Abstract
A variety of embryonal tumors of the central nervous system, typically malignant and occurring in young individuals, are recognized in humans and animals. This report describes an invasive subdural but predominantly extramedullary primitive neuroectodermal tumor developing at the lumbosacral junction in a 6-month-old Brahman crossbred calf. The tumor was composed of spindloid embryonal cells organized in interlacing fascicles. The cells had oval to elongate or round hyperchromic nuclei, single to double nucleoli, and scant discernible cytoplasm. Immunohistochemical staining for neuron-specific enolase, synaptophysin, and S-100 protein and formation of pseudorosettes suggested neuronal and possibly ependymal differentiation.
Primary neoplasms of the spinal cord are rare in domestic animals. 2 With the exception of neoplasms restricted to specific sites, such as the cerebellar medulloblastoma, most tumors that occur in the brain can also be found in the spinal cord. Neoplasms arising in the spinal cord in very young animals are even more infrequent. Neuroblastoma has the potential to develop in the spinal cord or spinal ganglia. 2 Another rare tumor is the thoracolumbar spinal tumor nephroblastoma of young dogs. 2, 6
A 6-month-old Brahman crossbred bull calf with posterior paralysis and recumbency for 19 days was submitted alive to the Departmento de Patología, Universidad Nacional, Heredia, for evaluation and postmortem examination. The calf was humanely killed and a necropsy performed. Gross lesions of diagnostic significance were limited to the spinal cord. At the lumbosacral junction, there was focal thickening of the spinal cord to almost twice normal size. The area of thickening was localized and centered on the spinal nerve rootlets at this level of the cord. On cut section, the spinal cord was compressed and displaced laterally by a homogeneous white-to-tan subdural mass. The mass was relatively well demarcated from the distorted spinal cord, but, in some areas, the demarcation was indistinct. Additional gross lesions included decubitus ulcers, atrophy and pallor of hind limb muscles, and a distended urinary bladder.
Multiple sections of spinal cord at the level of the subdural mass were fixed in 10% neutral buffered formalin, processed, and embedded in paraffin according to accepted histologic technique. Five micron sections were stained routinely with hematoxylin and eosin (HE). Immunohistochemical studies were performed using the avidin-biotin-peroxidase complex (ABC) method (Vectastain Elite ABC Kit, Vector Laboratories, Burlingame, CA).
The primary antibodies used were mouse monoclonal antibodies against neuron-specific enolase (NSE, 1 : 400; DAKO, Carpinteria, CA), vimentin (1 : 300, DAKO), and synaptophysin (1 : 400, DAKO) and rabbit antibodies against S-100 protein (1 : 300; DAKO), glial fibrillary acidic protein (GFAP, 1 : 800; DAKO), and chromagranin A (1 : 400, DAKO). Negative-control sections were incubated in nonimmune serum in place of the primary antibodies.
Microscopic examination showed a subdural but largely extramedullary densely cellular mass. In most areas, the mass was separated from the spinal cord by the collagenous connective tissue of the leptomeninges. All funiculi, central gray matter, the central canal, and the ventral spinal artery were compressed and distorted by the mass, but all these structures were identifiable in all sections. In focal areas of some sections, the mass extended through the leptomeninges and into the white matter of the lateral funiculus but did not progress to the central canal or gray matter of the spinal cord.
Neoplastic cells also extended into the dura mater and along spinal nerve rootlets to form solid, small epidural aggregates. The mass was composed predominantly of compact to loosely organized interlacing fascicles of spindloid cells (Fig. 1). In some areas, the cells were more round or polyhedral, at times with apparent palisading of cells in a radial fashion about small blood vessels forming pseudorosettes (Fig. 2). Formation of neuroblastic (Homer-Wright) or ependymal rosettes was not evident. The cells had oval to elongate or round hyperchromic nuclei, single to double nucleoli, and scant discernible cytoplasm. The mitotic rate was 3 per 10 400× fields. Small capillaries and bands of collagen traversed the mass, the latter being most obvious in areas with a loosely organized cellular arrangement.
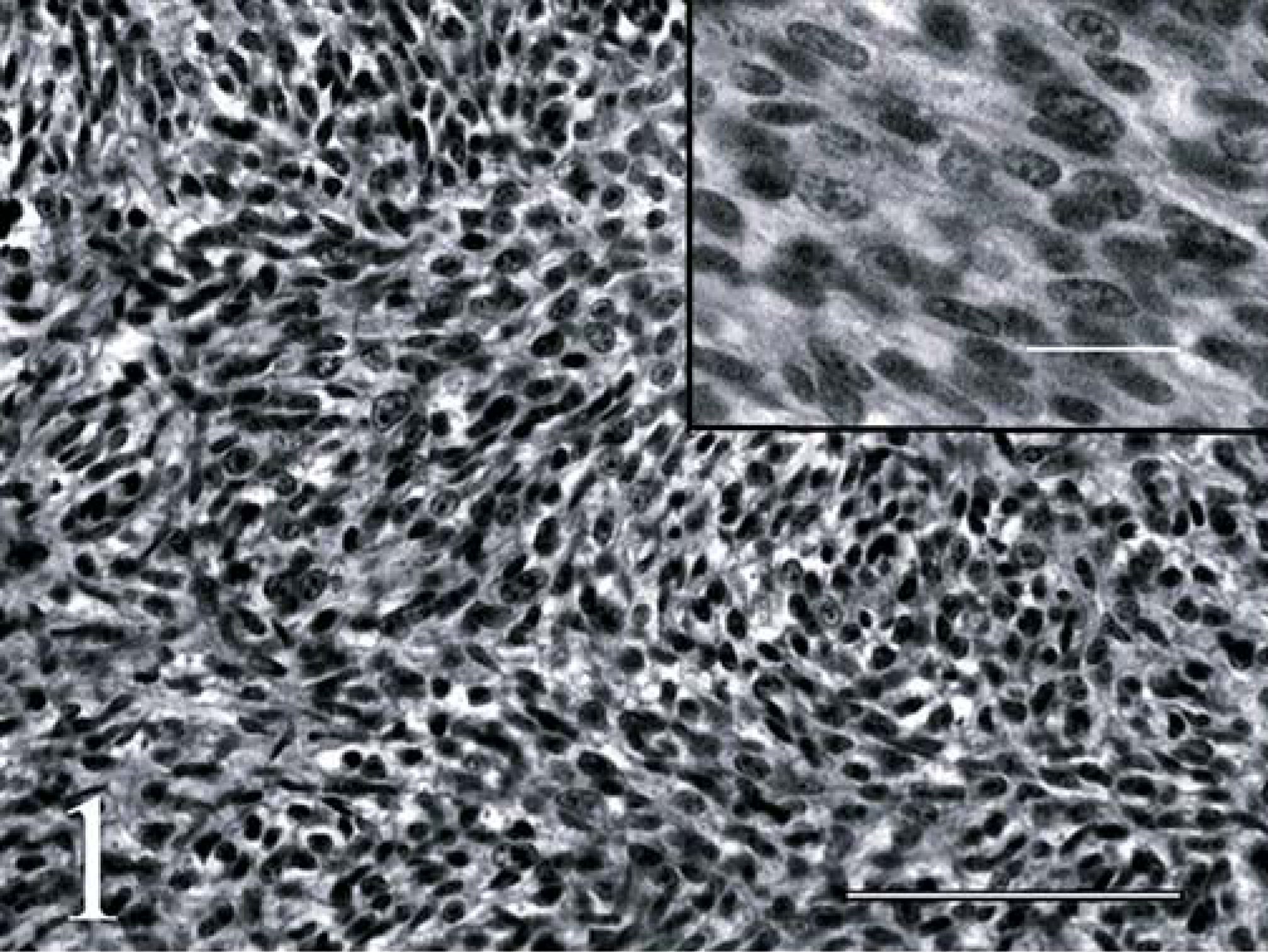
Spinal cord, primitive neuroectodermal tumor (PNET); calf. Interlacing fascicles of primitive embryonal cells from a spinal cord PNET in a calf. Hematoxylin and eosin (H&E). Bar = 100 µm. Inset; higher magnification of cells in the PNET. H&E. Bar = 20 µm.
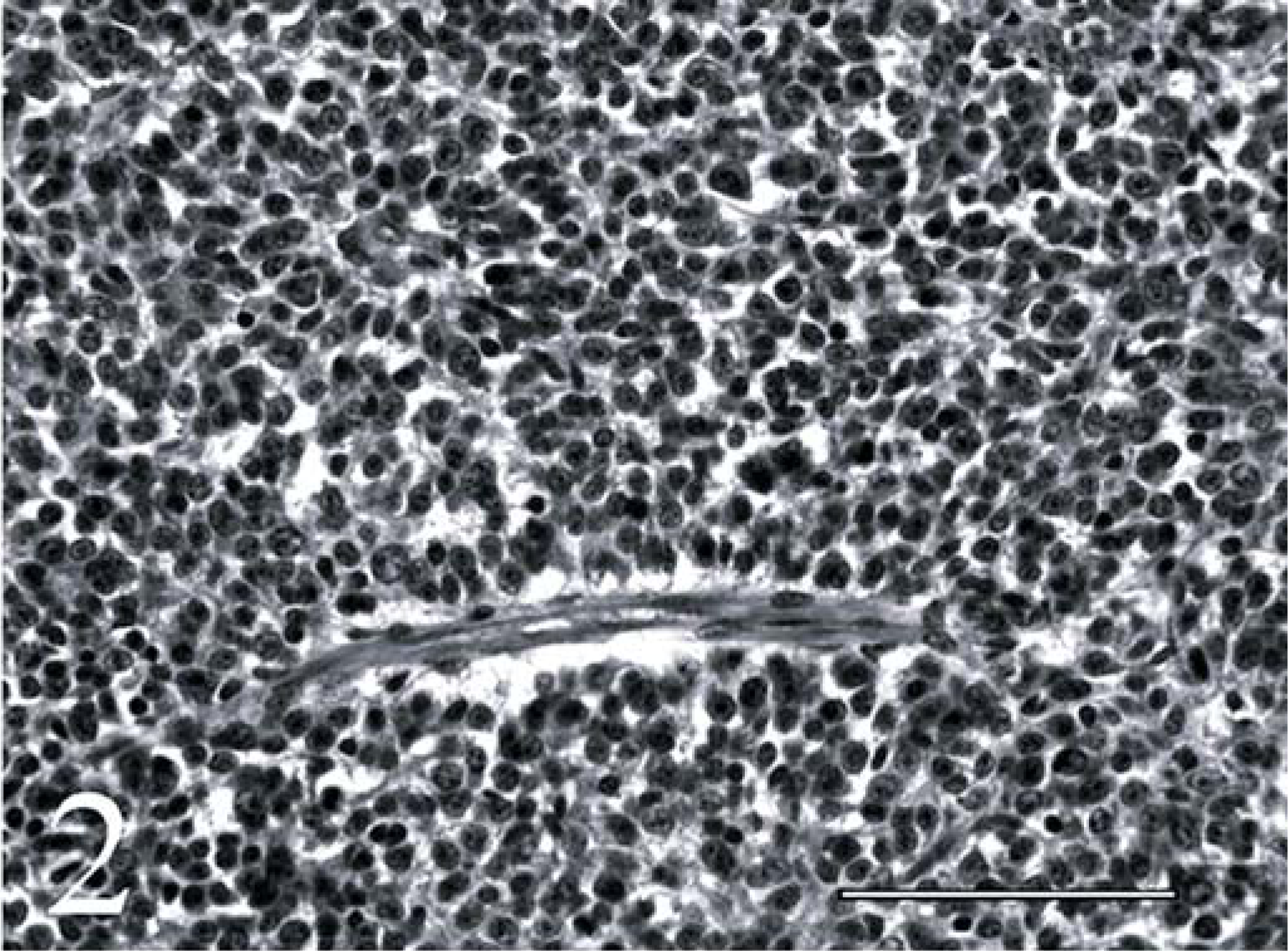
Spinal cord, primitive neuroectodermal tumor; calf. Palisading of primitive embryonal cells around a blood vessel forming a pseudorosette. Hematoxylin and eosin. Bar = 100 µm.
Immunohistochemical staining for S-100 and NSE was weak to moderate, but definitely positive in a large percentage of cells throughout the mass in both loosely organized and compact cellular areas (Figs. 3, 4).
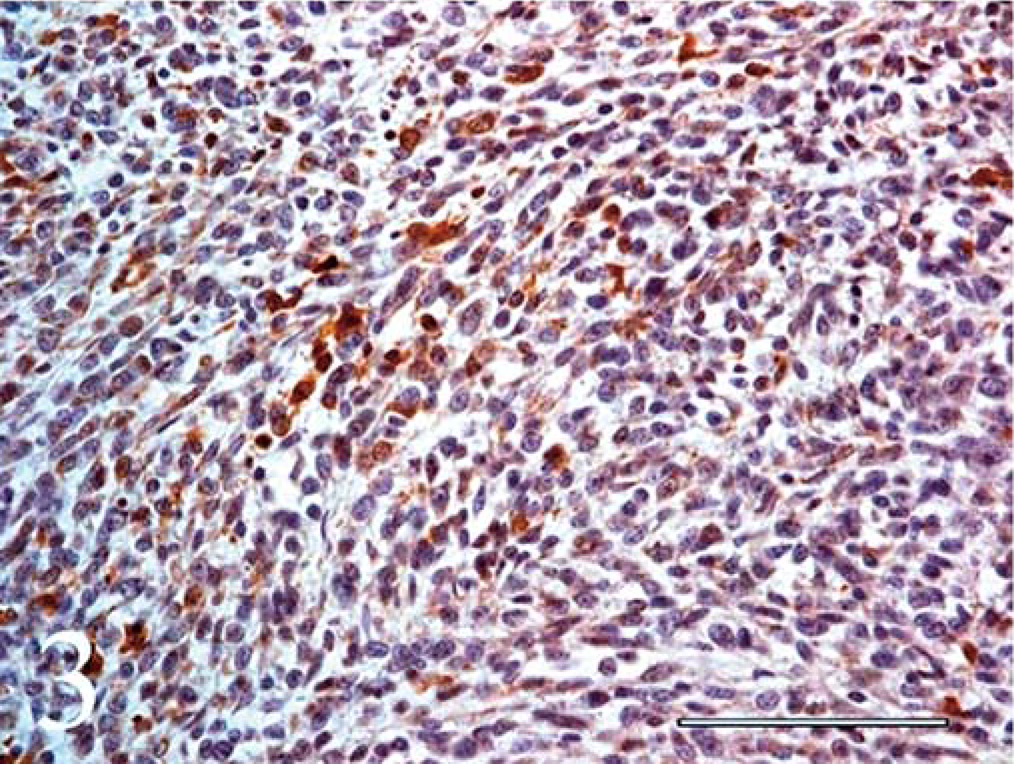
Spinal cord, primitive neuroectodermal tumor (PNET); calf. Positive staining for NSE from a spinal PNET in a calf. Avidin-biotin-peroxidase complex method. Hematoxylin counterstain. Bar = 100 µm.
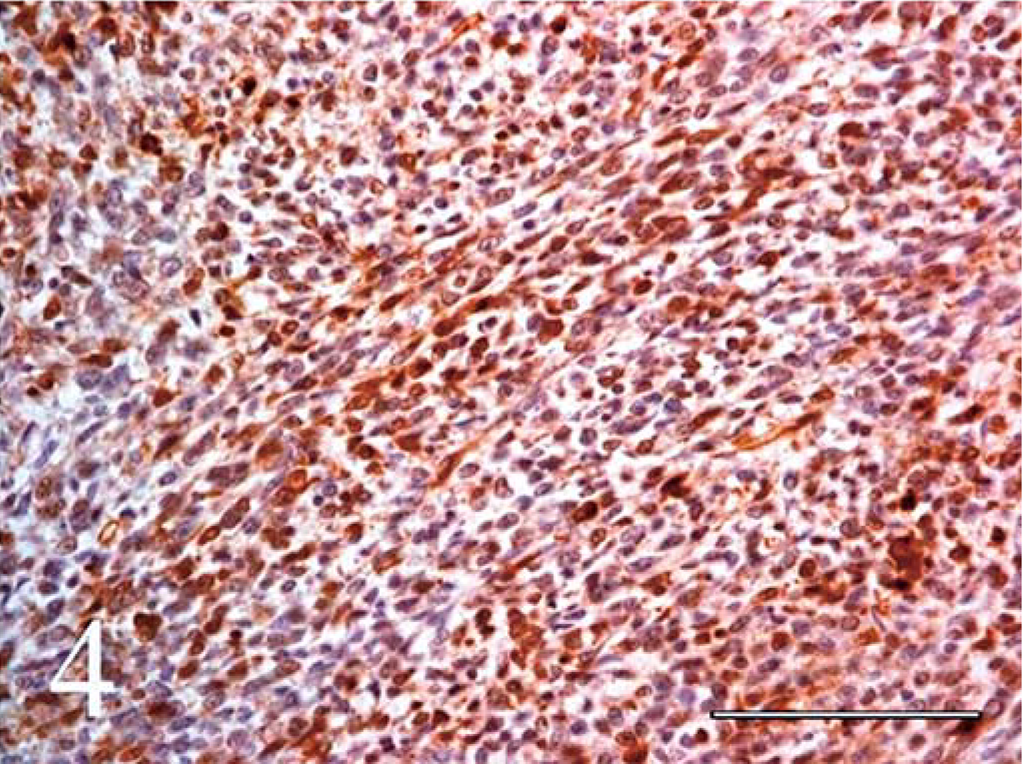
Spinal cord, primitive neuroectodermal tumor (PNET); calf. Positive staining for S-100 protein from a spinal PNET in a calf. Avidin-biotin-peroxidase complex method. Hematoxylin counterstain. Bar = 100 µm.
Positive staining for synaptophysin was present in a minority of neoplastic cells, mainly in more compact areas of the mass. Strong vimentin staining was confined to capillaries and bands of stroma. Weak staining for vimentin was present in a small percentage of neoplastic cells. Staining for GFAP and chromagranin A was not present in the neoplastic cells. A diagnosis of primitive neuroectodermal tumor (PNET) was made based on cellular morphology and organization and immunohistochemical staining.
A variety of embryonal neoplasms thought to be of primitive neuroectodermal origin are recognized in the central nervous system in humans and some have been reported in animals. 2, 3, 6 These include medulloblastoma, neuroblastoma, medulloepithelioma, ependymoblastoma, and PNET. 3 Common characteristics are typical occurrence at a young age, a high degree of malignancy, and morphologic features of small cells having round, oval, or elongate hyperchromic nuclei and scant cytoplasm. Results of immunohistochemical staining can indicate multiple lines of neuroectodermal cellular differentiation, primarily neuronal, ependymal, and glial. 3, 5
There is confusion and controversy regarding use of the term PNET. While all of the above tumors could be considered PNETs in a general sense (usually with modifiers to indicate the primary line of differentiation), some are distinct entities based on characteristic patterns of cellular organization and/or development in characteristic locations. Thus, the current World Health Organization and other classifications prefer to separate cerebellar medulloblastoma (which could be considered the prototype PNET), neuroblastoma, medulloepithelioma, and/or ependymoblastoma as distinct entities. 2, 3, 6 The use of PNET as a specific diagnosis could then be reserved for those tumors lacking distinctive patterns of cellular organization or developing in uncharacteristic locations. There are also reports of PNETs developing in peripheral, nonneural tissues.
Using the term PNET in this more restricted sense, most human PNETs of neural origin develop in the cerebral hemispheres. 3 Involvement of the spinal cord is rare. 1, 3, 8 Formation of neuroblastic (Homer-Wright) rosettes is a distinctive feature suggestive of neuronal differentiation but may not be present. These rosettes are formed by radially oriented, elongate cells encircling an eosinophilic fibrillary core of cellular processes. Immunohistochemical staining often indicates multiple lines of differentiation, as noted previously.
Primitive neuroectodermal tumors are being reported with increasing frequency in animals, including cattle, 4, 7 probably in part due to the increased utilization of immunohistochemical stains for routine diagnosis.
Previous reports of PNET in cattle indicated positive staining for vimentin and NSE 4 and neurofilament protein as well as NSE but no staining for vimentin and S-100 protein. 7 In the current case, staining for NSE and synaptophysin would suggest neuronal differentiation. The formation of pseudorosettes along with the positive staining for S-100 protein in many cells might indicate concurrent ependymal differentiation of the tumor in this calf. Staining for GFAP, most commonly observed in pseudorosettes, and vimentin can also indicate ependymal differentiation but is not present in all cases. 3 Positive staining for vimentin was weak and only observed in a minority of neoplastic cells in this calf, while GFAP was negative. Ganglioneuroblastoma originating in a spinal ganglion was also a consideration in this case. In humans, positive immunohistochemical staining for NSE and S-100 protein is observed but typically in morphologically distinct subpopulations of cells, whereas in the present case, both NSE and S-100 staining occurred in morphologically similar embryonal cells. 3 Additionally, the bulk of the tumor was within the confines of the dura mater and no continuity could be established with spinal ganglia or the gray matter of the spinal cord.
Footnotes
Acknowledgements
We would like to thank Dr. Horold Marin for submission of this case and Dr. Andy Ambrus for assistance with immunohistochemistry.
