Abstract
Congenital renal dysplasia (RD) is a severe form of congenital renal malformation characterized by disruption of normal renal development with cyst formation, reduced or absent nephrons, and impaired renal growth. The authors previously identified that matrilysin (matrix metalloproteinase–7) was overexpressed in a microarray gene expression analysis of human RD compared to normal control kidneys. They now find that active matrilysin gene transcription and protein synthesis occur within dysplastic tubules and epithelial cells lining cysts in human RD by RT-PCR and immunohistochemistry. Similar staining patterns were seen in obstructed kidneys of pouch opossums that show histological features similar to that of human RD. In vitro, matrilysin inhibits formation of branching structures in mIMCD-3 cells stimulated by bone morphogenetic protein–7 (BMP-7) but does not inhibit hepatocyte growth factor–stimulated branching. BMP-7 signaling is essential for normal kidney development, and overexpression of catalytically active matrilysin in human embryonic kidney 293 cells reduces endogenous BMP-7 protein levels and inhibits phosphorylation of BMP-7 SMAD signaling intermediates. These findings suggest that matrilysin expression in RD may be an injury response that disrupts normal nephrogenesis by impairing BMP-7 signaling.
Congenital renal dysplasia (RD) represents a collection of disorders in which the kidney begins to form but fails to differentiate normal nephron and collecting duct structures and is an important cause of end-stage renal disease in childhood (Woolf et al. 2004; Winyard and Chitty 2008). Dysplastic kidneys are variably cystic with reduced or absent nephrons and impaired growth. RD pathology is characterized by abnormally thick parenchymal vessels and primitive collecting ducts and tubules surrounded by characteristic smooth muscle whorls and, variably, presence of cartilage. About half of children with congenital RD have obstructive nephropathy (Woolf and Thiruchelvam 2001). Animal models of urinary tract obstruction during renal development in rabbits, opossums, sheep, swine, and, more recently, newborn rats and mice have recapitulated histological aspects of human RD (Peters 2001; Bascands and Schanstra 2005). Insights gained from genetically engineered mice have linked several genes with dysplasia phenotypes. For example, Wt1, Pax2, Gdnf, Cret, At1r, Glyp3, Wnt4, and Bmp7 mutant mice have absent, hydronephrotic, or dysplastic kidneys (Lipschutz 1998; Liapis 2003). Studies of syndromic human RD have associated renal agenesis and/or RD with a variety of associated heritable syndromes with multiorgan involvement, suggesting that kidney damage may result not from a primary single constitutional gene mutation but rather from defective local gene control of renal outgrowth (Woolf et al. 2004). Recently, mutations in individual single genes, such as TCF2/hepatocyte nuclear factor 1β (HNF1β), PAX2, RET, and ROB02, were described, but these are very rare (Winyard and Chitty 2008).
We previously used an unbiased microarray approach to investigate gene transcription associations in congenital human RD (Jain et al. 2007). One gene that was highly upregulated in dysplastic kidneys compared to normal controls was the gene for matrilysin (MMP7), a secreted member of the matrix metalloproteinase (MMP) family of proteolytic enzymes that had not previously been associated with developmental abnormalities in the kidney or other tissues. Matrilysin is not detected in normal kidneys in humans or in mice; however, we found matrilysin to be highly expressed in polycystic kidney disease and hydronephrosis. Expression was markedly induced in collecting duct epithelium by ureteral obstruction or folic acid nephropathy in mice (Surendran et al. 2004). In the respiratory and gastrointestinal tracts, matrilysin expression is induced by acute epithelial injury and functions in repair to promote re-epithelialization (Dunsmore et al. 1998; McGuire et al. 2003; Swee et al. 2008). However, matrilysin may also contribute to pathology. Studies with matrilysin-null mice have shown that matrilysin may promote excessive inflammation (Li et al. 2002), and elevated matrilysin expression has been associated with fibrosis in the lung and liver (Lichtinghagen et al. 2001; Li et al. 2002; Zuo et al. 2002; Huang et al. 2005; Rosas et al. 2008).
Matrilysin is tightly associated with cell surface heparan sulfated proteoglycans (Yu and Woessner 2000), and this interaction localizes the proteinase with heparin-binding matrilysin substrates such as heparin-binding epidermal growth factor and C type lectin member family member 3A (Yu et al. 2002; Cheng et al. 2007; Lynch et al. 2007; Tsunezumi et al. 2009). Bone morphogenetic protein (BMP-7) and hepatocyte growth factor (HGF) are heparin-binding proteins that are critical in normal renal development (Davies and Fisher 2002; Irie et al. 2003; Takada et al. 2003), and our prior microarray study demonstrated that BMP7 gene expression and BMP-7 tissue levels were reduced in dysplastic kidneys compared to normal controls (Jain et al. 2007). Thus, we hypothesized that an inappropriate increase in matrilysin during renal tubular development could activate an “injury response” that may disrupt normal nephrogenesis and contribute to RD, possibly by disrupting signaling of heparin-binding growth factors such as BMP-7 and HGF. To address this, we characterized matrilysin expression in human RD and in an opossum model of experimental RD and used a cell-based system to evaluate matrilysin effects on growth factor–stimulated renal tubular morphogenesis in vitro.
Materials and Methods
Tissue Samples
This study was approved by the Washington University Human Studies Committee (IRB 03–104). Kidneys (n=10) were selected from nephrectomy or fetal autopsy specimens. Nephrectomies were performed because of severe cystic disease complicated by either recurrent infection or severe renal scarring (atrophy) at the time of renal transplantation. Tissues were collected within 1 hr of resection. A 10 × 10-mm section was frozen, embedded in OCT medium (Tissue-Tek, Sakura USA, Torrance, CA), and stored in the Washington University Department of Pathology tissue bank. Samples were anonymized of individual patient identifiers. All but 1 patient (case 3) had sporadic kidney disease. All patients had clinical history of obstructive nephropathy, and all were male. Three patients had unilateral kidney disease, and 2 had bilateral hydronephrosis (1 of whom was a patient with type 1 neurofibromatosis). A section from the frozen block was stained with hematoxylin and eosin for histological assessment. Control kidneys were selected from mid-late gestation fetal autopsies when nephrogenesis is active. All control kidneys were grossly normal. Gestational age was assessed by counting glomerular generations as previously described (Jain et al. 2007).
To evaluate matrilysin expression in experimental RD, existing sections of kidneys collected from North American opossum pups that had been subjected to 14 days of unilateral ureteral obstruction beginning at 21 days of life were used (Liapis et al. 2001).
Cell Culture
Mouse inner medullary collecting duct cells (mIMCD-3, ATCC CRL-2123) and human embryonic kidney (HEK) 293 cells (ATCC CRL-1573) were obtained from American Type Culture Collection (ATCC, Manassas, VA) and cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum and penicillin/streptomycin. The m-IMCD3 cell line is derived from the terminal inner renal medullary collecting duct of the SV40 transgenic mouse, and mIMCD-3 cells retain several differentiated characteristics of the collecting duct nephron segment (Rauchman et al. 1993). Unless otherwise specified, culture media and supplements were obtained from Mediatech (Manassas, VA). BMP-7 was kindly supplied by Dr. Keith Hruska (Washington University, St. Louis, MO). The HGF was obtained from Sigma-Aldrich (St. Louis, MO). Recombinant active human matrilysin was obtained from Calbiochem (San Diego, CA). Type 1 rat collagen was obtained from BD Biosciences (San Jose, CA).
An auto-activating human matrilysin construct was used to transfect HEK 293 cells (ATCC cell line CRL-1573). This construct contains a furin cleavage site (RRKKR) inserted at the junction between the pro- and catalytic domains of the full-length human pro-matrilysin gene (kindly provided by Dr. David Madtes, MD, Fred Hutchinson Cancer Research Center, Seattle, WA). HEK 293 cells were cultured in DMEM supplemented with 10% fetal bovine serum (Mediatech) in a 37C incubator with 5% C02. Transfections were done with Lipofectamine 2000 (Invitrogen, Carlsbad, CA) using manufacturer’s protocol. Several single colonies were isolated from a 100-mm culture dish with 150 µl cloning cylinders (Sigma-Aldrich). Selection was maintained with 0.5 mg/ml Geneticin (Invitrogen). Expression of human MMP-7 was analyzed by reverse transcriptase polymerase chain reaction (RT-PCR) with primers for human MMP-7 cDNA. Multiple clones expressing MMP-7 mRNA were detected by RT-PCR, and 2 clones were selected for further experiments based on detection of high levels of the 19 kD active MMP-7 protein in the conditioned medium by Western blotting. Detection of pro- and active MMP-7 was done by Western blotting of conditioned media collected from the transfected cells using rabbit anti-human MMP-7 antibody AB-4 (EMD Biosciences, Gibbstown, NJ).
RNA Analysis
At least 10 µg of total RNA was isolated from each kidney sample using Trizol reagent following the supplier’s protocol (Invitrogen). RNA was further purified using the RNeasy total RNA clean up protocol (Qiagen, Valencia, CA). RNA was quantified by fiber optic UV spectroscopy (Nanodrop Technologies, Wilmington, DE). Total RNA was isolated from HEK 293 cells with Qiagen RNeasy according to supplier’s protocol and quantified by UV spectroscopy. For semiquantitative RT-PCR analysis, 1 µg of total kidney RNA was reverse transcribed with random primers using Sigma Enhanced Avian RT kit (Sigma-Aldrich) followed by PCR according to manufacturer’s protocol. The primers used for specific PCR amplification were as follows: for matrilysin (MMP-7), 5’-ACAGGTGCAGCTCAGGAAGG-3’; reverse, 5’-GTGA-AGGACGCAGGAGTGAAC-3’; for glyceraldehyde 3- phosphate dehydrogenase (GAPDH), forward, 5’-CACTGA-CACGTTGGCAGTGG-3’; reverse, 5’-CATGGAGAAGG-CTGGGGCTC-3.’ The primers for each gene were designed from different exons so as to distinguish between products made by genomic DNA amplification versus cDNA amplification, and control PCR reactions were done without template cDNA (data not shown). Conditions were optimized to ensure that reactions were in the linear range, and PCR products were resolved on agarose gels, stained with ethidium bromide, and recorded using a Kodak EDAS 290 gel imaging system. Densitometry calculations were done using ImageJ software (NIH, Bethesda, MD). For real-time reverse-transcription PCR (qRT-PCR), 250 ng total RNA was reverse transcribed with a High-Capacity cDNA Archive kit (ABI-Applied Biosystems, Foster City, CA). Unlabeled PCR primers and TaqMan FAM dye-labeled probes for target genes were obtained from ABI. qRT-PCR reactions were performed according to the manufacturer’s protocol with an ABI HT7900 Real-Time PCR System using TaqMan reagents for 40 cycles. The threshold cycle (Ct) for each probe set/cDNA was obtained from triplicate samples and averaged, and the ΔCt was the calculated difference between the average Ct for the target gene and the average Ct for glyceraldehyde-3-phosphate dehydrogenase as a control for total starting RNA quantity. Gene expression relative to control (RQ) was determined as 2-ΔΔCt.
Immunohistochemistry
Sections were stained for matrilysin as described (McGuire et al. 2003; Jain et al. 2007). Monoclonal antibody against human pro-matrilysin (clone MAB907) was obtained from R & D Systems (Minneapolis, MN). Briefly, sections (5 µm) were collected on lysine-coated slides, rehydrated, rinsed in Tris-buffered saline (TBS) for 10 min, incubated in 3% H202 in methanol to quench endogenous peroxidase activity, blocked, incubated (overnight at 4C) with the antibody, washed, incubated with biotinylated secondary antibody (Vector Labs, Burlingame, CA) followed by streptavidin peroxidase-conjugated tertiary antibody, rinsed in TBS, and finally developed with diaminobenzidine (Vector Labs) and counterstained with hematoxylin for visualization. Opossum tissues were stained with anti-human matrilysin antibodies as for human tissue samples, and antibodies cross-reacted with opossum tissues. For lectin staining, fixed, rehydrated sections were incubated with rhodamine-conjugated peanut agglutinin lectin (arachis hypogaea [AH], Vector Labs) or FITC-conjugated winged pea (lotus tetragonolobus [LT]) lectin (Vector) at 10 µg/mL for 30 min at 37C. Indirect immunofluorescence microscopy was used to image fluorescent markers.
In Vitro Branching Morphogenesis Assay
The in vitro renal tubule branching morphogenesis assay was performed as described by Piscione et al. (1997). Collagen gels were prepared by mixing 16 µl of 1M HEPES (Sigma-Aldrich), 32 µl of 1M NaHC03, 160 µl DMEM-F12, 800 µl rat type I collagen, and 1 × 106 mIMCD-3 cells. Aliquots (50 µl/well = 5 × 104 cells/well) were seeded in 96-well round-bottomed culture plates, and gels were allowed to solidify at 37C; 100 µl of DMEM-F12 containing 5% fetal bovine serum was added to each well, and cultures were maintained at 37C in 5% C02 for 48 hr. For stimulation experiments, BMP-7, HGF, and matrilysin were diluted into culture media to achieve desired final concentrations immediately prior to adding media to wells with collagen gels. After 48 hr in culture, gels were fixed in neutral buffed formalin for 10 min at room temperature. Fixed gels were then washed 4 times in PBS and stored at 4C. After fixation, gels were imaged by differential interference contrast microscopy using an Olympus BX51 microscope and at least 4 low-power field (lpf) images s/gel photographed with an Olympus DP70 digital camera with the ×10 objective. Images were then imported into Adobe Photoshop and overlaid with a counting grid dividing each lpf image into 16 equal divisions. Quantification of branching morphogenesis was determined by counting the total number of continuous, elongated, linear structures per photograph and averaging 4 separate lpf images per gel for each culture condition.
Western Blot Analysis
Antibodies against anti-human Smad-1 (#9512) and rabbit anti-human-phospho-Smad1 (Ser463/465)/Smad5 (Ser463/465)/Smad8 (Ser426/428) (#9511) were obtained from Cell Signaling Technology (Danvers, MA). Sheep polyclonal anti-human-BMP-7 (OP-1) antibody was obtained from Dr. Keith Hruska at Washington University in St. Louis, Missouri. Cultured cells were lysed in RIPA lysis buffer (30 mM NaCl, 50 mM Tris-HCl, pH 7.4, 5 mM EDTA, 1% Igepal CA630, 1% deoxycholic acid, 0.1% SDS, with Roche Complete protease inhibitor cocktail), and protein content in lysates quantified by the Bradford assay. Equal quantities (10 µg) of protein/sample were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), transferred to Hybond nitrocellulose membranes (Amersham Pharmacia Biotech, Buckhamshire, UK), and blocked overnight with 5% milk in TBS with 0.05% Tween-20. Blots were probed with above antibodies at manufacturers’ recommended concentrations and visualized by enhanced chemiluminescence system (Amersham Pharmacia Biotech).
In Vitro Cleavage Assay
Human recombinant BMP-7 and matrilysin were incubated in MMP cleavage buffer (10 mM HEPES, pH 7.4, 5 mM CaC12, 150 mM NaCl) at 37C at a 1:1 molar ratio. At 0, 0.5, 1, 2, 4, 8, and 24 hr after initiation of reaction, 10 µL aliquots of the reaction mixture were removed and the reaction stopped by addition of EDTA (2 mM final concentration). For the zero hour time point, EDTA was added at the start of the incubation period. An equal amount of 2X SDS-PAGE loading buffer was added to each sample, and samples were resolved by SDS-PAGE (net mass of 1 µg of BMP-7 per lane), followed by staining gels with Coomassie blue, and imaging with a digital gel imaging system. Subsequent reactions were performed at varying molar ratios of proteinase to substrate (ranging from 1:1 to 1:1000) and analyzed by SDS-PAGE. The net concentration of BMP-7 in each reaction was 100 µg/mL, and concentrations for MMP-7 ranged from 100 µg/mL (at 1:1 molar ratio) to 100 ng/mL (at 1:1000 molar ratio).
Results
Matrilysin Expression in Human Congenital RD
To verify matrilysin mRNA expression, differences previously reported in our microarray expression study of human RD (Jain et al. 2007), matrilysin mRNA expression was assessed in samples of midgestational fetal and postnatal dysplastic kidneys and normal control fetal kidneys by semiquantitative RT-PCR and quantitative real-time RT-PCR (qPCR). Matrilysin mRNA expression was detected in 4 of 5 RD samples by RT-PCR and was expressed at low levels in only 2 of 5 control samples (Fig. 1A,B), and average matrilysin expression in RD samples was significantly higher than in controls (p<0.05). Similar results were obtained using qPCR (Fig. 1C). Therefore, by both Affymetrix microarray and 2 RT-PCR methods of RNA analysis, matrilysin gene expression is significantly increased in human RD as compared to normal control kidneys.
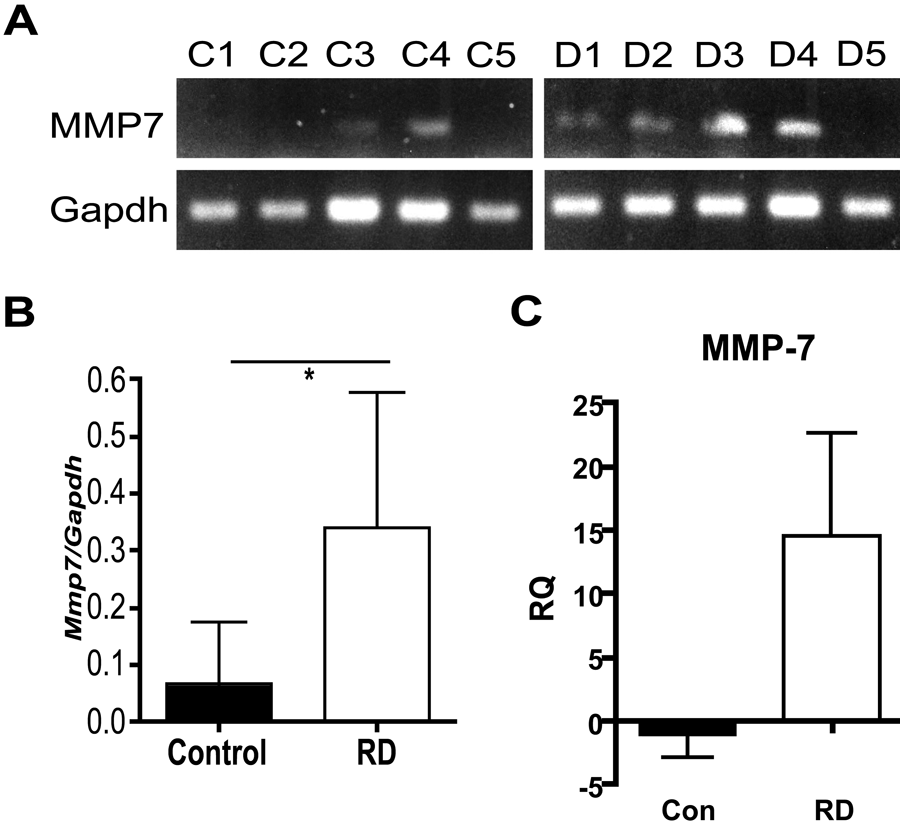
Matrilysin gene expression is significantly increased in human renal dysplasia (RD). (A) Total mRNA from samples of human dysplastic kidneys (D1-D5) and control fetal kidneys (C1-C5) was isolated and matrilysin (Mmp7) and glyceraldehyde-3-phosphodehydrogenase (Gapdh) gene expression determined by semiquantitative RT-PCR in individual samples. (B) Densitometry analysis shows average matrilysin expression in RD samples to be significantly greater than in control kidneys (normalized to Gapdh, mean ± SD, *p<0.05 by Student’s t-test). (C) Matrilysin expression as assessed by quantitative real-time PCR shows markedly higher average levels in RD samples compared to control (RQ = relative expression related to control sample 1).
Immunohistochemistry was used to localize matrilysin protein expression in RD tissue sections. Prominent matrilysin staining was observed in 7 of 8 RD samples assessed and localized to epithelial cells lining cysts and dilated tubules (Fig. 2A,C). In atrophic tubules, strong matrilysin staining was seen at the apical/lumenal surface and was associated with intralumenal proteinaceous debris (Fig. 2A, inset). Although low levels of matrilysin mRNA were detected in 2 normal fetal kidney samples by RT-PCR, matrilysin protein was not detected in normal fetal kidney tissue sections (n=3) (Fig. 2B). Consistent with our reported observations in autosomal-dominant polycystic kidney disease and hydronephrosis (Surendran et al. 2004), staining was not seen in interstitial or inflammatory cells. Sections stained for the distal tubule/collecting duct–specific rhodamine-conjugated peanut agglutinin lectin (AH lectin) and the proximal tubule–specific FITC-conjugated LT lectin showed that matrilysin expression in RD tissues was limited to epithelial cells originating from the distal nephron (Fig. 2C,D).
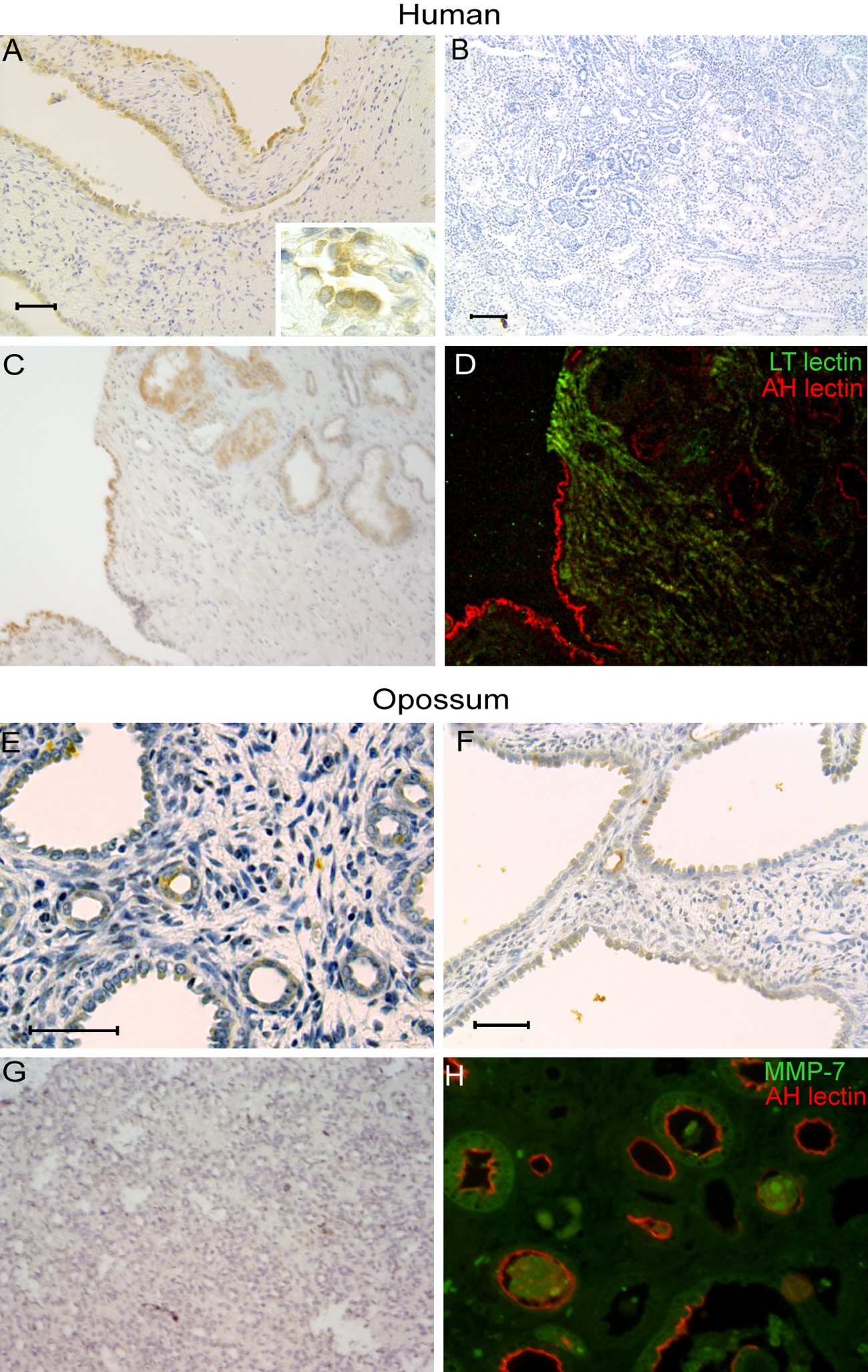
Matrilysin localizes to atrophic tubules and epithelium lining cysts and dilated tubules in human renal dysplasia (RD) and experimental kidney obstruction in pouch opossums. Samples of human RD and opossum kidneys at 14 days after unilateral ureteral obstruction were stained for matrilysin. (A, C) Strong matrilysin staining is seen in epithelium lining cysts and dilated tubules (peroxidase with diaminobenzidine stain and hematoxylin nuclear counter stain). (B) Lack of matrilysin staining in normal fetal human kidney. (C, D) Serial section from panel (B) stained with FITC-conjugated lotus tetragonolobus lectin (green) and rhodamine-conjugated peanut agglutinin (red) shows that matrilysin staining is mainly associated with epithelium derived from distal tubules/collecting ducts. (E-G) Matrilysin staining pattern in obstructed opossum kidney is similar to that seen in human RD tissues and is not seen in contralateral unobstructed pouch opossum kidney. (H) Staining for matrilysin (green) and peanut agglutinin (AH, red) in obstructed opossum kidney shows matrilysin localizing to epithelium of distal nephron origin. Bars: A, E, F = 50 µm; B = 100 µm.
Complete unilateral ureteral obstruction in North American opossum pups produces pathology with histological features that mimic that of human RD with obstruction resulting in fewer glomerular generations and distinct patterns of interstitial fibrosis characterized by concentric collars of mesenchyme around primitive ducts (Liapis et al. 2001; Steinhardt et al. 1994). Thus, to assess if matrilysin expression was induced in a prospective model of renal obstruction during development, sections of pouch opossum kidneys that were obstructed at 21 days of age were stained for matrilysin by immunohistochemistry at 14 days after unilateral ureteral obstruction. Similar to the pattern seen in human RD, matrilysin staining was observed in epithelial cells lining dilated tubules and cystic structures, and prominent staining was associated with atrophic tubules (Fig. 2E,F). Matrilysin staining was not observed in sham operated pouch opossum kidneys (Fig. 2G). As was observed in human MDCK sections, localization of matrilysin in opossum kidney with rhodamine-conjugated peanut agglutinin (AH lectin) showed matrilysin was expressed in tubules of distal nephron origin (Fig. 2H). These findings demonstrate that matrilysin expression is induced in a model in which the response to early fetal urinary tract obstruction leads to a dysplastic phenotype.
Matrilysin Inhibits BMP-7 Induced Tubulogenesis In Vitro
To evaluate the effects of matrilysin on renal tubular function, we used a well-characterized in vitro branching morphogenesis model in which mouse inner medullary collecting duct cells (mIMCD-3) cultured in collagen gels are induced with nephrogenic growth factors to form three-dimensional chord-like tubular structures (Piscione et al. 1997; Sakurai and Nigam 1998; Karihaloo et al. 2001). In this model, HGF stimulates branching and BMP-7 has dose-dependent effects, with low doses stimulating branching and high doses inhibiting branching morphogenesis (Cantley et al. 1994; Piscione et al. 1997). To assess whether matrilysin affected growth factor–stimulated tubulogenesis in this system, 5 × 104 mIMCD-3 cells/well were seeded into collagen gels in 96-well tissue culture plates and stimulated with medium supplemented with BMP-7 (low dose = 10 ng/mL or high dose = 40 ng/mL) or HGF (10 ng/ml) with or without the addition of matrilysin (25 or 200 ng/mL). Consistent with results reported by others (Cantley et al. 1994; Piscione et al. 1997), low-dose BMP-7 and HGF stimulated formation of elongated, branched tubule-like structures above that induced by serum alone, and high-dose BMP-7 modestly inhibited tubule formation (Fig. 3, 4). Matrilysin had no effect on branching morphogenesis induced by HGF, but matrilysin inhibited BMP-7 induced effects in a dose-dependent manner, with the higher concentration of matrilysin completely inhibiting BMP-7 morphogenic effects (Fig. 4). These observations indicate that matrilysin did not have a generalized inhibitory or toxic effect on mIMCD-3 cells, but rather it specifically inhibited BMP-7–dependent stimulation of in vitro tubulogenesis.

Matrilysin inhibits BMP-7 stimulated branching morphogenesis in vitro. mIMCD-3 murine collecting duct epithelial cells were seeded in collagen gels and stimulated for 48 hr with medium with fetal bovine serum alone or supplemented with BMP-7 (10 ng/mL) or HGF (10 ng/mL). Gels were imaged by differential interference contrast microscopy. Both BMP-7 and HGF stimulate formation of branched tubular structures, but addition of MMP-7 inhibits branching-induced BMP-7 with no effect on HGF-stimulated branching. Insets in each panel show higher magnification views of each condition tested. Bars = 50 µm.
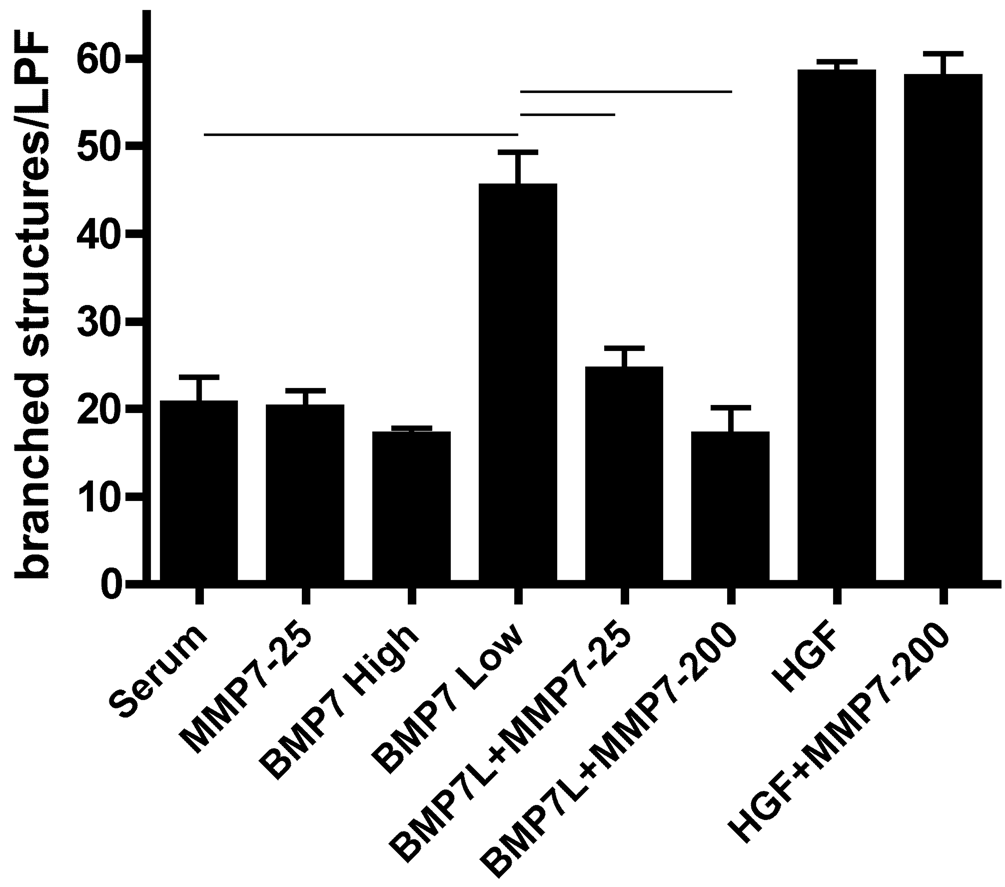
Matrilysin inhibition of BMP-7 branching morphogenesis is dose dependent. In vitro branching morphogenesis was quantified by counting number of branched structures per low power field (lpf) using a counting grid. BMP-7 low (L) = 10 ng/mL, BMP-7 high = 40 ng/mL, HGF = 10 ng/mL, MMP-7 = 25 or 200 ng/mL (p<0.001 for indicated comparisons by ANOVA with Bonferroni’s multiple comparisons posttest; N=4 gels/per condition, and data shown are representative of 3 independent experiments).
Matrilysin Inhibits Endogenous BMP-7 Signaling in Human Embryonal Kidney 293 Cells
We assessed whether expression of active matrilysin in cells that express endogenous BMP-7 alters BMP-7 signaling. HEK 293 cells express BMP-7 RNA and protein but do not express endogenous matrilysin. Therefore, HEK 293 cells were transfected to stably express an auto-activating mutant human matrilysin with a furin cleavage site inserted between the prodomain and active enzyme such that the zymogen is activated in the secretory pathway by furin (Fig. 5A). The furin cleavage site auto-activating matrilysin (FCS-aMat) was used because prior studies show that ectopically expressed promatrilysin is not efficiently activated in vitro in epithelial cells (Rudolph-Owen et al. 1998; Hotary et al. 2000). Expression of FCS-aMat reduced levels of BMP-7 protein in HEK 293 cells but did not alter BMP-7 mRNA levels as assessed by qRT-PCR (Fig. 5B,C). BMP-7, like other TGF-β superfamily ligands, binds to cell surface receptors and signals by phosphorylation of SMADs-1, 5, and 8. Consequently, activation of SMAD phosphorylation is a reliable readout of BMP signaling activity (Macias-Silva et al. 1998; Miyazono et al. 2000). Concomitant with reduction in BMP-7 protein levels, expression of active matrilysin in 293 cells reduced phosphorylation of SMAD-1 as assessed by Western blotting with a phospho-SMAD specific antibody (Fig. 5). Total SMAD-1 protein levels were unchanged, indicating that the reduction in phospho-SMAD was due to reduced activation rather than to SMAD protein degradation or reduced SMAD protein production. Taken together, these observations show that matrilysin inhibits endogenous BMP-7/SMAD signaling in HEK 293 cells.
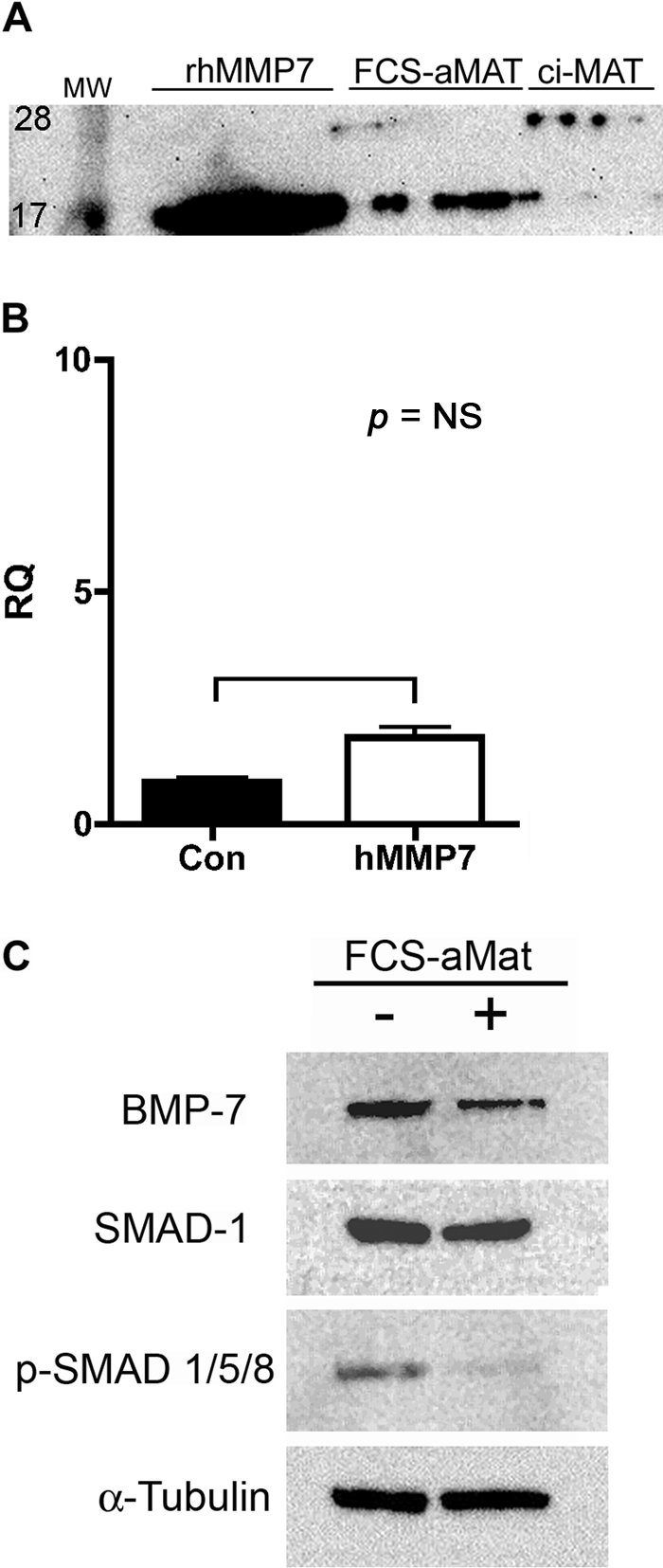
Matrilysin reduces BMP-7 protein expression and inhibits BMP-SMAD signaling. Human embryonal kidney (HEK) 293 cells were transfected to express a furin-cleavage site containing catalytically active human matrilysin (FCS-aMAT). (A) Immunoblotting of condition medium shows secretion of active matrilysin (FCS-aMAT) at same molecular weight (19 kD) as that of recombinant human matrilysin protein (rhMMP7). A catalytically inactive MMP7 is secreted at the predicted 29 kD molecular weight of promatrilysin. (B) Matrilysin expression did not significantly alter BMP-7 mRNA expression in HEK 293 cells as assessed by qRT2-PCR. (C) Compared to vector-transfected cells, FCS-aMAT cells had lower BMP-7 protein expression and reduced levels of SMAD-1/5/8 phosphorylation. Total SMAD-1 and alpha-tubulin were used as protein loading controls. Data are representative of 2 independent clones and 3 separate experiments.
Because matrilysin expression reduced BMP-7 protein levels and SMAD signaling without direct changes in BMP-7 mRNA expression, an in vitro cleavage assay was used to evaluate if matrilysin can directly cleave BMP-7. Incubation of purified human recombinant BMP-7 with activated human matrilysin at equimolar concentrations in an MMP cleavage assay buffer at physiologic pH and temperature was done for various time points, and reactions were stopped by addition of EDTA. Staining reactions products with Coomassie blue showed that matrilysin cleaved BMP-7 within 0.5 hr and nearly completely degraded BMP-7 within 8 hr (Fig. 6A). Inhibition of cleavage by EDTA indicated that BMP-7 proteolysis was dependent on MMP-7 activity. A separate gel was run and transferred to PVDF membrane for Western blotting, and an anti–BMP-7 antibody detected the monomeric recombinant BMP-7 at the expected molecular of approximately 17 kD (Fig. 6B). As expected, there was no reaction with the matrilysin lane. At 0.5 hr after the start of the reaction, an approximately 15-kD band was visible, which correlated with the major intermediate cleavage product seen on the Coomassie stained gel (indicated by asterisk). Lower molecular weight BMP-7 fragments at 5 to 7 kD were seen on the gel, but these were not recognized by the antibody. As expected, the MMP-7 band was not recognized by the anti-BMP-7 antibody. To assess matrilysin affinity for BMP-7 as a potential substrate, reactions were prepared at MMP-7:BMP-7 molar ratios ranging from 1:1 to 1:1000. After a 2-hr incubation, the 1:1 and 1:10 samples showed complete cleavage of the BMP-7 to the 15-KD product. At the ratios, the MMP-7 quantity was below the detection limit of the Coomassie stain; however, the 1:100 lane showed greater than 50% cleavage, and the 1:1000 lane showed a faint band at 15 kD. These data indicate that matrilysin efficiently cleaves BMP-7 in vitro, even at low proteinase to substrate ratios, and raise the possibility that BMP-7 is a direct substrate of matrilysin in vivo.
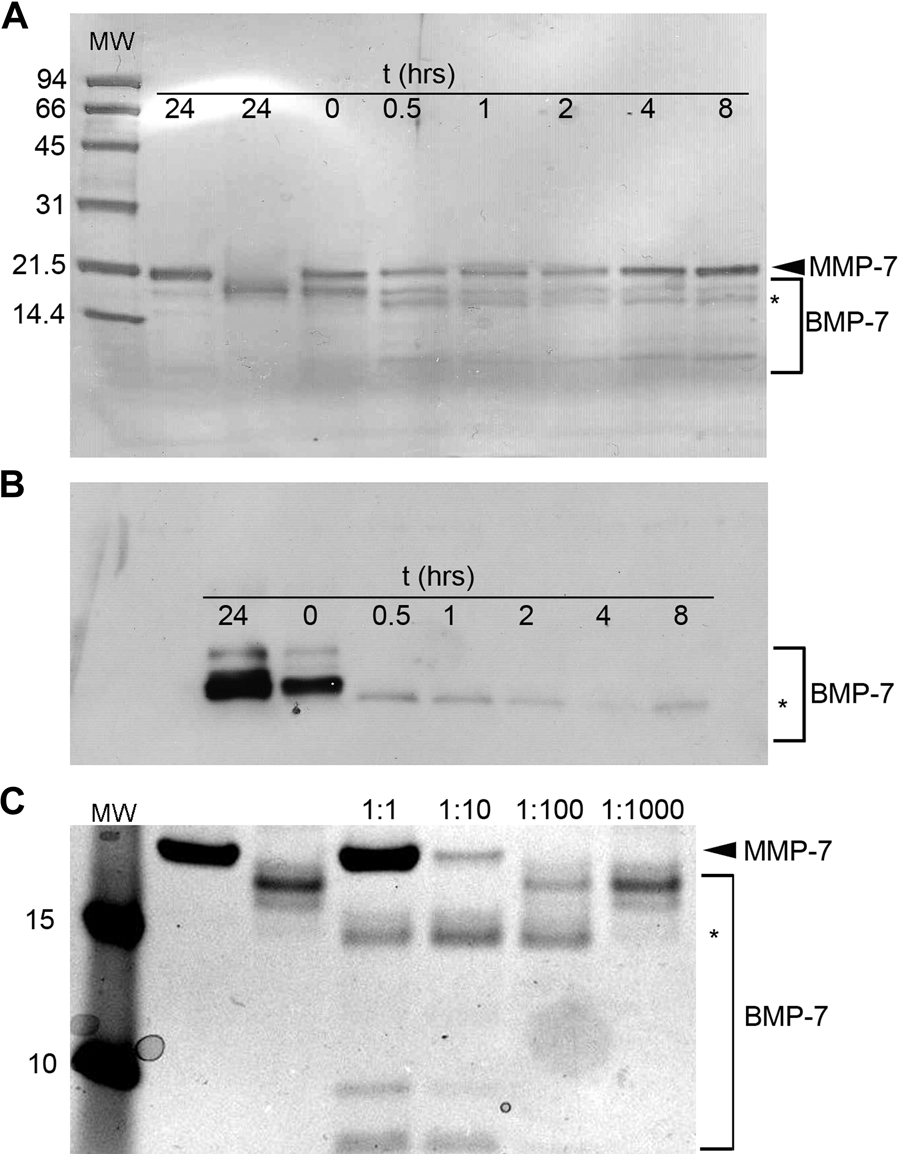
Matrilysin cleaves BMP-7 protein. (A) An in vitro cleavage assay was performed, reaction products were resolved by SDS-PAGE, and the gel was stained with Coomassie blue and digitally imaged. Matrilysin cleaved BMP-7 by 0.5 hr and produced lower molecular weight cleavage products with near complete degradation of the 17-kD BMP-7 monomer within 8 hr. Matrilysin and BMP-7 alone are stable over a 24-hr time course of incubation in reaction buffer. (B) Western blotting of reaction products shows that an approximately 15-kD BMP-7 fragment is recognized by the anti–BMP-7 antibody (noted by asterisk). (C) Reactions were set up using progressively lower ratios of proteinase to substrate and incubated for 2 hr. Matrilysin efficiently cleaved BMP-7 at a ratio as low as 1:100.
Discussion
Matrilysin is constitutively expressed in epithelial cells lining ducts of human exocrine glands, but it is generally not expressed in other normal, healthy human tissues, including the kidney. Our new data confirm our previous microarray observations of increased matrilysin mRNA expression in human RD and localize matrilysin protein expression in RD tissues to epithelial cells lining cysts and to atrophic tubules. Matrilysin is induced in the distal nephron in a variety of renal tubular disease processes in humans and in experimental renal tubular injury in mice (Surendran et al. 2004), suggesting that induction of matrilysin expression is a generalized response to injury in kidney collecting ducts. Experimental renal injury induced by complete ureteral obstruction in the developing pouch opossum kidney results in tubular and glomerular cysts, interstitial fibrosis, and impaired renal development. The pattern of matrilysin staining in the fetal opossum kidney at 2 weeks after obstruction is strikingly similar to that seen in human RD samples. Hence, an induction of matrilysin is likely an “injury response” in the developing nephron in RD patients. Although we did not investigate the mechanisms responsible for inducing matrilysin expression in RD, matrilysin is a target gene of the beta catenin pathway (Crawford et al. 1999; Crawford et al. 2001), and a recent report showing that elevation of beta catenin levels causes dysplasia in mice (Bridgewater et al. 2011) raises the possibility that matrilysin activity could be a downstream effector mechanism of pathological beta catenin signaling.
We have previously shown that matrilysin contributes to re-epithelialization in airways and in colonic mucosal epithelium (McGuire et al. 2003; Chen et al. 2008; Swee et al. 2008). However, matrilysin physiological functions require that the enzyme be expressed and activated at the appropriate time and place; and when activity is dysregulated or uncontrolled, matrilysin can contribute to pathology by promoting apoptosis, abnormal differentiation, or hyperproliferation (Ii et al. 2006; McCaig et al. 2006; Sawey et al. 2007). We speculate that high expression of matrilysin during nephrogenesis, a time and location in which it is either typically absent or present at very low levels in normal kidneys, interferes with normal developmental programs by contributing to the pathological tissue remodeling in RD.
BMP-7 has pleotropic functions during kidney development, including transformation of mesenchyme to epithelia and growth and maturation of the collecting ducts, and is essential for normal kidney development (Dudley et al. 1995; Luo et al. 1995; Archdeacon and Detwiler 2008). Observations made in Bmp7-null mice demonstrate that BMP-7 signaling is not necessary for normal inductive tissue interactions but that in the absence of BMP-7, there is a reduction in ureteric bud branching and loss of metanephric mesenchyme via apoptosis. Thus, BMP-7 is necessary for continuous growth and morphogenesis during later stages of renal development. Our results show that matrilysin inhibits BMP-7 dependent branching morphogenesis, reduces BMP7 levels in HEK 293 cells, and impairs BMP signaling in vitro and raise this as a potential mechanism contributing to RD pathogenesis. Matrilysin, BMP-7, and HGF are also heparin-binding proteins, and cell surface heparan-sulfated proteoglycans may regulate interactions between matrilysin and potential substrates. However, a lack of effect on HGF-induced branching morphogenesis in vitro suggests that the effect is specific for BMP-7. Our prior microarray data showed significantly reduced BMP-7 mRNA expression in RD samples compared to that of normal controls (Jain et al. 2007). In the new data presented here, matrilysin expression did not directly reduce BMP-7 gene expression in epithelial cells; however, our data suggest that matrilysin may inhibit BMP-7 effects by proteolytic degradation, a possibility supported by our observations of reduced BMP-7 staining in RD tissues (Jain et al. 2007) and reduced levels of BMP-7 protein in HEK 293 cells overexpressing matrilysin. Thus, BMP-7 in RD could be reduced by both transcriptional and post-transcriptional mechanisms.
Taken together, our data indicate that matrilysin may contribute to RD pathology by disrupting BMP-7 dependent effects on normal tubular development. Matrilysin can cleave multiple substrates in the pericellular environment, including growth factors, proteoglycan core proteins, matrix proteins, and cell adhesion receptors (Li et al. 2002; Yu et al. 2002; McGuire et al. 2003; Nakamura et al. 2005). Elucidating the specific processes and pathways by which matrilysin inhibits BMP-7 effects in the developing kidney and determining whether BMP-7 is a direct substrate of matrilysin proteolytic activity is the focus of ongoing work in our laboratory.
Footnotes
Acknowledgements
We thank Drs. William C. Parks, Peter Chen, Anne Manicone, and Keith Hruska for helpful discussions; Dr. Hruska for providing reagents and cell lines; and David Madtes for the auto-activating matrilysin construct.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the National Institutes of Health (HL 68780).
