Abstract
Netrin-1 is a potent axonal and neuronal guidance cue in the developing nervous system. Netrin-1 functions are mediated by its receptors, such as deleted in colorectal cancer (DCC) present on axons and neurons. Localization of DCC and Netrin-1 on various types of enteric neurons and their role in the mature enteric nervous system is unknown. The results of our study revealed that almost all enteric neurons and processes express DCC and Netrin-1 in the adult mice. Netrin-1-like-immunoreactivity (IR) was detected in the cytoplasm of neurons with some showing strong or weak staining. The majority of Netrin-1-like-immunoreactive enteric neurons were choline acetyltransferase (ChAT)-positive. However, ~19% of neurons were strongly Netrin-1-like-positive but ChAT-negative while ~8% of neurons were Netrin-1-like-negative but strongly ChAT-positive. In contrast, almost all nitric oxide synthase (nNOS)-positive enteric neurons displayed strong Netrin-1-like-IR. This differential intensity of Netrin-1 expression in the myenteric neurons might determine major neuronal subtypes regulating intestinal motility, ChAT-IR excitatory, and nNOS-IR inhibitory muscle motor and interneurons. This is the first study demonstrating the localization of DCC and Netrin-1 in the colonic myenteric plexus of the adult mice and their expression level determining two major neuronal subtypes regulating intestinal motility.
Keywords
Introduction
Netrin-1 is a laminin-related protein that was initially discovered by examining diffusible cues secreted by the floor plate cells during spinal cord development. 1 Since then it has been known as a potent axonal and neuronal guidance cue in the developing central nervous system (CNS). 2
The hallmark of Netrin-1 function is that it serves as either a chemoattractant or chemorepellent for different types of axons and neurons.3,4 These opposing roles of Netrin-1 are mediated by a particular type of Netrin-1 receptors, such as deleted in colorectal cancer (DCC) and UNC5 homologues (UNC5H), present on axons and neurons. 5 For example, DCC-expressing axons elicit chemoattractive activity toward Netrin-1 molecules. On the contrary, UNC5H expressing axons mediate chemorepulsive activity in the presence of Netrin-1. This dynamic expression of Netrin-1 receptors on axons and neurons is directed to find their appropriate targets or prevented from aberrantly growing under Netrin-1 guidance. 6
In addition to the expression of Netrin-1 and its receptors and their bifunctional roles in the developing CNS, the expression of DCC in the developing enteric nervous system (ENS) has been noted since early 2000. 7 Seaman et al. have demonstrated that DCC expression is present in the peripheral nervous system (PNS) of the developing mouse, in particular, at the early phase of development of the ENS. 7 This finding highlighted that DCC signaling is not limited to the CNS.
Subsequent studies have described that vagal neural crest-derived precursors, which colonize the gut and form the ENS, were found to express DCC. 8 During the colonization, precursors initially establish the myenteric plexus and then the subset of those cells undergo a perpendicular secondary migration toward mucosa to form the submucosal plexus and pancreatic ganglia. The migration of this subset was guided by Netrin-1 that originated from the mucosal layer of the gut and pancreas. 8 In addition to vagal neural crest-derived precursors, vagal sensory axons were found to upregulate the expression of DCC at a certain developmental period, so that the sensory afferent innervation to the fetal gut could be mediated by Netrin-1 guidance. 9 It was further demonstrated that the sensory afferent innervations to the gut were only possible in the ganglionic regions, whereas no progressive innervations were made when the fibers reached the aganglionic regions.
Importantly, in vitro studies have revealed that the enteric neural crest-derived cells (ENCC) do not express Netrin-1, but they synthesize Netrin-1 once they give rise to enteric neurons. 10 Therefore, DCC-expressing vagal sensory afferent fibers were able to connect with the intrinsic enteric neurons expressing Netrin-1. 10
The intrinsic nerve circuits of the ENS contain 14 to 20 types of enteric neurons depending on species and the region of the gut. 11 A vast number of neurotransmitters and neuropeptides have been identified in enteric neurons at different stages of the development. 12 The neurochemical coding of the enteric neurons has revealed a useful distinction between the enteric neurons in relation to their functional types. 11 As consequence, diverse subtypes of enteric neurons have been generated based on neurochemical phenotypes.
Certain neurochemical codes have been highly conserved between species. The conserved chemical codes are mostly for excitatory and inhibitory muscle motor neurons that are essential for gut motility. These include choline acetyltransferase (ChAT, an enzyme synthesizing acetylcholine) for excitatory muscle motor neurons and interneurons, 13 and neuronal nitric oxide synthase (nNOS, an enzyme synthesizing nitric oxide) for inhibitory muscle motor neurons and interneurons. 14
It has been indicated that DCC/Netrin-1 signaling plays a pivotal role in establishing the ENS during development, and subsequent studies have indicated that Netrin-1 expression is present in the neural unit of the adult gut. 15 However, the localized expression of DCC and Netrin-1 and the role of Netrin-1 in the mature ENS have not been fully unraveled.
This study aimed to determine the localization of DCC and Netrin-1 expression in the various types of colonic myenteric neurons of the healthy adult mice.
Materials and Methods
Animals
Male Balb/c mice aged 6 to 10 weeks (18–25 g) were purchased from the Animal Resources Centre (WA, Australia). Mice were housed in a controlled environment where autoclaved cages, bedding, food, and water were provided. They were maintained on a daily 12-hr dark and light cycle at 22C. Mice were acclimatized for a minimum of 3 days before commencing any experiments. All surgical and experimental procedures were conducted in accordance with the guidelines approved by the Victoria University Animal Experimentation Ethics Committee (AEETH21/11).
Wholemount Immunofluorescence
The distal colon segments were collected from mice. Samples were placed in a phosphate buffered saline (PBS) solution (pH 7.2) containing an L-type calcium channel blocker, nicardipine (3 μM), and supplied with carbogen for 20 min to hinder smooth muscle contraction. Samples were cut open along the mesenteric border, maximally stretched, and pinned into a silicon coated dish after clearing the bowel contents. Tissues were fixed in a Zamboni’s fixative (2% formaldehyde and 0.2% picric acid) solution at 4C overnight. Tissues were then washed for 3 × 10 min in dimethyl sulfoxide (Sigma-Aldrich, Australia) followed by 3 × 10 min in PBS to remove fixative. Subsequently, tissues were dissected to expose myenteric plexus and they were incubated with 10% donkey serum (Merck Millipore, Burlington, MA) diluted in 0.1% PBS-tween 20 for 1 hr at room temperature. The myenteric plexus in wholemount preparations were incubated overnight at 4C in the primary antibodies diluents (listed in Table 1). When double- or triple-labeling was conducted in the wholemount preparations, the primary antibodies that were raised in different species were used. Following 3 × 10 min washes in a 0.1% PBS-tween 20 solution, the wholemount preparations were incubated for 1 hr at room temperature in the species-specific secondary antibodies labeled with different fluorophores (listed in Table 2). The wholemount preparations were washed 3 × 10 min in PBS-tween 20 (0.1%). Before the final washing step, the fluorescent nucleic acid stain 4′-6-dimamidino-2-phenylinodole (DAPI, 14 nM, D1306; ThermoFisher, Australia) was introduced to the wholemount preparation for 1 min, and they were mounted onto glass slides with fluorescent mounting medium (DAKO).
List of Primary Antibodies Used in Immunofluorescence Assays.
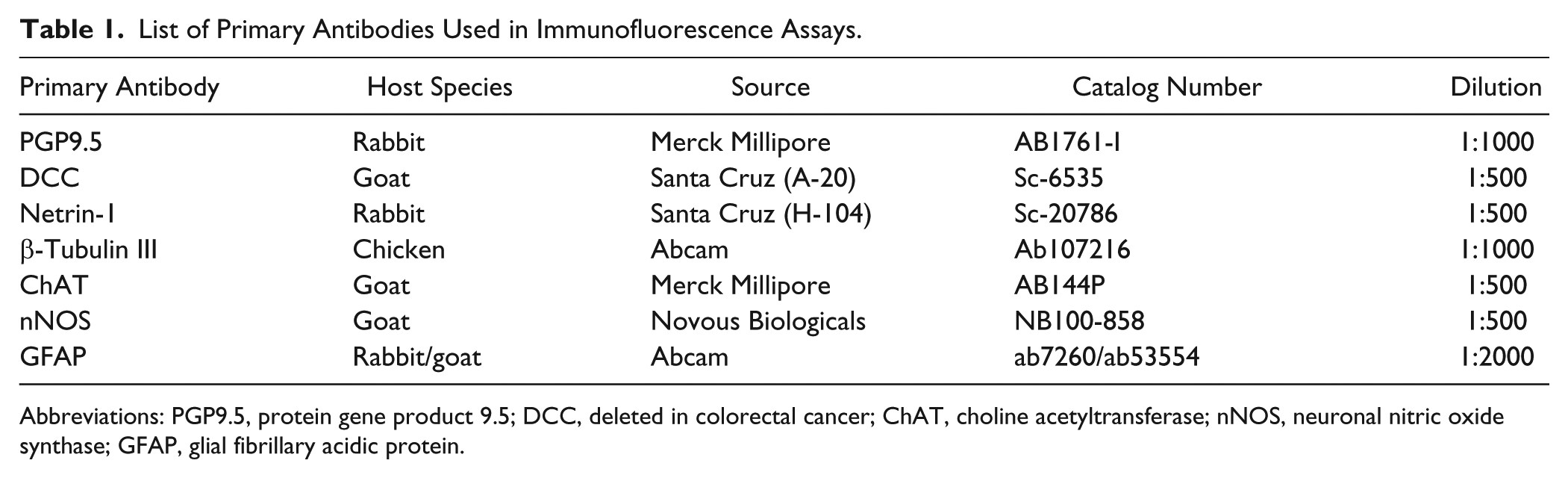
Abbreviations: PGP9.5, protein gene product 9.5; DCC, deleted in colorectal cancer; ChAT, choline acetyltransferase; nNOS, neuronal nitric oxide synthase; GFAP, glial fibrillary acidic protein.
List of Secondary Antibodies Used in Immunofluorescence Assays.
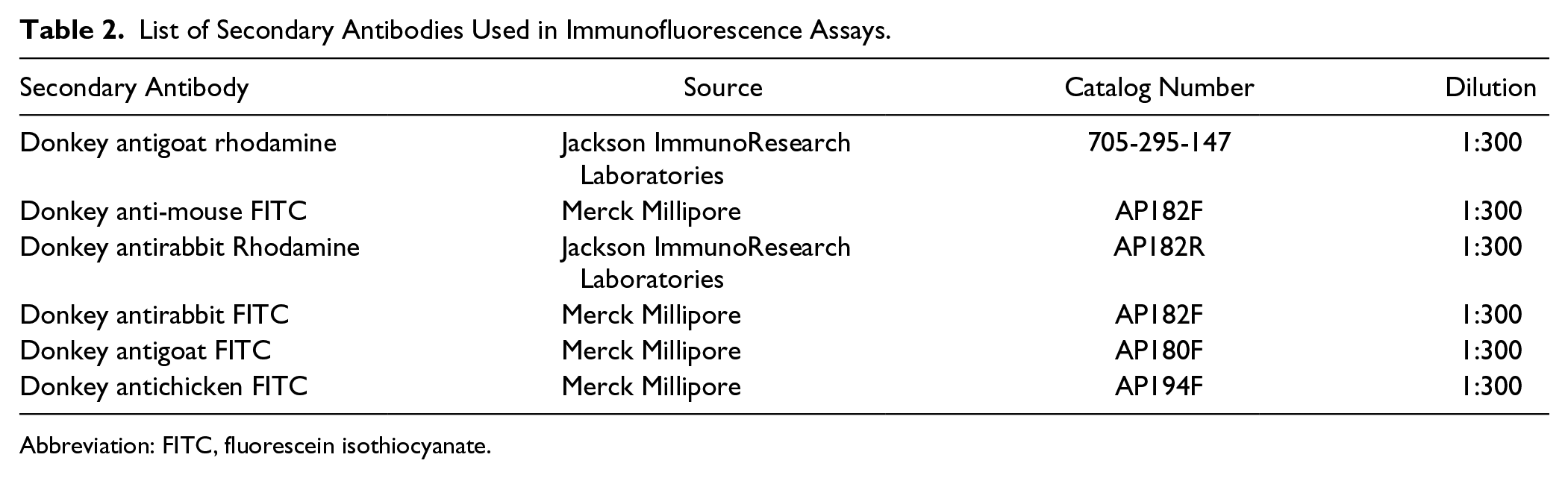
Abbreviation: FITC, fluorescein isothiocyanate.
For secondary antibody control, myenteric plexus in wholemount preparations were incubated in a single primary antibody diluent, namely, a chicken anti-neurofilament heavy chain (NF-H) antibody at 4C overnight. Either anti-DCC or anti-Netrin-1 antibody was omitted in the primary antibody incubation while corresponding species-specific secondary antibodies were added during the secondary antibody incubation for 1 hr at room temperature (Supplemental Fig. 1). In addition, absorption control was conducted using Netrin-1 blocking peptide (Catalog number X1868b, Exalpha Biologicals). 16 The wholemount preparations were incubated in the diluents of anti-NF-H antibody and the preabsorbed Netrin-1 antibody overnight at 4C. After washing, the wholemount preparations were incubated for 1 hr at room temperature in the species-specific secondary antibodies labeled with different fluorophores: donkey antichicken FITC (fluorescein isothiocyanate) and donkey antirabbit rhodamine (Table 2). Preabsorbed Netrin-1 antibody was produced by mixing anti-Netrin-1 antibody (200 μg/mL) with Netrin-1 blocking peptide (1 mg/mL), at a working dilution of 1:10 (antibody:immunogen) ratio, and the mixture was incubated overnight at 4C with a gentle agitation. A double labeling of NF-H and Netrin-1 was conducted parallel to the absorption control (Supplemental Fig. 2). The immunoblot control of DCC antibody specificity was performed in SW480 cells transfected with plasmids including Neo-Bam (empty vector plasmid), DCC-Mutant (DCC gene missing entire intracytoplasmic domain), and DCC-Full (DCC gene full length). The specificity of DCC antibody was also confirmed by using Basic Local Alignment Search Tool (BLAST; Supplemental Fig. 3).
In addition to using Netrin-1 and DCC antibodies from Santa Cruz Biotechnology, the wholemount preparations of the colon tissues were also labeled with antibodies from different sources: a chicken polyclonal anti-Netrin-1 antibody (Aves Labs, Cat #: NET), as well as a rabbit polyclonal anti-DCC antibody (Abnova, Cat #: PAB17539) (Supplemental Fig. 4). As no differences in the pattern of Netrin-1-like-immunoreactivity (IR) and DCC-IR were observed by using these additional antibodies, the antibodies from Santa Cruz Biotechnology (listed in Table 1) were used in this study.
Imaging
The images of wholemount preparations were taken using an Eclipse Ti confocal laser scanning microscope (Nikon, Lidcombe, Australia) with NIS element software. Fluorophores were visualized using a 488 nm excitation filter for FITC, a 561 nm excitation filter for Alexa 594, a 405 nm excitation filter for DAPI, and a 640 nm excitation filter for Alexa 647. Three-dimensional (Z-series) images of the wholemount preparations were acquired at a normal thickness of 0.5 μm, with 20× (dry, 0.75) or 40× (oil immersion, 1.3) or 100× (oil immersion, 1.45) objective lenses.
Neural Cell Counting and Quantitative Analysis
The immunoreactive neuronal cell counting was performed according to the method published previously. 17 The IR of DCC, Netrin-1-like, ChAT, and neuronal nNOS-positive neurons was assessed against IR of β-Tubulin III positive neurons in the myenteric plexus in total area of 2 mm2. Eight randomly chosen images per preparation were taken with a 20× objective. All acquired images were captured with an identical acquisition exposure setting, and they were analyzed blindly. Thresholding for each channel was determined based on a single fluorophore labeling, and the calibrated threshold was applied to analyze double-labeled preparations. The strong and weak intensity of immunostaining was distinguished by setting a constant threshold using MatLab. In the case of Netrin-1 and ChAT double labeling, the merge images were threshold by subtracting one channel from the other channel. As a result, the images show strongly stained neurons, but remove weakly stained neurons. In the case of Netrin-1 and nNOS double labeling, the threshold for Netrin-1 channel was based on histogram settings at a constant value: channel Min = 20 to channel Max = 70. This allowed us to collect the images showing neurons expressing a strong Netrin-1-like-IR. The immunoreactive neurons were counted manually per ganglion in images (512 × 512 pixels per image) using ImageJ plugins cell counter tool (average 30 ganglia in total area of 2 mm2 per preparation).
Statistical Analysis
Results are presented as mean ± SD from three to four animals. GraphPad Prism 7 software was used for statistical analysis. One-way ANOVA followed by Tukey’s multiple comparisons test or Kruskal–Wallis and Dunn’s multiple comparison tests and two-way ANOVA followed by Tukey’s multiple comparisons test were used; p value less than 0.05 was considered significant.
All raw data can be provided upon requests.
Results
Localization of Netrin-1-like and DCC Immunoreactivity in the Myenteric Plexus of the Colon From Healthy Adult Mice
To investigate whether or not Netrin-1 is expressed in the mature ENS, wholemount preparations of the adult mice colon tissues revealing the myenteric plexus were labeled with Netrin-1. Our results showed that the Netrin-1 is distinctively expressed in the mature myenteric plexus. The Netrin-1-like-IR was found to be co-localized with a neuronal marker, protein gene product 9.5 (PGP9.5) that labels neuronal cell bodies and processes (Fig. 1A). Also, Netrin-1-like-IR was clearly shown in the internodal strands forming a nerve plexus. This was detected by the colocalization of Netrin-1 staining combined with another neuronal marker specific to the NF-H (Fig. 1B). The Netrin-1-like-IR was primarily detected in the cytoplasm of the nerve cell bodies, and those Netrin-1-like positive neurons showed both strong and weak staining in their cell bodies. There were many punctate structures surrounding individual neurons within a ganglion (Fig. 1C). To see whether those punctate structures of Netrin-1-IR were associated with glial cells, intermediate filament glial fibrillary acidic protein (GFAP) was costained with Netrin-1. As expected, Netrin-1-like-positive neurons and processes were outlined by the GFAP-IR (Fig. 1D). However, the IR for GFAP and Netrin-1 markers did not appear to be colocalized (Fig. 1E). This suggests that Netrin-1 is specifically expressed in myenteric neurons.
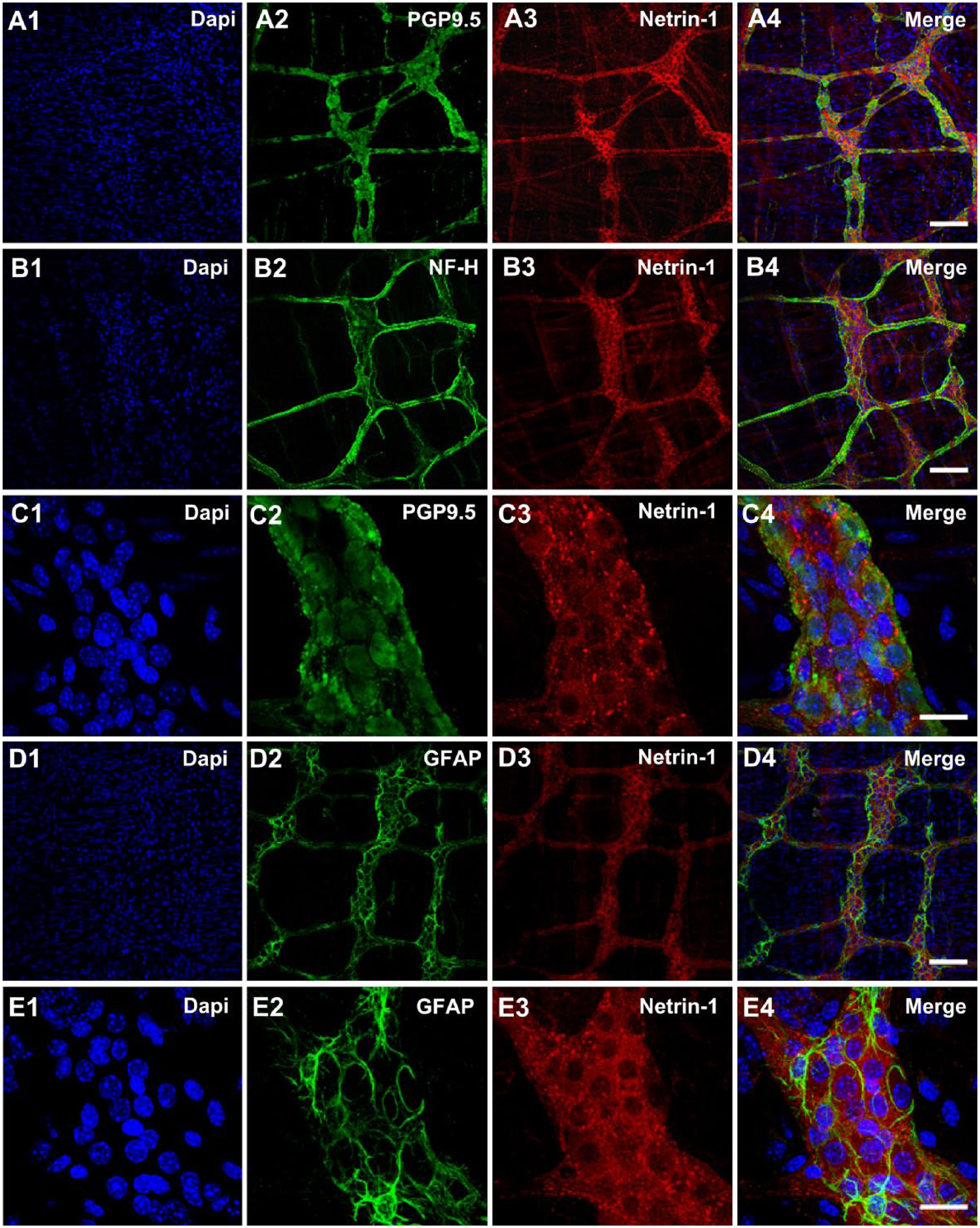
Localization of Netrin-1 in the myenteric plexus of the colon from healthy young adult mice. The myenteric plexus of the colon tissues were stained with DAPI, nuclei staining (A1–E1), neuronal markers, PGP9.5 (A2 and C2), and NF-H (B2), as well as a glial marker, GFAP (D2 and E2). Netrin-1 (A3–E3) was labeled and the merged images (A4–E4) demonstrate the colocalization of neuronal and glial markers with Netrin-1. N=5 animals. Scale bars: 100 μm (A1–4, B1–4, D1–4); 25 μm (C1–4, E1–4). Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole, PGP9.5, protein gene product 9.5; NF-H, neurofilament-heavy chain; GFAP, glial fibrillary acidic protein.
Next, we examined whether or not Netrin-1 receptor, DCC expression is remained in the adult ENS. Our results revealed that DCC is also expressed in the mature myenteric plexus with a distinctively similar pattern to Netrin-1 expression. DCC staining was co-localized with the neuronal markers, PGP9.5 (Fig. 2A), and NF-H (Fig. 2B), suggesting that DCC expression is localized in neuronal cell bodies and nerve processes. Similar to Netrin-1 expression, DCC expression was shown in the cytoplasm of the neuronal cell bodies. A similar pattern of Netrin-1-like-IR and DCC-IR was also observed by using additional antibodies from different sources (Supplemental Fig. 4). This supports the findings that Netrin-1 and DCC are present in the myenteric plexus of the colons in the adult mice. The expression of DCC was found to be evenly distributed among individual neurons (Fig. 2C). In addition, co-labeling of DCC and GFAP was tested to see if this observed DCC expression was specific to the myenteric neurons or was shared with glial cells. The GFAP-IR was shown to outline the DCC-immunoreactive myenteric neurons and internodal strands (Fig. 2D). At a high magnification, some small punctuated structures were exhibited along where the glial cell fibers embraced the neurons (Fig. 2E, arrowheads). However, it appeared some glial cell fibers lack the DCC-IR (Fig. 2E, arrows). Collectively, these data suggested that DCC receptors were primarily expressed in myenteric neuronal cell bodies.
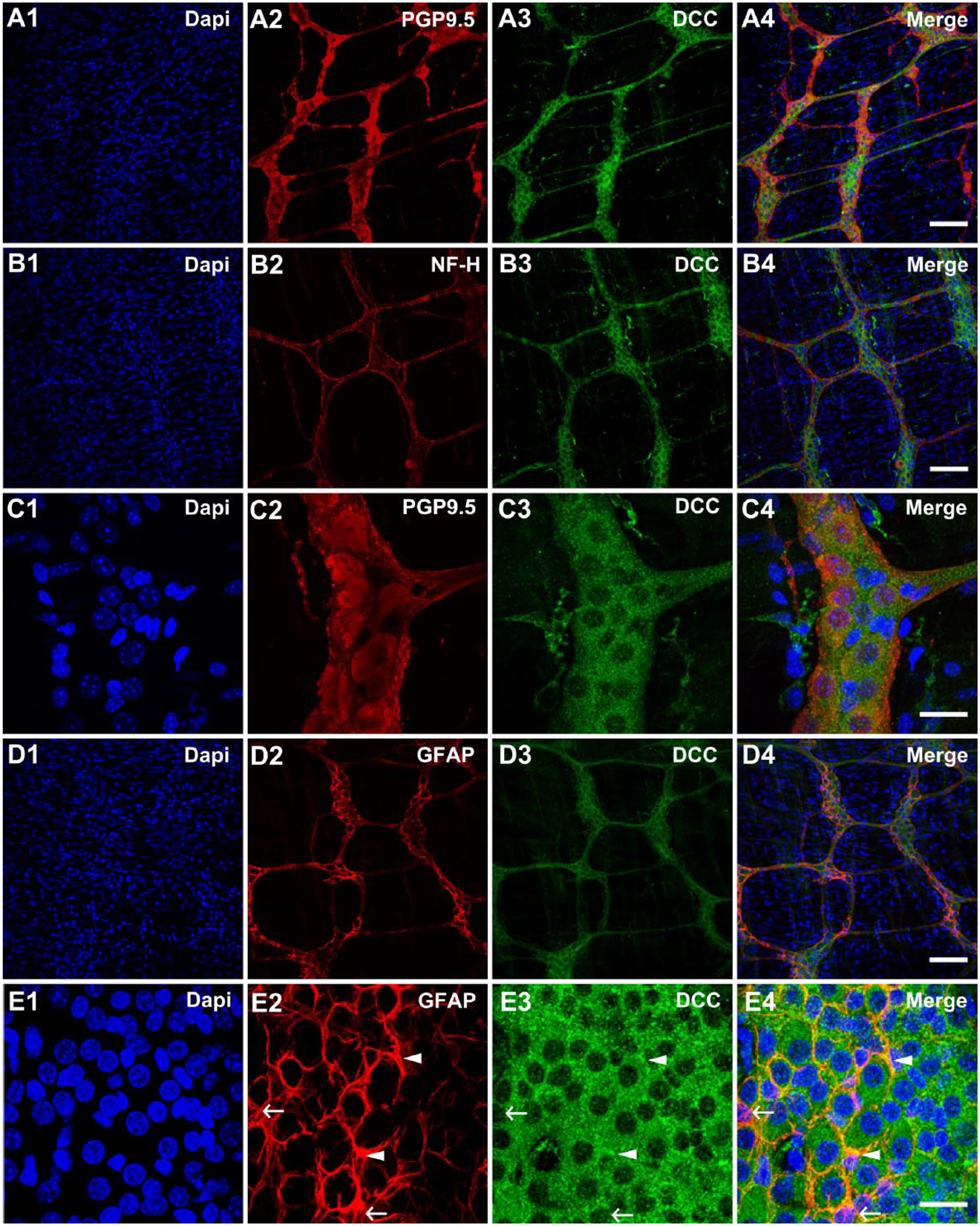
Localization of DCC receptors in the myenteric plexus of the colon from healthy young adult mice. The myenteric plexus of the colon tissues were stained with DAPI, nuclei staining (A1–E1), neuronal markers, PGP9.5 (A2 and C2) and NF-H (B2), as well as a glial marker, GFAP (D2 and E2). DCC receptors (A3–E3) were labeled and the merged images (A4–E4) demonstrate the colocalization of neuronal and glial markers with DCC receptors. Filled arrowheads in E2–4 indicate the colocalization regions of GFAP and DCC markers, while arrows indicate lack of colocalization of GFAP and DCC makers (E2–4). N=5 animals. Scale bars: 100 μm (A1–4, B1–4, D1–4); 25 μm (C1–4, E1–4). Abbreviations: DCC, deleted in colorectal cancer; DAPI, 4′,6-diamidino-2-phenylindole; PGP9.5, protein gene product 9.5; NF-H, neurofilament-heavy chain; GFAP, glial fibrillary acidic protein.
As a similar pattern of the expression between Netrin-1 and DCC in the myenteric plexus was observed, a double labeling of Netrin-1 and DCC was conducted to assess their colocalization in the mature myenteric plexus (Fig. 3A–C). Although DCC-IR was evenly distributed in individual neuronal cytoplasm within ganglia, it was evident that some neurons exhibited stronger Netrin-1-IR in their cytoplasm than others within a ganglion (Fig. 3B). Also, it revealed a strong IR of Netrin-1-like protein in the border of individual neurons (Fig. 3B, B3′) and the varicose axons (Fig. 3C, C3′). Taken together, these observations prompted us to investigate whether Netrin-1 expression is associated with certain neuronal subtypes. In the following section, the data concerning expression of Netrin-1 in relation to the expression of ChAT and nNOS in the colonic myenteric plexus are presented.
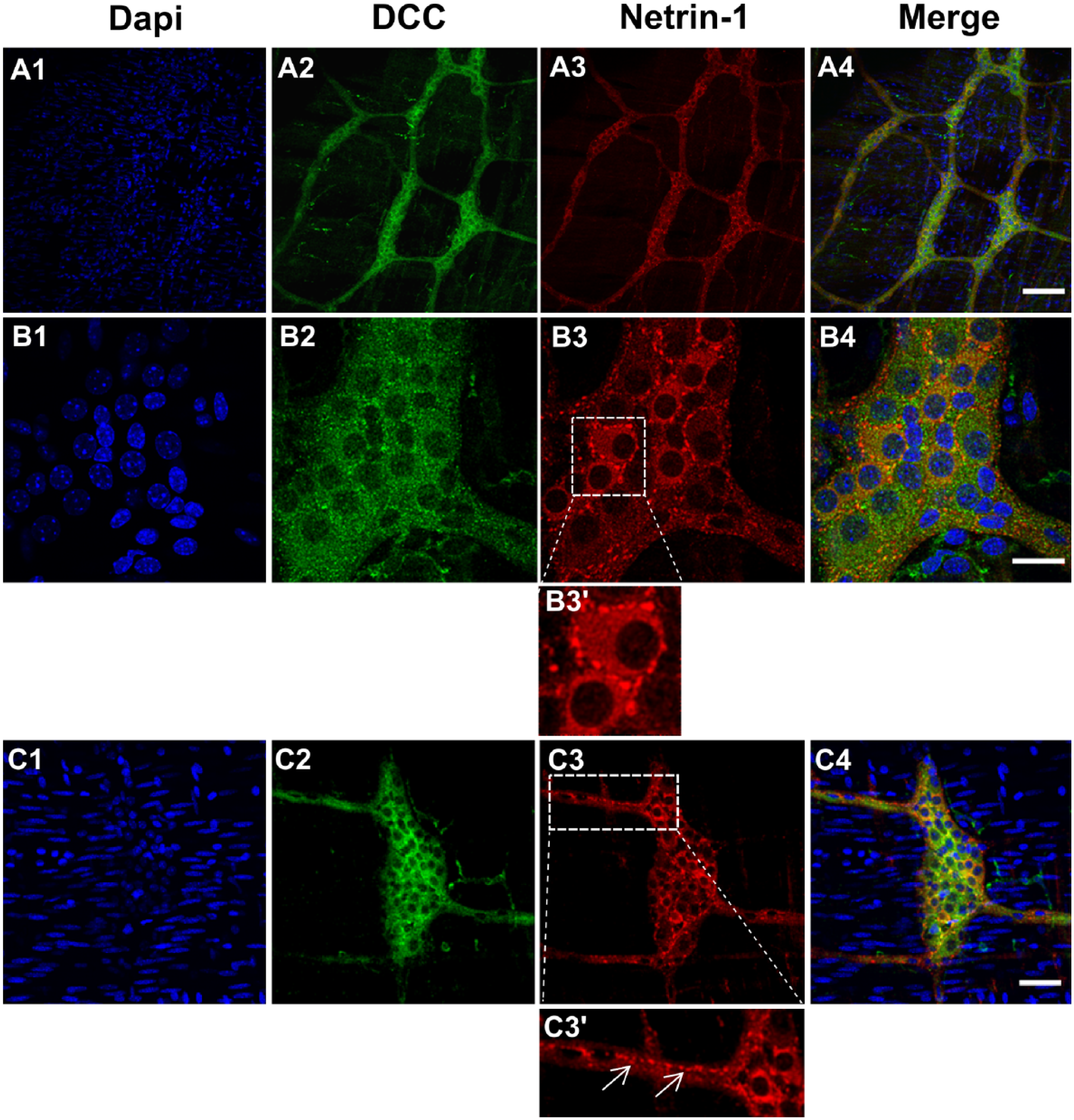
Colocalization of DCC-IR and Netrin-1-IR in the myenteric plexus of the colon from healthy young adult mice. The myenteric plexus of colon tissues were stained with DAPI, nuclei staining (A1–C1), DCC receptors (A2–C2), and Netrin-1 (A3–C3). The merged images (A4–C4) demonstrate the colocalization of DCC receptors and Netrin-1. The inserted box area of B3 is shown with a high magnification view (B3′) and it highlights the punctated structure around the cell body. The arrows in C3′ indicate the IR of Netrin-1 on varicosities. N=5 animals/group. Scale bars: 100 μm (A1–4); 25 μm (B1–4), 50 μm (C1–4). Abbreviations: DCC, deleted in colorectal cancer; IR, immunoreactivity; DAPI, 4′,6-diamidino-2-phenylindole.
Coexpression of Netrin-1-like Protein With ChAT and nNOS in the Myenteric Plexus of the Colon From Healthy Adult Mice
The motility of the gut is controlled by excitatory and inhibitory motor neurons, which are identified by neurochemical coding such as ChAT and nNOS, respectively.18,19 These ChAT and nNOS markers were used to test whether or not the Netrin-1-like-positive neurons overlap with either of those two markers in the myenteric plexus of the colon in healthy young adult mice. As discussed earlier, the Netrin-1-like-IR was found in the myenteric plexus, and this time it was indicated by showing the colocalization of Netrin-1-like protein with a pan-neuronal marker, β-Tubulin III (Fig. 4A). Also, the IR of ChAT was shown to be colocalized with the β-Tubulin III marker, and the majority of myenteric neurons were labeled with the ChAT marker in the myenteric plexus (Fig. 4B). Similar to what had seen in the case of Netrin-1-like-positive neurons, there were either strongly or weakly labeled ChAT-positive neuronal cell bodies in ganglia. The colabeling of Netrin-1 with ChAT was conducted in the myenteric plexus of the colon tissues to confirm their overlapping population (Fig. 4C). Although the majority of the Netrin-1-like-positive and ChAT-positive neurons were shown to overlap, four different combinations of stained neurons were observed and were indicated as arrows and arrowheads in Fig. 4C, C1′ to C3′. These were grouped as neurons that were (1) strongly Netrin-1-like-positive but ChAT-negative (N1++/ChAT–, filled arrowheads); (2) Netrin-1-like-negative but strongly ChAT-positive (N1–/ChAT++, empty arrowheads); (3) strongly Netrin-1-like-positive but also weakly positive for ChAT (N1++/ChAT+, filled arrows); (4) weakly Netrin-1-like-positive but also strongly positive for ChAT (N1+/ChAT++, empty arrows). The groups 3 and 4 were combined into one group, namely, N1+(+)/ChAT+(+), as those neurons expressed both Netrin-1 and ChAT markers (Fig. 4D).
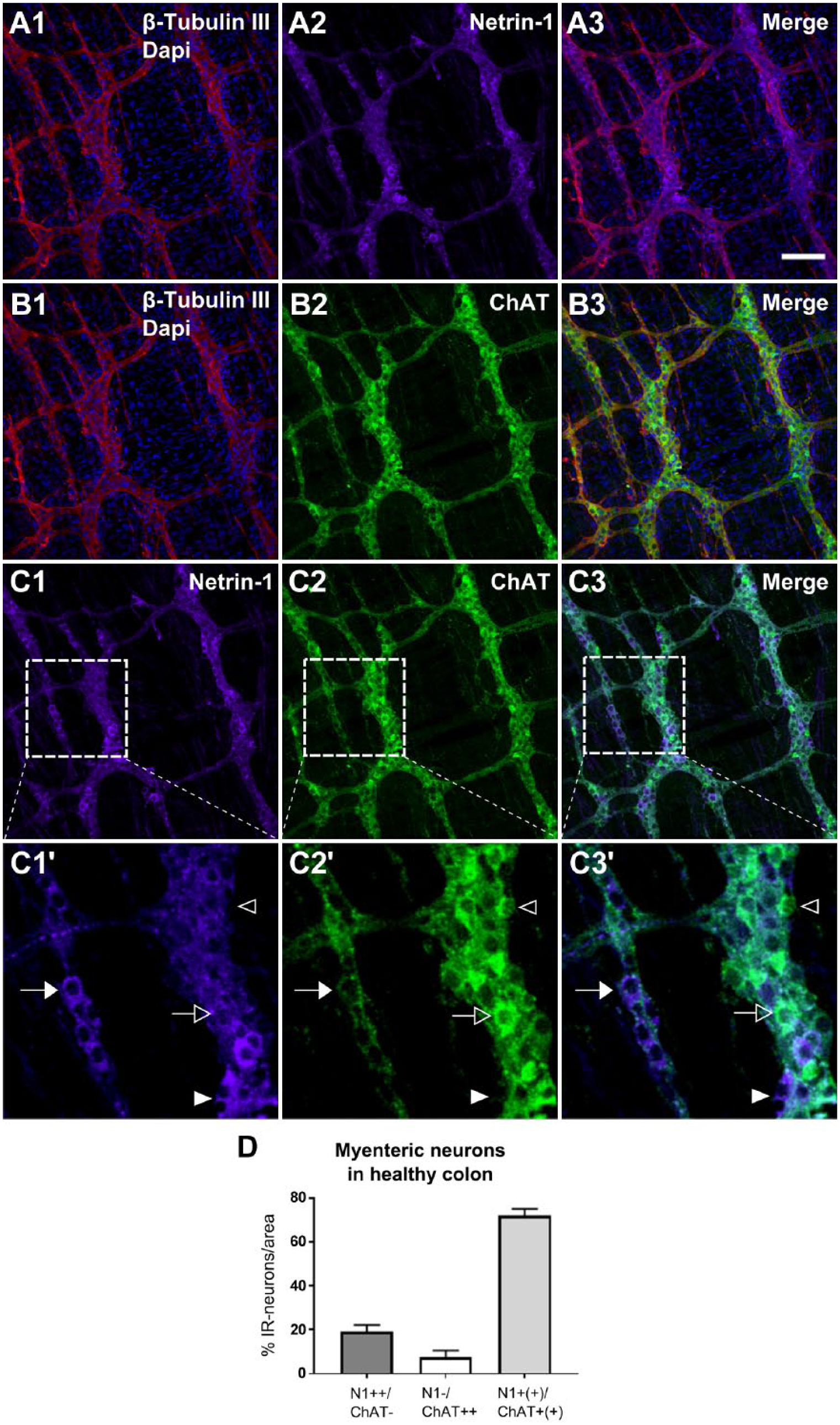
Colocalization of Netrin-1-IR and ChAT-IR in the myenteric plexus of the colon from healthy young adult mice. The myenteric plexus of the colon tissues were triple-labeled with β-Tubulin III, Netrin-1, and ChAT, and the images indicate labeling of β-Tubulin III and Netrin-1 (A1–3), β-Tubulin III and ChAT (B1–3), and Netrin-1 and ChAT (C1–3). The inserted box areas of C1 to 3 are shown with a high magnification view (C1′–3′). In C1′ to 3′, the filled arrowhead indicates the neuronal cell bodies that are strongly Netrin-1-positive, but ChAT-negative (N1++/ChAT–). The empty arrowhead in C1′ to 3′ indicates the neuron that is Netrin-1-negative but strongly ChAT-positive (N1–/ChAT++). The filled arrow in C1′ to 3′ indicates the neuron that is not only strongly Netrin-1-positive but also weakly positive for ChAT (N1++/ChAT+). The empty arrow in C1′ to 3′ indicates the neuron that is not only weakly Netrin-1-positive but also strongly positive for ChAT (N1+/ChAT++). Scale bar: 50 μm. The graph (D) indicates the proportion of the immunoreactive neurons for N1++/ChAT–, N1–/ChAT++, and N1+(+)/ChAT+(+) were measured in the total area of 2 mm2. The population of N1+(+)/ChAT+(+) represents the populations of both N1+/ChAT++ and N1++/ChAT+. Data represented as mean ± SD (n=3 animals). Abbreviations: IR, immunoreactivity; ChAT, choline acetyltransferase; N1, Netrin-1; +, weakly positive; ++, strongly positive; +(+), both weakly and strongly positive; –, negative.
The quantified results showed that either weakly or strongly colabeled neurons for both Netrin-1 and ChAT markers, N1+(+)/ChAT+(+), were found to be 73.3 ± 3.1%. On the contrary, the non-overlapping population which means the neurons positive only for either Netrin-1-like or ChAT marker were shown to be 26.7 ± 3.1%. This included the population of N1++/ChAT– (19.2 ± 3.0%) and the population of N1–/ChAT++ (7.5 ± 3.1%).
Next, to examine whether or not the strong Netrin-1-like-positive neurons overlap with nNOS-positive neurons in the myenteric plexus of the colon from healthy young adult mice, the nNOS marker was co-labeled with Netrin-1 marker (Fig. 5C). The result showed that the proportion of neurons, that were strongly positive for Netrin-1-like protein, was 43.1 ± 3.8% (Fig. 5D). The proportion of neurons that were positive for nNOS was 32.0 ± 3.6%. Around 67% of neurons expressing strong Netrin-1-like protein were also positive for nNOS (N1++/nNOS+), which is 30.2 ± 4.5% of the total number of neurons. This suggests that a high expression of Netrin-1 is associated with the nNOS phenotype in the myenteric plexus of the colon.
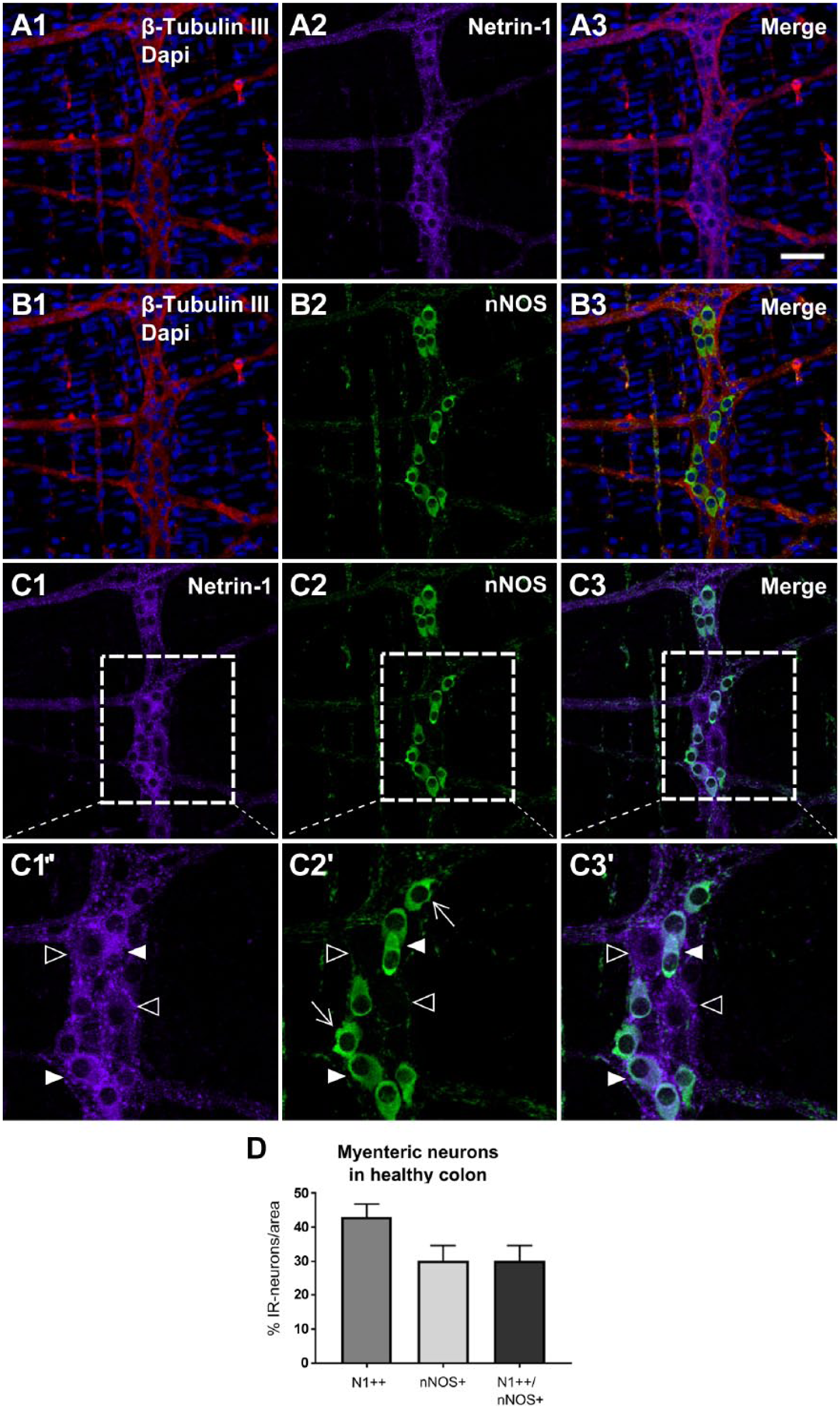
Colocalization of Netrin-1-IR and nNOS-IR in the myenteric plexus of the colon from healthy young adult mice. The myenteric plexuses of the colon tissues were triple-labeled with β-Tubulin III, Netrin-1, and nNOS, and the images indicate labeling of β-Tubulin III and Netrin-1 (A1–3), β-Tubulin III and nNOS (B1–3), and Netrin-1 and nNOS (C1–3). The inserted box areas of C1 to 3 are shown with a high magnification view (C1′–3′). In C1′ to 3′, the filled arrowhead indicates a strong Netrin-1-immunoreactive neuron that was costained with nNOS (N1++/nNOS+). The empty arrowhead in C1′ to C3′, indicates a weak Netrin-1-immunoreactive neuron that did not show nNOS-IR (N1+/nNOS–). The arrow in C2′ indicates a nNOS-positive neuron with flat lamella dendrites. This is known as a characteristic of mature nNOS enteric neurons. Scale bar: 50 μm. The graph (D) indicates the proportion of the immunoreactive neurons for N1++, nNOS+ and N1++/nNOS+ were measured within the total area of 2 mm2. Data represented as mean ± SD (n=3 animals). Abbreviations: IR, immunoreactivity; nNOS, neuronal nitric oxide synthase; N1, Netrin-1; +, weakly positive; ++, strongly positive; +(+), either weakly or strongly positive; –, negative.
Discussion
Intrinsic Expression of Netrin-1 and DCC in the Colonic Myenteric Plexus of Healthy Adult Mice
The work of Ratcliffe et al. 20 has indicated that the Netrin-1 receptor, DCC was found to express in the growth cones of the vagal sensory axons during the embryonic development of mouse gut. These axons were migrated toward the intrinsic enteric neurons where Netrin-1 was thought to be synthesized. Hence, a neural network between the CNS and the ENS is formed during development.9,10 However, the expression of DCC and Netrin-1 and their role in the mature ENS of the adult gut have not been elucidated.
To the best of our knowledge, this is the first study to present the localization of DCC and Netrin-1 in the mature ENS. The immunofluorescence labeling of colonic wholemount preparations indicated that Netrin-1 and the chemoattractive Netrin-1 receptor, DCC, were expressed in the myenteric plexus of the colon in healthy adult mice. In addition to DCC, the chemorepulsive Netrin-1 receptor, UNC5H2, was also examined in our study. Although some IR of UNC5H2 in myenteric ganglia was detected, the staining of UNC5H2 was rather indefinable (data not shown).
The vast majority of all myenteric neurons were positive for DCC and Netrin-1. Also, double labeling of Netrin-1 and DCC showed that both markers were clearly colocalized in the cytoplasm of the neuronal cell bodies and processes.
Previously, the colocalization of Netrin-1 and DCC was found in the cell bodies of neuronal precursor cells from the ventricular zone of the hippocampus in the developing murine brain. A decrease or increase in the level of Netrin-1 or DCC is found to affect neuronal precursor cell survival, regulation of neurogenesis and differentiation in developing murine hippocampus. 21
In the case of developing mammalian cerebral cortex, immunohistochemical labeling revealed that Netrin-1 and DCC localization overlaps in the excitatory synapses during the development and the early postnatal period of synaptogenesis. It had been demonstrated that manipulating gain or loss of Netrin-1 function in the synapses resulted in regulation of the number and stabilization of presynapse and postsynapse formation. The authors suggested that Netrin-1 is a potent synaptogenic molecule for cortical neuron excitatory synapse formation. 22
In the adult CNS, Netrin-1 expression is found in motor neurons and dorsal interneurons of the rodent spinal cord.23,24 Particularly, Netrin-1 expression is mostly enriched in periaxonal myelin membranes, and Netrin-1 is thought to serve as a short-range cue to maintain the appropriately formed network structure in the adult mammalian CNS.25,26 A short-range role of Netrin-1 can be mediated in cell-to-cell interactions. 27 Transient cell-to-cell interactions in the vertebrate olfactory system were shown to mediate patterning of neuronal connectivity by some cells acting as scaffolds for axon guidance and synaptogenesis. 28
Furthermore, in vitro studies have indicated that Netrin-1 can serve as an autocrine factor, and this promotes focal adhesions of the cells thereby slowing cell migration. 29 The autocrine and paracrine functions of Netrin-1 have been shown to promote osteoclast differentiation. 30
Our study demonstrates the colocalization of Netrin-1-like protein and DCC expression in the mature myenteric plexus in the murine colon tissues. At this stage, we can only speculate that this colocalization may be due to the consequence of neuronal precursor cell survival and/or differentiation. Ultimately, it may suggest a stabilization of neuronal synaptic formation in the mature enteric neurons. Determining the relationship of Netrin-1 and DCC coexpression in the mature enteric neurons would be greatly beneficial to understand a functional role of Netrin-1 in the adult ENS.
Particularly, our results showed that Netrin-1-like-positive neurons exhibited both strong and weak staining in their cytoplasm. This observation led us to examine whether or not there was any potential relationship between Netrin-1 expression with specific subtypes of neurons, such as excitatory and inhibitory motor neurons and interneurons. This is discussed in more detail in the next section.
Moreover, some punctate structures from Netrin-1-like-IR were notably observed on the edges of the neuronal cell bodies and along the processes; possibly they are varicosities of the axons where neurotransmitters are contained and released. 31 Often successive varicosities are distributed along an axon. 32 Synaptophysin is a synaptic vesicle marker that has been found in myenteric ganglia and nerve fibers including varicosities in human and rat ENS. 33 The vesicular acetylcholine transporter (VAChT) is a neurotransmitter transporter that functions to load acetylcholine (ACh) into synaptic vesicles in the cleft. 34 Previous studies have shown synaptophysin and VAChT immunolabeling in enteric neurons which appear as granular and/or punctated structures within the neuronal fibers and enteric glial cells,35,36 similar to Netrin-1-like-IR found in this study.
In recent years, Netrin-1 function has been found to be involved in nerve regeneration in the adult CNS and PNS.24,37,38 Studies concerning middle cerebral artery occlusion (MCAO) have indicated that forced up-regulation of Netrin-1 in the peri-ischemic area of the adult rat’s ischemic brain resulted in the upregulation of DCC receptors. 39 The Netrin-1/DCC signaling pathway is found to activate the JNK1/c-Jun pathway that facilitates axonal regeneration and synaptic formation after the MCAO. 39 In particular, Netrin-1 has been implicated in neural plasticity and neuronal regeneration in the adult PNS following injury.37,38 The expression of Netrin-1 receptors such as DCC and UNC5B was significantly upregulated in the injured sciatic nerve when compared with a normal sciatic nerve. 37 After sciatic nerve crush in rats, bone marrow mesenchymal stem cells (BMSCs) producing Netrin-1 were transplanted and these cells showed a significant improvement of nerve recovery by promoting axon regeneration, when compared with the rats transplanted with BMSCs alone. 37
In our study, colabeling of DCC or Netrin-1 and a glial cell marker, GFAP was tested to see whether or not glial cells also express DCC and/or Netrin-1. It appeared that the Netrin-1-like protein is specifically expressed by myenteric neurons, while the DCC is primarily expressed by myenteric neurons as well as some glial cells. GFAP was chosen because its expression is known to be rich in mature enteric glial cells.17,40 However, additional glial cell markers such as calcium binding protein, S100β, or transcription factor SRY box-containing gene 10 (SOX10) 41 may help to further clarify this question.
Correlation Between Netrin-1 Expression and the Phenotypes of ChAT- and nNOS-positive Neurons in the Myenteric Plexus
The ENS is composed of many different functional types of enteric neurons in mammals. 18 Motility of the gut is regulated by the neuronal circuitry of the myenteric plexus. The subtypes of myenteric neurons in the mouse have been characterized by neurochemistry and electrophysiology.42,43 The subtypes include inhibitory and excitatory motor neurons, ascending and descending interneurons and intrinsic sensory neurons. Among those, two major subtypes of myenteric neurons are involved in regulating motility: excitatory motor neurons and interneurons expressing ChAT and inhibitory motor neurons and interneurons expressing nNOS.
Here, we investigated the expression of ChAT and nNOS in relation to Netrin-1 expression in the myenteric neurons in adult healthy mice. It was revealed that the majority of Netrin-1-like expressing neurons also expressed ChAT. However, approximately 19% of neurons were strongly Netrin-1-like-positive but ChAT-negative. In contrast, a small population, around 8% of neurons, were Netrin-1-like-negative but strongly ChAT-positive.
In the case of nNOS-expressing subtype, almost all of nNOS-positive enteric neurons were found to also be strongly Netrin-1-like-IR. This suggests that there is a prominent correlation between Netrin-1 and nNOS expression in the colonic myenteric plexus of the healthy adult mice.
A differential intensity of IR in the retinal ganglion cells has been monitored during normal development and after optic tract lesion in rats. 44 The authors noted that changes in the immunostaining pattern of ganglion cells were involved in retinal neuronal survival, differentiation, and synaptogenesis.
Moreover, the correlation between cancer antigen (CA-125) expression and pathological parameters in endometrial carcinoma has been evaluated by immunohistochemistry. 45 It was noted that the intensity of positively labeled cells (i.e., strong or weak or negative labeling) is correlated with the stages of cancer invasiveness.
Our unpublished data revealed that the intensity of Netrin-1-like-IR and ChAT was modulated in the myenteric plexus of the colon from mice with colorectal cancer. The contribution of Netrin-1 to phenotypic changes in enteric neurons in pathological conditions, such as cancer, may be worth investigating in the future.
Our results revealed that Netrin-1 and DCC are co-expressed in the mature myenteric plexus in the murine colon. This may suggest that Netrin-1 could serve as either short-range cue for cell-to-cell interactions or autocrine and paracrine functions for survival/differentiation and/or stabilizing neural connectivity in the ENS. The direct relationship of Netrin-1 and DCC co-expression in the myenteric neurons needs to be elucidated in the future. In addition, the expression of Netrin-1 is found to be associated with major neuronal subtypes, such as ChAT and nNOS-expressing myenteric neurons. Investigating the nerve regeneration role of Netrin-1 in the adult ENS under the pathological conditions involving an enteric nerve injury would be worth examining to find a potential therapeutic use of Netrin-1 in the adult ENS.
Supplemental Material
DS_10.1369_0022155418819821 – Supplemental material for Netrin-1-like-immunoreactivity Coexpresses With DCC and Has a Differential Level in the Myenteric Cholinergic and Nitrergic Neurons of the Adult Mouse Colon
Supplemental material, DS_10.1369_0022155418819821 for Netrin-1-like-immunoreactivity Coexpresses With DCC and Has a Differential Level in the Myenteric Cholinergic and Nitrergic Neurons of the Adult Mouse Colon by Suh Youn Ko, John T. Price, Gregory L. Blatch and Kulmira Nurgali in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
The authors thank the animal technicians at Victoria University for their assistance in animal husbandry. Also, we thank Ms. Valentina Jovanovska and Dr. Aaron Harwood for assisting in imaging analysis. We acknowledge Biological Optical Microscopy Platform at the University of Melbourne for microscopic software training.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SYK performed and analyzed all experiments, and wrote the manuscript. KN designed the research and wrote the manuscript. The manuscript was reviewed and edited by KN, GLB, and JTP. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was supported by the College of Health and Biomedicine at Victoria University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
