Abstract
Netrin-1, a survival factor, is highly induced and excreted after renal injury in animal experiments. We aimed to research the relationship between serum netrin-1 and parameters of renal tubule function as well as circulating inflammatory cytokines and to evaluate effect factors for netrin-1 in humans with type 2 diabetes mellitus (T2DM) undergoing different levels of albuminuria. In total, 81 T2DM patients were included and divided into three groups according to their amount of 24-h urine albumin excretion (UAE) after receiving consent. Plasma netrin-1, interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) were quantified by commercially available enzyme-linked immunosorbent assay kits and the data were analyzed to assess whether serum netrin-1 correlates significantly with disease progression. The results showed that plasma netrin-1 level in patients with macroalbuminuria was significantly higher than that in those with microalbuminuria and normoalbuminuria. Plasma netrin-1 level was significantly associated with UAE, estimated glomerular filtration rate (eGFR), IL-6, and TNF-α independently of age, sex, diabetes duration, and glycosylated hemoglobin (HbA1c). However, no correlation was found between netrin-1 and β2-microglobulin (β2-MG). Our studies may suggest that serum netrin-1 concentrations are increased with diabetic nephropathy progression, particularly in patients with macroalbuminuria, which are associated with renal insufficiency and compensatory responses after inflammation.
Introduction
A recent study suggested that, in subjects with diabetes, microalbuminuria could predict more effectively for the occurrence of macroalbuminuria and declined renal function. 1 High albuminuria has been identified as a real risk marker in these patients, while its onset is just an early sign of diabetic nephropathy (DN). 2 Moreover, it is increasingly believed that the renal tubule impairment is involved in the development of DN, with detection of several metabolic injury factors associated with sustained diabetes disease. 3 Moreover, the chronic subclinical inflammation may have an important effect on the initiation and progress of DN, which increase the glomerular permeability of intravascular protein in the glomerulus by activating protein kinase cascade and transcription factors.4,5
Netrin-1 is highly induced after acute and/or chronic kidney injury and excreted in urine in both animal and human experiments. Urine netrin-1 concentration has recently received attention as a potential biomarker independently predicting the development of diabetes mellitus and chronic kidney disease.6,7 In these, a majority of study subjects were those who had already developed chronic renal failure (CRF) with damaged renal function or without diabetes history or just on animal experiments; meanwhile, there were limited data available about the mutual relation between the plasma levels of netrin-1 and inflammation cytokines. The relationship between DN development and plasma netrin-1 levels is unknown.
In this study, we aimed to investigate the effects of netrin-1 on type 2 diabetic kidney complications and possible associations between the presence of netrin-1 and inflammation cytokines and to evaluate determinant factors for serum netrin-1 in patients with degree of albuminuria.
Patients and methods
This study was performed in the Department of Endocrinology, Nanjing First Hospital, affiliated to Nanjing Medical University. The study was approved by the appropriate independent ethics committees and regulatory authorities. Informed consent forms compatible with the Declaration of Helsinki and International Conference on Harmonization Good Clinical Practice guidelines were received from the participants prior to the study.
We enrolled 81 patients aged 38–88 years with type 2 diabetes mellitus (T2DM) with different levels of albuminuria from February to August 2016. These subjects were classified into three groups according to their amount of 24-h urine albumin excretion (UAE): patients with normoalbuminuria (Nor-MA; <30 mg/24 h; N = 37), microalbuminuria (Micro-MA; 30–300 mg/24 h; N = 23), and macroalbuminuria (Macro-MA; >300 mg/24 h; N = 21). DN was defined as T2DM patients having chronic kidney disease, UAE ⩾ 30 mg/24 h, and/or estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2. 8
Patients with a history of severe uncontrolled hyper-tension (systolic blood pressure (SBP) ⩾ 180 mmHg and/or diastolic blood pressure (DBP) ⩾ 110 mmHg) and severe dyslipidemia (total cholesterol > 400 mg/day), with the acute infection or chronic inflammatory conditions, and those with diagnosed liver failure, type 1 diabetes mellitus, using anti-infective drugs, with kidney diseases except DN such as kidney transplantation, and coexistence of other renal diseases (except for nephrosclerosis) were excluded from the study.
Blood samples were taken from the antecubital vein after 10 h of overnight fasting. These blood samples were centrifuged within 30 min at 3000 r/min for 10 min and subsequently stored in aliquots without preservatives at −80°C. Netrin-1, interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) levels were measured by commercially available human enzyme-linked immunosorbent assay (ELISA) kits (netrin-1: catalog no. bsk00458; IL-6: catalog no. bsk00040; and TNF-α: catalog no. bsk00162, all from Bioss, Burlington, ON, Canada). The lowest limit of detectability of netrin-1, IL-6, and TNF-α was 70 pg/mL, 2 ng/L, and 8 pg/L, respectively. The intra- and interassay coefficients of variation of the assay were 5% and 10%, respectively. Sodium concentrations, fasting plasma glucose levels, liver and kidney function, and HbA1c levels were assessed using routine hospital standard techniques.
Body mass index (BMI) was calculated as weight (kg)/square of height.
CKD-EPI Cr: estimated glomerular filtration rate (eGFR, mL/min/1.73 m2) = 141 × (Scr/K)a × (0.993)age × 1.018(female); K: female = 0.7, male = 0.9; a: female, Scr ⩽ 0.7, a = –0.329, Scr > 0.7, a = –1.209; male, Scr ⩽ 0.9, a = –0.411, Scr > 0.9, a = –1.209.
Statistical analysis
Non-normal distribution data were assessed and appropriate transformations were made when necessary. Median (interquartile range) values were reported for non-normal data and mean ± standard deviation (SD) is reported otherwise. Chi-square tests were used to assess the relationship between the control and albuminuria groups with categorical variables. Student’s t test was used to compare the measurements of a particular variable of the two separate groups and for normal scattered groups, and the Mann–Whitney U test was used for non-normal distribution. The one-way analysis of variance (ANOVA) was used to compare multiple groups. Pearson correlation analysis was used to determine the relationship between numerical variables for normally distributed groups, and Spearman test was used for abnormally distributed groups. The correlation coefficient (r) between 0.000 and 0.249 was considered as weak relation, from 0.250 to 0.499 as moderate, from 0.500 to 0.749 as strong, and that between 0.750 and 1.000 as very strong. All statistical tests were performed using SPSS (Statistical Package for Social Sciences) for Windows 20.0 statistical software. The
Results
Baseline characteristics
The average age of the 81 individuals who participated in our study was 63.94 ± 11.64 years and the BMI average was 24.29 ± 3.95 kg/m2. The individuals with type 2 diabetes in the three groups were equal in terms of age and gender. Demographics and clinical features according to the albuminuria categories are presented in Table 1. With increasing albuminuria, patients had higher SBP, duration of diabetes, blood urea nitrogen (BUN), serum creatinine (Scr), and Cl levels, whereas they had lower levels of serum albumin, eGFR, and HbA1c. DBP was also comparable among the three groups. No differences were observed in alanine transarninase (ALT), aspartate transaminase (AST), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), Na, K, and uric acid (UA) among the three groups. Naturally, HbA1c levels of all study patients were above the normal values, but HbA1c was lower in the Macro-MA group than the other two groups (
General clinical profiles between the three groups.

Nor-MA: normoalbuminuria; Micro-MA: microalbuminuria; Macro-MA: macroalbuminuria; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; UAE: urinary albumin excretion; HbA1c: glycosylated hemoglobin; ALT: alanine transarninase; AST: aspartate transaminase; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; BUN: blood urea nitrogen; Scr: serum creatinine; TC: total cholesterol; TG: triglycerides; UA: uric acid; ALB: plasma albumin.
Continuous variables were expressed as mean (standard deviation) and non-normally distributed variables were expressed as median (interquartile range).
Student’s t test was used to compare the measurements of a particular variable of the two separate groups and for the normal scattered groups.
Mann–Whitney U test was used for non-normal distribution.
Levels of serum netrin-1 and cytokines according to the albuminuria stage
Serum netrin-1 median was statistically significantly higher (
Plasma cytokines levels of patients according to the albuminuria categories.
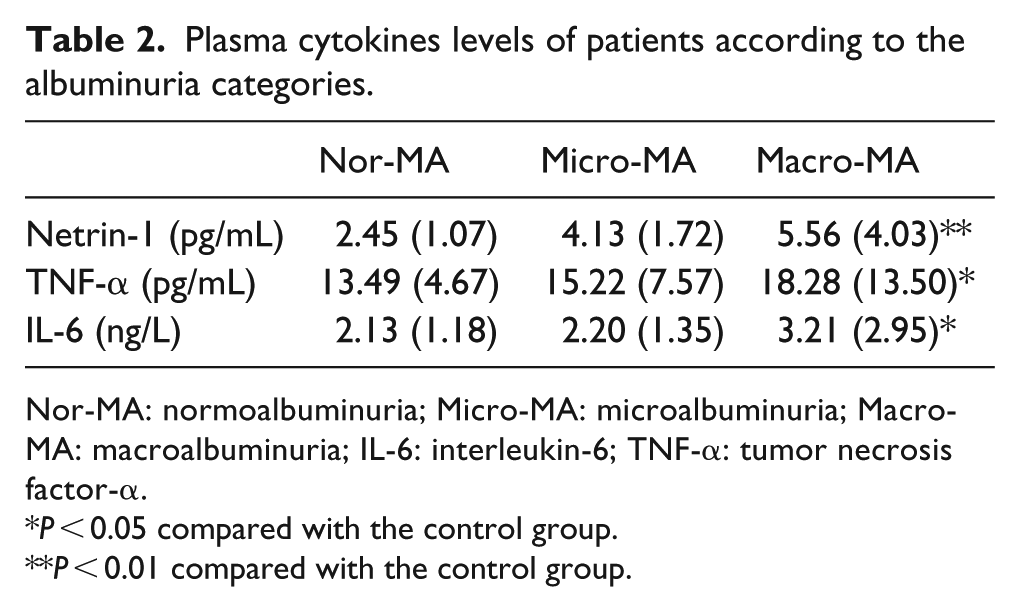
Nor-MA: normoalbuminuria; Micro-MA: microalbuminuria; Macro-MA: macroalbuminuria; IL-6: interleukin-6; TNF-α: tumor necrosis factor-α.
Effect factors of netrin-1 level in DN
The correlations of netrin-1 with 20 parameters are depicted in Table 3. A positive statistically significant correlation was found between netrin-1 level and IL-6, TNF-α, UAE, NAG, and RBP. There were a slightly strong correlation and a negative statistically significant difference between netrin-1 level and eGFR. There were a very strong correlation between netrin-1 level and TNF-α, a strong correlation with IL-6, and a mediocre strong correlation between UAE, NAG, and RBP (Table 3). After adjustment for age, sex, diabetes duration, and HbA1c, the concentrations of TNF-α, IL-6, eGFR, and UAE still correlated independently with netrin-1 (
Univariate correlation analysis between serum netrin-1 and the clinical parameters.
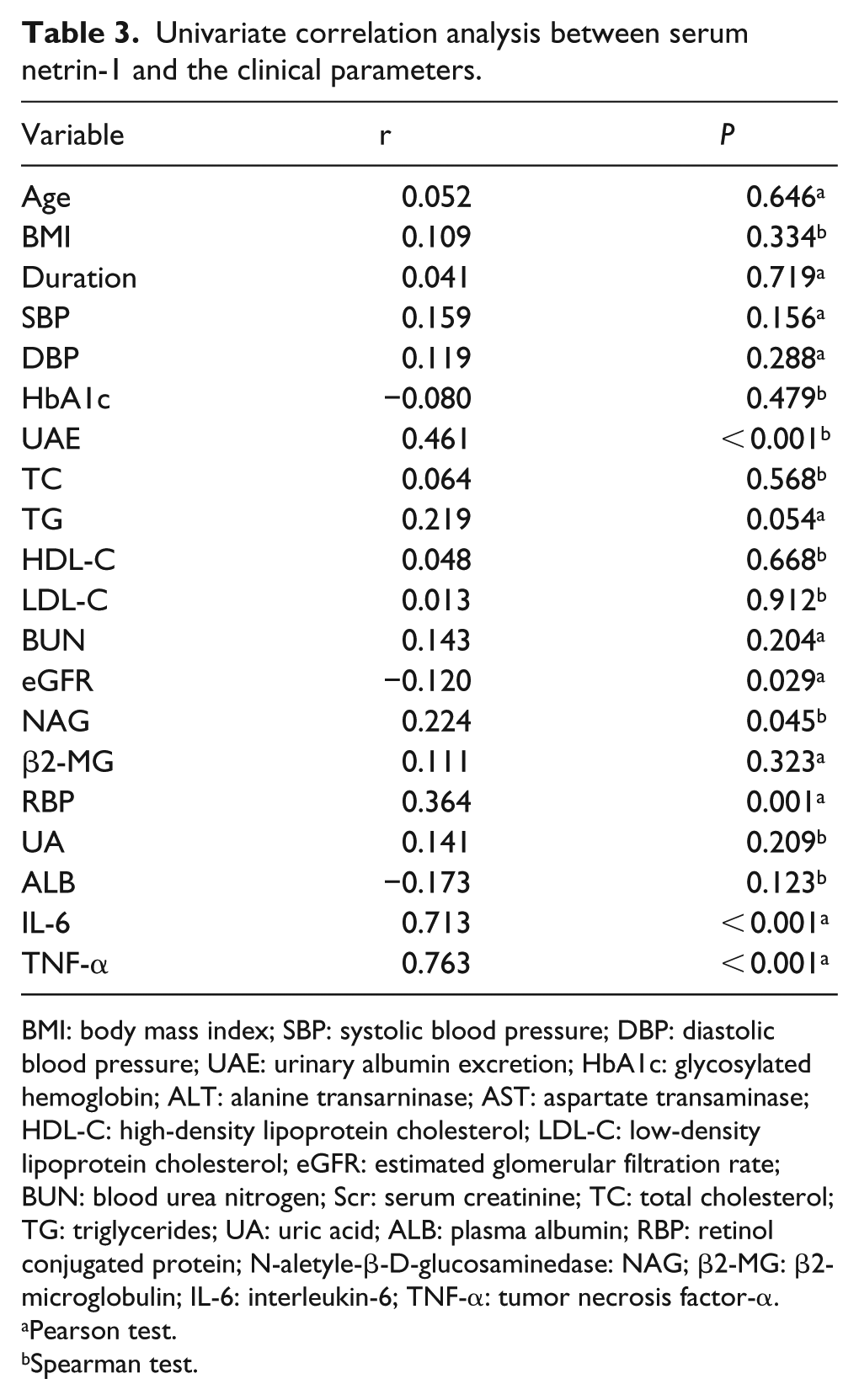
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; UAE: urinary albumin excretion; HbA1c: glycosylated hemoglobin; ALT: alanine transarninase; AST: aspartate transaminase; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; eGFR: estimated glomerular filtration rate; BUN: blood urea nitrogen; Scr: serum creatinine; TC: total cholesterol; TG: triglycerides; UA: uric acid; ALB: plasma albumin; RBP: retinol conjugated protein; N-aletyle-β-D-glucosaminedase: NAG; β2-MG: β2-microglobulin; IL-6: interleukin-6; TNF-α: tumor necrosis factor-α.
Pearson test.
Spearman test.
Discussion
We did not expect the progress of DN to increase netrin-1 levels, but there are several possible explanations for this finding. First, the high levels of albumin trigger production of netrin-1 from proximal tubular epithelial cells via extracellular signal–regulated kinase (ERK) and Akt kinase pathways to enhance translation.
9
Second, netrin-1 production is a compensatory mechanism for defective reduced capacity of the proximal tubules in early diabetes. More recently, impaired tubular reabsorption of albumin plays a major role in the development of DN. Our results are in line with recent reports that several tubular damage markers are increased in CKD. NAG and RBP levels were statistically higher in patients as albuminuria increased (all
Experiments on renal injury mice have shown that the major source of netrin-1 in acute kidney injury was proximal tubular epithelial cells. In addition, netrin-1 is minimally expressed in tubular epithelial cells of the normal kidney, while it is widely expressed in the damaged kidney. Tu et al. 11 found elevated netrin-1 in septic acute kidney injury, and no changes of netrin-1 level were detected in healthy volunteer urine samples. NAG is a molecular weight lysosomal enzyme, which is present in tubular cells and induced directly by kidney injury, which need not be accompanied by a reduction in glomerular filtration rate. RBP, a low-molecular-weight protein, is filtered by the glomeruli and then reabsorbed and catalyzed by the proximal tubules. An increased level of RBP occurs with altered tubular function and when tubular reabsorptive capacity is exceeded. 12 β2-MG is a polypeptide with a molecular weight of 11.8 kDa. The concentration of β2-MG increases progressively, which is in parallel to the decrease in glomerular filtration rate in patients with CKD. In this study, the levels of NAG and RBP were positively correlated with netrin-1. However, no correlation was found between netrin-1 and β2-MG. We may say that rising netrin-1 levels in plasma represent a helpful biomarker to show the tubular damage. Moreover, overexpression of netrin-1 could increase albumin uptake in proximal tubular epithelial cells and decreased excretion of albuminuria, which was not altering the normal kidney function.
In conclusion, serum proximal tubular marker netrin-1 elevated in DN patients, particularly in those with Macro-MA, and which was augmented in a compensatory way with the progression of inflammation. Further randomized prospective studies with larger samples are required to support our findings.
Footnotes
Acknowledgements
We would like to thank the members of the Central Laboratory of Nanjing First Hospital affiliated to Nanjing Medical University and the Department of Endocrinology for sharing ideas. We would also like to thank all the patients and staff who participated in this study.
Ethical approval
The studies were approved by the local ethics committee or institutional review boards and in accordance with the Declaration of Helsinki, and are consistent with the International Conference on harmonization-Good Clinical Practices and applicable amendments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by grants from the National Natural Science Foundation of China (81200594) and Jiangsu Planned Projects of Postdoctoral Research Funds.
