Abstract
Summary
We previously demonstrated that among various histological types of human testicular germinal cell tumors (GCTs), embryonal carcinoma (EC) preferentially expresses low-sulfated keratan sulfate (KS) consisting of repeating N-acetyllactosamine (LacNAc) disaccharide units composed of galactose and 6-O-sulfated N-acetylglucosamine (GlcNAc), which is recognized by the R-10G antibody. Recently, we generated another anti-low-sulfated KS monoclonal antibody, 294-1B1. Immunohistochemical analysis of testicular GCTs (n=83) revealed that the low-sulfated KS recognized by 294-1B1 is also preferentially expressed in EC but minimally in other GCT histological types. Moreover, immunolabeling with R-10G and 294-1B1 antibodies was resistant to peptide-N-glycosidase F digestion, and EC was not stained with the MECA-79 antibody, indicating that low-sulfated KS expressed in EC contains mucin-type core 2 O-glycans carrying GlcNAc-6-O-sulfated oligo-LacNAc. Double immunofluorescence staining showed that R-10G and 294-1B1 antibody signals colocalized with those for podocalyxin (PODXL). Furthermore, western blot analysis of recombinant human PODXL•IgG fusion proteins secreted from low-sulfated KS-expressing human embryonic kidney 293T cells revealed that PODXL functions as a core protein for low-sulfated KS. Taken together, these findings strongly suggest that the PODXL glycoform decorated with low-sulfated KS is preferentially expressed in human testicular EC and may therefore serve as a diagnostic marker for this malignancy
Introduction
Keratan sulfate (KS), present in the cornea, cartilage, and central nervous system, is a member of the glycosaminoglycan family, which includes chondroitin sulfate (CS), heparan sulfate (HS), dermatan sulfate, and heparin. In general, these long linear glycosaminoglycan chains are covalently attached to core proteins to form proteoglycans. Glycosaminoglycans interact with a variety of bioactive proteins and play important roles in proteoglycan functions such as cell proliferation, cell migration, and cell adhesion. 1
KS consists of repeating N-acetyllactosamine (LacNAc) disaccharide units composed of galactose (Gal) and N-acetylglucosamine (GlcNAc), both usually 6-O-sulfated. 2 Unlike other canonical glycosaminoglycans such as CS and HS, which are covalently attached to their core proteins via a unique tetrasaccharide linker to form proteoglycans, 3 KS is attached to its core proteins via N- and/or O-glycosidic bonds to form both proteoglycans and glycoproteins. 4 Thus, short KS or sulfated oligo-LacNAc, which would not be considered genuine KS glycosaminoglycans, may be more widely present in normal and pathological human tissues as “KS glycoproteins” than was predicted. 5
As noted, both Gal and GlcNAc residues comprising KS are usually 6-O-sulfated, but KS content is heterogeneous depending on its origin. 6 In this regard, KS (or regions of that glycosaminoglycan) can be classified into highly sulfated (with both GlcNAc and Gal residues 6-O-sulfated) or low sulfated (only GlcNAc residues 6-O-sulfated) types (or regions).5,6 The 5D4 monoclonal antibody, which recognizes a linear hexa-sulfated tetra-LacNAc octasaccharide, has long been used to detect highly sulfated KS glycosaminoglycans.7,8 Almost a decade ago, Kawabe et al. established the R-10G monoclonal antibody, which binds selectively to human embryonic stem (ES) and induced pluripotent stem (iPS) cells. 9 In a follow-up study, they determined its epitope to be GlcNAc-6-O-sulfated di-LacNAc tetrasaccharide, which constitutes low-sulfated KS. 10
Kawabe et al. also revealed that the core protein of R-10G-reactive low-sulfated KS in iPS cells was podocalyxin (PODXL). 9 PODXL is a CD34-related transmembrane glycoprotein expressed on iPS cells but originally identified in podocytes surrounding glomerular capillary walls.11,12 PODXL is reportedly decorated with numerous sialylated/sulfated O-glycans to form sialomucin/sulfomucin, and the negative charge of these glycans functions to keep the urinary filtration barrier open by separating adjacent foot processes. 13 PODXL is also expressed on vascular endothelial cells and functions as an L-selectin ligand when decorated with sialyl 6-sulfo Lewis x oligosaccharide. 14
We previously demonstrated that, in addition to ES and iPS cells, human testicular embryonal carcinoma (EC) cells also express R-10G-reactive low-sulfated KS 15 but did not determine the glycostructure of R-10G-reactive low-sulfated KS expressed in EC cells. Recently, we also generated a new anti-low-sulfated KS monoclonal antibody 294-1B1, whose epitope is at least two, and possibly three or more, tandem GlcNAc-6-O-sulfated LacNAc units. 6 The epitopes of R-10G and 294-1B1 antibodies overlap and their reactivity to low-sulfated KS is similar. Thus, we thought that 294-1B1 could be used to corroborate expression of low-sulfated KS in EC cells.
In the present study, we performed immunohistochemical analysis of testicular EC with 294-1B1 antibody and a series of endoglycosidases and revealed that the low-sulfated KS expressed in human testicular EC is displayed on core-2-branched mucin-type O-glycans. Because such low-sulfated KS is preferentially expressed in EC but minimally in other histological types of germinal cell tumor (GCT), 294-1B1 may serve as a diagnostic marker for EC. We also conducted western blot analysis of recombinant human PODXL fused to human IgG constant region (PODXL•IgG) and showed that PODXL functions as a scaffold protein of low-sulfated KS recognized by both R-10G and 294-1B1 antibodies.
Materials and Methods
Human Tissue Samples
Formalin-fixed, paraffin-embedded (FFPE) tissue blocks of surgical specimens of testicular GCT (n=83) containing components of EC (n=39), yolk sac tumor (n=23), teratoma (n=16), choriocarcinoma (n=5), and/or seminoma (n=42) were obtained from the pathology archives of University of Fukui Hospital and its affiliated hospitals. Pathological diagnosis of each component was re-evaluated and confirmed by a urological pathologist (A.M.), as described previously. 15 Analysis of human testicular GCT tissues was approved by the Research Ethics Committee of University of Fukui (reference number 20210003, approved on April 12, 2021).
Antibodies
The following antibodies served as primary antibodies: JCM182 (mouse IgG, undiluted; Nichirei Biosciences, Tokyo, Japan) recognizing human CD30; goat anti-human PODXL polyclonal antibody (catalog no. AF1658, diluted 1:100; R&D Systems, Minneapolis, MN); R-10G (mouse IgG, diluted 1:400; Tokyo Chemical Industry, Tokyo, Japan) recognizing low-sulfated KS9,10; 294-1B1 (mouse IgM, undiluted culture medium of hybridoma cells) recognizing low-sulfated KS 6 ; 5D4 (mouse IgG, diluted 1:100; Seikagaku Corporation, Tokyo, Japan) recognizing highly sulfated KS 7 ; and MECA-79 (rat IgM, diluted 1:100; BD Biosciences, Franklin Lakes, NJ) recognizing GlcNAc-6-O-sulfated LacNAc attached to extended core 1 O-glycans.16,17
Endoglycosidase Digestion
To cleave internal β1,4-galactosidic linkages in poly-LacNAc and low-sulfated KS, FFPE tissue sections were incubated with 50 µg/ml (dissolved in 30 mM sodium acetate, pH 5.2) of endo-β-galactosidase from Citrobacter freundii, purified as described,18,19 at 37C for 90 min in a humidified chamber. Similarly, to cleave internal β1,3-N-acetylglucosaminic linkages in KS, sections were incubated with 50 µg/ml of recombinant keratanase II from Bacillus circulans (Tokyo Chemical Industry). To remove N-glycans, sections were incubated with 5000 U/ml (dissolved in 50 mM sodium acetate, pH 6.0) of peptide-N-glycosidase F (PNGase F) from Flavobacterium meningosepticum (catalog no. P0704S; New England BioLabs, Ipswich, MA).20,21
Immunohistochemistry
Immunohistochemical staining for PODXL and MECA-79 was performed using an indirect method as described.22,23 Immunohistochemical staining for other antigens was performed using the Histofine system (Nichirei Biosciences), as described. 20 Briefly, deparaffinized sections were immersed 30 min in absolute methanol containing 0.3% H2O2 to quench endogenous peroxidase activity, and then autoclaved at 105C for 15 min in 10 mM Tris/HCl buffer (pH 8.0) containing 1 mM ethylenediaminetetraacetic acid to retrieve antigens. After blocking 15 min with 1% bovine serum albumin in Tris-buffered saline (TBS), sections were incubated with primary antibodies at 4C overnight. After washing with TBS, sections were incubated 30 min with Histofine Simple Stain MAX-PO (MULTI; Nichirei Biosciences). The color reaction was developed using a Betazoid DAB Chromogen Kit (Biocare Medical, Pacheco, CA). Sections were briefly counterstained with hematoxylin. Immunostained slides were scanned using a NanoZoomer S60 (Hamamatsu Photonics, Hamamatsu, Japan) at 20× magnification in single layer mode to obtain whole slide images (WSIs), and WSIs were observed using NDP.view2 software (Hamamatsu Photonics). In each GCT component, an Allred score was determined as described. 24 Briefly, first, a proportion score representing the fraction of stained cells among tumor cells was assigned (0, none; 1, less than one-hundredth; 2, one-hundredth to one-tenth; 3, one-tenth to one-third; 4, one-third to two-thirds; 5, greater than two-thirds). An intensity score representing the average signal intensity of stained cells was then assigned (0, none; 1, weak; 2, intermediate; 3, strong). The overall expression level of the antigen of interest was expressed as the sum of the above two scores (ranging from 0 to 8), and GCT components with a score of 2 or greater were considered positive. Intensity of immunolabeling before and after endoglycosidase digestion was quantified using a previously described method. 20 Double immunofluorescence staining was performed essentially as described. 25
Construction of the PODXL•IgG Expression Vector
DNA fragments encoding the extracellular domain (amino acid residues 1–425) of human PODXL were PCR-amplified with oligonucleotides 5′-ACCCAAgCTggCTAgCCgACgACACgATgCgCTg-3′ (5′-primer; NheI site underlined) and 5′-CTCACCCTCgggATCCCggTCCTCggCCTCCTCC-3′ (3′-primer; BamHI site underlined) using human kidney first-strand cDNA (Clontech Laboratories, Mountain View, CA) as template. PCR products were inserted into pcDNA3.1/Hygro-IgG, which harbors the sequence of the human IgG constant region adjacent to the multiple cloning site, 23 using In-Fusion Snap Assembly Master Mix (Takara Bio, Kusatsu, Japan) according to the manufacturer’s instructions, resulting in pcDNA3.1/Hygro-PODXL•IgG.
Expression of PODXL•IgG Fusion Proteins
Human embryonic kidney (HEK) 293T cells, which endogenously express glycosyltransferases functioning in biosynthesis of core 1 and core 2 O-glycans capped by sialic acid, 26 were engineered to express low-sulfated KS. Briefly, cells were stably transfected with cDNA encoding β1,3-N-acetylglucosaminyltransferase 7 (GlcNAcT-7) and corneal GlcNAc-6-O-sulfotransferase (C-GlcNAc6ST), as described,6,27 resulting in HEK 293T/GC cells. Cells were then transiently transfected with pcDNA3.1/Hygro-PODXL•IgG (described above) and cultured in HyClone CDM4HEK293 serum-free medium (Thermo Fisher Scientific, Waltham, MA) supplemented with 2 mM L-glutamine (Thermo Fisher Scientific). Forty-eight hr later, conditioned medium was recovered.
Western Blot Analysis
PODXL•IgG fusion proteins secreted from 293T/GC and control 293T cells were purified by incubating with Protein A-Sepharose 4B Fast Flow from Staphylococcus aureus (Sigma-Aldrich, St. Louis, MO) on a rotator (at 145 rpm) at 4C for 90 min. After three washes in TBS supplemented with 0.05% Tween-20 followed by washing with distilled water, beads were suspended in sample buffer. After incubating 2 min at 100C, samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto a polyvinylidene difluoride membrane. After blocking with 5% skim milk in TBS for 60 min, the membrane was incubated with R-10G or 294-1B1 at 4C overnight. After washing with TBS supplemented with 0.05% Tween-20, the membrane was incubated with horseradish peroxidase (HRP)-conjugated anti-mouse IgG (for R-10G) or anti-mouse IgM (for 294-1B1) (Jackson ImmunoResearch, West Grove, PA). To detect PODXL•IgG, the membrane was incubated directly with HRP-conjugated anti-human IgG (Jackson ImmunoResearch) without primary antibodies. Immunoreactive bands were visualized using SuperSignal West Dura Extended Duration Substrate (Thermo Fisher Scientific) and a luminescent image analyzer LAS-4000 (GE Healthcare, Chicago, IL).
Statistical Analysis
Differences between groups were statistically analyzed by one-way analysis of variance with a Tukey–Kramer post hoc test using GraphPad Prism 7 software (GraphPad Software, San Diego, CA). The sensitivity and specificity of the antibodies to detect EC tissue were determined by Fisher’s exact test using JMP Pro 17 software (SAS Institute, Cary, NC). p values of less than 0.05 were considered statistically significant.
Results
Human Testicular EC Cells Express Low-sulfated KS
As shown in Fig. 1A (left panel), EC tissue is composed of primitive large pleomorphic epithelial cells with vesicular nuclei and conspicuous nucleoli, forming solid tumor cell nests with cribriform glands. The plasma membrane of tumor cells was CD30-positive (Fig. 1A, middle panel) and the luminal surface of glandular structures was PODXL-positive (Fig. 1A, right panel), consistent with a previous report. 28 EC cells were also R-10G-positive, and the luminal surface of glandular structures was particularly intensely stained (Fig. 1B, left panel), as reported. 15 To corroborate that those signals originated from low-sulfated KS, we performed immunohistochemical staining with 294-1B1, a different anti-low-sulfated KS monoclonal antibody that we recently generated. 6 As shown in Fig. 1B (middle panel), the 294-1B1 staining pattern was similar to that of R-10G, although signal intensity was slightly weaker. Importantly, R-10G- and 294-1B1-positive EC cells were completely devoid of immunolabeling with 5D4, which recognizes highly sulfated KS (Fig. 1B, right panel). These results overall indicate that human testicular EC cells exclusively express low-sulfated KS.
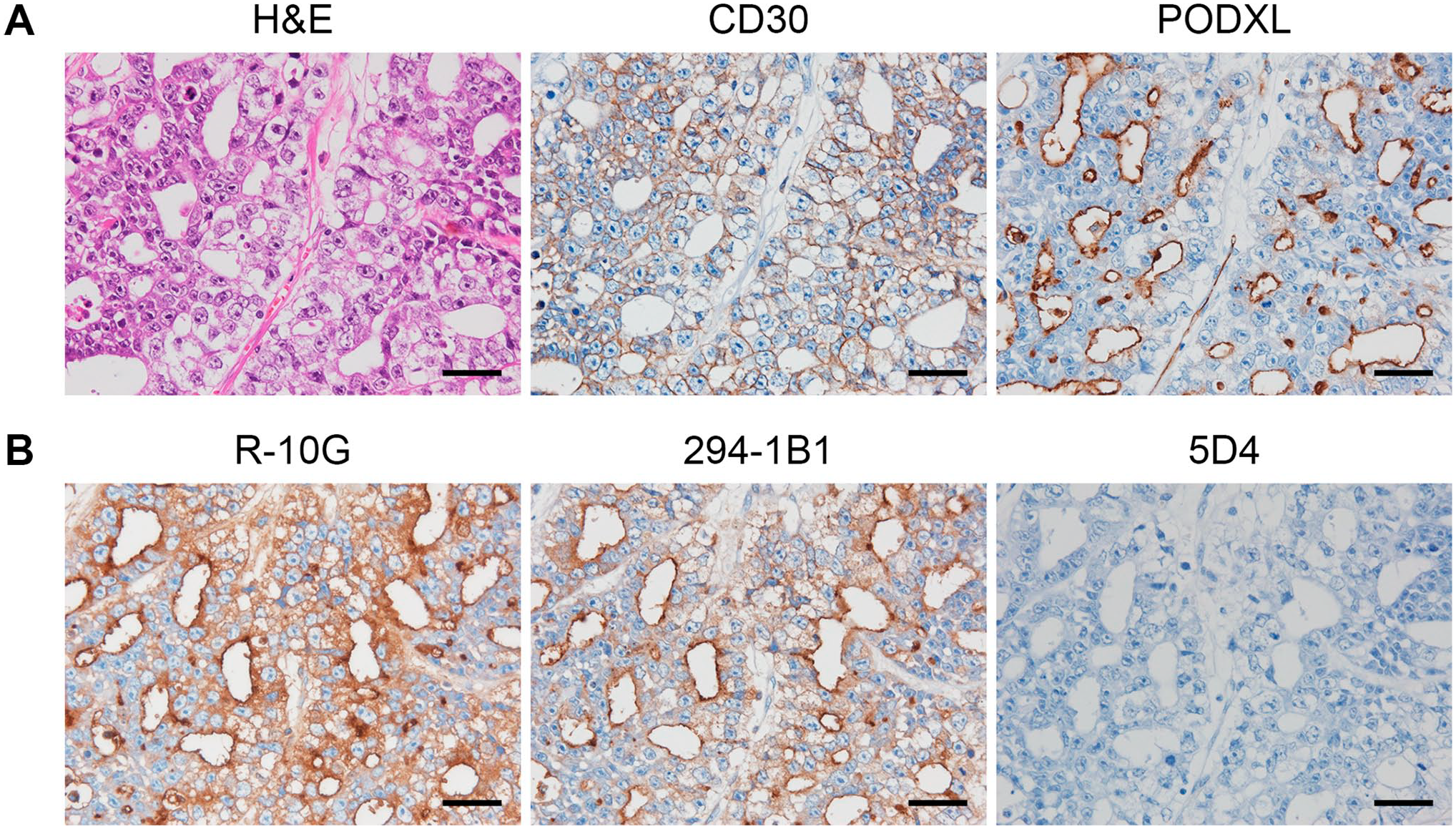
Expression of low-sulfated keratan sulfate (KS) in a representative case of human testicular embryonal carcinoma (EC). Histology of testicular EC and expression of EC markers. Serial tissue sections of testicular EC were stained with hematoxylin and eosin (H&E) or immunostained for CD30 or podocalyxin (PODXL; Panel A). Immunoreactivities of testicular EC to a series of anti-KS monoclonal antibodies (Panel B). Serial tissue sections were immunostained for R-10G, 294-1B1, or 5D4. In Panels A and B, signals were visualized with 3,3′-diaminobenzidine (brown) and tissues were counterstained with hematoxylin. Bar = 50 µm.
PODXL Is Expressed Primarily in EC and to Some Extent in Yolk Sac Tumors and Teratomas
As noted, PODXL is expressed on the apical surface of glandular structures formed in EC. We next determined whether PODXL is expressed exclusively in EC or also expressed in other histological types of GCT. As shown in Fig. 2 (second column from left), in yolk sac tumors, tumor cells forming a honeycomb-like meshwork (arrows) as well as those comprising Schiller–Duval bodies (arrowheads) were PODXL-positive. In teratomas, the luminal aspect of neural tubes, which are immature neuroectodermal structures recapitulating those seen in the early embryonic central nervous system (asterisks), was PODXL-positive. Mature glandular structures similar to those seen in the gastrointestinal tract were also occasionally PODXL-positive (data not shown). On the other hand, cytotrophoblasts and syncytiotrophoblasts comprising choriocarcinoma were completely devoid of PODXL immunolabeling, and in seminoma, only occasional small dot-like staining was observed in very limited areas. These findings indicate that PODXL is expressed primarily in EC and to some extent in yolk sac tumors and teratomas but not in choriocarcinoma and seminoma.
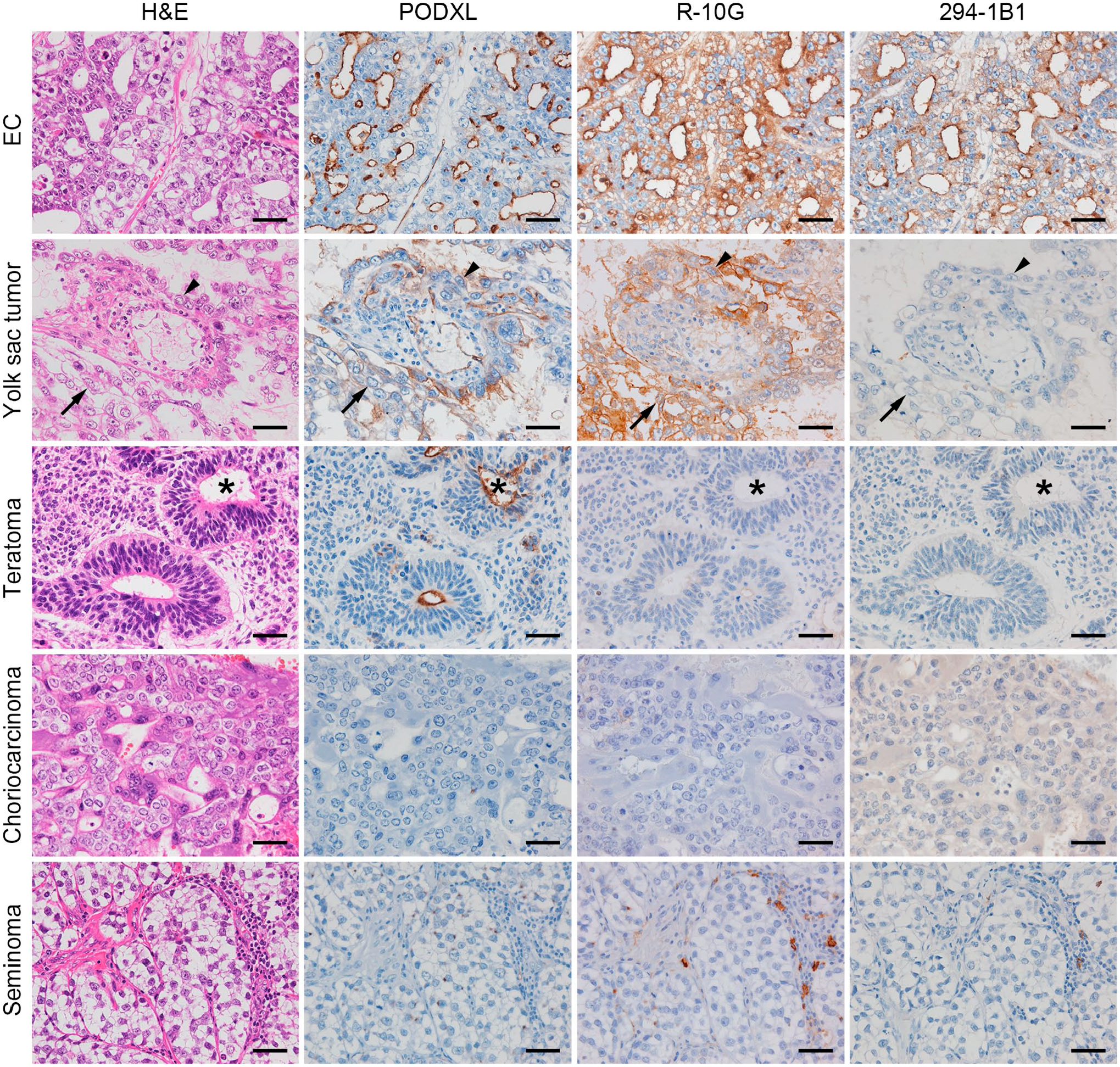
Characteristic histology and expression patterns of podocalyxin (PODXL) and low-sulfated keratan sulfate (KS) in representative cases of various histological types of testicular germ cell tumor, namely, embryonal carcinoma (EC), yolk sac tumor, teratoma, choriocarcinoma, and seminoma. Serial tissue sections were stained with hematoxylin and eosin (H&E) or immunostained for PODXL, R-10G, or 294-1B1. Signals were visualized with 3,3′-diaminobenzidine (brown) and tissues were counterstained with hematoxylin. Photomicrographs of EC in the top row are the same as their counterparts shown in Fig. 1. Arrows and arrowheads in the second row (yolk sac tumors) indicate tumor cells forming a honeycomb-like meshwork and those comprising Schiller–Duval bodies, respectively, and asterisks in the third row indicate neural tubes. Bar = 50 µm.
294-1B1 May Be a Diagnostic Marker for EC, Comparable to CD30
Consistent with our previous report, 15 R-10G-reactive low-sulfated KS was expressed not only in EC but also in yolk sac tumors and in a fraction of lymphocytes comprising the lymphoid stroma of seminoma (Fig. 2, third column from left). On the other hand, 294-1B1-reactive low-sulfated KS was preferentially expressed in EC and minimally in other histological types of GCT (Fig. 2, right column), suggesting that 294-1B1 could be a diagnostic marker for EC. To confirm this possibility, we determined an Allred score for 294-1B1 staining for each of the 125 various GCT components and compared median scores for EC, non-EC non-seminomatous GCT (NSGCT), and seminoma. As shown in Fig. 3A, the median Allred score in EC (=5) was greater than that seen in non-EC NSGCT (=0) and seminoma (=0) with high statistical significance (both p<0.001). These findings are comparable to those for the established EC marker CD30, with a median Allred score in EC (=7) significantly greater than that seen in both non-EC NSGCT (=0) and seminoma (=0; both p<0.001; Fig. 3B). As for R-10G, because the median Allred score in EC (=7) was significantly greater than that seen in non-EC NSGCT (=4) and seminoma (=4; both p<0.001), a substantial number of cases with non-EC NSGCT as well as seminoma showed rather high Allred scores (Fig. 3C).
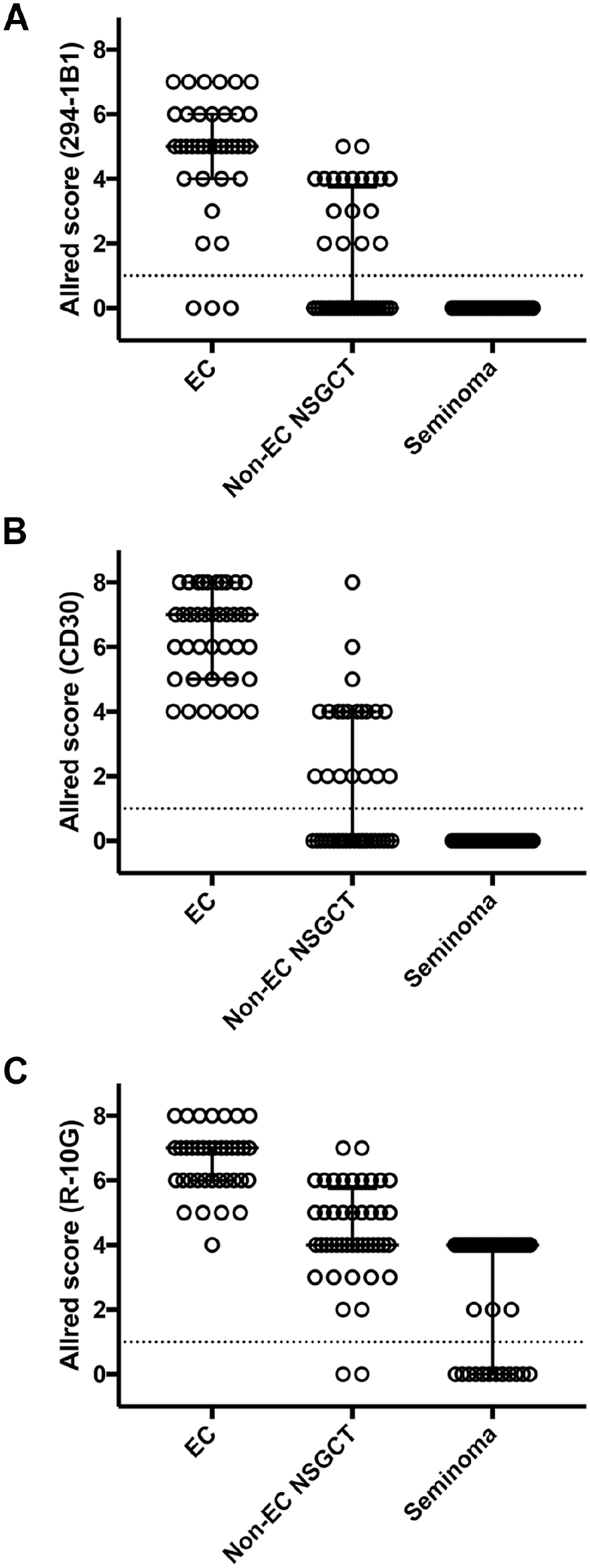
Scatter plots showing the Allred score for 294-1B1 (Panel A), CD30 (Panel B), and R-10G (Panel C) immunohistochemistry in embryonal carcinoma (EC), non-EC non-seminomatous germ cell tumor (non-EC NSGCT), and seminoma. Median and interquartile ranges are overlaid on plots for each histological type. Dotted line at 1 indicates the threshold between positive and negative cases. In Panels A, B, and C, p-values were less than 0.001 for all combinations of EC, non-EC NSGCT, and seminoma.
We then determined the sensitivity and specificity of the above three antibodies to detect EC. The results showed that the sensitivity of 294-1B1, CD30, and R-10G were 95.8%, 100%, and 100%, respectively. On the other hand, the specificity of 294-1B1, CD30, and R-10G were 66.7%, 66.1%, and 35.8%, respectively, indicating that the specificity of 294-1B1 is higher than that of R-10G and comparable to that of CD30, a widely used diagnostic marker for EC. Taken together, these findings confirm that 294-1B1 could serve as a diagnostic marker for EC comparable to CD30.
Low-sulfated KS Expressed in EC Is Displayed on Core 2-Branched Mucin-Type O-Glycans
To define the glycan structure of low-sulfated KS expressed in testicular EC, we treated sections with a series of endoglycosidases, including KS-degrading enzymes (Fig. 4), before immunostaining for R-10G and 294-1B1. As shown in Fig. 5A (second column from left) and Fig. 6, digestion with endo-β-galactosidase completely eliminated immunoreactivity of both R-10G and 294-1B1 antibodies, indicating that low-sulfated KS recognized by these two antibodies contains endo-β-galactosidase-sensitive glycosequences, namely, di-LacNAc tetrasaccharide without Gal-6-O-sulfation. On the other hand, whereas both R-10G and 294-1B1 signals quantified by densitometry were significantly reduced by keratanase II digestion (p=0.005 for R-10G and p<0.001 for 294-1B1), a large proportion of R-10G and 294-1B1 immunolabeling (~80% and ~70%, respectively) was resistant to keratanase II digestion (Fig. 5A, third column from left and Fig. 6), strongly suggesting that low-sulfated KS recognized by these two antibodies is not genuine KS glycosaminoglycans but is likely short low-sulfated KS or GlcNAc-6-O-sulfated oligo-LacNAc. Moreover, both R-10G immunolabeling and 294-1B1 immunolabeling were resistant to PNGase F digestion (Fig. 5A, right column and Fig. 6), indicating that low-sulfated KS recognized by R-10G and 294-1B1 antibodies is displayed on O- rather than N-glycans. O-glycans with LacNAc-containing structures exist as either extended core 1- (Fig. 5B, left structure) or core 2-branched O-glycans (Fig. 5B, right structure). Here, testicular EC cells were negative for MECA-79 (Fig. 5C), which recognizes GlcNAc-6-O-sulfated LacNAc attached to extended core 1 O-glycans (see Fig. 5B, left). 17 These findings overall indicate that the low-sulfated KS expressed in testicular EC is likely GlcNAc-6-O-sulfated oligo-LacNAc attached to core 2-branched mucin-type O-glycans (Fig. 5D).
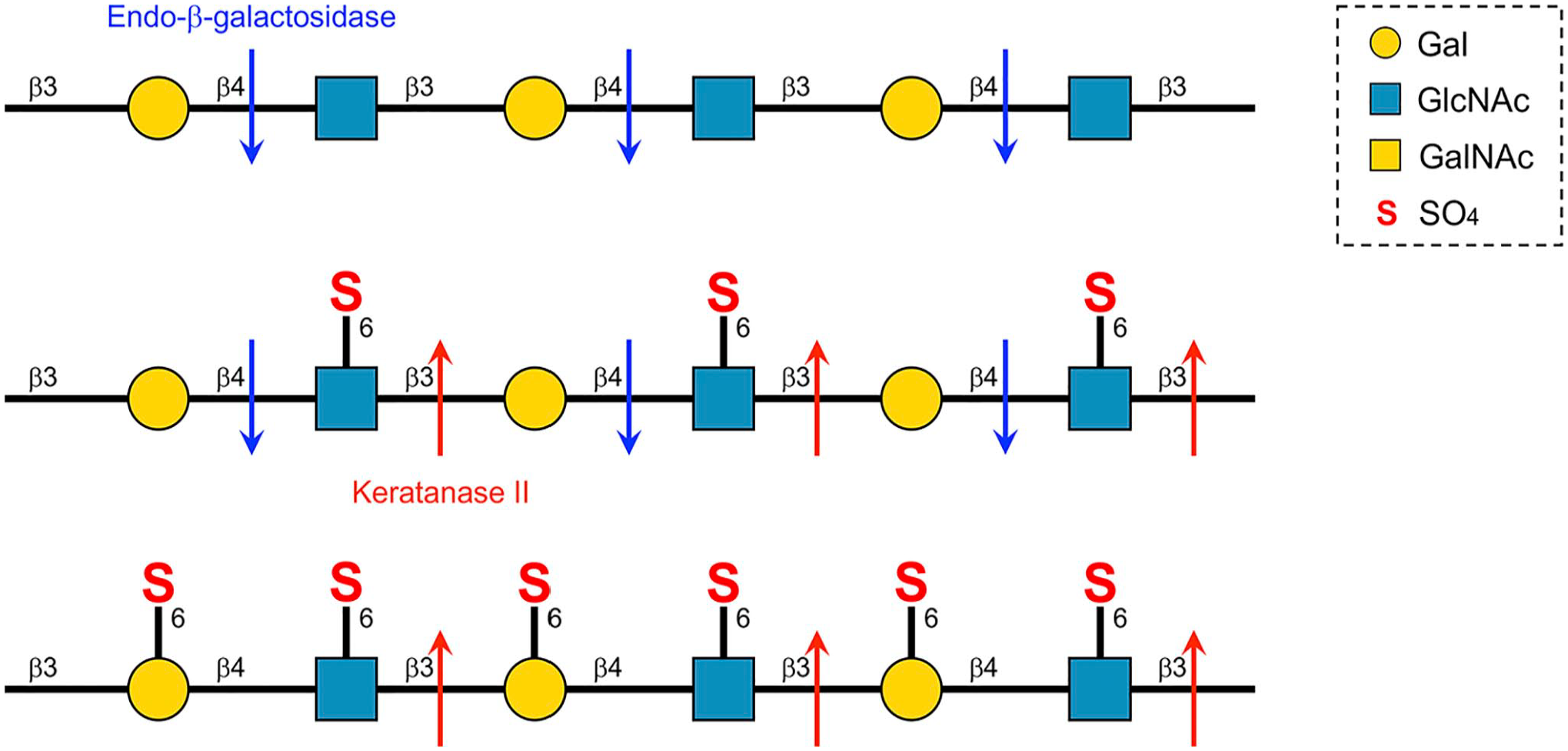
Schematic representation of the specificity of endo-β-galactosidase (blue arrows) and keratanase II (red arrows) for keratan sulfate (KS) with different sulfation modifications, that is poly-N-acetyllactosamine (so-called unsulfated KS; top structure), low-sulfated KS (middle structure), and highly sulfated KS (bottom structure). Arrows indicate that the enzyme cleaves glycosidic bonds.
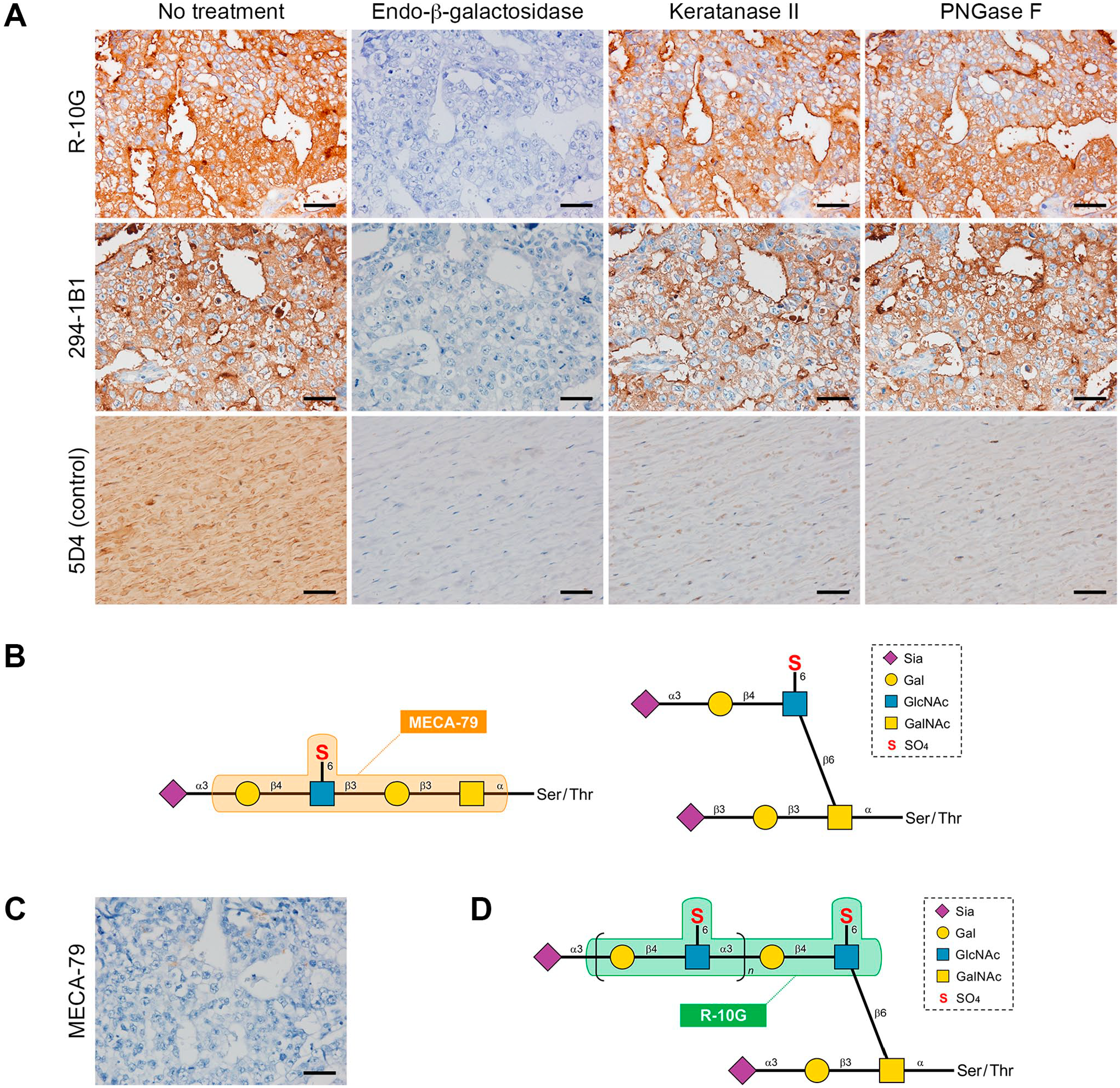
Glycoanalysis of low-sulfated keratan sulfate (KS) expressed in human testicular embryonal carcinoma (EC). Susceptibility of low-sulfated KS to a series of endoglycosidases (Panel A). Serial tissue sections of testicular EC were treated with or without (no treatment) indicated endoglycosidases and then immunostained with R-10G (top row) or 294-1B1 (middle row) antibodies. As a control (bottom row), porcine corneal tissue sections were similarly treated and stained with 5D4 antibody. Schematic diagram of sialylated N-acetylglucosamine (GlcNAc)-6-O-sulfated N-acetyllactosamine (LacNAc) attached to extended core 1- (left) or core 2-branched O-glycans (right). Orange box indicates the epitope of the MECA-79 antibody (Panel B). Immunohistochemical staining of testicular EC with MECA-79 antibody (Panel C). Schematic of the putative carbohydrate structure of low-sulfated KS expressed in testicular EC (Panel D). GlcNAc-6-O-sulfated oligo-LacNAc is displayed on mucin-type core 2-branched O-glycans via the 6-arm of GalNAc, and presumably capped by sialic acid. Green box indicates R-10G antibody epitopes. The 294-1B1 epitope is presumed to be a slightly longer GlcNAc-6-O-sulfated oligo-LacNAc containing the R-10G epitope (ref.6). In Panels A and C, signals were visualized with 3,3′-diaminobenzidine (brown) and tissues were counterstained with hematoxylin. Bar = 50 µm. Abbreviation: PNGase F, peptide-N-glycosidase F.
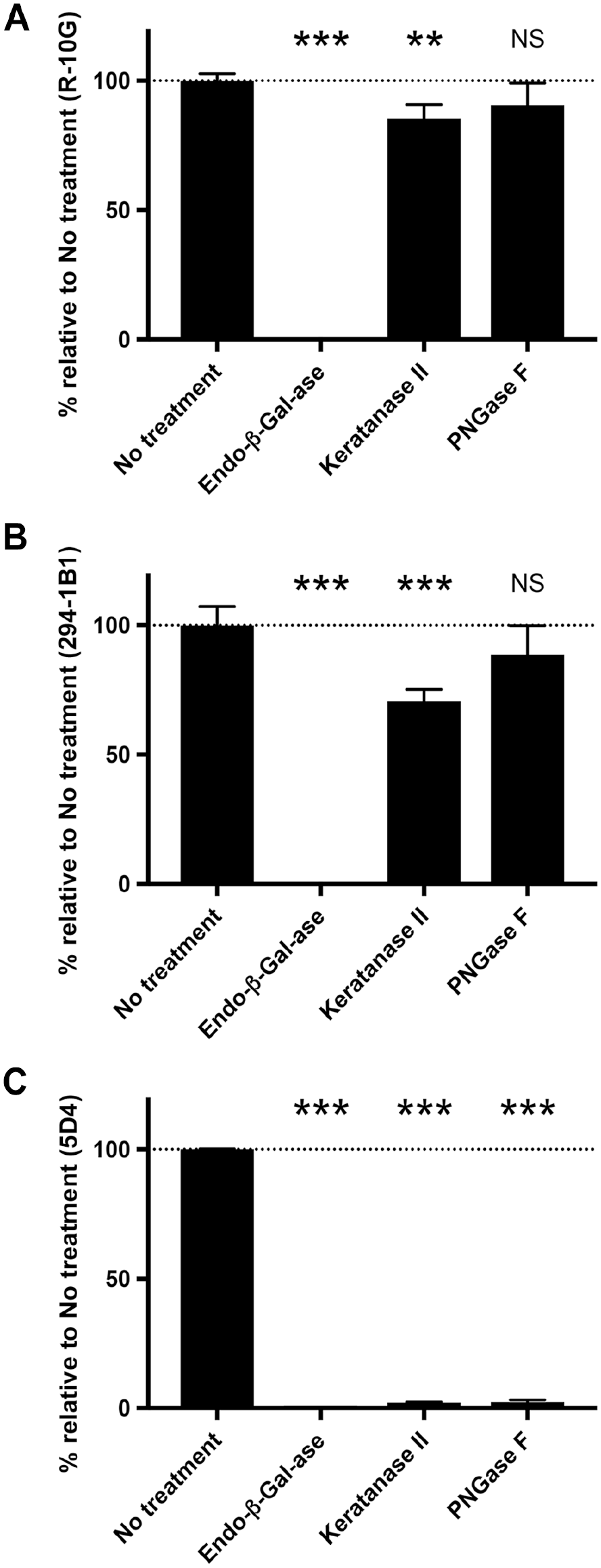
Quantification of signal intensity of R-10G (Panel A), 294-1B1 (Panel B), and control 5D4 (Panel C) immunohistochemistry in the absence (no treatment) or presence of indicated endoglycosidase pretreatments shown in Fig. 5A. Data are presented as means with SD of three segments of each image. The y-axis represents the percentage relative to no treatment (defined as 100% and indicated by the dotted line). Abbreviations: PNGase F, peptide-N-glycosidase F; NS, not significant versus no treatment. **p<0.01 versus no treatment, ***p<0.001 versus no treatment.
PODXL Serves as a Core Protein for Low-sulfated KS
Kawabe et al. previously showed that the amino acid sequence of several peptides obtained from trypsin-digested R-10G-positive bands matched that of PODXL, indicating that PODXL is a core protein of R-10G-positive low-sulfated KS in iPS cells. 9 To corroborate this finding, we first conducted double immunofluorescence staining for PODXL and either R-10G or 294-1B1. As shown in Fig. 7A, signals for both R-10G (upper panels) and 294-1B1 (lower panels), both of which are observed at the apical surface of EC glandular structures, colocalize with those for PODXL, confirming that PODXL is likely a core protein for low-sulfated KS recognized by both R-10G and 294-1B1 antibodies. To obtain direct evidence for this hypothesis, we conducted western blot analysis of a soluble form of PODXL (PODXL•IgG) secreted from HEK 293T/GC cells that express low-sulfated KS. 6 As shown in Fig. 7B, immunoblotting for human IgG (left two panels) resulted in a 100–300 kDa smear and a faint band migrating at 400 kDa in both control 293T and 293T/GC cells, indicating that PODXL•IgG fusion proteins are secreted from both cell lines. On the other hand, immunoblotting with R-10G (third panel from left) or 294-1B1 (right panel) revealed two apparently overlapping 200–400 and 400–600 kDa smears only in 293T/GC cells but not in parental 293T cells. These findings overall strongly suggest that PODXL serves as a core protein for low-sulfated KS recognized by both R-10G and 294-1B1 antibodies (Fig. 7C).
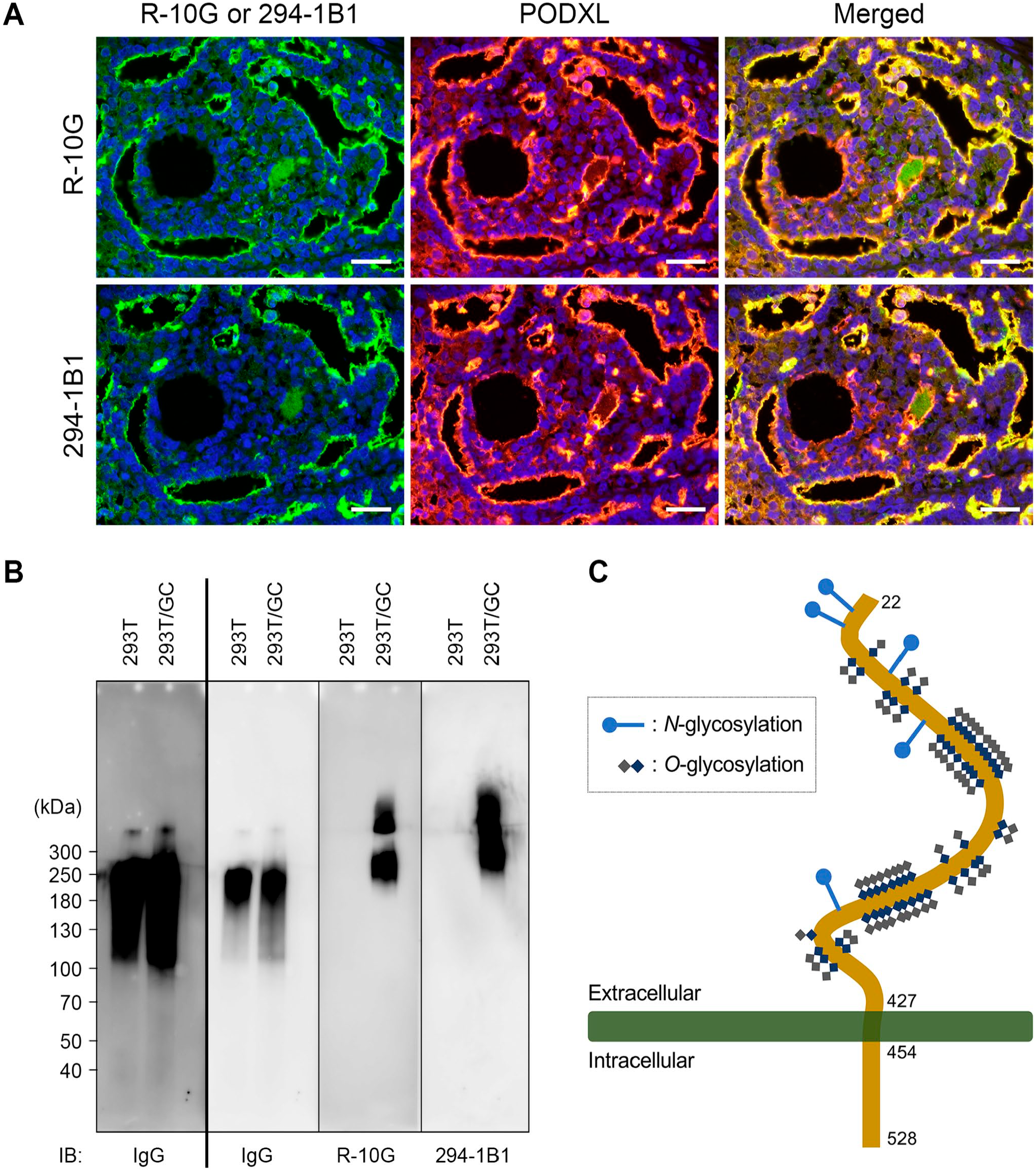
Podocalyxin (PODXL) is a core protein for low-sulfated keratan sulfate recognized by R-10G and 294-1B1 antibodies. Double immunofluorescence of testicular embryonal carcinoma for either R-10G (upper left panel) or 294-1B1 (lower left panel) (both green) and PODXL (middle panels; red) antibodies. Yellow signals in merged images (right panels) indicate antigen colocalization. Sections were counterstained with 4′,6-diamidino-2-phenylindole (blue; Panel A). Western blot analysis of PODXL•IgG fusion proteins secreted from HEK 293T/GC and parental 293T cells (controls; Panel B). Blot was probed with anti-human IgG antibody (left two panels), R-10G (third panel from left), or 294-1B1 (right panel) antibodies. The left two panels (separated by bold line) are from the same blot, but the panel at the far left was exposed longer to better detect a faint band at 400 kDa. Numbers at left indicate molecular mass (kDa). Schematic showing membrane topology of human PODXL (Panel C). Potential N- and O-glycosylation sites are shown. Bar = 50 µm. Abbreviations: IB, immunoblot; IgG, immunoglobulin G.
Discussion
Here, we demonstrate that human testicular EC cells express low-sulfated KS recognized not only by R-10G but by 294-1B1. In particular, 294-1B1-positive low-sulfated KS was preferentially expressed in EC and rarely in other histological types of GCT. A series of endoglycosidase-based glycan analyses indicated that the putative structure of such low-sulfated KS is likely core 2-branched mucin-type O-glycans carrying GlcNAc-6-O-sulfated oligo-LacNAc. We also confirmed that PODXL functions as a core protein of such low-sulfate KS.
Our findings strongly suggest that 294-1B1 could serve as a histological diagnostic marker of EC. Currently, CD30 is widely accepted as an authentic marker of EC, 29 and we previously showed that R-10G can also serve as a marker of this malignancy. 15 Thus, we evaluated 125 various GCT components for 294-1B1 or CD30 immunoreactivity as well as that of R-10G. Accordingly, median Allred scores of the all three markers in EC were significantly greater than those seen in non-EC NSGCT and seminoma, but scores for R-10G staining in non-EC NSGCT and seminoma were greater than those for 294-1B1 and CD30 (see Fig. 3). Moreover, the specificity of 294-1B1, CD30, and R-10G were 66.7%, 66.1%, and 35.8%, respectively. These results combined suggest that 294-1B1 could be a better diagnostic marker than R-10G for EC, one comparable to the established EC marker CD30.
In this study, we also found that PODXL is expressed not only in EC but also in yolk sac tumors and some components of teratoma, but not in choriocarcinoma or seminoma. Originally, PODXL was characterized as a stem cell marker expressed in testicular EC.28,30 However, it was not known whether PODXL is expressed in GCTs other than EC. Interestingly, both EC and yolk sac tumors coexpressed PODXL and R-10G-reactive sulfated glycans, and choriocarcinoma and seminoma, both of which are PODXL-negative, were also negative for both R-10G and 294-1B1. These findings suggest that PODXL is a potential core protein of R-10G-reactive low-sulfated KS in testicular EC cells.
Our western blot analysis of PODXL•IgG secreted from 293T/GC cells showed that only the high-molecular mass portion (200–600 kDa) was positive for R-10G and 294-1B1, whereas the low-molecular mass portion (100–200 kDa) seen in human IgG immunoblots was negative for both antibodies, indicating that PODXL•IgG decorated with R-10G- and 294-1B1-reactive low-sulfated KS is of higher molecular weight than PODXL•IgG not decorated with these glycans.
PODXL is translated as a 528-amino acid protein, and after removal of the 21-amino acid signal peptide, resultant PODXL protein is calculated to have a molecular mass of 54 kDa. 12 By contrast, the PODXL•IgG protein used in this study has a molecular mass of 69 kDa. Both forms of PODXL harbor five potential N-glycosylation sites, and the extracellular region (from amino acids 22 to 295) exhibits high serine/threonine content (39%), providing numerous potential O-glycosylation sites (see Fig. 7C). 12 Assuming that all potential N- and O-glycosylation sites are decorated with the putative low-sulfated KS shown in Fig. 5D (where n=1), the molecular mass of the PODXL•IgG glycoform is calculated to be 291 kDa, consistent with our western blot analysis. If the PODXL•IgG protein is decorated with putative low-sulfated KS with extended LacNAc (e.g., n=5 in Fig. 5D), its molecular mass is calculated to be 513 kDa, which is consistent with the molecular mass of the upper portion of the R-10G- and 294-1B1-reactive bands (400–600 kDa). Interestingly, however, the signal intensity of the corresponding band probed with anti-IgG antibody was much weaker (or under detection levels). A possible explanation for this finding is that the great majority of PODXL•IgG proteins secreted from 293T/GC cells are decorated with short low-sulfated KS (e.g., n=1 in Fig. 5D) and only a small fraction of PODXL•IgG proteins are decorated with low-sulfated KS with such longer extended LacNAc repeats. Moreover, whereas numerous binding sites for R-10G and 294-1B1 antibodies are present on dozens of KS side chains attached to the extracellular domain of PODXL molecule (which is also included in PODXL•IgG), only a single IgG Fc domain is present at the C-terminus of the PODXL•IgG fusion protein molecule, which may contribute to the difficulty in detecting this molecule by anti-IgG antibody compared with anti-KS antibodies.
As it was not possible to analyze fresh EC tissue in the present study, an alternative approach was to purify endogenous PODXL from EC cell lines and analyze the sulfated glycans on that protein. To this end, we first searched the Dependency Map (DepMap) portal for NSGCT lineage subtypes and identified three cell lines (1618K, NCCIT, and SuSa) that express the PODXL gene. However, for reasons unknown, these cell lines had very low, if any, expression levels of genes involved in KS biosynthesis, namely GlcNAc6STs and GlcNAcT-7. In addition, the SuSa cells were apparently of teratoma origin, 31 which is consistent with the finding shown in the present study that teratomas express the PODXL protein but were not immunolabeled with 294-1B1 and R-10G. For these reasons, we conclude that the use of these cell lines is not suitable for this study.
Here, whereas low-sulfated KS expressed in testicular EC was completely eliminated by endo-β-galactosidase digestion, it was only partially (~20–30%) digested by keratanase II (see Figs. 5A and 6). A similar result was obtained by Kawabe et al., who demonstrated that R-10G antigen isolated from human iPS cell lysates was completely eliminated by keratanase I and endo-β-galactosidase digestion but only partially by keratanase II. 9 This finding suggests that keratanase II-resistant R-10G-reactive sulfated glycans are not genuine KS glycosaminoglycans but likely short low-sulfated KS or GlcNAc-6-O-sulfated oligo-LacNAc; however, the number of LacNAc repeats remains to be determined.
Although not formally proven, the minimal keratanase II-sensitive glycan sequence is believed to be a linear di-LacNAc tetrasaccharide with GlcNAc-6-O-sulfation, namely, Galβ1-4GlcNAc(6S)β1-3Galβ1-4GlcNAc(6S), which is identical to the R-10G epitope. 10 Thus, theoretically, keratanase II digestion should completely remove the R-10G epitope. However, what if the reducing terminal of this sequence is bound to GalNAc residue comprising mucin-type O-glycans via a β1-6 linkage to form a core 2 structure? Because this linkage would create a branching structure (see Fig. 5D, where n=1), even if a keratanase II-sensitive sequence was present, it might not fit into the catalytic pocket of the enzyme, which might result in resistance to this endoglycosidase. Further studies with chemically synthesized sulfated oligosaccharides of this type are required to test this hypothesis.
Expression of KS glycosaminoglycans may be associated with tumor cell differentiation in testicular tumors. Here, we showed that yolk sac tumors are positive for R-10G but not for 294-1B1 antibodies. R-10G antibody recognizes GlcNAc-6-O-sulfated di-LacNAc,9,10 whereas 294-1B1 antibody recognizes at least two, and possibly three or more, tandem GlcNAc-6-O-sulfated LacNAc units. 6 Thus, this finding indicates that yolk sac tumors express GlcNAc-6-O-sulfated oligo-LacNAc, which is too short to be recognized by 294-1B1 antibody. Because yolk sac tumor is thought to be more differentiated than EC,32,33 it is possible that the length of oligo-LacNAc chains shortens as the tumor differentiates.
Kato et al. previously demonstrated that highly sulfated KS detected by 5D4 antibody was expressed in 68% of glioblastoma cases and 43% of anaplastic astrocytoma cases, whereas such KS was rarely detected in diffuse astrocytoma cases, suggesting that expression of highly sulfated KS is associated with astrocytoma grade and may be used as a prognostic marker. 34 It is therefore tempting to speculate that low-sulfated KS may also have a potential role in tumorigenesis and/or progression of EC. This is a topic for our future research.
Footnotes
Acknowledgements
We thank Mr. Hisataka Kato and Ms. Maiko Yamanaka for technical assistance, and Dr. Elise Lamar for critical reading of the manuscript. Part of the work was presented as a poster at the Annual Meeting of the Society for Glycobiology, held in Waikoloa, Hawaii, November 5–8, 2023.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to this article as follows: AM designed and performed the research, analyzed the data, and cowrote the manuscript; HH designed and performed the research, and analyzed the data; SI cowrote the manuscript; MM cowrote the manuscript; TOA designed and performed the research, and analyzed the data; NT organized the research team; MK conceived of and designed the research, analyzed the data, and wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid for Scientific Research (C, grant no. 22K06977; to HH) and (B, grant no. 21H02702; to MK) from the Japan Society for the Promotion of Science.
