Abstract
Soluble epoxide hydrolase (sEH) is a phase-I xenobiotic metabolizing enzyme having both an N-terminal phosphatase activity and a C-terminal epoxide hydrolase activity. Endogenous hydrolase substrates include arachidonic acid epoxides, which have been involved in regulating blood pressure and inflammation. The subcellular localization of sEH has been controversial. Earlier studies using mouse and rat liver suggested that sEH may be cytosolic and/or peroxisomal. In this study we applied immunofluorescence and confocal microscopy using markers for different subcellular compartments to evaluate sEH colocalization in an array of human tissues. Results showed that sEH is both cytosolic and peroxisomal in human hepatocytes and renal proximal tubules and exclusively cytosolic in other sEH-containing tissues such as pancreatic islet cells, intestinal epithelium, anterior pituitary cells, adrenal gland, endometrium, lymphoid follicles, prostate ductal epithelium, alveolar wall, and blood vessels. sEH was not exclusively peroxisomal in any of the tissues evaluated. Our data suggest that human sEH subcellular localization is tissue dependent, and that sEH may have tissue- or cell-type-specific functionality. To our knowledge, this is the first report showing the subcellular localization of sEH in a wide array of human tissues.
E
sEH possesses an N-terminal phosphatase activity and a C-terminal epoxide hydrolase activity (Cronin et al. 2003; Newman et al. 2003). Endogenous hydrolase substrates include arachidonic acid epoxides, which have multiple biological functions (Capdevila et al. 2000) including blood pressure regulation (Sinal et al. 2000; Imig et al. 2002) and inflammation (Node et al. 1999). Hence, sEH has become a potential therapeutic target for treatment of hypertension (Jung et al. 2005) and modulation of inflammation (Schmelzer et al. 2005).
Initially, sEH was mitochondrial based on enzyme activity in different subcellular fractions from mouse liver (Gill and Hammock 1981a, b). Subsequent experiments in mice using isopycnic subfractionation and enzyme markers revealed both a peroxisomal and cytosolic localization (Waechter et al. 1983; Kaur and Gill 1986). This was followed, however, by immunocytochemical experiments showing the localization of sEH solely in peroxisomes of mouse and rat liver. The authors attributed this discrepancy to limitations of the subcellular fractionation approach such as peroxisomal fragmentation and contamination from other fractions (Hollinshead and Meijer 1988). Later, mice hepatocyte digitonin permeabilization studies showed the majority of sEH to be localized in the cytosol (Eriksson et al. 1991). Arand et al. (1991) suggested that the dual compartmentalization was due to an impaired peroxisomal targeting sequence. More recently, analysis of the sEH peroxisomal targeting sequence (PTS-1) and localization via immunofluorescence microscopy in mammalian and plant cells provided evidence suggesting that the dual localization is due to the existence of two distinct forms of sEH localized in separate compartments (Mullen et al. 1999).
Recently, sEH has been found to have a broad distribution in human tissues (Enayetallah et al. 2004); however, its subcellular localization in different human tissues has not been evaluated. Immunohistochemistry staining for sEH in human tissues showed diffuse staining patterns suggestive of cytosolic localization in tissues with low abundance or devoid of peroxisomes such as renal distal tubule and pancreatic islet cells. Punctate patterns suggestive of peroxisomal localization were found in cells possessing abundant large-sized peroxisomes such as hepatocytes and renal proximal tubular (RPT) cells (Enayetallah et al. 2004). In this study we tested the hypothesis that human sEH subcellular localization is tissue or cell dependent. Colocalization of sEH with various subcellular compartmentalization markers was evaluated by confocal laser scanning microscopy using human tissue microarray slides.
Materials and Methods
Human Tissues, Antibodies, and Fluorescent Probes
Human tissue samples were obtained from the Cooperative Human Tissue Network, International Bioresearch Solutions (Tucson, AZ), and human tissue microarrays were obtained from Zymed Laboratories (South San Francisco, CA).
For all experiments, 4-μm sections of paraffin-embedded tissues were used. The tissue microarrays provided a high throughput method for evaluation of a large number of tissues and ensured identical experimental conditions and reagent concentrations for all tissues on the same slide.
Polyclonal anti-human sEH rabbit serum (Dr. B. Hammock, University of California, Davis) was used as previously described (Enayetallah et al. 2004). Sheep polyclonal antihuman catalase (Biodesign; Saco, ME) and monoclonal antihuman mitochondrial porin antibody (Molecular Probes; Eugene, OR) were used as peroxisomal and mitochondrial markers, respectively. Propidium iodide was used for counterstaining as well as a nuclear marker. Vectastain Elite ABC secondary antibody avidin-biotin kits were purchased from Vector Laboratories (Burlingame, CA). Streptavidin Alexa Fluor 488 and 647 conjugates (Molecular Probes) were used for fluorescence avidin-biotin detection.
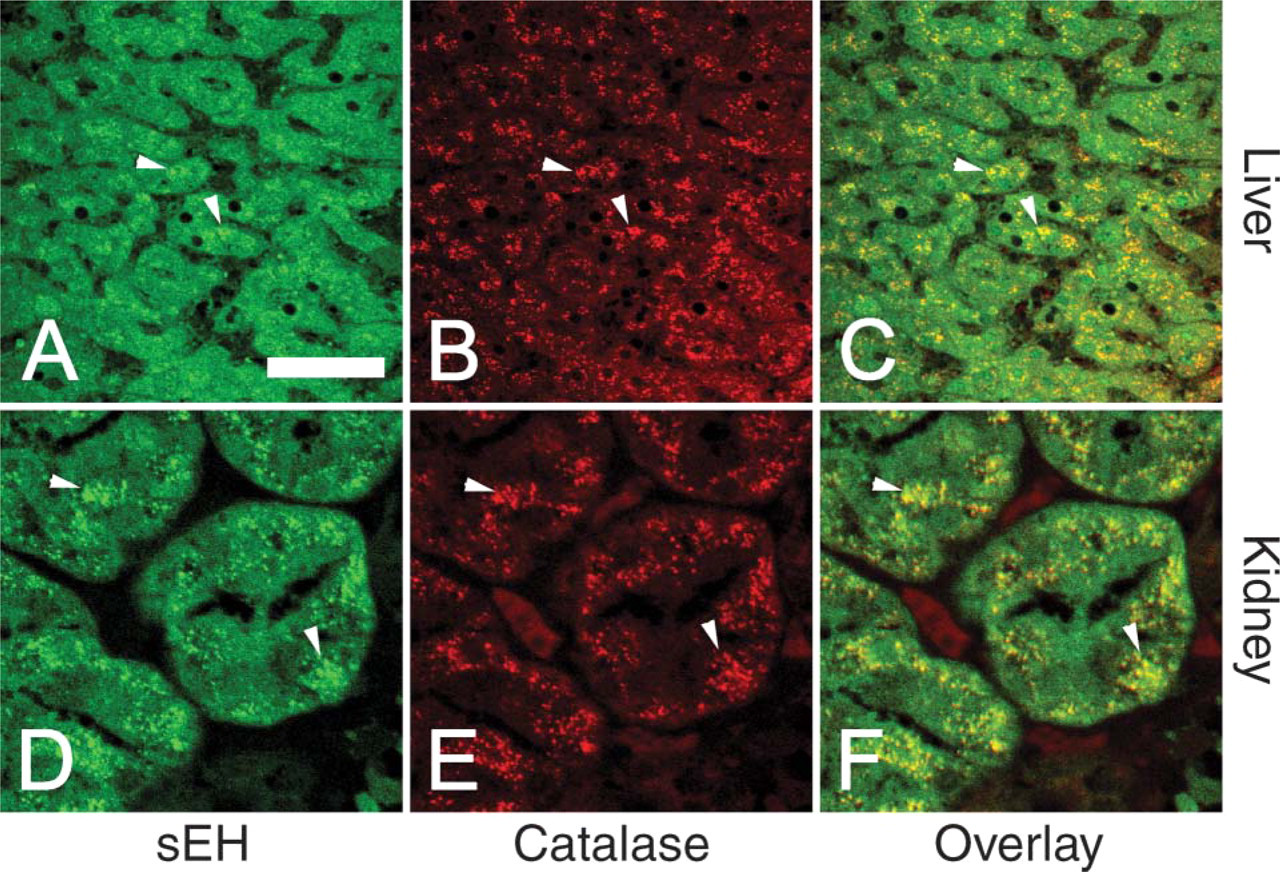
Soluble epoxide hydrolase (green,
Immunofluorescence Labeling
Liver and kidney sections were used as positive controls for all antibodies separately before the dual-labeling studies (Pacifici et al. 1988; Espeel and Van Limbergen 1995; Lustbader et al. 2004). Negative control studies were performed in which the primary antibodies were replaced by preimmune sera or IgGs corresponding to each antibody at the relevant concentration and incubation period. For dual immunofluorescence, no cross labeling was detected in the negative controls where the first or second primary antibodies were replaced by preimmune serum or IgG, confirming the efficiency of the blocking step. The double-immunofluorescence labeling protocol and the concentration of different reagents used in this study were essentially those recommended by the manufacturers. In brief, tissue slides were deparaffinized in ethanol and serial dilutions of xylene, followed by antigen retrieval using Retrieve-All (Signet; Dedham, MA). The slides were then washed in Tris buffer (0.096 M Tris-HCl, 0.029 M Tris-base, 0.347 M NaCl with 0.025% Triton-X 100, pH 7.6) for 10 min. The same buffer was also used for washing between steps. Sections were incubated with 5% normal blocking serum in Tris buffer for 30 min and then with the first primary antibody. Primary antibody dilutions and incubation periods were determined based on titration and optimization experiments. Sections were incubated for 30 min with biotinylated secondary antibody followed by the streptavidin fluor conjugate 488. Avidin-biotin blocking reagent was added (Vector Laboratories), and the above-described steps were repeated using the second primary antibody and streptavidin fluor conjugate 647. Slides were then coverslipped using vectashield fluorescence mounting media (Vector Laboratories). A Leica multi-channel confocal microscope and software (Leica Microsystems; Bannockburn, IL) was used for fluorescence detection.
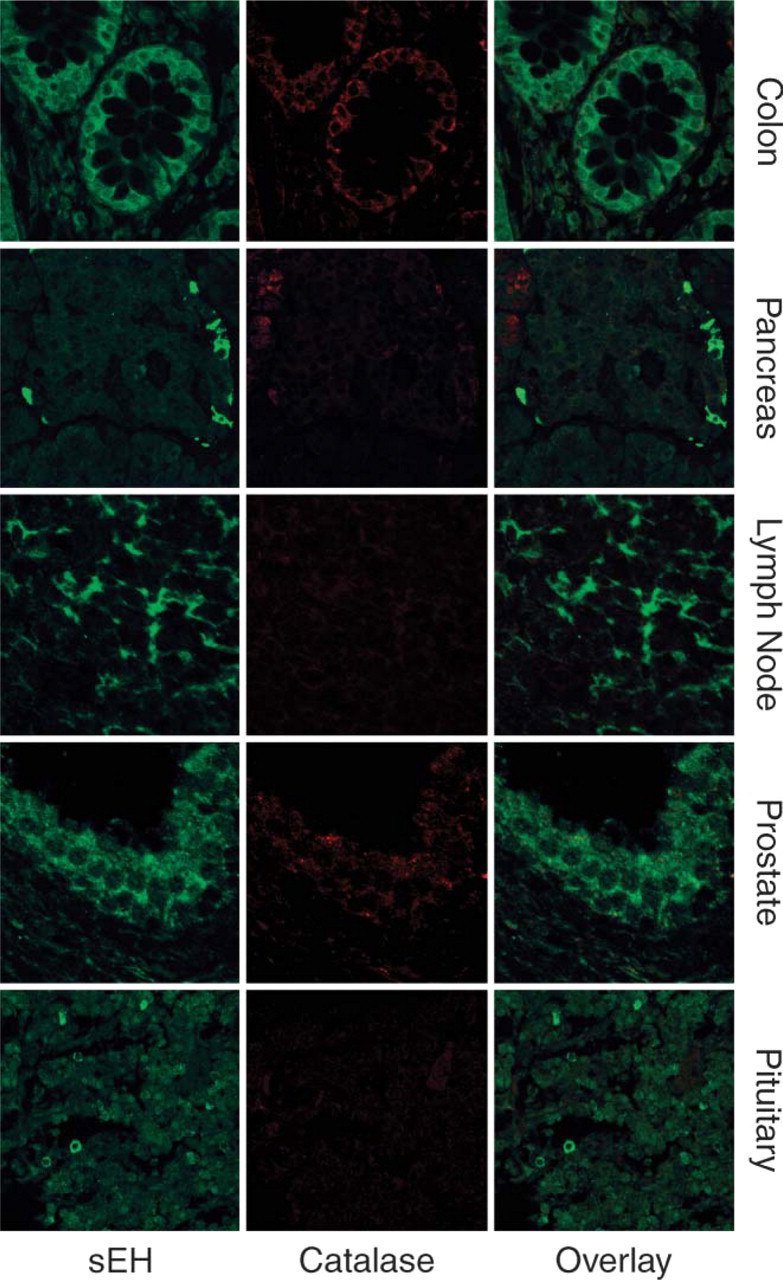
Diffuse cytosolic localization of sEH (green) in cells of the intestinal epithelium, pancreatic islets, germinal centers of lymphoid tissues, prostate ductal epithelium, and pituitocytes. Catalase staining (red) is seen in the intestinal epithelium, prostatic ductal epithelium, and pancreatic ducts but negative staining in the lymphoid germinal center and pituitocytes.
Results
We examined a number of different human tissues using confocal laser scanning microscopy. Composite images of sEH and the peroxisomal marker catalase in hepatocytes and RPTs showed colocalization of the two enzymes in peroxisomes (Figure 1). However, in addition to the prominent peroxisomal component, sEH showed a diffuse distribution consistent with cytosolic subcellular localization as well (Figure 1). Renal distal tubules (not shown) showed only diffuse cytosolic staining for sEH, consistent with the previous finding of distal tubules being devoid of peroxisomes (Litwin et al. 1988).
Other human tissues evaluated in this study include pancreas, lymphoid tissues, anterior pituitary, prostate, intestinal epithelium (Figure 2), lung, and blood vessels (Figure 3), all of which previously expressed sEH (Enayetallah et al. 2004). These tissues showed diffuse cytosolic staining of sEH with no colocalization with the peroxisomal marker catalase. Unlike hepatocytes and renal proximal tubules, these tissues showed no prominent peroxisomes, consistent with previous findings (Krisans 1996). The negativity of peroxisomes for sEH in these tissues was confirmed by sEH/catalase dual staining (Figure 2). This is clearly demonstrated in the intestinal epithelium where numerous small sEH-negative peroxisomes are shown (Figure 2). However, lack of prominent peroxisomes in these tissues was not correlated to the intensity of sEH staining. For example, the intensity of the diffuse cytosolic staining in the cells at the periphery of the pancreatic islets (Figure 2) was significantly higher than that in hepatocytes or RPTs (Figure 1), which have been previously described to possess the highest sEH expression.
The cell-specific distribution of sEH was another intriguing finding, such as the cells at the periphery of the pancreatic islets, the germinal centers of lymphoid follicles, and differential staining intensities of cells in the anterior pituitary gland (Figure 2); however, further identification of these cell populations is required.
Finally, we excluded any detectable mitochondrial and/or nuclear compartmentalization of sEH in any of the tissues evaluated, as represented by nuclear counterstaining (Figure 3) and sEH/porin dual immunofluorescence (Figure 4).
Discussion
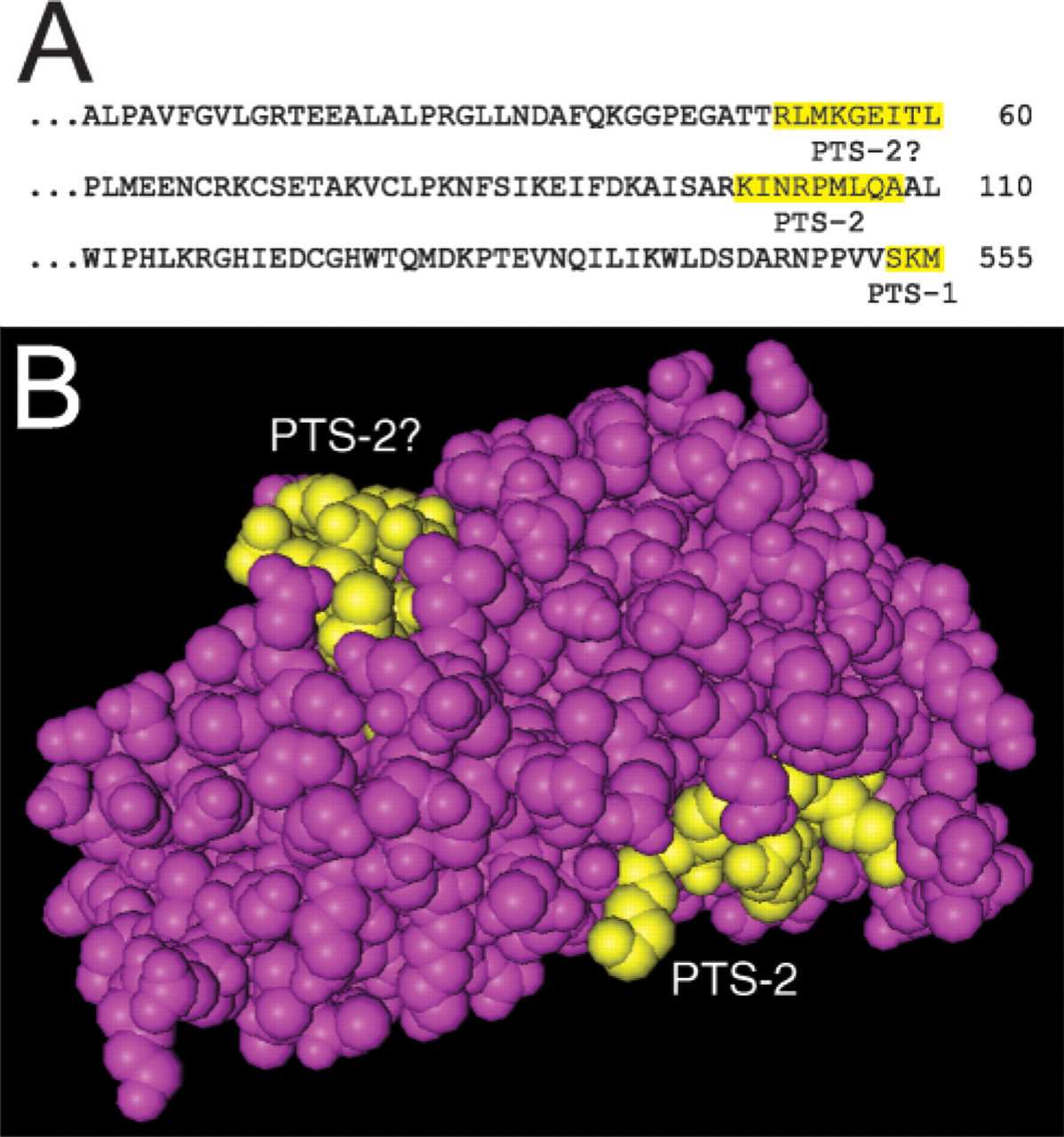
Previously, a tripeptide PTS-1 (Gould et al. 1990; Subramani 1993) was found at the C-terminal end of sEH; SKI in rat (Arand et al. 1991) and mouse sEH (Grant et al. 1993) or SKM in human sEH (Beetham et al. 1993). However, the rodent SKI was described as an impaired targeting sequence as a result of the previous conflicting data showing sEH to be cytosolic and/or peroxisomal (Arand et al. 1991). In agreement with the impaired sequence hypothesis, it has been demonstrated that transiently introduced rat and mouse wild-type sEH-SKI was not targeted to the peroxisomes but rather accumulated exclusively in the cytosol (Mullen et al. 1999). Analysis of the human sEH sequence revealed a potential PTS-2 sequence KI(X5)QA at the N-terminus of the enzyme (Figure 5), which matches the PTS-2 consensus nine amino acid bipartite sequence (R/K)(L/V/I)(X5)(H/Q)(L/A) (Elgersma et al. 1998; Mukai et al. 2002). Another possible PTS-2 was also found, RL(X5)(T?)L; however, this sequence lacks the H/Q consensus motif (Figure 5). The presence of multiple putative peroxisomal targeting sequences in human sEH requires further investigation. However, it is interesting that an enzyme with multiple putative peroxisomal targeting sequences still localizes to the cytosol in all analyzed tissues and cells. This may be suggestive of variable efficiencies of the peroxisomal targeting sequences in different cell types.
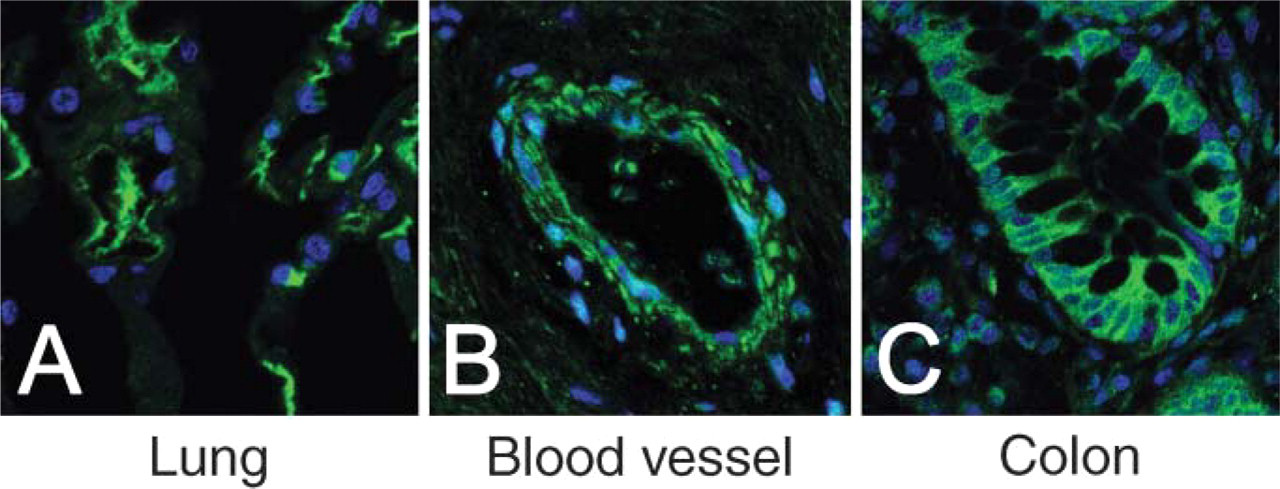
Diffuse cytosolic distribution of sEH (green) with nuclear counterstaining (blue) in pneumocytes (
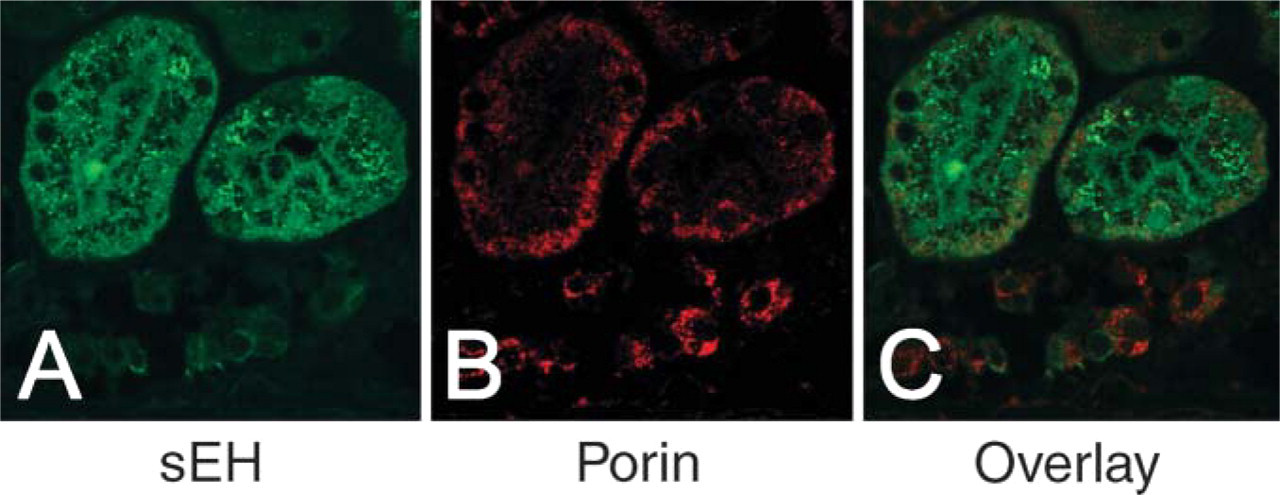
Renal cortex stained for sEH (green,
Zellweger syndrome (ZS) is one of the peroxisomal disorders characterized by absence of intact peroxisomes and consequent cytosolic degradation of peroxisomal proteins (Purdue and Lazarow 1996). Measuring sEH activity in different subcellular fractions showed a 40% reduction of sEH activity in ZS patients as compared with control. This was, however, attributed to loss of peroxisomal EH, whereas cytosolic EH activity in ZS remained similar to that of control (Pahan et al. 1996). This supports the existence of a distinct cytosolic isoform that does not undergo the degradation fate of the peroxisomal isoform in ZS. Results reported in this study may be suggestive of cell-type-specific isoforms that may account for different subcellular localization. This is supported by the recent identification of ovary-specific epoxide hydrolase that matches sEH (Hennebold et al. 2005).
It is not uncommon that limitations of the subcellular fractionation approach may contribute to the discrepancy met with sEH subcellular localization studies (Hollinshead and Meijer 1988). It has been reported that ∼75% of cytosolic NADP-dependent isocitrate dehydrogenase is present in the cytosolic fraction after differential centrifugation (Leighton et al. 1968); however, subsequent immunocytochemical studies clearly demonstrated exclusive peroxisomal localization (Yoshihara et al. 2001). Also, subcellular localization studies of enzymes involved in cholesterol biosynthesis demonstrated exclusive peroxisomal localization of enzymes previously believed to be cytosolic based on the results of fractionation studies (Biardi and Krisans 1996; Krisans 1996). Levels of peroxisomal enzymes involved in cholesterol biosynthesis and cholesterol synthetic capacity were related to the levels of peroxisomes in different cells and tissues (Krisans 1996). However, our results show that sEH is different from other peroxisomal enzymes not only for having dual localization but also because, unlike many peroxisomal enzymes, levels of sEH were not related to levels of peroxisomes in different cells. In fact, some cells with exclusive cytosolic localization showed very high levels of sEH, such as cells at the periphery of the pancreatic islets (Figure 2). Similar to sEH, catalase has a C-terminal PTS-1 (Purdue and Lazarow 1996). Initially, the existence of extraperoxisomal catalase was attributed to methodological artifacts. However, cytochemical analysis confirmed this in sheep, rhesus monkey, and guinea pig liver (Roels 1976; Roels et al. 1977).
The dual functionality of sEH (Cronin et al. 2003; Newman et al. 2003) and the dual localization shown here may support the rationale that the many biological roles of sEH may take place in different subcellular compartments. In addition, sEH has a wide range of substrates that may be suggestive of cell- or compartment-specific substrates.
(
In conclusion, we report that sEH has tissue-dependent subcellular localization, both peroxisomal and cytosolic in hepatocytes and RPTs and exclusively cytosolic in other sEH-containing human tissues evaluated in this study. Interestingly, sEH was cytosolic in all evaluated tissues despite the existence of two putative peroxisomal targeting sequences. Our results may be suggestive of the existence of two distinct isoforms of the enzyme in different tissues, namely, peroxisomal and cytosolic. Further investigation is required to identify the nature of pre- and/or post-translational modification(s) or mechanism(s) involved. Finally, results from this study warrant further investigation of possible tissue-or compartment-specific enzyme functionality.
Footnotes
Acknowledgements
This study was supported by NIH Grants ES-011630 and GM-56708.
We are grateful to Dr. B. Hammock (University of California, Davis) for providing the polyclonal anti-human soluble epoxide hydrolase rabbit serum. We are also grateful to Dr. Michael Arand (Institute of Pharmacology and Toxicology, University of Würzburg, Germany) for providing the rat sEH baculovirus.
