Abstract
The arrangement of immature germ cells changes regularly and periodically along the axis of the seminiferous tubule, and is used to describe the progression of spermatogenesis. This description is based primarily on the changes in the acrosome and the nuclear morphology of haploid spermatids. However, such criteria cannot be applied under pathological conditions with arrested spermatid differentiation. In such settings, the changes associated with the differentiation of premeiotic germ cells must be analyzed. Here, we found that the unique bipolar motor protein, KIF11 (kinesin-5/Eg5), which functions in spindle formation during mitosis and meiosis in oocytes and early embryos, is expressed in premeiotic germ cells (spermatogonia and spermatocytes). Thus, we aimed to investigate whether KIF11 could be used to describe the progression of incomplete spermatogenesis. Interestingly, KIF11 expression was barely observed in haploid spermatids and Sertoli cells. The KIF11 staining allowed us to evaluate the progression of meiotic processes, by providing the time axis of spindle formation in both normal and spermatogenesis-arrested mutant mice. Accordingly, KIF11 has the potential to serve as an excellent marker to describe spermatogenesis, even in the absence of spermatid development.
Keywords
Introduction
Mammalian spermatogenesis proceeds within seminiferous tubules in the testis (Fig. 1A). The process of spermatogenesis is strictly controlled in a spatiotemporal manner. During spermatogenesis, the differentiating germ cells simultaneously move spirally toward the center of the tubules and forward along the tubules (Fig. 1B). Consequently, the arrangements of the germ cells in the cross-sections of the seminiferous tubules change periodically (Fig. 1C). The resultant cycles in mice are subdivided into 12 stages, I–XII, which are identified primarily based upon the changes in the acrosome and the nuclear morphology of spermatids (defined as steps 1–16), 1 as illustrated in Fig. 1C. 2
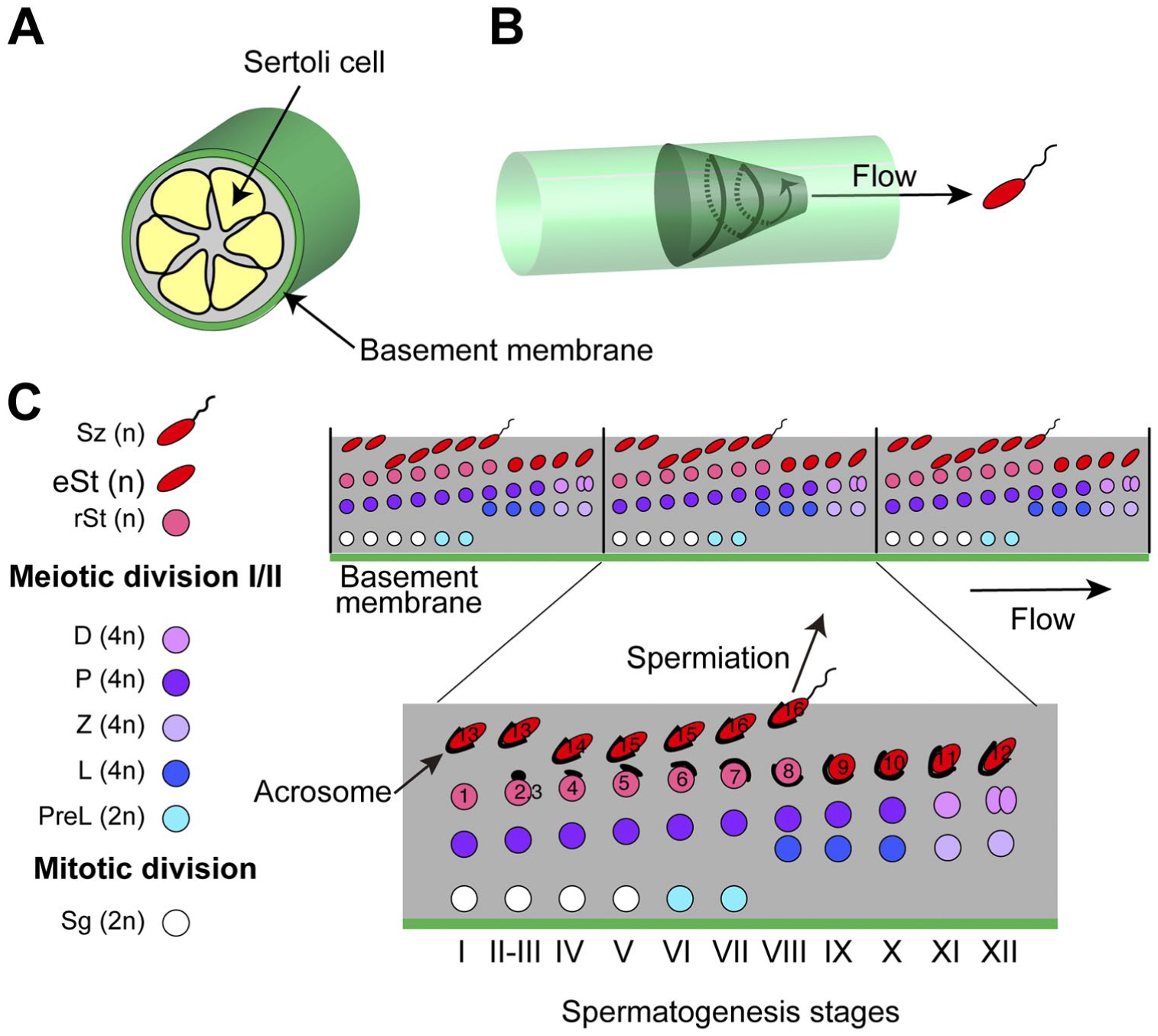
Schematic drawing of spermatogenesis stages. (A) Mammalian spermatogenesis proceeds within the tubular seminiferous epithelium, composed of somatic Sertoli cells. (B) The spiral and forward movement of a differentiating germ cell, starting from the periphery of the tubules, is depicted as a spiral arrow on a circular truncated cone. When mature sperms reach the lumen in the central region, they flow from the testis to the epididymis. (C) The initial spermatogonia are present near the basement membrane, which is a specialized form of the extracellular matrix that exists around the seminiferous tubule. The spermatogonia include stem cells, which give rise to spermatocytes after several mitotic divisions. One diploid spermatocyte goes through a meiotic division to generate four haploid round spermatids, which transform into elongated spermatids. The elongated spermatids are released as spermatozoa into the lumen of the seminiferous tubule, in a process called spermiation. Successive events of spermatogenesis lead to periodical changes in the arrangement of the germ cells along the seminiferous tubule. One of the resultant repeating units is expanded to show the stages of mouse spermatogenesis (I–XII). The developing acrosomes on spermatids are colored black. 3 Sg (2n), spermatogonia (diploid); PreL (2n), preleptotene spermatocytes (diploid); L/Z/P/D (4n), leptotene/zygotene/pachytene/diprotene spermatocytes (tetraploid); rSt (n), round spermatids (haploid); eSt (n), elongating/elongated spermatids (haploid); and Sz (n), spermatozoa (haploid). The changes in the acrosome and the nuclear morphology of spermatids are defined as steps 1–16. 1 The PNA-positive acrosomal granules (steps 2–4), acrosomal rims (steps 5, 6), and acrosomal caps (steps 7–16) are shown in black lines. Abbreviation: PNA, peanut agglutinin.
The stage-identification of each cross-section of the seminiferous tubule allows the tracking of a molecule of interest according to the time series of spermatogenesis, by an immunohistochemical analysis. Even in cases where spermatid development is incomplete, such as in some spermatogenesis-arrested mutant mice, it is very important to describe the outline of spermatogenesis. In these settings, markers capable of identifying all of the premeiotic immature germ cells in cryosections will be effective. Since mitotic and meiotic divisions take place prior to the production of spermatids, the events associated with cell division will also provide important information.
Kinesin family member 11 (KIF11/kinesin-5/Eg5) is a plus-end directed bipolar motor protein, 4 which plays a crucial role in stabilizing and separating the bipolar spindle in somatic division 5 and meiotic cell division of oocytes and early embryos.6,7 KIF11 may also play an important role in meiosis in the testis, but the expression of KIF11 during spermatogenesis has not been analyzed.
In the present study, we aimed to investigate whether KIF11 could be utilized to describe the progression of spermatogenesis. Surprisingly, we found that KIF11 is expressed in premeiotic germ cells (spermatogonia and spermatocytes), but is hardly detected in spermatids and Sertoli cells. This downregulation suggested the tight coupling between the expression of KIF11 and the meiotic events, and was supported by the analysis of the KIF11-positive spermatocytes in spermatogenesis-arrested mutant mice.
Materials and Methods
Mice
The generation of β1,4GalNAc-T-deficient mice was described previously. 8 All animal experiments were approved by the Institutional Animal Care and Use Committees of Juntendo University and Tokyo Medical and Dental University.
Immunohistochemistry
Mice (C57BL/6, 7-week old) were anesthetized and transcardially perfused with PBS-buffered 4% paraformaldehyde. The testes were further fixed with 4% paraformaldehyde, and the sections were prepared as described previously. 9 The sections were washed with PBS, blocked with blocking solution (1% bovine serum albumin in PBS), and incubated for 2 h with anti-KIF11 (1:100, Sigma HPA010568, rabbit), anti-Scp3 (1:100, Santa Cruz SC-74569, mouse), anti-LIN28A (1:100, Cell Signaling #3978, rabbit, or 1:100, Proteintech 60344-1-Ig, mouse) or anti-vimentin (1:100, PROGEN DS-041002, guinea pig) antibodies. After washing with blocking solution, the sections were incubated for 1 h with Alexa 488-, Alexa 555- or Alexa 647-conjugated secondary antibodies diluted with the blocking solution. Some sections were double-stained with Alexa 488-conjugated PNA lectin (1:200; Thermo Fisher Scientific, Tokyo, Japan) to detect developing acrosomes. Immunoreactivity was not observed when the section was stained with only the secondary antibody. Images were acquired using a Leica TCS SP8 inverted confocal microscope with an 63× oil immersion lens. Essentially, the same results were obtained in an independent experiment.
Preparation of Testicular Cells and Flow Cytometry
The testes were collected in Hank’s balanced salt solution (HBSS), and the tunica albuginea was removed. The seminiferous tubules were kept in HBSS for 10 min on ice, to exclude the Leydig cells that did not sediment. Seminiferous epithelia were treated with collagenase Type I and DNase for 30 min at 37℃, and then with 0.25% trypsin/ EDTA for 15 min at 37℃. After quenching the trypsin reaction by adding fetal bovine serum, the tubular fragments were filtered through a 40 µm cell strainer. For flow cytometry, the dispersed cells were precipitated and fixed by the high citrate fixative methods described by Mays-Hoopes et al. 10 Finally, the cells were stained with propidium iodide and analyzed by flow cytometry, using a FACSCalibur flow cytometer (BD Biosciences, Tokyo, Japan).
Results
KIF11 Is Expressed in Spermatocytes But Downregulated in Spermatids During Acrosome Development Within Seminiferous Tubules
To investigate the distribution of KIF11-expressing cells within the seminiferous tubules, we co-stained the testis sections with an anti-KIF11-antibody, an anti-Scp3-antibody, and Alexa-conjugated peanut agglutinin (PNA). Scp3 is a member of the synaptonemal complexes formed between homologous chromosomes during meiotic division,11,12 and thus Scp3 is a specific meiotic marker. PNA-lectin stains developing acrosomes on spermatids.3,13 As shown in Fig. 2, the KIF11-positive cells were located near the basement membrane in the cross-sections of different stages. In the majority of the KIF-11 positive cells, the nuclei were also stained with the anti-Scp3-antibody (among 61, 89, and 108 KIF11 positive cells, 49, 81, and 103 were Scp3 positive cells, in Fig. 2J to L, respectively). In contrast, the KIF11-positive cells did not costain with PNA-lectin, suggesting that KIF11 is expressed in spermatocytes and downregulated in spermatids with formed acrosomes.
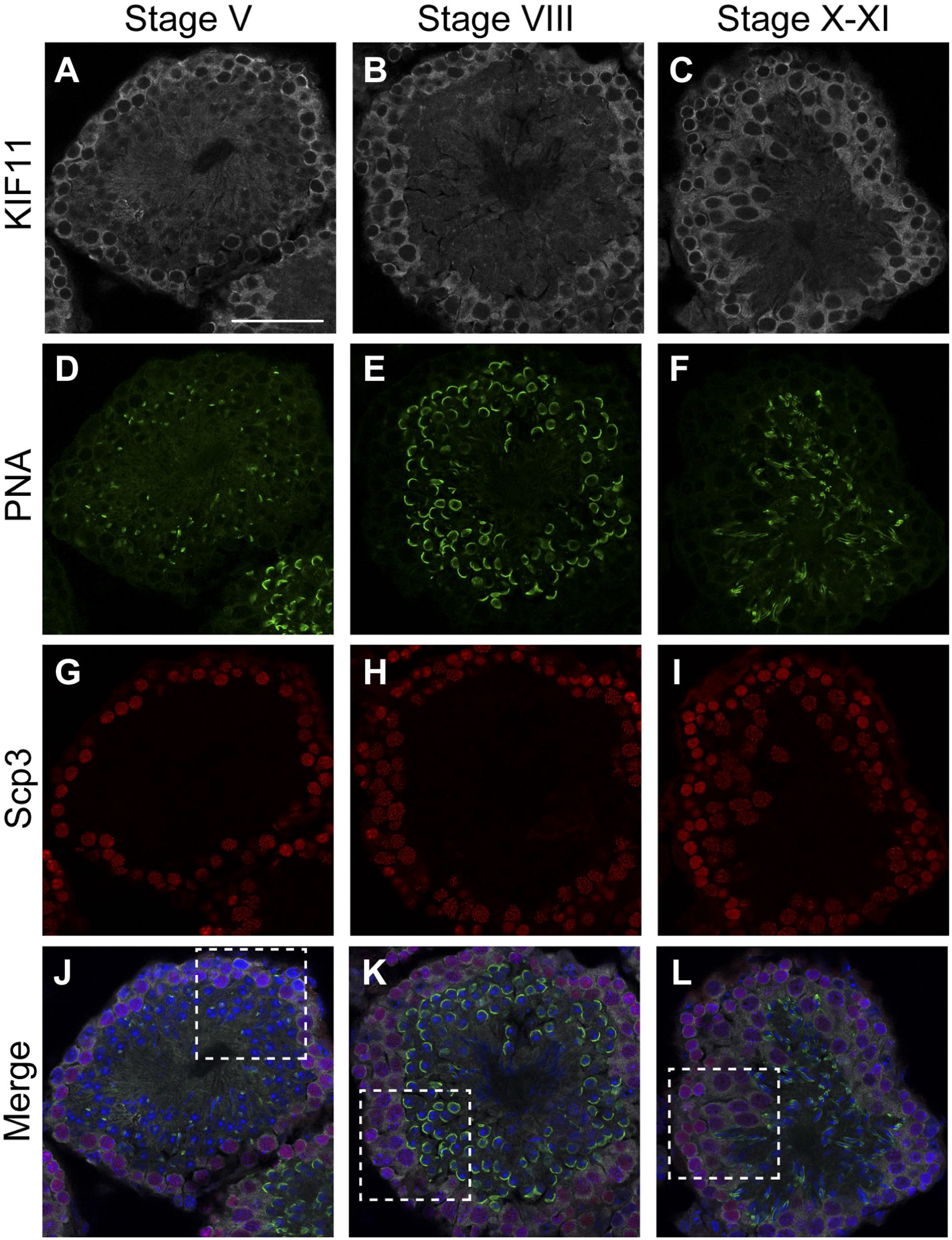
KIF-11 is expressed in spermatocytes but not in spermatids. Paraformaldehyde-fixed cryo-sections of mouse testes were triple-stained for KIF11 (gray), PNA (green), and Scp3 (red), with nuclear counterstaining using Hoechst 33342 (blue). The immunofluorescent images of KIF11 (A–C), PNA (D–F), Scp3 (G–I), and their overlays with Hoechst 33342 (J–L, respectively) in stages V (A, D, G, J), VIII (B, E, H, K), and X–XI (C, F, I, L) are shown. The areas enclosed by the dashed rectangles in J–L are enlarged in Fig. 3. Scale bar, 50 µm. Abbreviations: KIF, Kinesin family member; PNA, peanut agglutinin.
In response to the changes in the Scp3-staining within the nuclei of spermatocytes during spermatogenesis, 11 the cytoplasmic KIF11-staining also underwent alterations, likely reflecting the different cell size of spermatocytes (Fig. 3). Pachytene spermatocytes had a wider fringe of KIF11 staining than leptotene or zygotene spermatocytes. The surrounding area of KIF11 became even wider and oval-shaped in diplotene spermatids. These changes in the KIF11-staining of the spermatocytes are schematically summarized in Fig. 3J.
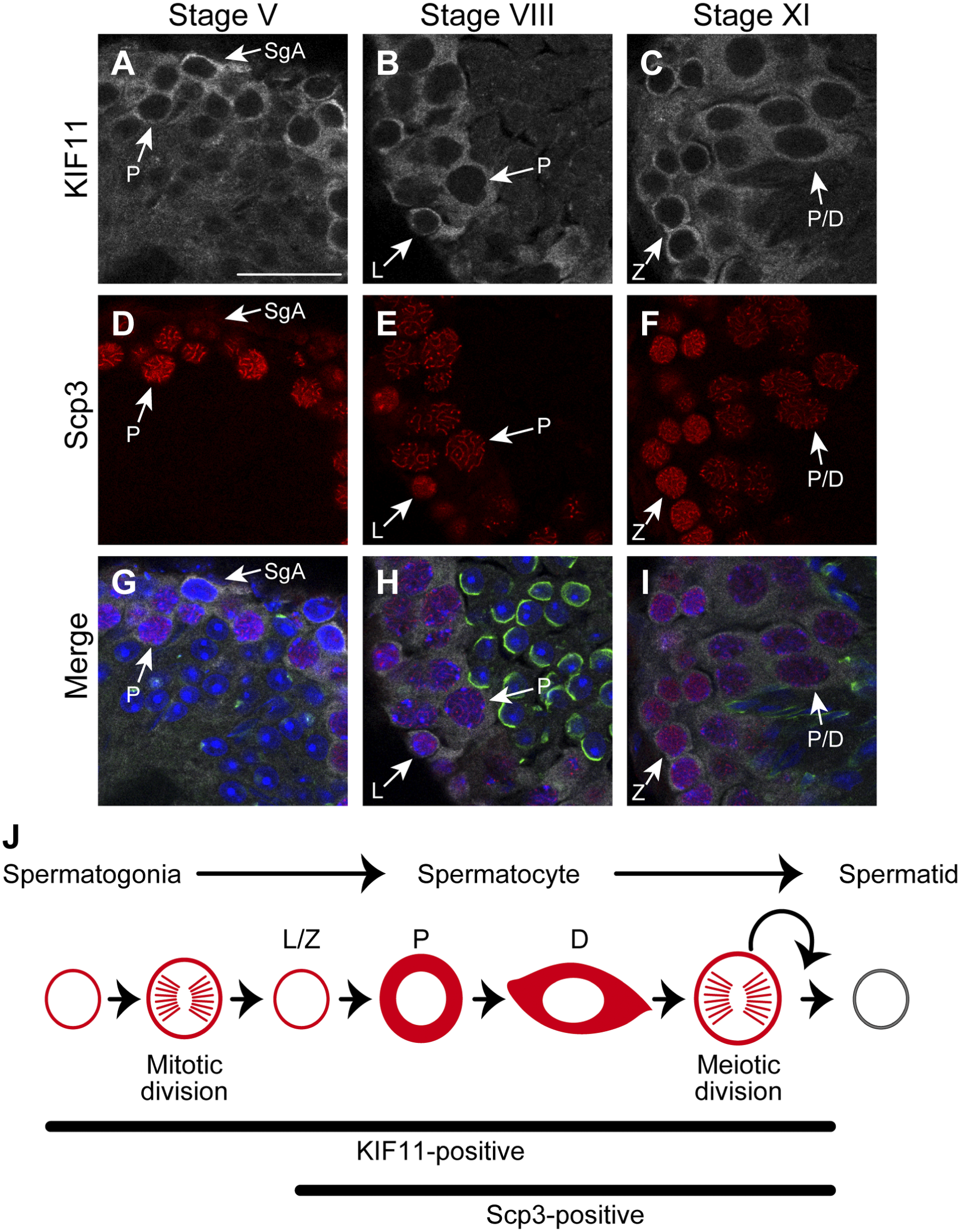
Expression profile of KIF-11 during spermatogenesis. (A–I) The areas enclosed by the dashed rectangles in J–L in Fig. 2 are enlarged: A, D, G (corresponding to Fig. 2J), B, E, H (corresponding to Fig. 2K), C, F, I (corresponding to Fig. 2L). SgA, type A spermatogonia; L/Z/P/D, leptotene/zygotene/pachytene/diplotene spermatocytes. Scale bar, 25 µm. (J) Schematic drawing of KIF11-positive cells. KIF11 is probably expressed in type A, intermediate, and type B spermatogonia. KIF11 is not expressed in spermatids with developing acrosomes. Abbreviation: KIF, Kinesin family member.
KIF11 Is Not Expressed in Sertoli Cells Within Seminiferous Tubules
We next investigated whether the Sertoli cells, which are unique somatic cells within the seminiferous tubules, express KIF11 to the same extent as the germ cells. Sertoli cells were identified based on their immunoreactivity toward vimentin, a component of Sertoli cell-specific intermediate filaments. 14 As shown in Fig. 4, the KIF11-positive cells were not stained with the anti-vimentin antibody, indicating that the KIF11-staining within the seminiferous tubules is confined to the germ cells.
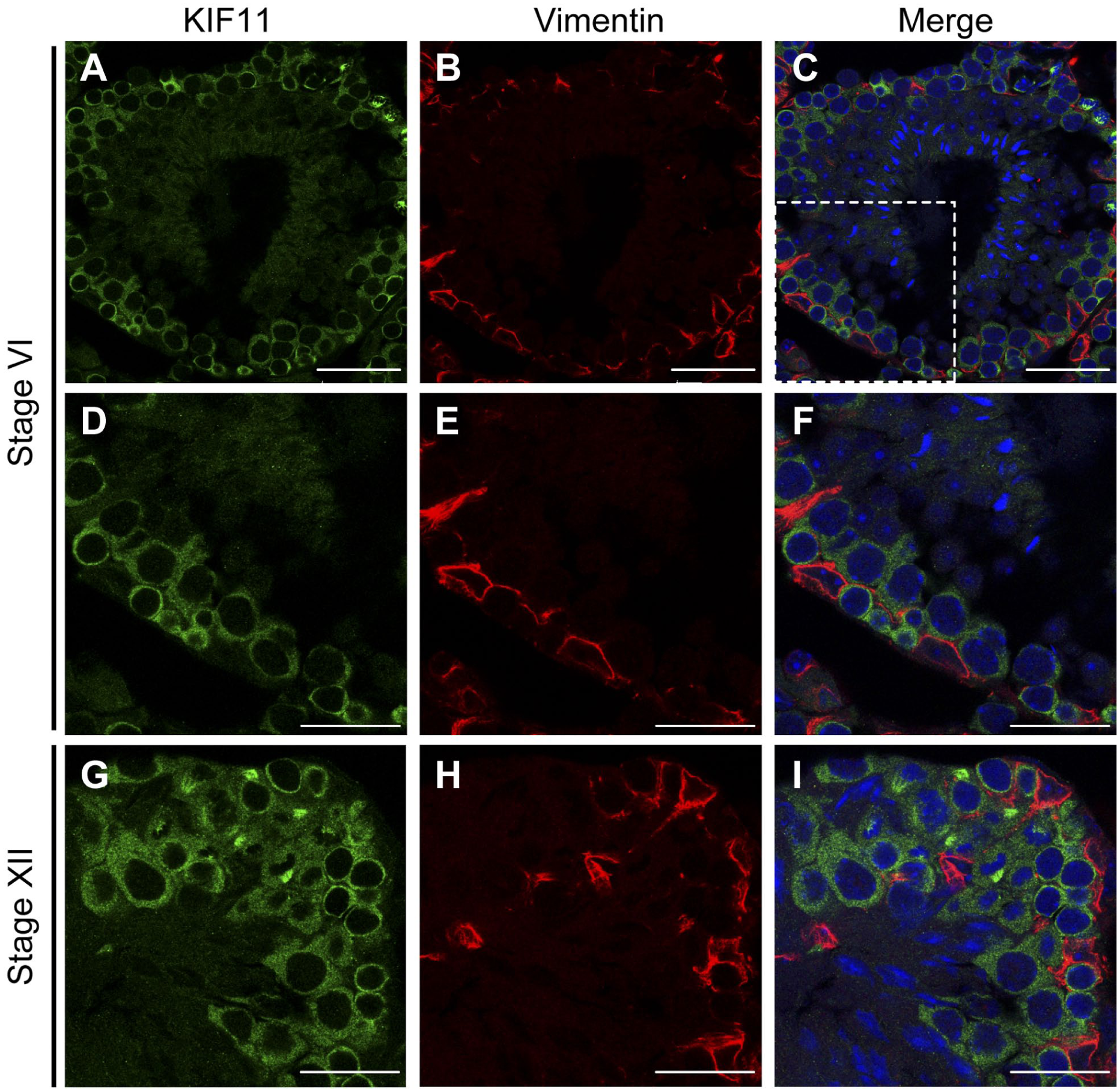
KIF-11 is not expressed in sertoli cells. Paraformaldehyde-fixed cryo-sections of mouse testes were double-stained for KIF11 (green) and vimentin (red), with nuclear counterstaining using Hoechst 33342 (blue). The immunofluorescent images of KIF11 (A, D, and G), vimentin (B, E, and H), and their overlays with Hoechst 33342 (C, F, I, respectively) in stages VI (A–F) and XII (C–I) are shown. The areas enclosed by the dashed rectangles in C are enlarged in D–F. Scale bars, 50 µm (A–C) and 25 µm (D–I). Abbreviation: KIF, Kinesin family member.
KIF11 Identifies the Cross-section of Seminiferous Tubules Undergoing Mitotic or Meiotic Division
As shown in Fig. 5, the KIF11-staining identified the cross-section in which spindle formation occurs. The formation was observed near the basement membrane (Fig. 5A to F) and in areas about 20-30 µm away from the basement membrane (Fig. 5G to O). Since one generation of spermatocytes (pachytene spermatocytes) was dominant in the cross-section (Fig. 5B), the former shows the mitosis of spermatogonia. Judging from the presence of pachytene/diplotene spermatocytes (Fig. 5H), the latter shows the meiotic cell division. The double-staining for KIF11 and Scp3 revealed that Scp3 was present in the cytoplasm when the meiotic spindle is formed (Fig. 5J to O). There were two staining patterns detected with and without prominent puncta of Scp3 on the chromosome (Fig. 5J to O, respectively). It presently remains to be elucidated whether these two situations correspond to the first and second meiotic divisions or the continuous phenomena in the course of either division. In the cell with the Scp3 puncta, we observed tubulin staining closely resembling that of KIF11 (Fig. S1), further confirming the formation of the spindle.
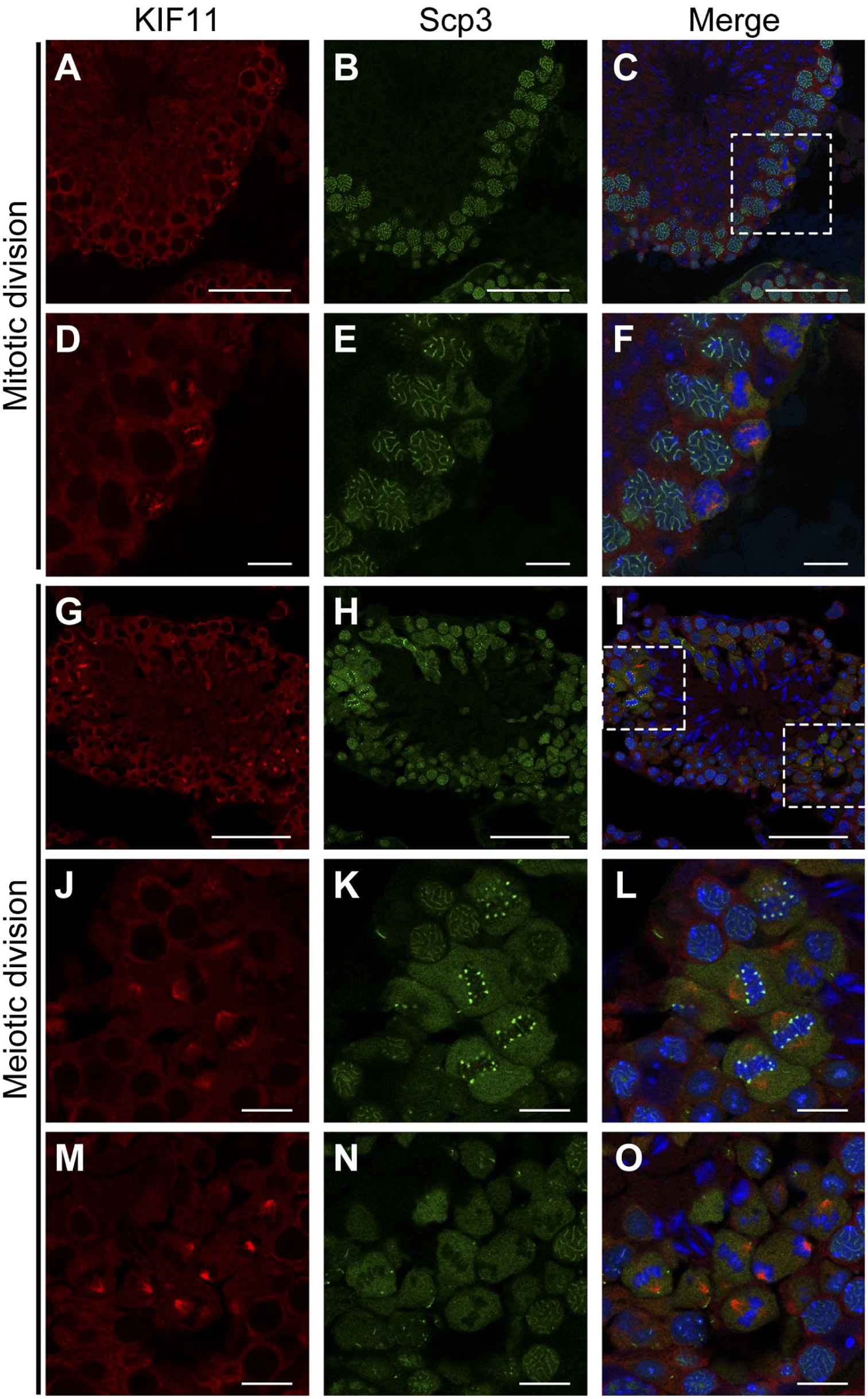
KIF-11 is involved in spindle formation during mitosis and meiosis within the seminiferous tubules. Paraformaldehyde-fixed cryo-sections of mouse testes were double-stained for KIF11 (red) and Scp3 (green), with nuclear counterstaining using Hoechst 33342 (blue). The immunofluorescent images of KIF11 (A, D, G, J, and M), Scp3 (B, E, H, K, and N), and their overlays with Hoechst 33342 (C, F, I, L, and O, respectively) in stages VI (A–F), VIII, and XII (G–O) are shown. The areas enclosed by the dashed rectangles in C and I are enlarged in D–F and J–O, respectively. Scale bars, 50 µm (A–C, G–I) and 10 µm (D–F, J–O). Abbreviation: KIF, Kinesin family member.
KIF11 Is Expressed in Spermatogonia Within Seminiferous Tubules
The most peripherally located cells include spermatogonia, which can be classified as type A, intermediate, and type B. 1 Type A spermatogonia contain a population responsible for the self-renewal of spermatogonial stem cells. In contrast, intermediate and type B spermatogonia are in the course of generating spermatocytes. LIN28A is an RNA-binding protein involved in the control of microRNA synthesis, and is located in type A spermatogonia. 15 Some of the cells that were negative for both Scp3 and vimentin were positive for LIN28A (Fig. S2A to D). The peripheral germ cells that were negative for both Scp3 and LIN28A are therefore considered to be intermediate or type B spermatogonia (shown by white arrows in Fig. S2G). Since the peripherally located cells in cross-sections of seminiferous tubules were stained by either the anti-KIF11-antibody or anti-vimentin-antibody, KIF11 should be expressed in most types of spermatogonia.
Collectively, these results revealed for the first time that KIF11 is expressed in premeiotic germ cells (spermatogonia to spermatocytes) and is downregulated when the germ cells become spermatids, which no longer undergo cell division (Fig. 3J).
The Immunolocalization of KIF11 Suggests the Dysregulation of Meiosis Progression in Spermatogenesis-arrested Mice
We sought to evaluate whether the KIF11-staining could be applied to describe the process of incomplete spermatogenesis. For this purpose, we intended to focus on mutant mice that become male infertile due to a deficiency of a factor that seemingly is not directly involved in spermatogenesis. β1,4-N-acetylgalactosaminyltransferase (β1,4-GalNAc-T, EC2.4.1.94) is a key enzyme in the synthesis of glycosphingolipids. 8 Mice deficient in β1,4-GalNAc-T (hereafter referred to as the mutant mice) are viable and do not show an appreciable phenotype; however, they are male infertile.16,17 These mutant mice do not produce elongating and elongated spermatids within the seminiferous tubules.16,17 However, the defects in the premeiotic cells have not been specified.
When testicular cells from the wild-type and mutant mice were compared by flow cytometry, the populations of haploid, diploid (including Sertoli cells), and tetraploid cells were 48%, 15%, and 37% in the wild-type mice and 27%, 25%, and 46% in the mutant mice, respectively (Fig. 6). In addition to the decrease in the population of haploid cells, these results revealed that the tetraploid cells include additional cell groups that are not present in the wild-type mice (shown by asterisks in Fig. 6B).
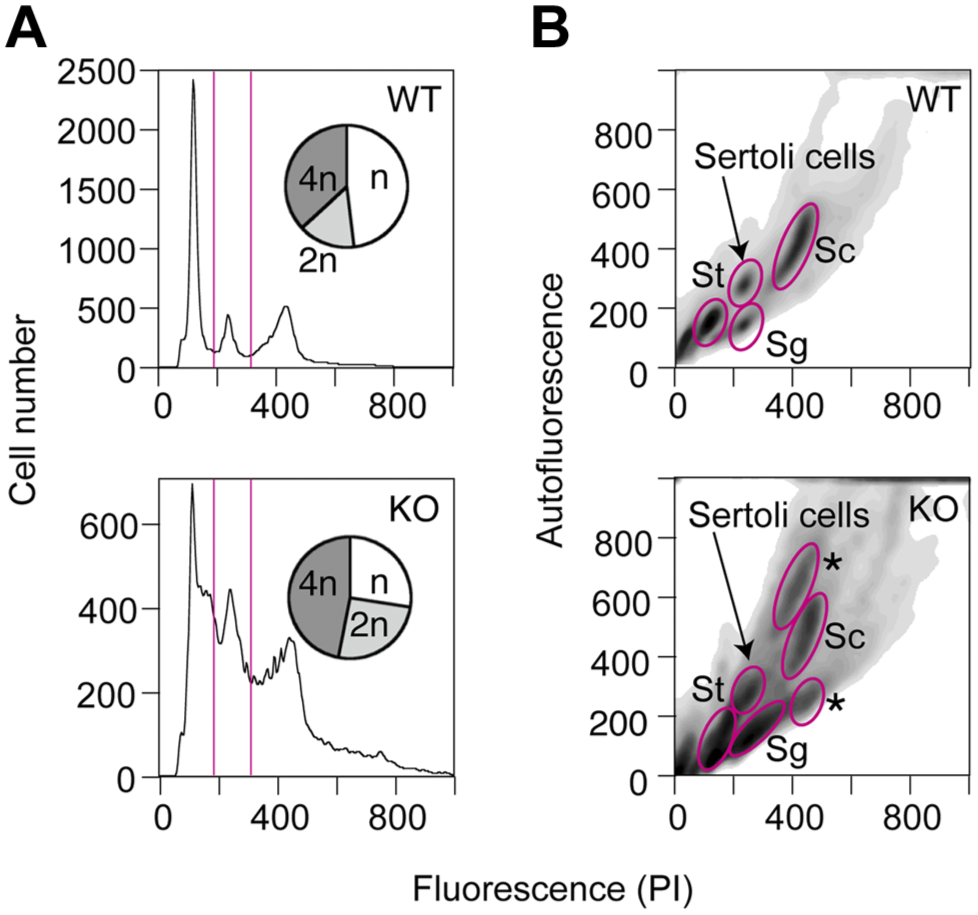
Abnormal tetraploid cells are present in the β1,4GalNAc-T-deficient mice. Testicular cells were prepared from the seminiferous tubules of the wild-type (8-week old) and mutant mice (17 week old, similar results were obtained in 10 ~11-week old mice). The cells were fixed by the high citrate method, stained with propidium iodide (PI), 10 and analyzed by flow cytometry. (A) The number of cells (vertical axis) was plotted against the staining with PI (horizontal axis). The populations of cells with different ploidies are shown in the inserted pie charts. (B) Two-dimensional separations, with PI staining on the horizontal axis and green autofluorescence on the vertical axis, are shown. St, spermatids (n); Sg, spermatogonia (2n); Sc, leptotene, zygotene, and pachytene spermatocytes (4n). The tetraploid cells in the mutant mice included additional cell groups that were not present in the wild-type mice (marked by asterisks). Abbreviations: PI, propidium iodide; WT, wild-type; KO, knockout.
Therefore, we co-stained the testis sections from the mutant mice with an anti-KIF11 antibody, an anti-Scp3 antibody, and PNA-lectin (Fig. 7). In agreement with the previous study,16,17 multinuclear giant cells composed of round spermatids were observed (Fig. 7J and K) and there was minimal development of acrosomal caps (Fig. 7D to F). The cells occupying the central part in the mutant mice appeared to be pachytene spermatocytes, based on the Scp3-staining. The KIF11-staining suggested that they were larger than the corresponding cells observed in the wild-type mice, which might be relevant to the abnormal population of tetraploid cells, which have a larger cell size (Fig. 6B).
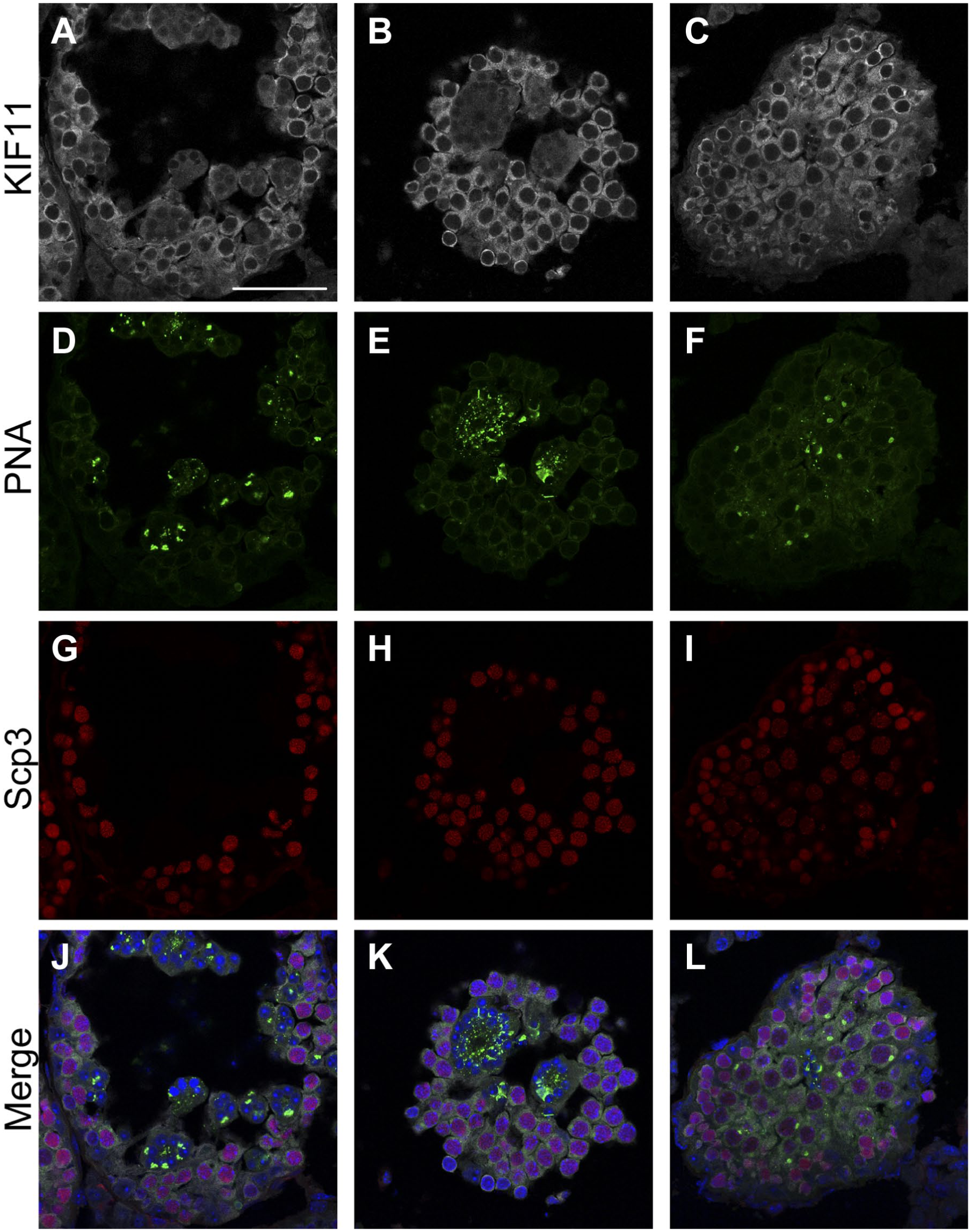
The KIF11-positive cells are not restricted to the periphery of the seminiferous tubules in the β1,4GalNAc-T-deficient mice. Paraformaldehyde-fixed cryo-sections of mouse testes from the mutant mice (7 week old) were triple-stained for KIF11 (gray), PNA (green), and Scp3 (red), with nuclear counterstaining using Hoechst 33342 (blue). The immunofluorescent images of KIF11 (A–C), PNA (D–F), Scp3 (G–I), and their overlays with Hoechst 33342 (J–L, respectively) are shown. Scale bar, 50 µm. Abbreviations: KIF, Kinesin family member; PNA, peanut agglutinin.
As shown in Fig. 8, meiotic spindles were formed in the cells with the cytosolic Scp3 in the wild-type mice (shown by white arrows in Fig. 8E). In contrast, in the case of the mutant mice, the cells showing the cytosolic staining-pattern for Scp3 were found both with and without the formation of the spindle (shown by white arrows and arrowheads, respectively, in Fig. 8F). In addition, although the zygotene spermatocytes were found near the cells undergoing meiotic cell division in the case of the wild-type mice (shown by orange arrows in Fig. 8E and also as the insets in Fig. 8C), the Scp3-staining suggested that the cells located in the corresponding position in the mutant mice are distinct from the zygotene spermatocytes (shown by orange arrowheads in Fig. 8F and also as the insets in Fig. 8D), which might be relevant to another abnormal population of tetraploid cells with a smaller cell size (Fig. 6B). However, the cytosolic staining of KIF11 revealed a slight oval shape in both cases (the insets in Fig. 8A and B), indicating that these cells are probably in the preparatory stages of meiosis, similar to diplotene spermatocytes (Fig. 3J). These results suggested that the spindle formation and the synaptonemal complex formation/disruption do not occur simultaneously in the mutant mice. Accordingly, the immunolocalization of KIF11 allows the description of abnormalities in the process of spermatogenesis prior to meiotic division in the mutant mice.
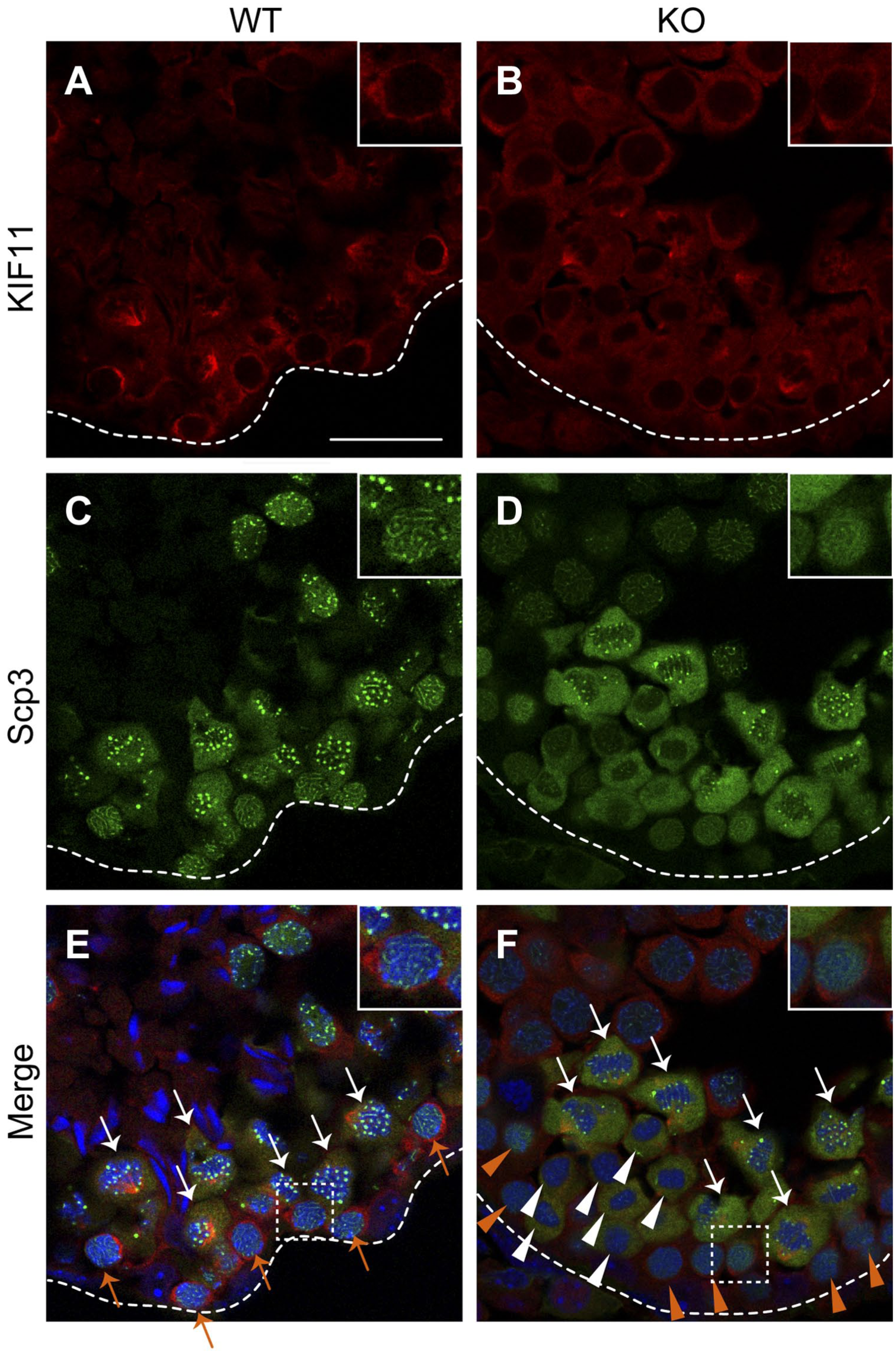
KIF11-staining suggests that the differentiation of premeiotic cells is delayed by improper spindle formation in the β1,4GalNAc-T-deficient mice. Paraformaldehyde-fixed cryo-sections of mouse testes (7 week old) from the wild-type (A, C, and E) and mutant (B, D, and F) mice were double-stained for KIF11 (red) and Scp3 (green), with nuclear counterstaining using Hoechst 33342 (blue). The immunofluorescent images of KIF11 (A and B), Scp3 (C and D), and their overlays with Hoechst 33342 (E, F, respectively) are shown. Outlines of cross-sections of the seminiferous tubules are shown with dashed lines. The cells with cytosolic Scp3-staining and spindle formation are shown with white arrows (E and F) and those with cytosolic Scp3-staining but without spindle formation are shown with white arrowheads (F). The zygotene spermatocytes are shown with orange arrows (E). The cells that were positive for KIF11 but had diffuse Scp3-staining are shown with orange arrowheads (F). The areas enclosed by the dashed rectangles in E and F are enlarged in the insets in A, D, E and B, D, F, respectively. Scale bar, 25 µm. Abbreviation: KIF, Kinesin family member.
Discussion
Analyses of spermatogenesis-arrested conditions, caused either by a loss of particular molecules or a drug treatment, are necessary to promote research on infertility. For this purpose, the stage-identification of each cross-section of the seminiferous tubules provides a basic description. However, in the spermatogenesis-arrested situation, the stage-identification without information about the acrosome and the nuclear morphology of spermatids remained as a cumbersome task.
KIF11 is a unique motor protein that is responsible for mitotic spindle formation.4,5 In the present study, we demonstrated the drastic decline in the expression of KIF11 after meiosis within the seminiferous tubules. Furthermore, the transition of KIF11 from its diffuse cytoplasmic localization to the spindle microtubules is likely to be regulated by several kinases. 5 Thus, it is conceivable that the expression of KIF11 is tightly coupled to the proper progression of meiosis during spermatogenesis. In support of our hypothesis, the KIF11 staining revealed that the synchronization between the spindle formation and the nuclear meiotic events is altered in the spermatogenesis-arrested mutant mice. Thus, KIF11 can serve as a marker of premeiotic germ cells to describe their temporal changes during spermatogenesis. This characteristic of KIF11 will be quite valuable to describe the incomplete spermatogenesis in the absence of spermatids.
The situations in the testis germ cells may be relevant to those in oocytes 6 and early embryos, in which the importance of KIF11 declines during preimplantation development. 7 Further work is necessary to elucidate the molecular mechanism underlying the downregulation of KIF11 after the completion of meiosis.
Supplemental Material
DS_10.1369_0022155419871027 – Supplemental material for KIF11 as a Potential Marker of Spermatogenesis Within Mouse Seminiferous Tubule Cross-Sections
Supplemental material, DS_10.1369_0022155419871027 for KIF11 as a Potential Marker of Spermatogenesis Within Mouse Seminiferous Tubule Cross-Sections by Miki Hara-Yokoyama, Hidetake Kurihara, Shozo Ichinose, Hironori Matsuda, Shizuko Ichinose, Masaru Kurosawa, Norihiro Tada, Chihiro Iwahara, Kazue Terasawa, Katarzyna A. Podyma-Inoue, Koichi Furukawa and Kazuhisa Iwabuchi in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We thank Yoshihiko Araki, at the Institute for Environmental and Gender-Specific Medicine of Juntendo University Graduate School of Medicine, and Tetsuro Watabe, at the Department of Biochemistry, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, for valuable discussions.
Author Contributions
MHY, HK, KAP-I, KF, and KI designed the research and wrote the paper; HM carried out the flow cytometric analysis. MHY, HK, S (Shozo) I and S (Shizuko) I performed the immunohistochemistry; MK, NT, CI, and KT analyzed the data. All authors have read and approved the final manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a Grant-in-Aid (S1311011) from the Foundation of Strategic Research Projects in Private Universities, Initiative for Realizing Diversity in the Research Environment and the High-Tech Research Center Project for Juntendo University: matching fund subsidy from the Ministry of Education, Culture, Sports, Science, and Technology, and the collaborative research fund program for women researchers from the Tokyo Medical and Dental University, funded by “Initiative for Realizing Diversity in the Research Environment,” from MEXT, Japan, in 2015: JSPS KAKENHI Grant Number JP17K12005 (to M.H.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
