Abstract
Primary obstructive megaureter (POM) morphogenesis is not fully known. The aim of the study was to evaluate the appearance of different factors that might take part in the pathogenesis of POM. Megaureter tissues of 14 children were stained with hematoxylin and eosin as well as with immunohistochemistry for protein gene product 9.5, nerve growth factor receptor, transforming growth factor beta 1 (TGFβ1), fibroblast growth factor receptor 1 (FGFR1), matrix metalloproteinase 2 (MMP-2), angiotensin 2 receptor type 2, and sonic hedgehog (SHH) protein. Apoptosis was detected by terminal dUTP nick-end labeling reaction. POM tissues revealed transitional epithelium with scattered vacuolization, submucosa with inflammatory cells, and focally vacuolized and chaotically organized muscle layers. Apoptosis, appearance of MMP-2, FGFR1, and SHH prevailed, but TGFβ1 positive cell number was lower in patients. Correlation between MMP-2 in epithelium and endothelium, FGFR1 and MMP-2 in epithelium, and TGFβ1 in epithelium and connective tissue in patients was detected. POM morphopathogenesis involves an apoptotic cell death of epithelium and smooth muscle as well as tissue degradation in epithelium and connective tissue of the ureter wall. The decrease of tissue growth through diminished TGFβ1 expression and stimulation of FGFR1 and MMP-2 suggests a disbalance of tissue remodelation in the megaureter wall:
Introduction
Megaureter is enlargement of the ureter greater than 7 mm in diameter 1 with estimated incidence at 0.3 in 1000 newborns worldwide. 2 It is believed to represent 23% of children with urinary tract obstruction. 3 Megaureter can lead to functional obstruction with adynamic juxta vesical or intramural ureteral ending segment. 4
Primary obstructive megaureter (POM) morphogenesis is not fully known. Some studies showed that there are increased amounts of connective tissue, atrophy of inner longitudinal muscle layer, and hypertrophy of outer muscle layer in ureteral wall.5–7
Protein gene product 9.5 (PGP 9.5) is deubiquitinating enzyme expressed mostly in neurons and neuroendocrine cells, and it is used as a biomarker to identify peripheral nervous system axons. 8 It has been detected that approximately 80% of ureter nervous fibers contain PGP 9.5. 9 Lack of PGP 9.5 expression can be associated with axonal atrophy. 10
Nerve growth factor (NGF) is a growth factor of neurotrophin family proteins. It has been noticed that a disturbance of the NGF has an impact on different urinary tract disorders, for example, bladder outlet obstruction. 11 The signaling of nerve growth factor receptor (NGFR) is involved in the pathophysiology of many renal diseases, and the proapoptotic role of NGFR is well known. 12 A kidney organogenesis requires functional NGFR. 13
Transforming growth factor beta (TGFβ) is a growth factor that is produced by most cells, including fibroblasts and macrophages.14,15 TGFβ1 is a profibrotic cytokine which stimulates mesenchymal cell proliferation and extracellular matrix deposition.16–18 In organ culture, exogenous TGFβ postpones the ureterovesical smooth muscle proliferation and affects the muscular structure. 19
Fibroblast growth factor (FGF) is a group of growth factors that plays role in a kidney and lower urinary tract development, for instance, nephron progenitor differentiation in nephrons and development of ureteric epithelium. 20 Fibroblast growth factor receptor 1 (FGFR1) is an FGF receptor with tyrosine kinase activity. It has been thought to be necessary for embryonic kidney and urinary tract development. 21 FGFRs are highly expressed in the lower urinary tract. 20
Matrix metalloproteinase 2 (MMP-2) is a type IV collagenase expressed by smooth muscles in broad types of organs. Main substrates of this enzyme are collagen type I and IV, that are found in extracellular matrix. 22 MMP-2 may have pathogenic role in renal interstitial fibrosis. 23
Angiotensin 2 (AT2) is a peptide hormone with the main effect of vasoconstriction and vascular remodulation. 24 AT2 mediates the early kidney and urinary tract morphogenesis. Disruption in receptor functions promotes development of congenital anomalies of the kidney and urinary tract.25,26
Sonic hedgehog (SHH) is protein secreted by numerous embryonic cells and is believed to be involved in stem cell differentiation. Some studies have shown variations of SHH expression in urinary tract as well as in kidney development, which could be linked to development of different pathologies.27–29 SHH, described in the ureteric epithelium, 30 stimulates peri-urothelial mesenchymal cell proliferation and regulates smooth muscle cell differentiation. 17 Deficiency of SHH resulted in reduced mesenchymal proliferation and delay smooth muscle cell differentiation. 31
Terminal dUTP nick-end labeling (TUNEL) assay is a marker that provides the information about cell’s DNA integrity. TUNEL binds oligonucleosomal-sized fragments that are prevalent in apoptotic cells. Some studies show increase of apoptotic epithelial, smooth muscle, and fibroblast cells in obstructive uropathy caused by ureteral obstruction or congenital ureteropelvic junction obstruction.32–35
The aim of the study was to evaluate the appearance and distribution of different factors that participate in regulation, innervation, vascularization, and growth/development of ureters and might take part of pathogenesis in development of POM.
Materials and Methods
The study population consisted of 11 children (8 boys, 3 girls) aged from 1 month to 15 years. Material was taken bilateral (2) or unilateral from the right side (2) as well as unilateral from the left side (7). Material was obtained either from distal parts of ureter (9), middle part (1), or both middle and distal parts (1) (Table 1). Three control samples were obtained from children not associated with megaureters from their distal part of the ureter. Age of control children (two boys and one girl) was 13 months, 2 years, and 10 years, respectively. The tissue material was obtained from the historical tissue material collection of the Institute of Anatomy and Anthropology, Riga Stradins University. On May 10, 2007, the study was approved by the Ethics Committee of the Riga Stradins University.
Study Population.
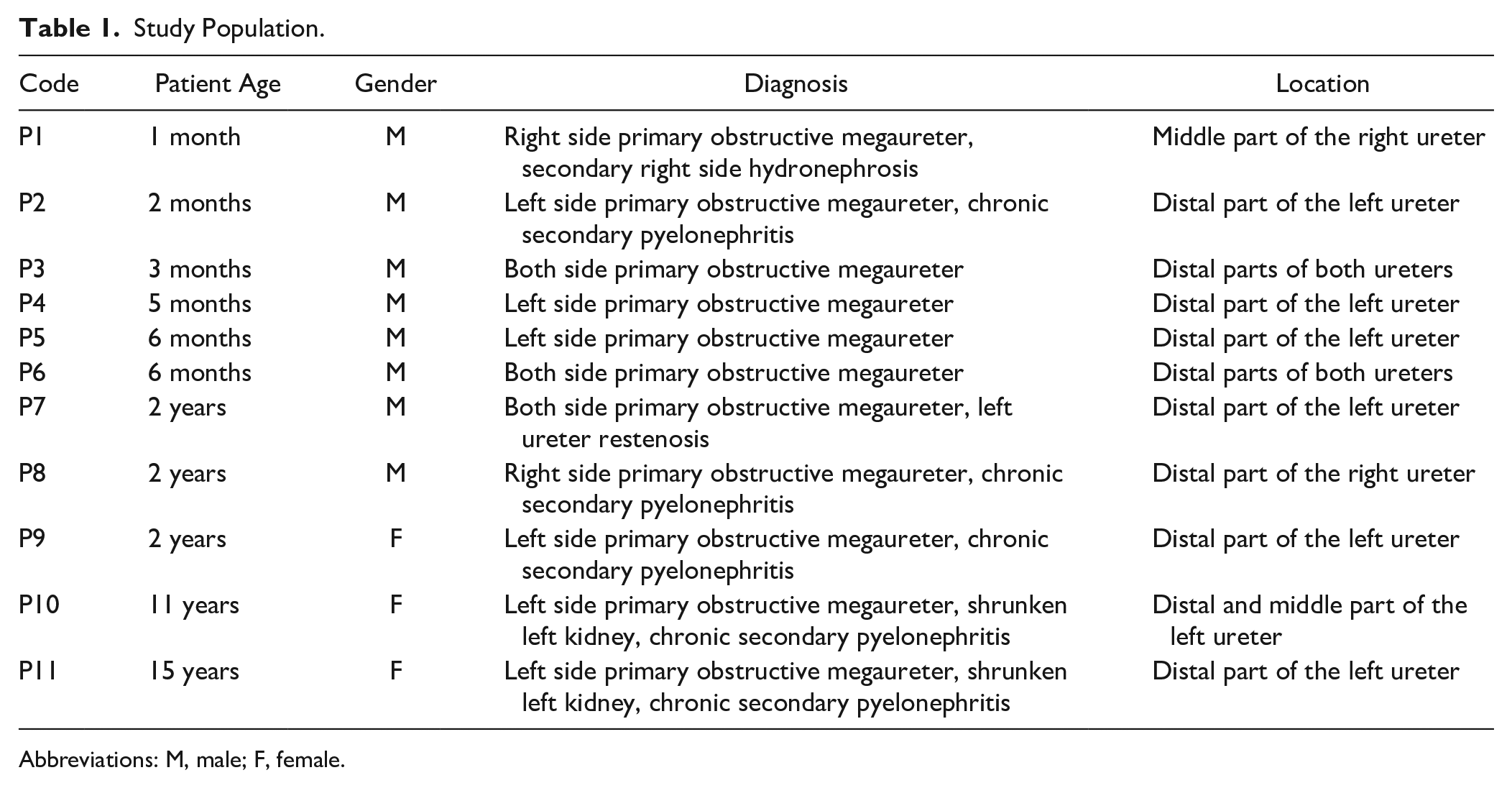
Abbreviations: M, male; F, female.
Tissue samples were fixed for 24 hr in Stefanini fixative—mixture of 2% formaldehyde and 0.2% picric acid in 0.1-M phosphate buffer (pH 7.2). 36 Then, samples were rinsed in Tyrode buffer (content: NaCl, KCl, CaCl2·2H2O, MgCl2·6H2O, NaHCO3, NaH2PO4·H2O, glucose) containing 10% saccharose for 12 hr and afterwards dehydrated and embedded in paraffin (sample processing scheme: t = 37C in thermostat, 70% ethanol for 1 hr, 80% ethanol for 1 hr, 96% ethanol I for 2 hr, 96% ethanol II for 2 hr, 96% ethanol III for 3 hr, xylol I for 1 hr, xylol II for 1 hr, paraffin I for 1 hr, paraffin II for 1.5 hr).
Three-µm-thin sections were cut, which were then stained with hematoxylin and eosin (xylol I for 5 min, xylol II for 5 min, xylol I for 5 min, 96% ethanol I for 3 min, 96% ethanol II for 3 min, 70% ethanol for 3 min, distilled water for rinsing, staining with hematoxylin for 10 min, rinsing with water, staining with eosin for 1 min, rinsing with water, 70% ethanol for 3 min, 96% ethanol I for 3 min, 96% ethanol II for 3 min, carbol/xylol mixture for 2 min, xylol I for 3 min, xylol II for 3 min, closing the sample with histological glue and coverslip) for routine morphological evaluation.
Immunohistochemistry
Manual staining method was used for immunohistochemistry [deparaffinizing and rehydration using heat-induced epitope retrieval with EDTA buffer for 5 min and rinse with distilled water, placing slides in peroxidase block for 10 min, rinse with distilled water 5×, apply the antibody and incubate, rinse with distilled water 5×, apply amplifier for mouse/rabbit antibodies (Lot. 000042022) and incubate for 10 min, rinse with distilled water for 5×, apply horseradish peroxidase polymer detector (Lot. 000041948), incubate for 10 min, rinse with distilled water for 5×, apply 3,3’-Diaminobenzidine (DAB) chromogen mixture (DAB buffer Lot. 000041948 and DAB chromogen Lot. 000025879), develop color for 4 min, then rinse the slides with distilled water 5×, counterstain with hematoxylin for 1 min, rinse with distilled water 5×, afterwards dehydrate the slides in 96% ethanol and apply the coverslip with histological glue]. Data on the antibodies used in the study have been summarized in Table 2.
Antibodies Used in Immunohistochemistry.
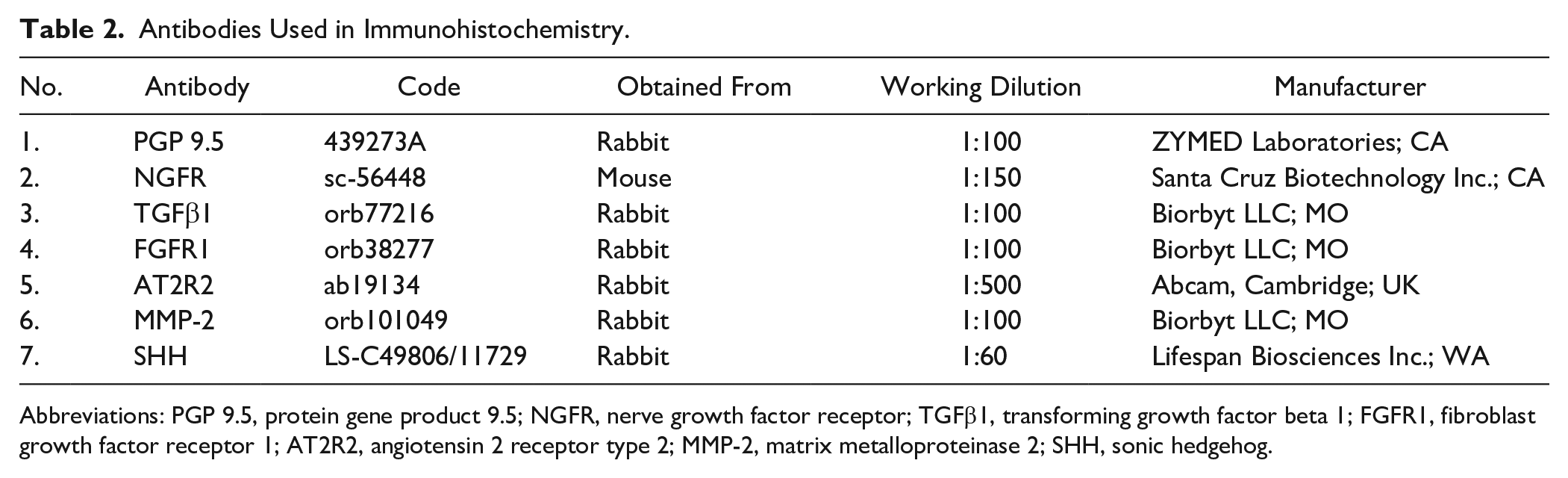
Abbreviations: PGP 9.5, protein gene product 9.5; NGFR, nerve growth factor receptor; TGFβ1, transforming growth factor beta 1; FGFR1, fibroblast growth factor receptor 1; AT2R2, angiotensin 2 receptor type 2; MMP-2, matrix metalloproteinase 2; SHH, sonic hedgehog.
Staining of apoptotic cells by TUNEL was processed by a standard in situ cell death detection kit, POD cat. no 11684817910, manufactured by Roche Diagnostics, in a working dilution of 1:10. 37
For each preparation series, positive controls of the tissues indicated by the manufacturer, which always have positive reaction, were prepared. As negative control, the parallel cuts of the preparation, where primary antibody was substituted by Antibody Diluent, were used. The stained preparations were examined using a light microscope (Leica DM500RB; Leica Biosystems Richmond Inc.), and afterward, the preparations were processed in the Image Pro Plus video analysis system and were fixed using a digital camera (Leica DC 300F; Leica Microsystem AG, Germany).
The slides were analyzed by light microscopy using non-parametric evaluation. The results were evaluated by grading the appearance of positively stained cells in the visual field. Structures in the visual field were labeled: 0 = no positive structures, 0/+ = occasional (<10) positive structures, + = few (10–15) positive structures, +/++ = few to moderate number (16–20) of positive structures, ++ = moderate number (21–35) of positive structures, ++/+++ = moderate to numerous (36–50) positive structures, +++ = numerous (51–70) positive structures, +++/++++ = numerous to abundant (71–90) positive structures, ++++ = abundance (>90) of positive structures in the visual field. 38 The number of positive structures was detected in 10 visual fields, and the rounded average was stated as the finding of the sample.
Statistical Methods
The data processing was performed with Statistical Package for the Social Sciences software, v26.0 (IBM Company; Chicago, IL). To evaluate the cross-compliance of two variables, Spearman’s rank correlation coefficient (rs) was calculated. 39 The acquired results were interpreted: rs = 0–0.2 was assumed as a very weak correlation, rs = 0.2–0.4 was assumed as a weak correlation, rs = 0.4–0.6 was assumed as a moderate correlation, rs = 0.6–0.8 was assumed as a strong correlation, and rs = 0.8–1.0 was assumed as a very strong correlation. To analyze control group versus patient data, Mann–Whitney U test was used. 40 The level of significance for all tests was chosen as 5% (p<0.05). To perform statistical studies, non-parametric evaluation grading was transformed to number labels: 0 = 0.00; 0/+ = 0.50; + = 1.00; +/++ = 1.50; ++ = 2.00; ++/+++ = 2.50; +++ = 3.00; +++/++++ = 3.50; ++++ = 4.00.
Results
The megaureter material contained transitional epithelium, submucosa, smooth muscle layers (inner longitudinal, middle circular, and outer longitudinal), and adventitia of ureter. Routine staining showed scattered epithelial vacuolization (Fig. 1A) and atrophy of smooth muscle layers with merged, different thickness layers and with some vacuolization of smooth muscle cells (Fig. 1B). Also, lymphatic follicles in submucosa (Fig. 1A) and blood vessel sclerosis were observed in some samples. In contrast, samples from the control group show no signs different from healthy tissue (Fig. 1C and D).
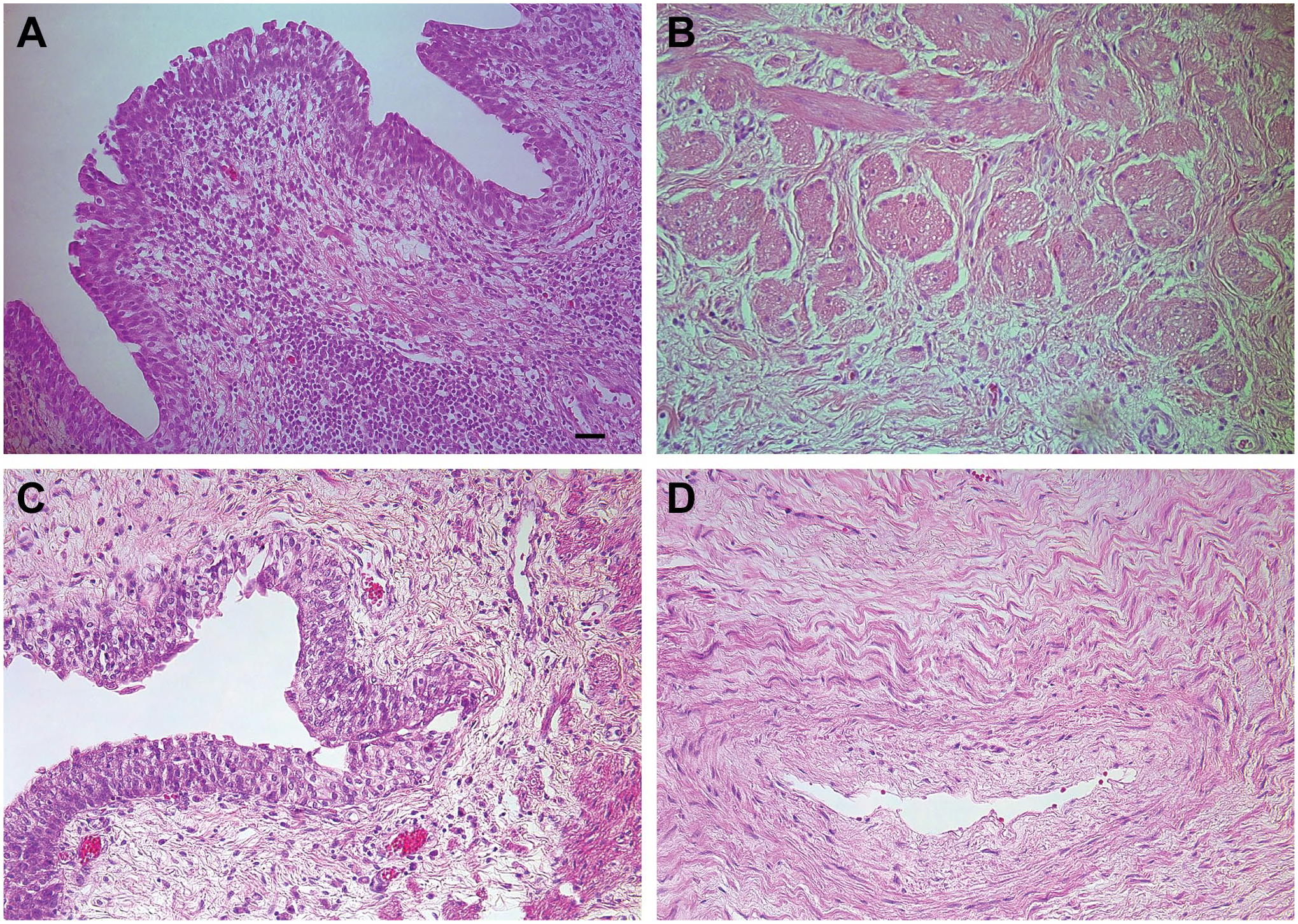
Morphological findings in megaureter-affected children and in the control group. (A) Epithelium vacuolization and lymphatic follicle in submucosa of a 2-month-old megaureter patient. H&E, ×200 (scale bar: 50 μm). (B) Muscle cell vacuolization and chaotic structure of a 5-month-old megaureter patient. H&E, ×250 (scale bar: 40 μm). (C) Normal epithelium and submucosa of a 13-month-old child in the control group. H&E, ×200 (scale bar: 50 μm). (D) Normal muscular layer with blood vessel of a 10-year-old child in the control group. H&E, ×250 (scale bar: 40 μm).
PGP 9.5-marked moderate to numerous positive structures were observed in six patients and few positive structures in three patients (Fig. 2A, Table 3). Meanwhile, a moderate number of PGP 9.5-positive nerve fibers were observed in the control group (Fig. 2B). There was no statistically significant difference between the groups. NGFR showed no difference between patient (Fig. 2C) and control group (Fig. 2D) with occasional positive neurocytes (Table 3).
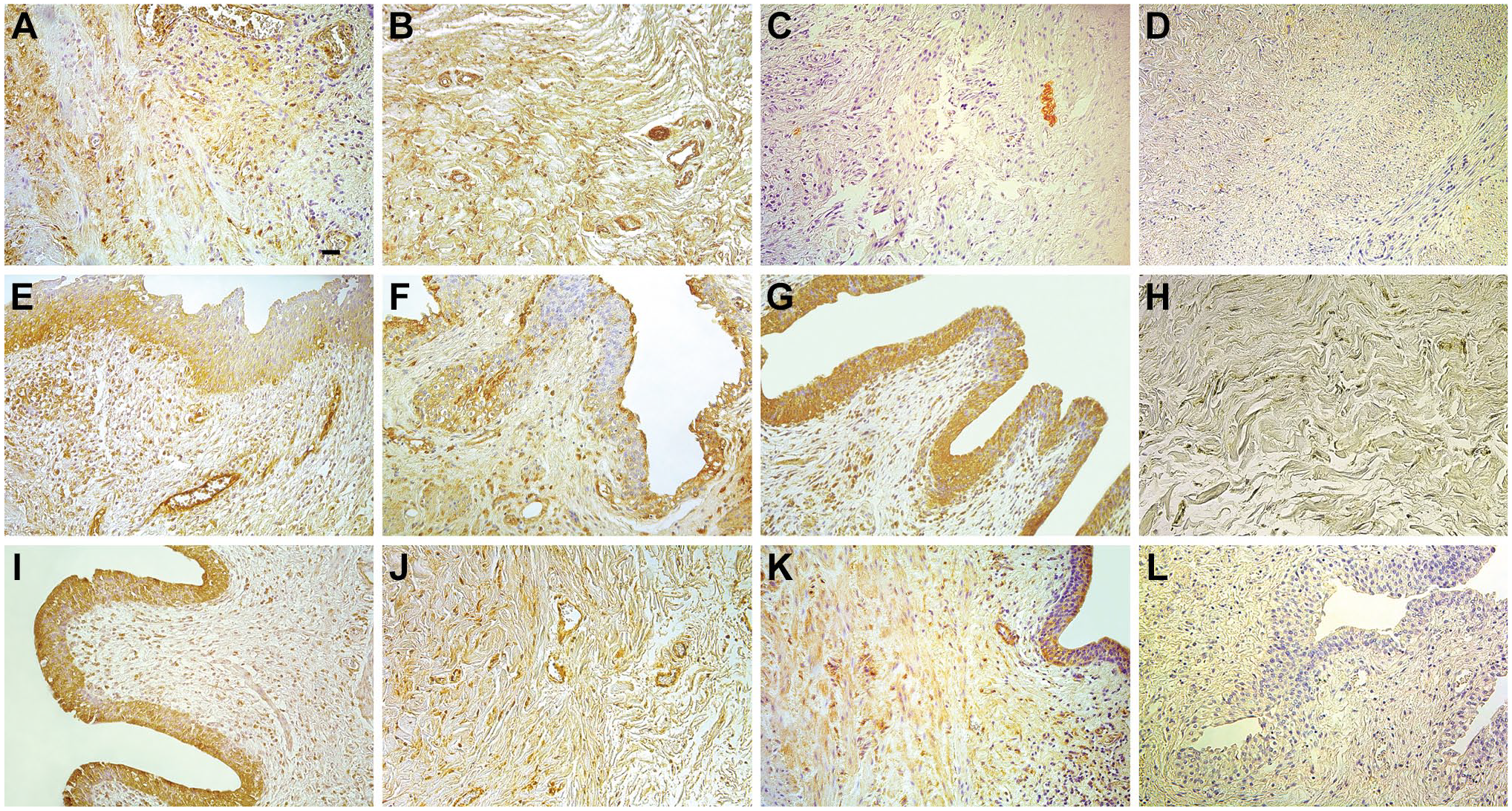
Immunoreactive structures in megaureter-affected children and in the control group. (A) Numerous PGP 9.5-positive nerve fibers in a 5-month-old megaureter patient. PGP 9.5 IMH, ×200 (scale bar: 50 μm). (B) Moderate PGP 9.5-positive nerve fibers of a 2-year-old child in the control group. PGP 9.5 IMH, ×200 (scale bar: 50 μm). (C) Few NGFR-positive neurons in a 2-month-old megaureter patient. NGFR IMH, ×200 (scale bar: 50 μm). (D) Occasional to few NGFR-positive cells of a 2-year-old child in the control group. NGFR IMH, ×200 (scale bar: 50 μm). (E) Few TGFβ1-positive cells in epithelium and connective tissue in a 15-year-old megaureter patient. TGFβ1 IMH, ×200 (scale bar: 50 μm). (F) Numerous TGFβ1-positive connective tissue cells of a 13-month-old child in the control group. TGFβ1 IMH, ×200 (scale bar: 50 μm). (G) Abundant FGFR1-positive epithelial cells, as well as moderate number of positive connective tissue cells in a 2-month-old megaureter patient. FGFR1 IMH, ×200 (scale bar: 50 μm). (H) Occasional to few FGFR1-positive cells in connective tissue of a 2-year-old child in the control group. FGFR1 IMH, ×200 (scale bar: 50 μm). (I) Numerous AT2R2-positive cells in a 2-month-old megaureter patient. AT2R2 IMH, ×200 (scale bar: 50 μm). (J) Moderate AT2R2-positive cells of a 2-year-old child in the control group. AT2R2 IMH, ×200 (scale bar: 50 μm). (K) Few MMP-2-positive cells in a 5-month-old megaureter patient. MMP-2 IMH, ×200 (scale bar: 50 μm). (L) Occasional MMP-2-positive cells of a 13-month-old child in the control group. MMP-2 IMH, ×200 (scale bar: 50 μm). Abbreviations: PGP 9.5, protein gene product 9.5; NGFR, nerve growth factor receptor; TGFβ1, transforming growth factor beta 1; FGFR1, fibroblast growth factor receptor 1; AT2R2, angiotensin 2 receptor type 2; MMP-2, matrix metalloproteinase 2
Morphological Findings in the Megaureter Tissue.

Abbreviations: E, epithelium; CT, connective tissue; IC, inflammatory cells; ET, endothelium; SM, smooth muscle; 0, no positive structures; 0/+, occasional positive structures; +, few positive structures; +/++, few to moderate number of positive structures; ++, moderate number of positive structures; ++/+++, moderate to numerous positive structures; +++, numerous positive structures; +++/++++, numerous to abundant structures; ++++, abundance of positive structures; Var, variable appearance of positive structures; PGP 9.5, protein gene product 9.5; NGFR, nerve growth factor receptor; TGFβ1, transforming growth factor beta 1; FGFR1, fibroblast growth factor receptor 1; AT2R2, angiotensin 2 receptor type 2; MMP-2, matrix metalloproteinase 2; SHH, sonic hedgehog; TUNEL, terminal dUTP nick-end labeling; *, statistically significant difference of the relative number in structures between the patient and control groups.
TGFβ1-marked few positive cells in epithelium and occasional positive cells in connective tissue were detected in the patient group (Fig. 2E). Compared with the control group, TGFβ1-positive structures in connective tissue were in higher counts, but there was no statistically significant difference between the groups (Table 3). Occasional TGFβ1-positive epithelial cells and moderate to numerous TGFβ1-positive connective tissue cells (Fig. 2F) were seen in the control group. Spearman’s rank coefficient revealed very strong positive correlation in the patient group between TGFβ1 epitheliocytes and fibroblasts (rs = 0.942; p<0.001), and strong positive correlation was observed between TGFβ1 epitheliocytes and FGFR1 endotheliocytes (rs = 0.745; p=0.005).
FGFR1 in epithelium had broad expression from moderate to abundant numbers of positive cells; in inflammatory cells, it varied from few to numerous quantities of positive structures; and in connective tissue, expression was from few to moderate (Fig. 2G). In control group, expression was occasional in structures mentioned above (Fig. 2H), but in endothelium, FGFR1 expression was moderate to numerous in both groups (Table 3). A statistically significant difference was seen between numbers of FGFR1-positive epithelial cells (U = 3.50; p=0.032) and FGFR1 inflammatory cells (U = 3.00; p=0.027). Spearman’s rank coefficient showed very strong positive correlation in the megaureter group between FGFR1- and MMP-2-positive epitheliocytes (rs = 0.805; p=0.005), strong positive correlation was observed between FGFR1 epitheliocytes and MMP-2 endothelial cells (rs = 0.755; p=0.005) as well as between FGFR1 and MMP-2 endotheliocytes (rs = 0.664; p=0.019).
Angiotensin 2 receptor type 2 (AT2R2) marked variable quantity of positive cells. Few to moderate positive epithelial and fibroblast cells were observed in six patient tissue samples, but moderate to numerous positive cells (Fig. 2I) were observed in five patient tissue samples. In the control group, moderate to numerous numbers of positive cells were observed (Fig. 2J). The number of positive structures in the megaureters and control group did not differ (no statistical difference was found; Table 3). Strong positive correlation was observed between AT2R2- and MMP-2-positive endotheliocytes (rs = 0.601; p<0.001).
The MMP-2 finding in the megaureter group was variable. Mostly (in eight patients) few to moderate epitheliocytes contained MMP-2, but some specimens (in three patients) showed numerous positive cells in epithelium. In seven specimens, moderate positive connective tissue cells were detected, but numerous MMP-2-positive connective tissue cells contained three specimens. In most patients, few to moderate number of MMP-2-positive endotheliocytes were found (Fig. 2K). Meanwhile, the control group has occasional positive structures in epithelium, connective tissue, and endothelium (Fig. 2L). The amount of MMP-2 found in the megaureter group was significantly higher in comparison to the control group: MMP-2-positive epitheliocytes (U = 0.00; p=0.009), MMP-2-positive fibroblasts (U = 0.00; p=0.007), and MMP-2 endotheliocytes (U = 1.50; p=0.014) (Table 3). Spearman’s rank coefficient revealed very strong positive correlation between MMP-2-positive epitheliocytes and endotheliocytes (rs = 0.867; p<0.001) in the patient group.
SHH expression showed few positive cells in the patient group and occasional positive cells in the control group. Statistical tests confirmed that significantly more SHH-positive structures (U = 0.00; p=0.007) were seen in the megaureter group (Table 3).
TUNEL marked 81.25 ± 41.22 epithelial cells, moderate number of connective tissue and smooth muscle cells, and few amounts of endothelium cells in eight patients, while the control group showed 9.89 ± 13.99 epithelial cells and few number of connective tissue, smooth muscle, and endothelium cells. A statistically significant difference was seen between numbers of apoptotic epithelial cells (U = 2.00; p=0.021), and apoptotic smooth muscle cells (U = 4.50; p=0.046) (Table 3). Strong positive correlation was observed between apoptotic fibroblasts and smooth muscle cells (rs = 0.669; p=0.017), and apoptotic fibroblasts and endotheliocytes (rs = 0.731; p=0.007).
Discussion
The anthropometrical parameters of the ureter vary according to age. Especially this relates to the ureters of the first postnatal years. But this covers mainly the anthropological measurements like size, length, and some other indices of ureters. In our research, both—younger and later age children—demonstrated similar morphological results. Thus, we believe that the comparison and interpretation of results is possible based on the morphopathogenesis of POMs and their morphological appearance. At the beginning, megaureters were classified into four categories—obstructed, refluxing, refluxing with obstruction, and non-refluxing/non-obstructing. 41 It is possible for many kinds of stimuli to produce the same response in the tissue levels. We have based our analysis on morphological appearance, which was described in the Pfistere Henderen classification: type I involved the distal ureter without associated hydronephrosis; type II extended to both ureter and pelvis; and type III was associated with severe hydroureteronephrosis. 42
Previous studies have revealed that congenital megaureters present as qualitative and quantitative anomalies of connective and muscular tissue, as well as structural modifications of nervous tissue. 43 In our study, routine staining showed scattered epithelial vacuolization and atrophy of smooth muscle layers with merged, different thickness layers and with some vacuolization of smooth muscle cells. Blood vessel sclerosis was observed in some samples, which is consistent with previous reports.43,44 Payabvash and colleagues have extensively investigated the role of vascular and neural supply of myocyte apoptosis in POMs. Congenital defective vascular development may result in myocyte apoptosis. 44 Empty spaces caused by apoptosis may then be filled with connective tissue, rather than slowly migrating smooth muscle cells. 45
Very few studies have investigated the immunohistochemical profile of POM. In this study, we detected the appearance and distribution of several factors that participate in the regulation, innervation, vascularization, and growth/development of ureters and might take part in the pathogenesis of POM.
Prior series have reported the decrease in nerve endings in obstructed ureteral endings of patients with POM compared with controls. 44 Although Wang and colleagues described the marked reduction of PGP 9.5 and NGFR staining in nerve fibers of the muscle layers in case of ureteropelvic junction obstruction and they suggested that defective innervation may have an important role in the pathogenesis of ureteropelvic junction obstruction, 46 in our study, the appearance of PGP 9.5 and NGFR was observed in equal frequencies between POM and the control group. Our results are consistent with those of Gündüz et al., 47 who reported that there were no differences observed in the immunohistochemical staining for PGP 9.5 in normal and ureteropelvic junction obstruction specimens.
In this study, statistically significant differences in NGFR findings between the study and control groups were not detected. These findings are supported by another publication that evaluated the regulation of NGF production and NGFR in the urinary tract. 48 These findings suggest a rather nonspecific role of NGFR in the pathogenesis of POM.
The pathogenic role of TGFβ is still a matter of speculation in the context of POM. A common finding is the abundance of connective tissue in the abnormal ureter. 49 TGFβ has been reported to be dysregulated in malformed ureters. 50 In this study, we detected few TGFβ-positive cells in the epithelium and occasional TGFβ-positive cells in the connective tissue in the megaureter group. Compared with the control group, TGFβ-positive structures in connective tissues were observed in lower counts, but there was no statistically significant difference between the groups. Our findings suggest that a diminished TGFβ expression may be partly responsible for decreased tissue growth. This hypothesis is supported by the findings of Ozturk and colleagues, who described that TGFβ is highest in embryonic ureters in vivo and decreases postnatally. 19 Positive TGFβ immunoreactions were detected in the ureter from 6- to 12-month-old patients with obstructed primary megaureter. Such reactions weakened progressively in those patients older than 1 year, becoming negative in all children older than 3 years. 16
Many studies have shown how FGFR signaling is critical for kidney and lower urinary tract patterning in humans. 20 FGFR1 has been known to be strongly expressed in the developing ureteric wall. 51 The FGFR finding in this study was variable. Statistical tests confirmed that significantly more FGFR-positive structures were seen in the megaureter group. Moreover, FGFR1 is involved in the pathogenesis of renal fibrotic diseases. 52 The disturbances in the FGFR1 signaling pathway cause the apoptosis of endotheliocytes. 53 FGFR1 has a key role in the regulation of cell migration, and it has been confirmed as a key inhibitor of TGFβ-induced endothelial mesenchymal transition. 54 Therefore, it could be assumed that the more prominent FGFR1 finding points out a disbalance of tissue remodelation.
FGFR1 may be a specific target for MMP-2 on the cell surface that may modulate the mitogenic and angiogenic activities. 55 This could explain the tight correlation between FGFR1 and MMP-2 cells at the site of the megaureter. We found a significant increase of MMP-2-positive structures in POM-affected ureters compared with the controls. This shows that ureter tissue is undergoing remodelation. Matrix accumulation is a consequence of imbalance between matrix production and its degradation. The increased expression of MMP-2 has been shown in an experimental model of obstructive nephropathy 56 and in children with intrinsic ureteropelvic junction obstruction. 57 Du et al. 23 showed that MMP-2 may have a pathogenic role in renal interstitial fibrosis, possibly through the induction of epithelial-to-mesenchymal transition and macrophage infiltration. This is approved also in other author’s studies, outlining that MMP-2 is important in the development of renal fibrosis.56,58
The results of Wang and colleagues indicate that AT2 can increase the expression of MMP-2. AT2 also increases the expression of AT2R2. 59 This study demonstrated similar results—a strong positive correlation between AT2R2- and MMP-2-positive endotheliocytes was observed. However, similar findings of AT2R2 in megaureter and control groups showed that this factor has no specific role in morphopathogenesis of POMs.
Apoptosis has shown to play an important role during urinary tract development. 60 However, we found an increased prevalence of apoptotic epithelial and smooth muscle cells in the megaureter group. Multiple authors have observed increased apoptosis of cells in obstructions of ureter. For instance, smooth muscle cell apoptosis was significantly greater at the site of congenital ureteropelvic junction obstruction.35,61 Apoptosis could be an early event that occurs before subsequent fibrosis. 62 Kajbafzadeh and colleagues described positive correlations between smooth muscle cell apoptosis and the amount of collagen and elastin fibers. 35 Excessive apoptosis decreased the number of renal tubular epithelial cells and accelerated the progression of renal interstitial fibrosis. 63 The balance between proliferation and apoptosis is of critical importance in the process of tubulointerstitial fibrosis in response to ureteral obstruction. 62
SHH signaling is essential for tissue growth, cell proliferation, and differentiation in early ureter development. 31 It regulates epithelial–mesenchymal interactions during the development. 64 Sheybani-Deloui et al. 65 observed upregulated HH signaling-related genes in obstructing ureter tissue segments in 38% of children with ureteropelvic junction obstruction. In this study, significantly more SHH-positive structures were observed in the megaureter group. It cannot be excluded SHH promotes fibrosis in case of megaureter development. HH signaling is an important mediator of progressive fibrosis 66 and its profibrogenic effects have been demonstrated in various tissues. 67
POM morphopathogenesis involves a dynamic apoptotic cell death of epithelium and smooth muscle as well as tissue degradation in epithelium, connective tissue, and blood vessels of the ureter wall. The decrease of tissue growth through diminished TGFβ expression and stimulation of FGFR1 and MMP-2 suggests a disbalance of tissue remodelation in the megaureter wall.
Footnotes
Acknowledgements
Our greatest thanks to Assoc. Prof. Jānis Dobelis and Dr Ainārs Ģīlis for providing the study specimen.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: conception and design (AJ, MP), collection and/or assembly of data (IS, AP), data analysis (AJ, IS), data interpretation (AJ, AP, MP), and article writing (AJ, IS, MP). All authors have read and approved the final article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
