Abstract
Smad proteins are signaling intermediates of the TGF-β superfamily and are involved in a range of biological activities including development and immune responses. We studied the expression of TGF-β-receptor activated Smads (Smad2 and Smad3), the common partner Smad (Smad4), an inhibitory Smad (Smad7), and the activated (phosphorylated) Smad2 (pSmad2) in developing and adult kidneys of humans and mice. These studies demonstrate associated expression of these Smads in multiple renal cell types in all developmental stages and in mature non-diseased kidneys. Smad expression is in general most widespread at the earliest stages of nephron development and diminishes as components of the nephrons become more differentiated. Paucity of Smad expression in mesangial cells in contrast to widespread expression of these Smads in glomerular visceral epithelial cells in both developing and mature kidneys was remarkable. Divergent and less extensive expression of Smad4, compared with other Smad proteins, was also demonstrated in tubules of human kidneys. Based on the observed expression patterns, these findings demonstrate, for the first time, expression of the TGF-β-receptor-activated Smad2 and Smad3, the common mediator Smad4, and the inhibitory Smad7 in the developing human fetal kidney, extending observations previously made in rodent systems to humans.
Keywords
M
We utilized immunohistochemistry (IHC), Western blotting, and reverse transcriptase (RT)- PCR to study tissue of humans and mice. These studies demonstrate associated expression of TGF-β-receptor activated Smad2, Smad3, the common mediator Smad4, and the inhibitory Smad7 in the development of the human fetal kidney. Continued constitutive expression of Smad2, 3, 4, and 7 can be demonstrated in mature non-diseased kidneys.
Materials and Methods
Source of Tissue
Normal human renal tissue was obtained from kidneys surgically excised because of the presence of a localized neoplasm. Tissue utilized from nephrectomies was obtained from macroscopically normal portions of kidney located at some distance from the neoplastic process. Human fetal kidneys (estimated gestational age ranging from 54 to 122 days) were obtained fresh from tissue examined after therapeutic abortion. Kidney samples were fixed in 10% neutral-buffered formalin for at least 12 hr, processed, paraffin-embedded, and sectioned using conventional techniques. These studies were performed in accordance with the Declaration of Helsinki (Carlson et al. 2004) under approval from the University of Washington Human Subjects Division (approval #01-8008-E-01) under conditions of tissue anonymity, whereby all study investigators remain masked to the specific identities of patients from whom tissues were obtained.
Time-mated pregnant Swiss-Webster mice were obtained from Simonsen Laboratories (Gilroy, CA) and housed at the Department of Comparative Medicine until tissues were used for processing. Mice were euthanized by CO2 asphyxiation and adult and embryonic tissues [embryonic days (ED) 17 and 19] were fixed in 4% paraformaldehyde, 0.1 M sodium phosphate buffer, pH 7.4, for 3 to 7 days prior to paraffin embedding. Animals were treated in accordance with the rules and regulations of the National Institutes of Health (NIH) Guidelines for the Care and Use of Laboratory Animals.
Antibodies
Smad2. An affinity-purified rabbit polyclonal antibody (cat. #51-1300; Zymed Laboratories, South San Francisco, CA) derived against the MH1 domain of human Smad2 was utilized. This antibody has previously been used for IHC detection of Smad2 in human (Jeruss et al. 2003b) and murine (Jeruss et al. 2003a) tissue.
Phospho Smad2. A synthetic phospho-peptide rabbit polyclonal antibody (cat. #AB3849; Chemicon International, Temecula, CA) that recognizes Smad2 only when dually phosphorylated at Ser465 and Ser467 was utilized.
Smad3. An affinity-purified rabbit polyclonal antibody (cat. #51-1500; Zymed Laboratories) derived against a central portion of the linker domain of human Smad3 was utilized. This antibody has previously been used for IHC detection of Smad3 in human (Jeruss et al. 2003b) and rodent tissue (Oxburgh and Robertson 2002; Furuse et al. 2004).
Smad4. An affinity-purified mouse monoclonal antibody (cat. #sc-7966; Santa Cruz Biotechnology, Santa Cruz, CA) raised against amino acids 1-552 representing full-length Smad4 of human origin was utilized. This antibody has previously been used for IHC detection of Smad4 in murine (Oxburgh and Robertson 2002; Furuse et al. 2004) and human tissue (Natsugoe et al. 2002; Han et al. 2005).
Smad7. A polyclonal antibody produced in goats raised against amino acids 320-398 was utilized (cat. #AF2029; R&D Systems, Minneapolis, MN).
IHC
IHC was performed in formalin-fixed, paraffin-embedded tissue as previously described by our laboratory (Alpers et al. 2002). Briefly, 4-μm tissue sections were deparaffinized in xylene and rehydrated in graded ethanol. Endogenous peroxidase was blocked with 3% hydrogen peroxide. To increase staining, sections were pretreated by steam heating for 30 min in Antigen Unmasking Solution (Vector Laboratories; Burlingame, CA), followed by treatment with avidin/biotin blocking (Avidin/Biotin Blocking Kit; Vector Laboratories). To block nonspecific background, the sections were incubated with non-immune goat serum or non-immune horse serum for 30 min. Sections were then incubated overnight at 4C with the primary antibody diluted in phosphate-buffered saline (PBS; 138 mM NaCl, 2.7 mM KCl, 2 mM Na2HPO4, 1.5 mM KH2PO4, pH 7.3) plus 1% bovine serum albumin (BSA; Sigma, St Louis, MO). Following washes in PBS, the sections were subsequently incubated with biotinylated host-specific secondary antibodies (Vector Laboratories) and with the ABC-Elite reagent (Vector Laboratories); 3,3′-diaminobenzidine with nickel chloride enhancement was used as chromogen. Sections were counterstained with methyl green or hematoxylin, dehydrated, and coverslipped. For all samples, negative controls consisted of substitution of the primary antibody with isotype-matched, irrelevant murine, goat, or rabbit antibodies, at the same total protein concentrations as the corresponding primary antibody.
Protein Preparation and Western Blotting
To confirm the specificity of the antibodies directed against Smad2, 3, 4, 7, and pSmad2 that were used for IHC, Western blotting was performed. Approximately 250 mg of frozen fetal and adult kidney tissue was minced and washed thoroughly in PBS. After centrifugation and removal of tissue debris, the suspension buffer (0.1 mM/liter NaCl, 0.01 mol/liter Tris-HCl, pH 7.6, 0.001 mol/liter EDTA, pH 8.0, 1 μg/ml aprotinin, and 100 μg/ml phenylmethylsulfonyl fluoride) was added, and the tissue was dispersed with a Tissuemizer (Tekmar; Cincinnati, OH). For Western reducing conditions, 2X sample buffer with sodium dodecyl sulfate (SDS) and 3-mercaptoethanol was added, the protein sample was boiled, and the pellet discarded. Samples were electrophoresed on an 8-16% Tris-Hepes polyacrylamide gel (ISC Bioexpress; Kaysville, UT) and then blotted onto 0.2-μm nitrocellulose membranes. Blots were blocked with 5% BSA, 1% non-fat milk, and 1% Tween 20 in Tris buffer (0.1 M Tris(hydroxymethyl)aminomethane) for 30 min and then incubated with anti-Smad2, anti-phospho Smad2, anti-Smad3, anti-Smad4, and anti-Smad7 antibodies or non-immune isotype-matched antisera, respectively, diluted in 1% BSA, 1% Tween 20 in Tris overnight at 4C. After washing, blots were incubated with horseradish peroxidase-conjugated host-specific secondary antibodies (Zymed) for 1 hr. Blots were visualized with SuperSignal West Pico chemiluminescent substrate (Pierce Chemical; Rockford, IL) and exposed on x-ray films.
RT-PCR Analysis
To demonstrate Smad2, 3, 4, and 7 gene expression in fetal and adult human kidney tissues, total cellular RNA from normal nephrectomies and fetal kidneys was extracted with RNAqueous-Midi kit (Ambion; Austin, TX) and stored at −70C until use. To reduce the risk of DNA contamination, samples were treated with RNase free DNase I (Invitrogen; Carlsbad, CA), followed by phenol/chloroform/isoamyl alcohol extraction (Gibco BRL; Gaithersburg, MD). Approximately 2 μg of RNA was mixed with the RT mixture per instructions in the Ambion RETROscript kit and then reverse transcribed with oligo(dT) primers for 1 hr at 42C. RNA without reverse transcriptase was used as a negative control. Sequences of the Smad2 primers were 5′-GTTCCTGCCTTTGCTGAGAC (forward) and 5′-TCTCTTTGCCAGGAATGCTT (reverse), for Smad3: 5′-TGCTGGTGACTGGATAGCAG (forward) and 5′-CTCCTTGGAAGGTGCTGAAG (reverse), for Smad4: 5′-CCATTTCCAATCATCCTGCT (forward), 5′-ACCTTTG CCTATGTGCAACC (reverse), for Smad7: 5′-TACCGTGCAGATCAGCTTTG (forward), 5′-TTTGCATGAAAAGCAAGCAC (reverse). After the hot start at 94C for 2 min, 37 amplification cycles were carried out with the following processes: denaturation at 94C for 30 sec, annealing at 56C for 30 sec, extension at 72C for 30 sec in each cycle, and 5 min at the last cycle. RT-PCR products were resolved on a 1% agarose gel containing ethidium bromide and visualized under ultraviolet illumination. RNA was replaced by nuclease-free water as negative control.
Results
RT-PCR
Smad2, 3, 4, and 7 mRNA expression in fetal and adult human kidney was analyzed by RT-PCR. A band corresponding to the expected length of Smad2 (220 bp), Smad3 (176 bp), Smad4 (221 bp), and Smad7 (200 bp) was identified in fetal and adult kidneys. No expression was detected in the negative controls (RT control and water control) (Figure 1).
Characterization of the Antibodies
Specificity of the antibodies to Smad2, phospho Smad2 (pSmad2), Smad3, Smad4, and Smad7 was characterized by Western immunoblotting of adult and fetal kidney tissue lysates. Specific immunoreactions of Smad2, pSmad2, Smad3, Smad4, and Smad7 were localized to bands at ∼55 kDa, 60 kDa, 45 kDa, 60 kDa, and 45 kDa in fetal and adult human tissue, respectively (Figure 2). These results were consistent with the known molecular sizes of these Smad proteins (Hao et al. 1999). Purified proteins corresponding to these entities were not available to use for competitive inhibition studies.
Smad2, 3, 4, 7, and pSmad2 Expression During Renal Development
Kidney development results from inductive interactions between ureteric bud epithelia and metanephric blastemal mesenchyme. Metanephric blastema refers to the subcapsular aggregate of densely populated, homogeneous, relatively undifferentiated cells with high nuclear/cytoplasmic ratios from which developing nephrons (glomeruli and tubules and their precursor forms) arise. The tips of ureteric buds branch and induce the kidney blastema to condense and form vesicles. These vesicles cavitate, elongate, and undergo regional differentiation to form both glomeruli and tubules (proximal and distal tubules and the loop of Henle). Extensions of the branched ureteric bud fuse with the tubular system of the developing nephron and contribute the collecting tubules and ducts of the fully formed nephron. Our analysis of Smad2, 3, 4, 7, and pSmad2 expression during renal development in human and mouse sequentially follows each of these steps, that is, expression in the ureteric bud epithelia, blastema, vesicle, glomerulogenesis, tubulogenesis, and the kidney interstitium. Developing human kidneys were obtained from fetuses of estimated 54 to 122 days gestational age. These kidneys demonstrated age-appropriate features of glomerulogenesis and tubulogenesis and uniformly lacked features of maldevelopment such as dysplasia, scarring, or cyst development. Wild-type mice were examined at two different developmental stages: ED17 and ED19. Smad expression at these time points showed no significant differences, and so the results are discussed together. Table 1 and Table 2 summarize the expression data in both the embryonic and adult kidney. Although Figure 3 and Figure 4 do not show every fetal structure for each antibody, complete results are detailed in Table 1 and Table 2.
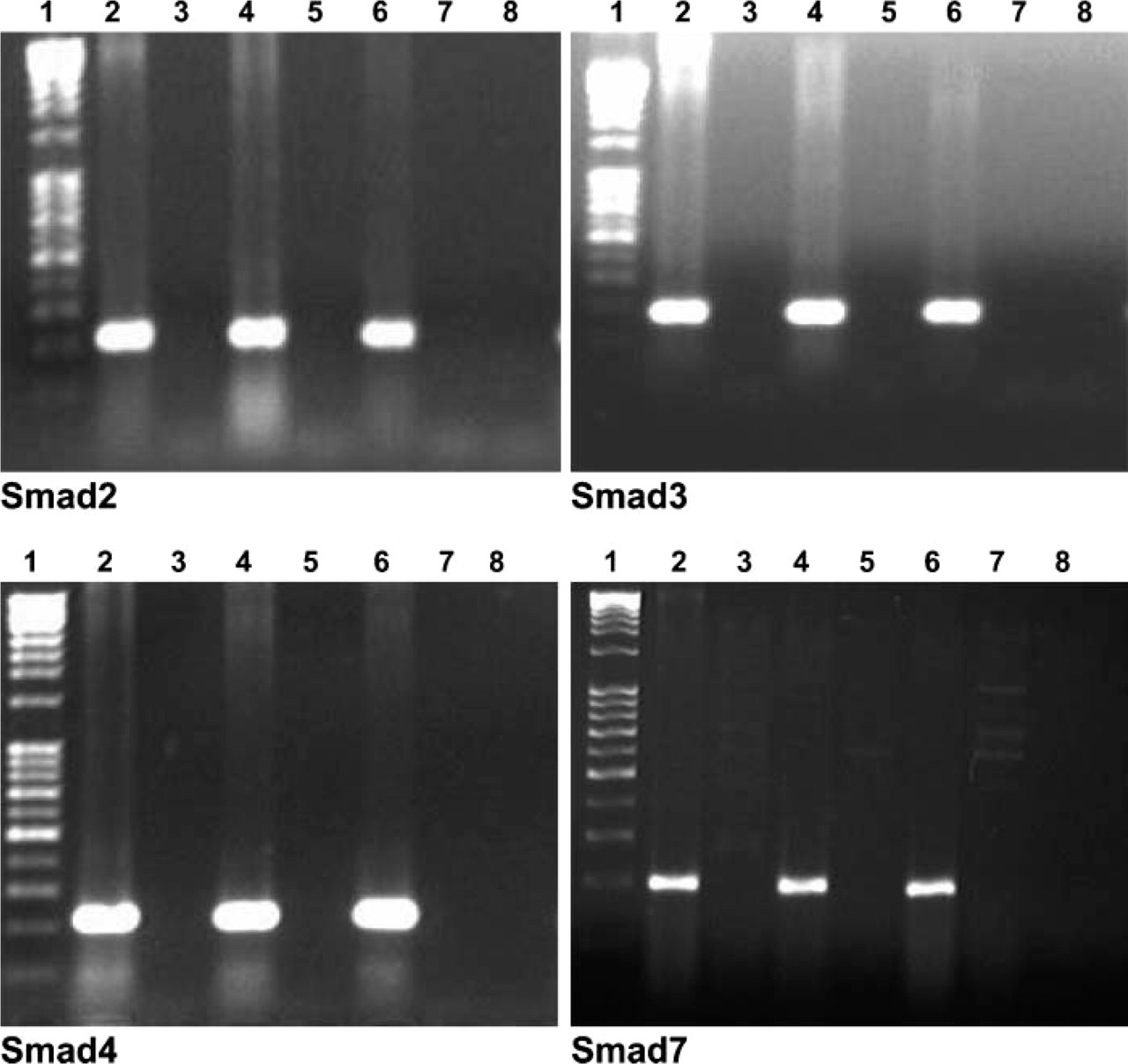
Reverse transcriptase-polymerase chain reaction (RT-PCR) analysis was used to detect Smad2, Smad3, Smad4, and Smad7 mRNA expression in human fetal (Lanes 2 and 4) and adult (Lane 6) kidney RNA. A band corresponding to the expected length of Smad2 (220 bp), Smad3 (176 bp), Smad4 (221 bp), and Smad7 (200 bp) is identified in fetal and adult kidneys. No expression is detected in the negative controls [RNA without RT (Lanes 3, 5, and 7) and water (Lane 8)]. Marker used was a 1-kb ladder.
Ureteric Bud and Derivatives
Expression of Smad2, 3, 4, 7, and the activated (phosphorylated) Smad2 protein (pSmad2) in the ureteric bud and derivatives (collecting tubules and urothelium) was seen in human as well as in mouse kidneys. Staining intensity obtained with the Smad2 and pSmad2 protein was more intense than that for the other Smad proteins, possibly reflecting differing levels of expression but possibly a consequence of technical performance characteristics of the antisera used for these studies. Smad2 and pSmad2 showed a strong expression in the mature collecting tubules and the urothelium and lower expression in embryonic ureteric buds. Smad3 also showed strong expression in the urothelium, moderate expression in ureteric buds, and faint expression in most of the collecting tubules. In contrast, Smad4 and 7 showed weak expression in collecting tubules and urothelium but showed strong expression in ureteric buds in mice and humans (Figure 3).
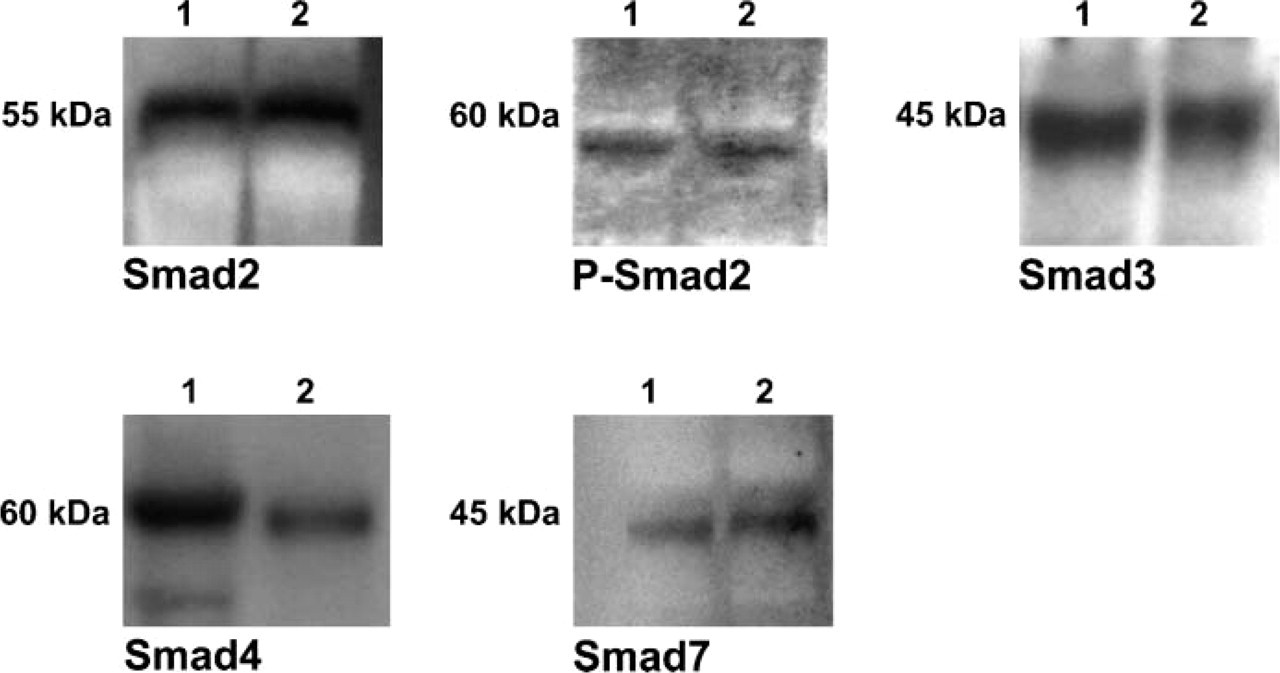
Western blot analysis utilizing anti-Smad2, anti-pSmad2, anti-Smad3, anti-Smad4, and anti-Smad7. Antibodies are reactive with fetal (Lane 1) and normal adult human kidney (Lane 2) protein extracts.
Smad expression in fetal human and mouse kidney
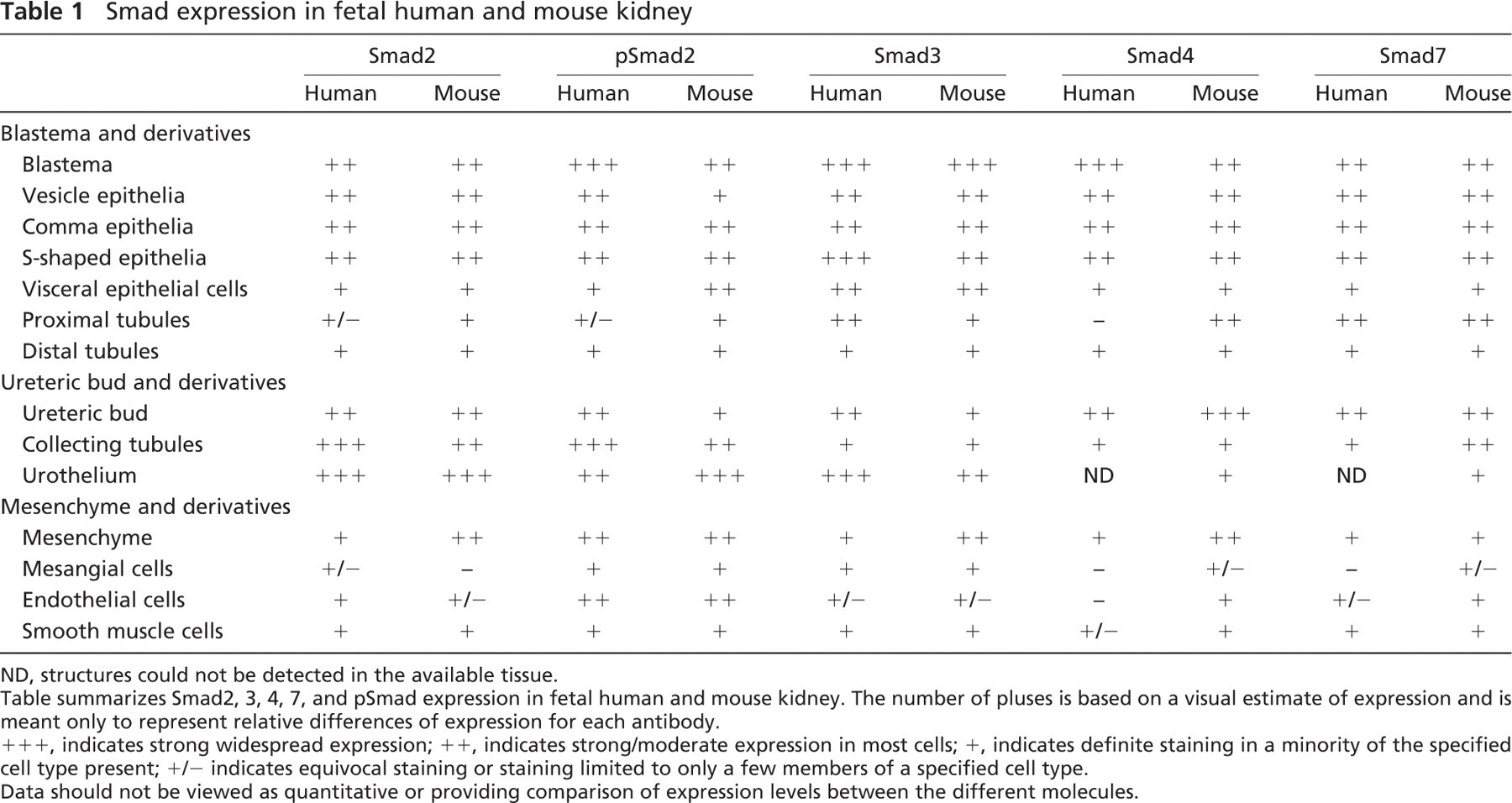
ND, structures could not be detected in the available tissue.
Table summarizes Smad2, 3, 4, 7, and pSmad expression in fetal human and mouse kidney. The number of pluses is based on a visual estimate of expression and is meant only to represent relative differences of expression for each antibody.
+++, indicates strong widespread expression; ++, indicates strong/moderate expression in most cells; +, indicates definite staining in a minority of the specified cell type present; +/- indicates equivocal staining or staining limited to only a few members of a specified cell type.
Data should not be viewed as quantitative or providing comparison of expression levels between the different molecules.
Blastema and Glomeruli
The blastema uniformly expresses high levels of Smad2, 3, 4, 7, and pSmad2 in humans and mice. In contrast, the vesicle epithelia show somewhat diffuse staining for Smad2, 3, 4, 7, and pSmad2 and progressively more irregular or less uniform staining of differentiating glomerular visceral epithelial cells as the fetal glomeruli mature. Some expression of Smad2, 3, 4, 7, and pSmad2 remains in the visceral epithelial cells in the most differentiated glomeruli within fetal kidneys in both humans and mice (Table 1). Only some cells with a mesangial cell-like central distribution in developing human and mouse glomeruli at early stages of differentiation, when the mature capillary vascular architecture is not yet developed, show detectable expression of pSmad2, Smad2, and Smad3 (Figure 3).
Smad expression in adult human and mouse kidney
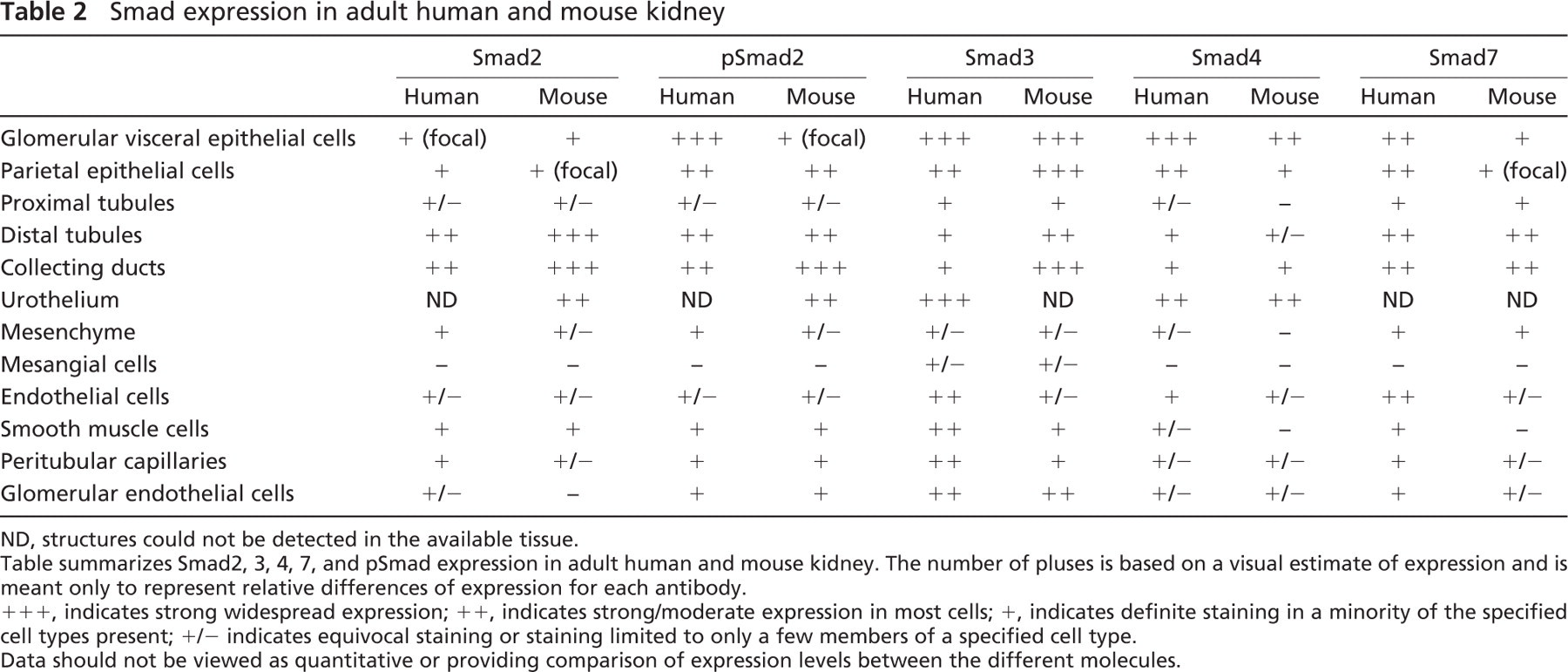
ND, structures could not be detected in the available tissue.
Table summarizes Smad2, 3, 4, 7, and pSmad expression in adult human and mouse kidney. The number of pluses is based on a visual estimate of expression and is meant only to represent relative differences of expression for each antibody.
+++, indicates strong widespread expression; ++, indicates strong/moderate expression in most cells; +, indicates definite staining in a minority of the specified cell types present; +/- indicates equivocal staining or staining limited to only a few members of a specified cell type.
Data should not be viewed as quantitative or providing comparison of expression levels between the different molecules.
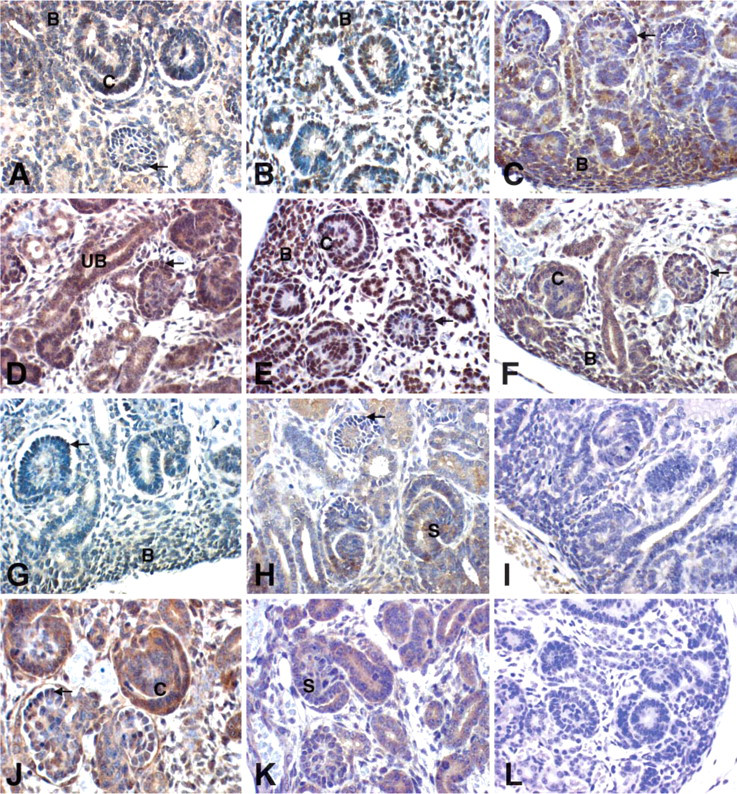
Immunolocalization of Smad2: (
Tubules
The distal portion of the vesicular epithelium elongates and forms the proximal and distal tubules, with the intervening ascending and descending tubules and the loop of Henle. Proximal tubules are readily identified by their large abundant eosinophilic cytoplasm. Expression of Smad2 and pSmad2 is barely detectable in human fetal proximal tubules. In contrast, focal expression in individual tubular cells of Smad2 and pSmad2 protein in proximal tubules could be seen in fetal mice. Smad3 and 7 protein in proximal tubules was detected in fetal human and mouse. Smad4 protein could not be detected in human fetal proximal tubules, although moderate expression was seen in mice. Distal tubules show focal expression of Smad2, 3, 4, 7, and pSmad2 in human and mouse. Tubular expression of all Smads was more intense in the medulla than in the cortex (Figure 3).
Immunolocalization of Smad2: (
Interstitium and Vascular Structures
Smad2, 3, 7, and pSmad2 are broadly expressed in the embryonic kidney interstitium. Expression of these Smad proteins seemed to be higher in the developing mouse kidney than in human. Expression of Smad2 and pSmad2 in human fetal kidney was higher in medullary than in cortical stroma. Remarkably, there was also widespread expression of Smad4 in medulla and cortex in the mouse fetus, in contrast to very low expression of Smad4 in the interstitium of the human fetus. Endothelium of human and mouse fetal arteries showed readily detectable expression of pSmad2 and lesser degrees of expression of Smad2, whereas expression of Smad3, 4, and 7 was either focal or equivocal in endothelial cells. In vascular smooth muscle cells, expression of Smad2, 3, 4, 7, and pSmad2 could be localized in both human and mouse developing kidneys (Figure 3).
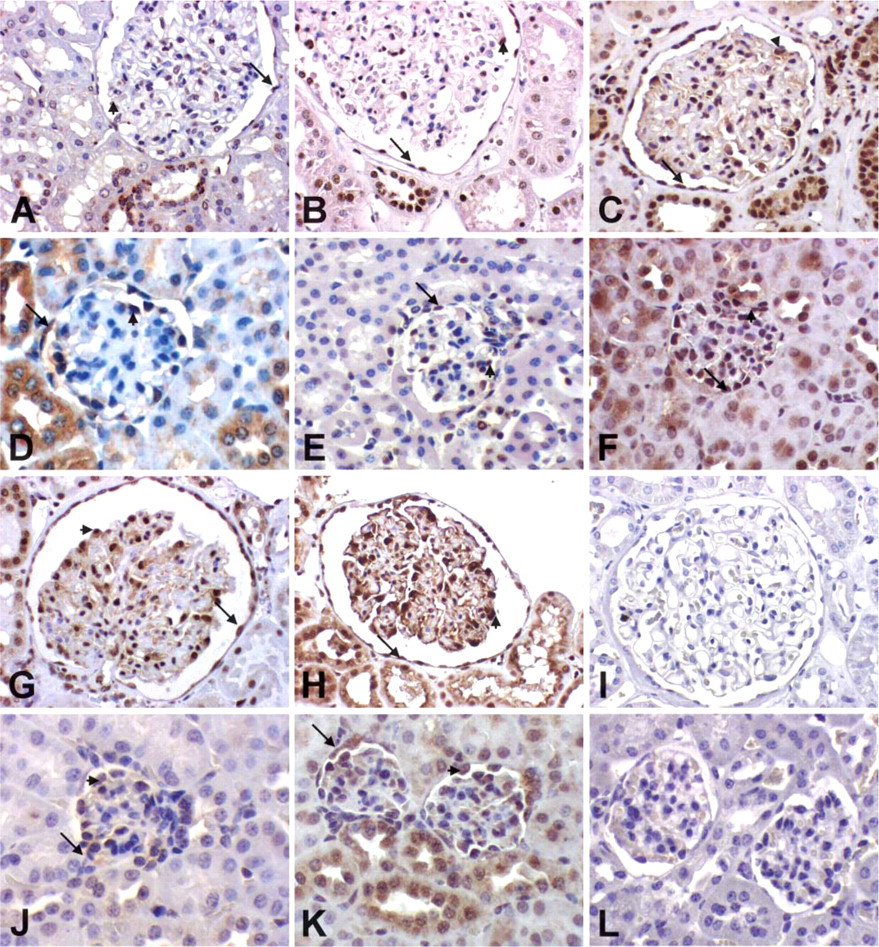
Smad2, 3, 4, 7, and pSmad2 Expression in Adult Kidney Glomeruli
All investigated Smads are expressed in the mature human and mouse glomerulus. They are expressed widely by glomerular visceral epithelial cells and more irregular in the endothelium. Smad2, 3, 4, 7, and pSmad2 showed strong expression in the glomerular visceral epithelial cells of mature human glomeruli. In mice, glomerular visceral epithelial cells also showed Smad2, 4, 7, and pSmad2 expression but at a lower staining intensity. Smad3 showed equivalent strong expression patterns in mice and humans. Expression of Smad2, 3, 4, 7, and pSmad2 was also detected in parietal epithelial cells in mice and humans. Only Smad3 protein could be detected in mesangial cells of mice and humans at low levels. These results are summarized in Table 2 and shown in Figure 4.
Smad Expression in Adult Tubules
Smad2, 3, 7, and pSmad2 are all expressed at low levels in proximal tubular cells both in humans and mice. Distal tubules and collecting ducts showed widespread strong expression of Smad2, 7, and pSmad2 in mice and humans, of Smad3 in mice, weak or undetectable expression of Smad3 in humans, and weak expression of Smad4 in both mice and humans. Expression was more prominent in the medulla than in the cortex of human and mouse adult kidneys (Figure 4).
Smad Expression in Adult Interstitium and Vascular Structures
In contrast to the developing kidneys, which showed strong expression of Smads in the mesenchyme, Smads could be detected only focally in the adult renal interstitium. Expression in vascular structures showed no significant differences between mice and humans. Whereas Smad3 and 7 were distinctly expressed by the endothelial cells of arterioles and arteries, Smad2, 4, and pSmad2 were detected only rarely and focally. Smooth muscle cells of arterioles and arteries showed high expression of Smad3 in humans and weak expression of Smad2 and pSmad2 in both mice and humans. Smad4 and 7 expression in smooth muscle cells of arterioles and arteries was only rarely detected (Figure 4).
Expression of Smad2, pSmad2, Smad3, Smad4, and Smad7 in Other Fetal Mouse Organs
The main focus of our study was to investigate the expression of the TGF-β-specific signaling intermediates Smad2, Smad3, Smad4, and Smad7 in the developing and mature kidney, but we were also interested in the distribution of these Smads in other organs of the developing mice. We investigated ED17 Swiss-Webster mice. We could detect expression of Smad2, pSmad2, Smad3, Smad4, and Smad7 in many sites in the developing fetus. Major sites of expression could be seen in organ structures arising from mesenchymal-epithelial interactions. Examples of localization include bronchial epithelium of the lungs, myocardium of the heart, whiskers, epidermis of the skin (particularly the basal layer), thymus, and epithelium of small bowel and colon (Figure 5). In the liver, expression was most prominent in megakaryocytes. A second site of strong expression was the central nervous system (data not shown).
Discussion
Genetic mutation studies have established the importance of Smads for the appropriate development of the fetus (Weinstein et al. 2000). Mice strains that harbor two mutated alleles for either Smad2, 3, or 4 have been described, but the Smad3 null mice are the only ones of these that survive through embryogenesis into adulthood (Datto and Wang 2000). Targeted gene disruptions in mice have shown that Smad2 and Smad4 are needed for gastrulation, Smad5 for angiogenesis, and Smad3 for establishment of the mucosal immune response and proper development of the skeleton (Weinstein et al. 2000). It is not clear from these studies whether specific Smad deletions impact renal development.
This study localizes sites of Smad2, 3, 4, 7, and pSmad2 protein and gene expression in human and mouse kidneys by RT-PCR, Western blotting, and IHC techniques and thereby is a first step in establishing a role for each of these Smads in renal development. This is the first comprehensive localization of Smad2, 3,4, 7, and pSmad2 in developing and mature human kidneys.
We detected protein expression of Smad2, 3, 4, and 7 in multiple renal cell types including glomerular visceral epithelial cells, parietal cells, endothelial cells, tubules, smooth muscle cells, and mesenchymal cells in all development stages (Table 1 and Table 2).
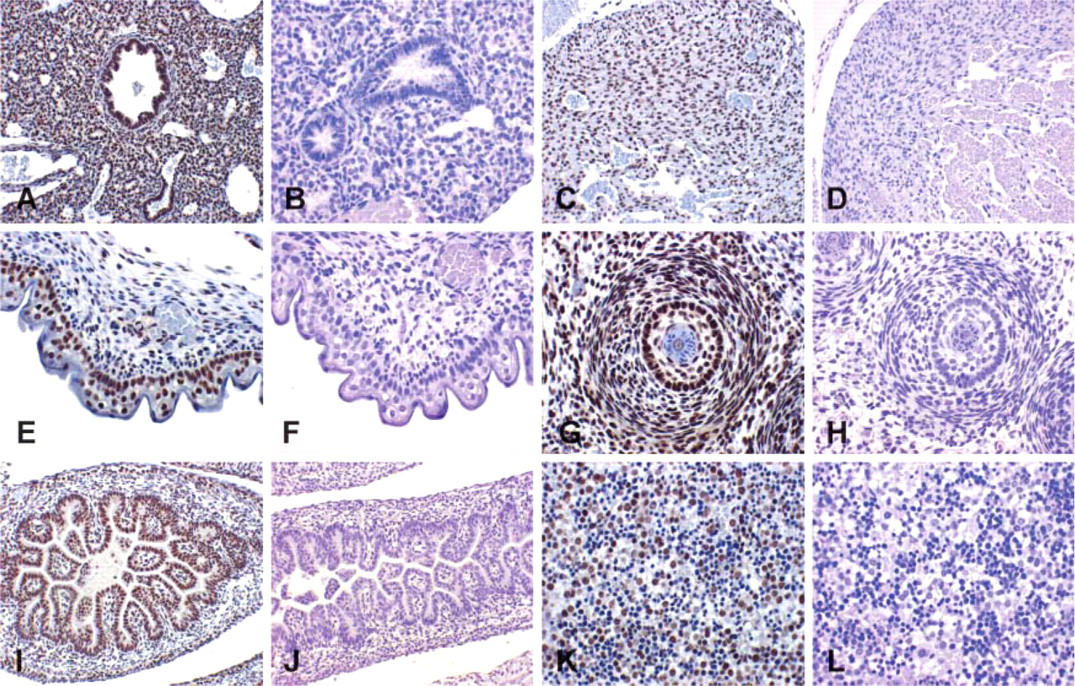
Immunolocalization of the active form of Smad2, pSmad2, in nuclei of mouse fetal organs (ED17). Major sites of expression could be seen in the bronchial epithelium of the lungs (
Our findings confirm the patterns of Smad expression detailed in previous studies in rodents (Oxburgh and Robertson 2002; Vrljicak et al. 2004) and extend them to humans. Smad expression is in general most widespread at earliest stages of nephron development and diminishes as components of the nephrons become more differentiated. Vrljicak et al. (2004) demonstrated that the R-Smads, Smad4, and the I-Smads are expressed in mouse kidney from ED12 until the end of nephrogenesis. They showed Smad mRNA expression in the nephrogenic zone by uninduced mesenchymal cells adjacent to the ureteric bud tips and by mesenchymal cells in the peripheral cortex that were destined to become stromal cells. In mesenchymal cells, adjacent to the ureteric bud tips, expression was downregulated once those cells began to undergo epithelialization. Oxburgh and Robertson (2002) could detect Smad1, 2, 3, 4, 5, and 8 protein expression in metanephric mesenchyme but described them as being downregulated in condensing mesenchyme and in derivative structures such as renal vesicles.
One major finding is that compared with the other Smads investigated there is less CoSmad (Smad4) staining in the human and mouse fetal and adult kidneys. Although previous studies support a pivotal role of Smad4 in TGF-β-mediated processes (Lagna et al. 1996), recent evidence from Smad4-deficient mouse embryos and cells indicate that certain responses to TGF-β signals may occur in the absence of Smad4.
Thus, chimeric mouse embryos containing Smad4-null epiblast cells form patterned mesoderm that gives rise to the heart, trunk somites, and lateral plate mesoderm, a phenotype different from that expected if both nodal and BMP signals are impaired (Chu et al. 2004). Murine fibroblast cells deficient in Smad4 still respond to TGFβ/activin-mediated growth inhibition and induction of extracellular matrix genes (Sirard et al. 2000). Human cell lines with depleted Smad4 only lose a subset of TGF-β-responsive gene expression and can undergo TGF-β-induced epithelial to mesenchymal transition (Levy and Hill 2005). These results imply that Smad4 may not be an obligatory component of all TGF-β signals and may be dispensable in particular TGF-β-responsive processes. In addition, studies in early frog development (Chang et al. 2006) and carp (Wang et al. 2005b) showed the existence of different types of Smad4.
Another major finding of our studies was the paucity of Smad expression, including the common mediator Smad4, in mesangial cells in both developing and mature kidneys. Constitutive expression of each of the Smads studied was generally low or absent in fetal mouse and human mesangium. In adult kidneys, only Smad3 protein could be detected in mesangial cells of mice and humans at low levels. This is supported by a previous study by Ostendorf et al. (2002) that could detect Smad7 expression in mesangial cells only after induction of anti-Thy 1.1 mesangioproliferative nephritis in rats. In the same study, Smad2 was expressed in peritubular and glomerular capillary and arterial endothelium in normal and diseased animals but not in mesangial cells. Previous in vitro studies stand in contrast to these findings. Schiffer et al. (2002) showed constitutive Smad7 expression in mouse mesangial cells that was not induced by TGF-β. In podocytes, Smad7 inhibited both Smad2- and Smad3-mediated TGF-β signaling, whereas in mesangial cells it inhibited Smad3 signaling only. Poncelet et al. (1999) showed Smad2, 3, and 4 protein expression in human mesangial cells and described that TGF-β1 downregulated Smad3 mRNA and protein expression, whereas Smad2 and 4 were less affected. In contrast to the Smads, TGF-β and its receptors are constitutively present in mesangial cells as shown in previous in vivo and in vitro studies (Hayashida et al. 1999; Hartner et al. 2003; Runyan et al. 2003; Schnaper et al. 2003; Tsuchida et al. 2003). Our findings indicate TGF-β signaling in mesangial cells requires either upregulation of relevant Smad proteins that are not constitutively present in high levels or signaling through Smad-independent pathways (Derynck and Zhang 2003).
There is growing evidence that TGF-β may promote podocyte injuries of apoptosis and matrix production (Schiffer et al. 2001; Chen et al. 2005), and the present study shows that both activatory and inhibitory signaling intermediates of TGF-β are in place to mediate this activity. The presence of phosphorylated Smad2 in these cells indicates that these cells in the normal state are being engaged by TGF-β.
Our findings of significant and constitutive expression of Smad2, 3, 4, and 7 in glomerular podocytes of fetal and adult human and mouse kidneys are in contrast to the low or undetectable expression of Smads in mesangial cells. Our findings are largely in accord with the studies of Schiffer et al. (2002) who found Smad7 expression in podocytes of normal mouse and human glomeruli, which could be further induced in podocytes in animal models and human glomerular diseases. However, the widespread constitutive podocyte expression of Smad7 that was demonstrated in our study appears to be inconsistent with the suggestion of Schiffer et al. (2002) that upregulated podocyte expression of this protein contributes to specific human diseases. Several studies have shown that TGF-β plays an important role in the function and injury response of podocytes. Conversely, podocyte injury can lead to increased TGF-β mRNA expression, as shown in in vitro studies of podocytes undergoing mechanical strain as a model for podocyte injury (Durvasula et al. 2004). Because TGF-β signals are largely transduced through Smad signaling pathways, the Smads constitutively expressed by podocytes likely assume a significant role in autocrine and paracrine signaling by these cells.
In summary, we provide data on the expression of multiple key mediators of TGF-β signaling in developing and mature rodent kidneys. We provide corresponding expression data in human kidneys and detail important similarities in expression that will help us better understand how the role of these molecules in murine development, injury, and repair can increase our understanding of human renal development and disease. The paucity of detectable baseline expression of many Smads in the mesangium, despite well-documented pleiotropic responses of these cells to TGF-β engagement, suggests the hypothesis that integrated induction of specific clusters of Smads is necessary for achieving specificity of TGF-β activity in these cells.
Footnotes
Acknowledgements
This work was supported by the National Institutes of Health (NIH), Grants DK-66802, DK-69912, and U24 DK-076126 (to CEA) and an Else Kröner Fresenius Foundation Grant (to MCB). We thank the Central Laboratory for Human Embryology at the University of Washington (supported by Grant HD-00836 from NIH) for assistance in providing fetal tissue.
