Abstract
Hepatocyte growth factor activator (HGFA) can activate the single-chain hepatocyte growth factor (HGF) required for embryonic development. We studied the immunohistochemical (IHC) localization of HGFA in adult mouse liver and its developmental changes from embryonic day 12 to postnatal day 30. A heterogeneous distribution of HGFA was observed in adult liver tissues. The hepatocytes around the hepatic veins were preferentially positive for HGFA, whereas those in other areas were negative. Depending on the vascular diameter, the hepatic veins were bordered by a one- to three-cell-thick layer of hepatocytes positive for HGFA, which showed evidence of cell–cell heterogeneity in staining intensity. Immunoelectron microscopy detected ubiquitous distribution of the gold particle reaction product for HGFA in the cytoplasm of these hepatocytes, especially in the rough endoplasmic reticulum. Developmental analysis indicated that there was hardly any staining of HGFA until postnatal day 0 and that noticeable staining was initially detected in the pericentral hepatocytes on postnatal day 3. Subsequently, immunoreactivity increased and the distinct staining pattern had been established by postnatal day 30. These results suggest that HGFA proteins are produced in the hepatocytes surrounding the efferent hepatic veins in the mouse and that development of the unique distributing pattern takes place postnatally.
HGFA, an activator of HGF, has been purified from fetal bovine (Shimomura et al. 1992) and human (Miyazawa et al. 1993) sera. Human HGFA is synthesized as an inactive protein and is activated to generate a heterodimer consisting of a short chain and a long chain linked together by a disulfide bond. The proteases of the HGFA family comprise the urokinase-type plasminogen activator, tissue-type plasminogen activator, blood coagulation factor XII, and plasma hyaluronan-binding protein (Naldini et al. 1992; Mars et al. 1993; Miyazawa et al. 1993; Shimomura et al. 1995; Choi–Miura et al. 1996; Hashimoto et al. 1997). HGFA is the most powerful serine protease encountered in vitro and its active form is generated exclusively in injured tissues. One of the functions of HGFA is to cleave the single-chain HGF into its active two-chain form, and it is the key enzyme for the regulation of HGF activity in the injured regions (Shimomura et al. 1992,1995; Miyazawa et al. 1996). HGF and its specific receptor c-Met, HGFA, and HGFA inhibitor type-1 (HAI-1) may also be important when interacting with other factors such as thrombin, tumor necrosis factor, transforming growth factor, and interleukins-1 and −6 for regeneration and repair of various tissues (Shimomura et al. 1993; Shinozuka et al. 1994; Michalopoulos and DeFrances 1997; Kataoka et al. 1999,2000). Recombinant human HGFA exogenously administered via the portal vein was found to enhance liver regeneration through the production of biologically active HGF in partially hepatectomized rats (Kaibori et al. 2002).
HGF has been identified a tissue organizer with multiple biological activities and has been isolated as the growth factor of hepatocytes (Michalopoulos et al. 1982; Nakamura et al. 1984,1987,1989; Russell et al. 1984; Thaler and Michalopoulos 1985; Gohda et al. 1988; Miyazawa et al. 1989). Moreover, it is known to act not only on hepatocytes but also on virtually every tissue in the body, ranging from the nervous system to the immune and reticuloendothelial systems (Zarnegar and Michalopoulos 1995; Matsumoto and Nakamura 1997). In the liver, HGF is expressed in non-parenchymal cells such as sinusoidal endothelial cells, Kupffer cells, and Ito cells, and acts on parenchymal cells expressing c-Met (Kinoshita et al. 1989; Noji et al. 1990; Tashiro et al. 1990; Sonnenberg et al. 1993). The proto-oncogene c-Met tyrosine kinase, the cell surface receptor for HGF, has been identified as a transmembrane protein (Cooper et al. 1984; Park et al. 1987; Bottaro et al. 1991; Naldini et al. 1991), and recent findings have demonstrated the immunolocalization of HGF and c-Met proteins during liver development including fetal and adult stages (Ishikawa et al. 2001). Mice lacking HGF, known as homozygous mutant embryos, have severely impaired placentas with markedly reduced numbers of labyrinthine trophoblast cells, and die in utero before birth. This mutation also affects the embryonic liver, which is reduced in size and shows extensive loss of parenchymal cells (Schmidt et al. 1995; Uehara et al. 1995; Kolatsi–Joannou et al. 1997). Therefore, HGF is essential for embryonic development, which means that HGF activators must be expressed during normal embryogenesis.
HGFA mRNA is mainly expressed in parenchymal cells of adult liver and its protein is secreted into the blood. A recent study reported that the expression of the HGFA gene was detected not only in the liver but also in the gastrointestinal tract of normal adult mice. In these mice, HGFA mRNA was strongly expressed in the liver as well as colon, moderately in the stomach, duodenum, jejunum, and ileum, weakly in the kidney, and almost imperceptibly in the brain, heart, testis, and ovary (Itoh et al. 2000). The level of HGFA mRNA increased in the liver after hepatotoxin or nephrotoxin treatment (Miyazawa et al. 1993; Okajima et al. 1997). As far as embryogenesis is concerned, HGFA has been shown to be a possible regulator of morphogenesis during fetal development of the rat gastrointestinal tract (Matsubara et al. 1998) and the mouse kidney (van Adelsberg et al. 2001), although the localization of HGFA during development of the liver is not clear.
To gain a better understanding of the role of HGFA in liver function and organogenesis, we employed monospecific antibodies to investigate the IHC distribution of HGFA in adult and developing mouse liver tissues.
Materials and Methods
Animals and Tissues
Liver tissues of ICR mice of different ages were examined: embryonic day 12, postnatal days 0, 3, 14, and 30, and postnatal week 8. Five embryos irrespective of gender and five male mice from each postnatal group were used. Adult male C57BL/6J mice were also used for verifying strain differences. All animals were purchased from SLC (Hamamatsu, Japan). They were sacrificed under deep anesthesia with pentobarbital sodium solution (0.04 mg/g body weight) in accordance with the Shinshu University School of Medicine Animal Care Committee guidelines.
Antibodies for HGFA
Three antibodies against HGFA were used for our study. N-19 goat polyclonal antibody for human HGFA was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). A-1 and P1-4 mouse monoclonal antibodies (MAbs) raised against human HGFA (Shimomura et al. 1993) were kindly donated by Dr. Miyazawa of Tokyo University.
Reverse Transcription-polymerase Chain Reaction (RT-PCR)
Total RNA from mouse liver obtained on postnatal day 8 was extracted with the RNeasy mini kit (Qiagen; Valencia, CA) according to the manufacturer's instructions. cDNA was synthesized from 1 μg of total RNA by means of Super-Script II reverse transcriptase (Invitrogen; Carlsbad, CA). cDNA samples were subjected to RT-PCR amplification using the GeneAmp PCR System 9700 (Applied Biosystems; Foster City, CA) with a primer set selective for mouse HGFA: the forward primer was 5′-CGGAGGCACCTGCCACC-3′, and the reverse primer was 5′-GCAGCGGTGCTGGCCAC-3′ (150 bp). Amplification consisted of a hot start at 94C for 3 min followed by 50 cycles of denaturation at 94C for 30 sec, annealing at 60C for 30 sec, and extension at 72C for 30 sec with a final 7-min extension at 72C. The PCR products were size-fractionated by 3% agarose gel electrophoresis and visualized with ethidium bromide staining. Sequencing used the ABI PRISM 310 Genetic Analyzer (Applied Biosystems) to confirm that the PCR product corresponded to the mouse HGFA cDNA sequence (van Adelsberg et al. 2001).
Immunoblotting Assay
ICR mouse plasma (Cosmo Bio; Tokyo, Japan) was heated at 100C for 4 min in a sample buffer solution containing 1% SDS under reducing conditions. It was then subjected to SDS-polyacrylamide slab gel electrophoresis with a small slab gel (6 × 7 cm) followed by immunoblotting analysis. After transfer to nitrocellulose membranes, blots were blocked with 5% bovine serum albumin for 1 hr and then incubated overnight at 4C with goat polyclonal antibody against HGFA diluted to 1:100 with 3% bovine serum albumin/150 mM sodium phosphate, pH 7.4. Blots were washed three times in PBS–Tween and incubated with appropriate peroxidase-labeled secondary antibodies diluted 1:200 for 30 min. Visualization was attained with the reaction using 4-chloro-1-naphthol.
Light Microscopic Immunohistochemistry
Small pieces of liver tissue were fixed for 24 hr in a Methacarn solution (methanol–chloroform–acetic acid 6:3:1) at room temperature or in 4% paraformaldehyde (PFA)/100 mM sodium phosphate, pH 7.4, at 4C, and embedded in paraffin. After deparaffinization and rehydration, 5-μm-thick serial sections were immunostained with anti-HGFA antibodies by employing the ABC method followed by DAB reaction. Before immunostaining on PFA-fixed specimens with the N-19 antibody, they were pretreated for antigen retrieval in 10 mM Na citrate, pH 6.0, in a microwave at 100C for 5 min (Shi et al. 1991). Specimens were observed with a Nikon Microphot-FXA light microscope after counterstaining with a methyl green solution. Control staining used the N-19 antibody preabsorbed with blocking peptides (Santa Cruz Biotechnology). The same specimens were immunostained with the rabbit polyclonal antibody against glutamine synthetase (GS; Santa Cruz Biotechnology) to compare the distribution of HGFA and GS.
Confocal Laser Scanning Microscopy (CLSM)
The sections of adult mouse liver tissues treated as described above were doubly stained with a mixture of antibodies for HGFA (N-19) and GS. The former antibody was detected by means of donkey anti-goat IgG conjugated with Alexa Fluor 488 and, after blocking treatment with normal goat serum, the latter was detected with the aid of goat anti-rabbit IgG conjugated with Alexa Fluor 568. Fluorescent labeled antibodies were purchased from Molecular Probes (Eugene, OR). Specimens were observed with an Olympus Fluo View CLSM (Olympus; Tokyo, Japan) equipped with Ar and He/Ne lasers.
Electron Microscopic Immunohistochemistry
Small pieces of liver tissue (approximately 1 mm × 0.5 mm × 0.5 mm) were fixed in 4% paraformaldehyde, 0.1% glutaraldehyde, and 100 mM sodium phosphate, pH 7.4, at 4C. After rinsing in a cold solution consisting of 100 mM lysine, 100 mM sodium phosphate, pH 7.4, and 150 mM NaCl and dehydration in a graded series of cold ethanol, they were embedded in Lowicryl K4M at −20C (Bendayan 1984). Ultrathin sections were cut on a Potter Blum MT2B ultramicrotome and collected on nickel grids with polyvinylformal coating, and immunostained with N-19 antibody and protein G–gold solution of 20-nm gold particles (ICN Biomedicals; Aurora, OH). Specimens were observed with a JEOL JEM 1200 electron microscope at an accelerating voltage of 80 kV after staining with uranyl acetate and lead citrate.
Results
Immunoblotting Assay
Immunoblot analysis demonstrated that the N-19 and A-1 antibodies were reactive with mouse HGFA (Figure 1A). These antibodies produced a single signal of 96 kD in response to mouse plasma, which suggests that the unactivated HGFA precursor in the circulation was detected. No discernible signal was obtained with the P1-4 antibody (data not shown).
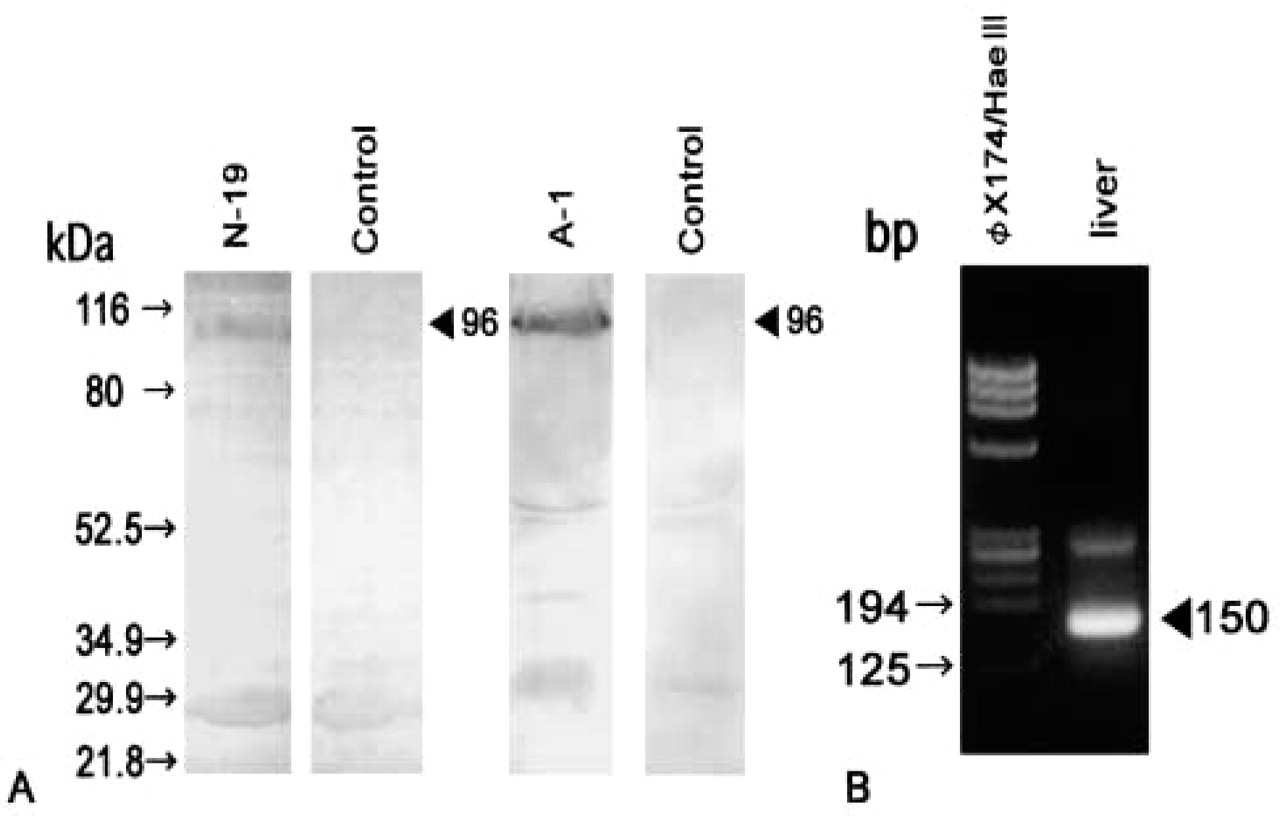
Immunoblotting analysis of plasma HGFA (
HGFA in Adult Liver
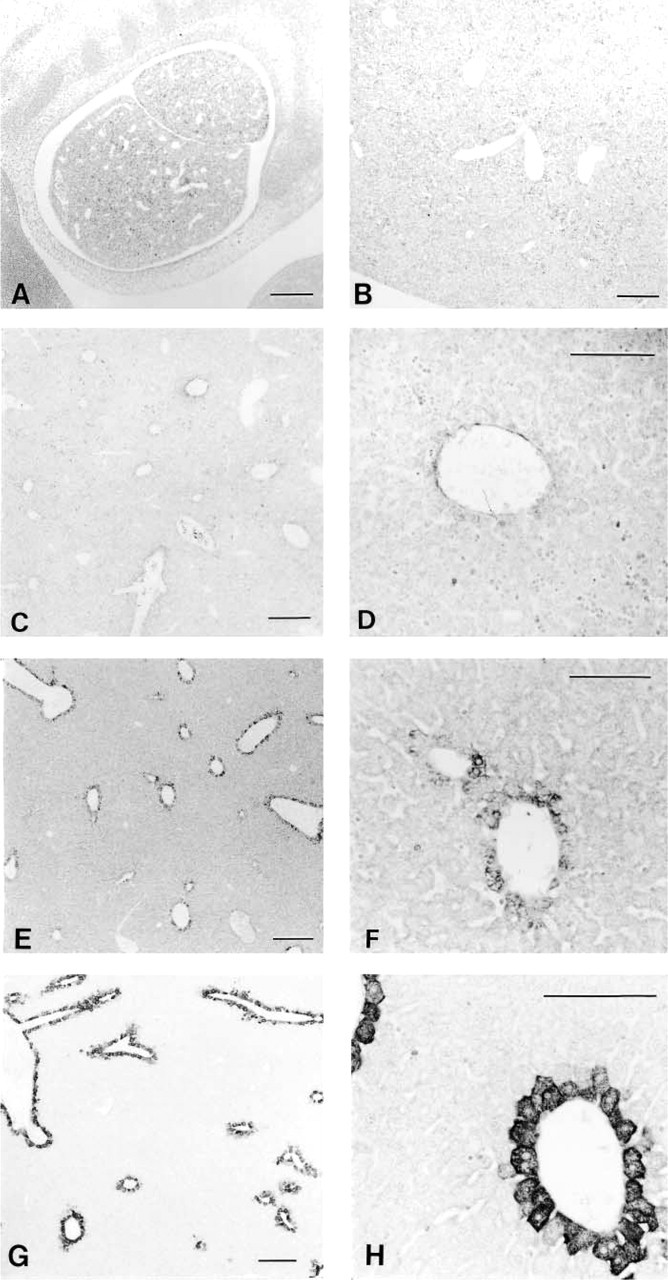
RT-PCR analysis demonstrated expression of HGFA mRNA in adult mouse livers. The predicted 150-bp fragment was consistently amplified from liver cDNA (Figure 1B). The PCR product was ascertained to correspond to the mouse HGFA cDNA sequence (data not shown). Light microscopic IHC using the N-19 antibody showed heterogeneous distribution of HGFA in the adult liver tissues at postnatal week 8 (Figure 2). Positive staining was restricted to the cytoplasm of hepatocytes adjacent to the efferent hepatic veins, including the central veins in the hepatic lobules, sublobular veins, and collecting veins (Figure 2A), but hepatocytes were mostly negative for HGFA in the other regions of liver tissue, including the periportal area. The largest hepatic veins were bordered by a one-cell-thick layer of HGFA-positive hepatocytes interspersed with HGFA-negative hepatocytes (Figures 2A and 2B). Around smaller efferent hepatic veins, only a one- or two-cell-thick layer of hepatocytes stained for HGFA, thus forming a lining of positive cells (Figures 2A and 2C). In the hepatic lobules, on the other hand, HGFA-positive hepatocytes were observed in a one- to three-cell-thick layer around the central veins (Figures 2A and 2D). The HGFA-positive hepatocytes showed cell–cell heterogeneity in staining intensity, ranging from faint to strong, with the strongly positive hepatocytes preferentially distributed near the hepatic veins. The control experiment using the absorbed antibody exhibited a marked reduction in staining intensity, thus confirming the specificity of the N-19 antibody (Figures 2E and 2F). All these findings were the same for both the ICR and C57BL/6J strains. Use of two other MAbs, A-1 and P1-4, resulted in the same staining pattern as that of N-19 antibody in PFA-fixed liver specimens (Figure 3). Electron microscopic IHC showed that the gold particles were ubiquitously distributed in the cytoplasm of the HGFA-positive hepatocytes, principally in the rough endoplasmic reticulum, whereas the nucleoplasm and cell organelles such as mitochondria and lysosomes of these cells were mostly negative (Figure 4).
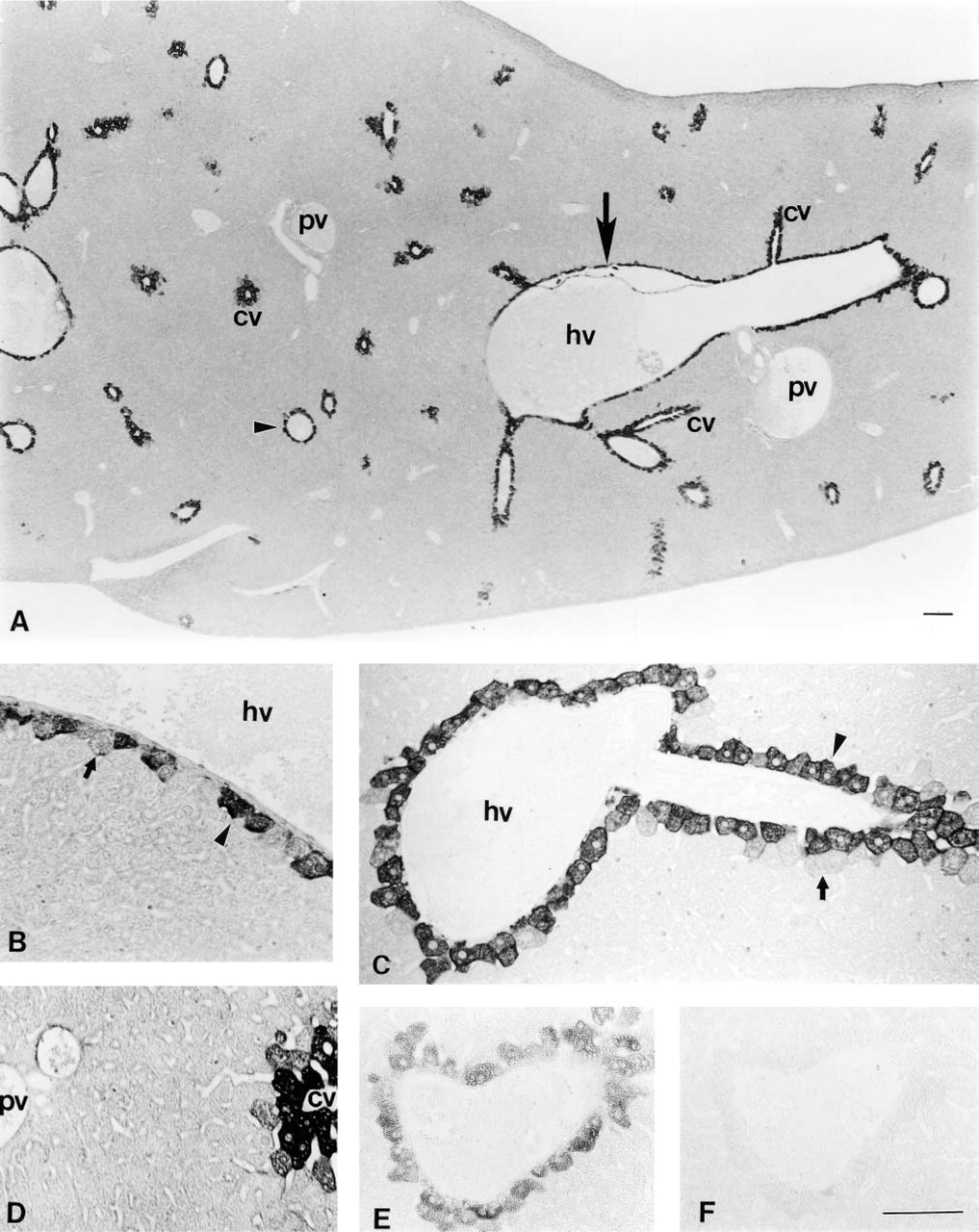
Immunohistochemical localization of HGFA in adult mouse liver tissues detected by light microscopy. (
Comparison of the staining pattern for HGFA in adult liver tissues in response to N-19 (
HGFA in Liver Development
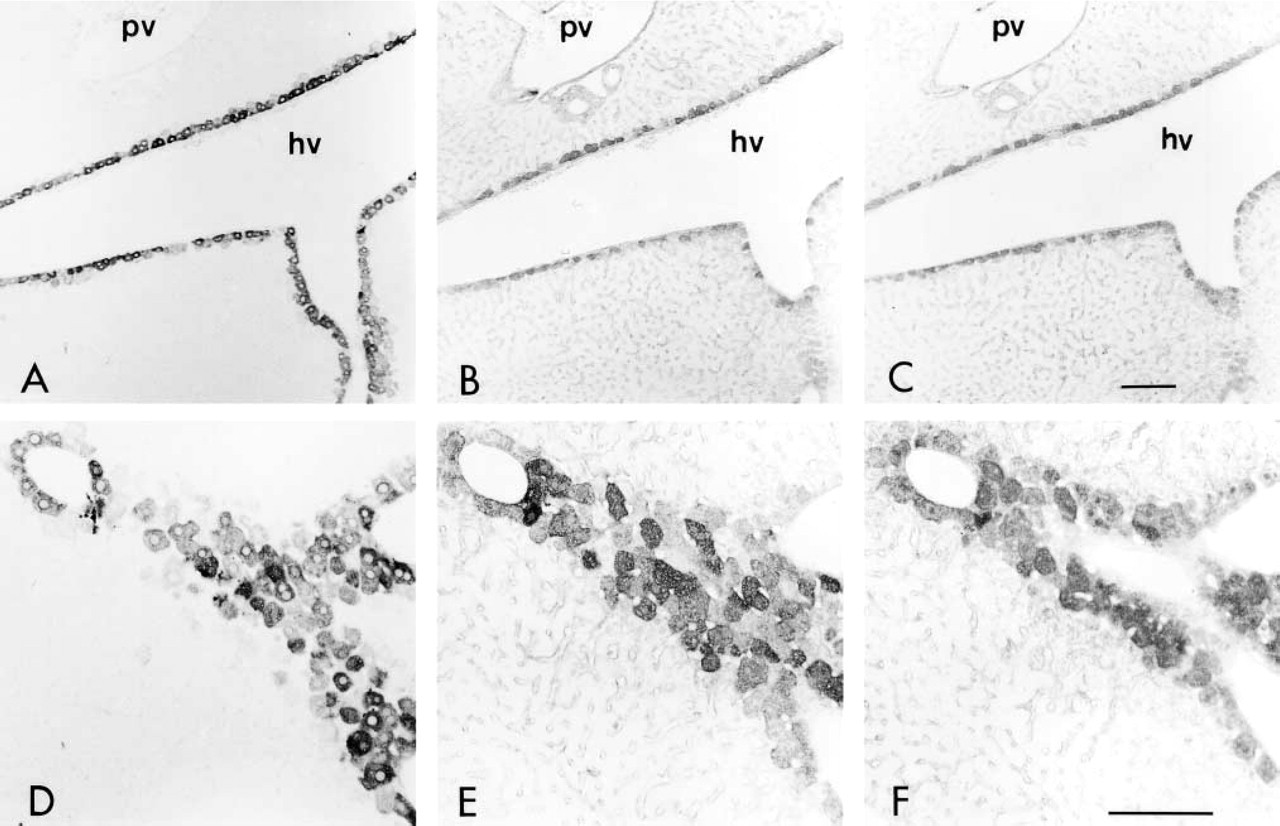
Immunoreactivity to HGFA and its localization were observed to change during liver development, while positive staining was not detected in the developing livers until postnatal day 0 (Figures 5A and 5B). On postnatal day 3, positive staining became somewhat detectable around the central veins in the hepatic lobules (Figures 5C and 5D). After this the immunoreactivity increased, and weak but detectable staining was observed on postnatal day 14 (Figures 5E and 5F). On postnatal day 30, positive staining was clearly observed in the hepatocytes surrounding the efferent hepatic veins (Figures 5G and 5H), similar to that seen in the adult liver. Throughout development of the liver, however, the periportal hepatocytes remained negative for HGFA.
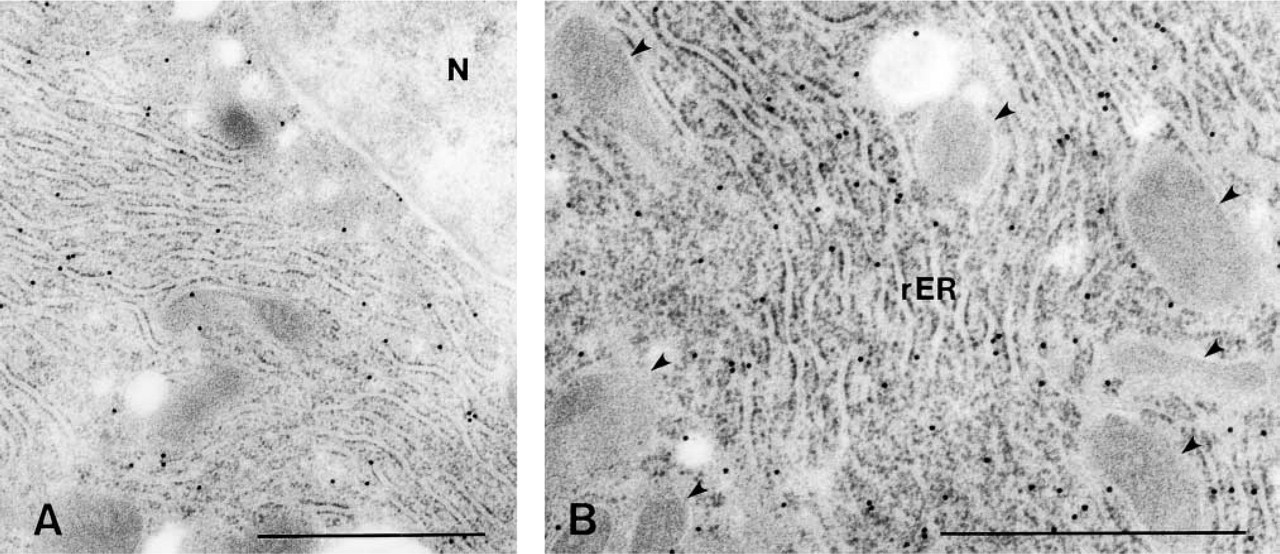
Subcellular localization of HGFA in hepatocytes. (
During liver development, HGFA exhibited a similar distribution pattern to that of GS, a representative enzyme synthesized by the pericentral hepatocytes (Figures 6A–6C). However, CLSM observation of adult liver tissues showed that the two antigens did not display a completely identical distribution, i.e., some pericentral hepatocytes were positive for GS but not for HGFA (Figures 6D–6F), indicating that HGFA has an intrinsic distribution pattern in the liver tissue different from that of GS.
Discussion
The study presented here showed that the localization of HGFA in the adult mouse liver was confined to the hepatocytes surrounding the efferent hepatic veins. The thickness of the HGFA-positive cell layer varied depending on the diameter of the hepatic vein branches.
HGFA immunoreactivity was mostly negative in the fetal and neonatal livers, became detectable in the hepatocytes adjacent to the pericentral veins by postnatal day 3, and was clearly observable by postnatal day 30. Use of the N-19 antibody cannot help determine whether the HGFA detected here is active or inactive, because both isoforms contain a long-chain subunit. Moreover, N-19 is an affinity-purified goat polyclonal antibody raised against an epitope mapping at the amino terminus of the long-chain subunit of human HGFA, while the peptide used to raise this antibody is identical in mouse and human HGFA (van Adelsberg et al. 2001). Because the use of the antibody absorbed with the peptide resulted in a significant decrease in staining intensity, HGFA or an identical epitope was shown to be present in the hepatocytes surrounding the efferent hepatic veins. Furthermore, two other antibodies, A-1 and P1-4, confirmed the unique distribution of HGFA. HGFA is supplied by circulating blood and by local expression in injured tissues. It is present in plasma as an inactive zymogen and is activated by thrombin and plasma kallikrein. HGFA selectively binds to the surface of epithelial cells expressing HAI-1 and its activity is suppressed during trapping by HAI-1. After shedding of HGFA, recovery of its activity begins to increase significantly. Strong immunoreactivity for HAI-1 has indeed been observed in regenerating tissues, but the HAI-1 message is not expressed in normal human hepatocytes (Shimomura et al. 1993; Kataoka et al. 1999,2000). Judging from the expression of mRNA in the liver, and the cytoplasmic localization of protein in the hepatocytes surrounding the efferent hepatic veins, HGFA detected here is considered to be the inactive HGFA precursor produced by those hepatocytes rather than ones binding to cell surfaces with HAI-1.
Developmental analysis of mouse liver HGFA on embryonic day 12 (
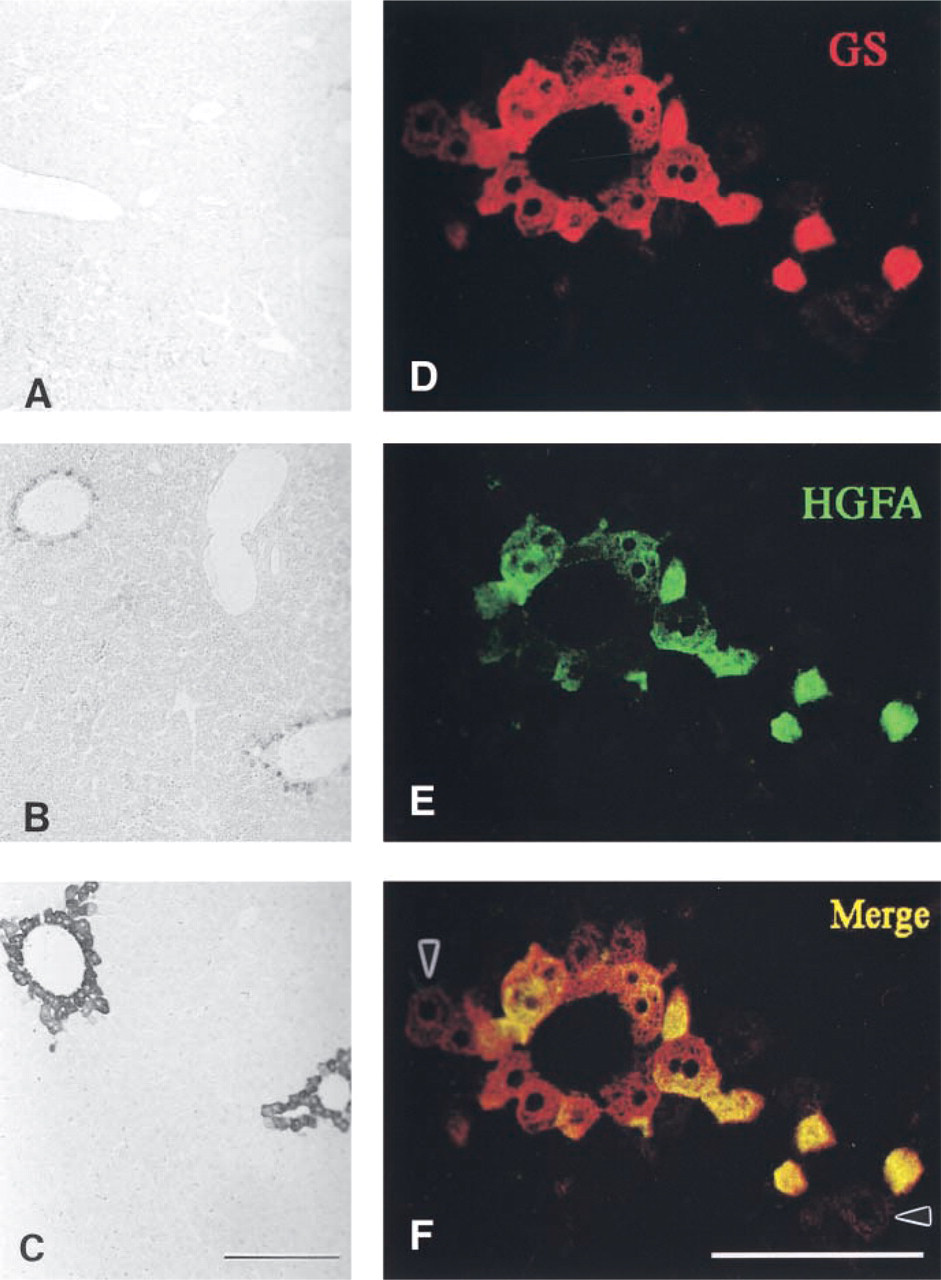
GS localization in developing liver tissues (
A similar localization pattern for HGFA was detected in GS of adult mouse livers, consistent with previously reported results for mouse and rat livers (Bennett et al. 1987; Gaasbeek Janzen et al. 1987; Shiojiri et al. 1995; Notenboom et al. 1997; Lamers et al. 1999). Some different patterns of antigen distribution and gene expression in adult mouse liver have also been reported. Carbamoylphosphate synthetase (CPS) has been found to occupy a 9- to 12-hepatocyte-thick layer around the afferent veins (Gaasbeek Janzen et al. 1987; Notenboom et al. 1997). Common liver-specific proteins such as albumin and α-1-antitrypsin are synthesized by all hepatocytes. The cytochrome P450, NADPH reductase, and glutathione-S-transferases, all enzymes involved in detoxification, are present either exclusively or predominantly in the 6–10-cell-deep layers of hepatocytes around the central vein (Bennett et al. 1987). HGFA has been identified as an acute-phase protein (Okajima et al. 1997) and glutamine as a regulator of acute-phase protein synthesis (Lavoinne et al. 1998). Our results indicate that HGFA is produced in part of the GS-producing hepatocytes. The functional relationship between HGFA, GS, and the other proteins, however, has remained completely unknown. On the other hand, a spatiotemporal relationship among HGF, c-Met, CPS, and GS has been reported in developing rat liver (Spijkers et al. 2001). With our fixation protocol, immunoreactivity for either HGFA or GS was detected from postnatal day 3, as also reported by Shiojiri et al. (1995). In contrast, some studies have suggested that the GS protein starts to accumulate in mouse hepatocytes as early as embryonic day 15 (Bennett et al. 1987; Notenboom et al. 1997). It has been suggested that this discrepancy depends on the effective antigen concentration of GS and its sensitivity to the fixation protocol.
A viable reason for the absence of detectable staining for HGFA in fetal and neonatal mouse livers is that HGFA is synthesized at too low a level for detection by the IHC technique employed. Another study found that the HGF mRNA level in the liver was very low during late gestation, increased remarkably in neonates, and reached a maximum 2 weeks after birth (Kagoshima et al. 1992). A possible explanation for this finding is that the amounts of HGF and/or HGFA necessary for liver development are very low, or that other HGF variants, which do not need the activator, are produced for development. In this connection, two natural splice variants of HGF were found to interact with a common c-Met receptor in vitro and in vivo (Miyazawa et al. 1991; Jakubczak et al. 1998; Otsuka et al. 2000). Another possibility is that the active HGF and HGFA are supplied by amniotic fluid, placenta, or maternal blood, because it was found that the placental villi and membranes show positive immunostaining for HGFA throughout gestation (Somerset et al. 2000b). HGF produced by the placental villous core might thus act to regulate trophoblast development or function through the c-Met, since the placenta was shown to contain the highest levels of HGF protein of all organs (Clark et al. 1996; Kauma et al. 1997; Kolatsi–Joannou et al. 1997). HGF levels in amniotic fluid are much higher than those in the umbilical cord blood and in the serum of pregnant women, especially during the second trimester (Horibe et al. 1995), and the level of HGF in fetal and first neonatal urine is much lower than that in amniotic fluid (Itakura et al. 1997; Okamura et al. 1998). The presence of an extremely high level of HGF in the amniotic fluid might also be involved in the maturation of fetal organs such as the lung and the digestive tract (Kurauchi et al. 1995). It has therefore been suggested that amniotic fluid HGF plays an important role in fetal growth. However, another study has stated the opposite view, i.e., that the placenta does not secrete HGF and that hepatic growth and development are independent of placental HGF (Somerset et al. 2000a). More work is clearly needed to verify the origin of HGF in embryos and to determine its actual activator.
The liver regenerates in a variety of proliferative patterns. In acute liver injuries of mice after CCl4 administration, the appearance of necrotic hepatocytes around the central veins was found to result in the random presence of proliferative hepatocytes in the lobulus. When mice are subjected to hepatectomy, the manner of hepatocyte proliferation varies depending on the amount of liver tissue removed. After 70% partial hepatectomy, a marked, random DNA synthesis by hepatocytes was observed in the lobulus, while after 30% partial hepatectomy, the cells undergoing DNA synthesis were located at the periportal zone of the lobulus (Ishiki et al. 1992). In rats with a two-thirds hepatectomy, the hepatic regenerative growth proceeded in a characteristic temporal and spatial pattern, in which several sequential waves of DNA synthesis and mitosis started in the periportal zone and ended in the pericentral zone (Rabes et al. 1976; Gebhardt 1988,1990). These different manners of HGF supply may contribute to the type of pattern of hepatocyte proliferation, i.e., the paracrine or endocrine mode. HGF supplied by the non-parenchymal liver cells affects the hepatocytes in the paracrine mode, whereas HGF from the distant organs does so in the endocrine mode. Differences in susceptibility to HGF between the pericentral and periportal hepatocytes may also be responsible for variations in liver regeneration. When the synthetic activity of DNA resulting from HGF stimulation was compared in cultured periportal and pericentral hepatocyte populations, the former showed a higher proliferative activity (Volk et al. 1995). The HGFA distribution pattern in the livers of adult and developmental mice therefore does not appear to be directly related to the proliferation pattern. The role and distribution of HGFA in hepatic regeneration therefore remain unknown and need to be further investigated.
Footnotes
Acknowledgements
Supported in part by the Kazato Research Foundation.
Mouse monoclonal antibodies for HGFA, P1-4, and A-1 were a generous gift from Dr Keiji Miyazawa (Department of Molecular Pathology, Graduate School of Medicine, University of Tokyo).
