Abstract
Fusobacterium necrophorum, a Gram-negative, anaerobic bacterium, is an opportunistic animal and human pathogen that causes a variety of infections termed necrobacillosis. There are 2 subspecies of F. necrophorum (subsp. necrophorum and subsp. funduliforme) that differ morphologically and biochemically and in virulence. Leukotoxin, a secreted protein, is considered to be the major virulence factor. In camelids, F. necrophorum causes a variety of infections, generally involving the lips, tongue, pharynx, interdigital spaces, foot pad, larynx, mandible, or maxillary bones. The objective of the current study was to characterize the presumptive Fusobacterium isolates from a variety of necrotic infections in llama (Lama glama) and alpaca (Vicugna pacos) and determine whether the strains possess leukotoxin activities. A total of 7 isolates from alpaca and 2 isolates from llama were characterized. Based on growth characteristics in broth culture, and biochemical and polymerase chain reaction analyses, all 9 isolates belonged to subsp. necrophorum and possessed the putative hemagglutinin gene. Western blot analysis with antileukotoxin antibodies raised in rabbit showed the presence of leukotoxin protein in the culture supernatant of all isolates. Furthermore, flow cytometry of the culture supernatants demonstrated cytotoxicity to bovine and alpaca polymorphonuclear leukocytes (PMNs). The extent of cytotoxicity to either alpaca or bovine PMNs differed among camelid strains. The cytotoxicity of many of the camelid strains was higher (P < 0.05) toward alpaca PMNs compared to bovine PMNs. Fusobacterium necrophorum isolates from llama and alpaca are similar to bovine isolates, and leukotoxin may be a major virulence factor.
Keywords
Fusobacterium necrophorum is a Gram-negative, anaerobic, and rod-shaped bacterium that causes a variety of suppurative and necrotic infections, generally called necrobacillosis, in animals and human beings. 9 Two subspecies of F. necrophorum are recognized. 12 The subsp. necrophorum is more frequently encountered in animal infections, 6 while clinical isolates from human infections have the characteristics of subsp. funduliforme. 13 The subsp. necrophorum is more virulent 6 because of the presence and/or increased production of potent virulence factors, such as leukotoxin, lipopolysaccharide, and hemagglutinin. 9 Of these factors, leukotoxin is considered to be the major virulence determinant in the pathogenesis of animal infections. 9
In wild and captive animals, necrobacillosis has been reported to cause infections of the oral cavity in white-tailed deer (Odocoileus virginianus), mule deer (Odocoileus hemionus), pronghorn antelopes (Antilocapra americana), and blue duikers (Philantomba monticola; syn. Cephalophus monticola fusicolor).2,3,11,16 In camelids, F. necrophorum infections occur on the lips, tongue, pharynx, interdigital spaces, foot pad, larynx, mandible, or maxillary bones. 5 In alpaca (Vicugna pacos), the organism has been reported to cause osteomyelitis of mandibles and many disseminated necrotic lesions. 5 Occasionally, the infection may be aspirated to the lung, causing severe necrotizing pneumonia, while infections in the interdigital space and foot pad can lead to lameness. 5 The current study examined presumptive Fusobacterium isolates from a variety of necrotic infections in llama (Lama glama) and alpaca to identify the species and subspecies, and to determine whether the strains possess leukotoxin activity.
A total of 9 isolates, 2 from llama (61557 and 69521) and 7 from alpaca (11034, 61715, 68690, 68699, 73556, 118610, and 1012060) were included in the study (Table 1). Fusobacterium necrophorum subsp. necrophorum strain A25 and subsp. funduliforme, strain B35, both previously isolated from liver abscesses of cattle, 6 served as reference cultures in all the tests conducted to characterize the llama and alpaca isolates. All strains were cultured in brain-heart infusion broth a (BHI) that was prereduced with 0.05% cysteine hydrochloride and anaerobically sterilized (PRAS). 15 The growth characteristic (sedimentation or no sedimentation) of the isolates in the PRAS-BHI broth was recorded, and the isolates were tested biochemically using a commercial kit. b
Source of Fusobacterium necrophorum isolates from llama and alpaca and polymerase chain reaction (PCR) analysis results.
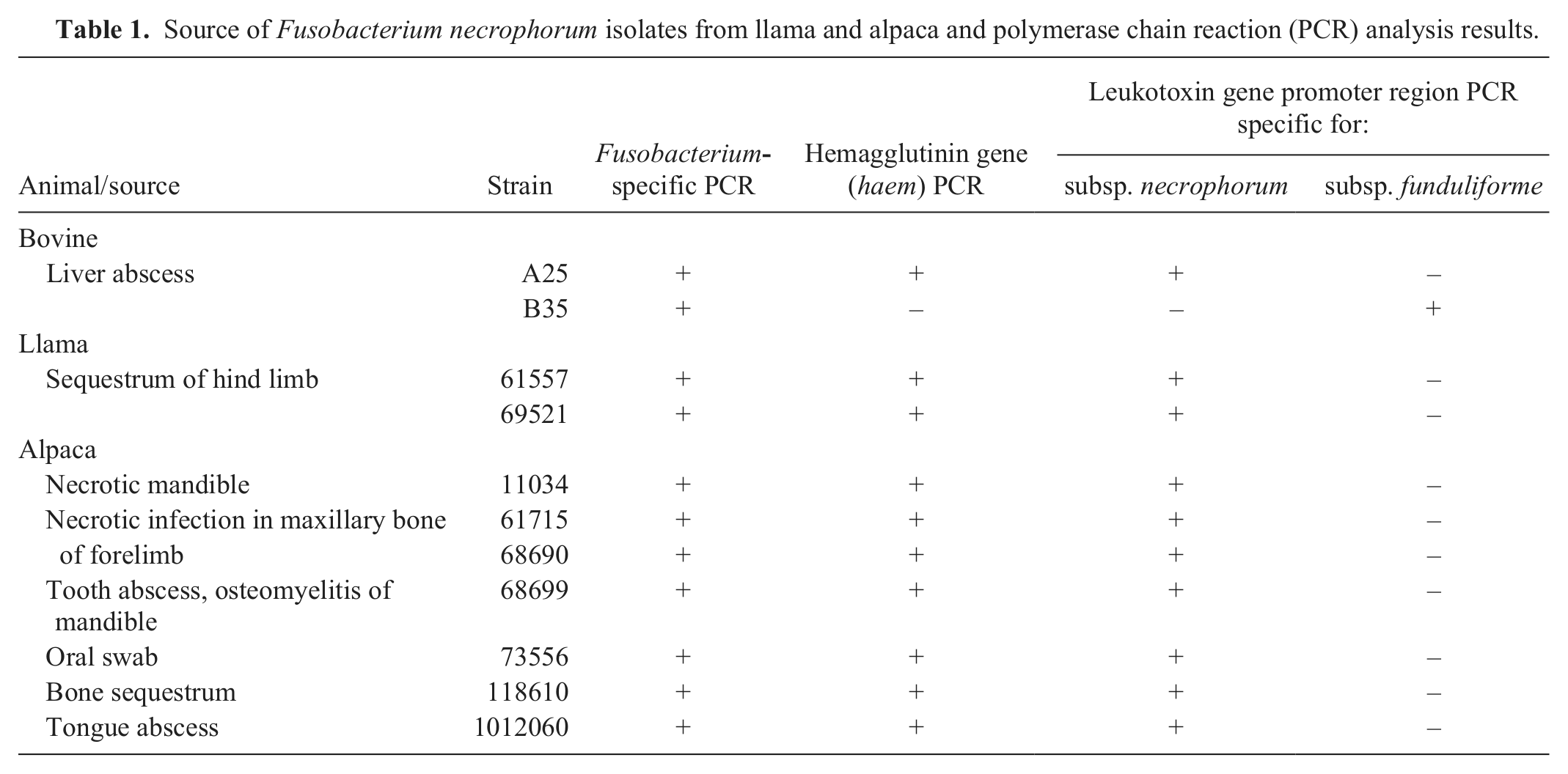
All isolates showed uniform turbidity in the broth cultures (no sedimentation) and, based on the commercial identification kit, all isolates were positive for indole production and alkaline phosphatase activity, similar to subsp. necrophorum, strain A25. Many of the isolates, except oral swab (73556) and tongue abscess (1012060) isolates, obtained from llama and alpaca were associated with infections involving the bone. The alkaline phosphatase activity may help the organism in bone resorption creating a deep wound, and thus making a more anaerobic environment for the bacterial growth. 17 The frequent association of F. necrophorum in camelids with necrotic infections in bone needs further study and explanation.
To identify the organisms at the molecular level, different polymerase chain reaction (PCR) assays were carried out using a PCR machine. c The genus-specific PCR assay based on the 16S ribosomal RNA gene 8 yielded an amplicon of 610 bp, confirming that all isolates belonged to genus Fusobacterium (Fig. 1A). The subspeciation of the isolates was based on PCR amplifications of hemagglutinin (haem) gene 1 and the leukotoxin (lkt) operon promoter–containing intergenic region. 18 The primer sequences, PCR running conditions, and amplicon sizes are shown in Table 2. The PCR specific for the hemagglutinin gene amplified a 315-bp product from all llama and alpaca isolates and in the bovine strain of subsp. necrophorum, but not in the bovine strain of subsp. funduliforme (Fig. 1B). The PCR assay designed to amplify the lkt promoter–containing intergenic region of the subsp. necrophorum amplified a product of 571 bp with all the isolates and that of the bovine strain of subsp. necrophorum (Fig. 1C). In contrast, the PCR assay designed to amplify the lktpromoter–containing intergenic region of subsp. funduliforme gave a negative result with all of the camelid isolates (Fig. 1D) and, as expected, amplified a product of 337 bp with the bovine strain of subsp. funduliforme. A summary of the PCR results is given in Table 1. Overall, the results revealed that the Fusobacterium isolates from alpaca and llama belonged to subsp. necrophorum. This is similar to a previous observation that necrotic infections caused byF. necrophorum in cattle are more often caused by subsp. necrophorum. 9
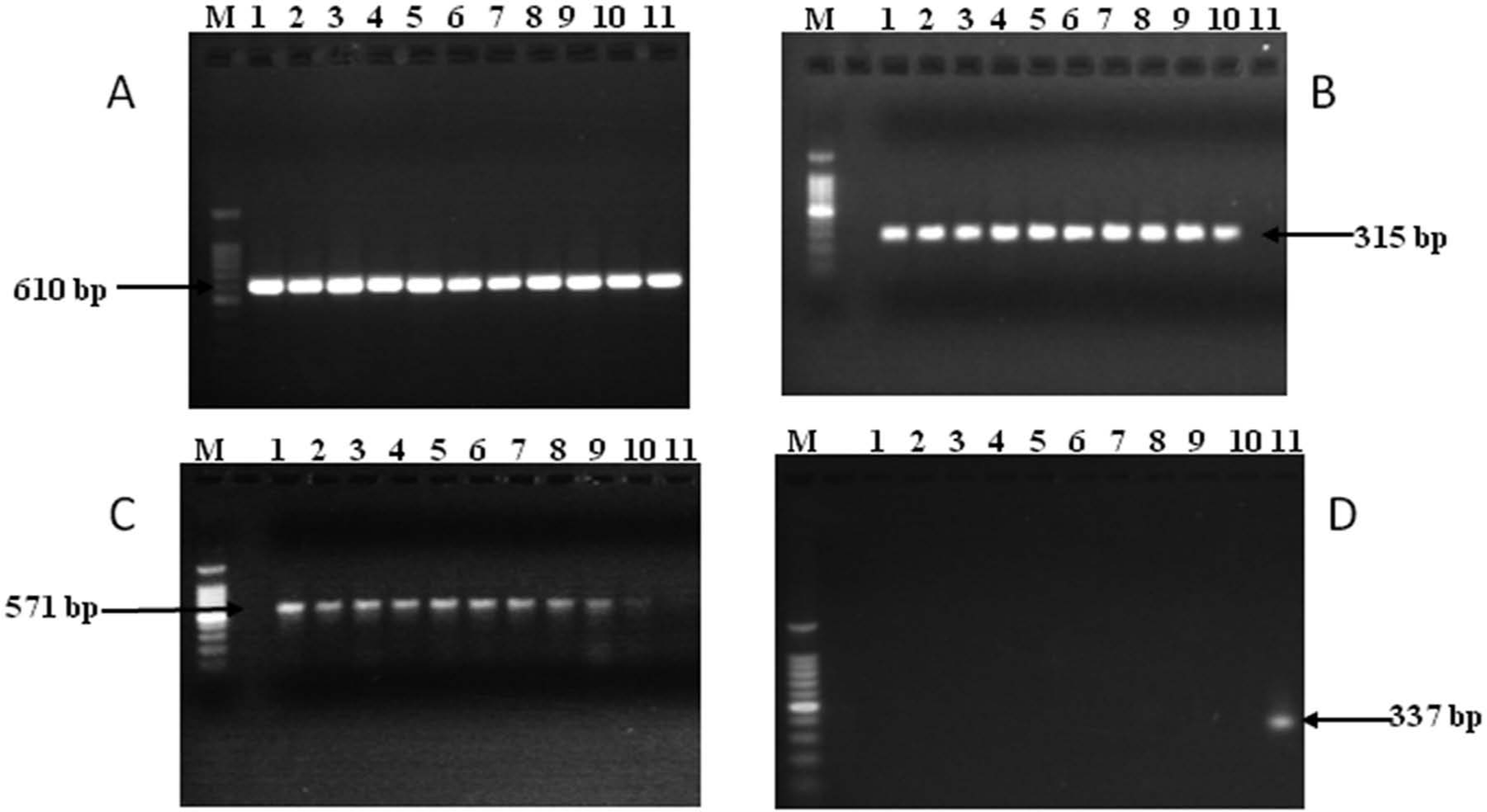
Polymerase chain reaction amplification of genes targeting Fusobacterium genus–specific 16S ribosomal DNA (
Primers, target genes, polymerase chain reaction (PCR) running conditions, and amplicon sizes.
The production of leukotoxin was tested by a Western blot assay for the leukotoxin protein in culture supernatant. An aliquot of each supernatant, containing 20 µg of protein as determined by Bradford assay, d was loaded into each lane. Rabbit polyclonal antiserum, raised against affinity-purified leukotoxin from a bovine strain of subsp. necrophorum, was used as the primary antibody 10 followed by incubation with goat antirabbit immunoglobulin G conjugated with alkaline phosphatase e as the secondary antibody. The culture supernatant was prepared by growing the isolates in PRAS-BHI broth to an absorbance of 0.60–0.65 at 600 nm, pelleting cells by centrifugation, filtering through a 0.22-µm filter, f and concentrating 60-fold with a 100-kDa molecular mass cutoff filter. f The Western blot with concentrated supernatants from all strains showed 110 and 90 kDa bands, similar to that of bovine strain of subsp. necrophorum A25 (Fig. 2). A 250-kDa band was observed in strains 1012060, 61715, 69521, and 73556, but the band was absent in 11034, 68690, 68699, and 118610 strains and was faint in the bovine strain of A25. The 150-kDa band was present in all strains except in strains 11034, 68690, 68699, and 118610. Bands of 130 and 125 kDa were present in all strains, including the bovine strain. In 2 strains, 68690 and 68699, all bands were generally of low intensity. One strain (1012060) showed additional high-intensity bands of 80, 60, and 40 kDa. Alpaca strains 61715 and 73556 showed 2 additional bands in the 30-kDa range, and the 30-kDa band was more prominent. These 2 bands were absent in all other strains (Fig. 2). The higher intensity of protein bands from strain 1012060 could be either due to difference in the promotor sequence leading to a higher expression of leukotoxin or to difference in amino acid sequence of the protein increasing its affinity to the antibody. However, the higher expression of leukotoxin by the strain 1012060 was not expected because the cytotoxicity of this strain on polymorphonuclear leukocytes (PMNs) was less compared to 2 other camelid strains (as described below). In addition, the multiple bands that appeared in the current analysis supports the previous observation that the leukotoxin from F. necrophorum is highly unstable.13,15 The difference in the banding pattern among the strains is most likely because of cleavage by proteolytic enzymes. 14 Likewise, the difference in intensities of similar bands in different strains could also be attributed to the difference in the proteolytic activity.
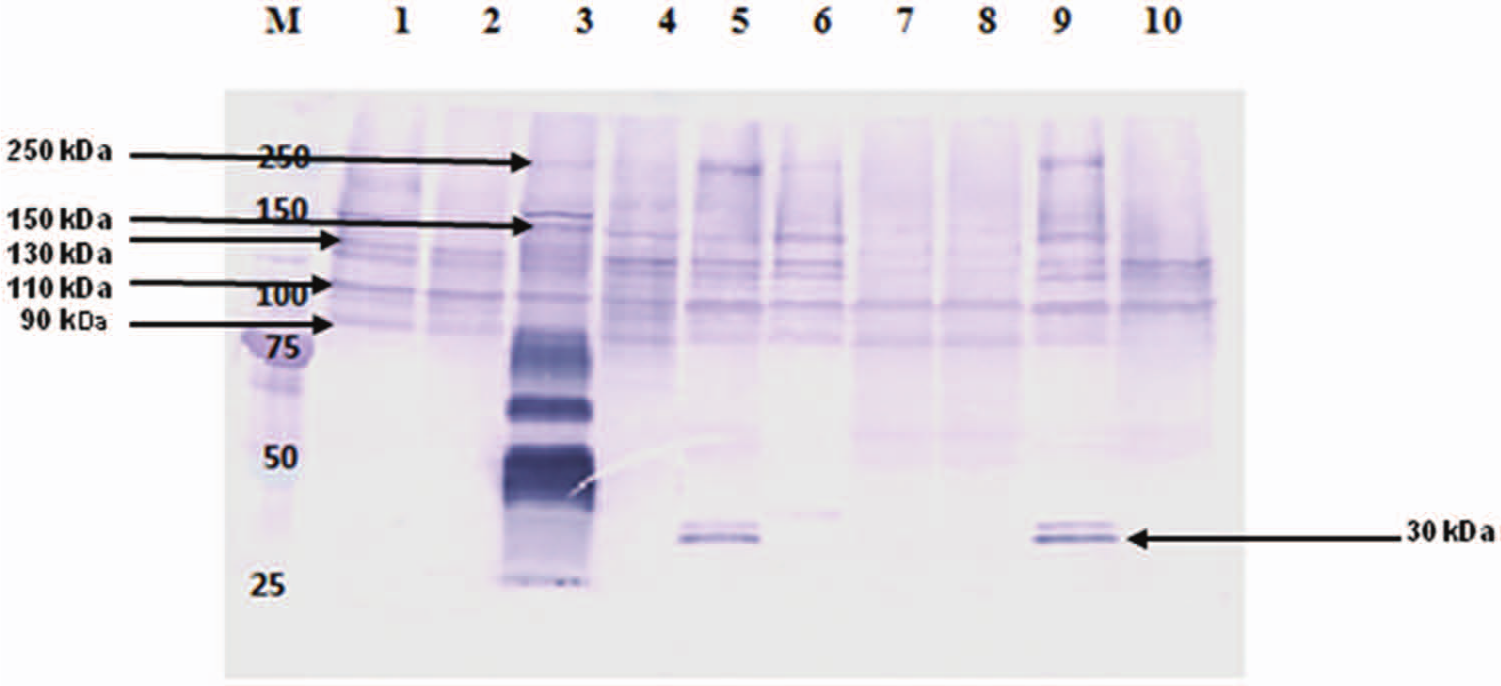
Western blot analysis of culture supernatants of Fusobacterium necrophorum strains from llama and alpaca. Lane M: protein ladder; lane 1: bovine strain, A25 (positive control); lane 2: 11034; lane 3: 1012060; lane 4: 61557; lane 5: 61715; lane 6: 69521; lane 7: 68690; lane 8: 68699; lane 9: 73556; lane 10: 118610.
The leukotoxicity of the culture supernatant was assessed by a cell viability assay using propidium iodide in a flow cytometer. 10 For the assay, PMNs collected from the peripheral blood of cattle and alpaca were used. 14 The viable PMN (1 × 106) cells were treated with culture supernatants for 45 min at 37°C and 5% CO2, washed twice with phosphate buffered saline (PBS), resuspended in PBS, and stained with 10 ml of propidium iodide (50 mg/ml) in the dark for 5 min. The PMNs in complete Roswell Park Memorial Institute (RPMI-1640 g ) medium and PMNs treated with supernatants autoclaved (to denature the protein) from each strain served as negative controls. The samples were processed on a flow cytometer, h and data were analyzed using commercial software. i The flow cytometry analysis showed that the culture supernatants from llama and alpaca isolates were cytotoxic to alpaca and bovine PMNs (Fig. 3). The statistical analysis of cytotoxicity was performed using a generalized linear model (PROC GENMOD). j For analyzing the cytotoxicity of the culture supernatants of the isolates to alpaca and bovine PMNs, a repeated measures analysis of variance model with a serial correlation structure was used. The values were assumed to be normal, and a generalized linear model was used to fit the model. Score statistics based on the differences in the least square means were used to assess the significance of strains, species, and their interactions. Results were considered significant at P < 0.05.
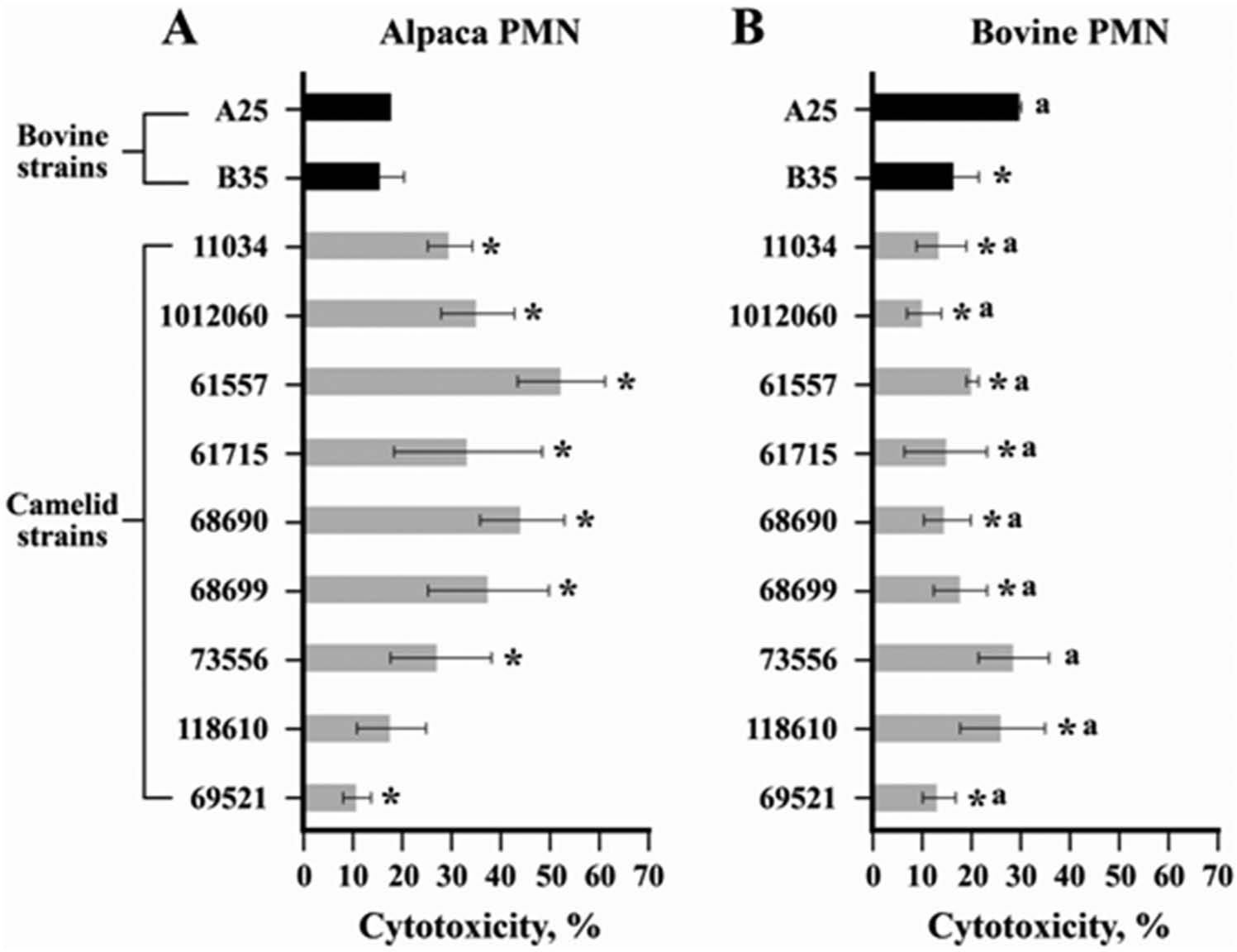
Cytotoxicity assay with culture supernatants of bovine and camelid strains of Fusobacterium necrophorum subsp. necrophorum and subsp. funduliforme using polymorphonuclear cells (PMNs) of alpaca (
The extent of cytotoxicity with either alpaca or bovine PMNs differed among camelid strains. The cytotoxicity of many of the camelid strains were higher (P < 0.05) with alpaca PMNs compared to bovine PMNs. Similarly, strain A25 (subsp. necrophorum) was more cytotoxic to bovine PMNs compared to alpaca PMNs. However, the cytotoxicity of the B35 strain (subsp. funduliforme) did not differ between alpaca and bovine PMNs. Also, the 2 bovine strains did not differ significantly in cytotoxicity with alpaca PMNs, but strain A25 was more cytotoxic than strain B35 with bovine PMNs. With alpaca PMNs, the cytotoxicities of 7 out of 9 camelid strains were higher than either of the 2 bovine strains, cytotoxicity of 1 strain (118610) was similar to, and that of another strain (69521) was lower than that of bovine strains (Fig. 3). With bovine PMNs, the cytotoxicities of all camelid strains, except strain 73556, were lower than that of the bovine strain, A25 (Fig. 3). There was no difference in the cell viabilities of PMNs treated with the autoclaved culture supernatant of either camelid or bovine strains with PMNs suspended in RPMI-1640 as negative control (data not shown), suggesting the toxin is a protein and not lipopolysaccharide.
The cell viability assay using alpaca and bovine PMNs showed that the culture supernatants of camelid strains were cytotoxic to PMNs of both alpaca and bovine. However, many of the strains of llama and alpaca isolates were more toxic to the alpaca PMNs compared to bovine PMNs, which may indicate some animal species specificity with regard to cytotoxicity. The species specificity of bovine F. necrophorum leukotoxin has been previously reported.4,15 The importance of leukotoxin as a virulence factor in fusobacterial infections in camelids needs to be assessed. There is a suggestion that leukotoxin is not a universal virulence factor of those strains of F. necrophorum responsible for necrotic infections in animals and human beings. 7 In the current study, all the isolates from llama and alpaca were shown to have a functional leukotoxin. It is possible that, in camelids, leukotoxin plays a major role in the establishment of necrotic infections. In conclusion, the present study has characterized Fusobacterium isolates of camelid origin. The Fusobacterium isolates from the necrotic infections in alpaca and llama belong to the subsp. necrophorum and the strains had leukotoxin activity, which could be a major virulence factor.
Footnotes
Acknowledgements
Mr. Sailesh Menon is thanked for his help and suggestions with manuscript preparation and submission. Dr. Greg Peterson is also thanked for laboratory support.
a.
BD, Franklin Lakes, NJ.
b.
RapID ANA II kit, Remel Inc., Lenexa, KS.
c.
Eppendorf Mastercycler Gradient, Fisher Scientific, Pittsburgh, PA.
d.
Bio-Rad Laboratories, Hercules, CA.
e.
Sigma-Aldrich, St. Louis, MO.
f.
Millipore Corp., Billerica, MA.
g.
Life Technologies, Grand Island, NY.
h.
FACSCalibur flow cytometer, BD Bioscience, San Jose, CA.
i.
Cell Quest Analysis Software, BD, Franklin Lakes, NJ.
j.
SAS v. 9.3, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declare no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The SUCCESS-FYI grant from the College of Veterinary Medicine at Kansas State University partially supported the study.
