Abstract
Various types of endogenous stem cells (SCs) participate in wound healing in the skin at different anatomical locations. SCs need to be identified through multiple markers, and this is usually performed using flow cytometry. However, immunohistological identification of endogenous stem cells in the skin at different anatomical locations by co-staining multiple SC markers has been seldom explored. We examined the immunohistological localization of four major types of SCs in wounded skin by co-staining for their multiple markers. Hematopoietic SCs were co-stained for Sca1 and CD45; mesenchymal SCs for Sca1, CD29, and CD106; adipose SCs for CD34, CD90, and CD105; and endothelial progenitor cells and their differentiated counterparts were co-stained for CD34, Tie2, and von Willebrand factor. We found Sca1+CD45+ SCs in the epidermis, dermis and hypodermis of wounded skin. Sca1+CD29+ and Sca1+CD106+ mesenchymal SCs, CD34+CD105+, CD34+CD90+, and CD90+CD105+ adipose SCs, as well as CD34+Tie2+ endothelial progenitor cells were also located in the epidermis, dermis, and hypodermis. This study demonstrates the feasibility of using immunohistological staining to determine the location of SCs in wounded skin and the intracellular distribution of their molecular markers.
Keywords
Introduction
Stem cells (SCs) are so-called because of their unique ability to self-renew and differentiate into a variety of cell lineages (Barrilleaux et al. 2006; He et al. 2009; Phinney and Prockop 2007; Raff 2003; Smith et al. 2007). Wounding recruits and activates SCs to home to wounded skin. SCs produce factors that modulate healing and proliferate and differentiate to replenish lost cells at the site. The typical endogenous SCs that contribute to skin wound healing include mesenchymal SCs (MSCs), adipose SCs (ASCs), endothelial progenitor cells (EpCs), hematopoietic SCs (HSCs), and epidermal SCs (Barrilleaux et al. 2006; Fathke et al. 2004; Hamou et al. 2009; Jeyarajah et al. 1986; Phinney and Prockop 2007; Plikus et al. 2012; Prockop 2009; Raff 2003; Seppanen et al. 2013; Smith et al. 2007). MSCs and ASCs promote healing and angiogenesis mainly by producing paracrine factors that act on neighboring cells; however, they can also differentiate into skin cells, including epithelial cells, endothelial cells, and/or fibroblasts (Badiavas et al. 2003; Brittan et al. 2005; Chen et al. 2008; Fathke et al. 2004; Hocking and Gibran 2010; Hong et al. 2013; Javazon et al. 2007; Jin et al. 2010; Khan et al. 2011; Kwon et al. 2008; Stepanovic et al. 2003; Tian et al. 2011; Wu et al. 2007). EpCs differentiate into endothelial cells during skin repair (Albiero et al. 2011; Nie et al. 2011; Ong and Sugii 2013) whereas HSCs differentiate into leukocytes (Jeyarajah et al. 1986).
One of the major hurdles hindering the elucidation of the precise roles of different SCs in wound healing is a lack of means to identify and locate diversified SCs in hair follicles, dermis, and hypodermis of wounded skin. In principle, each type of SC can be identified by staining for specific markers. SCs are readily identifiable using flow cytometry (Albiero et al. 2011; Bourin et al. 2013; Jensen et al. 2009; Rebelatto et al. 2008; Rostovskaya and Anastassiadis 2012; Sasaki et al. 2008; Tobita et al. 2011), which requires relatively large amounts of tissue to obtain sufficient numbers of cells (105-106 cells dispersed from as much as 1 g of tissue) (Marsee et al. 2010). In contrast, histological analysis can be performed on as few as tens of milligrams of tissue. Additionally, flow cytometry does not provide the location (within tens of micrometers) of SCs in wounded skin, or the cellular morphology or intracellular distribution of molecules within the cells. Western blotting and PCR can also be used to detect SCs in skin, but these techniques are also limited by their inability to localize cells anatomically.
Florescence-tagged SCs, infused or transplanted into animals, will mimic the endogenous untagged SCs and provide an opportunity to study the location of the cells. However, the validity of these results remains a concern, as the tags or tagging process may alter the behavior of the proteins or the SCs themselves (Michaelson and Philips 2006; Skube et al. 2010; Taghizadeh and Sherley 2008). Transgenic reporter mouse strains and lineage tracing experiments are well-established, definitive approaches to track SCs (Snippert and Clevers 2011). These tools are continuously used in wide range of SC research so long as the needed transgenic reporter mouse strains or lineage tracing animals are available. If not, the animals need to be first generated. As an alternative, an immunohistological method combined with confocal microscopy with multiple wavelength excitation and signal acquisition may offer a way to localize endogenous SCs in tissues, using less tissue samples as compared with other methods.
Protein markers expressed by SCs are critical for SC localization in wounded skin. Stem cell antigen 1 (Sca1) and CD45 are expressed by HSCs and are thus commonly used for HSC identification (Granick et al. 2013; Hamou et al. 2009; Meindl et al. 2006). In addition, the GFP-labeled MSC lineage populations have been used to examine epidermis and dermis wound repair processes (Borue et al. 2004; Harris et al. 2004; Tian et al. 2011; Wu et al. 2010). MSCs express high levels of CD29 or CD106 and Sca1, and therefore immunohistological co-staining of CD29 or CD106 and Sca1 can be used to localize MSCs in wounded skin. CD34, CD90, and CD105 are expressed by ASCs, but absent or weakly expressed by MSCs, and are thus considered as good markers for detecting ASCs (De Francesco et al. 2009; Festa et al. 2011; Ong and Sugii 2013; Rostovskaya and Anastassiadis 2012; Tobita et al. 2011; Zimmerlin et al. 2011). EpCs are usually identified by multiple markers, including CD34 and Tie2, using flow cytometry (Chen et al. 2013; Sheridan et al. 2006). Additionally, EpC differentiation into endothelial cells can be traced using CD34 plus identification of the endothelial marker, von Willebrand factor (vWF) (Albiero et al. 2011; Chen et al. 2013; Sheridan et al. 2006). In this report, we devised an immunohistological approach to localize and discriminate the four main types of endogenous stem cells (HSCs, MSCs, ASCs, and EpCs) in wounded skin by staining different combinations of antigens prominently present in the each type of SC. The resulting scheme should facilitate analysis of homing, recruitment flux, and fate of endogenous stem cells during wound healing under various pathophysiological and pharmacological conditions.
Materials & Methods
Animals and Procedures: Full-Thickness Incisional Wounding
Established protocols were used (Ashcroft et al. 1997), and studies were conducted in conformity with the Public Health Service Policy on Humane Care and Use of Laboratory Animals. Animal protocols were approved by the Institutional Animal Care and Use Committee and Institutional Review Board of Louisiana State University Health Sciences Center, New Orleans; and followed the National Institutes of Health guidelines for experimental animal use. For wounding, mice (~19 g body weight, C57BL/6, 10 weeks old) (Jackson Laboratories, Bar Harbor, ME) were anesthetized by intraperitoneal injection of ketamine hydrochloride (0.2 mg/Kg body weight) and xylazine (0.4 mg/Kg body weight). The hair on the dorsal side was removed by Veet depilating cream, and the area was cleaned with 70% ethanol. Two full-thickness incisions (each 20-mm length) on the dorsal side were created symmetrically across the midline between the 5th thoracic vertebrae and the ischium bone. Incisions were closed with two sutures (Ethilon 6-0). The sutures were removed at day 5 post-wounding. Hair was similarly removed from sham-treated controls but incisions were not made.
Collection and Immunohistology of Wounded Skin
At day 10 post-wounding, mice (n=6/group) were perfused via cardiac puncture with PBS−/− (without calcium and magnesium) at a flow rate of 5 ml/min for 5 min under anesthesia, and then sacrificed. Wounds and a 2-mm margin of normal surrounding skin were excised, fixed in 4% paraformaldehyde (4 hr, ~23C), incubated in 30% sucrose (12 hr, 4C), embedded in OCT, and then stored at -80C, as reported previously (Lu et al. 2010; Tian et al. 2010). Unwounded skin from the same position on the dorsal side from sham controls was similarly processed. Serial sagittal cryosections (7-µm-thick) were cut using a Leica CM3050s cryostat microtome (Buffalo Grove, IL) and transferred to Superfrost slides (Fisher Scientific, Pittsburgh, PA). Cryosections were treated sequentially as follows: incubated in methanol/acetone (1/1) (20 min, -20C), washed with 1× phosphate-buffered saline (PBS) for 10 min for the removal of OCT; incubated in citrate buffer (20 min, 90–100C), then in blocking buffer [5% Goat/Donkey serum (MP Biomedical, Solon, OH), and 0.3% trition-X-100 (Fluka, St. Louis, MO)] for 1 hr, ~23C; incubated with two anti-mouse primary antibodies that were raised from two different hosts and diluted as in Table 1 (4C, 12 hr), washed thrice with 1× PBS (10 min, ~23C), incubated with two secondary antibodies (labeled with Alexa Fluor 488 (green) and Alexa Fluor 568 (red), raised from compatible hosts) at 23C for 1 hr, and then washed thrice with 1× PBS (10 min, ~23C). Nuclei were counterstained with 4′-6-diamidino-2-phenylindole (DAPI, 1:2000, 10 min) (Life Technologies, Grand Island, NY). Slides were rinsed with 1× PBS (10 min, ~23°C, 3 times) to remove extra DAPI, mounted using mounting media (Vector Laboratories, Burlingame, CA), and imaged using a Zeiss 510 Meta confocal laser-scanning microscope (Carl Zeiss, Jena, Germany) with a 40× oil immersion objective (numerical aperture: 1.25). The images were analyzed by Zen 2011 software (Carl Zeiss) and annotated for presentation using Adobe Photoshop (Adobe Systems, Inc. San Jose, CA).
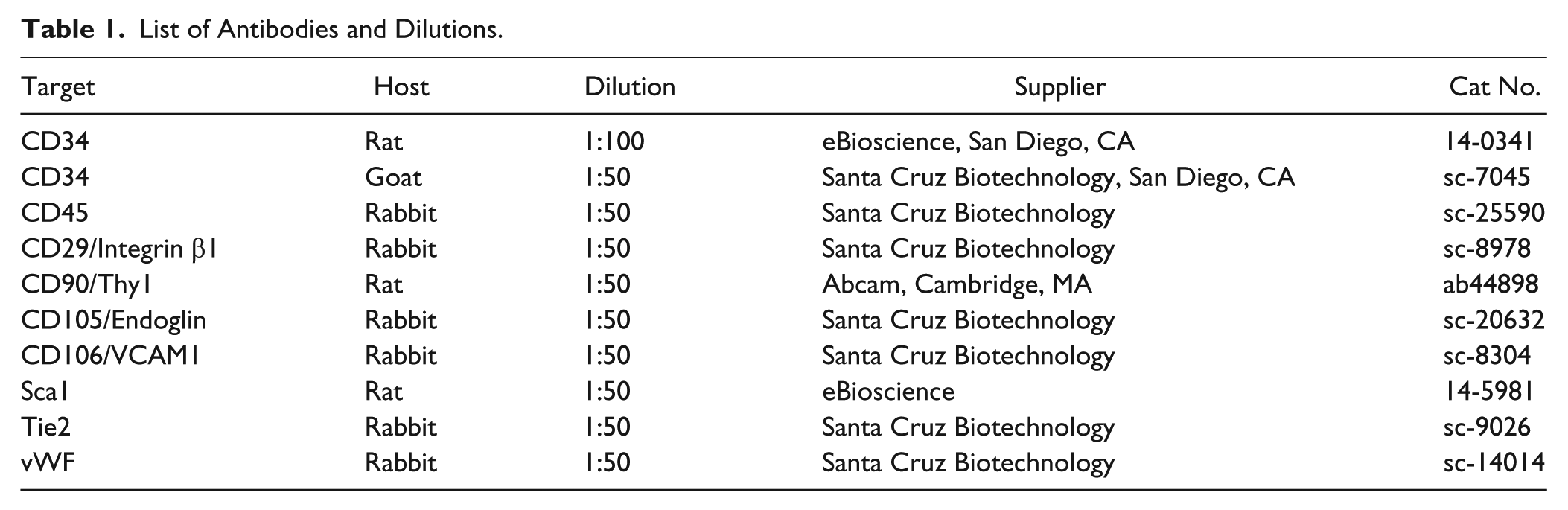
List of Antibodies and Dilutions.
Results
Immunohistological Localization of HSCs in Wounded Skin
Localization of typical HSCs was studied by staining cryosections of wounded skin for specific HSCs markers, CD45 and Sca1 (Table 2; Fig. 1). Confocal images revealed Sca1+CD45+ HSCs in the epidermis (Fig. 1A), dermis (Fig. 1A, 1B) and hypodermis (Fig. 1C) of wounded skin. Sca1+CD45+ HSCs were also found in the dermis and hypodermis (Fig. 1E, 1F), but not in epidermis (Fig. 1D) of unwounded skin. These results show that immunofluorescence staining and confocal imaging can uncover the anatomical locations of Sca1+CD45+ in wounded skin.
Specific Marker Expression in Different Types of Stem Cells.
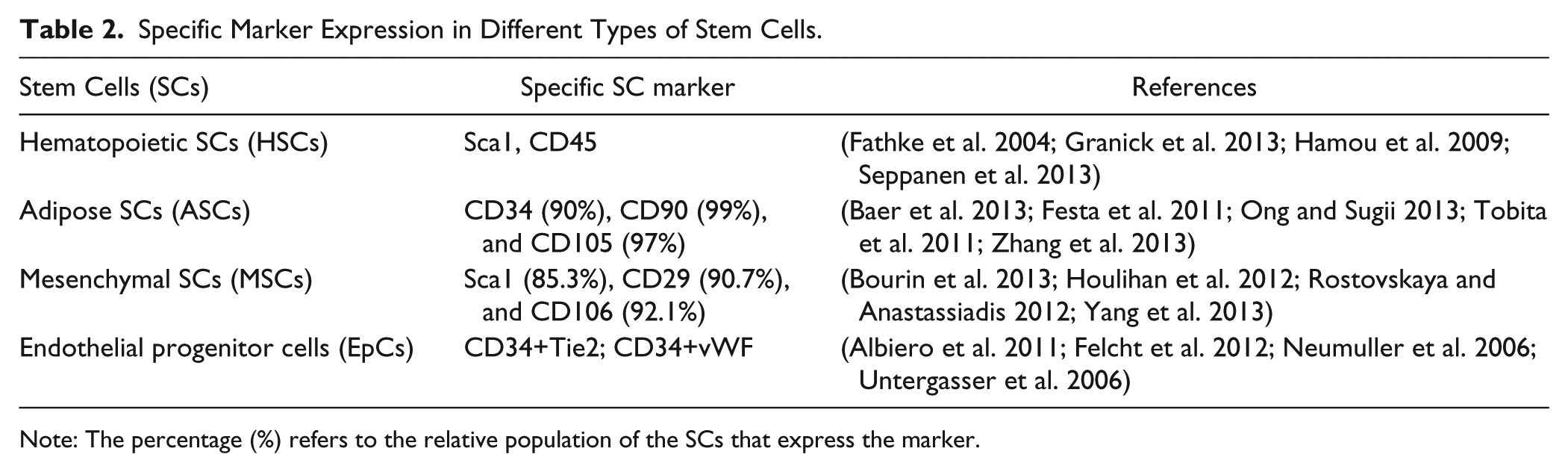
Note: The percentage (%) refers to the relative population of the SCs that express the marker.
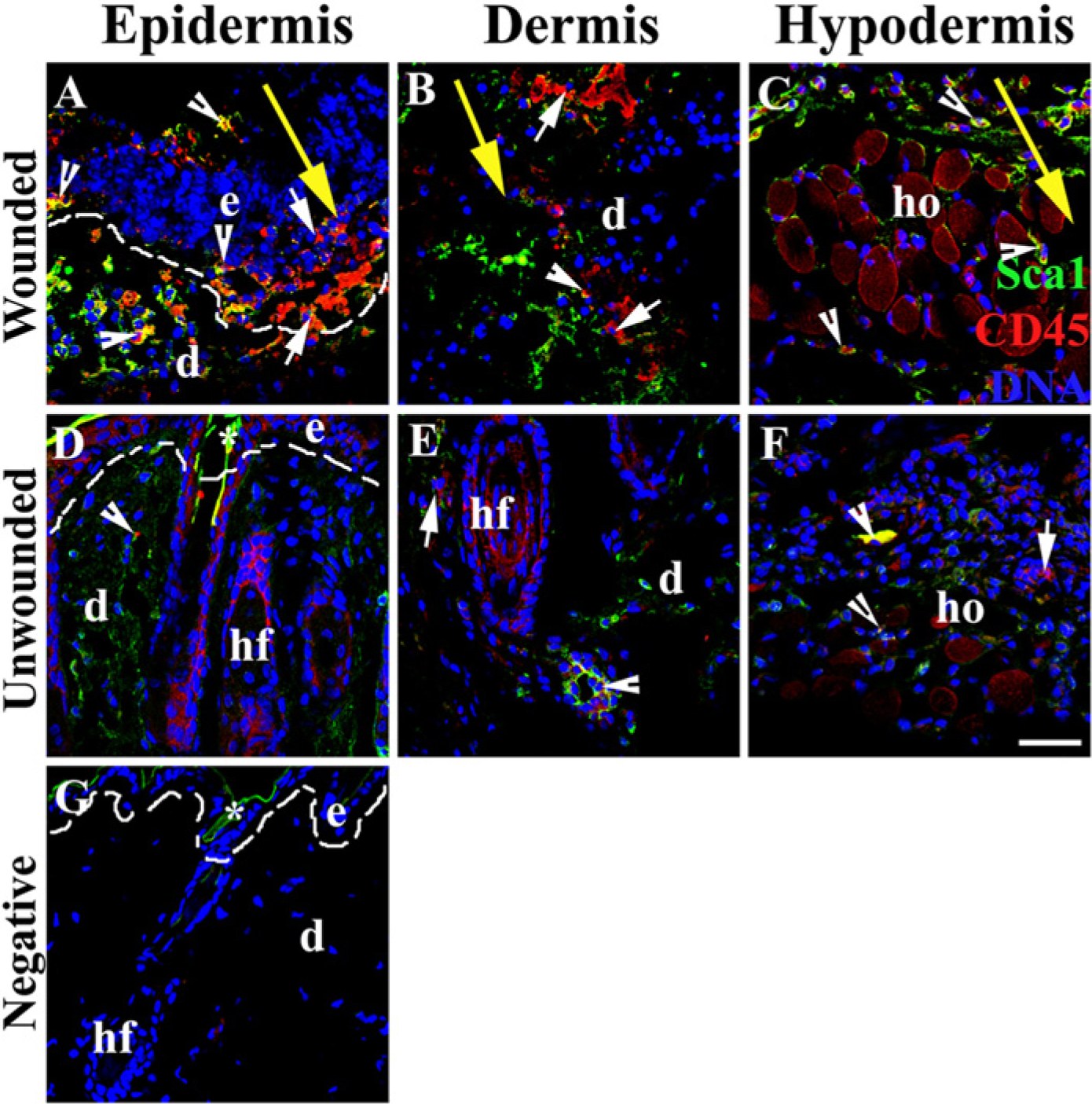
Localization and morphological observation of endogenous hematopoietic stem cells (HSCs) in wounded skin. Sca1+CD45+ cells (white short arrow) and Sca1−CD45+ cells (white arrowhead) resided in the epidermis (A), dermis (B) and hypodermis (C) of wounded skin. The Sca1+CD45+ and Sca1−CD45+ cells were also observed in the dermis (E) and hypodermis (F) of unwounded skin. (G) Image showing the non-specific binding of the secondary antibodies. Nuclei were counterstained with DAPI (blue). Abbreviations: bu-bulge; ho-hypodermis; d-dermis; e-epidermis; hf-hair follicle; dashed line (—)-border of dermis and epidermis. The large yellow arrow indicates the site of the incision wound, and marks the direction from the epidermis to the hypodermis. Asterisk (*) indicates non-specific binding of secondary antibodies. Bar, 38 µm.
Immunohistological Identification and Localization of Mesenchymal Stem Cells in Wounded Skin
MSCs are usually identified by flow cytometric analysis of multiple markers including Sca1, CD29, and CD106 (Table 2) (Rostovskaya and Anastassiadis 2012; Tobita et al. 2011). However, histological localization of endogenous SCs in situ is usually done by single-marker staining (Hong et al. 2009). To explore the feasibility of histological localization and morphological observation of multiple types of endogenous SCs in wounded skin, we double-stained skin cryosections with combinations of MSC markers Sca1, CD29, and CD106 (Fig. 2) (Rostovskaya and Anastassiadis 2012; Tobita et al. 2011). Sca1+CD29+ and Sca1+CD106+ MSCs were observed in wounded epidermis (Fig. 2A-2C, 2J-2L) and dermis (Fig. 2D-2F, 2M-2O) and in unwounded epidermis and dermis (Fig. 2G-2I & 2P-2R). The increased numbers of positively stained cells in the epidermis and dermis around the wound site suggest that endogenous MSCs may regenerate skin cells to heal the wound.
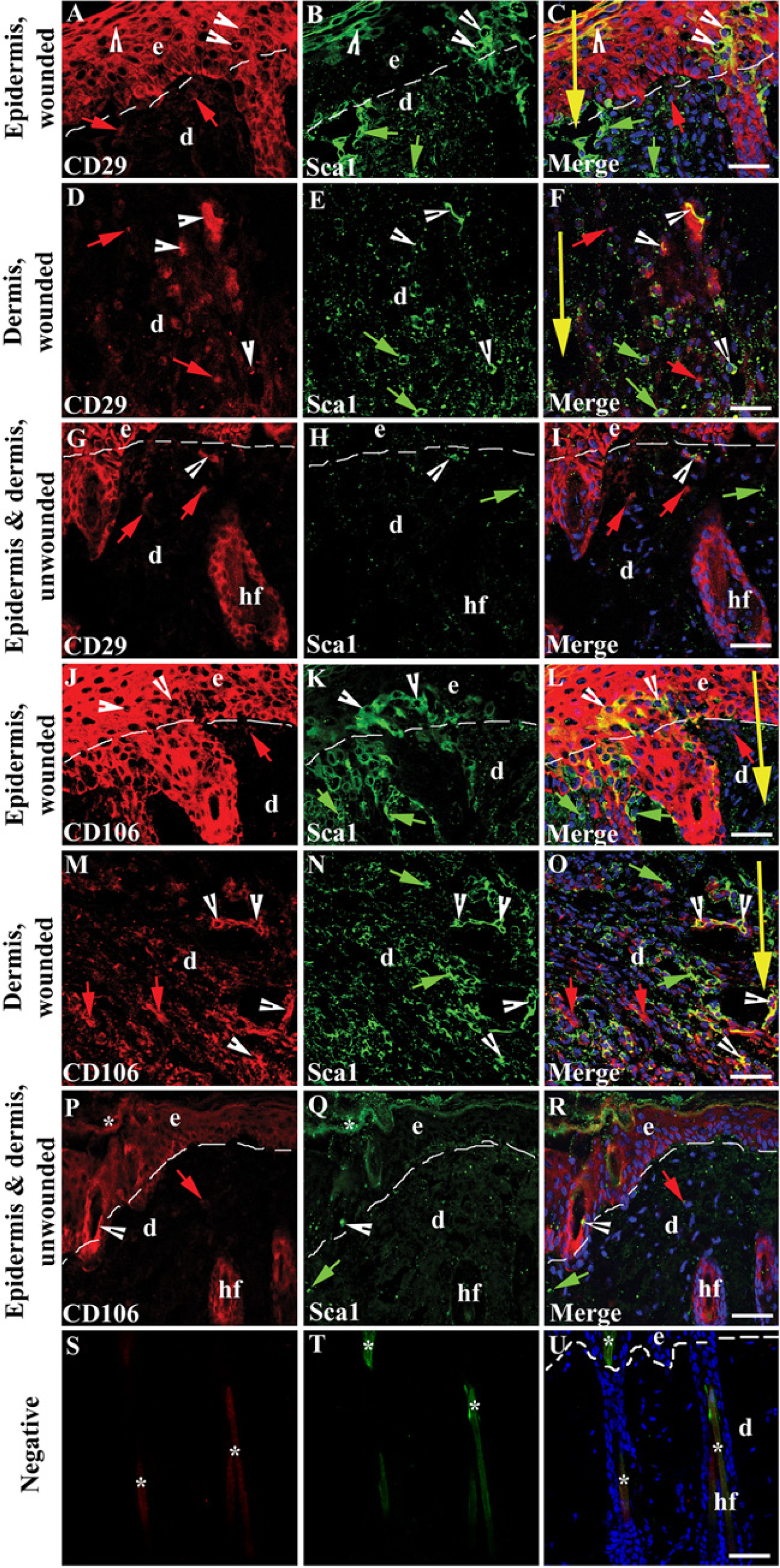
Localization of endogenous MSCs in wounded skin using combinations of typical MSC markers: (A, D, G) CD29 (red; red arrows); (J, M, P) CD106 (red; red arrows), and (B, E, H, N, Q) Sca1 (green; green arrows). (C, F, I, L, O, R) Merged images. White arrowheads show (A-F) CD29+Sca1+ cells and (J-O) CD106+Sca1+ cells in wounded skin epidermis and dermis. Fewer CD106+Sca1+, Sca1+, or CD106+ cells were present in unwounded skin (G-I; P-R) as compared with wounded skin (A-F; J-O). Negative staining (S-U) shows non-specific binding of secondary antibodies. Nuclei were counterstained with DAPI (blue). Abbreviations: e-epidermal; d-dermis; hf-hair follicle; dash line(—)-border of dermis and epidermis. The large yellow arrow indicates the site of the incision wound, and marks the direction from the epidermis to the hypodermis. Asterisk (*) indicates non-specific binding of secondary antibodies. Bars, (C & L) 42 µm; (F, I, O, R, U) 38 µm.
Adipose Stem Cells in Wounded Skin Localized by Immunohistological Co-staining of their Markers
ASCs are often identified by flow cytometric analysis using multiple markers, such as CD34, CD90, and CD105 (Table 2) (Zhang et al. 2013). Based on the flow cytometry method, we double-stained skin cryosections with combinations of ASC markers (Fig. 3) (Chen et al. 2007; Rostovskaya and Anastassiadis 2012; Tobita et al. 2011). CD34+CD90+ (Fig. 3A-3D), CD34+CD105+ (Fig. 3E, 3G, 3H), and CD90+CD105+ (Fig. 3I-3L) ASCs were observed in the dermis and hypodermis of wounded skin. Higher proportions of each ASC subgroup existed in the wounded dermis (Fig. 3A, 3E, 3I) and hypodermis (Fig. 3C, 3G, 3K) as compared with the unwounded dermis (Fig. 3B, 3F, 3J) and hypodermis (Fig. 3D, 3H, 3L). Cells that stained positively for only one marker are unlikely to be ASCs (Fig. 3).
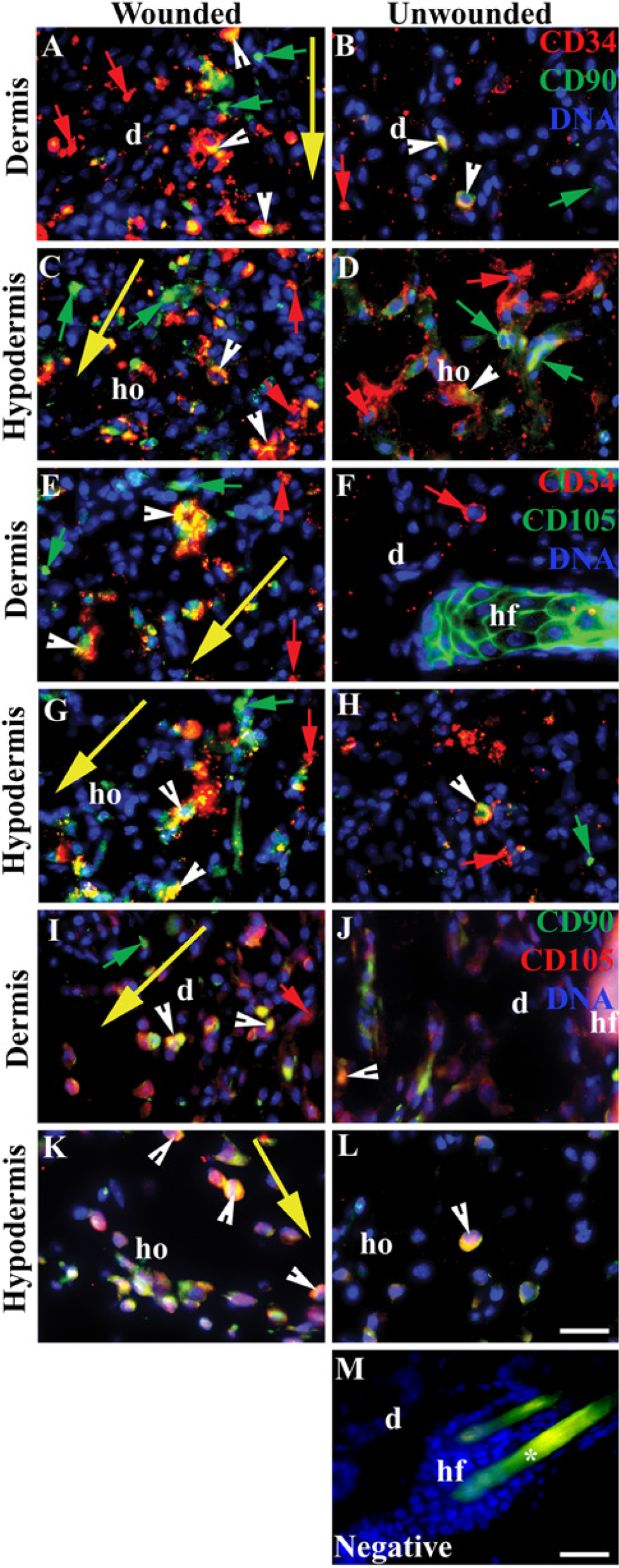
Localization of endogenous adipose stem cells (ASCs) in wounded skin using combinations of specific ASC markers: CD34, CD90, and CD105. Each cryosection was co-stained for two of the three ASC markers. (A-D) CD34 (red; red arrows) and CD90 (green; green arrows); (E-H) CD34 (red; red arrows) and CD105 (green; green arrows); (I-L) CD90 (green; green arrows) and CD105 (red; red arrows). Cells in the dermis and hypodermis positive for two markers are regarded as ASCs. Double-positive cells are yellow and indicated by white arrowheads. Negative staining (M) shows non-specific binding of the secondary antibodies. Nuclei were counterstained with DAPI (blue). Abbreviations: d-dermis, ho-hypodermis. The large yellow arrow indicates the site of the incision wound, and marks the direction from the epidermis to the hypodermis. Asterisk (*) indicates non-specific binding of secondary antibodies. Bar, 28 µm.
Localization of Endothelial Progenitor Cells and their Differentiated Counterparts in Wounded Skin
CD34 and Tie2 was used to identify EpCs (Table 2), and CD34 and vWF to identify endothelial cells differentiated from EpCs via flow cytometry (Albiero et al. 2011; Chen et al. 2013; Padfield et al. 2010; Sheridan et al. 2006). vWF is a marker of endothelial cells (Chen et al. 2013; Sheridan et al. 2006). Therefore, we stained the skin sections for CD34 (green) and Tie2 (Fig. 4A, 4B) or vWF (Fig. 4C, 4D) (red). The CD34+Tie2+ cells (Fig. 4A, shown by white arrowheads) are EpCs, and CD34+vWF+ cells (Fig. 4C,D) (white arrowhead) are endothelial cells differentiated from EpCs. These histological images demonstrate that there are more CD34+Tie2+ EpCs and CD34+vWF+ endothelial cells in wounded skin compared with unwounded skin (Fig. 4).
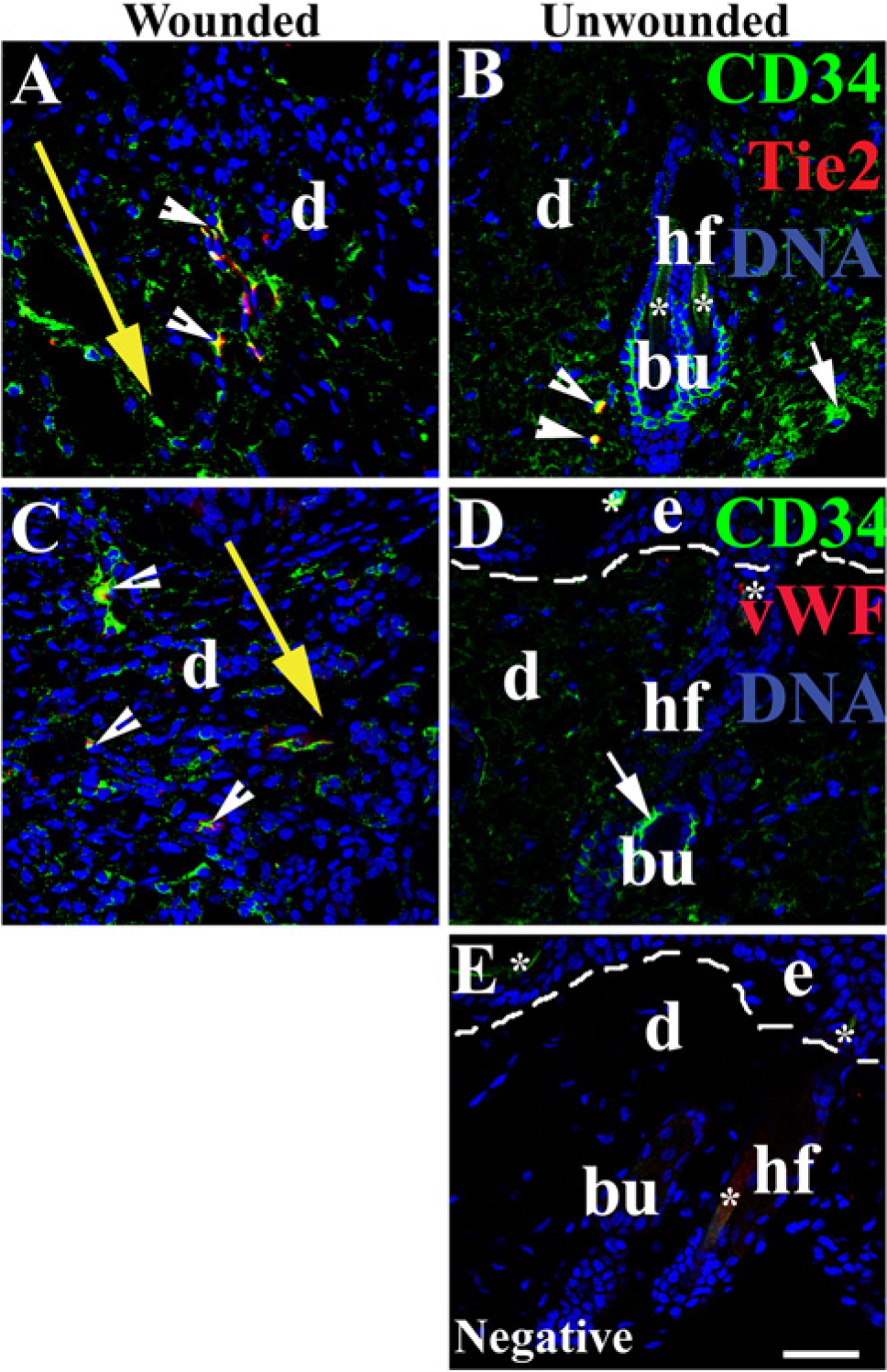
Localization of endothelial progenitor cells (EpCs) and differentiated endothelial cells in wounded skin. (A-B) EpCs were localized by co-staining CD34 and Tie2. (C-D) The differentiation of EpCs to endothelial cells was identified by co-staining CD34 and vWF. Negative staining image (E) shows unspecific binding of secondary antibodies. Double-positive cells are yellow and indicated by white arrowheads. Nuclei were counterstained with DAPI (blue). Abbreviations: e-epidermal; d-dermis; hf-hair follicle; bu-bulge; dash line(—)-border between epidermis and dermis. The large yellow arrow indicates the site of the incision wound, and marks the direction from the epidermis to the hypodermis. Asterisk (*) indicates non-specific binding of secondary antibodies. Bar, 38 µm.
Discussion
A variety of endogenous SCs participate in skin wound healing (Daley 2012; Plikus et al. 2012; Prasongchean and Ferretti 2012). However, the anatomical distribution, morphology, and intracellular molecular arrangements of different SCs during skin healing in vivo cannot be readily assessed by flow cytometry and are not well defined. We undertook a histological approach to identify properties of SCs in skin wound healing. We have identified and localized the major endogenous SCs (HSCs, MSCs, ASCs, and EpCs) in wounded skin by immunohistological analysis of sections co-stained for multiple markers.
The anatomical niche for endogenous MSCs in different parts of skin (epidermis, dermis, and hypodermis) is still not well understood. There are reports, however, indicating that MSCs are present in follicular connective sheath tissue and in the papilla of the dermis (Hoogduijn et al. 2006; Kruse et al. 2006; Lako et al. 2002; Richardson et al. 2005). It has also been reported that 4% of transplanted tagged-MSCs engraft into the epidermis (Borue et al. 2004; Harris et al. 2004). Other studies have provided evidence for the localization of dermal MSCs in skin independent of the hair follicles (Chen et al. 2007; Lorenz et al. 2008; Toma et al. 2005). All studies have been performed by flow cytometry in association with complementary immunohistological staining of single MSC markers (Hong et al. 2009). The flow cytometry used in these studies is a limiting factor, and cannot localize MSCs in the skin. Staining of only one marker may lead to a misidentification of SCs because of the potential off-target staining resulting from the expression of a single marker in non-MSCs. We observed that Sca1+CD29+ and Scal+CD106+ cells were co-localized in the epidermis and dermis, but that there were far fewer dual-positive cells than single-positive Scal+, CD29+, or CD106+ cells (Fig. 2). Sca1 is expressed by hematopoietic SCs. CD29 and CD106 are also expressed by epithelial cells (Hong et al. 2009; Kim et al. 2002). Therefore, to avoid mistakenly identifying HSCs, epithelial cells, or other cell types as MSCs, MSCs should be identified and localized in skin sections based on co-staining with the combination of Sca1 and CD29 or Sca1 and CD106. Similarly, cells expressing both Sca1 and CD29 are Sca1+CD29+ HSCs; however, cells expressing only one of Sca1 or CD45 are unlikely to be so.
Sca1 is a member of the Ly6A superfamily of glycophosphatidylinositol (GPI)-anchored membrane proteins (Holmes and Stanford 2007), involved in SC self-renewal, fate decisions, and lipid raft signaling via weak protein-protein interaction in HSCs (Holmes and Stanford 2007; Okada et al. 1992; Spangrude et al. 1988). CD45 is a hematopoietic cell marker present on the surface of HSCs and plays a critical role in survival, expansion, differentiation and migration of HSCs during the process of hematopoiesis (Kiel and Morrison 2008; Raaijmakers and Scadden 2008). In the present study, endogenous Sca1+CD45+ HSCs were identified in the epidermis, dermis, and hypodermis of wounded skin, but not in the epidermis of unwounded skin, implying a role for HSCs in wound healing of the epidermis. Further, Sca− CD45+ cells were located in the epidermis, dermis, and hypodermis in unwounded and wounded skin. Our observation of HSCs in skin wounds is consistent with the published research conducted by other groups (Etich et al. 2013; Meindl et al. 2006; Passier and Mummery 2003).
CD34 is a cell-surface glycoprotein thought to mediate the attachment of SCs to the extracellular matrix or directly to stromal cells (Nielsen and McNagny 2008). This implies that CD34 may play a role in the reparative function of CD34-containing SCs. We have localized ASCs to the dermis and hypodermis by co-staining with CD34, CD105, and CD90, which are reported to be maintained at high levels in ASCs (Table 2) (Chen et al. 2010; Folgiero et al. 2010; Tobita et al. 2011) and which do not occur, or occur at very low levels, in MSCs (Zhang et al. 2013). On the other hand, ASCs do not express, or express at very low levels, the MSC markers Sca1, CD29, and CD106 (Chen et al. 2010; Ong and Sugii 2013; Rostovskaya and Anastassiadis 2012; Tobita et al. 2011; Vaculik et al. 2012).
This study was aimed at differentiating among SC populations of ASCs, MSCs, and HSCs, and assessing their involvement in wounded skin qualitatively. To this end, we have explored the localization of the major endogenous stem cells—HSCs, MSCs, ASCs, and EpCs—by co-staining multiple typical markers of each type of SC in sections of wounded murine skin. We demonstrated the feasibility of immunohistological co-staining for localizing endogenous, untagged SCs in wounded skin, and for observing the intracellular distribution of the proteins. Although this report is limited by the small number of SC markers tested, additional SC markers could be assessed to confirm the localization of SCs, and to identify potential new SC phenotypes that may play roles in cutaneous wound healing. Staining of multiple SC markers of skin sections with more than three colors could be developed in combination with advanced confocal microscopic imaging. If successful, such multiplexed staining could offer more definitive localization and morphological understanding of SCs in wounded skin, which could in turn facilitate the development of new diagnostic methods for the treatment of chronic wounds. Our approach is complementary to methodology that uses the transgenic reporter mouse strains or lineage tracing experiments. For example, we can use this relative easy immunohistological approach to make the initial observation or narrow down the studies, then use the transgenic reporter mouse strains and lineage tracing experiments to make a final observation. Alternatively, if this type of immunohistological approach yields potential new SC markers, then the markers can be selected for lineage tracing experiments or for the development of new transgenic reporter mouse strains for definitive discovery. This report could provide a complementary tool that is more accessible, affordable, and realistic to investigators, especially to those in the wound repair community, who may be limited by the availability of transgenic animals for lineage tracing or by the small sample sizes of tissue when studying SCs in wounded skin.
Footnotes
Acknowledgements
Many thanks to Dr. Eric C.B. Milner for excellent editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by NIH grant 5R01DK087800 (S.H.).
