Abstract
Thus far, several monoclonal antibodies directed against cell-surface carbohydrate antigens have been generated. Among them, R-10G reportedly reacts selectively with human embryonic stem and induced pluripotent stem cells, but not with embryonal carcinoma (EC) cells. However, EC cells derived from patients’ EC tumors may exhibit varying levels of R-10G-reactive antigen expression. Thus, we asked whether human EC tissues or germ cell tumor (GCT) tissues other than EC express R-10G-reactive antigen. To do so, we quantitatively analyzed R-10G-reactive antigen expression in 83 testicular GCT surgical specimens containing a total of 125 various GCT components. Accordingly, in all EC components examined, the EC cell plasma membrane was immunolabeled with R-10G, while most seminoma components were R-10G-negative. In non-seminomatous GCT (NSGCT) other than EC (non-EC NSGCT), R-10G-reactive antigen expression was variable, but signal distribution was focal, and the average intensity was weaker than that seen in EC. The percentages of R-10G-positive cells in these three groups varied with high statistical significance (p<0.001 for all combinations). These findings indicate that the R-10G-reactive antigen is preferentially expressed in human testicular EC tissues and, thus, could be used as a diagnostic marker for this malignancy.
Introduction
In 1974, a pluripotent cell line was established from mouse “teratocarcinoma,” a malignant tumor composed of differentiated somatic tissues and undifferentiated malignant stem cells.1,2 Cells from this line formed clumps that grew into a simple embryoid body with epithelial cells surrounding a stem cell core.3,4 Further culture of this simple embryoid body resulted in formation of a cavity containing various intermixed cell types.3,4 Cells forming this structure, called a cystic embryoid body, came to be designated embryonal carcinoma (EC) cells.
Several human EC lines have also been established, but conflicting characteristics have been reported for each.5–7 This inconsistency led to problems in defining “normal” attributes of EC cells, but with the development of embryonic stem (ES) cells,8,9 the problem has become a thing of the past, as ES cells are generally considered more suitable than EC cells for most analyses of pluripotent cells. 4 Furthermore, induced pluripotent stem (iPS) cells, which are pluripotent cells derived from adult tissues, were subsequently established. 10 Currently, most experimental analyses of pluripotent cells employ ES or iPS cells, and EC cells are now rarely used for these studies.
Several monoclonal antibodies directed against cell-surface carbohydrate antigens on human pluripotent stem cells have been developed: Among them, TRA-1-60 and TRA-1-81 have been found to recognize a type of poly-N-acetyllactosamine (poly-LacNAc) composed of repeating disaccharide units of galactose and N-acetylglucosamine (GlcNAc) and displayed on podocalyxin.11–13 These antibodies react with human ES/iPS cells as well as EC cells, suggesting that antigens recognized by these antibodies are expressed on pluripotent cells and are not specific for either ES or iPS cells.
Kawabe et al. 14 established the R-10G monoclonal antibody, which binds to a type of keratan sulfate displayed on podocalyxin expressed on iPS cells. 15 Subsequently, the R-10G epitope was determined to be GlcNAc-6-O-sulfated di-LacNAc tetrasaccharide, which constitutes low-sulfated keratan sulfate.16,17 Given that R-10G reportedly binds preferentially to ES/iPS rather than EC cells, it has been suggested that R-10G could serve as a tool to evaluate and standardize human iPS/ES cells. 14 However, we note that EC cell lines, including 2102Ep line used in the study of Kawabe et al., 14 were derived from single cells obtained from patients’ EC tissues, and it is possible that cells obtained from those tissues may show varying levels of R-10G-reactive antigen expression. Indeed, in preliminary experiments, we found that surgical tissue from a patient with testicular EC, the second most common histological subtype of testicular germ cell tumor (GCT) after seminoma, stained positively with R-10G antibody. This finding prompted us to investigate (1) whether human testicular EC tissues generally express R-10G-reactive antigen and (2) whether GCTs other than EC express R-10G-reactive antigen. We also asked whether (3) R-10G could serve as a diagnostic marker of human testicular EC, a cancer with an estimated incidence of approximately 0.2 cases per 100,000 person-years and an overall 5-year survival rate of 88%.18–20
In this article, we report that the R-10G-reactive antigen is widely expressed in human testicular EC and, to a lesser extent, in non-seminomatous GCT (NSGCT) other than EC (non-EC NSGCT) but rarely in seminoma. These findings suggest that R-10G could serve as an unanticipated marker of human testicular EC.
Materials and Methods
Human Tissue Samples
Analysis of human testicular GCT tissues was approved by the Research Ethics Committee of University of Fukui (reference number 20210003, approved on April 12, 2021). Formalin-fixed, paraffin-embedded tissue blocks of surgical specimens of testicular GCT (n=83) were obtained from the pathology archives of University of Fukui Hospital and its affiliated hospitals. Histopathological diagnosis of each specimen was re-evaluated by a urological pathologist (AM) according to the latest (5th) edition of the World Health Organization classification. 18 Consequently, the following 125 components were recognized in the entire collection of specimens: EC (n=39), yolk sac tumor (n=23), choriocarcinoma (n=5), seminoma (n=42), and teratoma (n=16).
Monoclonal Antibodies
The following monoclonal antibodies served as primary antibodies: R-10G (mouse IgG, diluted 1:400; Tokyo Chemical Industry, Tokyo, Japan) recognizing GlcNAc-6-O-sulfated di-LacNAc tetrasaccharide; JCM182 (mouse IgG, undiluted [ready to use]; Nichirei Biosciences, Tokyo, Japan) recognizing human CD30; CS-56 (mouse IgM, diluted 1:400; Sigma-Aldrich, St. Louis, MO) recognizing chondroitin sulfate; 10E4 (mouse IgM, diluted 1:400; GeneTex, Irvine, CA) recognizing heparan sulfate; BLR055F (rabbit IgG, diluted 1:200; Abcam, Cambridge, UK) recognizing human CD30; SP86 (rabbit IgG, diluted 1:100; Abcam) recognizing human glypican 3 (GPC3); and YR145 (rabbit IgG, diluted 1:400; Abcam) recognizing human KIT. The optimal antibody concentration for R-10G was determined using serially diluted antibodies in twofold series from 1:50 to 1:800. For the other antibodies, the concentrations listed in the manufacturer’s data sheets were used.
Immunohistochemistry
Conventional immunohistochemical staining for R-10G, CD30, CS-56, and 10E4 was performed using the Histofine system (Nichirei Biosciences) as described. 21 Briefly, sections were immersed 30 min in absolute methanol containing 0.3% H2O2 to quench endogenous peroxidase activity and then autoclaved at 105C for 15 min in 10-mM Tris/HCl buffer (pH 8.0) containing 1-mM ethylenediaminetetraacetic acid to retrieve antigens. After blocking 15 min with 1% bovine serum albumin in Tris-buffered saline (TBS), sections were incubated with primary antibodies at 4C overnight. After washing with TBS, sections were incubated 30 min with Histofine Simple Stain MAX PO (MULTI) (Nichirei Biosciences). The color reaction was developed using a Betazoid DAB Chromogen Kit (Biocare Medical, Pacheco, CA), and sections were briefly counterstained with hematoxylin. Negative and positive controls for R-10G staining were established using respective keratanase-II-treated and keratanase-II-untreated porcine corneal tissue (Fig. 1A). Immunostained slides were scanned using a NanoZoomer S60 (Hamamatsu Photonics, Hamamatsu, Japan) at ×20 magnification in a single-layer mode to obtain whole-slide images (WSIs), and WSIs were observed using NDP.view2 software (Hamamatsu Photonics). The percentage of immunolabeled cells among all tumor cells in each GCT component was assessed by a urological pathologist (AM) and assigned a value ranging from 0% to 100% in 10% increments. Tissues containing >5% of immunolabeled cells were considered positive. In the case of teratoma, the R-10G signal seen in differentiated cartilage was excluded from this evaluation as it is known that differentiated cartilage expresses keratan sulfate. R-10G staining intensity was quantified by the same urological pathologist (AM) using the following scoring system set up with reference to the intensity score of the Allred scoring system22,23: score 0, weakly positive cell count of ≤20%; score 1, weakly positive cell count of >20% with no strongly positive cells; score 2, strongly positive cell count of ≤80%; and score 3, strongly positive cell count of >80%. Double immunofluorescence staining for R-10G and CD30, GPC3, or KIT was performed essentially as described previously. 24
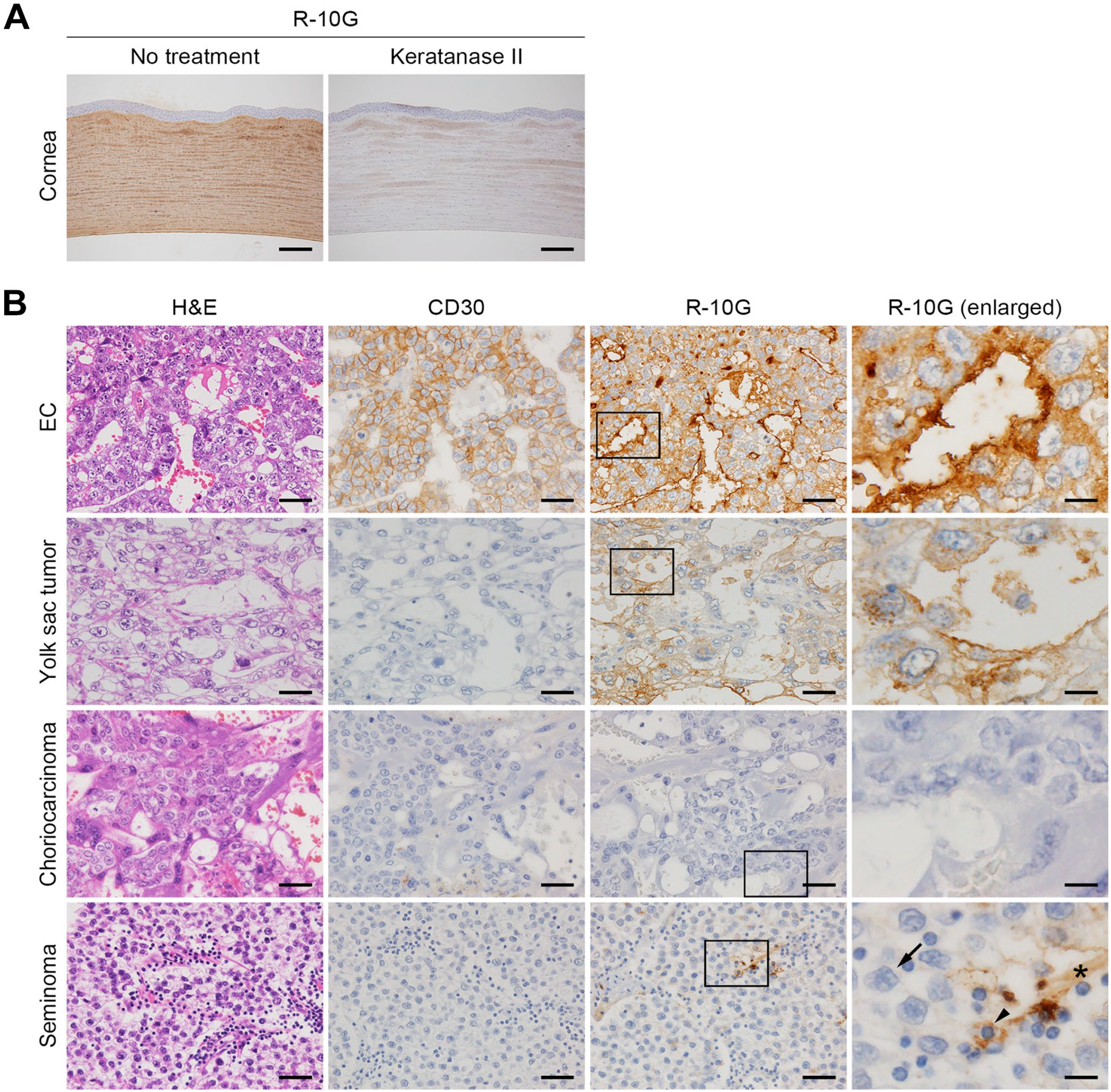
(A) Analysis of positive and negative controls for R-10G immunostaining using porcine corneal tissue rich in keratan sulfate glycosaminoglycans. Untreated (positive control) or keratanase-II-treated (negative control) sections were stained with R-10G. Signals were visualized with 3,3’-diaminobenzidine (DAB) (brown), and tissues were counterstained with hematoxylin. (B) Expression of R-10G-reactive antigen in tissues of indicated testicular GCTs, including EC, yolk sac tumor, choriocarcinoma, and seminoma. Tissue sections containing GCT components were stained with H&E (left column) or immunostained for CD30 (second column from left) or R-10G (right two columns). Boxed areas in column 3 are enlarged in adjacent images in column 4. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Representative cases are shown. In the lower right panel, arrows indicate seminoma cells, arrowheads indicate lymphocytes, and asterisks indicate fibrous septa. Bar A = 200 µm. Bar B = 50 µm for columns 1–3; 12.5 µm for column 4. Abbreviations: GCT, germ cell tumor; EC, embryonal carcinoma; H&E, hematoxylin and eosin.
Statistical Analysis
Differences in expression of R-10G-reactive antigen and CD30 among different GCT histological types were analyzed statistically by one-way analysis of variance with a Tukey–Kramer post hoc test. Differences in R-10G staining intensity between EC and yolk sac tumor were analyzed by Student’s t-test. IBM SPSS Statistics 22 (IBM, Armonk, NY) and GraphPad Prism 7 (GraphPad Software, La Jolla, CA) software were used for these analyses, and p values less than 0.05 were considered significant.
Results
Expression of R-10G-reactive Antigen in Human Testicular EC Tissue
To determine whether R-10G-reactive antigen is expressed in human testicular EC tissue, we conducted immunostaining of tissue specimens obtained from patients with testicular EC. As shown in a representative sample in Fig. 1B (top row), EC tissue was composed of undifferentiated neoplastic epithelial cells with abundant clear to granular cytoplasm, and these cells proliferated in sheets, forming gland-like structures. The plasma membrane of tumor cells stained positively for CD30, an established EC marker.18,25 Intriguingly, in all EC tissues examined, the EC cell plasma membrane, particularly the apical membrane of gland-like structures, was strongly immunolabeled with R-10G. Moreover, double immunofluorescence for R-10G and CD30 revealed colocalization of both antigens at the apical surface of gland-like structures (Fig. 2A, top row). These findings support the idea that human testicular EC tissues express R-10G-reactive antigens. We also determined whether EC tissues express glycosaminoglycans other than keratan sulfate by staining EC tissues with CS-56 antibody, which recognizes chondroitin sulfate, and with 10E4 antibody, which recognizes heparan sulfate. As shown in Fig. 2B, both CS-56 and 10E4 antibodies stained the extracellular matrix of EC tumor stroma, such as the walls of vessels (arrows), but not EC tumor cells themselves (arrowheads). This finding indicates that tumor cells in EC tissues express R-10G-reactive keratan sulfate but not other glycosaminoglycans.
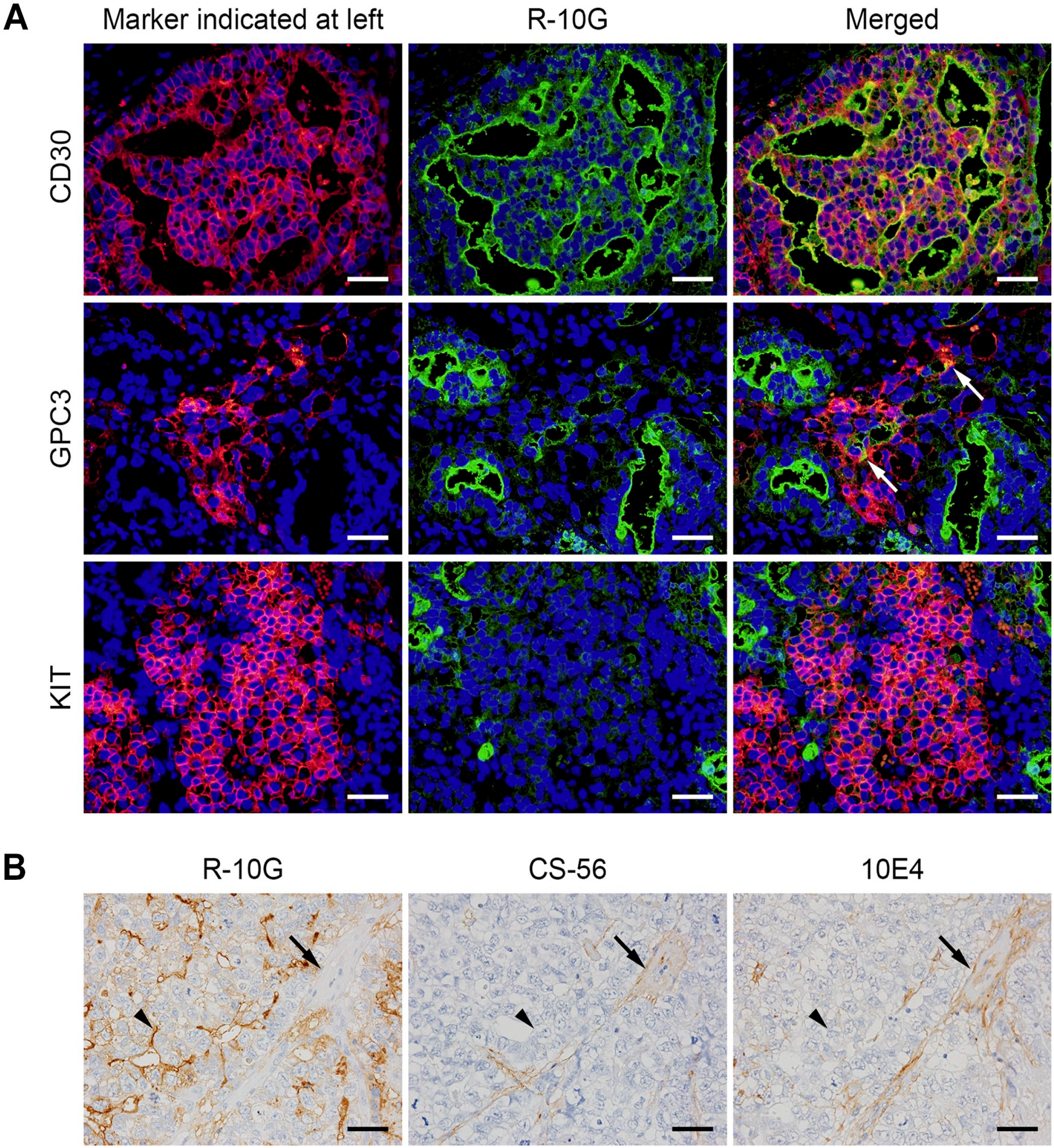
(A) Double immunofluorescence of testicular embryonal carcinoma (EC) (top row), yolk sac tumor plus EC (middle row), and seminoma plus EC (bottom row) using respective diagnostic markers (left column; red) and R-10G (middle column; green). Yellow signals in merged images (right column) indicate antigen colocalization. Tissues were stained with 4’,6-diamidino-2-phenylindole (DAPI) to mark nuclei. Arrows in GPC3 “Merged” indicate weak colocalization signals. (B) Expression patterns of glycosaminoglycans other than R-10G-reactive keratan sulfate in EC tissues. Serial EC sections were immunostained with R-10G, CS-56 (which recognizes chondroitin sulfate), or 10E4 (which recognizes heparan sulfate), as indicated. Signals were visualized with 3,3’-diaminobenzidine (DAB) (brown), and tissues were counterstained with hematoxylin. Arrows indicate vessel walls of the tumor stroma, and arrowheads indicate EC tumor cells. Bars A and B = 50 µm. GPC3, glypican 3.
GCT Subtypes Other Than EC Exhibit Weak, Focal Expression of R-10G-reactive Antigen
We next asked whether GCT subtypes other than EC express R-10G-reactive antigen. As shown in Fig. 1B (second row), a representative yolk sac tumor showing honeycomb-like reticular structures was immunolabeled with R-10G, but the signal intensity was weaker than that seen in EC. Quantitative analysis showed that the mean signal intensity score for yolk sac tumor (1.09 ± 0.49) was significantly lower than that of EC (2.23 ± 0.60; p<0.001). Moreover, double immunofluorescence revealed that only a portion of R-10G signals colocalized with signals from the yolk sac tumor marker GPC3 (arrows in Fig. 2A, middle row). 26 Choriocarcinoma, in which cytotrophoblasts and syncytiotrophoblasts proliferate against a background of hemorrhage and necrosis, was R-10G-negative (Fig. 1B, third row). Relevant to seminoma, as shown in Fig. 1B (bottom row), tumor cells with clear to pale cytoplasm and large, central nuclei exhibiting prominent nucleoli were R-10G-negative (arrows), while a small fraction of lymphocytes (arrowheads) and interspersed fibrous septa (asterisks) were weakly R-10G-positive. Consistent with this finding, double immunofluorescence staining for R-10G and KIT, the latter is a marker of seminoma, 18 did not show antigen colocalization (Fig. 2A, bottom row). Lastly, in teratoma, immature neuroectodermal structures (neural tubes) were devoid of R-10G immunolabeling (Fig. 3, top row), while mature gastrointestinal glandular structures (endoderm) were focally positive for R-10G (Fig. 3, middle row). Notably, mature cartilage (mesoderm) was R-10G-positive (Fig. 3, bottom row), consistent with the fact that cartilage tissue is rich in keratan sulfate as well as chondroitin sulfate. 27 In addition, other than EC, GCT subtypes noted earlier were devoid of CD30 immunolabeling, confirming that CD30 serves as an EC marker, as previously established.18,25 Taken together, these findings indicate that R-10G-reactive antigen is expressed in a fraction of testicular GCTs other than EC but that its expression levels are limited and lower than those seen in EC.
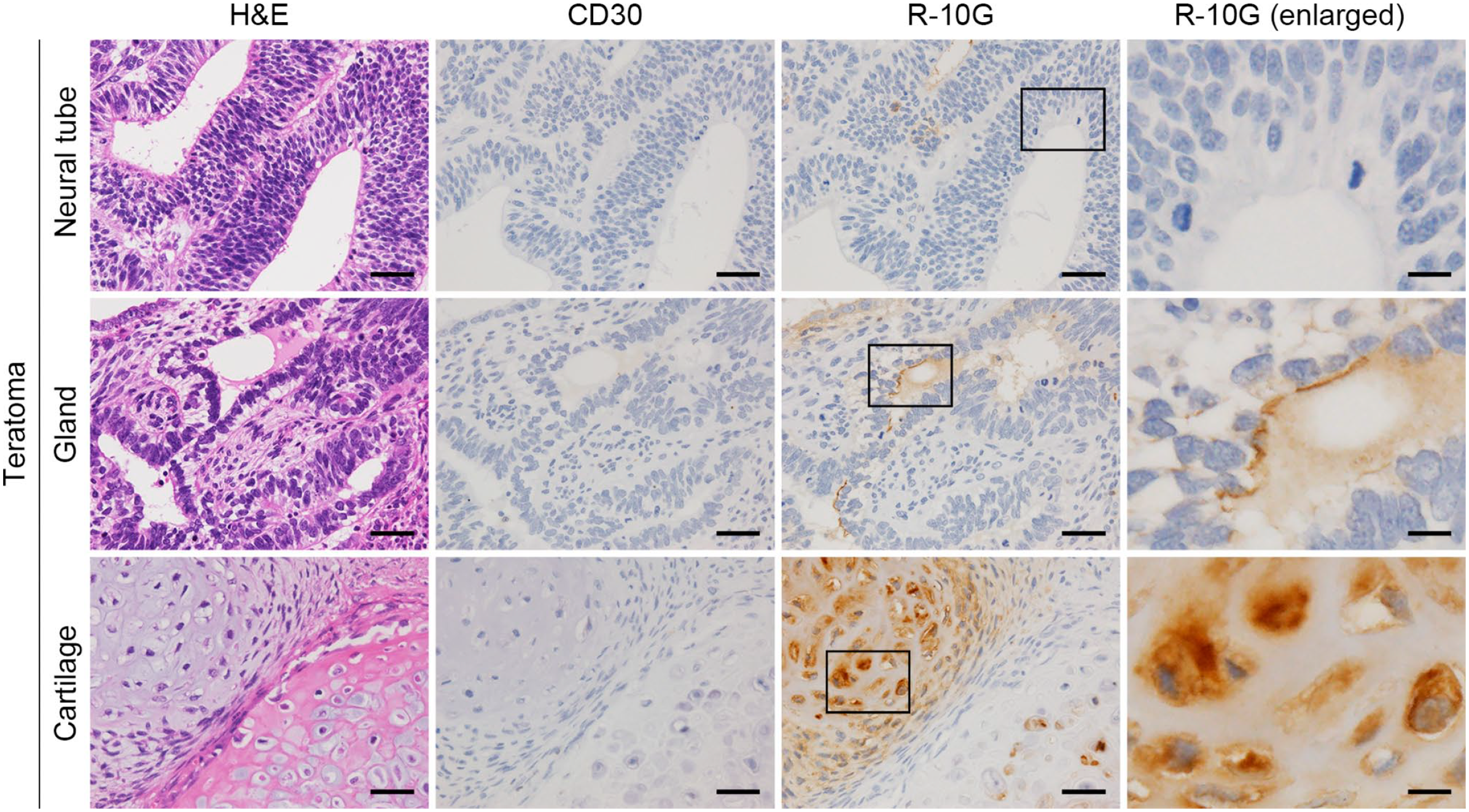
Expression of R-10G-reactive antigen in testicular teratoma. Tissue sections were stained with H&E (left column) or immunostained for CD30 or R-10G, as indicated. Images in right panels are enlarged from boxes in panels in third column from left. Signals were visualized with 3,3’-diaminobenzidine (DAB) (brown), and tissues were counterstained with hematoxylin. Shown are sections representing three germ layers (neural tube for ectoderm, gastrointestinal gland for endoderm, and cartilage for mesoderm). Bar = 50 µm for columns 1–3; 12.5 µm for column 4. Abbreviation: H&E, hematoxylin and eosin.
R-10G-reactive Antigen as a Potential Diagnostic Marker of EC
Because CD30 is widely accepted as a diagnostic marker of EC,18,25 we first evaluated differences in CD30 expression among different histological types of GCT. As shown in Fig. 4A, all 39 EC cases analyzed (100%) were immunolabeled with CD30, and the median percentage of CD30-positive cells in a tumor was 80%. Here, due to the relatively small number of cases of yolk sac tumor (n=23), choriocarcinoma (n=5), and teratoma (n=16), we combined these three histological types and classified them as non-EC NSGCT. Accordingly, while 13 of 44 (29.5%) non-EC NSGCT cases showed CD30 immunolabeling in a very small proportion of cells, the median percentage of CD30-positive cells in this group of tumors was 0%. For seminoma, none of the 42 cases (0%) were CD30-immunolabeled. Moreover, based on multiple comparisons, the percentage of CD30-positive cells in EC was significantly greater than that seen in non-EC NSGCT or seminoma (both p<0.001), but the percentage of CD30-positive cells between non-EC NSGCT and seminoma did not differ significantly (p=0.149). These results confirm that as a diagnostic marker, CD30 is capable of differentiating EC from non-EC NSGCT or seminoma.
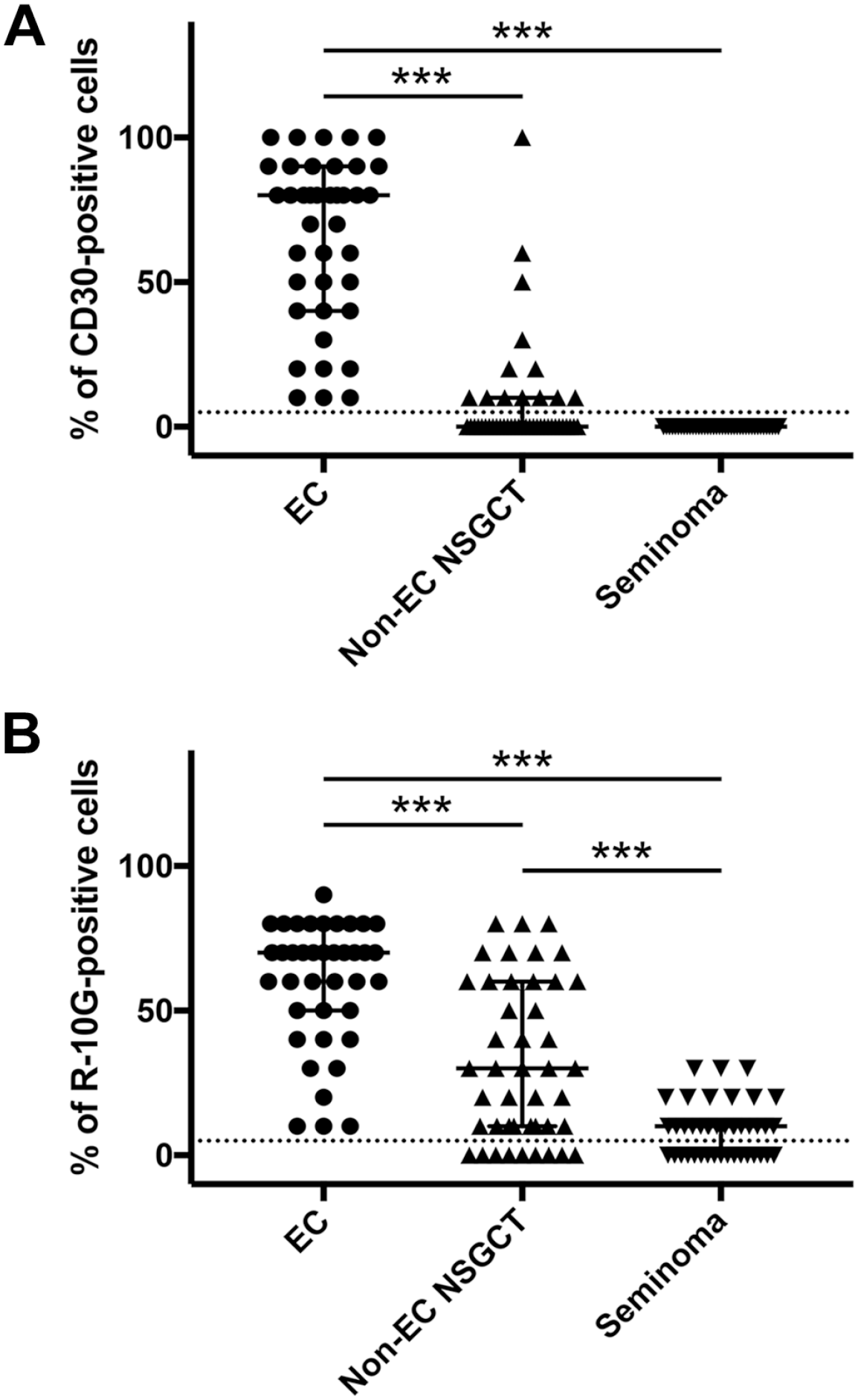
Scatter plot showing the percentage of CD30-positive (A) and R-10G-positive (B) cells in EC, non-seminomatous germ cell tumor other than EC (non-EC NSGCT) and seminoma. The percentage of CD30-positive or R-10G-positive cells was determined by dividing the number of immunolabeled cells by the total number of tumor cells. Median and interquartile ranges are overlaid on plots for each histological type. The dotted line at 5% indicates the threshold between positive and negative cases. Abbreviation: EC, embryonal carcinoma; NSGCT, non-seminomatous germ cell tumor. ***p<0.001.
Finally, we assessed differences in R-10G expression among different histological types of GCT. As shown in Fig. 4B, similar to CD30, all 39 EC cases (100%) were immunolabeled with R-10G, and the median percentage of R-10G-positive cells in a tumor was 70%. As for non-EC NSGCT, 35 of 44 (79.5%) cases were immunolabeled with R-10G, and the median percentage of R-10G-positive cells in this group of tumors was 30%. For seminoma, 25 of 42 (59.5%) cases were immunolabeled with R-10G, and the median percentage of R-10G-positive cells in a tumor was 10%. Based on multiple comparisons, the percentage of R-10G-positive cells in EC was significantly greater than that seen in non-EC NSGCT or seminoma (both p < 0.001), and the percentage of R-10G-positive cells in non-EC NSGCT was also significantly greater than that seen in seminoma (p < 0.001). These results are comparable to those of CD30, suggesting that R-10G may serve as a diagnostic marker for EC.
Discussion
In the present study, we demonstrate immunolabeling of human testicular EC tissue with R-10G antibody. Specifically, we showed that a proportion of non-EC NSGCT cases was also R-10G-positive, but signal distribution was focal, and its average intensity was weaker than that seen in EC. Seminoma was almost devoid of R-10G immunolabeling. To the best of our knowledge, this is the first report demonstrating expression of R-10G-reactive antigen in human testicular EC tissue, and our findings suggest that R-10G could serve as a histological marker of that malignancy.
Our conclusions differ to some extent from those of Kawabe et al., 14 who previously reported that the R-10G-reactive antigen is selectively expressed in human ES/iPS cells and minimally in EC cells. In establishing the antibody, that group selected iPS cell-reactive hybridoma cells, from which they excluded EC cell-reactive hybridoma cells, and used differential screening to obtain clones secreting antibodies distinguishing ES/iPS from EC cells. Only the 2102Ep EC line was used for the negative selection procedure although, in data reported as not shown, the authors reported low R-10G reactivity to the NCR-G3 EC line. 14 Moreover, analysis of a normal human tissue array containing adult and fetal samples revealed significant R-10G reactivity only in adult brain and cerebellum. 14 However, we previously reported constitutive expression of R-10G-reactive antigen in normal intrahepatic bile ducts, particularly at the apical membrane of small-sized ducts, and observed similar patterns in reactive and neoplastic counterparts.21,28 Several factors may underlie these discrepancies: For example, the tissue array analyzed by Kawabe et al. 14 may not have contained sufficient numbers of intrahepatic bile ducts. Alternatively, the R-10G antigen may be expressed focally in organs other than liver and testis, in either normal or pathological conditions. Further immunohistochemical analysis of normal and pathological human tissues is required to clarify these differences.
Although all EC tissue specimens examined here were R-10G-positive to some extent, R-10G signals were not observed in all cells found in EC tissue but were, as noted previously, more frequent in the apical membrane of EC cells that constitute gland-like structures (see Figs. 1B and 2A, both top row). This finding illustrates the importance of viewing cancer as an organized tissue, rather than a collection of tumor cells: Because EC tissues are composed of EC cells and extracellular matrix and stromal cells (including cancer-associated fibroblasts and tumor-associated macrophages), 29 stromal components may secrete humoral factor(s) or directly interact with carcinoma cells to induce apical expression of R-10G-reactive antigen.
This study indicates that as a diagnostic marker of EC, R-10G is comparable to CD30. Notably, all EC cases examined here were R-10G-positive. Thus, a key advantage of R-10G is that it may detect the rare EC cases that are CD30-negative. Also, when used in combination with CD30, analysis of R-10G positivity may make an EC diagnosis more reliable. Another advantage is that R-10G recognizes keratan sulfate. EC is rarely present in a pure form but instead is often a component of mixed GCTs that contain other GCT histological components, such as teratoma. 18 Teratomas contain cartilage, which expresses keratan sulfate (see Fig. 3, bottom row). Thus, R-10G positivity in such cartilage tissue could serve as an internal positive control in evaluating R-10G staining of EC cells within a tumor.
Fukuma et al. 25 previously generated the 4C4 monoclonal antibody directed against the human EC line NCR-G3. After biochemical analysis, they concluded that the 4C4 antigen is sialylated and fucosylated keratan sulfate, while the epitope was not completely determined. Interestingly, this antibody bound exclusively to EC and not to other types of GCT. 25 This finding is consistent with findings presented here and strongly suggests that the 4C4 epitope may overlap with the R-10G epitope. Future analysis should address the mechanism by which these sulfated glycans are preferentially expressed in EC tissue.
In conclusion, the R-10G-reactive antigen is preferentially expressed in human testicular EC tissues. Thus, paradoxically R-10G could serve as a diagnostic marker of EC.
Footnotes
Acknowledgements
We thank Dr. Mana Fukushima for useful discussion, Mr. Hisataka Kato and Ms. Maiko Yamanaka for skilled technical assistance, and Dr. Elise Lamar for critical reading of the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to this article as follows: designing the research (AM, MK), performing the research (AM, HH), analyzed data (AM, MK), writing the manuscript (AM, SI, MK), research conception (MK), organizing the research team (NT). All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Students Research from University of Fukui Life Science Innovation Center (to AM) and in part by a Grant-in-Aid for Scientific Research (B) 21H02702 from the Japan Society for the Promotion of Science (to MK).
