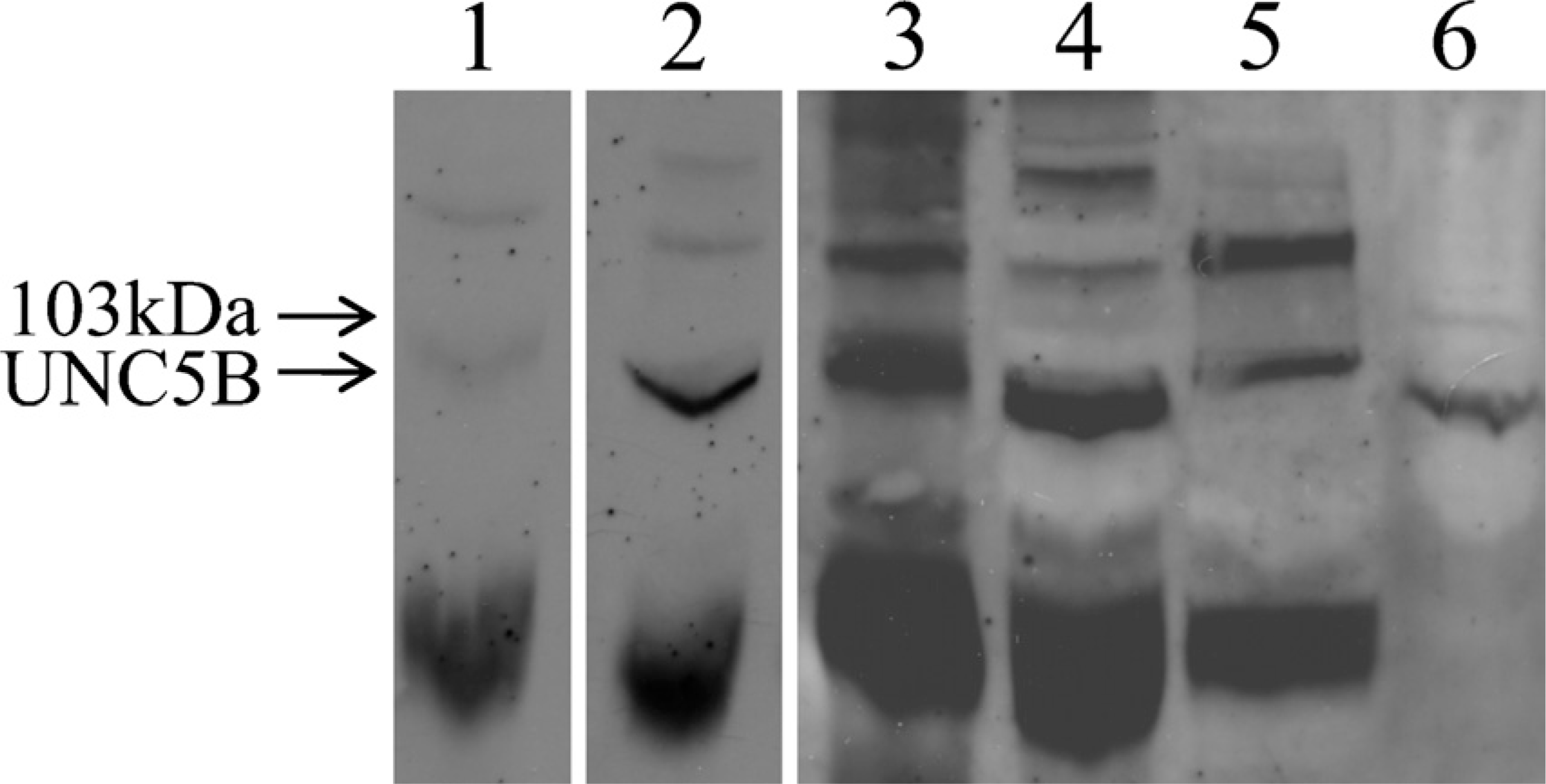
Western blot analysis of the netrin-1 receptor UNC5B. Western blot analysis of the netrin-1 receptor UNC5B was performed with 100 μg protein extracts. Lane 1, trophoblast tissue (10 weeks gestation) plus the UNC5B antigenic peptide UNC5H2; Lane 2, trophoblast tissue (10 weeks gestation); Lane 3, at term; Lane 4, trophoblast tissue (10 weeks gestation); Lane 5, 48-hr-cultured cytotrophoblasts; and Lane 6, UNC5B-transfected 293 T cells. The Western blot is representative of six separate experiments, each performed with samples prepared in different experiments and used only once.
DCC protein expression analysis in protein extracts from placental tissue, cultured cytotrophoblasts, and colon used as positive control (Shibata et al. 1996; Shin et al. 2007) showed a band of ~200 kDa (Figure 3) and a strong band at 120 kDa. Previous studies have also observed two bands at ~200 kDa and 120 kDa (Reale et al. 1994; Ren et al. 2008). The 120-kDa band seems to represent a proteolytic fragment of the full-length DCC molecule (Figure 3). The 200-kDa band signal increased with the amount of protein loaded in the lane (Figure 3, Lanes 5 and 6).
Localization of Netrin-1, UNC5B, and DCC in Placenta by Immunohistochemistry
To determine the cell types that express netrin-1, UNC5B, and DCC in human placenta, their immunohistochemical expression patterns were investigated in serial histological sections of first-trimester and term placentas. Immunoreactivity to cytokeratin 7 and c-ErbB2 antibodies was used to identify villous cytotrophoblasts, syncytiotrophoblasts, and extravillous cytotrophoblasts. Cytokeratin 7 staining identifies all villous and extravillous cytotrophoblasts (Frank et al. 2000), whereas the proto-oncogene product c-ErbB2 is present in the extravillous cells (Jokhi et al. 1994). Anti-vimentin antibodies were used to detect stromal cells (Kelley et al. 1990).
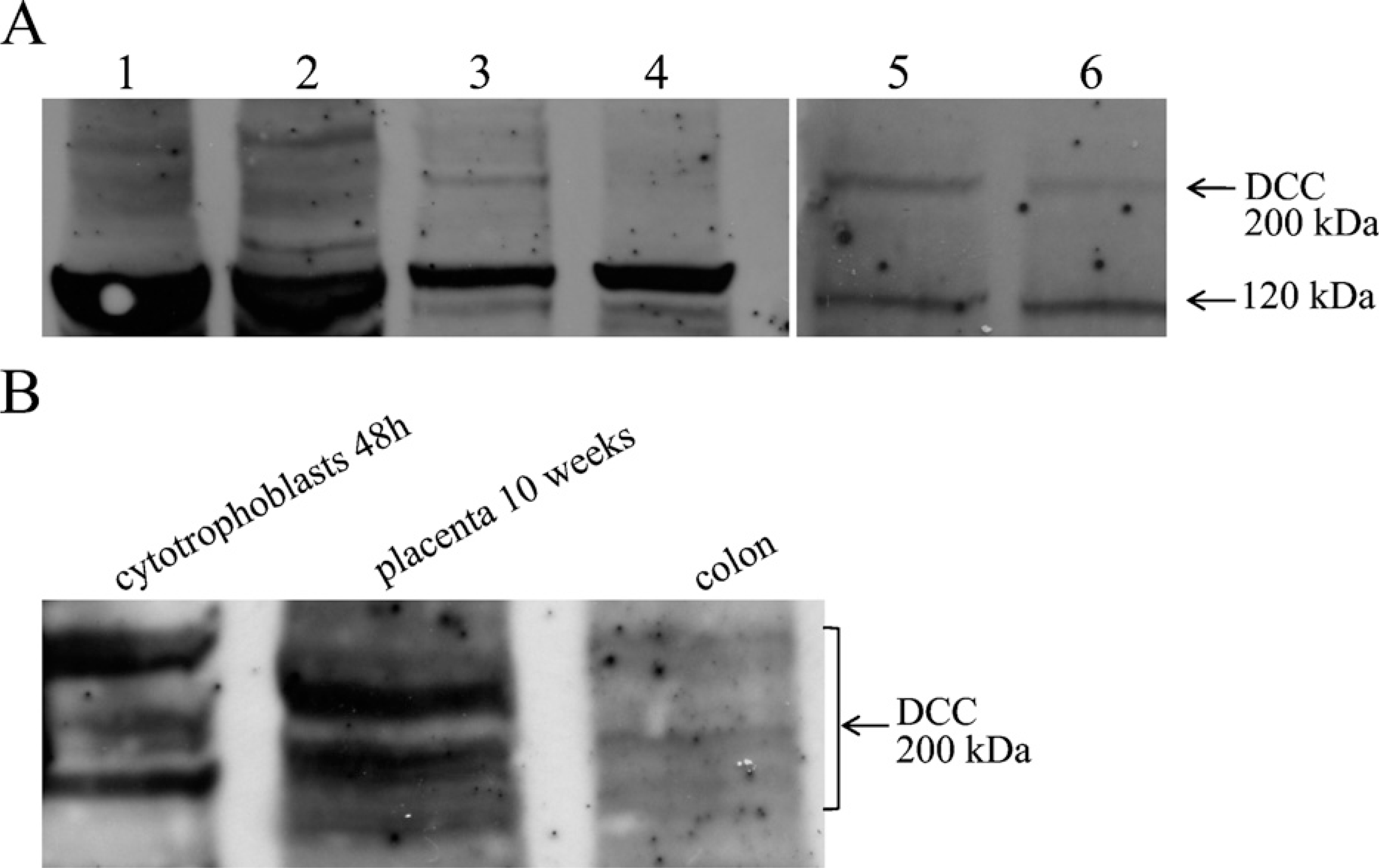
Western blot analysis of the netrin-1 receptor DCC. Western blot analysis of DCC was performed with 100 μg protein extracts, except for Lanes 5 and 6, which were performed with 70 μg and 35 mg, respectively. The 200-kDa band is the DCC receptor; the strong band at 120 kDa is a proteolytic fragment of the receptor. (
Immunohistochemistry for Netrin-1 in First-trimester and Term Placental Tissues. In first-trimester placenta, netrin-1 immunostaining with a goat antibody raised against the N-terminal portion of netrin-1 showed intense signal in the cytoplasm of villous and extravillous cytotrophoblasts, whereas syncytiotrophoblasts were stained at the cell surface. Furthermore, stromal cells were also stained by netrin-1 antibodies (Figure 4A).
In placenta at term, netrin-1 immunoreactivity was localized at the apical surface of villous syncytiotrophoblasts and in the decidual stromal cells at the basal plate (Figures 5G and 5I). In contrast, extravillous cytotrophoblast cells (immunoreactive for cytokeratin 7 and anti-c-ErbB2, Figures 5A and 5B) located at the basal plate were not labeled by netrin-1 antibody (Figure 5G). The same labeling pattern was found in first-trimester and term placental tissues with another anti-netrin-1 antibody raised against another epitope (rabbit antibody raised against the C-terminal portion of netrin-1, data not shown) indicating that nonspecific binding was unlikely.
Immunohistochemistry for UNC5B in First-trimester and Term Placental Tissues. Human first-trimester placenta showed UNC5B immunoreactivity in villous cytotrophoblasts and in extravillous cytotrophoblasts located proximal to anchoring areas, whereas no staining was detected in distal extravillous cytotrophoblasts and syncytiotrophoblasts (Figure 4C). In decidua of placenta at term, UNC5B immunostaining was observed in decidual cells and weakly in extra-villous cells (Figure 5E).
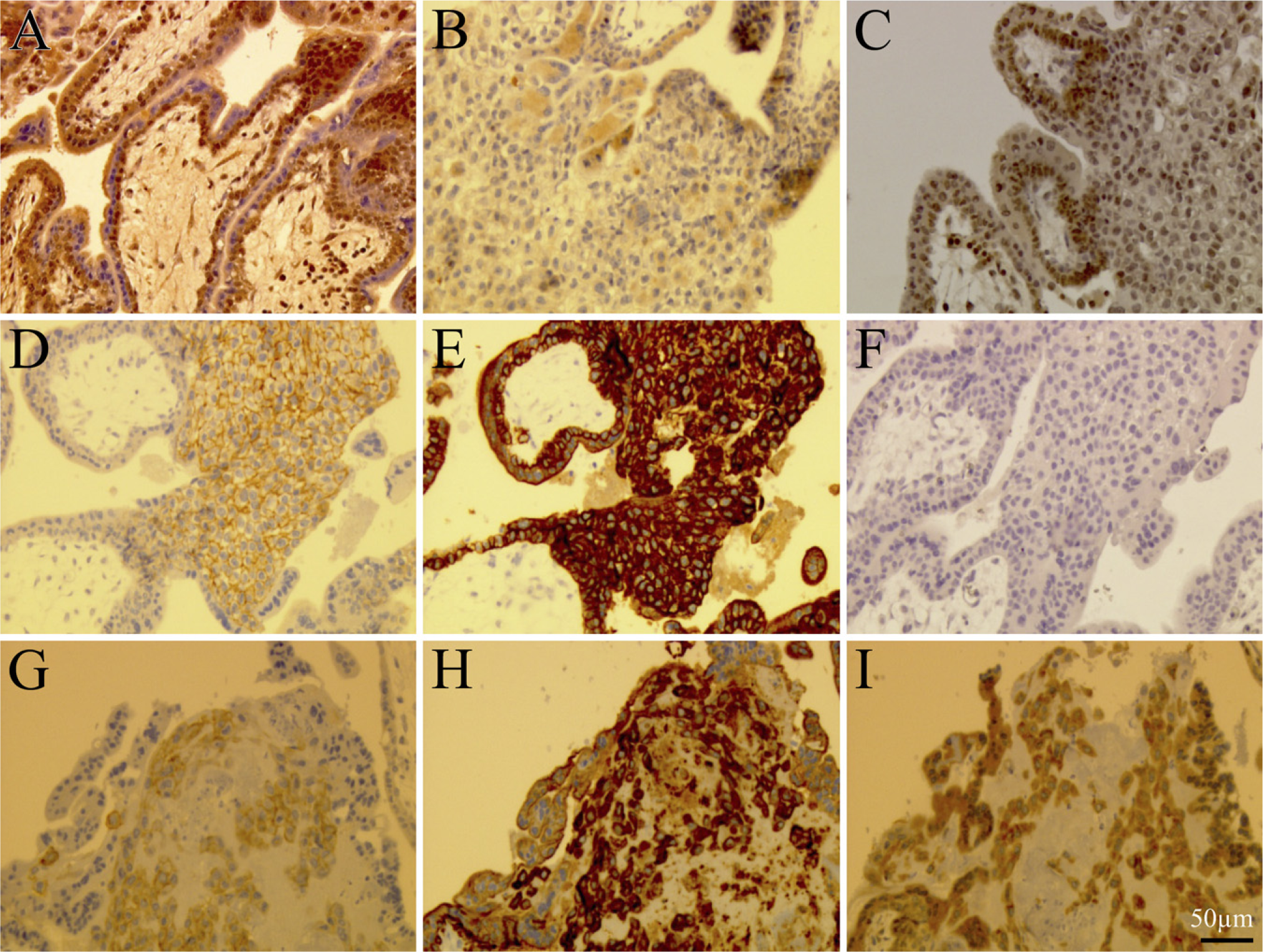
Immunohistology of netrin-1, UNC5B, and DCC in first-trimester human placental tissue. (
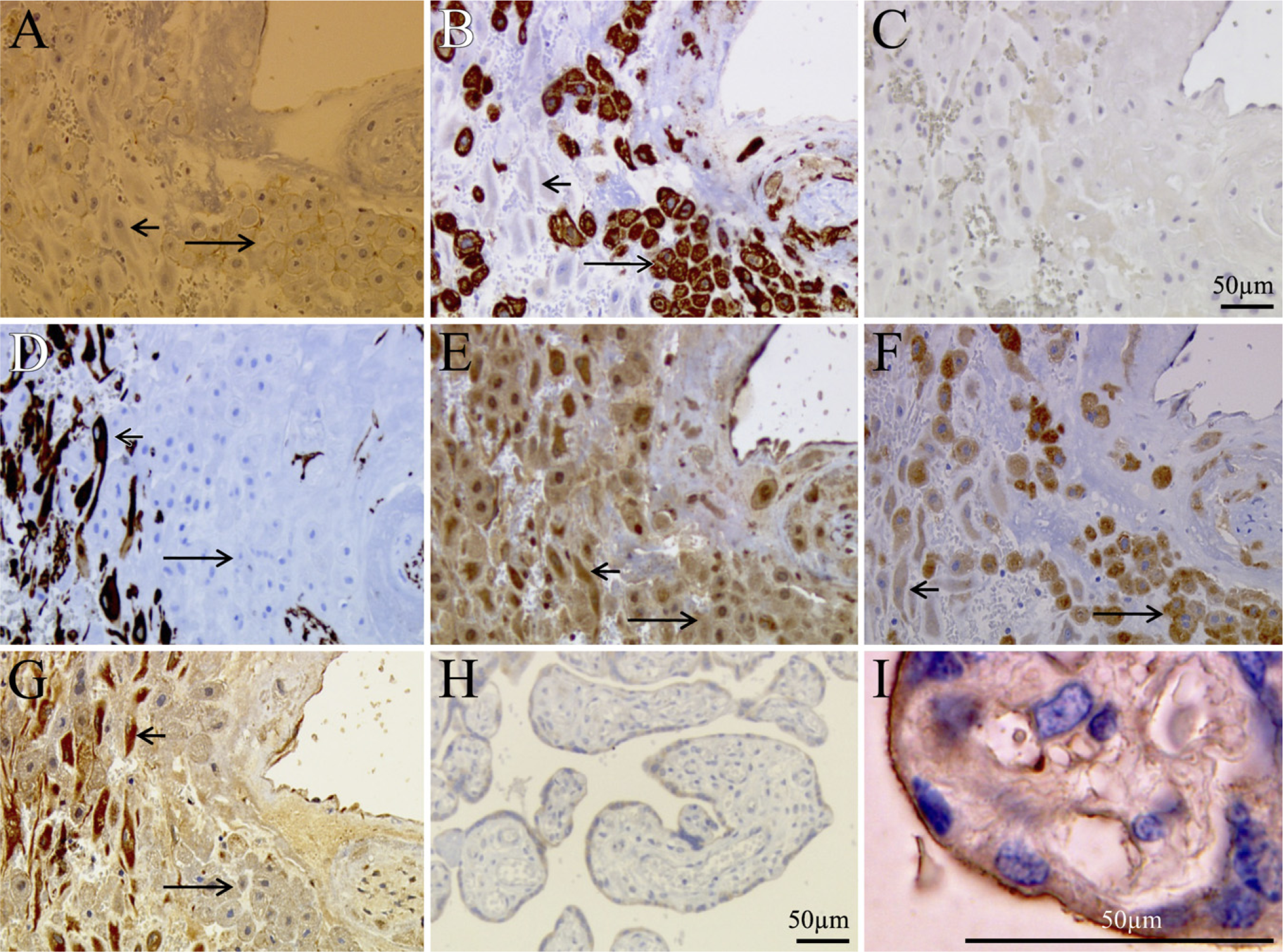
Immunohistochemical localization of netrin-1, UNC5B, and DCC in term human placental tissue. (
Immunohistochemistry for DCC in First-trimester and Term Placental Tissues. During the first trimester and at term, DCC was expressed by syncytiotrophoblasts (Figure 4B) and extravillous cytotrophoblasts (immunoreactive to both cytokeratin 7 and c-ErbB2, Figures 4H and 4G) located distally from the villous stroma (Figure 4I).
During the first trimester, villous cytotrophoblast and extravillous trophoblast cells located proximally to the villous stroma were not labeled for DCC, whereas extravillous cytotrophoblasts located distally from the villous stroma were stained weakly or negative for DCC (Figure 4B).
DCC immunostaining was found in the syncytiotrophoblasts and invasive extravillous cytotrophoblasts in term placenta (Figures 5F and 5H). Weak staining was also found in decidual cells. Table 3 summarizes the main data.
Discussion
Except for one study showing at term a slight decrease in netrin-1 in placenta from patients with preeclampsia (Yang et al. 2006), irrespective of the cell type involved, there are to date no reports describing expression of netrin-1 and its receptors DCC and UNC5B in human placenta. This is all the more surprising considering the number of reports ascribing to netrin-1 and its receptors key roles in the control of cell proliferation, differentiation, and guidance, and tissue development (Cirulli and Yebra 2007). Because in the above-mentioned study on preeclampsia (Yang et al. 2006), the cellular origin of netrin-1 was not described, we decided to investigate the distribution of netrin-1 and its receptors in human placenta.
Distribution of netrin-1 and its receptors in first-trimester and term placentas

Cytokeratin 7, c-ErbB2, and vimentin staining were used to ascertain the cell type.
In first-trimester placentas and at term, netrin-1 was expressed in the cytoplasm of villous cytotrophoblasts and was found at the surface of syncytiotrophoblasts. The present data showing absence of intracellular staining and strong surface staining suggest that upon differentiation, syncytiotrophoblasts lose netrin-1 synthesis capabilities. Other striking localizations in placenta include labeling of extravillous cytotrophoblasts located at the anchoring villi of first-trimester mesenchymal cells, and of term placenta decidual cells as well as term placenta endovascular cytotrophoblasts. Concerning netrin-1 receptors, UNC5B immunoreactivity was observed in villous cytotrophoblasts, in extravillous cytotrophoblasts located proximally to anchoring villi, in stromal decidual cells, and in endothelial cells. Extra-villous cytotrophoblast cells located at the basal plate and UNC5B-negative syncytiotrophoblasts were found to be stained by DCC receptor antibody. Western blots and RT-PCR results were in accordance with immunocytochemistry findings. UNC5B and DCC proteins were evidenced by Western blots in cultured cytotrophoblasts and placental extracts; UNC5B and DCC mRNAs were found in isolated trophoblast cells. The presence of DCC as shown by Western blots and RT-PCR in cultured cytotrophoblasts probably reflects the progressive syncytialization under our culture experimental conditions.
The netrin-1 pathways have potential roles in cell polarity, adhesion, apoptosis, migration, morphogenesis, and differentiation in various cell types. Recent studies have reported netrin-1 mRNA expression in epithelial cells during lung branching morphogenesis (Dalvin et al. 2003; Liu et al. 2004) and in pancreatic epithelium development (Yebra et al. 2003). Expression studies have reported that in addition to the nervous system, DCC is also expressed in the basal cell layer of many epithelia, such as gut, skin, lung, and bladder. cDCC (chicken homolog to a gene deleted in colorectal carcinoma) was shown to be an epithelial adhesion molecule expressed in the basal cells and involved in epithelial mesenchymal interaction (Chuong et al. 1994).
The present findings show that upon differentiation, syncytiotrophoblasts acquire DCC receptor synthesis capabilities. The presence of DCC in syncytiotrophoblasts, together with the strong netrin-1 labeling in villous cytotrophoblasts and at the surface of syncytiotrophoblasts, suggests that netrin-1 binds to syncytiotrophoblast DCC as a paracrine signal. The location of DCC at the apical surface of syncytiotrophoblasts suggests that morphogenesis and/or differentiation are linked to DCC appearance on the syncytiotrophoblast layer. In cytotrophoblast cells, expression of both netrin-1 and UNC5B suggests that autocrine mechanisms are involved in these cells. Autocrine and paracrine mechanisms have been demonstrated in colon cancer cells as well (Rodrigues et al. 2007).
The presence of DCC in distal invasive cytotrophoblasts and the strong netrin-1 labeling in decidual cells also suggest that the netrin-1-DCC pathway might play a role in the control of extravillous invasion or migration. Decidualizing stromal cells acquire the unique ability to regulate trophoblast invasion (Gellersen et al. 2007), and the present data suggest that the netrin-1-DCC pathway might be involved. It is now established that netrin-1 plays two opposite functions in the developing CNS. As a matter of fact, netrin-1 has been shown to either attract some axons through its DCC receptor or to mediate repulsion through both the DCC and UNC5B receptors (Kennedy et al. 1994; Serafini et al. 1994; Colamarino and Tessier-Lavigne 1995; Hong et al. 1999). In neural tissues, netrin-1 elicits migration of luteinizing hormone-releasing hormone neurons to the basal forebrain (Schwarting et al. 2001,2004). From these observations and the present data, it appears highly likely that the migration and/or invasion of DCC-positive extravillous cytotrophoblasts to the decidua are controlled by the netrin-1-expressing decidual cells. A recent study reported an increase in cell-cell adhesion with a simultaneous decrease in cell-matrix interaction by DCC when transfected in the intestinal HT-29 cell line (Martin et al. 2006), which is consistent with the blocking action of DCC on the latest steps of colorectal cancer progression associated with the metastatic phenotype (Kinzler and Vogelstein 1996; Chung 2000). Other reports investigating the angiogenesis process have shown that netrin-1, via the UNC5B receptor, functions as a repellant in the guidance events controlling vascular morphogenesis and remodeling (Lu et al. 2004; Eichmann et al. 2005). The presence in vascular structures of cells positive for DCC, cytokeratin 7, and c-ErbB2 shows that the cells expressing DCC are endovascular cytotrophoblasts. Netrin-1 was also present. Whether the presence of DCC and netrin-1 relates to invasion of uterine spiral arteries or to vascular remodeling in placenta remains elusive.
In conclusion, the present study shows in placenta a discrete pattern of distribution of netrin-1 and its two receptors, UNC5B and DCC. Villous cytotrophoblasts synthesize netrin-1 and the UNC5B receptor, suggesting an autocrine mechanism in these cells. Syncytiotrophoblasts express the DCC receptor, but have only extracellular netrin-1 labeling, suggesting a paracrine mechanism. The association of DCC expression with differentiation and/or with the development of particular cell types during organogenesis argues in favor of the involvement of DCC expression and DCC-mediated signaling in placental evolution throughout pregnancy. Further investigations are currently in progress to establish firmly the respective roles (and associated molecular mechanisms) not only of DCC but also of UNC5B and their ligands in normal placenta.
Footnotes
Acknowledgements
This work was supported by the Ministère de la Recherche, Université de Versailles Saint Quentin, Adenosine and by Grant BQR 2009 from Université de Versailles Saint Quentin.
We thank Dr. Patrick Mehlen for providing us UNC5B-transfected HEK 293cells.
