Abstract
Nitric oxide synthase (NOS) is responsible for the biological production of nitric oxide (NO) in several organs, including those of the reproductive tract. We investigated potential changes in NADPH-diaphorase (NADPH-d) activity (marker for NOS activity) and the presence and distribution of NOS in the porcine oviduct. Tissues were obtained from gilts (
N
Materials and Methods
Studies were carried out on 16 sexually mature, crossbred gilts that had exhibited at least two estrous cycles. The animals were kept in separate pens and checked daily for heat in the presence of a boar. The first day of behavioral estrus was established as Day 0 of the estrous cycle. Day of the estrous cycle was additionally confirmed by the morphological appearance of the ovaries after sacrifice (Akins and Morrisette 1968; Leiser et al. 1988). These observations allowed the classification of the oviductal tissues into (a) early proestrus, Days 16-18; (b) late proestrus, Days 18-20; (c) estrus; (d) postovulation, Days 2-4; (e) early luteal stage, Days 6-8; (f) midcycle, Days 8-12; and (g) late luteal stage, Days 13-15. Oviducts were collected immediately after sacrifice. One fallopian tube was used for histo- and immunohistochemistry and the other one for Western blotting analysis.
NADPH-d Staining and Immunocytochemistry for eNOS and iNOS Protein
Three portions of each oviduct (isthmus, ampulla, and infundibulum) were fixed for 6 hr in 4% paraformaldehyde (Fluka Chemie; Buchs, Switzerland) in 0.1 M phosphate buffer (PB). Fixed tissues were stored in 18% sucrose (POCH; Giliwice, Poland) in PB with 0.01% sodium azide (POCH). Cryostat sections (8 μm) were used for immunocytochemistry and histochemistry. To demonstrate NADPH-d activity, cryostat sections of fallopian tube were incubated at 37-38C in a freshly prepared solution of β-NADPH (5 mg/ml; Sigma, St Louis, MO), nitroblue tetrazolium (0.5 mg/ml; Sigma) and Triton X-100 (5 μl/ml; Merck, Darmstadt, Germany) in 0.1 M PB, pH 7.4, for 1 hr. Control sections were exposed to the staining solution without NADPH. All oviduct samples were assayed in parallel and at least three complete sets of samples were examined.
The activity of NADPH-d in porcine oviduct was measured as distribution of formazan deposits (endproduct of the histochemical reaction). The intensity of histochemical reaction in oviduct tissues was estimated by measuring the optical density using the PC-IMAGE system (Foster Findlay Associates; Oxford, UK). All values are reported as mean ±SEM. The data were subjected to one-way analysis of variance (ANOVA; InStat GraphPAD, San Diego, CA).
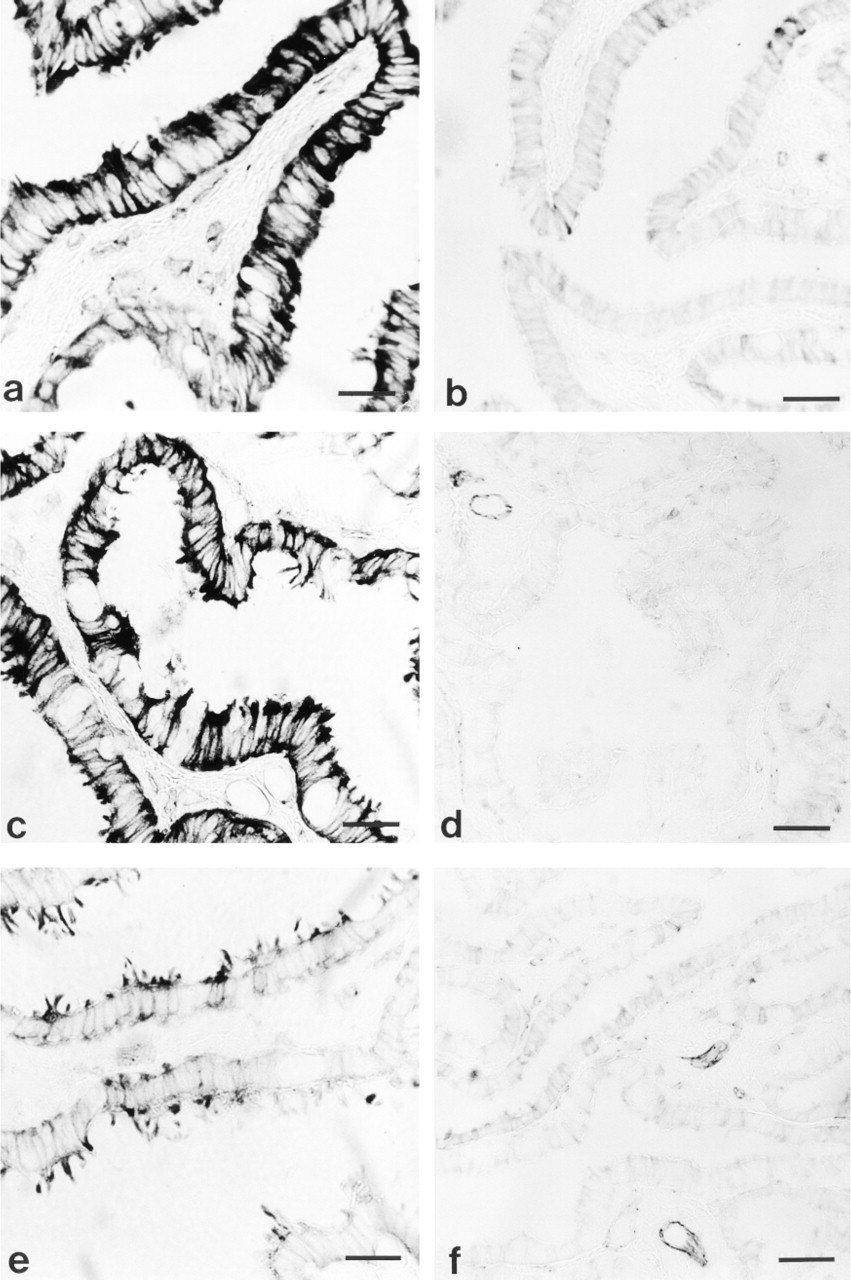
Histochemical reaction for NADPH-d in mucosal epithelium of porcine oviduct. (
For immunocytochemistry, consecutive sections were rinsed in 0.05 mol/liter Tris-hydroxylmethyl aminomethane (TBS; Sigma), then placed in ethanol in ascending concentration series (50%, 70%, 96%, absolute alcohol). The sections were treated with 1% H2O2 in methanol (Sigma-Aldrich; Steinhem, Germany) for 30 min to block endogenous peroxidases and then in 0.75% glycine (Sigma) in TBS for 30 min to block free aldehyde groups. After rinsing in TBS, the sections were incubated overnight with primary antibody against eNOS (diluted 1:50; a mouse monoclonal antibody directed against the amino acids 1030-1209 of human endothelial NO synthase; Transduction Laboratories, Lexington, KY). The consecutive sections were incubated with primary antibody against iNOS (diluted 1:50; a mouse monoclonal antibody directed against the amino acids 961-1144 of mouse macrophage inducible NO synthase; Transduction Laboratories). Antibody binding was detected with the ABC complex (Vectastain ABC kit from Vector Laboratories; Burlingame, CA). Peroxidase activity was revealed using 3,3′-diaminobenzidine (Sigma) as a substrate. Two types of controls were performed: (a) the primary antibody was omitted during the immunostaining procedure; (b) the primary antibody was substituted with nonspecific immunoglobulin G (IgG) during the immunostaining procedure. The observations and photographs were made using a light microscope (Olympus IMT-2; Tokyo, Japan).
NADPH-d staining of porcine myosalpinx. (
Western Blotting Analysis
Tissue samples were placed in ice-cold homogenization buffer [50 mM Tris-HCl, pH 7.4; 10 mM EDTA (Sigma) 150 mM NaCl, 1% Triton X-100 with 1 μM pepstatin A (Sigma) 5 μg/ml leupeptin (Sigma) 5 μg ml aprotinin (Sigma), 100 mM PMSF (Sigma) freshly made] and homogenized on ice. The homogenate was centrifuged at 5000 × g for 5 min at 4C and the pellet was discarded. The supernatant was then centrifuged at 29,000 × g for 30 min at 4C and the sediment suspended in PBS with proteinase inhibitors (concentration as above). The protein content of the resulting suspension was determined (Lowry et al. 1951). Equal amounts of protein (50-100 μg) were dissolved in SDS gel-loading buffer [50 mM Tris-HCl, pH 6.8, 2% SDS (BioRad, Hercules, CA), 10% glycerol (Sigma) 1% β-mercaptoethanol (Sigma) and heated at 95C for 4 min. Protein fractions were loaded on 10% SDS-PAGE and electrophoretically separated for 2 hr at constant current (30 mA). Separated proteins were electrotransferred onto 0.45-μm nitrocellulose membrane for 2 hr at 250 mA in transfer buffer (25 mM Tris-Cl, pH 8.3; 192 mM glycine; 20% methanol A; 0.02% SDS). Then the membrane was washed in TBS-T; (25 mM Tris-HCl, pH 7.5, 137 mM NaCl, and 0.05% Tween-20) and incubated in blocking buffer [1% BSA (Sigma) in TBS-T] for 1 hr at room temperature (RT). The presence of eNOS was confirmed by incubating the membrane in TBS containing eNOS antibody (a mouse monoclonal antibody directed against the amino acids 1030-1209 of human endothelial NO synthase; Transduction Laboratories) at a dilution of 1:2500 overnight at 4C. After incubation, the nitrocellulose membrane was washed three times for 10 min in TBS-T at RT. The washed blot was reincubated for 2 hr at RT with a 1:3000 dilution of biotinylated antimouse IgG (Vectastain ABC kit) washed three times in TBS for 10 min at RT, and incubated for 1 hr at RT with 1:3000 dilution of avdin-biotin-HRPO complex (Vectastain ABC kit). To develop color, TBS containing 0.01% H2O2 and 0.04% 3,3′-diaminobenzidine was used.
Results
NADPH-d Histochemistry
Porcine fallopian tube showed NADPH-d activity in mucosal epithelium, myosalpinx, nerve fibers, and endothelium of blood and lymph vessels in all regions of oviduct during the entire estrous cycle (Figures 1 and 2). However, the intensity of the histochemical reaction was different among regions of oviduct and stages of the estrous cycle. In general, NADPH-d activity was much higher in mucosal epithelium than in myosalpinx (Figures 1–4). The highest activity of NADPH-d was consistently found in the epithelium of the isthmus (Figures 1a and 3). The intensity of reaction (arbitrary units ± SEM) in this part of oviduct increased from postovulation (62.6 ± 5.8) till early proestrus (96.2 ± 11.2; Figures 1a and 3;
The most intense NADPH-d activity in myosalpinx of all parts of the oviduct was observed at the postovulatory stage of the estrous cycle (isthmus, 38.3 ± 2.5; ampulla, 35.6 ± 4.2; infundibulum, 24.7 ± 0.8; Figures 2a, 2b and 4) compared to estrus (isthmus, 12.4 ± 1.8;
The control specimens, in which β-NADPH was omitted during the histochemical procedure, showed no NADPH-d activity (Figure 2d).
NOS Immunohistochemistry
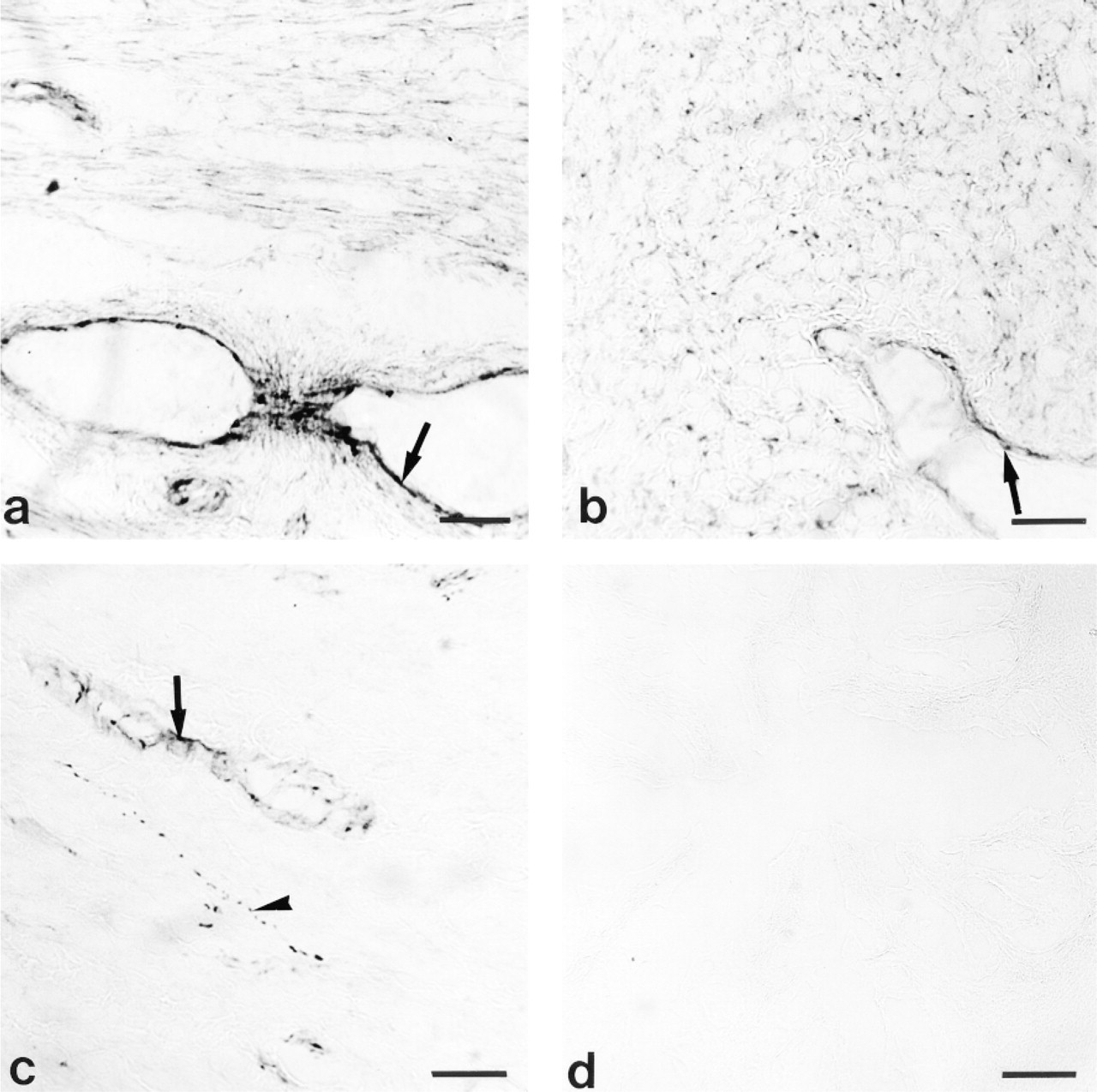
Figure 5 shows that the gilt oviduct was immunostained for the NOS. eNOS-like immunoreactivity was identified in epithelial cells of mucosa, endothelium of vascular tissues, and myosalpinx at all stages of the cycle (Figures 5a-5c). The positive reaction for the inducible calcium-independent isoform of NOS (iNOS) was restricted to the endothelium of lymph vessels and some blood vessels in both the mesosalpinx and the oviduct (Figures 5d and 5e, respectively).
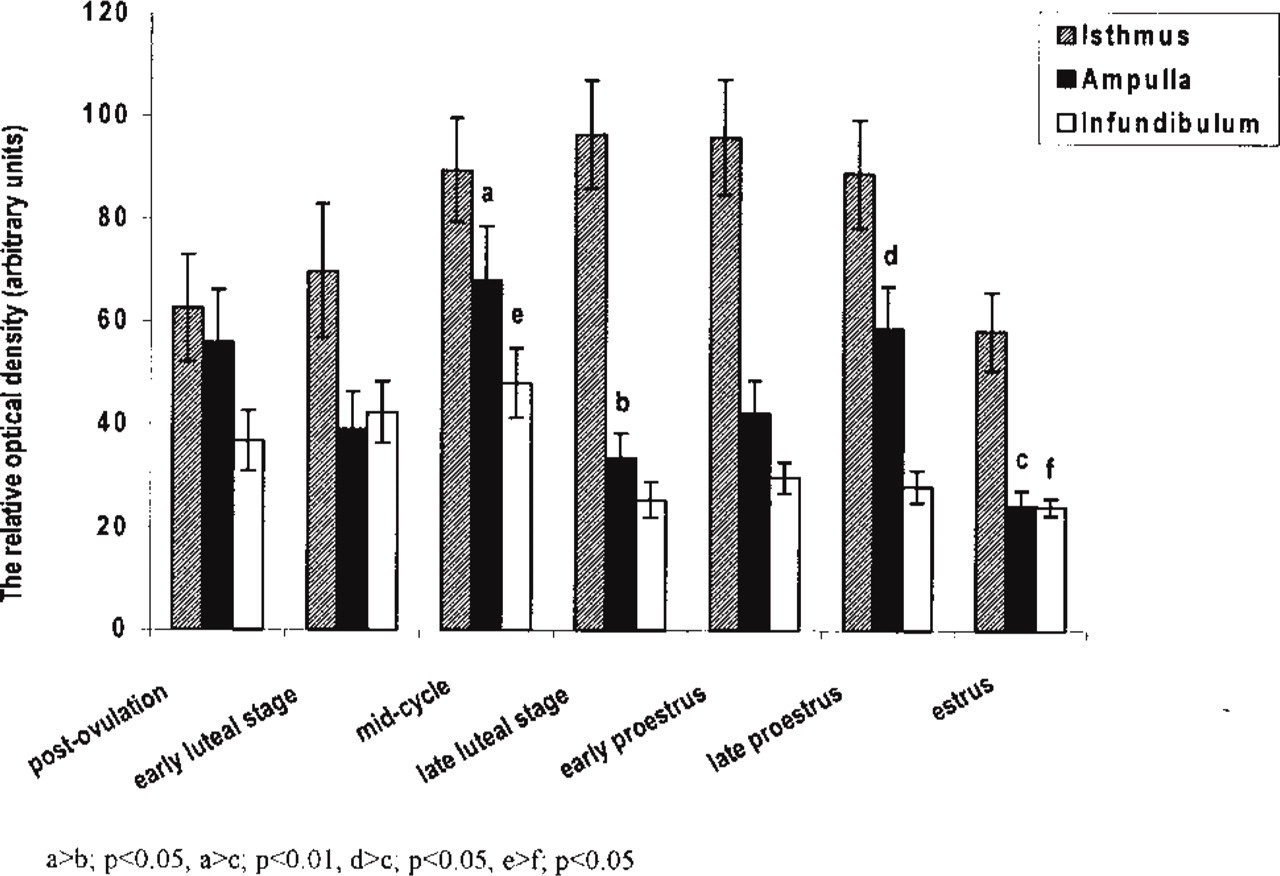
Quantitative analysis of NADPH-d activity in the mucosal epithelium of porcine oviduct. Columns represent the mean ± SEM of samples from separate experiments (
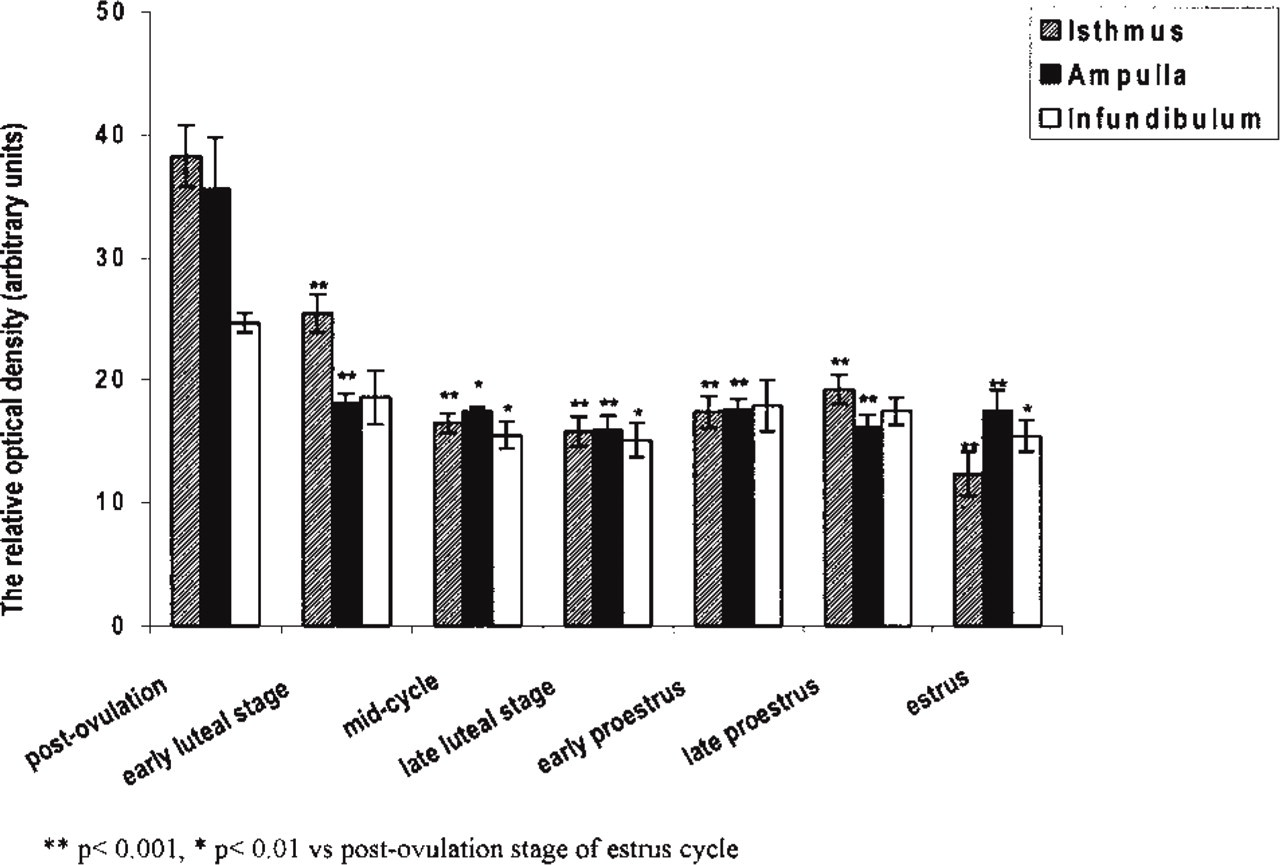
Quantitative analysis of NADPH-d activity in the myosalpinx of porcine oviduct. Columns represent the mean ± SEM of samples from separate experiments (
This immunostaining for eNOS and iNOS (data not shown) was absent when the receptor antibody was omitted or substituted with nonspecific immunoglobulin G (IgG) during the immunostaining procedure (Figure 5f).
Western Blotting Analysis
Figure 6 is a representative Western blot showing the presence of an eNOS-specific band in porcine oviduct. This band, with a molecular weight of about 135 kD, corresponds to endothelial NOS from human endothelial cell lysate (Figure 6, lane 2). In parallel immunoblots, we probed with a specific iNOS monoclonal antibody and iNOS was not detected in porcine fallopian tubes (not shown).
Discussion
This study demonstrates both the presence of NADPH-d activity in porcine oviduct and its fluctuation during the course of the estrous cycle. Furthermore, we documented the expression of NOS and assessed the molecular weight of eNOS in porcine fallopian tube.
NADPH-d co-localizes with all known NOS isoforms (Tracey et al. 1993; Sessa 1994; Bryant et al. 1995; Rosselli et al. 1996; Tschugguel et al. 1998). However, Tracey et al. (1993) suggested that NOS represents only a fraction of total cellular NADPH-d activity and that these activities are not always co-localized. Non-NOS NADPH-d activity can be removed by formaldehyde fixation because the process does not affect the NOS-related NADPH-d activity (Nakos and Gossrau 1994; Moorhead et al. 1995). Therefore, in our study we used paraformaldehyde-fixed tissues to ensure that NADPH-d activity coincided with NOS activity.
We observed the NADPH-d activity in porcine oviduct epithelium of mucous, myosalpinx, nerve fibers, and endothelium of blood and lymph vessels. The most intense staining was observed in the epithelium lining the porcine oviduct. The intensity of staining was variable, with the lowest intensity of reaction seen at estrus. This finding is in agreement with results obtained in rat oviduct (Bryant et al. 1995). NADPH-d staining was less dense in fallopian tube obtained from rats in late estrus and estrus than in samples taken at other stages of the estrous cycle. On the other hand, Tschugguel et al. (1998) observed only weak staining in the epithelial lining of the human oviduct. Rosselli et al. (1996) demonstrated that cultured bovine and human oviduct epithelial cells possessed areas of intense staining for NADPH-d and that this staining was not homogeneous. They suggested that NOS enzyme activity may be dependent on the differentiation of the cell.
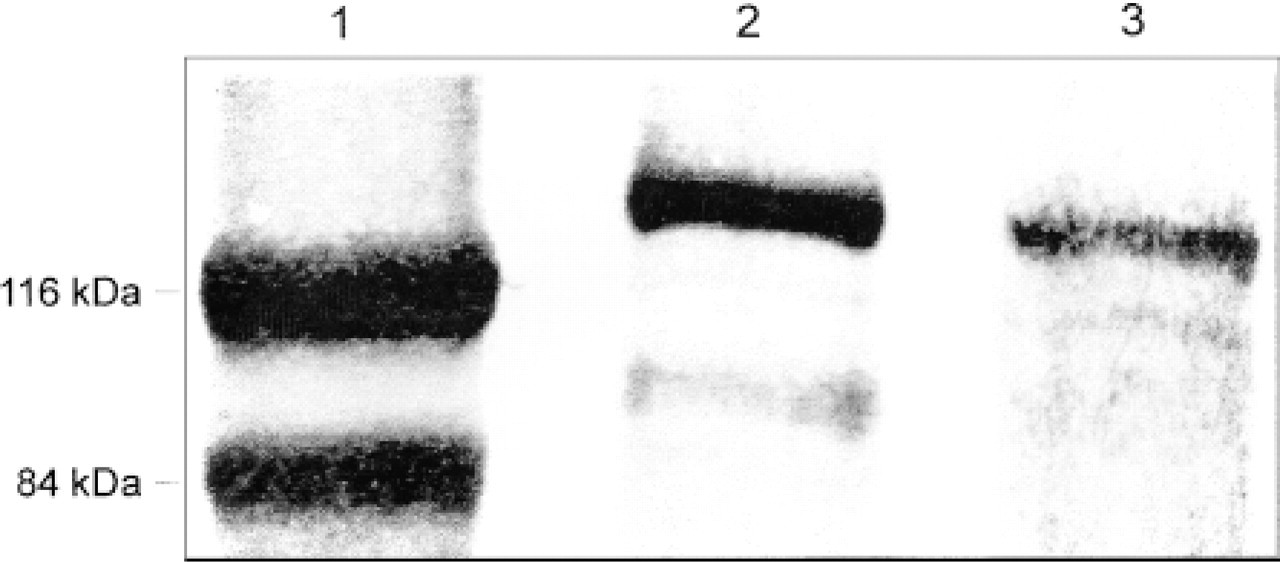
Representative Western blot of eNOS protein in porcine oviduct (Lane 3). Lane 1, marker of protein molecular weight; Lane 2, positive control in human endothelial cell lysate.
The results of our study indicate that the muscle layer of the porcine oviduct was almost clear of NADPH-d staining but that samples taken at the postovulatory stage of the estrous cycle showed marked blue formazan reaction product, mainly in the isthmic region. Bryant et al. (1995), examining rat oviduct, observed only light NADPH-d staining in the smooth muscle. It is interesting that porcine myosalpinx showed NADPH-d activity only at the postovulatory stage of the estrous cycle. Salvemini et al. (1993) showed that NO plays a critical role in the release of PGE2 by direct activation of cyclo-oxygenase. Prostaglandin E2 is a well-known factor that becomes increased at the postovulatory stage of the estrous cycle (Wijayagunawardane et al. 1998) and is responsible for relaxation of the oviduct in the presence of progesterone (Hunter 1988). Because progesterone can be locally concentrated in blood, reaching the oviduct immediately after ovulation (Pharazyn et al. 1991), it is possible that NO (apart from its own direct influence on oviduct; Ekerhovd et al. 1997) activates cyclo-oxygenase enzymes to increase the production of PGE2. The increase of NADPH-d activity in myosalpinx exclusively at the postovulatory stage of the estrous cycle can be connected with relaxation of the oviduct for passage of embryos to the uterus after fertilization.
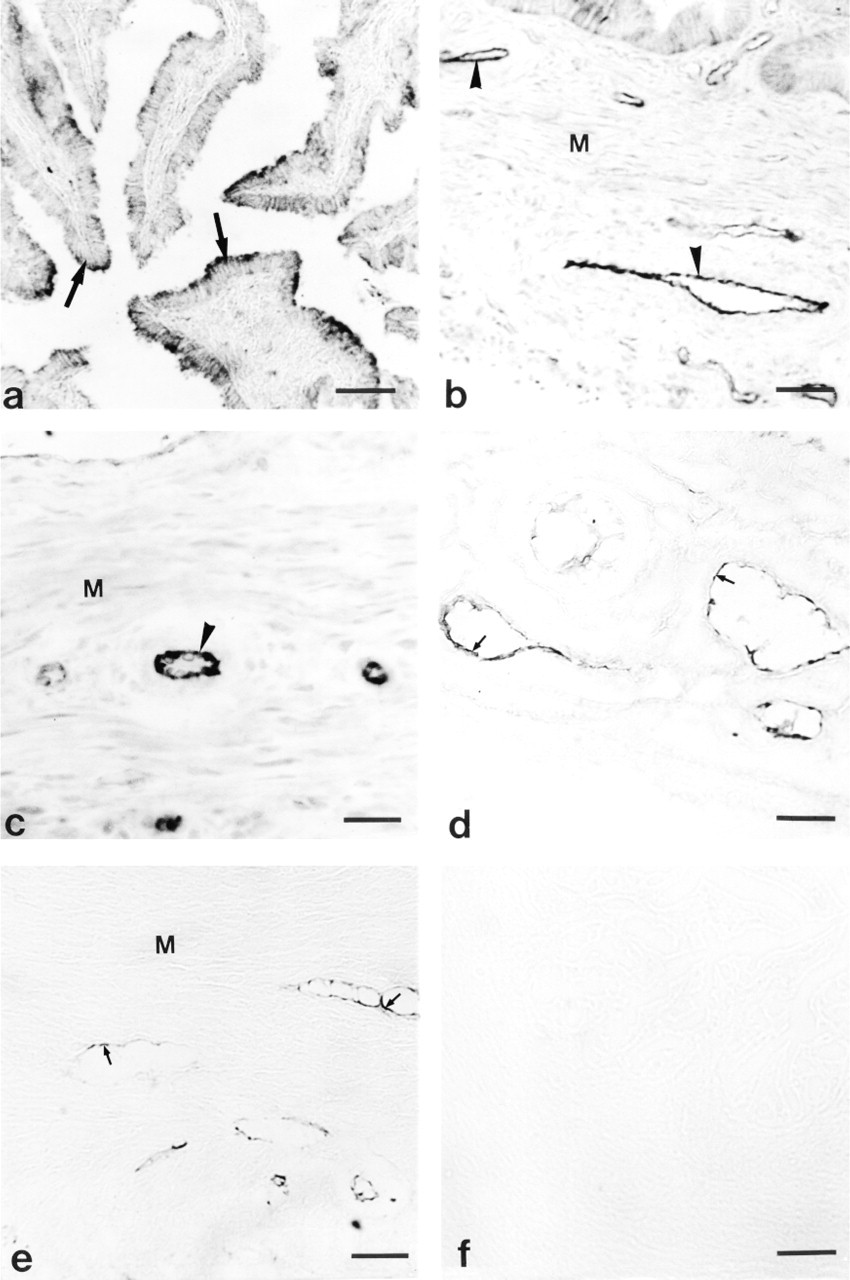
Immunohistochemical staining for NOS in porcine oviduct. Positive reaction for eNOS is shown in
Our immocytochemical study revealed that eNOS is the main isoform of NOS expressed in porcine oviduct. Immunolabeling for eNOS was found in epithelial cells of mucosa, endothelium of vascular tissues, and myosalpinx. Chatterjee et al. (1996) observed immunolabeling for eNOS in mucosal epithelium and muscule wall of rat oviduct. They suggested that maximal immunostaining for eNOS during proestrus and estrus may indicate the role of NO in quiescence for reception, retention, and fertilization of ovulated oocytes. In contrast, Bryant et al. (1995) did not observe apparent differences in the density of NOS labeling in rat oviduct at different stages of the estrous cycle. Rosselli et al. (1996) demonstrated positive staining for eNOS in human and bovine cultured epithelial cells.
The Western blotting analysis confirmed our immunocytochemical results. In homogenates of porcine oviduct, we detected a protein of approximately 135 kD which corresponded to the expected molecular weight of eNOS (Sessa 1994).
In contrast to eNOS, iNOS immunoreactivity was confined to endothelium of lymphatics and some blood vessels. These data are at variance with the results of others. Ekerhovd et al. (1997) observed positive staining for iNOS in smooth muscle, epithelium, vascular endothelium, and connective tissues of human oviduct. Labeling for iNOS in rat oviduct was restricted to the epithelial cell lining (Bryant et al. 1995; Martinez et al. 1997). The differences between those data and our results may result from species differences or may be attributable to low antigenicity of the primary antibody, which could not detect low concentration of iNOS in porcine oviduct.
Taking into account the method of fixation, the lack of the expected band using Western blotting analysis for iNOS, and single nNOS-immunoreactive nerve fibers running among smooth myocytes of the porcine oviductal isthmus (Majewski et al. 1995) it appears likely that observed NADPH-d activity coincides with eNOS activity. eNOS is commonly described as a constitutive isoform of NOS. Our results demonstrate that, in porcine oviduct, changes in NADPH-d/eNOS activity can be associated with hormonal changes during the estrous cycle.
In summary, we conclude that the different activity of NADPH-d in various regions of the oviduct, accompanied by changes of its activity during the course of the estrous cycle, can indicate a considerable role of NO in regulation of tubal function. The increase in NOS activity in myosalpinx of the isthmus up to Day 4 of the estrous cycle can inhibit motility of this part of oviduct, facilitating passage of embryos into the uterus after fertilization.
