Abstract
Diabetic nephropathy is characterized by decreased expression of bone morphogenetic protein-7 (BMP-7) and decreased podocyte number and differentiation. Extracellular antagonists such as connective tissue growth factor (CTGF; CCN-2) and sclerostin domain-containing-1 (SOSTDC1; USAG-1) are important determinants of BMP signaling activity in glomeruli. We studied BMP signaling activity in glomeruli from diabetic patients and non-diabetic individuals and from control and diabetic CTGF+/+ and CTGF+/− mice. BMP signaling activity was visualized by phosphorylated Smad1, −5, and −8 (pSmad1/5/8) immunostaining, and related to expression of CTGF, SOSTDC1, and the podocyte differentiation markers WT1, synaptopodin, and nephrin. In control and diabetic glomeruli, pSmad1/5/8 was mainly localized in podocytes, but both number of positive cells and staining intensity were decreased in diabetes. Nephrin and synaptopodin were decreased in diabetic glomeruli. Decrease of pSmad 1/5/8 was only partially explained by decrease in podocyte number. SOSTDC1 and CTGF were expressed exclusively in podocytes. In diabetic glomeruli, SOSTDC1 decreased in parallel with podocyte number, whereas CTGF was strongly increased. In diabetic CTGF+/− mice, pSmad1/5/8 was preserved, compared with diabetic CTGF+/+ mice. In conclusion, in human diabetic nephropathy, BMP signaling activity is diminished, together with reduction of podocyte markers. This might relate to concomitant overexpression of CTGF but not SOSTDC1.
Keywords
In the present study, we assessed glomerular BMP signaling activity in diabetic nephropathy in relation to SOSTDC1 and CTGF expression, and to podocyte number and differentiation.
Materials and Methods
Human Tissue
Renal tissue from five patients with diabetic nephropathy was selected from the archives of the Department of Pathology of the University Medical Center Utrecht in accordance with the Declaration of Helsinki and the guidelines of the University Medical Center Utrecht. Two out of five diabetic kidneys were transplant kidneys. Patients' age ranged from 54 to 75 years; three patients were women, and two were men. Mean disease duration was 10 years. As controls, tumor-free areas of five human kidneys obtained after surgical nephrectomy were used.
Immunohistochemistry
Tissue samples were formalin-fixed and paraffin-embedded (FFPE). Four-μm-thick sections were used for immunostaining. Primary antibody source, antibody references, antigen retrieval methods, and primary antibody dilutions are shown in Table 1. The PowerVision poly-HRP-anti-rabbit IgG or poly-HRP-anti-mouse/rabbit/rat IgG detection system (ImmunoLogic; Duiven, The Netherlands) was applied to enhance immunoreactivity, using DAB (ImmunoLogic) or NovaRed (Vector; Torrance, CA) as chromogens. For anti-nephrin, rabbit anti-guinea pig-HRP (P0141; Dako Netherlands, Heverlee, Belgium) was used as a secondary antibody before the application of PowerVision as tertiary antibody. Sections were counterstained with hematoxylin. Negative controls were performed by replacing the primary antibody with normal serum of the species in which the primary antibody was raised. Photographs were taken on a Nikon Eclipse E800 microscope with a Nikon DXM1200 digital camera using the Nikon ACT-1 software, version 2.70 (Nikon Netherlands; Lijnden, The Netherlands).
Immunofluorescence
Three-μm-thick FFPE tissue sections were used for immunofluorescent studies (for primary antibody source and dilutions, see Table 1). For anti-synaptopodin, goat anti-mouse FITC was used as secondary antibody (F0479; Dako Netherlands). All other primary antibodies were detected using PowerVision and the TSA tetramethylrhodamine system or TSA fluorescein system (PerkinElmer Life Sciences; Waltham, MA). Nuclear counterstain was performed with TO-PRO-3 iodide (Molecular Probes; Eugene, OR). Double-labeling immunofluorescence was performed as a sequence of two single-immunofluorescent protocols. Photographs were taken with a Leica TCS SP2 confocal microscope (Leica Microsystems; Rijswijk, The Netherlands).
Quantification
The cellular localization of immunoreactivity was examined by three investigators. Globally sclerosed glomeruli were excluded from analysis. Glomeruli were examined at random positions on the slides. The number of positive cells was counted in at least 20 glomeruli per subject. For correlations, WT1 and pSmad1/5/8, as well as WT1 and SOSTDC1, were counted on consecutive sections. The pSmad1/5/8 staining intensity and synaptopodin-positive and nephrin-positive glomerular tuft areas were calculated in at least 20 glomeruli using Adobe Photoshop software, version 10.0 (Adobe Systems; San Jose, CA) and ImageJ software, version 1.39u (National Institutes of Health; Bethesda, MD).
Primary antibodies, dilutions, and antigen retrieval methods for immunohistochemistry and immunofluorescence
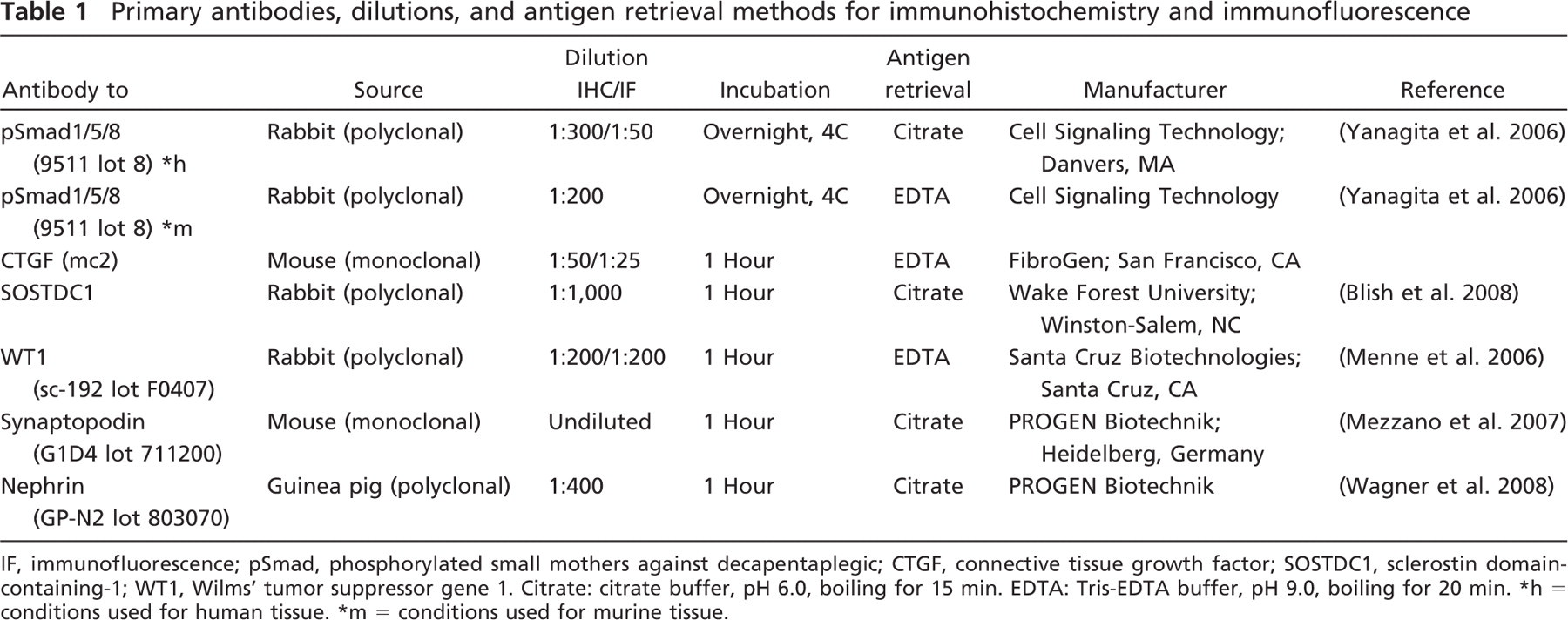
IF, immunofluorescence; pSmad, phosphorylated small mothers against decapentaplegic; CTGF, connective tissue growth factor; SOSTDC1, sclerostin domain-containing-1; WT1, Wilms' tumor suppressor gene 1. Citrate: citrate buffer, pH 6.0, boiling for 15 min. EDTA: Tris-EDTA buffer, pH 9.0, boiling for 20 min.
5 conditions used for human tissue.
= conditions used for murine tissue.
Animal Experiments
Outbred male BALBc/129Sv CTGF+/− mice, in which exon 1 of one CTGF allele had been replaced by a neomycin-resistant gene, were mated with female C57BL/6J mice. From the first offspring, female CTGF+/− mice and female CTGF+/+ littermates were used for this study. Diabetes was induced in five 16-week-old CTGF+/− mice and four CTGF+/+ mice by injection of streptozotocin. Nine control mice were injected with vehicle only. Mice were killed seventeen weeks after induction of diabetes (Nguyen et al. 2008a). The experiments were performed with the approval of the Experimental Animal Ethics Committee of the University of Utrecht.
Statistical Analysis
Data are presented as medians. Differences between control and diabetic subjects were calculated using the Mann-Whitney test. Association for WT1 and SOSTDC1 was calculated using linear regression. A
Results
Glomerular BMP Signaling Activity Is Decreased in Diabetic Nephropathy
Immunohistochemical nuclear staining of pSmad1/5/8 was much weaker in glomeruli of diabetic patients than in control kidneys (Figures 1A and 1B). The number of pSmad1/5/8-positive cells per glomerular cross section was clearly decreased in diabetes (9.2 versus 41.6 in controls,
Nephrin and Synaptopodin Are Decreased in Diabetic Nephropathy
Nephrin staining intensity showed a strong tendency (
Synaptopodin staining intensity showed a tendency (
CTGF Is Upregulated, Whereas SOSTDC1 Is Decreased in Diabetic Nephropathy
In control glomeruli, CTGF expression was weak, whereas SOSTDC1 staining was very prominent. Double-immunofluorescent staining of CTGF and WT1 indicated that in control as well as diabetic glomeruli, CTGF is present only in podocytes (Figure 3A). CTGF was upregulated in diabetes, as evidenced by increased staining intensity and number of positive cells per glomerulus (11.1 versus 3.8 in controls,
The number of SOSTDC1-positive cells correlated with and was almost identical to WT1 counts, both in control and in diabetic glomeruli (
In Experimental Diabetic Nephropathy, CTGF Inhibits BMP Signaling
As shown previously, CTGF expression was increased in podocytes of diabetic wild-type (CTGF+/+) mice, but not in diabetic CTGF+/− mice (Nguyen et al. 2008a). Also, albuminuria was less pronounced in diabetic CTGF+/− compared with diabetic wild-type mice (Nguyen et al. 2008a). To this we add that pSmad1/5/8 was mainly localized in podocytes. Consistent with the inhibitory effect of CTGF on BMP signaling, pSmad1/5/8 immunohistochemical staining was decreased only in diabetic wild-type mice, but preserved in diabetic CTGF+/− mice (Figure 5).
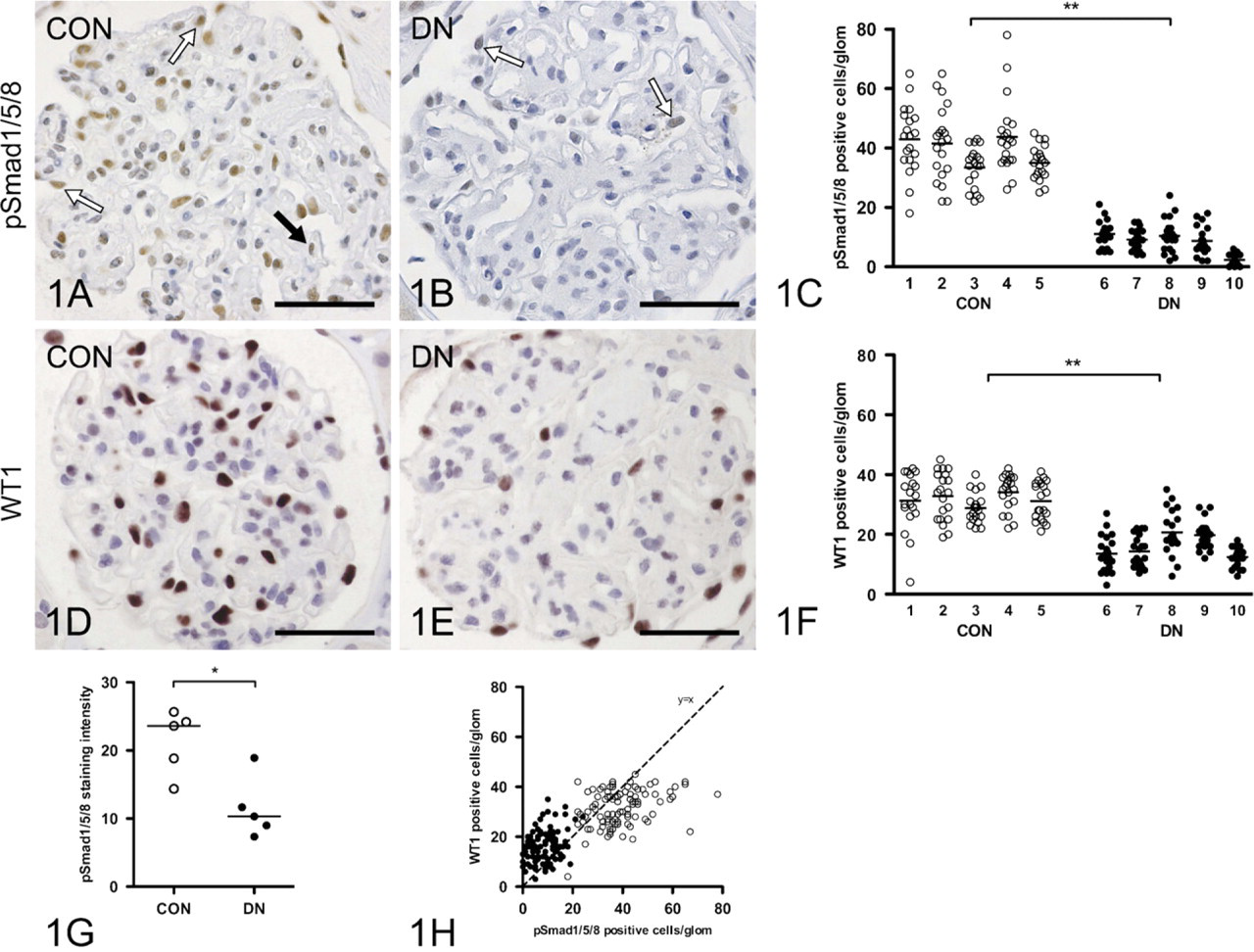
Bone morphogenetic protein (BMP) signaling activity is decreased in human diabetic glomeruli. Nuclear staining of phosphorylated Smad1, −5, and −8 (pSmad1/5/8) in human non-diabetic control (CON) glomerulus
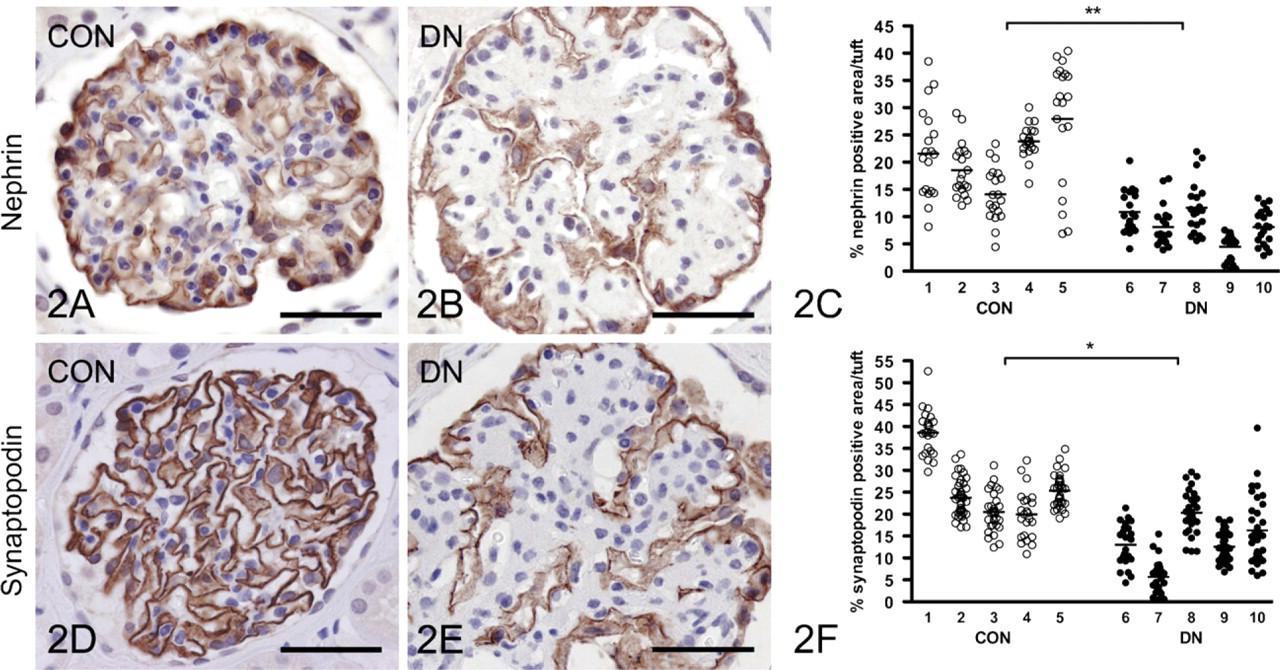
Loss of podocyte differentiation markers in human diabetic glomeruli. Nephrin-positive area and staining intensity is reduced in diabetic (DN) as compared with control (CON) glomeruli
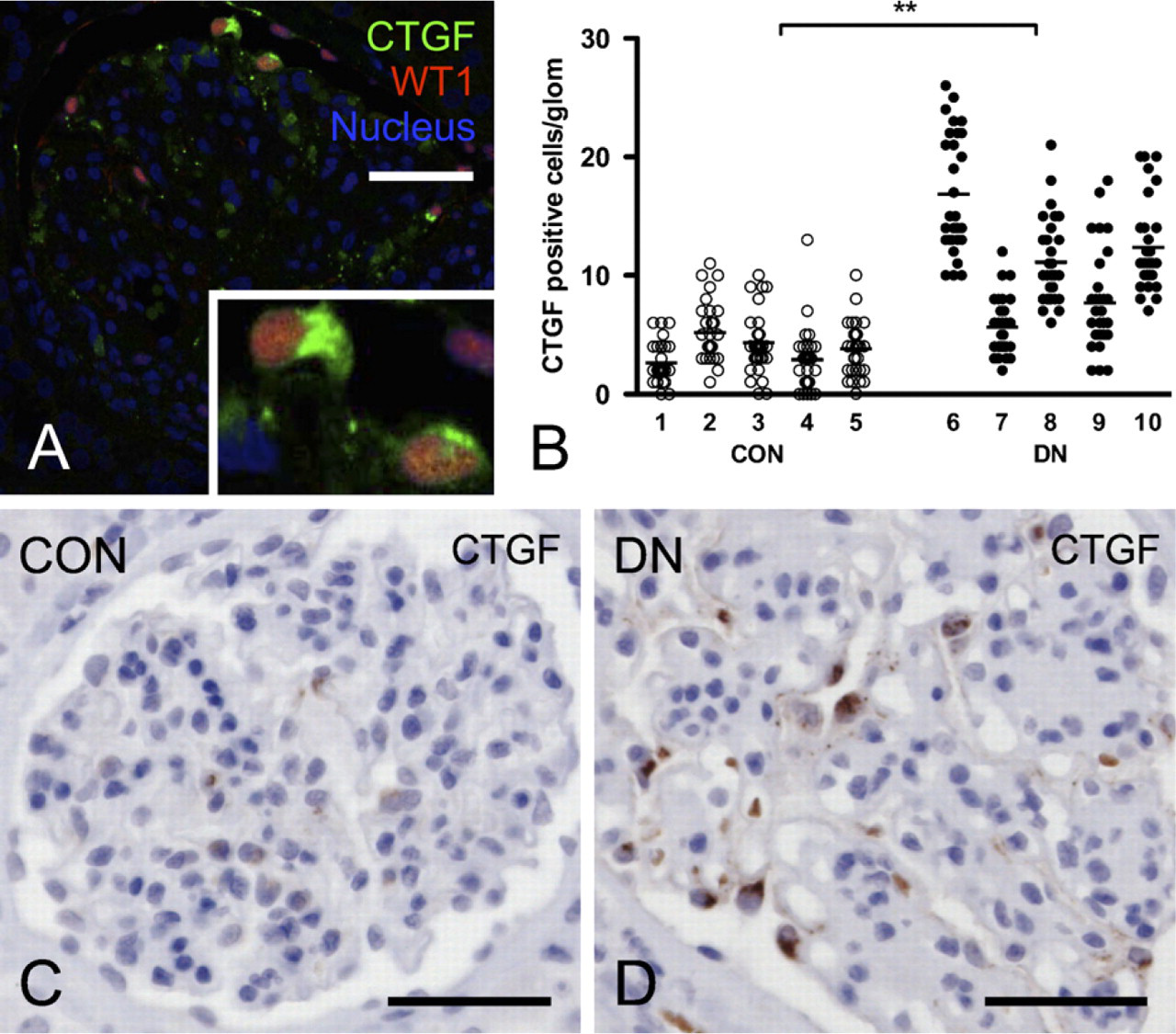
Connective tissue growth factor (CTGF) is upregulated in human diabetic glomeruli and localized in podocytes.
Discussion
The results of this study show that in human diabetic nephropathy, loss of podocytes and podocyte differentiation markers is associated with decreased BMP signaling activity, and that this might relate to increased CTGF expression. Strikingly, expression of SOSTDC1, another important antagonist of BMP signaling in the kidney, was found to be downregulated in diabetic glomeruli. In our specimens of diabetic nephropathy, the number of podocytes per glomerulus was significantly decreased, as evidenced by loss of WT1-positive nuclei. Similarly, the number of pSmad1/5/8-positive nuclei was decreased, indicating decreased BMP signaling activity. Also, the podocyte differentiation markers nephrin and synaptopodin were decreased in diabetic glomeruli. By immunohistochemistry, the two BMP antagonists known to be expressed in glomeruli, SOSTDC1 and CTGF, were found to be expressed primarily in normal and diabetic glomeruli, respectively. SOSTDC1, abundantly present in podocytes in control glomeruli, was downregulated in diabetic glomeruli, and therefore appears not to have a major role in decreased BMP signaling in diabetic nephropathy. CTGF, however, was hardly present in control glomeruli but strongly upregulated in podocytes in diabetic glomeruli. A possible role of CTGF in downregulation of BMP signaling in diabetic glomeruli was further emphasized by the preserved pSmad1/5/8 staining of podocyte nuclei that we observed in diabetic mice with hemizygous deletion of the CTGF gene (CTGF+/− mice). Together, these data suggest that CTGF rather than SOSTDC1 contributes to loss of BMP signaling activity in diabetic nephropathy.
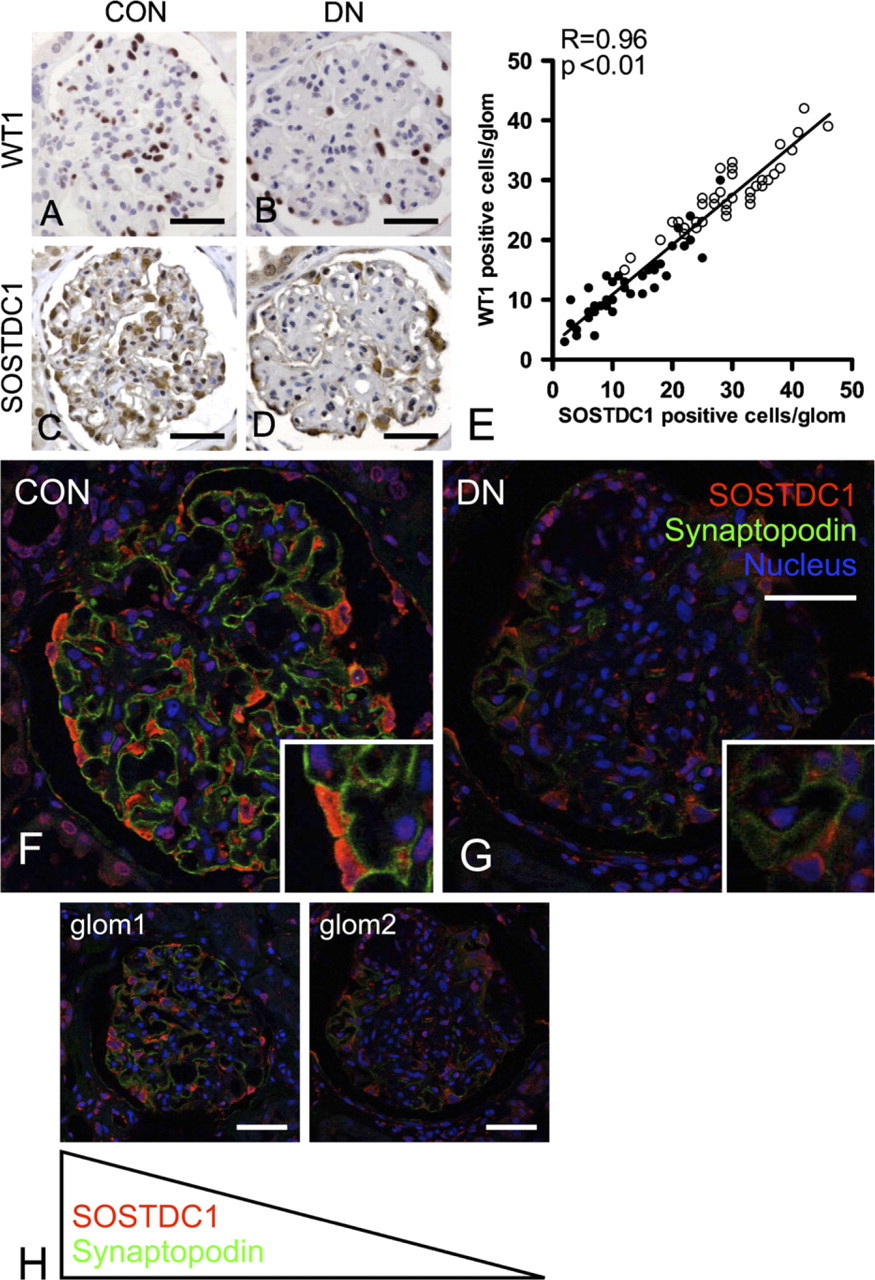
Sclerostin domain-containing-1 (SOSTDC1) is decreased in human diabetic glomeruli due to podocyte loss.
Decreased BMP signaling is an important determinant of adverse remodelling in renal response to injury, in particular diabetic nephropathy. BMP signaling activity is regulated at multiple levels. Major determinants include availability of various ligands and receptors, and extracellular modulators (Mitu and Hirschberg 2008). Phosphorylation of Smad1/5/8 is a central downstream element of BMP signal transduction, which makes it a good readout for this pathway. We show here that in human control glomeruli, pSmad1/5/8 is present mainly in podocytes. In diabetic glomeruli, we noticed a marked reduction of the number of pSmad1/5/8-positive cells. Not all of this reduction could be attributed to loss of podocytes. Control glomeruli contained more pSmad1/5/8-positive cells than podocytes. In contrast, in diabetic glomeruli, the number of podocytes exceeded that of pSmad1/5/8-positive cells. Moreover, the pSmad1/5/8 staining intensity was much lower in diabetic glomeruli.
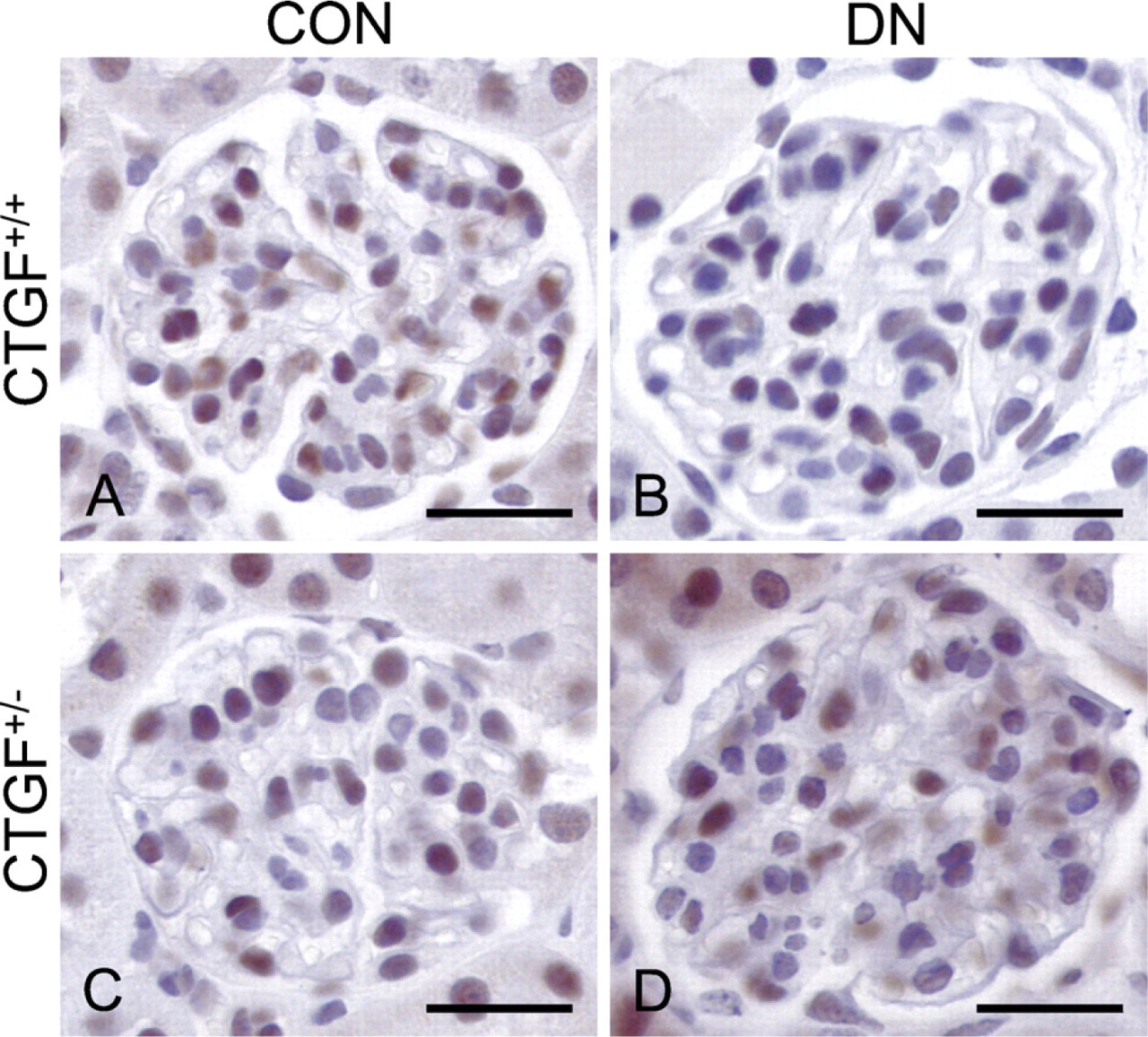
CTGF contributes to loss of BMP signaling in experimental diabetic nephropathy. In control (CON) CTGF+/− and CTGF+/+ mice, pSmad1/5/8 staining in podocytes is comparable
Both in vitro and in vivo studies have indicated that BMP-7 is important for maintenance of podocyte viability and differentiation (Wang et al. 2006; De Petris et al. 2007; Mitu et al. 2007). For example, in podocytes cultured under high glucose, Smad5 phosphorylation by BMP-7 led to increased podocyte survival, and BMP-7 treatment also restored synaptopodin and podocin expression (De Petris et al. 2007; Mitu et al. 2007). The loss of nephrin and synaptopodin we observed is consistent with previous studies of experimental and human diabetic nephropathy (Doublier et al. 2003; Benigni et al. 2004; Toyoda et al. 2004; Menne et al. 2006; Wang et al. 2006; De Petris et al. 2007; Wang et al. 2007). To this we add that in human diabetic nephropathy, loss of pSmad1/5/8 occurs in podocytes together with loss of the differentiation markers nephrin and synaptopodin, thus illustrating the relevance of the BMP signaling pathway in this disease.
There is growing evidence that several extracellular proteins, including SOSTDC1 and CTGF, can down-regulate BMP signaling activity in the kidney. SOSTDC1 is known to be expressed in tubuli and glomeruli of normal kidney (Blish et al. 2008). The role of SOSTDC1 in human tissues is largely unknown, but the observation that SOSTDC1 antagonized the Wnt pathway and that its expression was downregulated in renal cell carcinoma indicated strong antiproliferative activity of this protein (Blish et al. 2008). USAG-1, the rodent homolog of SOSTDC1, was first described as a negative regulator of the protective effects of BMP-7 in non-diabetic experimental nephropathies (Yanagita et al. 2006). Therefore, we postulated that SOSTDC1 might be up-regulated in human diabetic nephropathy, thus contributing to the decrease of glomerular pSmad1/5/8 activity. However, although the observed colocalization of SOSTDC1 with synaptopodin underscored the possible relevance of this BMP antagonist in podocyte biology, we observed a reduction of glomerular SOSTDC1 in human diabetic nephropathy. This suggests that in this disease, other factors might be more important in downregulation of BMP signaling activity.
We show that in glomeruli of human diabetic nephropathy, CTGF is upregulated and localized in podocytes, which is in accordance with previous reports (Wahab et al. 2005; Thomson et al. 2008). Although in this study, we found no clear evidence of CTGF staining in cells other than podocytes, we cannot exclude that CTGF is also present in, e.g., mesangial cells, as shown in some other studies (Ito et al. 1998; Rupérez et al. 2003; Wahab et al. 2005). Previous studies have shown that in both human and experimental diabetic nephropathy, glomerular CTGF is strongly upregulated and that plasma CTGF, as well as urine CTGF, might serve as a relevant biomarker in the progression of human diabetic nephropathy (Ito et al. 1998; Riser et al. 2000; Adler et al. 2001, 2002; Wahab et al. 2001; Nguyen et al. 2006, 2008b; Roestenberg et al. 2006; Guha et al. 2007; Yokoi et al. 2008). Exogenous recombinant human CTGF reduced pSmad1/5/8 level in the renal cortex, indicating that CTGF level significantly affects renal BMP signaling activity (Nguyen et al. 2008a). Moreover, inhibition of CTGF attenuated experimental diabetic nephropathy in at least three independent and fundamentally distinct studies involving neutralizing anti-CTGF antibodies, CTGF antisense oligonucleotides, and genetic deletion of one CTGF allele (CTGF+/−) (Flyvbjerg et al. 2004; Guha et al. 2007; Nguyen et al. 2008a; Yokoi et al. 2008). In a previous study, we observed a decrease of pSmad1/5/8 abundance in diabetic CTGF+/+ as compared with CTGF+/− mice in Western blots of total renal cortex (Nguyen et al. 2008a). The observation in the present study that nuclear pSmad1/5/8 staining is preserved in podocytes of diabetic CTGF+/− mice underscores the important role of CTGF in the loss of BMP signaling activity in the glomerulus. Apparently, the increase of BMP inhibition by CTGF exceeds the decrease of BMP inhibition by SOSTDC1.
We conclude that in addition to the previously observed downregulation of BMP-7 and BMP-receptor expression, overexpression of CTGF, rather than of SOSTDC1, might be an important determinant of loss of BMP signaling and of decreased podocyte number and differentiation in human diabetic nephropathy.
Footnotes
Acknowledgements
T.T. was supported by a European Renal Association–European Dialysis and Transplant Association research grant, J.W.L. was supported by a grant (C05.2144) from the Dutch Kidney Foundation, and T.Q.N. was supported by a Mozaïek grant (017.003.037) from the Dutch Organization for Scientific Research. S.V.T. was supported in part by Grant R21 CA-119181 from the National Institutes of Health and a gift from the Ben Mynatt family. R.G. has received research grants and consultancy fees from FibroGen, Inc.
The authors thank Dionne van der Giezen and Christine van Altena for technical assistance.
