Abstract
Malignant round cell neoplasia was identified in 12 llamas and 12 alpacas aged 0–23 years. Mean age of affected alpacas (3.1 years) was significantly less than that of affected llamas (8.0 years). Tumor cell morphology varied from large and often pleomorphic (11 tumors) to small and often homogeneous (13 tumors). Neoplastic lesions were multicentric in 12 cases. Other sites were gastric (5 cases), intra-abdominal (perirenal; 4 cases), intrathoracic (2 cases), and cervical (1 case). Immunohistochemistry with antibodies to CD79α, BLA36, and CD3 identified B-cell lymphoma (12 cases) and T-cell lymphoma (6 cases). Six tumors did not express any lymphoid marker and were further immunostained for neuron-specific enolase (NSE), synaptophysin, S-100, glial fibrillary acidic protein (GFAP), and chromogranin A. All 6 of these tumors were negative for GFAP and chromogranin A but expressed 1 or more of the neural markers NSE, synaptophysin, and S-100 and were classified as primitive malignant round cell tumors (PMRCT). Tumor types could not be distinguished on the basis of animal age, gross pathologic appearance, tumor morphology, or tumor location. All animals with lymphoma and 5 with PMRCT died or were euthanatized. One alpaca with a focal cervical PMRCT lived for at least 20 months after diagnosis. Results of this study indicate that malignant round cell tumors in llamas and alpacas are a heterogeneous group that cannot be distinguished on the basis of signalment, postmortem findings, or routine light microscopic findings. Immunohistochemistry is a valuable diagnostic procedure when evaluating malignant round cell neoplasia in llamas and alpacas.
Keywords
Introduction
Malignant round cell tumors in people are recognized to be a heterogeneous group of tumors, often referred to as small round blue cell tumors. 12, 20– 22, 30 The category of small round blue cell tumor includes lymphoma, primitive neuroectodermal tumor/Ewing's sarcoma, primitive neuroblastic tumors, rhabdomyosarcoma, and nephroblastoma. 12, 20– 22, 30 Malignant round cell tumors can arise in the central nervous system 12, 21 or in peripheral soft tissue, bone, or viscera. 12, 16, 20, 21 These tumors are particularly common in children and adolescents. 12, 21, 22, 30 Diagnosis is often challenging, and in many cases, tumor type cannot be accurately determined on routine histopathologic sections but is revealed by immunohistochemistry. 12, 21, 22, 30
Lymphoma is the most common malignant round cell tumor recognized in veterinary medicine. 14 There are occasional reports of peripheral primitive neuroblastic and primitive neuroectodermal tumors in animals. 2, 3, 5, 9, 17– 19, 24, 25 These tumors are most commonly reported in young dogs 2, 5, 17– 19, 24, 25 but have also been reported in ruminants. 3, 9 Malignant round cell neoplasia diagnosed as lymphoma is the most frequently reported neoplasm in camelids, but most reports are of a single case or a small series of cases.∗ Lymphoma was the most common malignant neoplasm affecting camelids in 2 of 3 survey studies 1, 28 and was the second most common malignant neoplasm in a third study. 32 Tumors often occurred in young animals. Of 23 reported cases of malignant round cell neoplasia in camelids, 14 were 2 years of age or younger, 1, 10, 13, 23, 26, 28 and 10 were less than 1 year of age. 1, 10, 26, 28
Diagnosis of lymphoma in most camelids relied on gross pathologic and routine light microscopic evaluation. Results of immunophenotyping are described in only 7 cases. 10, 23, 26 An immunohistochemical study of 4 alpacas with malignant round cell tumors found that 3 cases were B-cell lymphoma. One did not express lymphocyte markers but expressed the neural markers neuron-specific enolase (NSE) and synaptophysin, consistent with a primitive neural tumor. 26 Signalment, clinical findings, and gross pathologic findings were indistinguishable in these 4 cases, indicating that, as in people, malignant round cell tumors in camelids are a heterogeneous population and that immunohistochemistry is important for diagnosis.
The purpose of this retrospective study was to characterize pathologic and immunohistochemical findings in malignant round cell tumors diagnosed in camelids at Oregon State University.
Materials and Methods
Review of the Oregon State Veterinary Diagnostic Laboratory database from 1991 to 2008 revealed 24 llamas and alpacas diagnosed with malignant round cell neoplasia on the basis of routine histopathologic evaluation. Five cases were reported as lymphoma in a previous study on the basis of routine light microscopy. 32
Medical records of all camelids were examined for signalment and pathologic findings. Postmortem examination of 16 animals had been performed at the laboratory, samples were submitted from veterinarians performing field postmortem examinations in 7 cases, and 1 case was diagnosed from biopsy material obtained at the Oregon State University Veterinary Teaching Hospital.
Hematoxylin and eosin (HE)–stained tissue sections from all animals were examined. Tumor cell morphology was subjectively classified as large or small and pleomorphic or homogeneous, and mitotic indices were determined. Tumors were also examined for the presence or absence of a packeted pattern of tumor cells. Serial sections of formalin-fixed, paraffin-embedded tissues from all animals were prepared for immunostaining with a high-temperature antigen retrieval method with the use of target retrieval solution (Dako, Carpinteria, CA). Immunostaining was performed with an autostainer (Dako Autostainer Universal Staining System) with Nova Red chromogen (Vector Laboratories, Burlingame, CA) and Mayer's hematoxylin counterstain (Sigma, St. Louis, MO). The primary antibodies used were polyclonal rabbit anti-human CD3 (Dako, diluted 1 : 100), monoclonal mouse anti-human CD79α (Dako, diluted 1 : 100), and monoclonal mouse anti-human BLA36 (Dako, diluted 1 : 25). Normal camelid lymph node served as a positive control, and tumor sections incubated with non–immune serum served as negative controls. On the basis of the results, tumors were categorized as B-cell lymphoma, T-cell lymphoma, or indeterminate (negative for all 3 antigens). All indeterminate cases were further stained with argentaffin (Fontana-Masson) and argyrophil (Grimelius and Churkian-Schenk) silver stains and were further immunostained with the use of monoclonal mouse anti–human NSE (Dako, diluted 1 : 400), polyclonal rabbit anti-synaptophysin (Dako, diluted 1 : 200), polyclonal rabbit anti–glial fibrillary acidic protein (GFAP; Dako, diluted 1 : 500), polyclonal rabbit anti–S-100 (Dako, diluted 1 : 400), and polyclonal rabbit anti–chromogranin A (Dako, diluted 1 : 1,000). All procedures included high-temperature antigen retrieval with target retrieval solution (Dako) except for digestion with proteinase K (Dako) before incubation with GFAP antibodies, and high-temperature antigen retrieval with distilled water for NSE preparations. Positive control tissues were camelid brain (NSE, S-100, GFAP) and pancreatic islets and adrenal medulla (synaptophysin, chromogranin A). Negative controls were sections incubated with non–immune serum. Two indeterminate cases in which biopsy material was available were also stained with periodic acid–Schiff stain for glycogen.
Age of affected llamas and alpacas had a normal distribution (Kolmogorov–Smirnov test with Lilliefors correction). Mean age of llamas and alpacas, and of animals with different tumor types and locations, was compared by Student's t-test. Significance level was set at P < .05.
Results
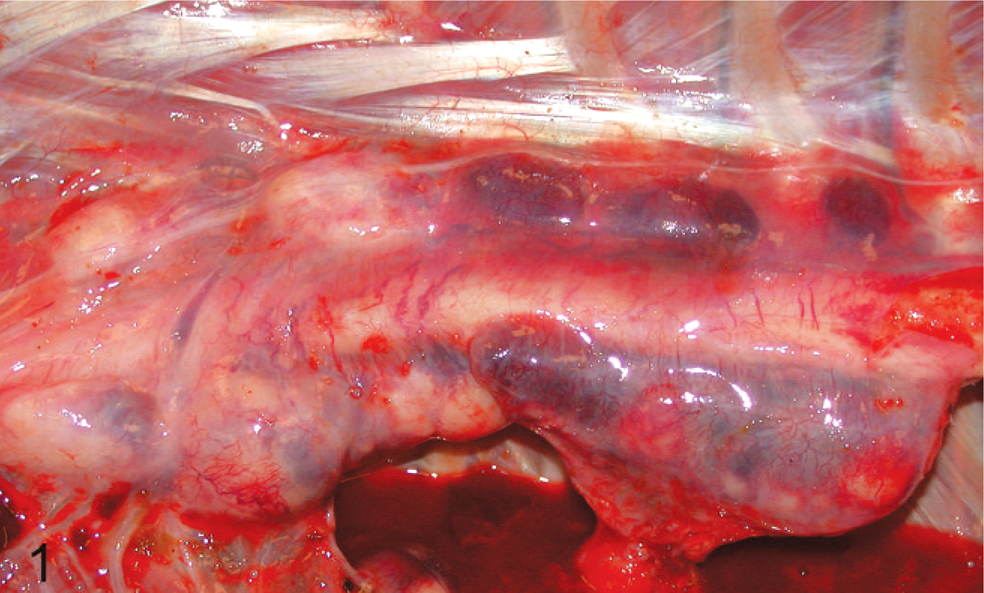
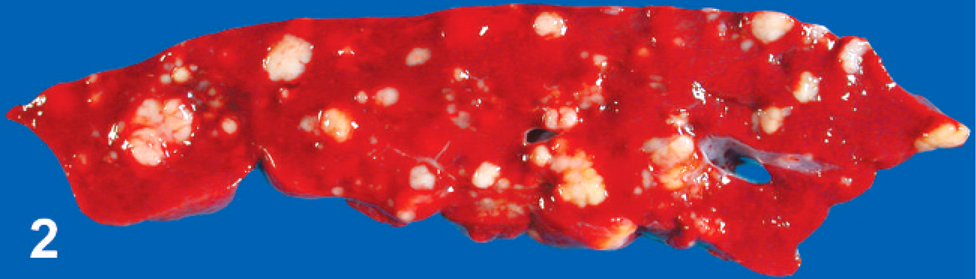
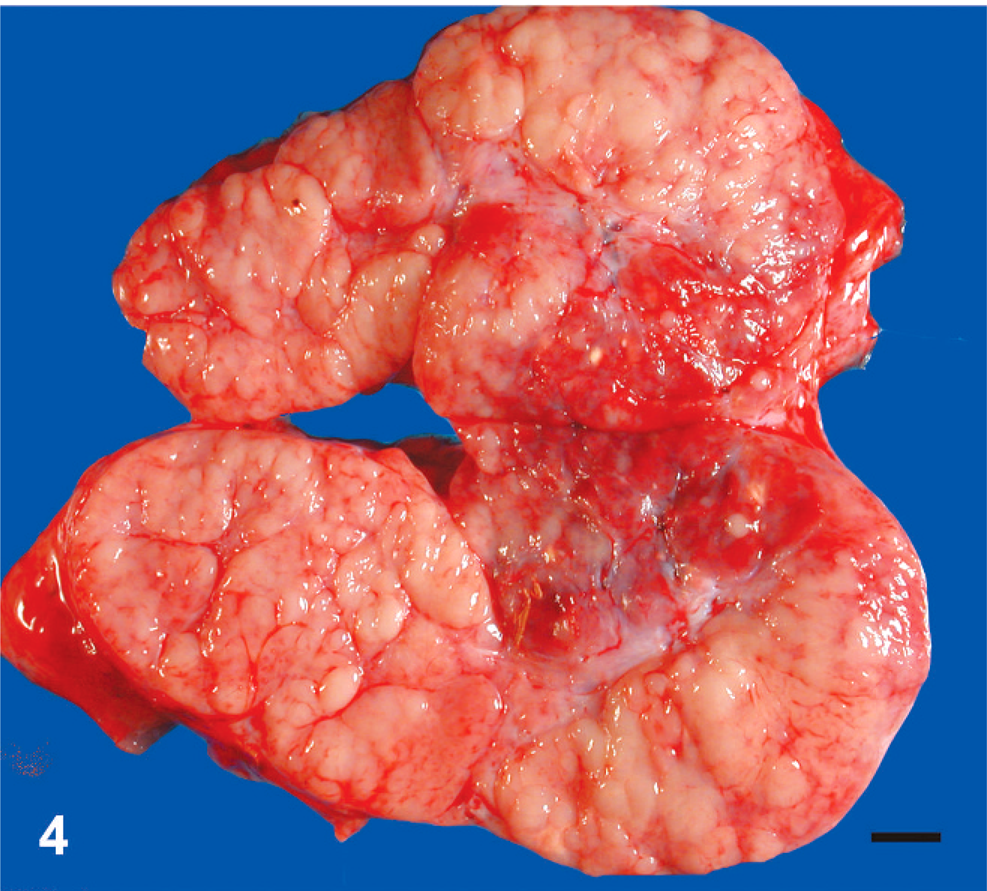
Malignant round cell tumors were identified in 12 llamas and 12 alpacas (Tables 1 and 2). Age ranged from fetal (late-stage gestation) to 23 years. No gender bias was detected. Body condition was described as thin to emaciated in 11 animals (Nos. 2, 4, 8, 12, 14, 16, 18, 19, 21, 22, 24), good in 7 animals (Nos. 3, 6, 7, 9, 10, 13, 17), and was not described in 6 animals. Tumors were soft, white to pale tan, solid and fleshy, and occasionally hemorrhagic or necrotic; formed masses within the abdomen or thorax (Fig. 1) and multifocal to locally extensive masses within visceral organs (Figs. 2, 3); and showed marked enlargement and effacement of architecture of affected lymph nodes (Fig. 4).
Cases of malignant round cell neoplasia in 24 llamas and alpacas diagnosed at Oregon State University from 1991 to 2008.*
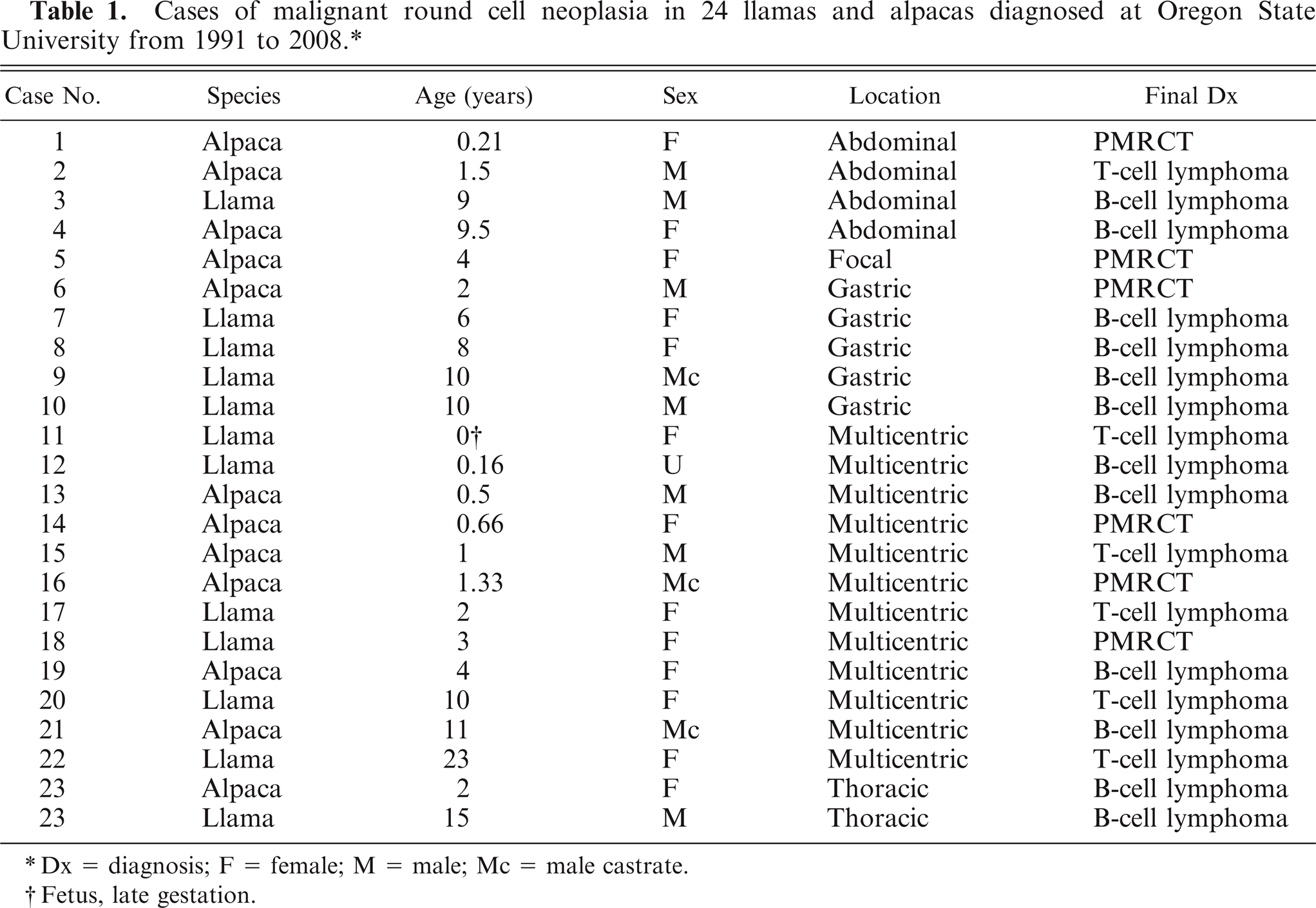
Dx = diagnosis; F = female; M = male; Mc = male castrate.
Fetus, late gestation.
Summary of tumor types, species, age, and gender of 24 llamas and alpacas with malignant round cell tumors.
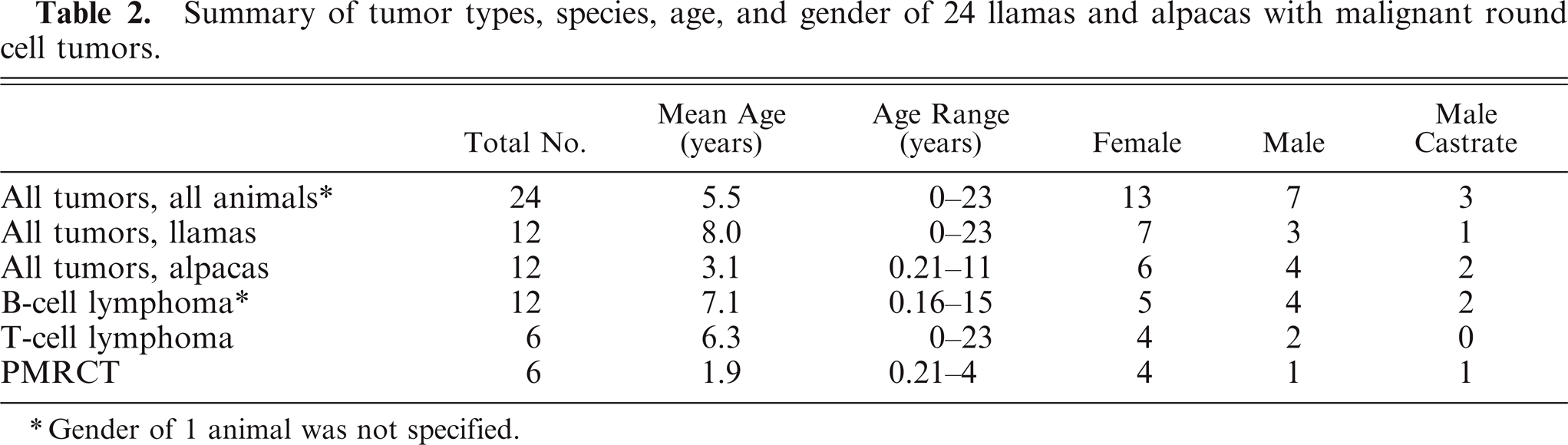
Gender of 1 animal was not specified.
Thorax; alpaca No. 14. Eight-month-old alpaca with multicentric primitive malignant round cell neoplasia. Multiple pale tan to hemorrhagic nodular masses surround the aorta and are adherent to the dorsal body wall forming paravertebral masses.
Liver; alpaca No. 14. Eight-month-old alpaca with multicentric primitive malignant round cell neoplasia. Multiple irregularly sized and shaped, pale tan, and slightly firm nodules are present throughout the parenchyma.
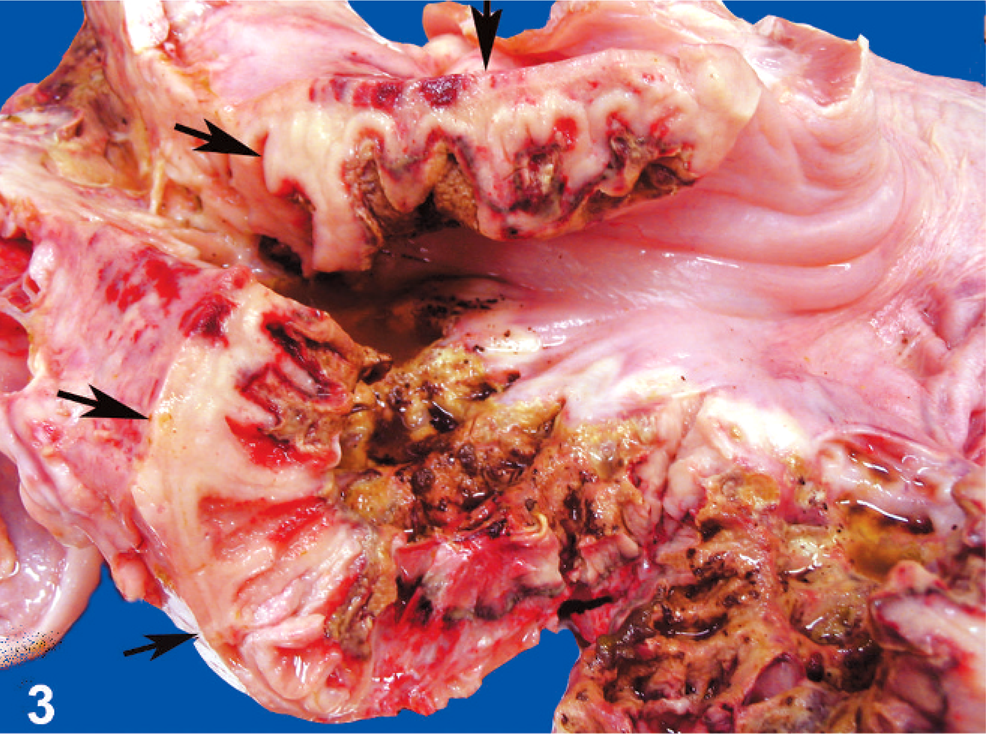
Stomach; llama No. 10. Ten-year-old llama with gastric B-cell lymphoma. The wall is markedly thickened because of transmural tumor cell infiltration (arrows). The mucosa is extensively ulcerated.
Peripheral lymph node; alpaca No. 14. Eight-month-old alpaca with multicentric primitive malignant round cell neoplasia. The node is markedly enlarged, and nodal architecture is effaced by multinodular tumor masses. Bar = 1 cm.
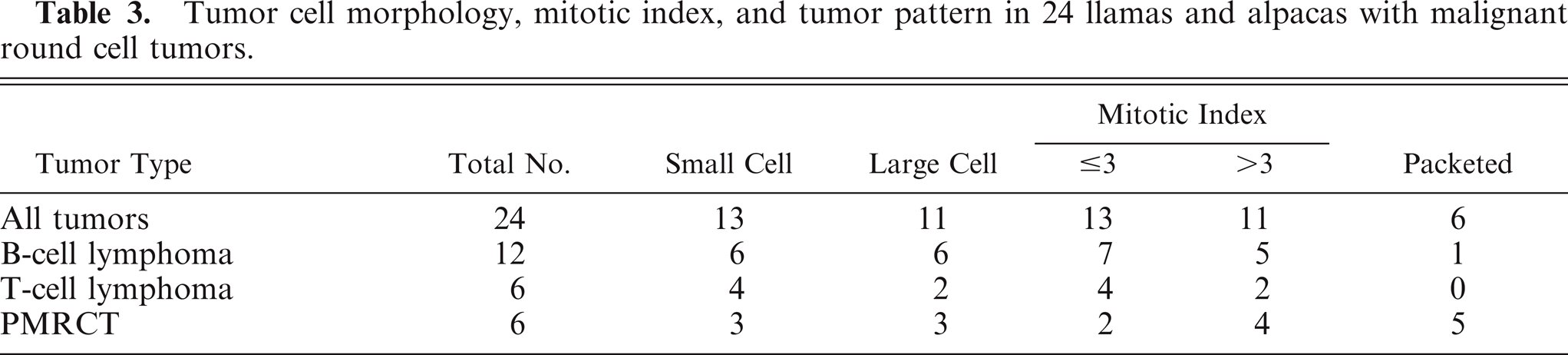
Tumor cell morphology, mitotic index, and pattern are summarized in Table 3, and tumor cell morphology is illustrated in Figs. 5–10. Cells were determined to be large in 11 tumors (Figs. 5, 6) and small in 13 tumors (Fig. 7) and were either entirely round (23 tumors) (Figs. 5–8) or also contained ovoid to slightly elongate cells (1 tumor; Fig. 9). Nuclear morphology varied from small and heterochromatic (Fig. 7) to large and euchromatic (Figs. 5, 6), from round (Figs. 6–8) to irregular (Figs. 5, 9, 10), and from central (Figs. 5–8) to eccentric (Figs. 9, 10). Tumor cell cytoplasm was either indistinct (Figs. 5–7) or eosinophilic (Figs. 5, 9, 10). Large-cell tumors often exhibited prominent cellular pleomorphism (Fig. 5), whereas small-cell tumors were typically composed of relatively homogeneous cells (Fig. 7). Vague packeting within fine reticular stroma was detected in 6 tumors (Fig. 8).
Tumor cell morphology, mitotic index, and tumor pattern in 24 llamas and alpacas with malignant round cell tumors.
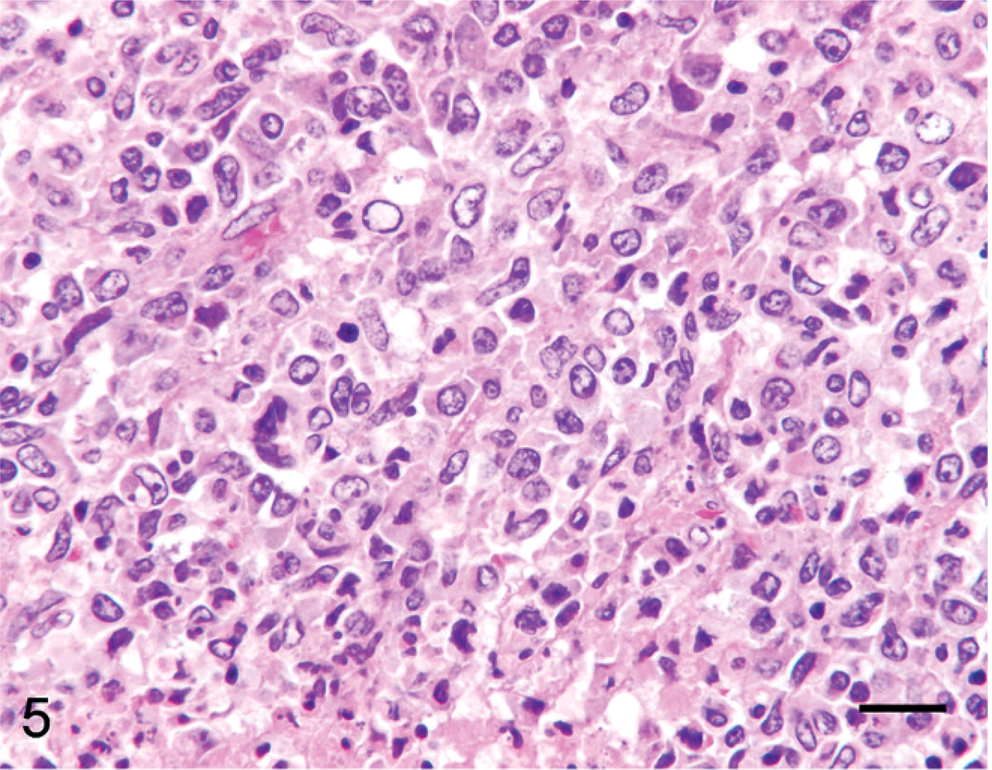
Lymph node; alpaca No. 19. Four-year-old alpaca with multicentric B-cell lymphoma. Cells are large and anaplastic with euchromatic nuclei and a variable amount of eosinophilic cytoplasm. HE. Bar = 50 µm.
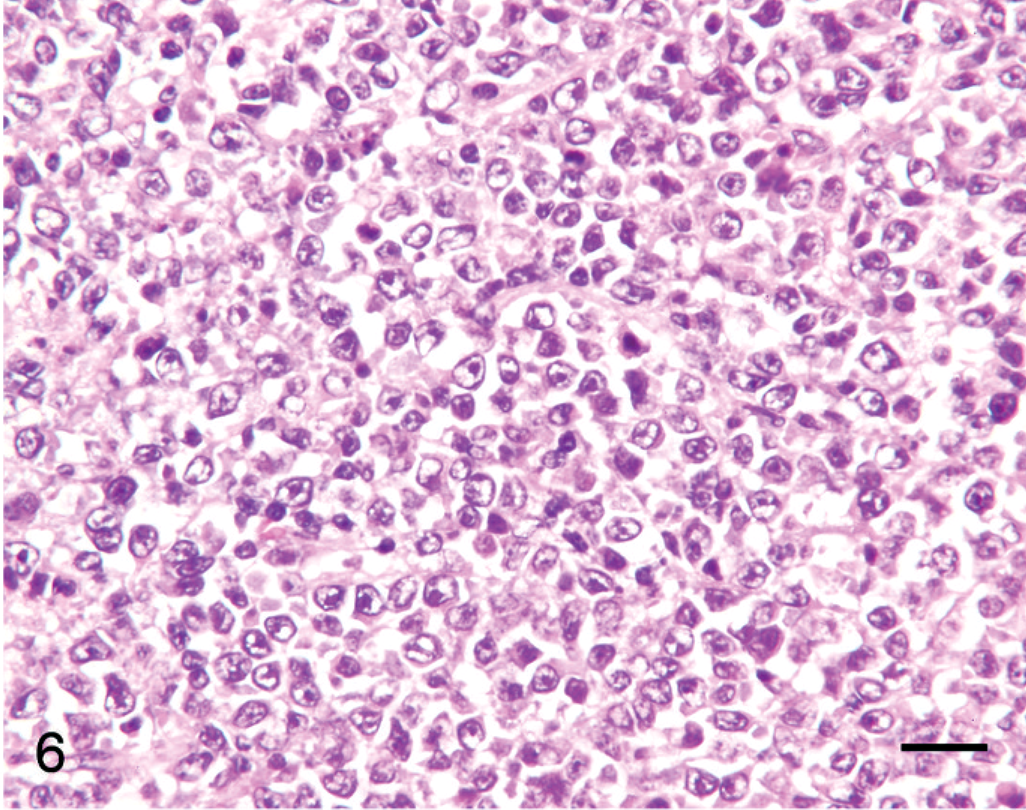
Gastric lymph node; llama No. 10. Ten-year-old llama with gastric B-cell lymphoma. Tumor cells are large and euchromatic with indistinct cytoplasm. HE. Bar = 50 µm.
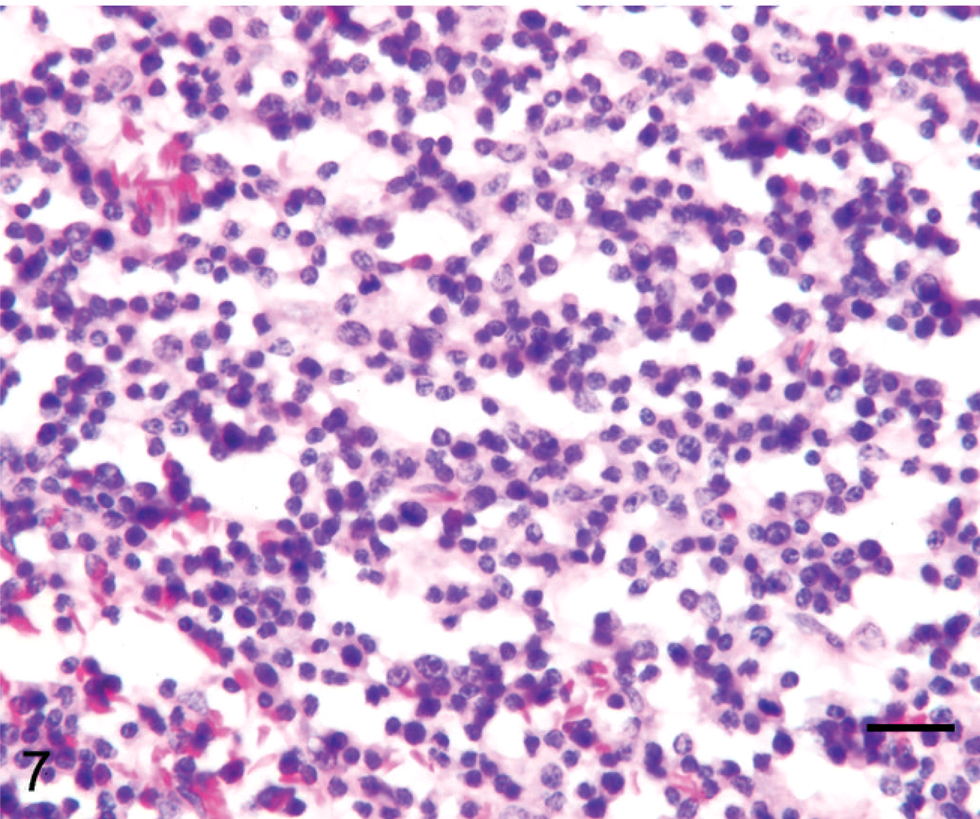
Lymph node; llama No. 17. Two-year-old llama with multicentric T-cell lymphoma. Tumor cells are small and relatively homogeneous, with granular chromatin. HE. Bar = 50 µm.
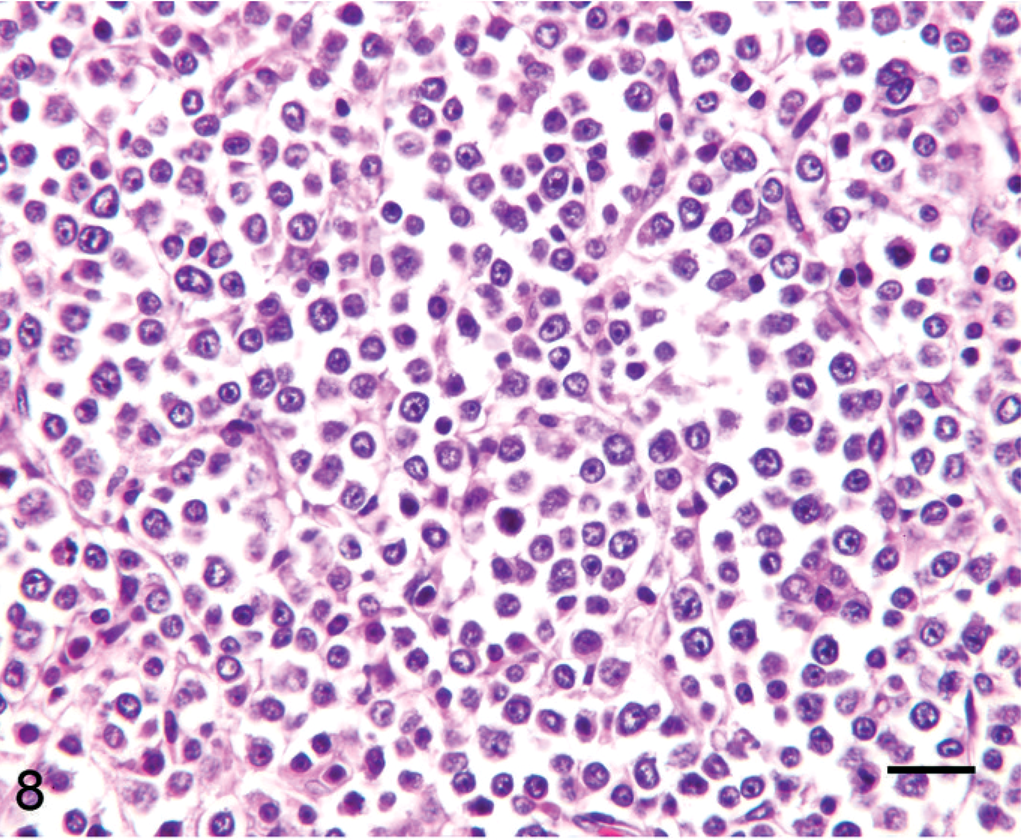
Lymph node; alpaca No. 14. Eight-month-old alpaca with multicentric primitive malignant round cell neoplasia. Tumor has relatively large and homogeneous round cells with indistinct cytoplasm and vague packeting. HE. Bar = 50 µm.
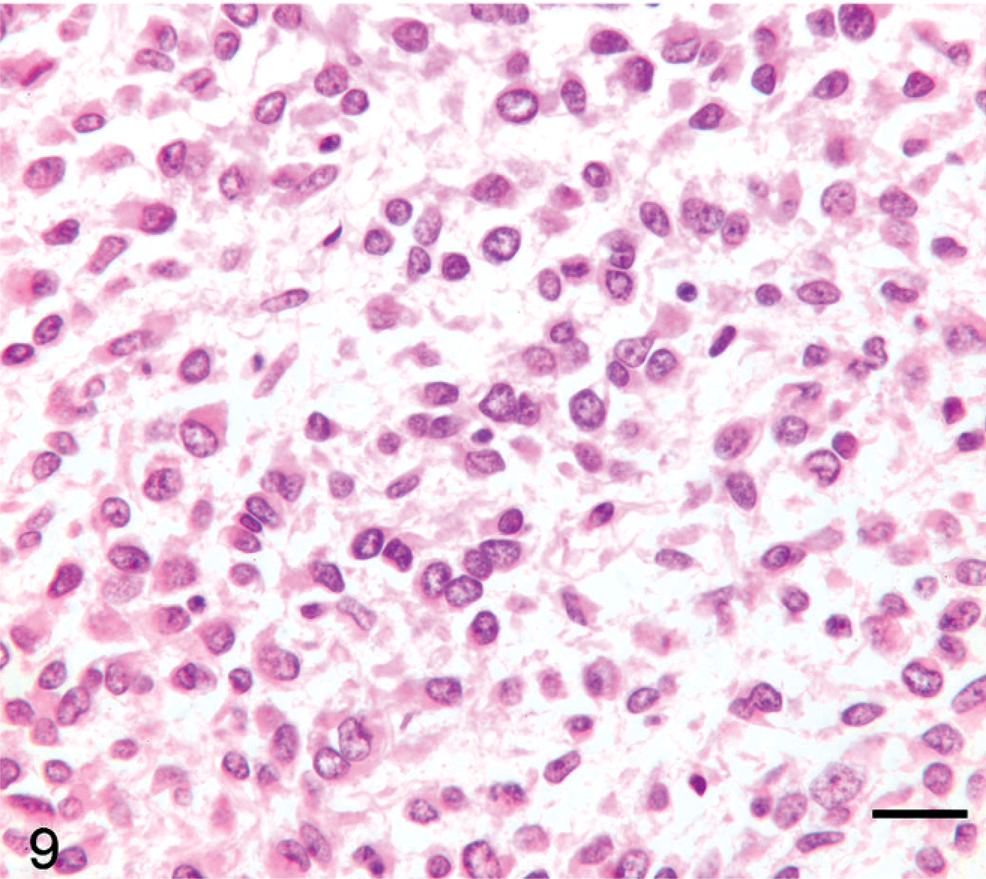
Lung; llama No. 18. Three-year-old llama with multicentric primitive malignant round cell neoplasia. Cells exhibit variable size and shape and are often ovoid to slightly elongate. Fibrillar stroma is faint. HE. Bar = 50 µm.
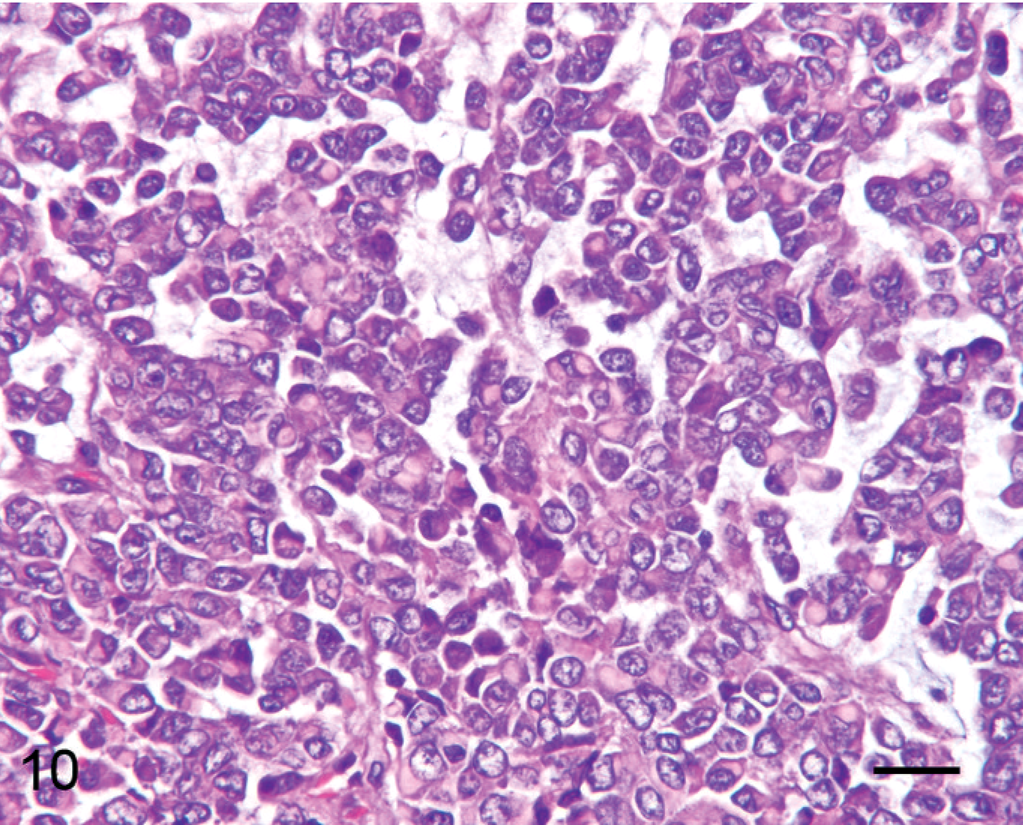
Subcutaneous cervical mass; alpaca No. 5. Four-year-old alpaca with focal primitive malignant round cell neoplasia. Tumor cells form sheets and small nests with pale basophilic myxoid matrix. Nuclei are large, euchromatic, often indented, and eccentric, and the eosinophilic hyaline cytoplasm is prominent. HE. Bar = 50 µm.
Immunohistochemical studies indicated that CD3 and CD79α appropriately labeled T and B lymphocytes, respectively, in normal camelid lymph node. BLA36 labeled some, but not all, cells within B-cell zones as well as cells interpreted to be dendritic macrophages within nodal sinuses. Neurons and glial cells in brain expressed S-100, NSE-labeled neurons, GFAP-identified astrocytes, and synaptophysin- and chromogranin A–labeled pancreatic islet cells and adrenal medullary cells.
On the basis of results of immunohistochemistry with lymphocyte markers, 18 tumors were determined to be lymphomas. Twelve tumors (7 llamas, 5 alpacas) were classified as B-cell lymphomas (Fig. 11), including 2 of the previously reported lymphoma cases (Nos. 19, 24). 32 Seven B-cell lymphomas expressed both CD79α and BLA36 (Nos. 3, 8, 12, 19, 21, 23, 24), 3 were CD79α positive and BLA36 negative (Nos. 9, 10, 13), and 2 tumors were CD79α negative and BLA36 positive (Nos. 4, 7). Two tumors, 1 CD79α+/BLA36+ (No. 23) and 1 CD79α−/BLA36+ (No. 4), were designated T-cell–rich B-cell lymphomas on the basis of the presence of a large number of small T lymphocytes mixed with larger atypical B cells. Six tumors (4 llamas, 2 alpacas) strongly expressed CD3 throughout and were designated T-cell lymphomas (Figs. 12, 13). T-cell lymphomas were most often composed of small, round, relatively homogeneous cells (Fig. 12), but marked cellular pleomorphism with scattered large binucleate to multinucleate cells was present in 1 T-cell lymphoma (Fig. 13). Necrosis of tumor cells was detected in 5 tumors and occurred in 3 B-cell lymphomas (Nos. 4, 19, 21) and 2 T-cell lymphomas (Nos. 2, 15). Mitotic index was not obviously different between tumor types, but statistics were not performed.
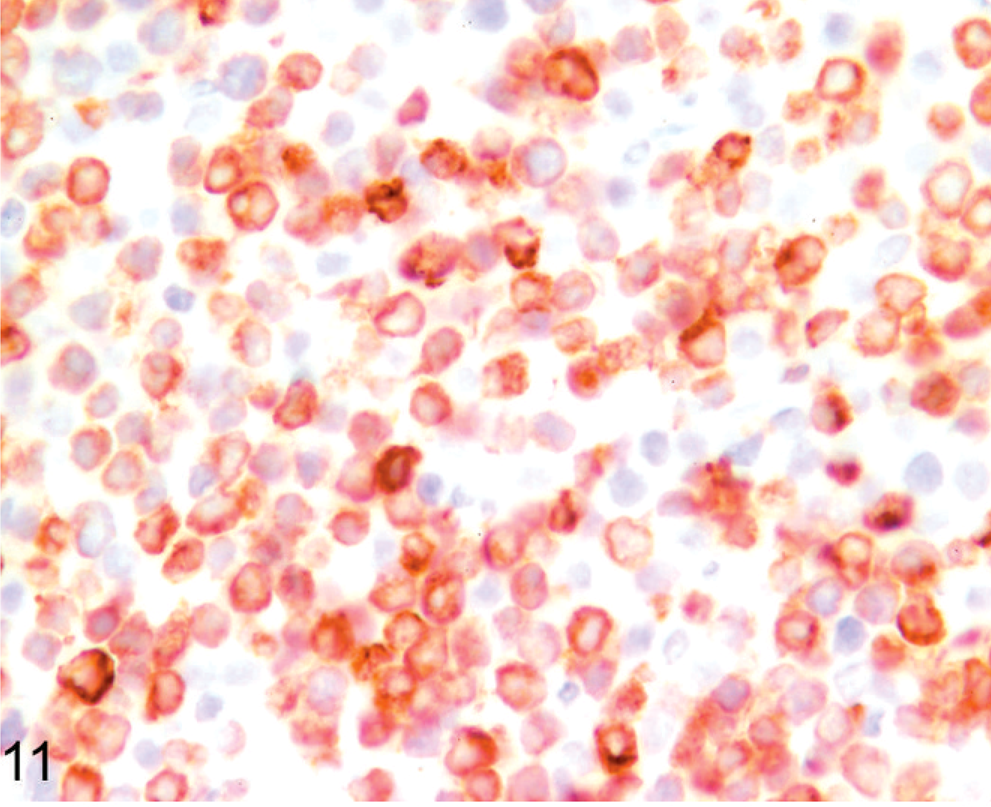
Lymph node; llama No. 12. Two-month-old llama with multicentric B-cell lymphoma. Most cells strongly express CD79α. Anti-CD79α immunohistochemistry, Nova Red chromogen, Mayer's hematoxylin counterstain.
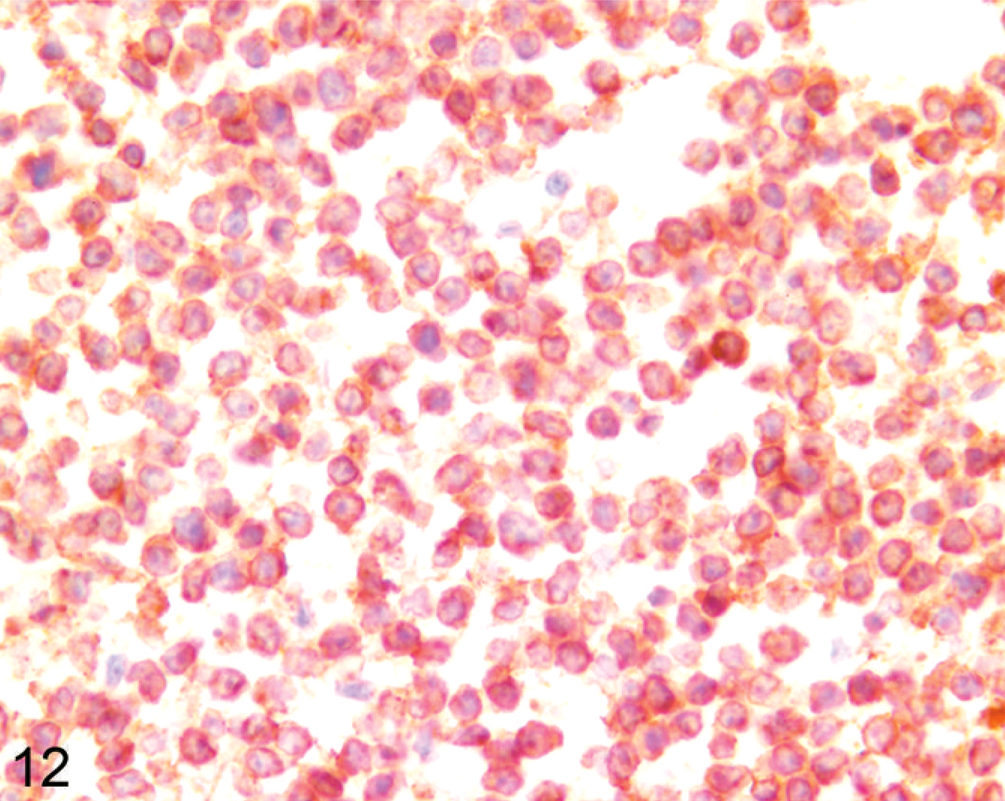
Lymph node; llama No. 20. Ten-year-old llama with multicentric T-cell lymphoma. Tumor cells are small and homogeneous and strongly express CD3. Anti-CD3 immunohistochemistry, Nova Red chromogen, Mayer's hematoxylin counterstain.
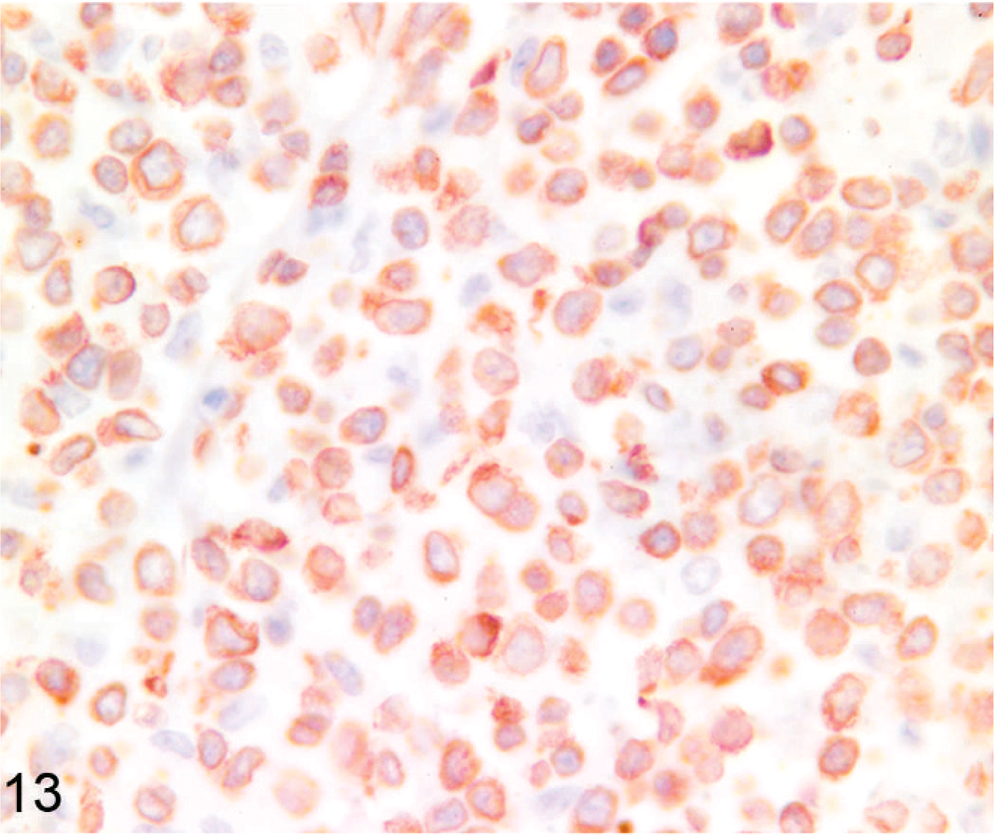
Lymph node; llama No. 22. Twenty-three–year-old llama with multicentric T-cell lymphoma. Cells are large and pleomorphic and most express CD3. Anti-CD3 immunohistochemistry, Nova Red chromogen, Mayer's hematoxylin counterstain.
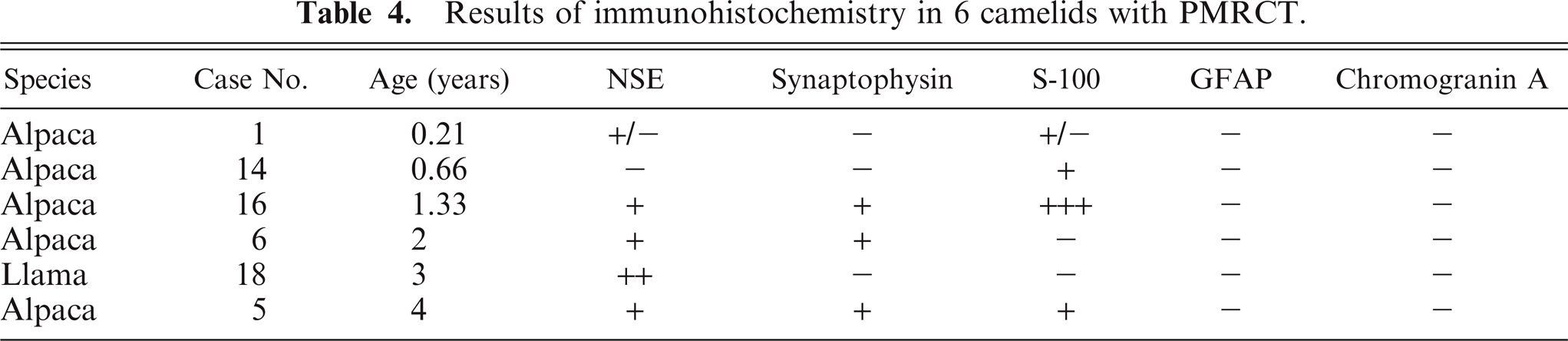
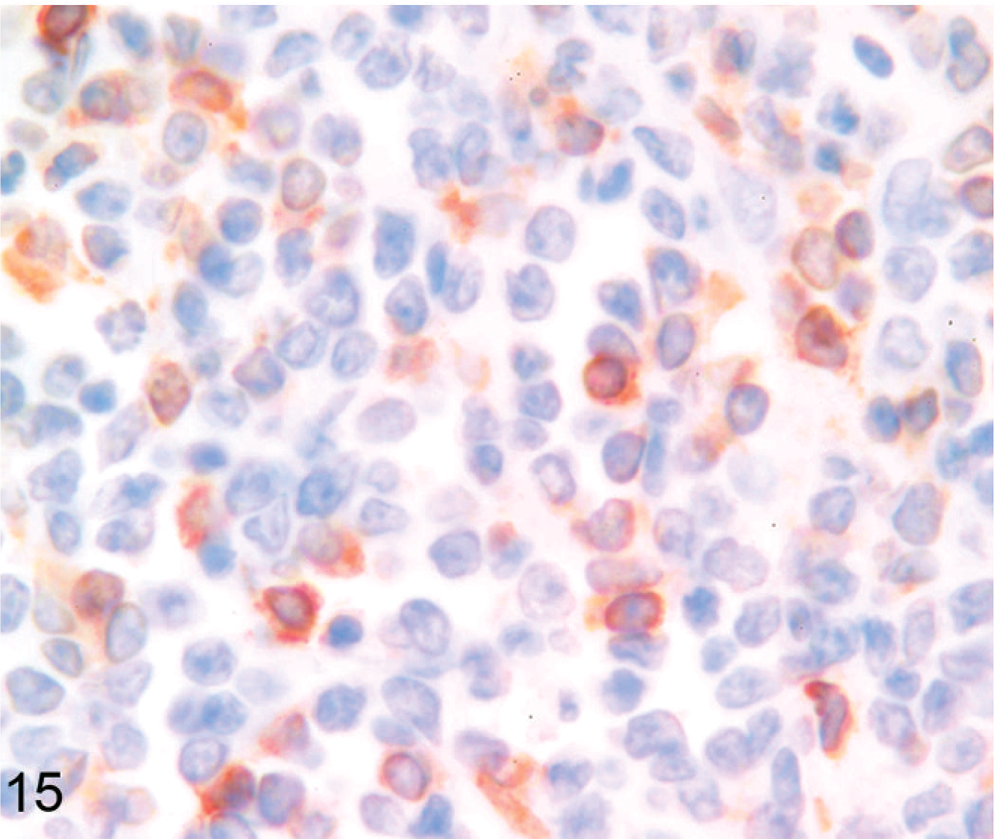
Six tumors (1 llama, 5 alpacas) did not express CD79α, BLA36, or CD3. These included 3 of 5 tumors previously reported as lymphomas. 32 These tumors were often vaguely packeted and were composed of either small cells or large cells. Four (Nos. 1, 6, 16, 18) were composed of a relatively homogeneous population of cells with heterochromatic nuclei and minimal cellular and nuclear pleomorphism (Fig. 8). Cells in 1 tumor were often slightly elongate with interspersed fibrillar material (Fig. 9). Two (Nos. 5 and 14) exhibited nuclear and cellular pleomorphism, 1 of which (No. 5) contained areas of necrosis and hemorrhage and areas with nests of cells with frequently indented and eccentric nuclei and hyaline eosinophilic cytoplasm mixed with basophilic myxoid matrix (Fig. 10). Results of additional immunohistochemical studies of these 6 tumors are summarized in Table 4. Immunostaining was graded as − (no tumor cells labeled), +/− (a small number of weakly positive tumor cells), + (a small number of moderately to strongly positive tumor cells), ++ (a moderate number of moderately to strongly positive tumor cells), and +++ (many strongly positive tumor cells). All tumors contained cells that labeled with NSE, synaptophysin, S-100, or a combination of these markers (Figs. 14–16). NSE labeling of the tumor with slightly elongate cells and fibrillar stroma revealed short cell processes (Fig. 14); this tumor was negative for all other markers. Chromogranin A and GFAP were not detected, and argentaffin and argyrophil stains did not reveal intracytoplasmic granules in any of the 6 indeterminate cases. Intracytoplasmic glycogen was not detected with PAS stain in the 2 cases examined. No evidence of neuronal differentiation, rosette formation, or perivascular pseudorosette formation was found. On the basis of these findings, a diagnosis of primitive malignant round cell tumor (PMRCT) was made.
Results of immunohistochemistry in 6 camelids with PMRCT.
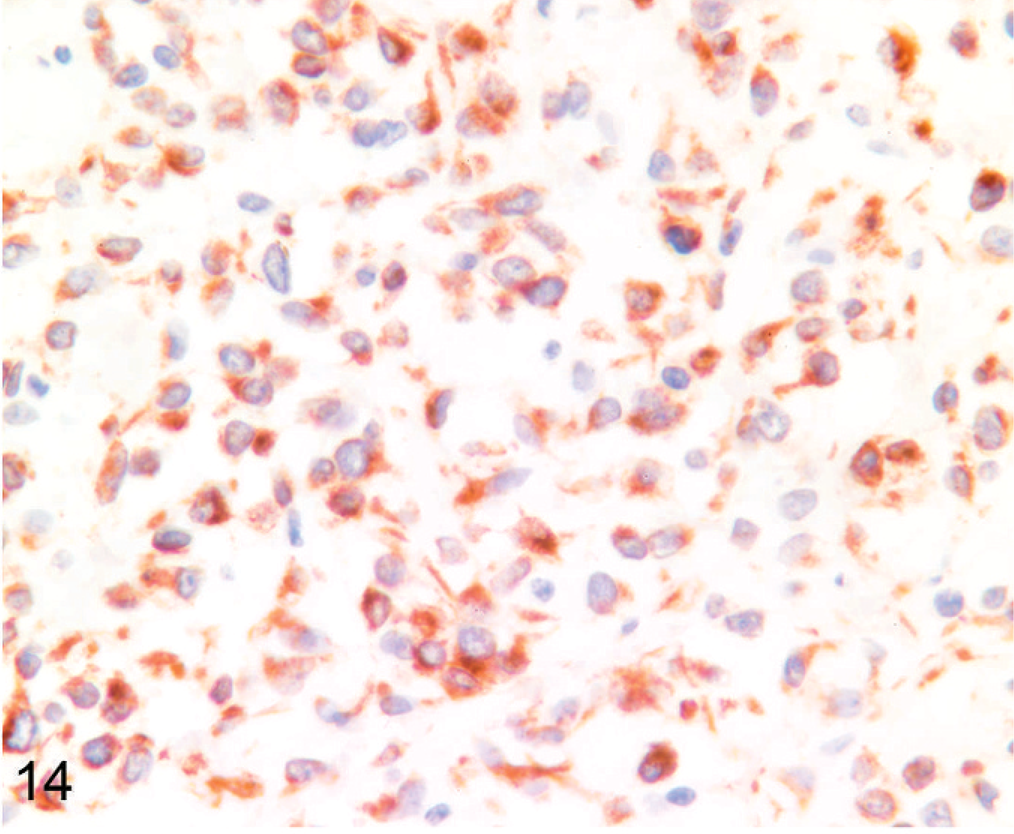
Lymph node; llama No. 18. Three-year-old llama with multicentric primitive malignant round cell neoplasia. Many cells express cytoplasmic NSE. Short cytoplasmic processes are evident. This tumor did not express synaptophysin or S-100. Anti-NSE immunohistochemistry, Nova Red chromogen, Mayer's hematoxylin counterstain.
Mesenteric lymph node; alpaca No. 6. Two-month-old alpaca with gastric primitive malignant round cell neoplasia. A moderate number of cells express synaptophysin within the cytoplasm. Tumor cells also expressed NSE but not S-100. Anti-synaptophysin immunohistochemistry, Nova Red chromogen, Meyer's hematoxylin counterstain.
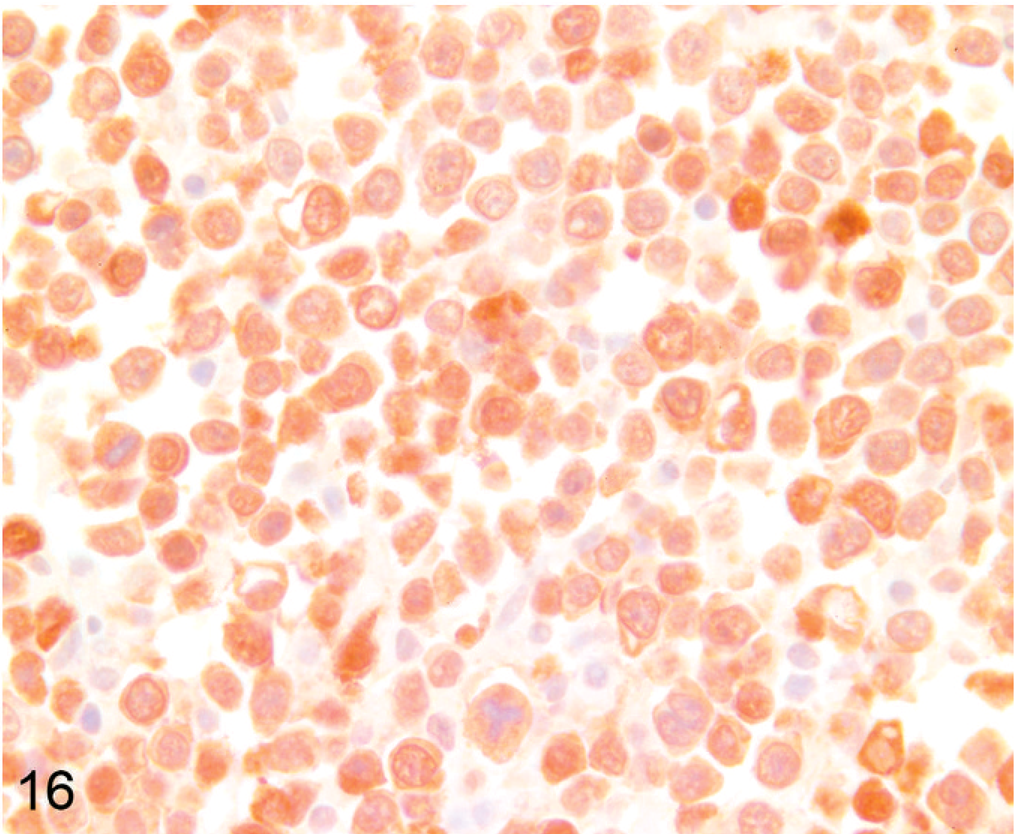
Lymph node; alpaca No. 16. Alpaca (1.33 years old) with multicentric primitive round cell neoplasia. Almost all cells strongly express nuclear and cytoplasmic S-100. Tumor cells also expressed synaptophysin and NSE. Anti-S-100 immunohistochemistry, Nova Red chromogen, Mayer's hematoxylin counterstain.
The mean age of alpacas with malignant round cell neoplasia (3.1 ± 3.6 years) was significantly less than that of llamas with malignant round cell neoplasia (8.0 ± 6.6 years; P = .034). Mean age of camelids with PMRCT (1.9 ± 1.4 years) was less than the mean age of camelids with lymphoma (6.8 ± 6.1 years), but this result did not attain significance (P = .066). Mean age of camelids with T-cell lymphoma (6.3 ± 9.0 years) was less than that of camelids with B-cell lymphoma (7.1 ± 4.6 years), but this result was not statistically significant (P = .792). Mean age of camelids with multicentric neoplasia (4.7 ± 6.8 years; range, 0–23 years) was less than that of animals with gastric neoplasia (7.2 ± 3.3 years; range, 2–10 years), abdominal neoplasia (5.1 ± 4.9 years; range, 0.21–9.5 years), and thoracic neoplasia (8.5 ± 9.2 years; range, 2–15 years), but this difference was not statistically significant (P = .787).
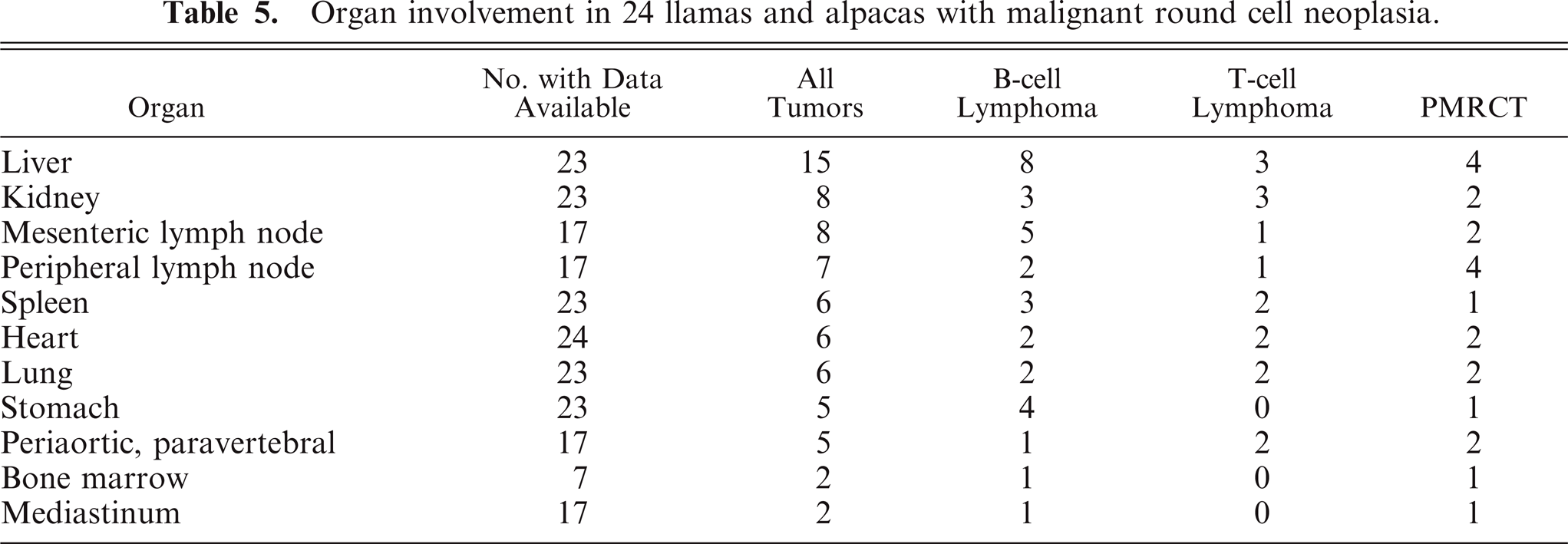
Organ involvement is summarized in Table 5. Multicentric neoplasia with no obvious primary lesion, involving peripheral, internal, or both kinds of lymph node, as well as thoracic and abdominal viscera, was the most common presentation and involved all tumor types. Primary gastric malignant round cell neoplasia arose in any of the 3 stomach compartments and was identified in 5 animals. Gastric tumors exhibited transmural infiltration with marked mural thickening (up to 6 cm thick) and extensive mucosal ulceration (Fig. 3). Primary gastric tumors were most often B-cell lymphoma (4 out of 5), but 1 tumor (No. 6) was a PMRCT. Metastasis was detected in all 5 tumors but was limited to gastric lymph node in 1 animal (No. 10). Tumors involving only the abdominal cavity (not including primary gastric tumors) were found in 4 animals (Nos. 1–4) and all occurred in perirenal tissue. Tumors involving only the thoracic cavity were identified in 2 animals (Nos. 23, 24). One unusual case of an apparently focal subcutaneous cervical PMRCT was diagnosed in a live animal (No. 5). The owners of this 4-year-old alpaca declined further workup and treatment; therefore, imaging of internal organs was not performed, and the animal was returned to the farm. Follow-up information obtained 20 months after diagnosis indicated that the animal was alive with no apparent clinical signs associated with the tumor, which had continued to slowly enlarge. Liver was the most commonly involved organ for all tumor types, detected in 15 animals (Nos. 1, 3, 4, 8, 9, 11–16, 18–21) (Fig. 2). Peripheral lymph node enlargement was detected in 7 animals (Nos. 6, 12, 14, 16, 18, 19, 22) and was often severe (Fig. 4). Bone marrow was examined in 7 animals (Nos. 8, 10, 13, 16, 18, 19, 22), and bone marrow involvement was detected in 1 animal with B-cell lymphoma (No. 13) and in 1 animal with a PMRCT (No. 18). Seven tumors, 2 PMRCTs (Nos. 14, 15), 3 T-cell lymphomas (Nos. 2, 15, 17), and 2 B-cell lymphomas (Nos. 3, 19) formed prominent multinodular periaortic and paravertebral tumor masses within the abdomen, thorax, or both (Fig. 1). Two tumors, 1 PMRCT (No. 16) and 1 B-cell lymphoma (No. 24), involved the mediastinum. Other tumor sites were diaphragm (Nos. 9, 16, 18), internal thoracic wall (Nos. 11, 17), trachea (No. 14), uterus (No. 18), adrenal gland (No. 8), distal colon (No. 2), and tongue (No. 21). Adrenal gland involvement in animal No. 8 was due to metastatic disease, and no tumor was identified as arising within adrenal glands.
Organ involvement in 24 llamas and alpacas with malignant round cell neoplasia.
Discussion
Results of this study confirm prior observations that, similar to the situation in people, 12, 15, 16, 20– 22, 30 malignant round cell neoplasms in camelids are a heterogeneous group and that immunohistochemical procedures are useful for differentiation of tumor types. 26 B-cell and T-cell markers allowed for differentiation of B-cell and T-cell lymphoma. It is possible that tumors that failed to express any of these markers could still be lymphoma, but the expression of 1 or more neural markers in tumors in this group makes this unlikely. Results of this and prior studies of camelid tumors suggest that B-cell lymphoma is more common than T-cell lymphoma 10, 23, 26 and that approximately 25% of malignant round cell tumors in camelids are PMRCTs. 26 PMRCTs were most likely to exhibit a packeted appearance on histopathology, and their cells were often a relatively homogeneous population of bland cells with minimal pleomorphism, as has been previously described. 26 However, the appearance of cells of PMRCTs was variable, and tumor morphology on routinely stained sections did not accurately differentiate tumor types in this study.
Liver and kidney were the most commonly involved organs in this study, occurring in 65 and 35% of cases, respectively. A prior study found that 80% of cases involved liver and 60% of cases involved kidney. 1 Tumor infiltration causing enlargement of peripheral lymph nodes was not as common in the present study (41% of cases in which this data was available) as in a previous report, in which peripheral lymph node enlargement occurred in 80% of cases. 1 Splenic involvement occurred in only 26% of the current cases and 20% of the cases in the prior case series. 1 The frequent involvement of cardiac structures (25% in this study, 40% in the prior study) is noteworthy, as is pulmonary involvement (26% in this study, 50% in the prior report). 1 Too few cases included examination of bone marrow to allow for determination of the prevalence of marrow involvement. Spinal cord compression leading to neurologic disease was described in 1 case in a prior study, 1 but central nervous system involvement was not detected in any of the cases in the current study.
The number of cases in this study allowed for classification of camelid malignant round cell neoplasia on the basis of tumor location. Results of this study and of prior studies indicate that multicentric neoplasia is the most common tumor distribution, occurring in 12 animals in the current series and in 13 of 23 reported cases. 1, 6, 10, 23, 26, 31 Primary gastric malignant round cell neoplasia with limited to widespread metastasis also occurs, and was identified in 5 animals in this study and in 2 animals in previous reports. 1, 29 Malignant round cell neoplasia confined to the abdomen, but not involving stomach, was identified in 5 animals in this current study and has been reported in 3 prior cases, 13, 26 1 of which also had axillary lymph node involvement. 26 Malignant round cell neoplasia confined to thoracic lymph nodes and organs occurred in 2 cases in this current report, and is described in 2 prior cases. 1, 26 A malignant round cell neoplasm described as a focal cervical mass, similar to the case described in this report, has been previously reported. 1 A complete postmortem examination was not performed in either of these cases, however, and it cannot be determined whether this was truly the only site of the tumors. Localization of 2 previously reported lymphoma cases was not possible on the basis of the case descriptions.
Immunohistochemistry proved to be extremely valuable in differentiating B- and T-cell lymphoma and nonlymphomatous PMRCTs in this study. Two tumors diagnosed as B-cell lymphoma expressed BLA36 but not CD79α. BLA36 identified dendritic macrophages as well as B lymphocytes within normal camelid lymph node, and it is possible that these tumors were of dendritic macrophage origin. Additional B-cell markers would be needed to confirm B-cell lymphoma in these 2 cases but were not available for this study. Of the few previously reported cases of camelid malignant round cell neoplasia in which immunophenotyping was performed, 10, 23, 26 4 were multicentric neoplasms that were determined to be B-cell lymphoma (2 tumors), 10, 26 1 tumor was a T-cell lymphoma, 23 and 1 was thought to express both T- and B-lymphocyte markers. 10 One intra-abdominal tumor was a B-cell lymphoma and 1 was a PMRCT. 26 One previously reported intrathoracic tumor was determined to be a B-cell lymphoma. 26
Malignant round cell neoplasia in camelids often occurs at a very young age, which mirrors the situation in people. 12, 15, 16, 20– 22, 30 Fetal lymphoma has been described in the horse 8, 14 and in cattle, 14 but this appears to be the first report of fetal lymphoma in camelids. Similar to findings of a survey of all neoplastic disease in llamas and alpacas, 32 the age of alpacas with malignant round cell neoplasia was significantly less than that of affected llamas. Other statistical analyses performed did not reveal significant differences, but a few trends are suggested by the findings of this study. Animals with PMRCT were typically younger than animals with lymphoma and were most often alpacas. Multicentric neoplasia and T-cell lymphoma were also more common in young animals. B-cell lymphoma, primary gastric tumors, and tumors confined to the abdomen or thoracic cavity were more common in older animals, and primary gastric neoplasia was most often B-cell lymphoma. The prolonged survival of the alpaca with a focal cervical mass diagnosed as a PMRCT suggests that, in some cases, PMRCT can remain localized for some time. It is worthy of note that this 4-year-old alpaca was the oldest animal diagnosed with a PMRCT.
Cytogenetic studies of tumor cells from malignant round cell tumors in people, including lymphoma, often detect specific genetic alterations. 12, 16, 20– 22 Despite a relatively small database regarding neoplasia in camelids, it is of interest to note that there are several reports of nonlymphomatous primitive tumors in South American camelids, including nephroblastoma, 4 retinoblastoma, 7 medulloepithelioma, 11, 27 and congenital hepatoblastoma. 33 It is possible that this population is predisposed to development of primitive tumors.
Primitive malignant round cell tumors in this study often involved caudal abdominal, periaortic, or paravertebral tissue or the mediastinum. Abdominal and thoracic autonomic ganglia and adrenal glands are common sites of origin of neuroblastic and neuroendocrine tumors in children 12, 15 and animals. 2, 3, 5, 9, 17– 19, 24, 25 In children, 35% of neuroblastic tumors arise in adrenal gland, 35% arise in the retroperitoneum, 20% arise in the mediastinum, and 5% occur as paravertebral masses. 12 None of the malignant round cell tumors in camelids in this or prior studies appeared to be of adrenal origin, and tumors with a similar distribution were also determined to be B-cell or T-cell lymphoma. Location did not accurately distinguish the tumor types in this study. Distinctive light microscopic features of neuroblastic and primitive neuroectodermal round cell neoplasia include formation of rosettes or perivascular pseudorosettes 15, 16, 20– 22 and ganglioneuronal differentiation in neural tumors. 12, 15, 21, 22 Argyrophilic intracytoplasmic granules can be detected in some of these tumors. 24 None of these features were detected in the PMRCTs in this study. Tumor cells of Ewing's sarcoma/primitive peripheral neuroectodermal tumors are characterized by intracytoplasmic glycogen, 12, 16, 21, 22 but glycogen was not detected in the 2 tumors in this study in which appropriate tissue was available for study.
The immunophenotype of the PMRCTs in this study is most suggestive of an origin from pleuripotential neural crest cells, 12, 15 as was suggested in a prior study. 26 Failure to express chromogranin A is consistent with an origin from primitive neuroblastic or primitive neuroectodermal cells. 15, 22 Expression of synaptophysin supports a neuroblastic rather than neuroectodermal origin. 16 However, no consistent immunohistochemical profile was identified, and it is not clear whether these PMRCTs are all the same tumor type with variable differentiation or whether they represent multiple distinct tumor types. Electron microscopy can detect neurosecretory granules to confirm a diagnosis of neuroendocrine or neuroectodermal neoplasia and can be useful in distinguishing the nature of malignant round cell tumors, 21, 22 but electron microscopy was not performed in this study.
Further studies, including electron microscopy, additional immunohistochemical studies and, ideally, cytogenetic studies of tumor cells are needed to better characterize the nature of malignant round cell tumors in camelids. Results of this and a prior study 26 suggest that, without immunohistochemical studies, a diagnosis of malignant round cell neoplasm might be more appropriate than diagnosis of lymphoma in camelid tumors with round cell morphology.
Footnotes
Acknowledgements
Supported in part by the Merck-Merial Veterinary Scholars Program. The authors thank Jermey Becker for photographic expertise; Dr. Stan Snyder, Dr. Jerry Heidel, and Dr. Olaf Hedstrom (deceased) for initial pathologic studies of some of these cases; and Misty Corbus and Renee Norred for histologic preparations.
